Effect of Maternal Docosahexaenoic Acid (DHA) Supplementation on Offspring Neurodevelopment at 12 Months in India: A Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Randomization and Masking
2.4. Procedures
2.5. Outcomes
2.6. Biomarkers
2.7. Statistical Analysis
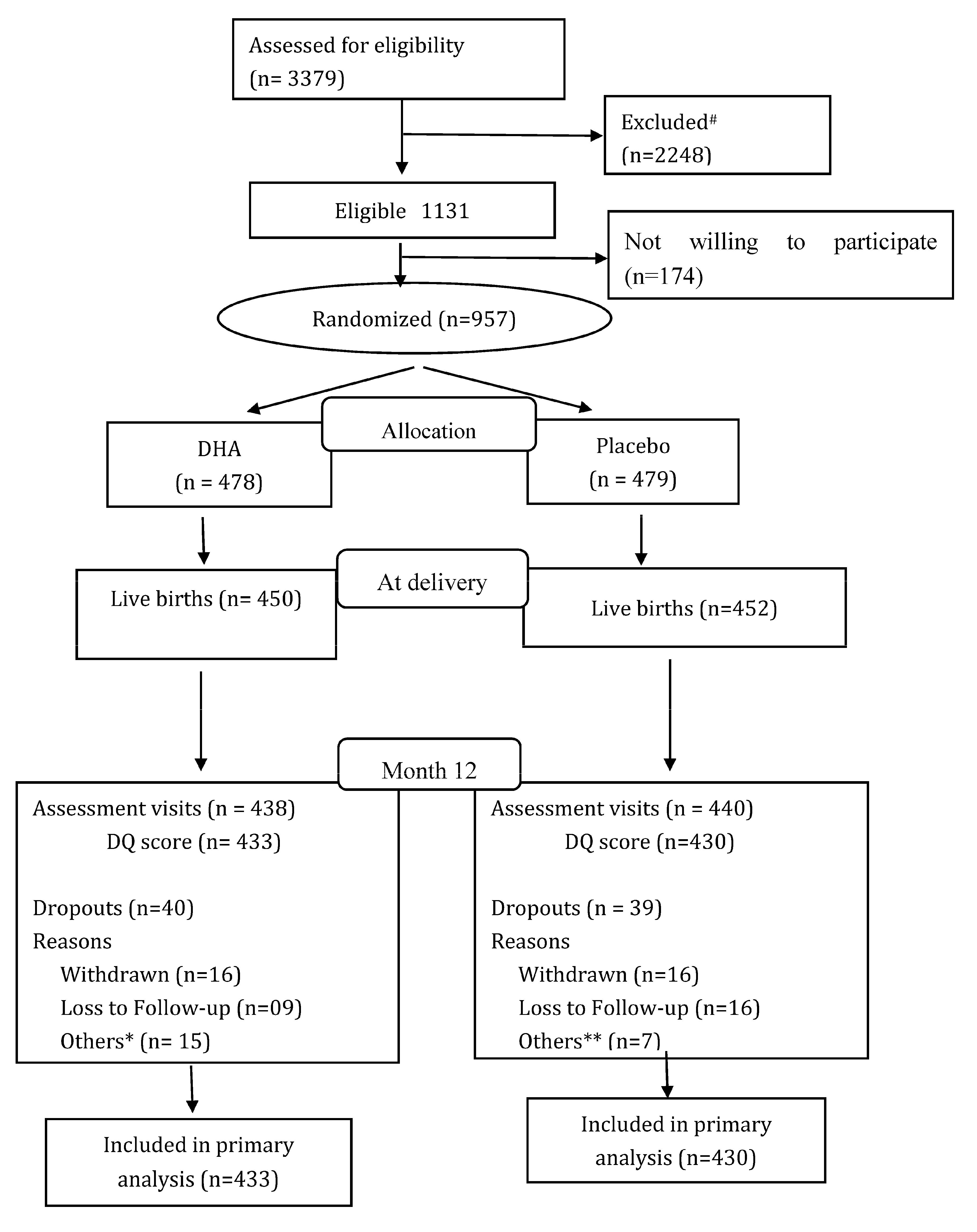
3. Results
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Sharing Statement
References
- Grantham-McGregor, S.; Cheung, Y.B.; Cueto, S.; Glewwe, P.; Richter, L.; Strupp, B. International Child Development Steering Group. Developmental potential in the first 5 years for children in developing countries. Lancet 2007, 369, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Nardelli, J. Cellular and molecular introduction to brain development. Neurobiol. Dis. 2016, 92 Pt A, 3–17. [Google Scholar] [CrossRef]
- Cusick, S.; Georgieff, M.K. The Role of Nutrition in Brain Development: The Golden Opportunity of the “First 1000 Days”. J. Pediatr. 2016, 175, 16–21. [Google Scholar] [CrossRef] [PubMed]
- Costello, A.M.D.L.; Osrin, D. Micronutrient Status during Pregnancy and Outcomes for Newborn Infants in Developing Countries. J. Nutr. 2003, 133 (Suppl. 2), 1757S–1764S. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Larqué, E.; Demmelmair, H.; Koletzko, B. Perinatal supply and metabolism of long-chain polyunsaturated fatty acids: Importance for the early development of the nervous system. Ann. New York Acad. Sci. 2002, 967, 299–310. [Google Scholar]
- Rahmanifar, A.; Kirksey, A.; Wachs, T.D.; McCabe, G.P.; Bishry, Z.; Galal, O.M.; Harrison, G.G.; Jerome, N.W. Diet during lactation associated with infant behavior and caregiver-infant interaction in a semirural Egyptian village. J. Nutr. 1993, 123, 164–175. [Google Scholar]
- Meldrum, S.; Karen, S. Docosahexaenoic Acid and Neurodevelopmental Outcomes of Term Infants. Ann. Nutr. Metab. 2016, 69 (Suppl. 1), 22–28. [Google Scholar] [CrossRef] [PubMed]
- Makrides, M. DHA supplementation during the perinatal period and neurodevelopment: Do some babies benefit more than others? Prostaglandins Leukot. Essent. Fat. Acids 2013, 88, 87–90. [Google Scholar] [CrossRef]
- Ryan, A.S.; Astwood, J.D.; Gautier, S.; Kuratko, C.N.; Nelson, E.B.; Salem, N. Effects of long-chain polyunsaturated fatty acid supplementation on neurodevelopment in childhood: A review of human studies. Prostaglandins Leukot. Essent. Fat. Acids 2010, 82, 305–314. [Google Scholar] [CrossRef]
- Makrides, M.; Smithers, L.G.; Gibson, R. Role of Long-Chain Polyunsaturated Fatty Acids in Neurodevelopment and Growth. Nestle Nutr. Workshop Ser. Pediatr Program 2010, 65, 123–136. [Google Scholar]
- Hibbeln, J.R.; Davis, J.M.; Steer, C.; Emmett, P.; Rogers, I.; Williams, C.; Golding, J. Maternal seafood consumption in pregnancy and neurodevelopmental outcomes in childhood (ALSPAC study): An observational cohort study. Lancet 2007, 369, 578–585. [Google Scholar] [CrossRef]
- Docosahexaenoic acid (DHA). Monograph. Altern. Med. Rev. 2009, 14, 391–399. [Google Scholar]
- Lauritzen, L.; Brambilla, P.; Mazzocchi, A.; Harsløf, L.B.S.; Ciappolino, V.; Agostoni, C. DHA Effects in Brain Development and Function. Nutrents 2016, 8, 6. [Google Scholar] [CrossRef]
- Carlson, S.E. Docosahexaenoic acid supplementation in pregnancy and lactation. Am. J. Clin. Nutr. 2009, 89, 678s–684s. [Google Scholar] [CrossRef]
- Makrides, M.; Gibson, R. Long-chain polyunsaturated fatty acid requirements during pregnancy and lactation. Am. J. Clin. Nutr. 2000, 71 (Suppl. 1), 307S–311S. [Google Scholar] [CrossRef]
- Koletzko, B.; Boey, C.C.; Campoy, C.; Carlson, S.E.; Chang, N.; Guillermo-Tuazon, M.A.; Joshi, S.; Prell, C.; Quak, S.H.; Sjarif, D.R.; et al. Current Information and Asian Perspectives on Long-Chain Polyunsaturated Fatty Acids in Pregnancy, Lactation, and Infancy: Systematic Review and Practice Recommendations from an Early Nutrition Academy Workshop. Ann. Nutr. Metab. 2014, 65, 49–80. [Google Scholar] [CrossRef]
- Weiser, M.J.; Butt, C.M.; Mohajeri, M.H. Docosahexaenoic Acid and Cognition throughout the Lifespan. Nutrients 2016, 8, 99. [Google Scholar] [CrossRef]
- Garg, P.; Pejaver, R.K.; Sukhija, M.; Ahuja, A. Role of DHA, ARA, & phospholipids in brain development: An Indian perspective. Clin. Epidemiology Glob. Health 2017, 5, 155–162. [Google Scholar] [CrossRef]
- Brenna, J.T. Animal studies of the functional consequences of suboptimal polyunsaturated fatty acid status during pregnancy, lactation and early post-natal life. Matern. Child Nutr. 2011, 7 (Suppl. 2), 59–79. [Google Scholar] [CrossRef]
- Wadhwani, N.; Patil, V.; Joshi, S.R. Maternal long chain polyunsaturated fatty acid status and pregnancy complications. Prostaglandins Leukot. Essent. Fat. Acids 2018, 136, 143–152. [Google Scholar] [CrossRef]
- Wierzejska, R.; Jarosz, M.; Wojda, B.; Siuba-Strzelińska, M. Dietary intake of DHA during pregnancy: A significant gap between the actual intake and current nutritional recommendations. Roczniki Państwowego Zakładu Higieny 2018, 69, 381–386. [Google Scholar] [CrossRef]
- Kris-Etherton, P.M.; Grieger, J.A.; Etherton, T.D. Dietary reference intakes for DHA and EPA. Prostaglandins Leukot. Essent. Fat. Acids 2009, 81, 99–104. [Google Scholar] [CrossRef]
- Huffman, S.L.; Harika, R.K.; Eilander, A.; Osendarp, S.J. Essential fats: How do they affect growth and development of infants and young children in developing countries? A literature review. Matern. Child Nutr. 2011, 7 (Suppl. 3), 44–65. [Google Scholar] [CrossRef]
- Shrimpton, R.; Huffman, S.L.; Zehner, E.R.; Darnton-Hill, I.; Dalmiya, N. Multiple Micronutrient Supplementation during Pregnancy in Developing-Country Settings: Policy and Program Implications of the Results of a Meta-Analysis. Food Nutr. Bull. 2009, 30 (Suppl. 4), S556–S573. [Google Scholar] [CrossRef]
- Muthayya, S.; Dwarkanath, P.; Thomas, T.; Ramprakash, S.; Mehra, R.; Mhaskar, A.; Mhaskar, R.; Thomas, A.; Bhat, S.; Vaz, M.; et al. The effect of fish and ω-3 LCPUFA intake on low birth weight in Indian pregnant women. Eur. J. Clin. Nutr. 2009, 63, 340–346. [Google Scholar] [CrossRef]
- Rees, A.; Sirois, S.; Wearden, A. Prenatal maternal docosahexaenoic acid intake and infant information processing at 4.5mo and 9mo: A longitudinal study. PLoS ONE 2019, 14, e0210984. [Google Scholar] [CrossRef]
- Colombo, J.; Shaddy, D.J.; Gustafson, K.M.; Gajewski, B.J.; Thodosoff, J.M.; Kerling, E.H.; Carlson, S.E. The Kansas University DHA Outcomes Study (KUDOS) clinical trial: Long-term behavioral follow-up of the effects of prenatal DHA supplementation. Am. J. Clin. Nutr. 2019, 109, 1380–1392. [Google Scholar] [CrossRef]
- Argaw, A.; Huybregts, L.; Wondafrash, M.; Kolsteren, P.; Belachew, T.; Worku, B.N.; Abessa, T.G.; Bouckaert, K.P. Neither n-3 Long-Chain PUFA Supplementation of Mothers through Lactation nor of Offspring in a Complementary Food Affects Child. Overall or Social-Emotional Development: A 2 × 2 Factorial Randomized Controlled Trial in Rural Ethiopia. J. Nutr. 2019, 149, 505–512. [Google Scholar] [CrossRef]
- Gould, J.F.; Yelland, L.N.; Smithers, L.G.; Makrides, M.; Treyvaud, K.; Anderson, V.; McPhee, A. Seven-Year Follow-up of Children Born to Women in a Randomized Trial of Prenatal DHA Supplementation. JAMA 2017, 317, 1173–1175. [Google Scholar] [CrossRef]
- Ramakrishnan, U.; Gonzalez-Casanova, I.; Schnaas, L.; DiGirolamo, A.; Quezada, A.D.; Pallo, B.C.; Hao, W.; Neufeld, L.M.; Rivera, J.A.; Stein, A.D.; et al. Prenatal supplementation with DHA improves attention at 5 y of age: A randomized controlled trial. Am. J. Clin. Nutr. 2016, 104, 1075–1082. [Google Scholar] [CrossRef]
- Makrides, M.; Gould, J.F.; Gawlik, N.R.; Yelland, L.N.; Smithers, L.G.; Anderson, P.J.; Gibson, R.A. Four-Year Follow-up of Children Born to Women in a Randomized Trial of Prenatal DHA Supplementation. JAMA 2014, 311, 1802–1804. [Google Scholar] [CrossRef]
- Carlson, S.E.; Colombo, J.; Gajewski, B.J.; Gustafson, K.M.; Mundy, D.; Yeast, J.; Georgieff, M.K.; Markley, L.A.; Kerling, E.H.; Shaddy, D.J. DHA supplementation and pregnancy outcomes. Am. J. Clin. Nutr. 2013, 97, 808–815. [Google Scholar] [CrossRef]
- Escolano-Margarit, M.V.; Ramos, R.; Beyer, J.; Csábi, G.; Parrilla-Roure, M.; Cruz, F.; Pérez-García, M.; Hadders-Algra, M.; Gil, Á; Decsi, T.; et al. Prenatal DHA Status and Neurological Outcome in Children at Age 5.5 Years Are Positively Associated. J. Nutr. 2011, 141, 1216–1223. [Google Scholar] [CrossRef]
- Ramakrishnan, U.; Stein, A.D.; Parra-Cabrera, S.; Wang, M.; Imhoff-Kunsch, B.; Juárez-Márquez, S.; Rivera, J.; Martorell, R. Effects of Docosahexaenoic Acid Supplementation During Pregnancy on Gestational Age and Size at Birth: Randomized, Double-Blind, Placebo-Controlled Trial in Mexico. Food Nutr. Bull. 2010, 31, S108–S116. [Google Scholar] [CrossRef]
- Gustafson, K.M.; Liao, K.; Mathis, N.B.; Shaddy, D.J.; Kerling, E.H.; Christifano, D.N.; Colombo, J.; Carlson, S.E. Prenatal docosahexaenoic acid supplementation has long-term effects on childhood behavioral and brain responses during performance on an inhibitory task. Nutr. Neurosci. 2020, 1–11. [Google Scholar] [CrossRef]
- Newberry, S.J.; Chung, M.; Booth, M.; Maglione, M.A.; Tang, A.M.; O’Hanlon, C.E.; Wang, D.D.; Okunogbe, A.; Huang, C.; Motala, A.; et al. Omega-3 Fatty Acids and Maternal and Child Health: An Updated Systematic Review. Évid. Rep. Assess. 2016, 2016, 1–826. [Google Scholar] [CrossRef][Green Version]
- Bergmann, R.L.; Haschke-Becher, E.; Klassen-Wigger, P.; Bergmann, K.E.; Richter, R.; Dudenhausen, J.W.; Grathwohl, D.; Haschke, F. Supplementation with 200 mg/day docosahexaenoic acid from mid-pregnancy through lactation improves the docosahexaenoic acid status of mothers with a habitually low fish intake and of their infants. Ann. Nutr. Metab. 2008, 52, 157–166. [Google Scholar] [CrossRef]
- Khandelwal, S.; Swamy, M.K.; Patil, K.; Kondal, D.; Chaudhry, M.; Gupta, R.; Divan, G.; Kamate, M.; Ramakrishnan, L.; Bellad, M.B.; et al. The impact of DocosaHexaenoic Acid supplementation during pregnancy and lactation on Neurodevelopment of the offspring in India (DHANI): Trial protocol. BMC Pediatr. 2018, 18, 261. [Google Scholar] [CrossRef]
- Longvah, T.; Bhaskarachary, K.; Venkaiah, K.; Longvah, T.; Aṉantaṉ, I. Indian food Composition Tables; National Institute of Nutrition: Telangana, India, 2017. [Google Scholar]
- Patni, B. Developmental Assessment Scales for Indian Infants (DASII). Ind. J. Prac. Pediatr. 2012, 14, 409–412. [Google Scholar]
- Ferreri, C.; Masi, A.; Sansone, A.; Giacometti, G.; LaRocca, A.V.; Menounou, G.; Scanferlato, R.; Tortorella, S.; Rota, D.; Conti, M.; et al. Fatty Acids in Membranes as Homeostatic, Metabolic and Nutritional Biomarkers: Recent Advancements in Analytics and Diagnostics. Diagnostics 2016, 7, 1. [Google Scholar] [CrossRef]
- Matthan, N.R.; Ooi, E.M.; Van Horn, L.; Neuhouser, M.L.; Woodman, R.; Lichtenstein, A.H. Plasma Phospholipid Fatty Acid Biomarkers of Dietary Fat Quality and Endogenous Metabolism Predict Coronary Heart Disease Risk: A Nested Case-Control Study Within the Women’s Health Initiative Observational Study. J. Am. Hear. Assoc. 2014, 3, e000764. [Google Scholar] [CrossRef]
- Rose, H.G.; Oklander, M. Improved Procedure for The Extraction of Lipids from Human Erythrocytes. J. Lipid Res. 1965, 6, 428–431. [Google Scholar]
- Lepage, G.; Roy, C.C. Direct transesterification of all classes of lipids in a one-step reaction. J. Lipid Res. 1986, 27, 114–120. [Google Scholar]
- Finley, D.A.; Lönnerdal, B.; Dewey, K.G.; E Grivetti, L. Breast milk composition: Fat content and fatty acid composition in vegetarians and non-vegetarians. Am. J. Clin. Nutr. 1985, 41, 787–800. [Google Scholar] [CrossRef]
- Ramakrishnan, U.; Stinger, A.; DiGirolamo, A.M.; Martorell, R.; Neufeld, L.M.; Rivera, J.A.; Schnaas, L.; Stein, A.D.; Wang, M. Prenatal Docosahexaenoic Acid Supplementation and Offspring Development at 18 Months: Randomized Controlled Trial. PLoS ONE 2015, 10, e0120065. [Google Scholar] [CrossRef]
- Van Der Merwe, L.F.; Moore, S.E.; Fulford, A.J.; Halliday, K.E.; Drammeh, S.; Young, S.; Prentice, A.M. Long-chain PUFA supplementation in rural African infants: A randomized controlled trial of effects on gut integrity, growth, and cognitive development. Am. J. Clin. Nutr. 2013, 97, 45–57. [Google Scholar] [CrossRef]
- Van Goor, S.A.; Dijck-Brouwer, D.J.; Erwich, J.J.H.; Schaafsma, A.; Hadders-Algra, M. The influence of supplemental docosahexaenoic and arachidonic acids during pregnancy and lactation on neurodevelopment at eighteen months. Prostaglandins Leukot. Essent. Fat. Acids 2011, 84, 139–146. [Google Scholar] [CrossRef]
- Helland, I.B.; Smith, L.; Sareem, K.; Saugstad, O.D.; Drevon, C.A. Maternal supplementation with very-long-chain n-3 fatty acids during pregnancy and lactation augments children’s IQ at 4 years of age. Pediatrics 2003, 111, e39–e44. [Google Scholar]
- Makrides, M.; Gibson, R.; McPhee, A.J.; Yelland, L.; Quinlivan, J.; Ryan, P. Effect of DHA Supplementation During Pregnancy on Maternal Depression and Neurodevelopment of Young Children. JAMA 2010, 304, 1675–1683. [Google Scholar] [CrossRef]
- Colombo, J.; Carlson, S.E.; Cheatham, C.L.; Gustafson, K.M.; Kepler, A.; Doty, T. Long-chain polyunsaturated fatty acid supplementation in infancy reduces heart rate and positively affects distribution of attention. Pediatr. Res. 2011, 70, 406–410. [Google Scholar] [CrossRef]
- Morton, S.U.; Vyas, R.; Gagoski, B.; Vu, C.; Litt, J.; Larsen, R.J.; Kuchan, M.J.; Lasekan, J.B.; Sutton, B.P.; Grant, P.E.; et al. Maternal Dietary Intake of Omega-3 Fatty Acids Correlates Positively with Regional Brain Volumes in 1-Month-Old Term Infants. Cereb. Cortex 2020, 30, 2057–2069. [Google Scholar] [CrossRef] [PubMed]
- Colombo, J.; Gustafson, K.M.; Gajewski, B.J.; Shaddy, D.J.; Kerling, E.H.; Thodosoff, J.M.; Doty, T.; Brez, C.C.; Carlson, S.E. Prenatal DHA supplementation and infant attention. Pediatr. Res. 2016, 80, 656–662. [Google Scholar] [CrossRef] [PubMed]
- Braarud, H.C.; Markhus, M.W.; Skotheim, S.; Stormark, K.M.; Frøyland, L.; Graff, I.E.; Kjellevold, M. Maternal DHA Status during Pregnancy Has a Positive Impact on Infant Problem Solving: A Norwegian Prospective Observation Study. Nutrients 2018, 10, 529. [Google Scholar] [CrossRef]
- Gould, J.F.; Smithers, L.G.; Makrides, M. The effect of maternal omega-3 (n-3) LCPUFA supplementation during pregnancy on early childhood cognitive and visual development: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2013, 97, 531–544. [Google Scholar] [CrossRef] [PubMed]
- Colombo, J.; Carlson, S.E. Is the measure the message: The BSID and nutritional interventions. Pediatrics 2012, 129, 1166–1167. [Google Scholar] [CrossRef] [PubMed]
- Jensen, C.L.; Voigt, R.G.; Llorente, A.M.; Peters, S.U.; Prager, T.C.; Zou, Y.L.; Rozelle, J.C.; Turcich, M.R.; Fraley, J.K.; Anderson, R.E.; et al. Effects of Early Maternal Docosahexaenoic Acid Intake on Neuropsychological Status and Visual Acuity at Five Years of Age of Breast-Fed Term Infants. J. Pediatr. 2010, 157, 900–905. [Google Scholar] [CrossRef]
- Bradshaw, J.L. Developmental Disorders of the Frontostriatal System: Neuropsychological, Neuropsychiatric and Evolutionary Perspectives. Developmental Disorders of the Frontostriatal System: Neuropsychological, Neuropsychiatric and Evolutionary Perspectives; Psychology Press: New York, NY, USA, 2001. [Google Scholar]
- Cheatham, C.L.; Colombo, J.; Carlson, S.E. N-3 fatty acids and cognitive and visual acuity development: Methodologic and conceptual considerations. Am. J. Clin. Nutr. 2006, 83, 1458S–1466S. [Google Scholar] [CrossRef][Green Version]
- Klevebro, S.; Juul, S.E.; Wood, T.R. A More Comprehensive Approach to the Neuroprotective Potential of Long-Chain Polyunsaturated Fatty Acids in Preterm Infants Is Needed—Should We Consider Maternal Diet and the n-6:n-3 Fatty Acid Ratio? Front. Pediatr. 2020, 7. [Google Scholar] [CrossRef]
- Santos, D.C.C.; Angulo-Barroso, R.; Li, M.; Bian, Y.; Sturza, J.; Richards, B.; Lozoff, B. Timing, duration, and severity of iron deficiency in early development and motor outcomes at 9 months. Eur. J. Clin. Nutr. 2018, 72, 332–341. [Google Scholar] [CrossRef]
- Morse, N.L. Benefits of Docosahexaenoic Acid, Folic Acid, Vitamin D and Iodine on Foetal and Infant Brain Development and Function Following Maternal Supplementation during Pregnancy and Lactation. Nutrents 2012, 4, 799–840. [Google Scholar] [CrossRef]
- Singh, M. Essential fatty acids, DHA and human brain. Indian J. Pediatr. 2005, 72, 239–242. [Google Scholar] [CrossRef] [PubMed]
- Dyall, S.C. Long-chain omega-3 fatty acids and the brain: A review of the independent and shared effects of EPA, DPA and DHA. Front. Aging Neurosci. 2015, 7, 52. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, K.T.; Cassells, R.C.; MacAllister, J.W.; Evans, G.W. The physical environment and child development: An international review. Int. J. Psychol. 2013, 48, 437–468. [Google Scholar] [CrossRef] [PubMed]
- Susane, A.A.; Santos, D.N.; Bastos, A.C.; Pedromonico, M.R.M.; de Almeida-Filho, N.; Barreto, M.L. Family environment and child’s cognitive development: An epidemiological approach. Rev. Saude Publica 2005, 39, 1–6. [Google Scholar]
- Nguyen, P.H.; DiGirolamo, A.M.; Young, M.; Kim, N.; Nguyen, S.; Martorell, R.; Ramakrishnan, U.; Gonzalez-Casanova, I. Influences of early child nutritional status and home learning environment on child development in Vietnam. Matern. Child Nutr. 2018, 14, e12468. [Google Scholar] [CrossRef]
- Pinares-Garcia, P.; Stratikopoulos, M.; Zagato, A.; Loke, H.; Lee, J. Sex: A Significant Risk Factor for Neurodevelopmental and Neurodegenerative Disorders. Brain Sci. 2018, 8, 154. [Google Scholar] [CrossRef]
- Lemmers-Jansen, I.L.J.; Krabbendam, L.; Veltman, D.J.; Fett, A.-K.J. Boys vs. girls: Gender differences in the neural development of trust and reciprocity depend on social context. Dev. Cogn. Neurosci. 2017, 25, 235–245. [Google Scholar] [CrossRef]
| DHA (N = 478) | Placebo (N = 479) | |
|---|---|---|
| Maternal age (years), mean ± SD | 23.5 ± 3.5 | 23.6 ± 3.7 |
| Gestational age at enrollment (weeks), median (p25, p75) | 15.0 (12, 18) | 15.0 (12, 18) |
| Primigravida, n (%) | 180 (37.7%) | 206 (43.0%) |
| Education, n (%) | ||
| College graduated and above | 88 (18.4%) | 82 (17.1%) |
| High school/secondary | 371 (77.6%) | 386 (80.6%) |
| Employed, n (%) | 119 (25.0%) | 104 (22.0%) |
| Household income (>Rs 20,000), n (%) | 65 (13.6%) | 47 (9.8%) |
| Dietary habits—vegetarian, n (%) | 73 (15.3%) | 87 (18.2%) |
| Consuming fish/seafood, n (%) | 258 (53.9%) | 202 (57.8%) |
| Anthropometric measurements | ||
| Height (cm), mean ± SD | 154.1 ± 5.6 | 153.9 ± 5.7 |
| Weight (kg), mean ± SD | 48.9 ± 9.0 | 48.9 ± 8.5 |
| BMI (kg/m2), mean ± SD | 20.5 ± 3.5 | 20.7 ± 3.6 |
| MUAC, (cm), mean ± SD | 24.3 ± 3.0 | 24.3 ± 3.1 |
| Biochemical measures | ||
| Hb (gm%), mean ± SD | 11.1 ± 1.3 | 11.2 ± 1.3 |
| DHA (mol % of fatty acid) *-, mean ± SD | 0.86 ± 0.78 | 0.88 ± 0.71 |
| At Delivery | DHA N = 450 | Placebo N = 452 |
|---|---|---|
| Live births, n (%) | 450 (94.1%) | 452 (94.3%) |
| Gestational age at the time of delivery (weeks), median (p25, p 75) | 39.0 (38.0, 40.0) | 39.0 (38.0, 40.0) |
| Delivery place—study center, n (%) | 372 (84.7%) | 375 (85.2%) |
| Delivery conducted by doctor, n (%) | 416 (94.5%) | 423 (96.1%) |
| Spontaneous labor, n (%) | 410 (93.2%) | 407 (92.5%) |
| Caesarean, n (%) | 154 (35.0%) | 175 (39.8%) |
| Male child, n (%) | 234 (52.0%) | 243 (54.0%) |
| DHA n/N (%) | Placebo n/N (%) | |
|---|---|---|
| Initiation of breastfeeding in 1 h, n (%) | 346/433 (79.9%) | 357/437 (81.7%) |
| Exclusively breastfed until 6 months, n (%) | 236/426 (55.4%) | 237/421 (56.3%) |
| Age at which complementary feeding initiated (months) *, mean ± SD | 5.4 ± 0.67 | 5.3 ± 0.83 |
| DHA | Placebo | Mean Difference * [95% CI] | p-Value | |
|---|---|---|---|---|
| DHA at baseline | N = 258 0.86 ± 0.78, 0.56 (0.32, 1.21) | N = 224 0.88 ± 0.71, 0.55 (0.37, 1.28) | 0.02 [0.11,0.15] | 0.77 |
| DHA at the time of delivery | N = 271 2.03 ± 1.75, 1.41 (0.61, 2.99) | N = 242 1.12 ± 0.86, 0.83 (0.42, 1.72) | −0.91 [−1.16, 0.67] | <0.001 |
| DHA in cord blood | N = 265 2.61 ± 1.45, 2.64 (1.38, 3.81) | N = 232 1.83 ± 0.90, 1.76 (1.16, 2.48) | −0.77 [−0.99, 0.56] | <0.001 |
| DHA in infant blood at 6 months | N = 263 1.94 ± 1.42, 1.65 (0.64, 3.08) | N = 240 0.84 ± 0.56, 0.77 (0.43, 1.12) | −1.09 [−1.29, −0.9] | <0.001 |
| DHA in infant blood at 12 months | N = 227 1.71 ± 1.18, 1.50 (0.60, 2.64) | N = 204 1.27 ± 0.93, 1.03 (0.43, 1.89) | −0.44 [−0.64, −0.24] | <0.001 |
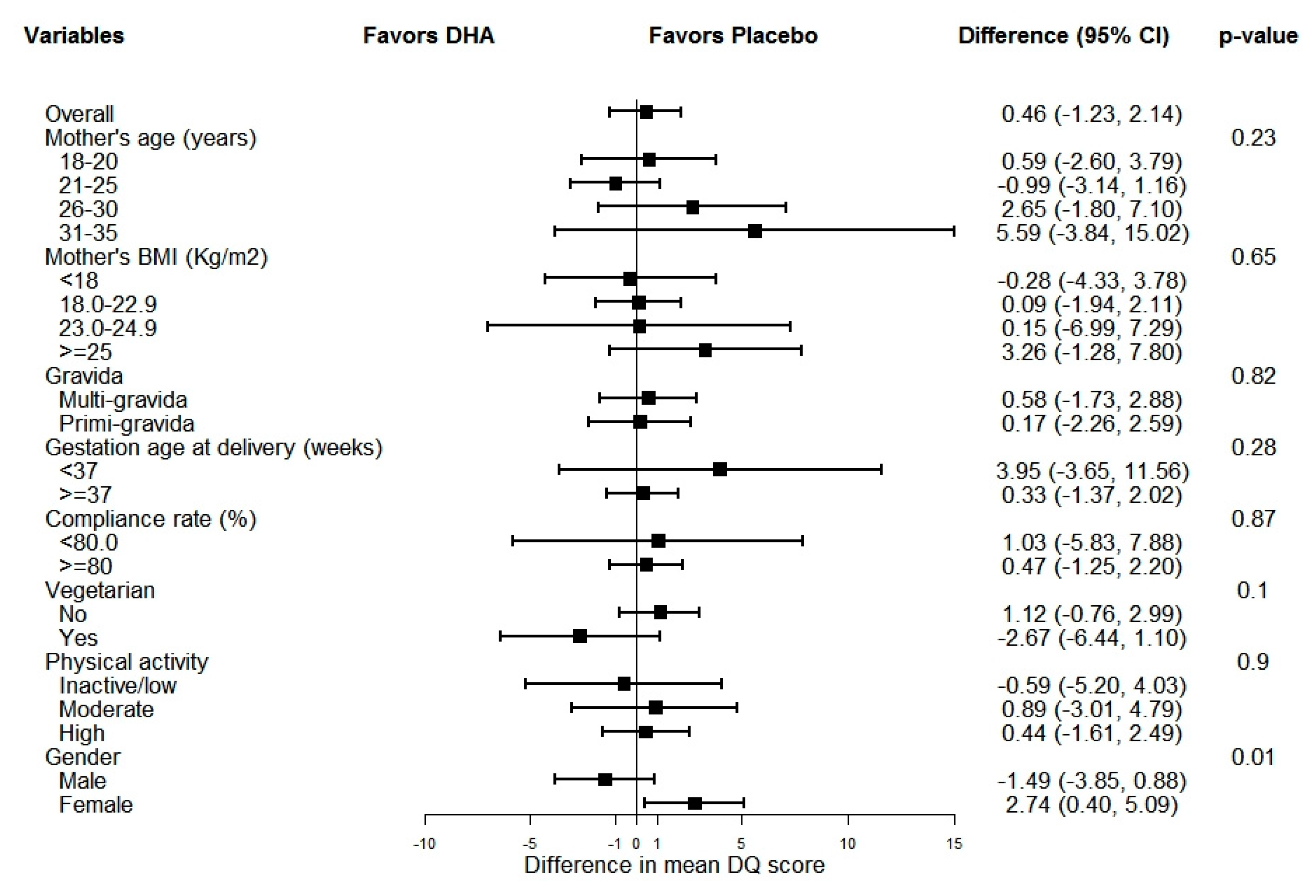
| At 12th Month | DHA (n = 433) | Placebo (N = 430) | Difference * [95% CI] | p-Value |
|---|---|---|---|---|
| DQ score | 96.6 ± 12.1 | 97.1 ± 13.0 | 0.46 [−1.23,2.14] | 0.60 |
| Motor score | 47.6 ± 3.7 | 47.6 ± 3.7 | 0.03 [−0.47,0.52] | 0.92 |
| Mental score | 106.0 ± 7.0 | 106.7 ± 7.6 | 0.63 [−0.35,1.61] | 0.21 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khandelwal, S.; Kondal, D.; Chaudhry, M.; Patil, K.; Swamy, M.K.; Metgud, D.; Jogalekar, S.; Kamate, M.; Divan, G.; Gupta, R.; et al. Effect of Maternal Docosahexaenoic Acid (DHA) Supplementation on Offspring Neurodevelopment at 12 Months in India: A Randomized Controlled Trial. Nutrients 2020, 12, 3041. https://doi.org/10.3390/nu12103041
Khandelwal S, Kondal D, Chaudhry M, Patil K, Swamy MK, Metgud D, Jogalekar S, Kamate M, Divan G, Gupta R, et al. Effect of Maternal Docosahexaenoic Acid (DHA) Supplementation on Offspring Neurodevelopment at 12 Months in India: A Randomized Controlled Trial. Nutrients. 2020; 12(10):3041. https://doi.org/10.3390/nu12103041
Chicago/Turabian StyleKhandelwal, Shweta, Dimple Kondal, Monica Chaudhry, Kamal Patil, Mallaiah Kenchaveeraiah Swamy, Deepa Metgud, Sandesh Jogalekar, Mahesh Kamate, Gauri Divan, Ruby Gupta, and et al. 2020. "Effect of Maternal Docosahexaenoic Acid (DHA) Supplementation on Offspring Neurodevelopment at 12 Months in India: A Randomized Controlled Trial" Nutrients 12, no. 10: 3041. https://doi.org/10.3390/nu12103041
APA StyleKhandelwal, S., Kondal, D., Chaudhry, M., Patil, K., Swamy, M. K., Metgud, D., Jogalekar, S., Kamate, M., Divan, G., Gupta, R., Prabhakaran, D., Tandon, N., Ramakrishnan, U., & Stein, A. D. (2020). Effect of Maternal Docosahexaenoic Acid (DHA) Supplementation on Offspring Neurodevelopment at 12 Months in India: A Randomized Controlled Trial. Nutrients, 12(10), 3041. https://doi.org/10.3390/nu12103041






