Role of Oral and Gut Microbiota in Dietary Nitrate Metabolism and Its Impact on Sports Performance
Abstract
1. Introduction
2. Nitrate Reduction in the Oral Cavity: The Role of Oral Microbiota
3. Gastrointestinal Implications of Dietary Nitrate: A Potential Role of the Gut Microbiota
4. Nitrate and the Oral and Gut-Muscle Axis: Potential Ergogenic Effects
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Engemann, A.; Focke, C.; Humpf, H.-U. Intestinal Formation ofN-Nitroso Compounds in the Pig Cecum Model. J. Agric. Food Chem. 2013, 61, 998–1005. [Google Scholar] [CrossRef] [PubMed]
- De Lima Bezerra, Á.D.; Costa, E.C.; Pacheco, D.A.; Souza, D.C.; Farias-Junior, L.F.; Ritti-Dia, R.M.; Grigolo, G.B.; de Bittencourt Júnior, P.I.H.; Krause, M.; Fayh, A.P.T. Effect of Acute Dietary Nitrate Supplementation on the Post-Exercise Ambulatory Blood Pressure in Obese Males: A Randomized, Controlled, Crossover Trial. J. Sports Sci. Med. 2019, 18, 118–127. [Google Scholar] [PubMed]
- Jones, A.M. Dietary Nitrate Supplementation and Exercise Performance. Sports Med. 2014, 44, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Petrick, J.L.; Steck, S.E.; Bradshaw, P.T.; Trivers, K.F.; Abrahamson, P.E.; Engel, L.S.; He, K.; Chow, W.-H.; Mayne, S.T.; Risch, H.A.; et al. Dietary intake of flavonoids and oesophageal and gastric cancer: Incidence and survival in the United States of America (USA). Br. J. Cancer 2015, 112, 1291–1300. [Google Scholar] [CrossRef] [PubMed]
- Song, P.; Wu, L.; Guan, W. Dietary Nitrates, Nitrites, and Nitrosamines Intake and the Risk of Gastric Cancer: A Meta-Analysis. Nutrients 2015, 7, 9872–9895. [Google Scholar] [CrossRef] [PubMed]
- EFSA. 2018. EFSA Explains Risk Assessment: Nitrites and Nitrates Added to Food. Available online: https://www.efsa.europa.eu/sites/default/files/corporate_publications/files/nitrates-nitrites-170614.pdf (accessed on 30 October 2020).
- Lundberg, J.O.; Carlström, M.; Larsen, F.J.; Weitzberg, E. Roles of dietary inorganic nitrate in cardiovascular health and disease. Cardiovasc. Res. 2011, 89, 525–532. [Google Scholar] [CrossRef] [PubMed]
- EFSA Nitrate in vegetables. Scientific Opinion of the Panel on Contaminants in the Food chain. EFSA J. 2008, 6, 689. [Google Scholar]
- McDonagh, S.T.J.; Wylie, L.J.; Thompson, C.; Vanhatalo, A.; Jones, A.M. Potential benefits of dietary nitrate ingestion in healthy and clinical populations: A brief review. Eur. J. Sport Sci. 2018, 19, 15–29. [Google Scholar] [CrossRef]
- Maughan, R.J.; Burke, L.M.; Dvorak, J.; Larson-Meyer, D.E.; Peeling, P.; Phillips, S.M.; Rawson, E.S.; Walsh, N.P.; Garthe, I.; Geyer, H.; et al. IOC consensus statement: Dietary supplements and the high-performance athlete. Br. J. Sports Med. 2018, 52, 439–455. [Google Scholar] [CrossRef]
- Australian Institute of Sport-AIS. AIS Sports Supplement Framework an Initiative of AIS Sports Nutrition Sports Gels. Available online: https://www.ais.gov.au/__data/assets/pdf_file/0004/698557/AIS-Sports-Supplement-Framework-2019.pdf (accessed on 30 October 2020).
- Valenzuela, P.L.; Morales, J.S.; Emanuele, E.; Pareja-Galeano, H.; Lucia, A. Supplements with purported effects on muscle mass and strength. Eur. J. Nutr. 2019, 58, 2983–3008. [Google Scholar] [CrossRef]
- Calvo, J.L.; Alorda-Capo, F.; Pareja-Galeano, H.; Jiménez, S.L. Influence of Nitrate Supplementation on Endurance Cyclic Sports Performance: A Systematic Review. Nutrients 2020, 12, 1796. [Google Scholar] [CrossRef] [PubMed]
- Larsen, F.J.; Schiffer, T.A.; Borniquel, S.; Sahlin, K.; Ekblom, B.; Lundberg, J.O.; Weitzberg, E. Dietary Inorganic Nitrate Improves Mitochondrial Efficiency in Humans. Cell Metab. 2011, 13, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Larsen, F.J.; Ekblom, B.; Sahlin, K.; Lundberg, J.O.; Weitzberg, E. Effects of Dietary Nitrate on Blood Pressure in Healthy Volunteers. N. Engl. J. Med. 2006, 355, 2792–2793. [Google Scholar] [CrossRef] [PubMed]
- Sobko, T.; Marcus, C.; Govoni, M.; Kamiya, S. Dietary nitrate in Japanese traditional foods lowers diastolic blood pressure in healthy volunteers. Nitric Oxide Biol. Chem. 2010, 22, 136–140. [Google Scholar] [CrossRef]
- Hughes, W.E.; Ueda, K.; Treichler, D.P.; Casey, D.P. Effects of acute dietary nitrate supplementation on aortic blood pressure and aortic augmentation index in young and older adults. Nitric Oxide Biol. Chem. 2016, 59, 21–27. [Google Scholar] [CrossRef]
- Velmurugan, S.; Gan, J.M.; Rathod, K.S.; Khambata, R.S.; Ghosh, S.M.; Hartley, A.; Van Eijl, S.; Sagi-Kiss, V.; Chowdhury, T.A.; Curtis, M.J.; et al. Dietary nitrate improves vascular function in patients with hypercholesterolemia: A randomized, double-blind, placebo-controlled study. Am. J. Clin. Nutr. 2016, 103, 25–38. [Google Scholar] [CrossRef]
- Larsen, F.J.; Weitzberg, E.; Lundberg, J.O.; Ekblom, B. Dietary nitrate reduces maximal oxygen consumption while maintaining work performance in maximal exercise. Free Radic. Biol. Med. 2010, 48, 342–347. [Google Scholar] [CrossRef]
- Bailey, S.J.; Winyard, P.; Vanhatalo, A.; Blackwell, J.R.; DiMenna, F.J.; Wilkerson, D.P.; Tarr, J.; Benjamin, N.; Jones, A.M. Dietary nitrate supplementation reduces the O2cost of low-intensity exercise and enhances tolerance to high-intensity exercise in humans. J. Appl. Physiol. 2009, 107, 1144–1155. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Moretti, C.; Benjamin, N.; Weitzberg, E. Symbiotic bacteria enhance exercise performance. Br. J. Sports Med. 2020. [Google Scholar] [CrossRef]
- McMahon, N.F.; Leveritt, M.D.; Pavey, T.G. The Effect of Dietary Nitrate Supplementation on Endurance Exercise Performance in Healthy Adults: A Systematic Review and Meta-Analysis. Sports Med. 2017, 47, 735–756. [Google Scholar] [CrossRef]
- Domínguez, R.; Cuenca, E.; Maté-Muñoz, J.L.; García-Fernández, P.; Serra-Paya, N.; Lozano-Estevan, M.D.C.; Veiga-Herreros, P.; Garnacho-Castaño, M.V. Effects of Beetroot Juice Supplementation on Cardiorespiratory Endurance in Athletes. A Systematic Review. Nutrients 2017, 9, 43. [Google Scholar] [CrossRef] [PubMed]
- Cutler, C.; Kiernan, M.; Willis, J.; Gallardo-Alfaro, L.; Casas-Agustench, P.; White, D.; Hickson, M.; Gabaldon, T.; Bescos, R. Post-exercise hypotension and skeletal muscle oxygenation is regulated by nitrate-reducing activity of oral bacteria. Free Radic. Biol. Med. 2019, 143, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Ashworth, A.; Cutler, C.; Farnham, G.; Liddle, L.; Burleigh, M.; Rodiles, A.; Sillitti, C.; Kiernan, M.; Moore, M.; Hickson, M.; et al. Dietary intake of inorganic nitrate in vegetarians and omnivores and its impact on blood pressure, resting metabolic rate and the oral microbiome. Free Radic. Biol. Med. 2019, 138, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Kapil, V.; Haydar, S.M.; Pearl, V.; Lundberg, J.O.; Weitzberg, E.; Ahluwalia, A. Physiological role for nitrate-reducing oral bacteria in blood pressure control. Free Radic. Biol. Med. 2013, 55, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Mitsui, T.; Harasawa, R. The effects of essential oil, povidone-iodine, and chlorhexidine mouthwash on salivary nitrate/nitrite and nitrate-reducing bacteria. J. Oral Sci. 2017, 59, 597–601. [Google Scholar] [CrossRef] [PubMed]
- Tribble, G.D.; Angelov, N.; Weltman, R.; Wang, B.-Y.; Eswaran, S.V.; Gay, I.C.; Parthasarathy, K.; Dao, D.-H.V.; Richardson, K.N.; Ismail, N.M.; et al. Frequency of Tongue Cleaning Impacts the Human Tongue Microbiome Composition and Enterosalivary Circulation of Nitrate. Front. Cell. Infect. Microbiol. 2019, 9, 39. [Google Scholar] [CrossRef]
- Joshipura, K.J.; Muñoz-Torres, F.; Fernández-Santiago, J.; Patel, R.P.; Lopez-Candales, A. Over-the-counter mouthwash use, nitric oxide and hypertension risk. Blood Press. 2019, 29, 103–112. [Google Scholar] [CrossRef]
- Rocha, B.; Correia, M.; Barbosa, R.; Laranjinha, J. A dietary-driven redox modulation of gut microbiome-host interactions: The rescue of epithelial barrier and mucus production during dysbiosis by dietary nitrate. Free Radic. Biol. Med. 2014, 75, S36–S37. [Google Scholar] [CrossRef]
- Nyakayiru, J.; Van Loon, L.J.; Verdijk, L. Could intramuscular storage of dietary nitrate contribute to its ergogenic effect? A mini-review. Free Radic. Biol. Med. 2020, 152, 295–300. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Weitzberg, E.; Cole, J.A.; Benjamin, N. Nitrate, bacteria and human health. Nat. Rev. Microbiol. 2004, 2, 593–602. [Google Scholar] [CrossRef]
- Scheiman, J.; Luber, J.M.; Chavkin, T.A.; Macdonald, T.; Tung, A.; Pham, L.-D.; Wibowo, M.C.; Wurth, R.C.; Punthambaker, S.; Tierney, B.T.; et al. Meta-omics analysis of elite athletes identifies a performance-enhancing microbe that functions via lactate metabolism. Nat. Med. 2019, 25, 1104–1109. [Google Scholar] [CrossRef] [PubMed]
- Ecerdá, B.; Epérez, M.; Pérez-Santiago, J.D.; Tornero-Aguilera, J.F.; Egonzález-Soltero, R.; Larrosa, M. Gut Microbiota Modification: Another Piece in the Puzzle of the Benefits of Physical Exercise in Health? Front. Physiol. 2016, 7, 51. [Google Scholar] [CrossRef]
- Sorrenti, V.; Fortinguerra, S.; Caudullo, G.; Buriani, A. Deciphering the Role of Polyphenols in Sports Performance: From Nutritional Genomics to the Gut Microbiota toward Phytonutritional Epigenomics. Nutrients 2020, 12, 1265. [Google Scholar] [CrossRef] [PubMed]
- Hezel, M.P.; Weitzberg, E. The oral microbiome and nitric oxide homoeostasis. Oral Dis. 2013, 21, 7–16. [Google Scholar] [CrossRef]
- Weitzberg, E.; Lundberg, J.O. Novel Aspects of Dietary Nitrate and Human Health. Annu. Rev. Nutr. 2013, 33, 129–159. [Google Scholar] [CrossRef]
- Farah, C.; Michel, L.Y.M.; Balligand, J.-L. Nitric oxide signalling in cardiovascular health and disease. Nat. Rev. Cardiol. 2018, 15, 292–316. [Google Scholar] [CrossRef]
- Doel, J.J.; Benjamin, N.; Hector, M.P.; Rogers, M.; Allaker, R.P. Evaluation of bacterial nitrate reduction in the human oral cavity. Eur. J. Oral Sci. 2005, 113, 14–19. [Google Scholar] [CrossRef]
- Kilian, M.; Chapple, I.L.C.; Hannig, M.; Marsh, P.D.; Meuric, V.; Pedersen, A.M.L.; Tonetti, M.S.; Wade, W.G.; Zaura, E. The oral microbiome–an update for oral healthcare professionals. Br. Dent. J. 2016, 221, 657–666. [Google Scholar] [CrossRef]
- Schreiber, F.; Stief, P.; Gieseke, A.; Heisterkamp, I.M.; Verstraete, W.; De Beer, D.; Stoodley, P. Denitrification in human dental plaque. BMC Biol. 2010, 8, 24. [Google Scholar] [CrossRef]
- Li, H.; Duncan, C.; Townend, J.; Killham, K.; Smith, L.M.; Johnston, P.; Dykhuizen, R.; Kelly, D.; Golden, M.; Benjamin, N.; et al. Nitrate-reducing bacteria on rat tongues. Appl. Environ. Microbiol. 1997, 63, 924–930. [Google Scholar] [CrossRef]
- Palmerini, C.A.; Palombari, R.; Perito, S.; Arienti, G. NO Synthesis in Human Saliva. Free Radic. Res. 2003, 37, 29–31. [Google Scholar] [CrossRef] [PubMed]
- Hyde, E.R.; Andrade, F.; Vaksman, Z.; Parthasarathy, K.; Jiang, H.; Parthasarathy, D.K.; Torregrossa, A.C.; Tribble, G.; Kaplan, H.B.; Petrosino, J.F.; et al. Metagenomic Analysis of Nitrate-Reducing Bacteria in the Oral Cavity: Implications for Nitric Oxide Homeostasis. PLoS ONE 2014, 9, e88645. [Google Scholar] [CrossRef] [PubMed]
- Mitsui, T.; Saito, M.; Harasawa, R. Salivary nitrate-nitrite conversion capacity after nitrate ingestion and incidence of Veillonella spp. in elderly individuals. J. Oral Sci. 2018, 60, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Burleigh, M.; Liddle, L.; Monaghan, C.; Muggeridge, D.J.; Sculthorpe, N.; Butcher, J.P.; Henriquez, F.L.; Allen, J.D.; Easton, C. Salivary nitrite production is elevated in individuals with a higher abundance of oral nitrate-reducing bacteria. Free Radic. Biol. Med. 2018, 120, 80–88. [Google Scholar] [CrossRef]
- Timby, N.; Domellöf, M.; Hernell, O.; Lönnerdal, B.; Nihlen, C.; Johanssson, I.; Weitzberg, E. Effects of age, sex and diet on salivary nitrate and nitrite in infants. Nitric Oxide Biol. Chem. 2019, 94, 73–78. [Google Scholar] [CrossRef]
- Hyde, E.R.; Luk, B.; Cron, S.; Kusic, L.; McCue, T.; Bauch, T.; Kaplan, H.; Tribble, G.; Petrosino, J.F.; Bryan, N.S. Characterization of the rat oral microbiome and the effects of dietary nitrate. Free Radic. Biol. Med. 2014, 77, 249–257. [Google Scholar] [CrossRef]
- Vanhatalo, A.; Blackwell, J.R.; L’Heureux, J.E.; Williams, D.W.; Smith, A.; Van Der Giezen, M.; Winyard, P.G.; Kelly, J.; Jones, A.M. Nitrate-responsive oral microbiome modulates nitric oxide homeostasis and blood pressure in humans. Free Radic. Biol. Med. 2018, 124, 21–30. [Google Scholar] [CrossRef]
- Duncan, C.; Li, H.; Dykhuizen, R.; Frazer, R.; Johnston, P.; Macknight, G.; Smith, L.; Lamza, K.; McKenzie, H.; Batt, L.; et al. Protection against oral and gastrointestinal diseases: Importance of dietary nitrate intake, oral nitrate reduction and enterosalivary nitrate circulation. Comp. Biochem. Physiol. Part A Physiol. 1997, 118, 939–948. [Google Scholar] [CrossRef]
- Lundberg, J.O.; Weitzberg, E. Biology of nitrogen oxides in the gastrointestinal tract. Gut 2013, 62, 616–629. [Google Scholar] [CrossRef]
- Björne, H.; Petersson, J.; Phillipson, M.; Weitzberg, E.; Holm, L.; Lundberg, J. Nitrite in saliva increases gastric mucosal blood flow and mucus thickness. J. Clin. Investig. 2004, 113, 106–114. [Google Scholar] [CrossRef]
- Matziouridou, C.; Rocha, S.D.C.; Haabeth, O.A.; Rudi, K.; Carlsen, H.; Kielland, A. iNOS and NOX1-dependent ROS production maintains bacterial homeostasis in the ileum of mice. Mucosal Immunol. 2018, 11, 774–784. [Google Scholar] [CrossRef] [PubMed]
- Jädert, C.; Phillipson, M.; Holm, L.; Lundberg, J.O.; Borniquel, S. Preventive and therapeutic effects of nitrite supplementation in experimental inflammatory bowel disease. Redox Biol. 2014, 2, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Tomasello, G.; Mazzola, M.; Leone, A.; Sinagra, E.; Zummo, G.; Farina, F.; Damiani, P.; Cappello, F.; Geagea, A.G.; Jurjus, A.; et al. Nutrition, oxidative stress and intestinal dysbiosis: Influence of diet on gut microbiota in inflammatory bowel diseases. Biomed. Pap. 2016, 160, 461–466. [Google Scholar] [CrossRef] [PubMed]
- Vermeiren, J.; Van De Wiele, T.; Van Nieuwenhuyse, G.; Boeckx, P.; Verstraete, W.; Boon, N. Sulfide- and nitrite-dependent nitric oxide production in the intestinal tract. Microb. Biotechnol. 2012, 5, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Roediger, W.E.W. Review article: Nitric oxide from dysbiotic bacterial respiration of nitrate in the pathogenesis and as a target for therapy of ulcerative colitis. Aliment. Pharmacol. Ther. 2008, 27, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Tiso, M.; Schechter, A.N. Nitrate reduction to nitrite, nitric oxide and ammonia by gut bacteria under physiological conditions. PLoS ONE 2015, 10, e0119712. [Google Scholar]
- Henning, S.M.; Yang, J.; Shao, P.; Lee, R.-P.; Huang, J.; Ly, A.; Hsu, M.; Lu, Q.-Y.; Thames, G.; Heber, D.; et al. Health benefit of vegetable/fruit juice-based diet: Role of microbiome. Sci. Rep. 2017, 7, 1–9. [Google Scholar] [CrossRef]
- Kina-Tanada, M.; Sakanashi, M.; Tanimoto, A.; Kaname, T.; Matsuzaki, T.; Noguchi, K.; Uchida, T.; Nakasone, J.; Kozuka, C.; Ishida, M.; et al. Long-term dietary nitrite and nitrate deficiency causes the metabolic syndrome, endothelial dysfunction and cardiovascular death in mice. Diabetologia 2017, 60, 1138–1151. [Google Scholar] [CrossRef]
- Conley, M.N.; Roberts, C.; Sharpton, T.J.; Iwaniec, U.T.; Hord, N.G. Increasing dietary nitrate has no effect on cancellous bone loss or fecal microbiome in ovariectomized rats. Mol. Nutr. Food Res. 2017, 61, 61. [Google Scholar] [CrossRef]
- Rocha, B.S.; Correia, M.G.; Pereira, A.; Henriques, I.; Da Silva, G.J.; Laranjinha, J. Inorganic nitrate prevents the loss of tight junction proteins and modulates inflammatory events induced by broad-spectrum antibiotics: A role for intestinal microbiota? Nitric Oxide Biol. Chem. 2019, 88, 27–34. [Google Scholar] [CrossRef]
- Natel, A.S.; Abdalla, A.L.; Araujo, R.; McManus, C.; Paim, T.D.P.; de Abdalla Filho, A.L.; Louvandini, H.; Nazato, C. Encapsulated nitrate replacing soybean meal changes in vitro ruminal fermentation and methane production in diets differing in concentrate to forage ratio. Anim. Sci. J. 2019, 90, 1350–1361. [Google Scholar] [CrossRef] [PubMed]
- Carlström, M.; Larsen, F.J.; Nyström, T.; Hezel, M.; Borniquel, S.; Weitzberg, E.; Lundberg, J.O. Dietary inorganic nitrate reverses features of metabolic syndrome in endothelial nitric oxide synthase-deficient mice. Proc. Natl. Acad. Sci. USA 2010, 107, 17716–17720. [Google Scholar] [CrossRef] [PubMed]
- Gusarov, I.; Nudler, E. NO-mediated cytoprotection: Instant adaptation to oxidative stress in bacteria. Proc. Natl. Acad. Sci. USA 2005, 102, 13855–13860. [Google Scholar] [CrossRef] [PubMed]
- Zumft, W.G. Cell biology and molecular basis of denitrification. Microbiol. Mol. Biol. Rev. 1997, 61, 61. [Google Scholar] [CrossRef]
- Bu, C.; Wang, Y.; Ge, C.; Ahmad, H.A.; Gao, B.; Ni, S.-Q. Dissimilatory Nitrate Reduction to Ammonium in the Yellow River Estuary: Rates, Abundance, and Community Diversity. Sci. Rep. 2017, 7, 6830. [Google Scholar] [CrossRef]
- Macfarlane, G.T.; Allison, C.; Gibson, S.A.W.; Cummings, J.H. Contribution of the microflora to proteolysis in the human large intestine. J. Appl. Bacteriol. 1988, 64, 37–46. [Google Scholar] [CrossRef]
- Mohan, S.B.; Schmid, M.; Jetten, M.; Cole, J. Detection and widespread distribution of the nrfA gene encoding nitrite reduction to ammonia, a short circuit in the biological nitrogen cycle that competes with denitrification. FEMS Microbiol. Ecol. 2004, 49, 433–443. [Google Scholar] [CrossRef]
- Kaldorf, M.; Von Berg, K.-H.L.; Meier, U.; Servos, U.; Bothe, H. The reduction of nitrous oxide to dinitrogen by Escherichia coli. Arch. Microbiol. 1993, 160, 432–439. [Google Scholar] [CrossRef]
- Rocha, B.S.; Laranjinha, J. Nitrate from diet might fuel gut microbiota metabolism: Minding the gap between redox signaling and inter-kingdom communication. Free Radic. Biol. Med. 2020, 149, 37–43. [Google Scholar] [CrossRef]
- Espey, M.G. Role of oxygen gradients in shaping redox relationships between the human intestine and its microbiota. Free Radic. Biol. Med. 2013, 55, 130–140. [Google Scholar] [CrossRef]
- Przewłócka, K.; Folwarski, M.; Kaźmierczak-Siedlecka, K.; Skonieczna-Żydecka, K.; Kaczor, J.J. Gut-Muscle AxisExists and May Affect Skeletal Muscle Adaptation to Training. Nutrients 2020, 12, 1451. [Google Scholar] [CrossRef] [PubMed]
- Lustgarten, M.S. The Role of the Gut Microbiome on Skeletal Muscle Mass and Physical Function: 2019 Update. Front. Physiol. 2019, 10, 1435. [Google Scholar] [CrossRef] [PubMed]
- Mika, A.; Van Treuren, W.; González, A.; Herrera, J.J.; Knight, R.; Fleshner, M. Exercise Is More Effective at Altering Gut Microbial Composition and Producing Stable Changes in Lean Mass in Juvenile versus Adult Male F344 Rats. PLoS ONE 2015, 10, e0125889. [Google Scholar] [CrossRef] [PubMed]
- Barton, W.; Penney, N.C.; Cronin, O.; Garcia-Perez, I.; Molloy, M.G.; Holmes, E.; Shanahan, F.; Cotter, P.D.; O’Sullivan, O. The microbiome of professional athletes differs from that of more sedentary subjects in composition and particularly at the functional metabolic level. Gut 2017, 67, 625–633. [Google Scholar] [CrossRef]
- Bressa, C.; Bailén-Andrino, M.; Pérez-Santiago, J.; González-Soltero, R.; Pérez, M.; Montalvo-Lominchar, M.G.; Maté-Muñoz, J.L.; Domínguez, R.; Moreno, D.; Larrosa, M. Differences in gut microbiota profile between women with active lifestyle and sedentary women. PLoS ONE 2017, 12, e0171352. [Google Scholar] [CrossRef]
- Okamoto, T.; Morino, K.; Ugi, S.; Nakagawa, F.; Lemecha, M.; Ida, S.; Ohashi, N.; Sato, D.; Fujita, Y.; Maegawa, H. Microbiome potentiates endurance exercise through intestinal acetate production. Am. J. Physiol. Metab. 2019, 316, E956–E966. [Google Scholar] [CrossRef]
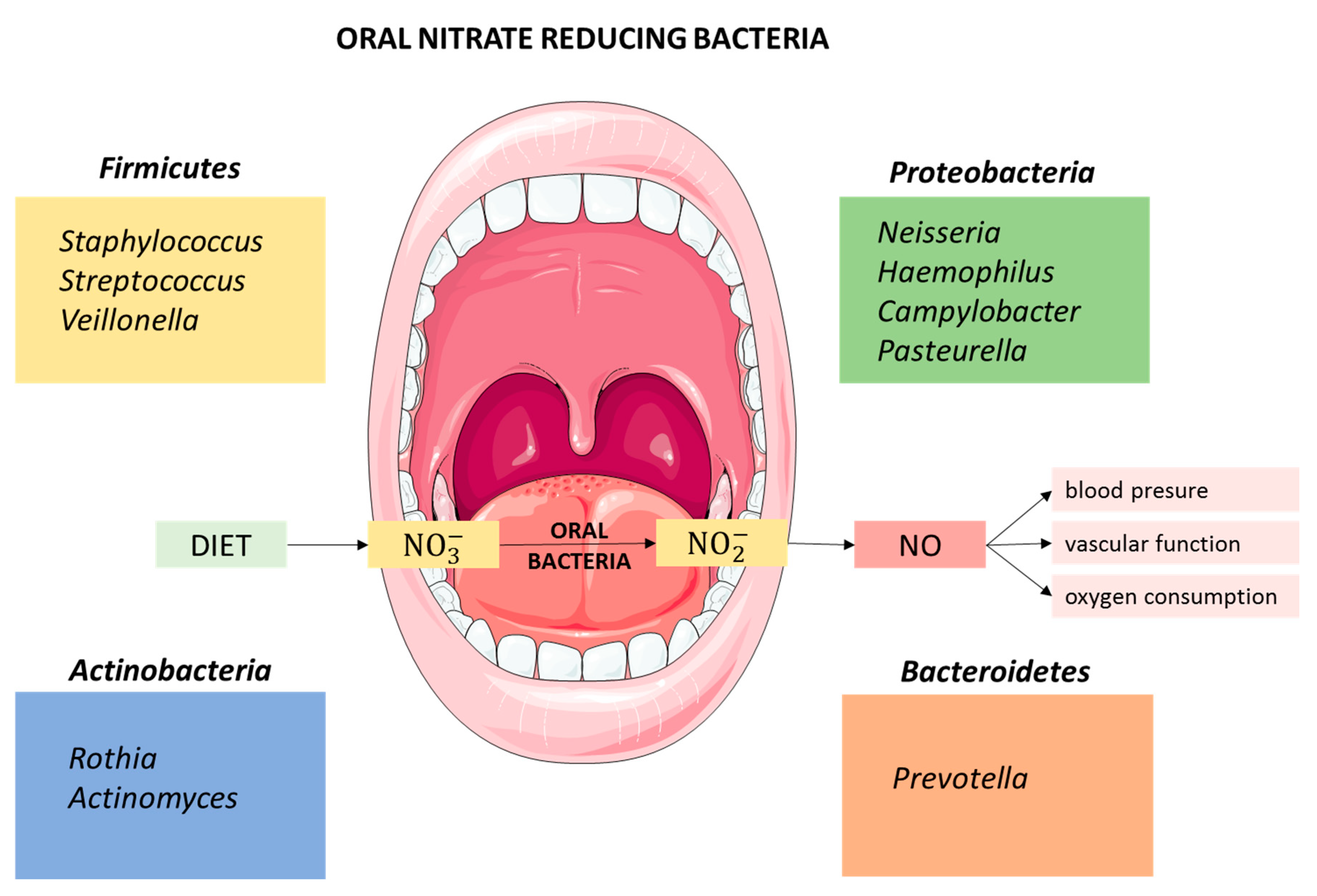
| Model | Participants | Age | Nitrate Supple- Mentation | Oral Nitrate-Reducing Bacteria | Reference |
|---|---|---|---|---|---|
| Human | hypercholesterolemic n = 69 | 18–80 | Yes (beetroot juice) | Neisseria, Rothia | [18] |
| Human | Females (n = 4) Males (n = 6) | 29 | No | Veillonella, Actinomyces, Rothia and Staphylococcus | [39] |
| Rat | Sprague- Dawley males (n = 5) | adults | No | Staphylococcus, Staphylococcus, Pasteurella spp. and Streptococcus spp. | [42] |
| Human | Females | 25–30 | No | Streptococcus | [43] |
| Human | n = 6 | >18 | No | Veillonella, Prevotella, Neisseria, and Haemophilus, Actinomyces | [44] |
| Human | Females (n = 12) Males (n = 12) | 70.3 ± 6.0 | No | Veillonella | [45] |
| Human | n = 25 | 27 ± 7 | No | Prevotella, Veillonella, Haemophilus, Neisseria, Rothia | [46] |
| Human | n = 240 | 2–12 months | No | Prevotella, Veillonella, Alloprevotella and Leptotrichia | [47] |
| Rat | Wistar rats (n = 8) | 7 weeks | Yes (NaNO3) | Streptococcus and Haemophilus | [48] |
| Human | Females (n = 6) Males (n = 3) | 70–79 | Yes (beetroot juice) | Rothia, Neisseria | [49] |
| Females (n = 5) Males (n = 4) | 18–22 |
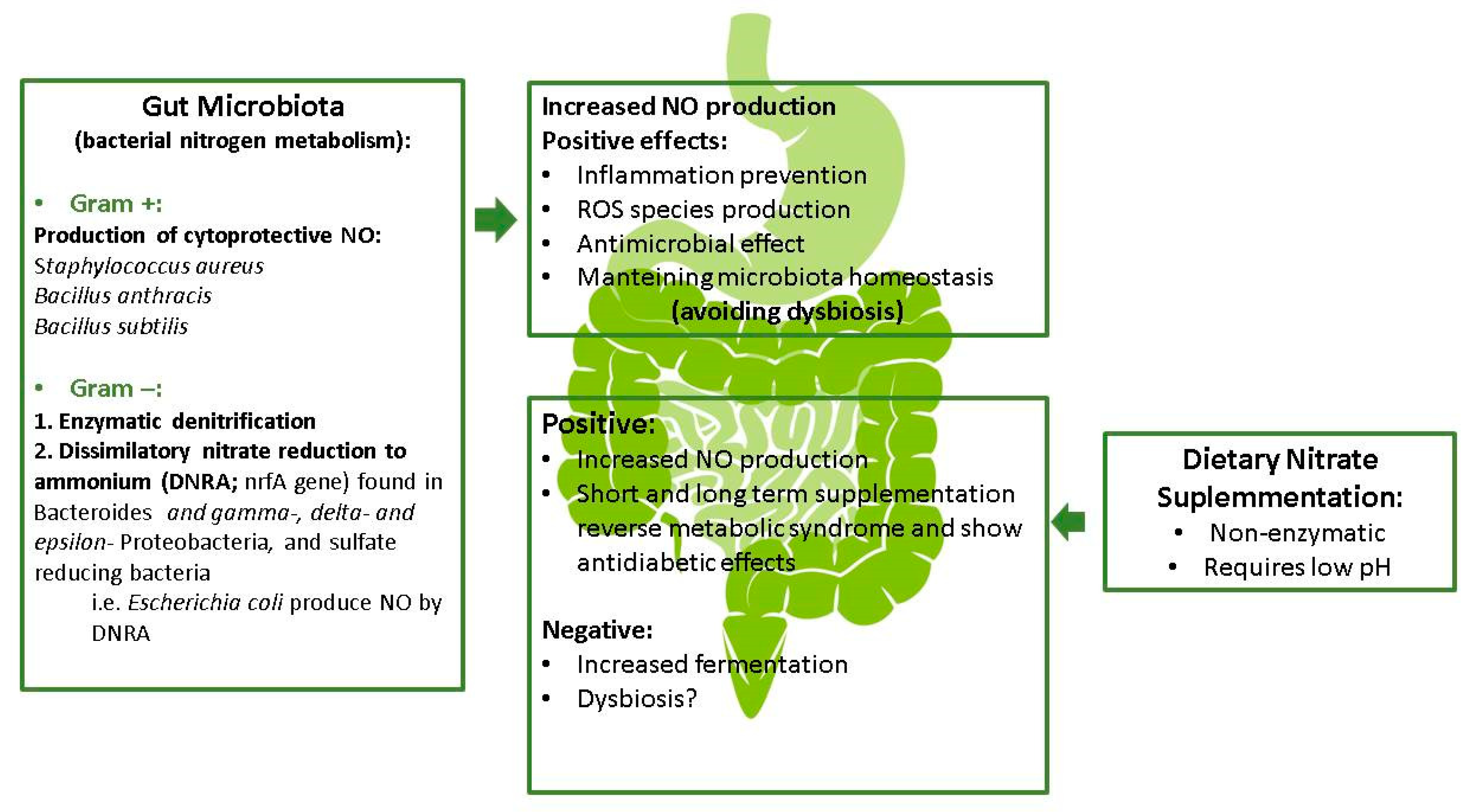
| Objective | Type of Study and Model | Observed Changes in the Metabolism of the Gut Microbiota | Reference |
|---|---|---|---|
| To test whether the formation of N-nitroso compounds is afforded by the gut microbiota | Pig cecum model | Following incubation with nitrate, the formation of NMOR, NPYR, NMU, and NEU * was detectable, with the microbiota being responsible for the reduction of nitrate to nitrite. After incubation of nitrite, a chemical formation of N-nitroso compounds was observed | [1] |
| To investigate whether dietary nitrate rescues gastrointestinal physiology during dysbiosis | Wistar rats were maintained in the animal facilities for 7 days during which, in addition to food, they had access to (1) water, (2) an antibiotic cocktail (neomycin, bacitracin, imipenem), (3) antibiotic cocktail plus 10 mM sodium nitrate, (4) sodium nitrate. | Nitrate prevented body weight loss under dysbiosis | [30] |
| To investigate whether dietary compounds can stimulate NO production | Representative cultures of human gut microbiota | Dietary compounds and the microbial community composition determine the conditions in the colon and hence the chemical production of NO. NO is formed by the reduction of nitrite with hydrogen sulfide, both products of microbial metabolism in the intestinal tract. Only small fractions of NO are released from nitrite, yet these amounts may impact the metabolism of colonocytes | [56] |
| To investigate whether bacterial nitrate reduction to ammonia, as well as the related NO formation in the gut, could be an important aspect of the overall mammalian nitrate/nitrite/NO metabolism and is yet another way in which the microbiota links diet and health | In vivo bacterial growth cultures | When supplied with exogenous nitrite, L. rhamnosus, L. acidophilus and B. longum infantis produce NO independent of added nitrate. Bacterial production of lactic acid causes medium acidification that, in turn, generates NO by non-enzymatic nitrite reduction | [58] |
| To investigate if changes in the intestinal microbiota induced by a nitrate-rich juice diet play an important role in its health benefits. | Twenty healthy adults consumed only vegetable/fruit juices (rich in nitrate) for 3 days followed by 14 days of customary diet | Between day 4 and 17 there were significant decreases in weight and body mass index Between day 4 and 17, the proportion of Firmicutes and Proteobacteria in stool was significantly decreased whereas Bacteroidetes and Cyanobacteria was increased An increase in NO in plasma was observed on day 4 A 3-day juice (rich in nitrate)-based diet changed the intestinal microbiota associated with weight loss, increased the vasodilator NO, and decreased lipid oxidation | [59] |
| To test if long-term dietary nitrite/nitrate deficiency induces the metabolic syndrome in mice | Experiments were performed in 6-week-old male C57BL/6J mice | Long-term dietary nitrite/nitrate deficiency gave rise to the metabolic syndrome, endothelial dysfunction and cardiovascular death in mice, indicating a novel pathogenic role of the exogenous NO production system in the metabolic syndrome and its vascular complications | [60] |
| To investigate the ability of dietary nitrate to improve NO bioavailability and reduce bone turnover and loss in ovariectomized (OVX) rats. | Six-month-old Sprague Dawley rats [30 OVX and 10 sham-operated (sham)] were randomized into three groups: (i) vehicle (water) control, (ii) low-dose nitrate (0.1 mmol nitrate/kg bw/day), or (iii) high-dose nitrate (1.0 mmol nitrate/kg bw/day) for three weeks | OVX (but not dietary nitrate) affects the fecal microbiome and the gut microbiome is associated with bone mass | [61] |
| To test dietary nitrate as a key component of functional foods with beneficial impact on gastric mucosal integrity during antibiotic therapy | Male Wistar rats followed 4 different treatments: (1) antibiotic cocktail (neomycin, bacitracin and imipenem), (2) antibiotic cocktail + sodium nitrate, (3) sodium nitrate, and (4) regular drinking water. | Dietary nitrate may be envisaged as a key component of functional foods with beneficial impact on gastric mucosal integrity during antibiotic therapy, but further studies are needed to better ascertain as to whether it modulates intestinal microbiota in terms of taxonomic and functional levels | [62] |
| To evaluate the effects of using encapsulated nitrate product (ENP) on ruminal fermentation | Ruminal fermentation and methane production in vitro using a semi-automatic gas production technique | ENP increases methane production | [63] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-Soltero, R.; Bailén, M.; de Lucas, B.; Ramírez-Goercke, M.I.; Pareja-Galeano, H.; Larrosa, M. Role of Oral and Gut Microbiota in Dietary Nitrate Metabolism and Its Impact on Sports Performance. Nutrients 2020, 12, 3611. https://doi.org/10.3390/nu12123611
González-Soltero R, Bailén M, de Lucas B, Ramírez-Goercke MI, Pareja-Galeano H, Larrosa M. Role of Oral and Gut Microbiota in Dietary Nitrate Metabolism and Its Impact on Sports Performance. Nutrients. 2020; 12(12):3611. https://doi.org/10.3390/nu12123611
Chicago/Turabian StyleGonzález-Soltero, Rocío, María Bailén, Beatriz de Lucas, Maria Isabel Ramírez-Goercke, Helios Pareja-Galeano, and Mar Larrosa. 2020. "Role of Oral and Gut Microbiota in Dietary Nitrate Metabolism and Its Impact on Sports Performance" Nutrients 12, no. 12: 3611. https://doi.org/10.3390/nu12123611
APA StyleGonzález-Soltero, R., Bailén, M., de Lucas, B., Ramírez-Goercke, M. I., Pareja-Galeano, H., & Larrosa, M. (2020). Role of Oral and Gut Microbiota in Dietary Nitrate Metabolism and Its Impact on Sports Performance. Nutrients, 12(12), 3611. https://doi.org/10.3390/nu12123611








