Maternal Docosahexaenoic Acid Status during Pregnancy and Its Impact on Infant Neurodevelopment
Abstract
1. Introduction
2. DHA and Its Metabolites: Effects on the Structure and Function of the Human Brain
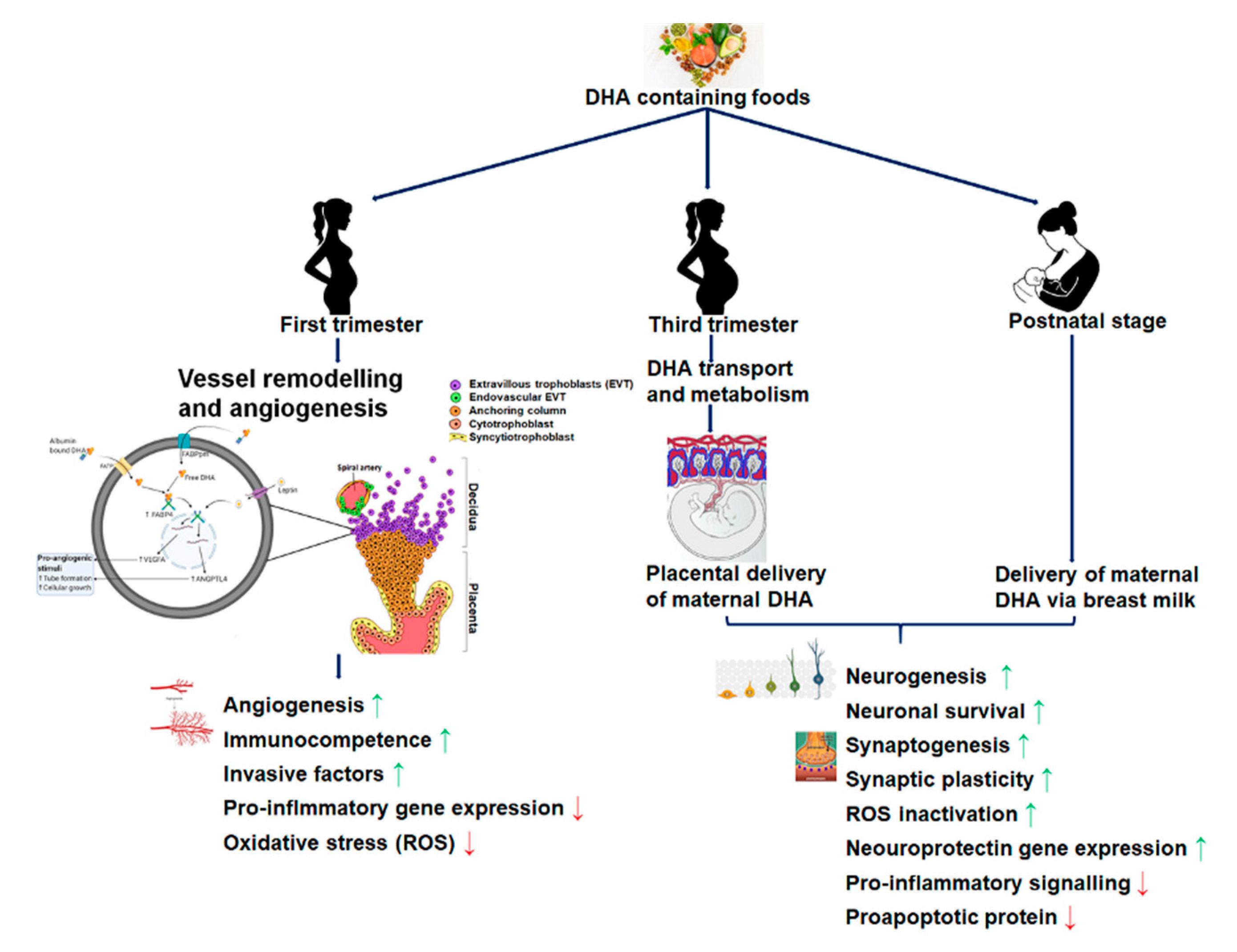
2.1. DHA Accretion, Supplementation, and Fetal Brain Development
2.2. Maternal DHA and Its Effects on the Placental Structure and Functional Development
2.3. Maternal Intakes of DHA and Its Impact on Fetal Brain Development
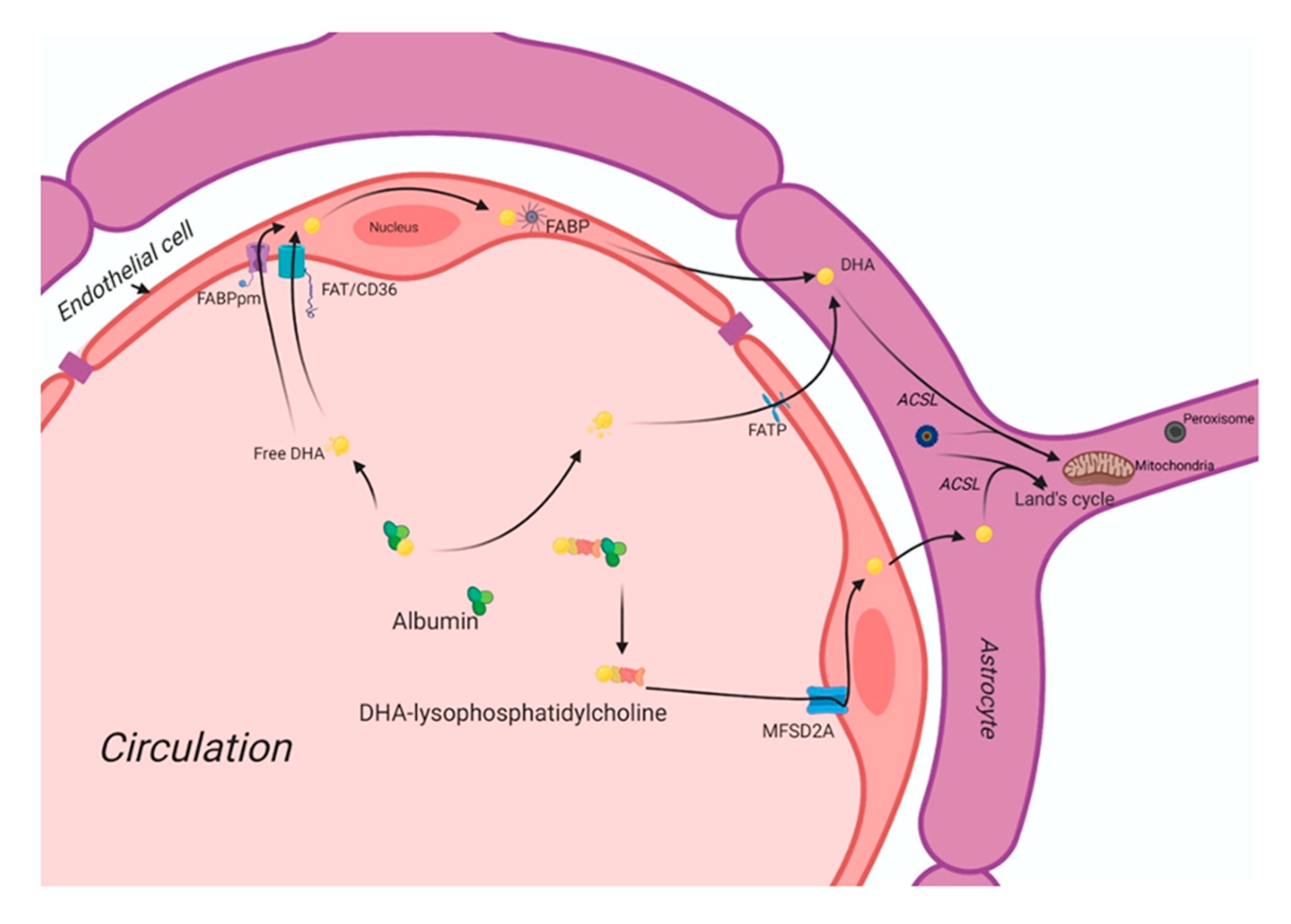
3. The DHA Uptake System in the Human Brain
DHA Deficiency during Fetal Brain Development and Its Impact on Cognitive Functions
4. Maternal DHA Supplementation and Brain Development
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Salem, N., Jr.; Litman, B.; Kim, H.Y.; Gawrisch, K. Mechanisms of action of docosahexaenoic acid in the nervous system. Lipids 2001, 36, 945–959. [Google Scholar] [CrossRef] [PubMed]
- Bourre, J.M. Dietary omega-3 fatty acids for women. Biomed. Pharm. 2007, 61, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Bradbury, J. Docosahexaenoic acid (DHA): An ancient nutrient for the modern human brain. Nutrients 2011, 3, 529–554. [Google Scholar] [CrossRef]
- Horrocks, L.A.; Yeo, Y.K. Health benefits of docosahexaenoic acid (DHA). Pharmacol. Res. 1999, 40, 211–225. [Google Scholar] [CrossRef]
- Quinn, J.F.; Raman, R.; Thomas, R.G.; Yurko-Mauro, K.; Nelson, E.B.; Van Dyck, C.; Galvin, J.E.; Emond, J.; Jack, C.R., Jr.; Weiner, M.; et al. Docosahexaenoic acid supplementation and cognitive decline in Alzheimer disease: A randomized trial. J. Am. Med. Assoc. 2010, 304, 1903–1911. [Google Scholar] [CrossRef]
- de Urquiza, A.M.; Liu, S.; Sjoberg, M.; Zetterstrom, R.H.; Griffiths, W.; Sjovall, J.; Perlmann, T. Docosahexaenoic acid, a ligand for the retinoid X receptor in mouse brain. Science 2000, 290, 2140–2144. [Google Scholar] [CrossRef]
- Ji, R.R.; Xu, Z.Z.; Strichartz, G.; Serhan, C.N. Emerging roles of resolvins in the resolution of inflammation and pain. Trends Neurosci. 2011, 34, 599–609. [Google Scholar] [CrossRef]
- Matesanz, N.; Park, G.; McAllister, H.; Leahey, W.; Devine, A.; McVeigh, G.E.; Gardiner, T.A.; McDonald, D.M. Docosahexaenoic acid improves the nitroso-redox balance and reduces VEGF-mediated angiogenic signaling in microvascular endothelial cells. Invest. Ophthalmol. Vis. Sci. 2010, 51, 6815–6825. [Google Scholar] [CrossRef] [PubMed]
- Kabaran, S.; Besler, H.T. Do fatty acids affect fetal programming? J. Health Popul. Nutr. 2015, 33, 14. [Google Scholar] [CrossRef] [PubMed]
- Basak, S.; Vilasagaram, S.; Duttaroy, A.K. Maternal dietary deficiency of n-3 fatty acids affects metabolic and epigenetic phenotypes of the developing fetus. Prostaglandins Leukot. Essent. Fatty Acids 2020, 158, 102109. [Google Scholar] [CrossRef]
- Lacombe, R.J.S.; Chouinard-Watkins, R.; Bazinet, R.P. Brain docosahexaenoic acid uptake and metabolism. Mol. Asp. Med. 2018, 64, 109–134. [Google Scholar] [CrossRef] [PubMed]
- Brenna, J.T.; Carlson, S.E. Docosahexaenoic acid and human brain development: Evidence that a dietary supply is needed for optimal development. J. Hum. Evol. 2014, 77, 99–106. [Google Scholar] [CrossRef] [PubMed]
- Niemoller, T.D.; Bazan, N.G. Docosahexaenoic acid neurolipidomics. Prostaglandins Other Lipid Mediat. 2010, 91, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Salem, N., Jr.; Eggersdorfer, M. Is the world supply of omega-3 fatty acids adequate for optimal human nutrition? Curr. Opin. Clin. Nutr. Metab. Care 2015, 18, 147–154. [Google Scholar] [CrossRef]
- Buckley, C.D.; Gilroy, D.W.; Serhan, C.N. Proresolving lipid mediators and mechanisms in the resolution of acute inflammation. Immunity 2014, 40, 315–327. [Google Scholar] [CrossRef]
- Serhan, C.N.; Dalli, J.; Colas, R.A.; Winkler, J.W.; Chiang, N. Protectins and maresins: New pro-resolving families of mediators in acute inflammation and resolution bioactive metabolome. Biochim. Biophys. Acta 2015, 1851, 397–413. [Google Scholar] [CrossRef]
- Makrides, M.; Gibson, R.A.; McPhee, A.J.; Yelland, L.; Quinlivan, J.; Ryan, P.; Team, D.O.I. Effect of DHA supplementation during pregnancy on maternal depression and neurodevelopment of young children: A randomized controlled trial. JAMA 2010, 304, 1675–1683. [Google Scholar] [CrossRef]
- Funk, C.D. Prostaglandins and leukotrienes: Advances in eicosanoid biology. Science 2001, 294, 1871–1875. [Google Scholar] [CrossRef]
- Latham, C.F.; Osborne, S.L.; Cryle, M.J.; Meunier, F.A. Arachidonic acid potentiates exocytosis and allows neuronal SNARE complex to interact with Munc18a. J. Neurochem. 2007, 100, 1543–1554. [Google Scholar] [CrossRef]
- Wu, A.; Ying, Z.; Gomez-Pinilla, F. The salutary effects of DHA dietary supplementation on cognition, neuroplasticity, and membrane homeostasis after brain trauma. J. Neurotrauma 2011, 28, 2113–2122. [Google Scholar] [CrossRef]
- Belayev, L.; Khoutorova, L.; Atkins, K.D.; Eady, T.N.; Hong, S.; Lu, Y.; Obenaus, A.; Bazan, N.G. Docosahexaenoic Acid therapy of experimental ischemic stroke. Transl. Stroke Res. 2011, 2, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Arita, M.; Bianchini, F.; Aliberti, J.; Sher, A.; Chiang, N.; Hong, S.; Yang, R.; Petasis, N.A.; Serhan, C.N. Stereochemical assignment, antiinflammatory properties, and receptor for the omega-3 lipid mediator resolvin E1. J. Exp. Med. 2005, 201, 713–722. [Google Scholar] [CrossRef] [PubMed]
- Marcheselli, V.L.; Hong, S.; Lukiw, W.J.; Tian, X.H.; Gronert, K.; Musto, A.; Hardy, M.; Gimenez, J.M.; Chiang, N.; Serhan, C.N.; et al. Novel docosanoids inhibit brain ischemia-reperfusion-mediated leukocyte infiltration and pro-inflammatory gene expression. J. Biol. Chem. 2003, 278, 43807–43817. [Google Scholar] [CrossRef]
- Gronert, K.; Maheshwari, N.; Khan, N.; Hassan, I.R.; Dunn, M.; Laniado Schwartzman, M. A role for the mouse 12/15-lipoxygenase pathway in promoting epithelial wound healing and host defense. J. Biol. Chem. 2005, 280, 15267–15278. [Google Scholar] [CrossRef]
- Serhan, C.N.; Gotlinger, K.; Hong, S.; Lu, Y.; Siegelman, J.; Baer, T.; Yang, R.; Colgan, S.P.; Petasis, N.A. Anti-inflammatory actions of neuroprotectin D1/protectin D1 and its natural stereoisomers: Assignments of dihydroxy-containing docosatrienes. J. Immunol. 2006, 176, 1848–1859. [Google Scholar] [CrossRef]
- Mukherjee, P.K.; Marcheselli, V.L.; Serhan, C.N.; Bazan, N.G. Neuroprotectin D1: A docosahexaenoic acid-derived docosatriene protects human retinal pigment epithelial cells from oxidative stress. Proc. Natl. Acad. Sci. USA 2004, 101, 8491–8496. [Google Scholar] [CrossRef]
- Chen, P.; Vericel, E.; Lagarde, M.; Guichardant, M. Poxytrins, a class of oxygenated products from polyunsaturated fatty acids, potently inhibit blood platelet aggregation. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2011, 25, 382–388. [Google Scholar] [CrossRef]
- Liu, M.; Boussetta, T.; Makni-Maalej, K.; Fay, M.; Driss, F.; El-Benna, J.; Lagarde, M.; Guichardant, M. Protectin DX, a double lipoxygenase product of DHA, inhibits both ROS production in human neutrophils and cyclooxygenase activities. Lipids 2014, 49, 49–57. [Google Scholar] [CrossRef]
- White, P.J.; St-Pierre, P.; Charbonneau, A.; Mitchell, P.L.; St-Amand, E.; Marcotte, B.; Marette, A. Protectin DX alleviates insulin resistance by activating a myokine-liver glucoregulatory axis. Nat. Med. 2014, 20, 664–669. [Google Scholar] [CrossRef]
- Stein, K.; Stoffels, M.; Lysson, M.; Schneiker, B.; Dewald, O.; Kronke, G.; Kalff, J.C.; Wehner, S. A role for 12/15-lipoxygenase-derived proresolving mediators in postoperative ileus: Protectin DX-regulated neutrophil extravasation. J. Leukoc. Biol. 2015, 99, 231–239. [Google Scholar] [CrossRef]
- Serhan, C.N.; Yang, R.; Martinod, K.; Kasuga, K.; Pillai, P.S.; Porter, T.F.; Oh, S.F.; Spite, M. Maresins: Novel macrophage mediators with potent antiinflammatory and proresolving actions. J. Exp. Med. 2009, 206, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Abdulnour, R.E.; Dalli, J.; Colby, J.K.; Krishnamoorthy, N.; Timmons, J.Y.; Tan, S.H.; Colas, R.A.; Petasis, N.A.; Serhan, C.N.; Levy, B.D. Maresin 1 biosynthesis during platelet-neutrophil interactions is organ-protective. Proc. Natl. Acad. Sci. USA 2014, 111, 16526–16531. [Google Scholar] [CrossRef] [PubMed]
- Nordgren, T.M.; Heires, A.J.; Wyatt, T.A.; Poole, J.A.; LeVan, T.D.; Cerutis, D.R.; Romberger, D.J. Maresin-1 reduces the pro-inflammatory response of bronchial epithelial cells to organic dust. Respir. Res. 2013, 14, 51. [Google Scholar] [CrossRef] [PubMed]
- Poorani, R.; Bhatt, A.N.; Dwarakanath, B.S.; Das, U.N. COX-2, aspirin and metabolism of arachidonic, eicosapentaenoic and docosahexaenoic acids and their physiological and clinical significance. Eur. J. Pharmacol. 2015, 785, 116–132. [Google Scholar] [CrossRef] [PubMed]
- Gurzell, E.A.; Teague, H.; Harris, M.; Clinthorne, J.; Shaikh, S.R.; Fenton, J.I. DHA-enriched fish oil targets B cell lipid microdomains and enhances ex vivo and in vivo B cell function. J. Leukoc. Biol. 2013, 93, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Anderson, G.J.; Connor, W.E.; Corliss, J.D. Docosahexaenoic acid is the preferred dietary n-3 fatty acid for the development of the brain and retina. Pediatric Res. 1990, 27, 89–97. [Google Scholar] [CrossRef]
- O’Brien, J.S.; Sampson, E.L. Fatty acid and fatty aldehyde composition of the major brain lipids in normal human gray matter, white matter, and myelin. J. Lipid Res. 1965, 6, 545–551. [Google Scholar]
- Brenna, J.T.; Diau, G.Y. The influence of dietary docosahexaenoic acid and arachidonic acid on central nervous system polyunsaturated fatty acid composition. Prostaglandins Leukot. Essent. Fat. Acids 2007, 77, 247–250. [Google Scholar] [CrossRef]
- Innis, S.M. Chapter 10 Essential fatty acid metabolism during early development. Biol. Grow. Anim. 2005, 3, 235–274. [Google Scholar] [CrossRef]
- Campoy, C.; Escolano-Margarit, M.V.; Anjos, T.; Szajewska, H.; Uauy, R. Omega 3 fatty acids on child growth, visual acuity and neurodevelopment. Br. J. Nutr. 2012, 107, S85–S106. [Google Scholar] [CrossRef]
- Montgomery, C.; Speake, B.K.; Cameron, A.; Sattar, N.; Weaver, L.T. Maternal docosahexaenoic acid supplementation and fetal accretion. Br. J. Nutr. 2003, 90, 135–145. [Google Scholar] [CrossRef] [PubMed]
- McCann, J.C.; Ames, B.N. Is docosahexaenoic acid, an n-3 long-chain polyunsaturated fatty acid, required for development of normal brain function? An overview of evidence from cognitive and behavioral tests in humans and animals. Am. J. Clin. Nutr. 2005, 82, 281–295. [Google Scholar] [CrossRef] [PubMed]
- Innis, S.M. Perinatal biochemistry and physiology of long-chain polyunsaturated fatty acids. J. Pediatrics 2003, 143, S1–S8. [Google Scholar] [CrossRef]
- Helland, I.B.; Saugstad, O.D.; Smith, L.; Saarem, K.; Solvoll, K.; Ganes, T.; Drevon, C.A. Similar effects on infants of n-3 and n-6 fatty acids supplementation to pregnant and lactating women. Pediatrics 2001, 108, E82. [Google Scholar] [CrossRef]
- Carlson, S.E.; Colombo, J.; Gajewski, B.J.; Gustafson, K.M.; Mundy, D.; Yeast, J.; Georgieff, M.K.; Markley, L.A.; Kerling, E.H.; Shaddy, D.J. DHA supplementation and pregnancy outcomes. Am. J. Clin. Nutr. 2013, 97, 808–815. [Google Scholar] [CrossRef] [PubMed]
- Decsi, T.; Campoy, C.; Koletzko, B. Effect of N-3 polyunsaturated fatty acid supplementation in pregnancy: The Nuheal trial. Adv. Exp. Med. Biol. 2005, 569, 109–113. [Google Scholar]
- Imhoff-Kunsch, B.; Stein, A.D.; Villalpando, S.; Martorell, R.; Ramakrishnan, U. Docosahexaenoic acid supplementation from mid-pregnancy to parturition influenced breast milk fatty acid concentrations at 1 month postpartum in Mexican women. J. Nutr. 2011, 141, 321–326. [Google Scholar] [CrossRef]
- Helland, I.B.; Saugstad, O.D.; Saarem, K.; Van Houwelingen, A.C.; Nylander, G.; Drevon, C.A. Supplementation of n-3 fatty acids during pregnancy and lactation reduces maternal plasma lipid levels and provides DHA to the infants. J. Matern. Fetal Neonatal Med. 2006, 19, 397–406. [Google Scholar] [CrossRef]
- Bergmann, R.L.; Haschke-Becher, E.; Klassen-Wigger, P.; Bergmann, K.E.; Richter, R.; Dudenhausen, J.W.; Grathwohl, D.; Haschke, F. Supplementation with 200 mg/day docosahexaenoic acid from mid-pregnancy through lactation improves the docosahexaenoic acid status of mothers with a habitually low fish intake and of their infants. Ann. Nutr. Metab. 2008, 52, 157–166. [Google Scholar] [CrossRef]
- Larque, E.; Krauss-Etschmann, S.; Campoy, C.; Hartl, D.; Linde, J.; Klingler, M.; Demmelmair, H.; Cano, A.; Gil, A.; Bondy, B.; et al. Docosahexaenoic acid supply in pregnancy affects placental expression of fatty acid transport proteins. Am. J. Clin. Nutr. 2006, 84, 853–861. [Google Scholar] [CrossRef]
- Duttaroy, A.K. Transport of fatty acids across the human placenta: A review. Prog. Lipid Res. 2009, 48, 52–61. [Google Scholar] [CrossRef] [PubMed]
- Dunstan, J.A.; Simmer, K.; Dixon, G.; Prescott, S.L. Cognitive assessment of children at age 2(1/2) years after maternal fish oil supplementation in pregnancy: A randomised controlled trial. Arch. Dis. Child. Fetal Neonatal Ed. 2008, 93, F45–F50. [Google Scholar] [CrossRef] [PubMed]
- Escolano-Margarit, M.V.; Ramos, R.; Beyer, J.; Csabi, G.; Parrilla-Roure, M.; Cruz, F.; Perez-Garcia, M.; Hadders-Algra, M.; Gil, A.; Decsi, T.; et al. Prenatal DHA status and neurological outcome in children at age 5.5 years are positively associated. J. Nutr. 2011, 141, 1216–1223. [Google Scholar] [CrossRef] [PubMed]
- Campoy, C.; Escolano-Margarit, M.V.; Ramos, R.; Parrilla-Roure, M.; Csabi, G.; Beyer, J.; Ramirez-Tortosa, M.C.; Molloy, A.M.; Decsi, T.; Koletzko, B.V. Effects of prenatal fish-oil and 5-methyltetrahydrofolate supplementation on cognitive development of children at 6.5 y of age. Am. J. Clin. Nutr. 2011, 94, 1880S–1888S. [Google Scholar] [CrossRef]
- Sanders, T.A.; Naismith, D.J. A comparison of the influence of breast-feeding and bottle-feeding on the fatty acid composition of the erythrocytes. Br. J. Nutr. 1979, 41, 619–623. [Google Scholar] [CrossRef]
- Clandinin, M.T.; Chappell, J.E.; Leong, S.; Heim, T.; Swyer, P.R.; Chance, G.W. Extrauterine fatty acid accretion in infant brain: Implications for fatty acid requirements. Early Hum. Dev. 1980, 4, 131–138. [Google Scholar] [CrossRef]
- Makrides, M.; Neumann, M.A.; Gibson, R.A. Perinatal characteristics may influence the outcome of visual acuity. Lipids 2001, 36, 897–900. [Google Scholar] [CrossRef]
- Innis, S.M.; Auestad, N.; Siegman, J.S. Blood lipid docosahexaenoic and arachidonic acid in term gestation infants fed formulas with high docosahexaenoic acid, low eicosapentaenoic acid fish oil. Lipids 1996, 31, 617–625. [Google Scholar] [CrossRef]
- Sanders, T.A.; Ellis, F.R.; Dickerson, J.W. Studies of vegans: The fatty acid composition of plasma choline phosphoglycerides, erythrocytes, adipose tissue, and breast milk, and some indicators of susceptibility to ischemic heart disease in vegans and omnivore controls. Am. J. Clin. Nutr. 1978, 31, 805–813. [Google Scholar] [CrossRef]
- Innis, S.M.; Gilley, J.; Werker, J. Are human milk long-chain polyunsaturated fatty acids related to visual and neural development in breast-fed term infants? J. Pediatrics 2001, 139, 532–538. [Google Scholar] [CrossRef]
- Lauritzen, L.; Brambilla, P.; Mazzocchi, A.; Harslof, L.B.; Ciappolino, V.; Agostoni, C. DHA Effects in Brain Development and Function. Nutrients 2016, 8, 6. [Google Scholar] [CrossRef] [PubMed]
- Colombo, J.; Kannass, K.N.; Shaddy, D.J.; Kundurthi, S.; Maikranz, J.M.; Anderson, C.J.; Blaga, O.M.; Carlson, S.E. Maternal DHA and the development of attention in infancy and toddlerhood. Child. Dev. 2004, 75, 1254–1267. [Google Scholar] [CrossRef] [PubMed]
- Malcolm, C.A.; McCulloch, D.L.; Montgomery, C.; Shepherd, A.; Weaver, L.T. Maternal docosahexaenoic acid supplementation during pregnancy and visual evoked potential development in term infants: A double blind, prospective, randomised trial. Arch. Dis. Child. Fetal Neonatal Ed. 2003, 88, F383–F390. [Google Scholar] [CrossRef] [PubMed]
- Lauritzen, L.; Jorgensen, M.H.; Mikkelsen, T.B.; Skovgaard l, M.; Straarup, E.M.; Olsen, S.F.; Hoy, C.E.; Michaelsen, K.F. Maternal fish oil supplementation in lactation: Effect on visual acuity and n-3 fatty acid content of infant erythrocytes. Lipids 2004, 39, 195–206. [Google Scholar] [CrossRef] [PubMed]
- Helland, I.B.; Smith, L.; Saarem, K.; Saugstad, O.D.; Drevon, C.A. Maternal supplementation with very-long-chain n-3 fatty acids during pregnancy and lactation augments children’s IQ at 4 years of age. Pediatrics 2003, 111, e39–e44. [Google Scholar] [CrossRef] [PubMed]
- Hibbeln, J.R.; Davis, J.M.; Steer, C.; Emmett, P.; Rogers, I.; Williams, C.; Golding, J. Maternal seafood consumption in pregnancy and neurodevelopmental outcomes in childhood (ALSPAC study): An observational cohort study. Lancet 2007, 369, 578–585. [Google Scholar] [CrossRef]
- Oken, E.; Wright, R.O.; Kleinman, K.P.; Bellinger, D.; Amarasiriwardena, C.J.; Hu, H.; Rich-Edwards, J.W.; Gillman, M.W. Maternal fish consumption, hair mercury, and infant cognition in a U.S. Cohort. Environ. Health Perspect. 2005, 113, 1376–1380. [Google Scholar] [CrossRef]
- Birch, E.E.; Hoffman, D.R.; Castaneda, Y.S.; Fawcett, S.L.; Birch, D.G.; Uauy, R.D. A randomized controlled trial of long-chain polyunsaturated fatty acid supplementation of formula in term infants after weaning at 6 wk of age. Am. J. Clin. Nutr. 2002, 75, 570–580. [Google Scholar] [CrossRef]
- Hoffman, D.R.; Theuer, R.C.; Castaneda, Y.S.; Wheaton, D.H.; Bosworth, R.G.; O’Connor, A.R.; Morale, S.E.; Wiedemann, L.E.; Birch, E.E. Maturation of visual acuity is accelerated in breast-fed term infants fed baby food containing DHA-enriched egg yolk. J. Nutr. 2004, 134, 2307–2313. [Google Scholar] [CrossRef]
- Birch, E.E.; Garfield, S.; Hoffman, D.R.; Uauy, R.; Birch, D.G. A randomized controlled trial of early dietary supply of long-chain polyunsaturated fatty acids and mental development in term infants. Dev. Med. Child. Neurol. 2000, 42, 174–181. [Google Scholar] [CrossRef]
- Birch, E.E.; Garfield, S.; Castaneda, Y.; Hughbanks-Wheaton, D.; Uauy, R.; Hoffman, D. Visual acuity and cognitive outcomes at 4 years of age in a double-blind, randomized trial of long-chain polyunsaturated fatty acid-supplemented infant formula. Early Hum. Dev. 2007, 83, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Auestad, N.; Scott, D.T.; Janowsky, J.S.; Jacobsen, C.; Carroll, R.E.; Montalto, M.B.; Halter, R.; Qiu, W.; Jacobs, J.R.; Connor, W.E.; et al. Visual, cognitive, and language assessments at 39 months: A follow-up study of children fed formulas containing long-chain polyunsaturated fatty acids to 1 year of age. Pediatrics 2003, 112, e177–e183. [Google Scholar] [CrossRef] [PubMed]
- Birch, E.E.; Carlson, S.E.; Hoffman, D.R.; Fitzgerald-Gustafson, K.M.; Fu, V.L.; Drover, J.R.; Castaneda, Y.S.; Minns, L.; Wheaton, D.K.; Mundy, D.; et al. The DIAMOND (DHA Intake And Measurement Of Neural Development) Study: A double-masked, randomized controlled clinical trial of the maturation of infant visual acuity as a function of the dietary level of docosahexaenoic acid. Am. J. Clin. Nutr. 2010, 91, 848–859. [Google Scholar] [CrossRef] [PubMed]
- Henriksen, C.; Haugholt, K.; Lindgren, M.; Aurvag, A.K.; Ronnestad, A.; Gronn, M.; Solberg, R.; Moen, A.; Nakstad, B.; Berge, R.K.; et al. Improved cognitive development among preterm infants attributable to early supplementation of human milk with docosahexaenoic acid and arachidonic acid. Pediatrics 2008, 121, 1137–1145. [Google Scholar] [CrossRef]
- Fang, P.C.; Kuo, H.K.; Huang, C.B.; Ko, T.Y.; Chen, C.C.; Chung, M.Y. The effect of supplementation of docosahexaenoic acid and arachidonic acid on visual acuity and neurodevelopment in larger preterm infants. Chang. Gung Med. J. 2005, 28, 708–715. [Google Scholar]
- Meldrum, S.J.; D’Vaz, N.; Simmer, K.; Dunstan, J.A.; Hird, K.; Prescott, S.L. Effects of high-dose fish oil supplementation during early infancy on neurodevelopment and language: A randomised controlled trial. Br. J. Nutr. 2012, 108, 1443–1454. [Google Scholar] [CrossRef]
- Isaacs, E.B.; Ross, S.; Kennedy, K.; Weaver, L.T.; Lucas, A.; Fewtrell, M.S. 10-year cognition in preterms after random assignment to fatty acid supplementation in infancy. Pediatrics 2011, 128, e890–e898. [Google Scholar] [CrossRef]
- Faldella, G.; Govoni, M.; Alessandroni, R.; Marchiani, E.; Salvioli, G.P.; Biagi, P.L.; Spano, C. Visual evoked potentials and dietary long chain polyunsaturated fatty acids in preterm infants. Arch. Dis. Child. Fetal Neonatal Ed. 1996, 75, F108–F112. [Google Scholar] [CrossRef]
- Lassek, W.D.; Gaulin, S.J. Linoleic and docosahexaenoic acids in human milk have opposite relationships with cognitive test performance in a sample of 28 countries. Prostaglandins Leukot. Essent. Fat. Acids 2014, 91, 195–201. [Google Scholar] [CrossRef]
- Yamashima, T. Dual effects of the non-esterified fatty acid receptor ‘GPR40’ for human health. Prog. Lipid Res. 2015, 58, 40–50. [Google Scholar] [CrossRef]
- Zamberletti, E.; Piscitelli, F.; De Castro, V.; Murru, E.; Gabaglio, M.; Colucci, P.; Fanali, C.; Prini, P.; Bisogno, T.; Maccarrone, M.; et al. Lifelong imbalanced LA/ALA intake impairs emotional and cognitive behavior via changes in brain endocannabinoid system. J. Lipid Res. 2017, 58, 301–316. [Google Scholar] [CrossRef] [PubMed]
- Malerba, G.; Schaeffer, L.; Xumerle, L.; Klopp, N.; Trabetti, E.; Biscuola, M.; Cavallari, U.; Galavotti, R.; Martinelli, N.; Guarini, P.; et al. SNPs of the FADS gene cluster are associated with polyunsaturated fatty acids in a cohort of patients with cardiovascular disease. Lipids 2008, 43, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Caspi, A.; Williams, B.; Kim-Cohen, J.; Craig, I.W.; Milne, B.J.; Poulton, R.; Schalkwyk, L.C.; Taylor, A.; Werts, H.; Moffitt, T.E. Moderation of breastfeeding effects on the IQ by genetic variation in fatty acid metabolism. Proc. Natl. Acad. Sci. USA 2007, 104, 18860–18865. [Google Scholar] [CrossRef] [PubMed]
- Harbild, H.L.; Harslof, L.B.; Christensen, J.H.; Kannass, K.N.; Lauritzen, L. Fish oil-supplementation from 9 to 12 months of age affects infant attention in a free-play test and is related to change in blood pressure. Prostaglandins Leukot Essent Fatty Acids 2013, 89, 327–333. [Google Scholar] [CrossRef]
- Ryan, A.S.; Nelson, E.B. Assessing the effect of docosahexaenoic acid on cognitive functions in healthy, preschool children: A randomized, placebo-controlled, double-blind study. Clin. Pediatrics (Phila) 2008, 47, 355–362. [Google Scholar] [CrossRef]
- Parletta, N.; Cooper, P.; Gent, D.N.; Petkov, J.; O’Dea, K. Effects of fish oil supplementation on learning and behaviour of children from Australian Indigenous remote community schools: A randomised controlled trial. Prostaglandins Leukot. Essent. Fat. Acids 2013, 89, 71–79. [Google Scholar] [CrossRef]
- Milte, C.M.; Parletta, N.; Buckley, J.D.; Coates, A.M.; Young, R.M.; Howe, P.R. Eicosapentaenoic and docosahexaenoic acids, cognition, and behavior in children with attention-deficit/hyperactivity disorder: A randomized controlled trial. Nutrition 2012, 28, 670–677. [Google Scholar] [CrossRef]
- Williams, P.J.; Bulmer, J.N.; Innes, B.A.; Broughton Pipkin, F. Possible roles for folic acid in the regulation of trophoblast invasion and placental development in normal early human pregnancy. Biol. Reprod. 2011, 84, 1148–1153. [Google Scholar] [CrossRef]
- Khong, Y.; Brosens, I. Defective deep placentation. Best Pract. Res. Clin. Obstet. Gynaecol 2011, 25, 301–311. [Google Scholar] [CrossRef]
- Mainigi, M.A.; Olalere, D.; Burd, I.; Sapienza, C.; Bartolomei, M.; Coutifaris, C. Peri-implantation hormonal milieu: Elucidating mechanisms of abnormal placentation and fetal growth. Biol. Reprod. 2014, 90, 26. [Google Scholar] [CrossRef]
- Alfaidy, N.; Hoffmann, P.; Boufettal, H.; Samouh, N.; Aboussaouira, T.; Benharouga, M.; Feige, J.J.; Brouillet, S. The multiple roles of EG-VEGF/PROK1 in normal and pathological placental angiogenesis. BioMed Res. Int. 2014, 2014, 451906. [Google Scholar] [CrossRef] [PubMed]
- Johnsen, G.M.; Basak, S.; Weedon-Fekjaer, M.S.; Staff, A.C.; Duttaroy, A.K. Docosahexaenoic acid stimulates tube formation in first trimester trophoblast cells, HTR8/SVneo. Placenta 2011, 32, 626–632. [Google Scholar] [CrossRef] [PubMed]
- Innis, S.M. Essential fatty acids in growth and development. Prog. Lipid Res. 1991, 30, 39–103. [Google Scholar] [CrossRef]
- Basak, S.; Duttaroy, A.K. Effects of fatty acids on angiogenic activity in the placental extravillious trophoblast cells. Prostaglandins Leukot. Essent. Fat. Acids 2013, 88, 155–162. [Google Scholar] [CrossRef]
- Spencer, L.; Mann, C.; Metcalfe, M.; Webb, M.; Pollard, C.; Spencer, D.; Berry, D.; Steward, W.; Dennison, A. The effect of omega-3 FAs on tumour angiogenesis and their therapeutic potential. Eur. J. Cancer 2009, 45, 2077–2086. [Google Scholar] [CrossRef]
- Jing, K.; Wu, T.; Lim, K. Omega-3 polyunsaturated fatty acids and cancer. Anticancer Agents Med. Chem. 2013, 13, 1162–1177. [Google Scholar] [CrossRef]
- Calviello, G.; Di Nicuolo, F.; Gragnoli, S.; Piccioni, E.; Serini, S.; Maggiano, N.; Tringali, G.; Navarra, P.; Ranelletti, F.O.; Palozza, P. n-3 PUFAs reduce VEGF expression in human colon cancer cells modulating the COX-2/PGE2 induced ERK-1 and -2 and HIF-1alpha induction pathway. Carcinogenesis 2004, 25, 2303–2310. [Google Scholar] [CrossRef]
- Hasan, A.U.; Ohmori, K.; Konishi, K.; Igarashi, J.; Hashimoto, T.; Kamitori, K.; Yamaguchi, F.; Tsukamoto, I.; Uyama, T.; Ishihara, Y.; et al. Eicosapentaenoic acid upregulates VEGF-A through both GPR120 and PPARgamma mediated pathways in 3T3-L1 adipocytes. Mol. Cell. Endocrinol. 2015, 406, 10–18. [Google Scholar] [CrossRef]
- Sapieha, P.; Stahl, A.; Chen, J.; Seaward, M.R.; Willett, K.L.; Krah, N.M.; Dennison, R.J.; Connor, K.M.; Aderman, C.M.; Liclican, E.; et al. 5-Lipoxygenase metabolite 4-HDHA is a mediator of the antiangiogenic effect of omega-3 polyunsaturated fatty acids. Sci. Transl. Med. 2011, 3, 69ra12. [Google Scholar] [CrossRef]
- Dutta-Roy, A.K. Transport mechanisms for long-chain polyunsaturated fatty acids in the human placenta. Am. J. Clin. Nutr. 2000, 71, 315S–322S. [Google Scholar] [CrossRef]
- Campbell, F.M.; Bush, P.G.; Veerkamp, J.H.; Dutta-Roy, A.K. Detection and cellular localization of plasma membrane-associated and cytoplasmic fatty acid-binding proteins in human placenta. Placenta 1998, 19, 409–415. [Google Scholar] [CrossRef]
- Campbell, F.M.; Dutta-Roy, A.K. Plasma membrane fatty acid-binding protein (FABPpm) is exclusively located in the maternal facing membranes of the human placenta. FEBS Lett. 1995, 375, 227–230. [Google Scholar] [CrossRef]
- Campbell, F.M.; Gordon, M.J.; Dutta-Roy, A.K. Placental membrane fatty acid-binding protein preferentially binds arachidonic and docosahexaenoic acids. Life Sci. 1998, 63, 235–240. [Google Scholar] [CrossRef]
- Sánchez-Campillo, M.; Ruiz-Palacios, M.; Ruiz-Alcaraz, A.J.; Prieto-Sánchez, M.T.; Blanco-Carnero, J.E.; Zornoza, M.; Ruiz-Pastor, M.J.; Demmelmair, H.; Sánchez-Solís, M.; Koletzko, B.; et al. Child Head Circumference and Placental MFSD2a Expression Are Associated to the Level of MFSD2a in Maternal Blood During Pregnancy. Front. Endocrinol. 2020, 11, 38. [Google Scholar] [CrossRef] [PubMed]
- Prieto-Sanchez, M.T.; Ruiz-Palacios, M.; Blanco-Carnero, J.E.; Pagan, A.; Hellmuth, C.; Uhl, O.; Peissner, W.; Ruiz-Alcaraz, A.J.; Parrilla, J.J.; Koletzko, B.; et al. Placental MFSD2a transporter is related to decreased DHA in cord blood of women with treated gestational diabetes. Clin. Nutr. 2017, 36, 513–521. [Google Scholar] [CrossRef]
- Duttaroy, A.K. Fatty acid-activated nuclear transcription factors and their roles in human placenta. Eur. J. Lipid Sci. Technol. 2006, 108, 70–83. [Google Scholar] [CrossRef]
- Olsen, S.F.; Joensen, H.D. High liveborn birth weights in the Faroes: A comparison between birth weights in the Faroes and in Denmark. J. Epidemiol. Community Health 1985, 39, 27–32. [Google Scholar] [CrossRef]
- Greenberg, J.A.; Bell, S.J.; Ausdal, W.V. Omega-3 Fatty Acid supplementation during pregnancy. Rev. Obstet. Gynecol. 2008, 1, 162–169. [Google Scholar]
- Dhobale, M.V.; Wadhwani, N.; Mehendale, S.S.; Pisal, H.R.; Joshi, S.R. Reduced levels of placental long chain polyunsaturated fatty acids in preterm deliveries. Prostaglandins Leukot. Essent. Fat. Acids 2011, 85, 149–153. [Google Scholar] [CrossRef]
- Koletzko, B.; Cetin, I.; Brenna, J.T. Dietary fat intakes for pregnant and lactating women. Br. J. Nutr. 2007, 98, 873–877. [Google Scholar] [CrossRef]
- Carlson, S.E. Docosahexaenoic acid supplementation in pregnancy and lactation. Am. J. Clin. Nutr. 2009, 89, 678S–684S. [Google Scholar] [CrossRef] [PubMed]
- Innis, S.M. Fatty acids and early human development. Early Hum. Dev. 2007, 83, 761–766. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, U.; Stein, A.D.; Parra-Cabrera, S.; Wang, M.; Imhoff-Kunsch, B.; Juarez-Marquez, S.; Rivera, J.; Martorell, R. Effects of docosahexaenoic acid supplementation during pregnancy on gestational age and size at birth: Randomized, double-blind, placebo-controlled trial in Mexico. Food Nutr. Bull. 2010, 31, S108–S116. [Google Scholar] [CrossRef] [PubMed]
- Innis, S.M.; Friesen, R.W. Essential n-3 fatty acids in pregnant women and early visual acuity maturation in term infants. Am. J. Clin. Nutr. 2008, 87, 548–557. [Google Scholar] [CrossRef] [PubMed]
- Larque, E.; Gil-Sanchez, A.; Prieto-Sanchez, M.T.; Koletzko, B. Omega 3 fatty acids, gestation and pregnancy outcomes. Br. J. Nutr. 2012, 107, S77–S84. [Google Scholar] [CrossRef]
- Mantzioris, E.; James, M.J.; Gibson, R.A.; Cleland, L.G. Dietary substitution with an alpha-linolenic acid-rich vegetable oil increases eicosapentaenoic acid concentrations in tissues. Am. J. Clin. Nutr. 1994, 59, 1304–1309. [Google Scholar] [CrossRef]
- Imhoff-Kunsch, B.; Briggs, V.; Goldenberg, T.; Ramakrishnan, U. Effect of n-3 long-chain polyunsaturated fatty acid intake during pregnancy on maternal, infant, and child health outcomes: A systematic review. Paediatr. Perinat. Epidemiol. 2012, 26, 91–107. [Google Scholar] [CrossRef]
- Olsen, S.F.; Secher, N.J. Low consumption of seafood in early pregnancy as a risk factor for preterm delivery: Prospective cohort study. BMJ 2002, 324, 447. [Google Scholar] [CrossRef]
- Rogers, I.; Emmett, P.; Ness, A.; Golding, J. Maternal fish intake in late pregnancy and the frequency of low birth weight and intrauterine growth retardation in a cohort of British infants. J. Epidemiol. Community Health 2004, 58, 486–492. [Google Scholar] [CrossRef]
- Olsen, S.F.; Sorensen, J.D.; Secher, N.J.; Hedegaard, M.; Henriksen, T.B.; Hansen, H.S.; Grant, A. Randomised controlled trial of effect of fish-oil supplementation on pregnancy duration. Lancet 1992, 339, 1003–1007. [Google Scholar] [CrossRef]
- Helland, I.B.; Smith, L.; Blomen, B.; Saarem, K.; Saugstad, O.D.; Drevon, C.A. Effect of supplementing pregnant and lactating mothers with n-3 very-long-chain fatty acids on children’s IQ and body mass index at 7 years of age. Pediatrics 2008, 122, e472–e479. [Google Scholar] [CrossRef] [PubMed]
- Harris, W.S.; Baack, M.L. Beyond building better brains: Bridging the docosahexaenoic acid (DHA) gap of prematurity. J. Perinatol. 2015, 35, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Holman, R.T.; Johnson, S.B.; Hatch, T.F. A case of human linolenic acid deficiency involving neurological abnormalities. Am. J. Clin. Nutr. 1982, 35, 617–623. [Google Scholar] [CrossRef]
- Bjerve, K.S.; Fischer, S.; Alme, K. Alpha-linolenic acid deficiency in man: Effect of ethyl linolenate on plasma and erythrocyte fatty acid composition and biosynthesis of prostanoids. Am. J. Clin. Nutr. 1987, 46, 570–576. [Google Scholar] [CrossRef]
- Burdge, G.C.; Jones, A.E.; Wootton, S.A. Eicosapentaenoic and docosapentaenoic acids are the principal products of alpha-linolenic acid metabolism in young men*. Br. J. Nutr. 2002, 88, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, M.; DeMar, J.C., Jr.; Ma, K.; Chang, L.; Bell, J.M.; Rapoport, S.I. Docosahexaenoic acid synthesis from alpha-linolenic acid by rat brain is unaffected by dietary n-3 PUFA deprivation. J. Lipid Res. 2007, 48, 1150–1158. [Google Scholar] [CrossRef]
- Uauy, R.; Mena, P.; Valenzuela, A. Essential fatty acids as determinants of lipid requirements in infants, children and adults. Eur. J. Clin. Nutr. 1999, 53, S66–S77. [Google Scholar] [CrossRef]
- Ciappolino, V.; Mazzocchi, A.; Botturi, A.; Turolo, S.; Delvecchio, G.; Agostoni, C.; Brambilla, P. The Role of Docosahexaenoic Acid (DHA) on Cognitive Functions in Psychiatric Disorders. Nutrients 2019, 11, 769. [Google Scholar] [CrossRef] [PubMed]
- McNamara, R.K.; Vannest, J.J.; Valentine, C.J. Role of perinatal long-chain omega-3 fatty acids in cortical circuit maturation: Mechanisms and implications for psychopathology. World J. Psychiatry 2015, 5, 15–34. [Google Scholar] [CrossRef]
- Guxens, M.; Mendez, M.A.; Molto-Puigmarti, C.; Julvez, J.; Garcia-Esteban, R.; Forns, J.; Ferrer, M.; Vrijheid, M.; Lopez-Sabater, M.C.; Sunyer, J. Breastfeeding, long-chain polyunsaturated fatty acids in colostrum, and infant mental development. Pediatrics 2011, 128, e880–e889. [Google Scholar] [CrossRef]
- Bernard, J.Y.; Armand, M.; Garcia, C.; Forhan, A.; De Agostini, M.; Charles, M.A.; Heude, B.; Group, E.M.-C.C.S. The association between linoleic acid levels in colostrum and child cognition at 2 and 3 y in the EDEN cohort. Pediatric Res. 2015, 77, 829–835. [Google Scholar] [CrossRef] [PubMed]
- Innis, S.M. Polyunsaturated fatty acids in human milk: An essential role in infant development. Adv. Exp. Med. Biol. 2004, 554, 27–43. [Google Scholar] [PubMed]
- Simopoulos, A.P. Importance of the omega-6/omega-3 balance in health and disease: Evolutionary aspects of diet. World Rev. Nutr. Diet. 2011, 102, 10–21. [Google Scholar]
- FAO. Fats and Fatty Acids in Human Nutrition. Report of an Expert Consultation; FAO Food and Nutrition Paper; FAO: Geneva, Switzerland, 2010; Volume 91, pp. 1–166. [Google Scholar]
- Niinivirta, K.; Isolauri, E.; Laakso, P.; Linderborg, K.; Laitinen, K. Dietary counseling to improve fat quality during pregnancy alters maternal fat intake and infant essential fatty acid status. J. Nutr. 2011, 141, 1281–1285. [Google Scholar] [CrossRef] [PubMed]
- Hautero, U.; Laakso, P.; Linderborg, K.; Niinivirta, K.; Poussa, T.; Isolauri, E.; Laitinen, K. Proportions and concentrations of serum n-3 fatty acids can be increased by dietary counseling during pregnancy. Eur. J. Clin. Nutr. 2013, 67, 1163–1168. [Google Scholar] [CrossRef] [PubMed]
- Oken, E.; Guthrie, L.B.; Bloomingdale, A.; Platek, D.N.; Price, S.; Haines, J.; Gillman, M.W.; Olsen, S.F.; Bellinger, D.C.; Wright, R.O. A pilot randomized controlled trial to promote healthful fish consumption during pregnancy: The Food for Thought Study. Nutr. J. 2013, 12, 33. [Google Scholar] [CrossRef]
- Emmett, R.; Akkersdyk, S.; Yeatman, H.; Meyer, B.J. Expanding awareness of docosahexaenoic acid during pregnancy. Nutrients 2013, 5, 1098–1109. [Google Scholar] [CrossRef]
- Miles, E.A.; Noakes, P.S.; Kremmyda, L.S.; Vlachava, M.; Diaper, N.D.; Rosenlund, G.; Urwin, H.; Yaqoob, P.; Rossary, A.; Farges, M.C.; et al. The Salmon in Pregnancy Study: Study design, subject characteristics, maternal fish and marine n-3 fatty acid intake, and marine n-3 fatty acid status in maternal and umbilical cord blood. Am. J. Clin. Nutr. 2011, 94, 1986S–1992S. [Google Scholar] [CrossRef]
- Bascunan, K.A.; Valenzuela, R.; Chamorro, R.; Valencia, A.; Barrera, C.; Puigrredon, C.; Sandoval, J.; Valenzuela, A. Polyunsaturated fatty acid composition of maternal diet and erythrocyte phospholipid status in Chilean pregnant women. Nutrients 2014, 6, 4918–4934. [Google Scholar] [CrossRef]
- Decsi, T.; Campoy, C.; Demmelmair, H.; Szabo, E.; Marosvolgyi, T.; Escolano, M.; Marchal, G.; Krauss-Etschmann, S.; Cruz, M.; Koletzko, B. Inverse association between trans isomeric and long-chain polyunsaturated fatty acids in pregnant women and their newborns: Data from three European countries. Ann. Nutr. Metab. 2011, 59, 107–116. [Google Scholar] [CrossRef]
- Lagarde, M.; Bernoud, N.; Brossard, N.; Lemaitre-Delaunay, D.; Thies, F.; Croset, M.; Lecerf, J. Lysophosphatidylcholine as a preferred carrier form of docosahexaenoic acid to the brain. J. Mol. Neurosci. 2001, 16, 201–204. [Google Scholar] [CrossRef]
- Chen, C.T.; Kitson, A.P.; Hopperton, K.E.; Domenichiello, A.F.; Trepanier, M.O.; Lin, L.E.; Ermini, L.; Post, M.; Thies, F.; Bazinet, R.P. Plasma non-esterified docosahexaenoic acid is the major pool supplying the brain. Sci. Rep. 2015, 5, 15791. [Google Scholar] [CrossRef] [PubMed]
- Demar, J.C., Jr.; Ma, K.; Chang, L.; Bell, J.M.; Rapoport, S.I. alpha-Linolenic acid does not contribute appreciably to docosahexaenoic acid within brain phospholipids of adult rats fed a diet enriched in docosahexaenoic acid. J. Neurochem. 2005, 94, 1063–1076. [Google Scholar] [CrossRef] [PubMed]
- Chan, J.P.; Wong, B.H.; Chin, C.F.; Galam, D.L.A.; Foo, J.C.; Wong, L.C.; Ghosh, S.; Wenk, M.R.; Cazenave-Gassiot, A.; Silver, D.L. The lysolipid transporter Mfsd2a regulates lipogenesis in the developing brain. PLoS Biol. 2018, 16, e2006443. [Google Scholar] [CrossRef]
- Kuipers, R.S.; Luxwolda, M.F.; Offringa, P.J.; Boersma, E.R.; Dijck-Brouwer, D.A.; Muskiet, F.A. Fetal intrauterine whole body linoleic, arachidonic and docosahexaenoic acid contents and accretion rates. Prostaglandins Leukot. Essent. Fat. Acids 2012, 86, 13–20. [Google Scholar] [CrossRef]
- Talamonti, E.; Sasso, V.; To, H.; Haslam, R.P.; Napier, J.A.; Ulfhake, B.; Pernold, K.; Asadi, A.; Hessa, T.; Jacobsson, A.; et al. Impairment of DHA synthesis alters the expression of neuronal plasticity markers and the brain inflammatory status in mice. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2020, 34, 2024–2040. [Google Scholar] [CrossRef]
- Labrousse, V.F.; Leyrolle, Q.; Amadieu, C.; Aubert, A.; Sere, A.; Coutureau, E.; Grégoire, S.; Bretillon, L.; Pallet, V.; Gressens, P.; et al. Dietary omega-3 deficiency exacerbates inflammation and reveals spatial memory deficits in mice exposed to lipopolysaccharide during gestation. Brain Behav. Immun. 2018, 73, 427–440. [Google Scholar] [CrossRef]
- Kitajka, K.; Sinclair, A.J.; Weisinger, R.S.; Weisinger, H.S.; Mathai, M.; Jayasooriya, A.P.; Halver, J.E.; Puskás, L.G. Effects of dietary omega-3 polyunsaturated fatty acids on brain gene expression. Proc. Natl. Acad. Sci. USA 2004, 101, 10931–10936. [Google Scholar] [CrossRef]
- Bhatia, H.S.; Agrawal, R.; Sharma, S.; Huo, Y.X.; Ying, Z.; Gomez-Pinilla, F. Omega-3 fatty acid deficiency during brain maturation reduces neuronal and behavioral plasticity in adulthood. PLoS ONE 2011, 6, e28451. [Google Scholar] [CrossRef]
- Tian, C.; Fan, C.; Liu, X.; Xu, F.; Qi, K. Brain histological changes in young mice submitted to diets with different ratios of n-6/n-3 polyunsaturated fatty acids during maternal pregnancy and lactation. Clin. Nutr. 2011, 30, 659–667. [Google Scholar] [CrossRef]
- Dragano, N.R.V.; Solon, C.; Ramalho, A.F.; de Moura, R.F.; Razolli, D.S.; Christiansen, E.; Azevedo, C.; Ulven, T.; Velloso, L.A. Polyunsaturated fatty acid receptors, GPR40 and GPR120, are expressed in the hypothalamus and control energy homeostasis and inflammation. J. Neuroinflamm. 2017, 14, 91. [Google Scholar] [CrossRef] [PubMed]
- Coti Bertrand, P.; O’Kusky, J.R.; Innis, S.M. Maternal dietary (n-3) fatty acid deficiency alters neurogenesis in the embryonic rat brain. J. Nutr. 2006, 136, 1570–1575. [Google Scholar] [CrossRef]
- Carrie, I.; Clement, M.; de Javel, D.; Frances, H.; Bourre, J.M. Specific phospholipid fatty acid composition of brain regions in mice. Effects of n-3 polyunsaturated fatty acid deficiency and phospholipid supplementation. J. Lipid Res. 2000, 41, 465–472. [Google Scholar]
- Giltay, E.J.; Gooren, L.J.G.; Toorians, A.W.F.T.; Katan, M.B.; Zock, P.L. Docosahexaenoic acid concentrations are higher in women than in men because of estrogenic effects. Am. J. Clin. Nutr. 2004, 80, 1167–1174. [Google Scholar] [CrossRef]
- Bondi, C.O.; Taha, A.Y.; Tock, J.L.; Totah, N.K.; Cheon, Y.; Torres, G.E.; Rapoport, S.I.; Moghaddam, B. Adolescent behavior and dopamine availability are uniquely sensitive to dietary omega-3 fatty acid deficiency. Biol. Psychiatry 2014, 75, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Auguste, S.; Sharma, S.; Fisette, A.; Fernandes, M.F.; Daneault, C.; Des Rosiers, C.; Fulton, S. Perinatal deficiency in dietary omega-3 fatty acids potentiates sucrose reward and diet-induced obesity in mice. Int. J. Dev. Neurosci. Off. J. Int. Soc. Dev. Neurosci. 2018, 64, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Harauma, A.; Moriguchi, T. Dietary n-3 fatty acid deficiency in mice enhances anxiety induced by chronic mild stress. Lipids 2011, 46, 409–416. [Google Scholar] [CrossRef]
- Kawakita, E.; Hashimoto, M.; Shido, O. Docosahexaenoic acid promotes neurogenesis in vitro and in vivo. Neuroscience 2006, 139, 991–997. [Google Scholar] [CrossRef]
- Moriguchi, T.; Salem, N., Jr. Recovery of brain docosahexaenoate leads to recovery of spatial task performance. J. Neurochem. 2003, 87, 297–309. [Google Scholar] [CrossRef]
- Braarud, H.C.; Markhus, M.W.; Skotheim, S.; Stormark, K.M.; Froyland, L.; Graff, I.E.; Kjellevold, M. Maternal DHA Status during Pregnancy Has a Positive Impact on Infant Problem Solving: A Norwegian Prospective Observation Study. Nutrients 2018, 10, 529. [Google Scholar] [CrossRef]
- Jensen, C.L.; Voigt, R.G.; Llorente, A.M.; Peters, S.U.; Prager, T.C.; Zou, Y.L.; Rozelle, J.C.; Turcich, M.R.; Fraley, J.K.; Anderson, R.E.; et al. Effects of early maternal docosahexaenoic acid intake on neuropsychological status and visual acuity at five years of age of breast-fed term infants. J. Pediatrics 2010, 157, 900–905. [Google Scholar] [CrossRef] [PubMed]
- Ogundipe, E.; Tusor, N.; Wang, Y.; Johnson, M.R.; Edwards, A.D.; Crawford, M.A. Randomized controlled trial of brain specific fatty acid supplementation in pregnant women increases brain volumes on MRI scans of their newborn infants. Prostaglandins Leukot. Essent. Fat. Acids 2018, 138, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Colombo, J.; Shaddy, D.J.; Gustafson, K.; Gajewski, B.J.; Thodosoff, J.M.; Kerling, E.; Carlson, S.E. The Kansas University DHA Outcomes Study (KUDOS) clinical trial: Long-term behavioral follow-up of the effects of prenatal DHA supplementation. Am. J. Clin. Nutr. 2019, 109, 1380–1392. [Google Scholar] [CrossRef] [PubMed]
- Ostadrahimi, A.; Salehi-Pourmehr, H.; Mohammad-Alizadeh-Charandabi, S.; Heidarabady, S.; Farshbaf-Khalili, A. The effect of perinatal fish oil supplementation on neurodevelopment and growth of infants: A randomized controlled trial. Eur. J. Nutr. 2018, 57, 2387–2397. [Google Scholar] [CrossRef]
- Mulder, K.A.; Elango, R.; Innis, S.M. Fetal DHA inadequacy and the impact on child neurodevelopment: A follow-up of a randomised trial of maternal DHA supplementation in pregnancy. Br. J. Nutr. 2018, 119, 271–279. [Google Scholar] [CrossRef]
- Smuts, C.M.; Borod, E.; Peeples, J.M.; Carlson, S.E. High-DHA eggs: Feasibility as a means to enhance circulating DHA in mother and infant. Lipids 2003, 38, 407–414. [Google Scholar] [CrossRef]
- Judge, M.P.; Harel, O.; Lammi-Keefe, C.J. A docosahexaenoic acid-functional food during pregnancy benefits infant visual acuity at four but not six months of age. Lipids 2007, 42, 117–122. [Google Scholar] [CrossRef]
- Judge, M.P.; Harel, O.; Lammi-Keefe, C.J. Maternal consumption of a docosahexaenoic acid-containing functional food during pregnancy: Benefit for infant performance on problem-solving but not on recognition memory tasks at age 9 mo. Am. J. Clin. Nutr. 2007, 85, 1572–1577. [Google Scholar] [CrossRef]
- Atalah, S.E.; Araya, B.M.; Rosselot, P.G.; Araya, L.H.; Vera, A.G.; Andreu, R.R.; Barba, G.C.; Rodriguez, L. Consumption of a DHA-enriched milk drink by pregnant and lactating women, on the fatty acid composition of red blood cells, breast milk, and in the newborn. Arch. Latinoam. Nutr. 2009, 59, 271–277. [Google Scholar]
- Echeverria, F.; Valenzuela, R.; Catalina Hernandez-Rodas, M.; Valenzuela, A. Docosahexaenoic acid (DHA), a fundamental fatty acid for the brain: New dietary sources. Prostaglandins Leukot. Essent. Fat. Acids 2017, 124, 1–10. [Google Scholar] [CrossRef]
- Eilander, A.; Hundscheid, D.C.; Osendarp, S.J.; Transler, C.; Zock, P.L. Effects of n-3 long chain polyunsaturated fatty acid supplementation on visual and cognitive development throughout childhood: A review of human studies. Prostaglandins Leukot. Essent. Fat. Acids 2007, 76, 189–203. [Google Scholar] [CrossRef] [PubMed]
- Jasani, B.; Simmer, K.; Patole, S.K.; Rao, S.C. Long chain polyunsaturated fatty acid supplementation in infants born at term. Cochrane Database Syst. Rev. 2017, 3, CD000376. [Google Scholar] [CrossRef] [PubMed]
- Khandelwal, S.; Swamy, M.K.; Patil, K.; Kondal, D.; Chaudhry, M.; Gupta, R.; Divan, G.; Kamate, M.; Ramakrishnan, L.; Bellad, M.B.; et al. The impact of DocosaHexaenoic Acid supplementation during pregnancy and lactation on Neurodevelopment of the offspring in India (DHANI): Trial protocol. BMC Pediatrics 2018, 18, 261. [Google Scholar] [CrossRef]
- Khandelwal, S.; Kondal, D.; Chaudhry, M.; Patil, K.; Swamy, M.K.; Metgud, D.; Jogalekar, S.; Kamate, M.; Divan, G.; Gupta, R.; et al. Effect of Maternal Docosahexaenoic Acid (DHA) Supplementation on Offspring Neurodevelopment at 12 Months in India: A Randomized Controlled Trial. Nutrients 2020, 12, 3041. [Google Scholar] [CrossRef]
- Clandinin, M.T.; Van Aerde, J.E.; Merkel, K.L.; Harris, C.L.; Springer, M.A.; Hansen, J.W.; Diersen-Schade, D.A. Growth and development of preterm infants fed infant formulas containing docosahexaenoic acid and arachidonic acid. J. Pediatrics 2005, 146, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Baack, M.L.; Puumala, S.E.; Messier, S.E.; Pritchett, D.K.; Harris, W.S. Daily Enteral DHA Supplementation Alleviates Deficiency in Premature Infants. Lipids 2016, 51, 423–433. [Google Scholar] [CrossRef]
| Subject, Sample Size, Location | Dosages, Duration | Primary Outcome | References |
|---|---|---|---|
| Pregnant women, n = 350, USA | DHA 600 mg per day, <20 wk to delivery | Gestational duration ↑ Birth size ↑ | Carlson et al., 2013 [45] |
| Pregnant women, n = 315, Germany, and others | DHA 500 mg and EPA 150 mg per day, <20 wk to delivery | Visual coordination 2.5 yr. children ↑ Cognitive development 5.5 yr children↑ | Dunstan et al., 2008 Escolano et al., 2011 [52] |
| Pregnant women, n = 300, UK | DHA 300 mg, EPA 42 mg, ARA 8.4 mg per day for 12 wks from the third trimester | MRI of infant (n = 86) at birth show a correlation with DHA and brain volume ↑ | Ogundipe et al., 2018 [163] |
| Pregnant women, n = 271, Canada | DHA 400 mg per day, 16 wk to delivery | Maternal DHA correlates with language and short-term memory development of 5.79 yr children ↑ | Mulder et al., 2018 [166] |
| Pregnant women, n = 1094, Mexico | DHA 400 mg per day, 18–22 wk to delivery | Birth size and head circumference at birth ↑ The attention of 5 yr pre-school children↑ | Ramkrishnan et al., 2010 [113] |
| Pregnant women, n = 2399, Australia | DHA 800 mg per day, <21 wk to delivery | No effects on cognitive and language development in 1.2 yr infant | Makrides et al., 2010 [17] |
| Pregnant women, n = 301, USA | DHA 600 mg per day, 14.5 wk to delivery | Cognitive behavior 10 mo to 6 yr; Visual attention ↑ No long-term beneficial effects | Colombo et al., 2019 [164] |
| Pregnant women, n = 143, Norway | DHA 1183 mg and EPA 803 mg per 10 mL per day, 18 wk to post-delivery 3 mo | Mental processing score at 4 and 7 yr age ↑ No effects on BMI at 7 yr age | Helland et al., 2003 [65] |
| Pregnant women, n = 150, Iran | DHA 120 mg and EPA 180 mg per day, 20 wk to post-delivery 1 mo. | Primary neurodevelopment outcome of 4–6 mo Infant ↑ | Ostadrahim et al., 2018 [165] |
| Pregnant women, n = 98, Australia | DHA 2200 mg and EPA 1100 mg per day, 20 wk to delivery | Visual and coordination 2.5 yr children ↑ | Dunstan et al., 2008 [52] |
| Pregnant women, n = 30, USA | DHA 214 mg as a functional food, 24 wk to delivery | Visual acuity 4 mo infant ↑ | Judge et al., 2007 [168] |
| Subject, Sample Size, Location | Dosages, Duration | Measured Outcome | References |
|---|---|---|---|
| Term formula-fed infant, n = 343, USA | DHA (0.32%–0.96%), ARA (0.64%) from 1–9 day to 1 yr | DHA (0.32%) group visual acuity ↑ | Birch et al., 2010 [73] |
| Term infant, n = 420, Australia | DHA 250 mg and EPA 60 mg per day, birth to 6 mo | Accretion of DHA ↑ Early development of language and communication skills ↑ | Meldrum et al., 2012 [76] |
| Infant BW < 1.5 kg, n = 141, Norway | DHA 32 mg, ARA 31 mg per 100 mL human milk per day, 1 to 9 wk after birth | Memory recognition and problem-solving skills of 6 mo infant ↑ | Henriksen et al., 2008 [74] |
| Pre-term infant, n = 107, UK | DHA (0.5%) in the supplemented formula from birth to 9 mo | Verbal and intellectual coefficient of 9 yr girl ↑ | Isaacs et al., 2011 [77] |
| Pre-school healthy children, n = 175, USA | DHA 400 mg per day for 4 mo of 4-yr-old children | Higher blood DHA correlates comprehension, punctuation, and vocabulary abilities ↑ | Ryan et al., 2008 [85] |
| Indigenous school children three to13 yr, n = 409, Australia | DHA 750 mg, GLA 60 mg per day for 20 school week | Scholar performance in 7–12 yr children ↑ | Parletta et al., 2013 [86] |
| Term infant, n = 227, USA | Algal DHA 200 mg per day for 4 mo after delivery | Cognitive abilities in 5 yr children ↑ | Jensen et al., 2010 [162] |
| Pre-term infant, n = 361, USA | Algal DHA oil (17 mg/100 kcal), fungal ARA oil (34 mg/100 mL) from birth to 4 mo | Growth and development of pre-term infant till 118 wk ↑ | Clandinin et al., 2005 [176] |
| Pre-term 23–24 wk infant, n = 90, USA | Algal DHA 50 mg per day from 1 to 6–7 wk at discharge | DHA levels of pre-term infant comparable to term placebo ↑ | Baack et Al, 2016 [177] |
| ADHD 7–12 yr children, n = 90, Australia | DHA 1032 mg or 108 mg, EPA 264 mg or 1109 mg for 4 mo | Reading and spelling correlated DHA levels in the blood ↑ | Milte et Al, 2012 [87] |
| Larger pre-term 30–37 wk, BW > 2 kg, n = 27, Taiwan | DHA (0.05%), ARA(0.1%) infant formula for 6 mo after birth | Mental development index at 6–12 mo ↑ | Fang et Al, 2005 [75] |
| Breastfed 6 mo infant, n = 25–26, USA | DHA 115 mg/100 g baby food from 6 mo to 12 mo of the breastfed infant | Maturation of retina and visual cortex at 12 mo ↑ | Hoffman et al., 204 [69] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Basak, S.; Mallick, R.; Duttaroy, A.K. Maternal Docosahexaenoic Acid Status during Pregnancy and Its Impact on Infant Neurodevelopment. Nutrients 2020, 12, 3615. https://doi.org/10.3390/nu12123615
Basak S, Mallick R, Duttaroy AK. Maternal Docosahexaenoic Acid Status during Pregnancy and Its Impact on Infant Neurodevelopment. Nutrients. 2020; 12(12):3615. https://doi.org/10.3390/nu12123615
Chicago/Turabian StyleBasak, Sanjay, Rahul Mallick, and Asim K. Duttaroy. 2020. "Maternal Docosahexaenoic Acid Status during Pregnancy and Its Impact on Infant Neurodevelopment" Nutrients 12, no. 12: 3615. https://doi.org/10.3390/nu12123615
APA StyleBasak, S., Mallick, R., & Duttaroy, A. K. (2020). Maternal Docosahexaenoic Acid Status during Pregnancy and Its Impact on Infant Neurodevelopment. Nutrients, 12(12), 3615. https://doi.org/10.3390/nu12123615







