Sedum takesimense Protects PC12 Cells against Corticosterone-Induced Neurotoxicity by Inhibiting Neural Apoptosis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of WEST
2.2. Cell Culture and Treatment
2.3. Cell Viability Assay
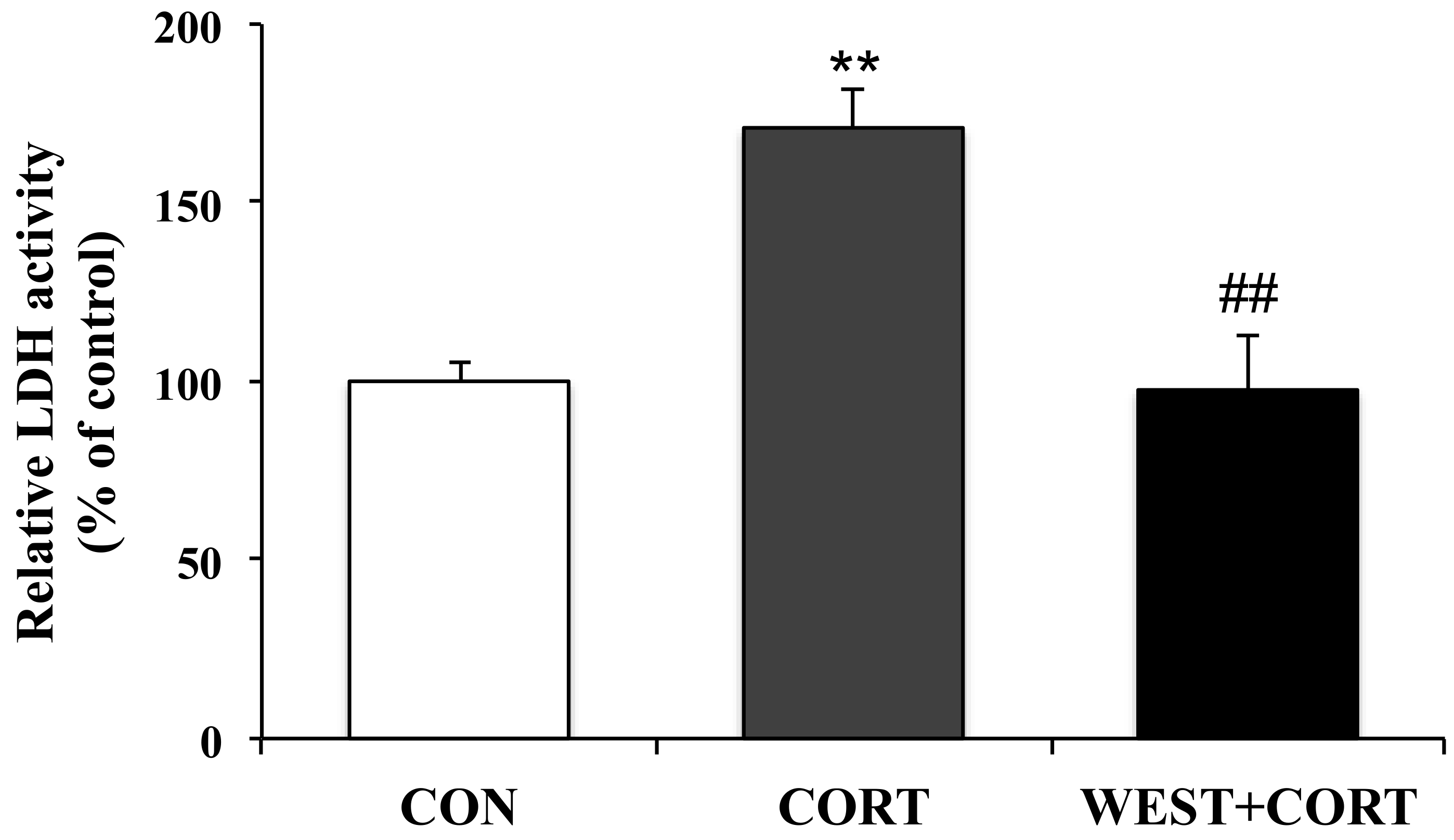
2.4. LDH Leakage Assay
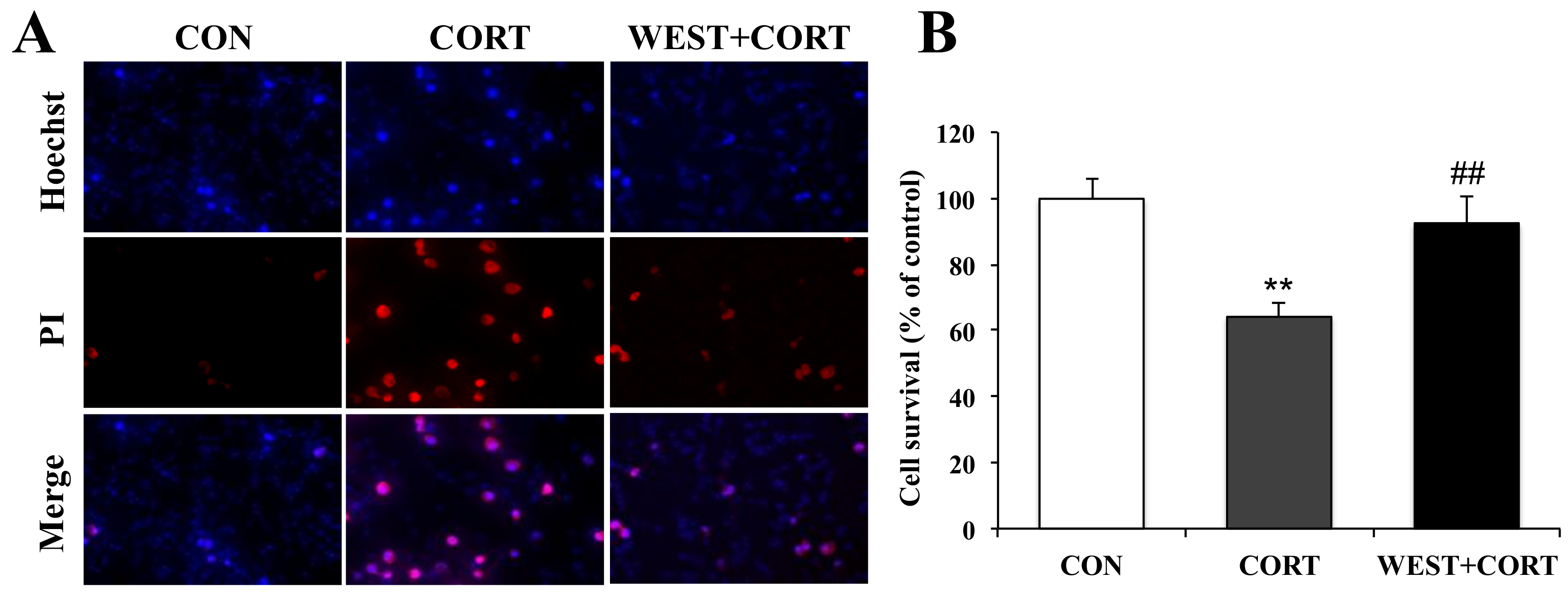
2.5. Hoechst 33342 and PI Double Staining
2.6. TUNEL Staining
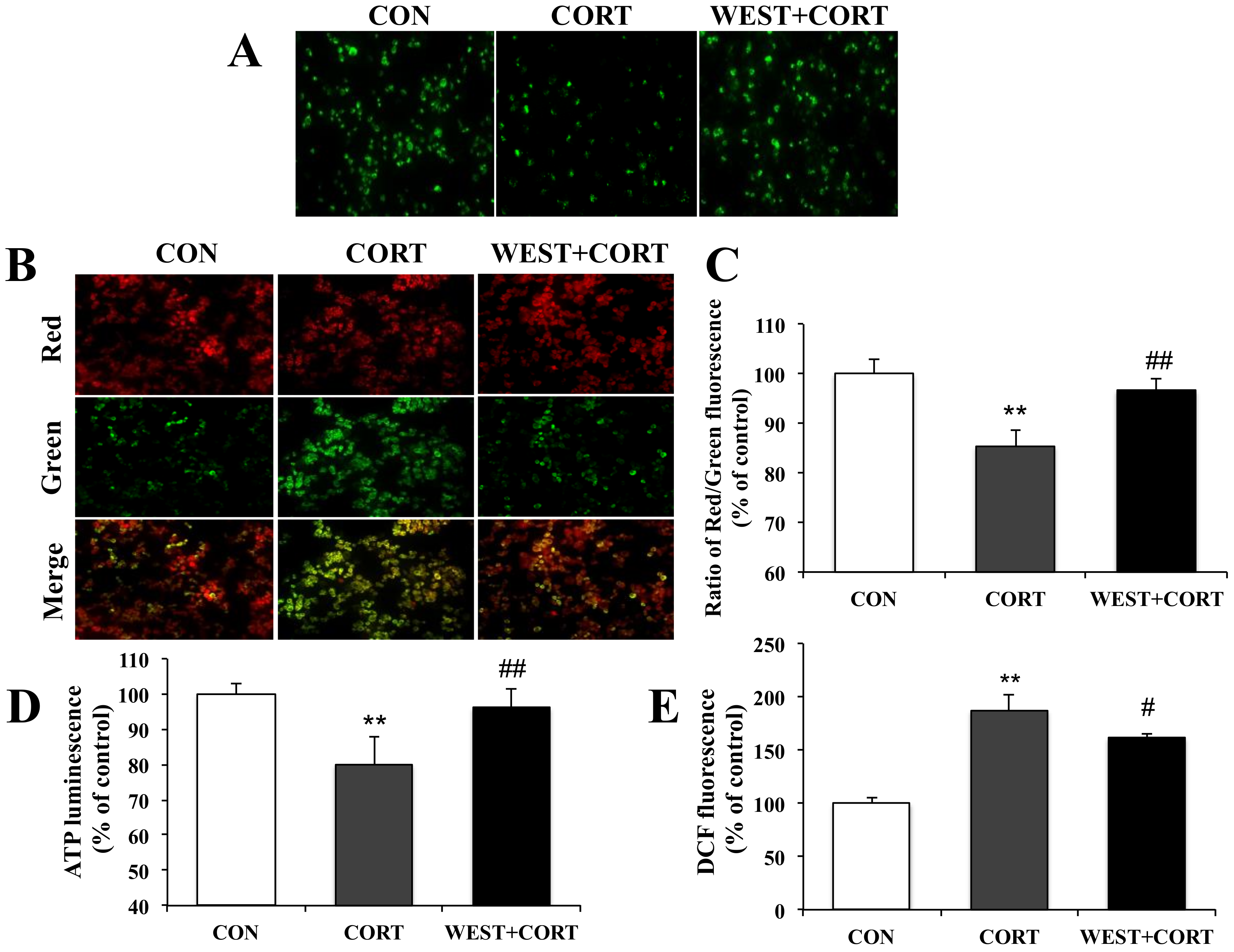
2.7. Intracellular ROS Level Assay
2.8. Intracellular Ca2+ Level Assay
2.9. Detection of mPTP Opening
2.10. Measurement of MMP
2.11. ATP Detection Assay
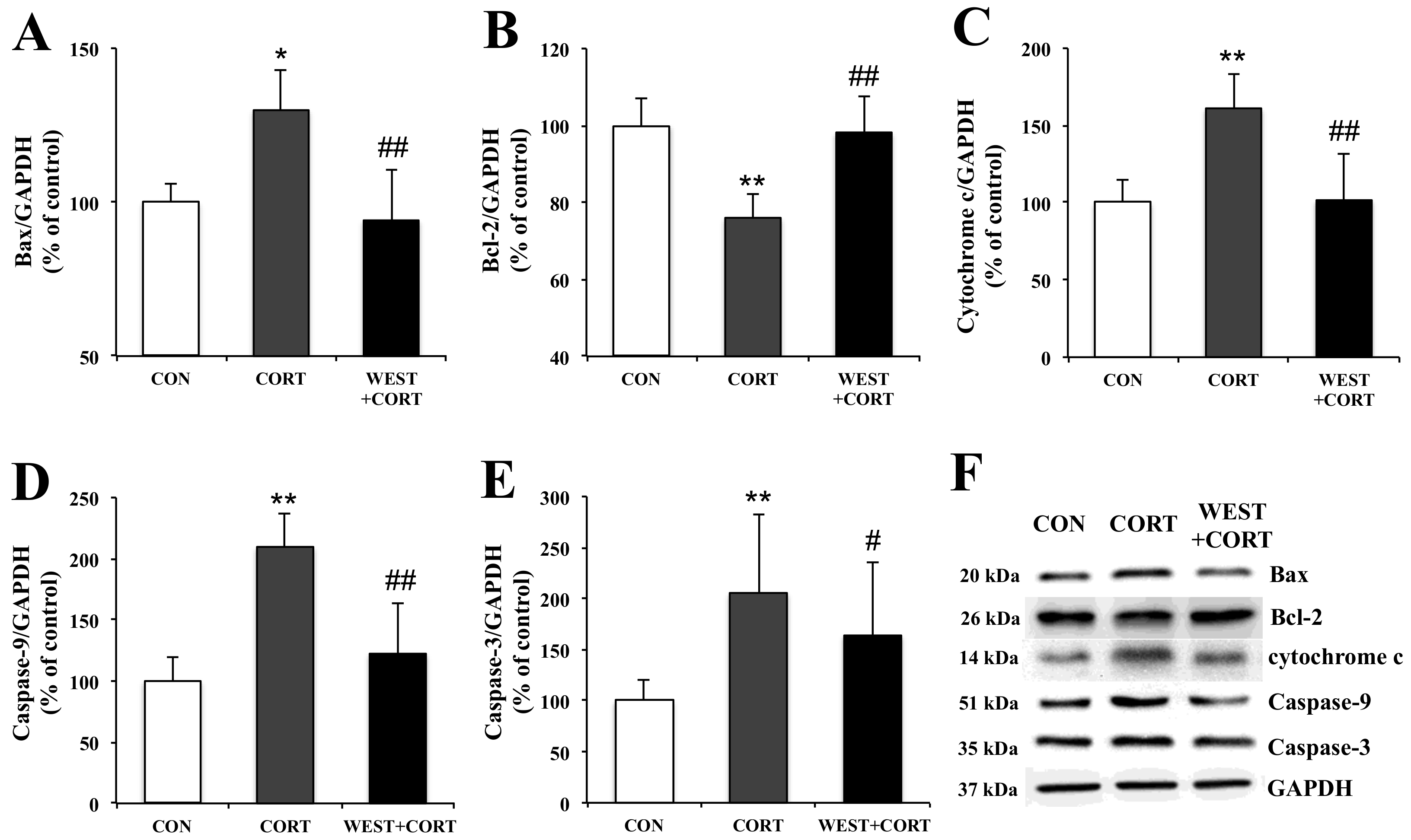
2.12. Western Blot Analysis
2.13. Statistical Analysis
3. Results
3.1. WEST Alleviates Corticosterone-Induced Injury in PC12 Cells
3.2. WEST Inhibits Apoptosis Induced by Corticosterone in PC12 Cells
3.3. WEST Prevents Corticosterone-Induced ER Stress in PC12 Cells
3.4. WEST Protects PC12 Cells against Mitochondrial Dysfunction Following Cell Damage
3.5. WEST Modulates the Expression of Apoptosis-Related Proteins in Corticosterone-Treated PC12 Cells
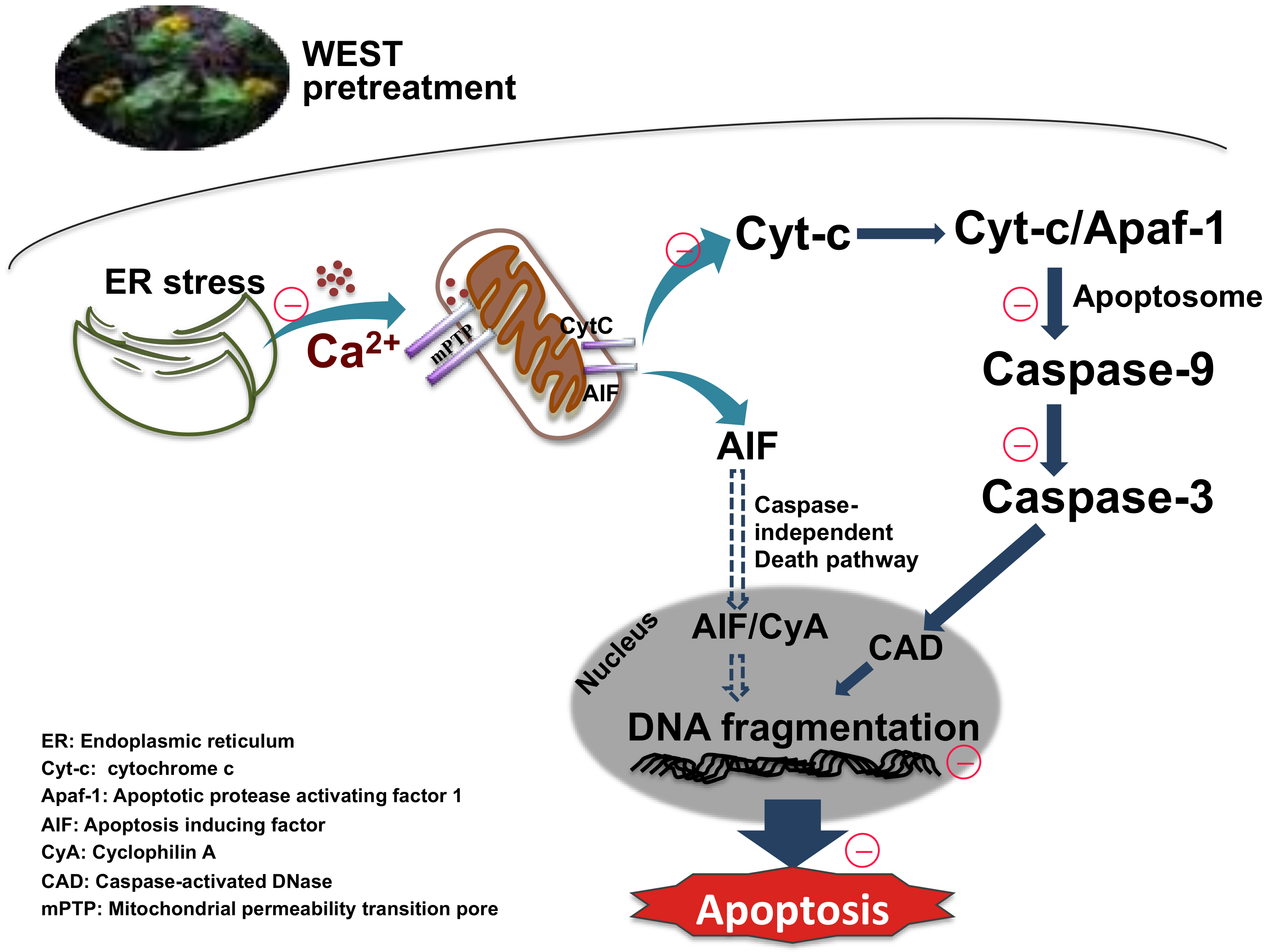
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Zhou, B.; Tan, J.; Zhang, C.; Wu, Y. Neuroprotective effect of polysaccharides from Gastrodia elata Blume against corticosterone-induced apoptosis in PC12 cells via inhibition of the endoplasmic reticulum stress-mediated pathway. Mol. Med. Rep. 2018, 17, 1182–1190. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bereshchenko, O.; Bruscoli, S.; Riccardi, C. Glucocorticoids, sex hormones, and immunity. Front. Immunol. 2018, 9, 1332. [Google Scholar] [CrossRef] [PubMed]
- Pasieka, A.M.; Rafacho, A. Impact of Glucocorticoid Excess on Glucose Tolerance: Clinical and Preclinical Evidence. Metabolites 2016, 6, 24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Numakawa, T.; Odaka, H.; Adachi, N. Actions of brain-derived neurotrophic factor and glucocorticoid stress in neurogenesis. Int. J. Mol. Sci. 2017, 18, 2312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hunter, R.G.; Seligsohn, M.; Rubin, T.G.; Griffiths, B.B.; Ozdemir, Y.; Pfaff, D.W.; Datson, N.A.; McEwen, B.S. Stress and corticosteroids regulate rat hippocampal mitochondrial DNA gene expression via the glucocorticoid receptor. Proc. Natl. Acad. Sci. USA 2016, 113, 9099–9104. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.Y.; Zhao, Y.N.; Wang, Z.L.; Huang, Y.F. Chronic corticosterone exposure reduces hippocampal glycogen level and induces depression-like behavior in mice. J. Zhejiang Univ. Sci. B. 2015, 16, 62–69. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.Y.; Jiang, Y.M.; Liu, Y.M.; Guo, Z.; Shen, S.N.; Liu, X.M.; Pan, R.L. Saikosaponin D acts against corticosterone-induced apoptosis via regulation of mitochondrial GR translocation and a GR-dependent pathway. Prog. Neuropsychopharmacol. Biol. Psychiatry 2014, 53, 80–89. [Google Scholar] [CrossRef]
- Li, Z.Y.; Guo, Z.; Liu, Y.M.; Chang, Q.; Liao, Y.H.; Pan, R.L. Neuroprotective effects of total saikosaponins of Bupleurum yinchowense on corticosterone-induced apoptosis in PC12 cells. J. Ethnopharmacol. 2013, 148, 794–803. [Google Scholar] [CrossRef] [Green Version]
- Jin, W.; Xu, X.; Chen, X.; Qi, W.; Lu, J.; Yan, X.; Zhao, D.; Cong, D.; Li, X.; Sun, L. Protective effect of pig brain polypeptides against corticosterone-induced oxidative stress, inflammatory response, and apoptosis in PC12 cells. Biomed. Pharmacother. 2019, 115, 108890. [Google Scholar] [CrossRef]
- Latt, H.M.; Matsushita, H.; Morino, M.; Koga, Y.; Michiue, H.; Nishiki, T.; Tomizawa, K.; Matsui, H. Oxytocin Inhibits Corticosterone-induced Apoptosis in Primary Hippocampal Neurons. Neuroscience 2018, 379, 383–389. [Google Scholar] [CrossRef] [Green Version]
- Jiang, B.P.; Liu, Y.M.; Le, L.; Si, J.Y.; Liu, X.M.; Chang, Q.; Pan, R.L. Cajaninstilbene acid prevents corticosterone-induced apoptosis in PC12 cells by inhibiting the mitochondrial apoptotic pathway. Cell Physiol. Biochem. 2014, 34, 1015–102611. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wolf, O.T.; Atsak, P.; Quervain, D.J.; Roozendaal, B.; Wignenfeld, K. Stress and Memory: A Selective Review on Recent Developments in the Understanding of Stress Hormone Effects on Memory and Their Clinical Relevance. J. Neuroendocrinol. 2016, 28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pivonello, R.; Simeoli, C.; De Martino, M.C.; Cozzolino, A.; De Leo, M.; Iacuaniello, D.; Pivonello, C.; Negri, M.; Pellecchia, M.T.; Iasevoli, F.; et al. Neuropsychiatric disorders in Cushing’s syndrome. Front Neurosci. 2015, 9, 129. [Google Scholar] [CrossRef] [PubMed]
- Andela, C.D.; van Haalen, F.M.; Ragnarsson, O.; Papakokkinou, E.; Johannsson, G.; Santos, A.; Webb, S.M.; Biermasz, N.R.; Wee, N.J.; Pereira, A.M. Mechanisms in endocrinology: Cushing’s syndrome causes irreversible effects on the human brain: A systematic review of structural and functional magnetic resonance imaging studies. Eur. J. Endocrinol. 2015, 173, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhao, T.T.; Shin, K.S.; Park, H.J.; Yi, B.R.; Lee, K.E.; Lee, M.K. Effects of (-)-sesamin on chronic stress-induced anxiety disorders in mice. Neurochem. Res. 2017, 42, 1123–1129. [Google Scholar] [CrossRef]
- Wang, C.N.; Gong, S.N.; Guan, W.; Wang, J.L.; Gao, T.T.; Wang, Y.; Sun, F.; Jiang, B. Hippocampal overexpression of chordin protects against the chronic social defeat stress-induced depressive-like effects in mice. Brain Res. Bull. 2020, 158, 31–39. [Google Scholar] [CrossRef]
- Griffin, C.E.; Kaye, A.M.; Bueno, F.R.; Kaye, A.D. Benzodiazepine pharmacology and central nervous system–mediated effects. Ochsner. J. 2013, 13, 214–223. [Google Scholar]
- Kim, J.H.; Hart, H.T.; Stevensa, J.F. Alkaloids of some Asian Sedum species. Phytochemistry 1996, 41, 1319–1324. [Google Scholar] [CrossRef]
- Korul’kin, D.Y. Chemical composition of certain Sedum species of Kazakhstan. Chem. Nat. Comp. 2001, 37, 219–223. [Google Scholar] [CrossRef]
- Stevens, J.F.; Hart, H.; Elema, E.T.; Bolck, A. Flavonoid variation in eurasian Sedum and Sempervivum. Phytochemistry 1996, 41, 503–512. [Google Scholar] [CrossRef]
- Szewczyk, K.; Smolarz, H.D.; Gawlik-Dziki, U. Antioxidant properties of three species from Crassulaceae family. Int. J. Pharm. Pharm. Sci. 2012, 4, 107–108. [Google Scholar]
- Vu, T.T.; Kim, J.C.; Choi, Y.H.; Choi, G.J.; Jang, K.S.; Choi, T.H.; Yoon, T.M.; Lee, S.W. Effect of gallotannins derived from Sedum takesimense on tomato bacterial wilt. Plant Dis. 2013, 97, 1593–1598. [Google Scholar] [CrossRef] [Green Version]
- Jang, J.H.; Jung, H.K.; Ko, J.H. Anti-inflammatory effect of Sedum takesimense Nakai water extract in RAW 264.7 cells. Korean J. Med. Corp. Sci. 2016, 24, 228–236. [Google Scholar] [CrossRef]
- Thuong, P.T.; Kang, H.J.; Na, M. Anti-oxidant constituents from Sedum takesimense. Phytochemistry 2007, 68, 2432–2438. [Google Scholar] [CrossRef]
- Jia, H.; Liu, Y.; Yu, M. Neuroprotective effect of cyperi rhizome against corticosterone-induced PC12 cell injury via suppression of Ca2+ overloading. Metabolites 2019, 9, 244. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Y.Z.; Li, X.; Gong, W.X. Protective effect of isoliquiritin against corticosterone-induced neurotoxicity in PC12 cells. Food Funct. 2017, 8, 1235–1244. [Google Scholar] [CrossRef]
- Gong, S.; Zhang, J.; Guo, Z.; Fu, W. Senkyunolide A protects neural cells against corticosterone-induced apoptosis by modulating protein phosphatase 2A and α-synuclein signaling. Drug Des. Devel. Ther. 2018, 12, 1865–1879. [Google Scholar] [CrossRef] [Green Version]
- Schwarz, D.S.; Blower, M.D. The endoplasmic reticulum: Structure, function and response to cellular signaling. Cell Mol. Life Sci. 2016, 73, 79–94. [Google Scholar] [CrossRef] [Green Version]
- Bravo-Sagua, R.; Rodriguez, A.E.; Kuzmicic, J. Cell death and survival through the endoplasmic reticulum-mitochondrial axis. Curr. Mol. Med. 2013, 13, 317–329. [Google Scholar] [CrossRef]
- Malhotra, J.D.; Kaufman, R.J. ER stress and its functional link to mitochondria: Role in cell survival and death. Cold Spring Harb. Perspect. Biol. 2011, 3, a004424. [Google Scholar] [CrossRef]
- Tabas, I.; Ron, D. Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress. Nat. Cell Biol. 2011, 13, 184–190. [Google Scholar] [CrossRef]
- Noh, Y.; Cheon, S.; Kim, I.H.; Kim, I.; Lee, S.-A.; Kim, D.-H.; Jeong, Y. The protective effects of ethanolic extract of Clematis terniflora against corticosterone-induced neuronal damage via the AKT and ERK1/2 pathway. BMB Rep. 2018, 51, 400–405. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Bai, C.; Lu, D.; Wu, X.; Gao, L.; Zhang, W. Endoplasmic reticulum stress and autophagy participate in apoptosis induced by bortezomib in cervical cancer cells. Biotechnol. Lett. 2016, 38, 357–365. [Google Scholar] [CrossRef]
- Park, K.-W.; Kim, G.E.; Morales, R.; Moda, F.; Moreno-Gonzalez, I.; Concha-Marambio, L.; Lee, A.S.; Hetz, C.; Soto, C. The endoplasmic reticulum chaperone GRP78/BiP modulates prion propagation in vitro and in vivo. Sci. Rep. 2017, 7, 44723. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Liu, L.; Naik, I.; Braunstein, Z.; Zhong, J.; Ren, B. Transcription factor C/EBP homologous protein in health and diseases. Front. Immunol. 2017, 8, 1612. [Google Scholar] [CrossRef]
- Kornmann, B.; Currie, E.; Collins, S.R. An ER-mitochondria tethering complex revealed by a synthetic biology screen. Science 2009, 325, 477–481. [Google Scholar] [CrossRef] [Green Version]
- Stacchiotti, A.; Favero, G.; Lavazza, A.; Garcia-Gomez, R.; Monsalve, M.; Rezzani, R. Perspective: Mitochondria-ER contacts in metabolic cellular stress assessed by microscopy. Cells 2019, 8, 5. [Google Scholar] [CrossRef] [Green Version]
- Stacchiotti, A. Exploring cellular stress response and chaperones. Cells 2019, 8, 408. [Google Scholar] [CrossRef] [Green Version]
- Baumgartner, H.K.; Gerasimenko, J.V.; Thorne, C. Calcium elevation in mitochondria is the main Ca2+ requirement for mitochondrial permeability transition pore (mPTP) opening. J. Biol. Chem. 2009, 284, 20796–20803. [Google Scholar] [CrossRef] [Green Version]
- Malli, R.; Frieden, M.; Trenker, M.; Graier, W.F. The role of mitochondria for Ca2+ refilling of the endoplasmic reticulum. J. Biol. Chem. 2005, 280, 12114–12122. [Google Scholar] [CrossRef] [Green Version]
- Deniaud, A.; Sharaf, O.; Maillier, E. Endoplasmic reticulum stress induces calcium-dependent permeability transition, mitochondrial outer membrane permeabilization and apoptosis. Oncogene 2008, 27, 285–299. [Google Scholar] [CrossRef] [Green Version]
- Kosten, T.A.; Galloway, M.P.; Duman, R.S.; Russell, D.S.; D’Sa, C. Repeated unpredictable stress and antidepressants differentially regulate expression of the bcl-2 family of apoptotic genes in rat cortical, hippocampal, and limbic brain structures. Neuropsychopharmacology 2008, 33, 1545–1558. [Google Scholar] [CrossRef]
- Nutt, L.K.; Pataer, A.; Pahler, J. Bax and Bak promote apoptosis by modulating endoplasmic reticular and mitochondrial Ca2+ stores. J. Biol. Chem. 2002, 277, 9219–9225. [Google Scholar] [CrossRef] [Green Version]
- Rong, Y.P.; Bultynck, G.; Aromolaran, A.S. The BH4 domain of Bcl-2 inhibits ER calcium release and apoptosis by binding the regulatory and coupling domain of the IP3 receptor. Proc. Nat. Acad. Sci. USA 2009, 106, 14397–14402. [Google Scholar] [CrossRef] [Green Version]
- White, C.; Li, C.; Yang, J. The endoplasmic reticulum gateway to apoptosis by Bcl-X(L) modulation of the InsP3R. Nature Cell Biol. 2005, 7, 1021–1028. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; He, Y.; Deng, N.; Chen, Y.; Huang, J.; Xie, W. Protective effect of resveratrol against corticosterone-induced neurotoxicity in PC12 cells. Transl. Neurosci. 2019, 10, 235–240. [Google Scholar] [CrossRef] [Green Version]
- Hajnóczky, G.; Csordás, G.; Das, S.; Garcia-Perez, C. Mitochondrial calcium signalling and cell death: Approaches for assessing the role of mitochondrial Ca2+ uptake in apoptosis. Cell Calcium. 2006, 40, 553–560. [Google Scholar] [CrossRef] [Green Version]
- Yang, Y.; Gao, H.; Zhou, H.; Liu, Q.; Qi, Z.; Zhang, Y.; Zhang, J. The role of mitochondria-derived peptides in cardiovascular disease: Recent updates. Biomed. Pharmacother. 2019, 117, 109075. [Google Scholar] [CrossRef]
- Kroemer, G.; Galluzzi, L.; Brenner, C. Mitochondrial membrane permeabilization in cell death. Physiol. Rev. 2007, 87, 99–163. [Google Scholar] [CrossRef]
- Flippo, K.H.; Strack, S. Mitochondrial dynamics in neuronal injury, development and plasticity. J. Cell Sci. 2017, 130, 671–681. [Google Scholar] [CrossRef] [Green Version]
- Lenaz, G.; Genova, M.L. Structural and functional organization of the mitochondrial respiratory chain: A dynamic super-assembly. Int. J. Biochem. Cell Biol. 2009, 41, 1750–1772. [Google Scholar] [CrossRef]
- Jiang, Y.; Li, Z.; Liu, Y. Neuroprotective effect of water extract of Panax ginseng on corticosterone-induced apoptosis in PC12 cells and its underlying molecule mechanisms. J. Ethnopharmacol. 2015, 159, 102–112. [Google Scholar] [CrossRef]
- Lechardeur, D.; Drzymala, L.; Sharma, M. Determinants of the nuclear localization of the heterodimeric DNA fragmentation factor (ICAD/CAD). J. Cell Biol. 2000, 150, 321–334. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Shen, S.; Li, Z. Cajaninstilbene acid protects corticosterone-induced injury in PC12 cells by inhibiting oxidative and endoplasmic reticulum stress-mediated apoptosis. Neurochem. Int. 2014, 78, 43–52. [Google Scholar] [CrossRef]
- Beytut, E.; Yilmaz, S.; Aksakal, M.; Polat, S. The possible protective effects of vitamin E and selenium administration in oxidative stress caused by high doses of glucocorticoid administration in the brain of rats. J. Trace. Elem. Med. Biol. 2018, 45, 131–135. [Google Scholar] [CrossRef]
- Lee, Y.M.; Bae, J.H.; Jung, H.Y.; Kim, J.H.; Park, D.S. Antioxidant activity in water and methanol extracts from Korean edible wild plants. J. Korean Soc. Food Sci. Nutr. 2011, 40, 29–36. [Google Scholar] [CrossRef]
- Wang, Y.G.; Li, Y.; Wang, C.Y. L-3-n-Butylphthalide protects rats’ cardiomyocytes from ischaemia/reperfusion-induced apoptosis by affecting the mitochondrial apoptosis pathway. Acta Physiol. 2014, 210, 524–533. [Google Scholar] [CrossRef]
- Di Meo, F.; Cuciniello, R.; Margarucci, S. Ginkgo biloba prevents oxidative stress-induced apoptosis blocking p53 activation in neuroblastoma cells. Antioxidants 2020, 9, 279. [Google Scholar] [CrossRef] [Green Version]
- Reyes-Fermín, L.M.; Aparicio-Trejo, O.E.; Avila-Rojas, S.H.; Gómez-Sierra, T.; Martínez-Klimova, E.; Pedraza-Chaverri, J. Natural antioxidants’ effects on endoplasmic reticulum stress-related diseases. Food Chem. Toxicol. 2020, 138, 111229. [Google Scholar] [CrossRef]



| Compounds | |
|---|---|
| ferulic acid caffeic acid gallic acid methyl gallate myricetin quercetin luteolin rhodalin | rhodalidin arbutin 2,6-di-O-galloylarbutin luteolin-7-O-β-D-glucoside gossypetin-8-O-β-D-xylopyranoside 1-(4-hydroxyphenyl)-2-(3,5-dihydroxyphenyl)-2-hydroxyethanone |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yun, H.-Y.; Jeong, Y. Sedum takesimense Protects PC12 Cells against Corticosterone-Induced Neurotoxicity by Inhibiting Neural Apoptosis. Nutrients 2020, 12, 3713. https://doi.org/10.3390/nu12123713
Yun H-Y, Jeong Y. Sedum takesimense Protects PC12 Cells against Corticosterone-Induced Neurotoxicity by Inhibiting Neural Apoptosis. Nutrients. 2020; 12(12):3713. https://doi.org/10.3390/nu12123713
Chicago/Turabian StyleYun, Hea-Yeon, and Yoonhwa Jeong. 2020. "Sedum takesimense Protects PC12 Cells against Corticosterone-Induced Neurotoxicity by Inhibiting Neural Apoptosis" Nutrients 12, no. 12: 3713. https://doi.org/10.3390/nu12123713




