Influence of Fermented Red Clover Extract on Skeletal Muscle in Early Postmenopausal Women: A Double-Blinded Cross-Over Study
Abstract
1. Introduction
2. Methods
2.1. Ethical Approval
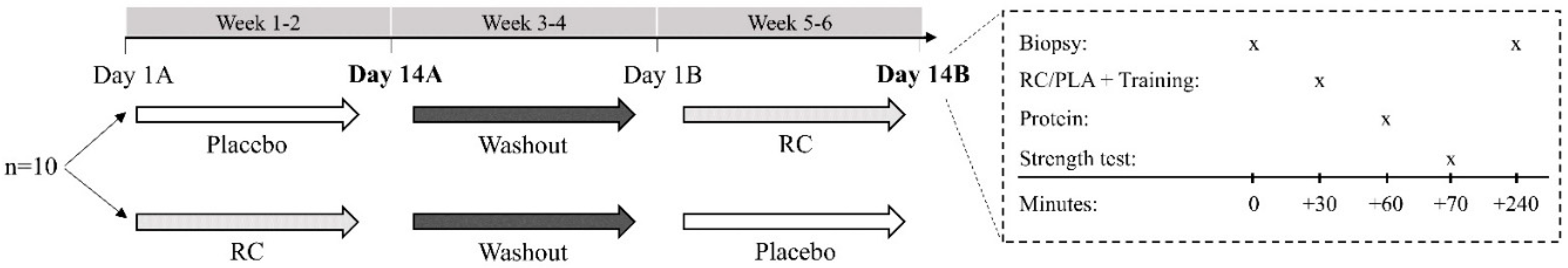
2.2. Design
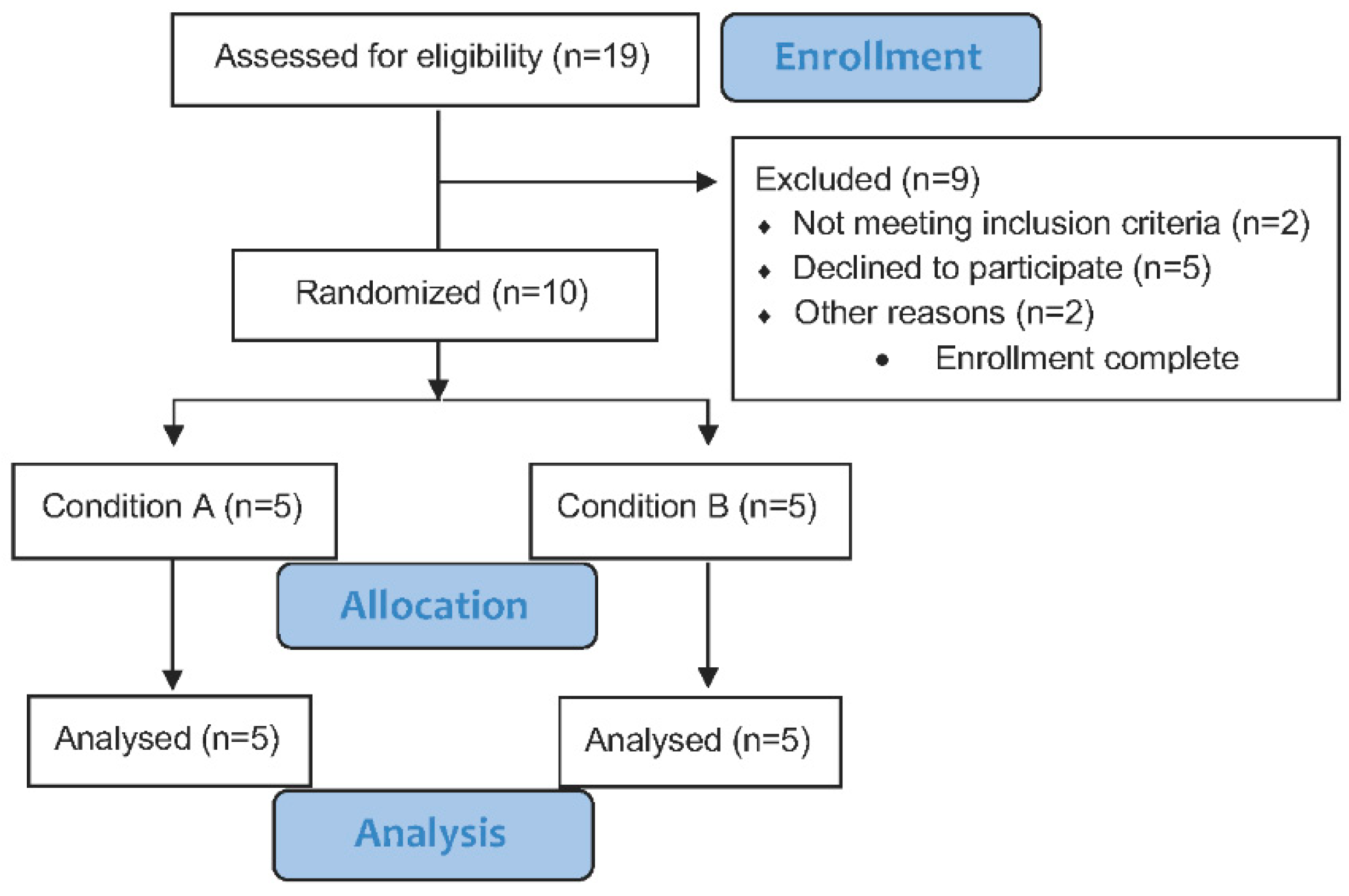
2.3. Participants
2.4. Experimental Drinks
2.5. Muscle Biopsy
2.6. Resistance Exercise
2.7. Maximal Isometric Grip Strength
2.8. Maximal Isometric Elbow Flexor Strength
2.9. Western Blotting
2.10. Statistical Analyses
3. Results
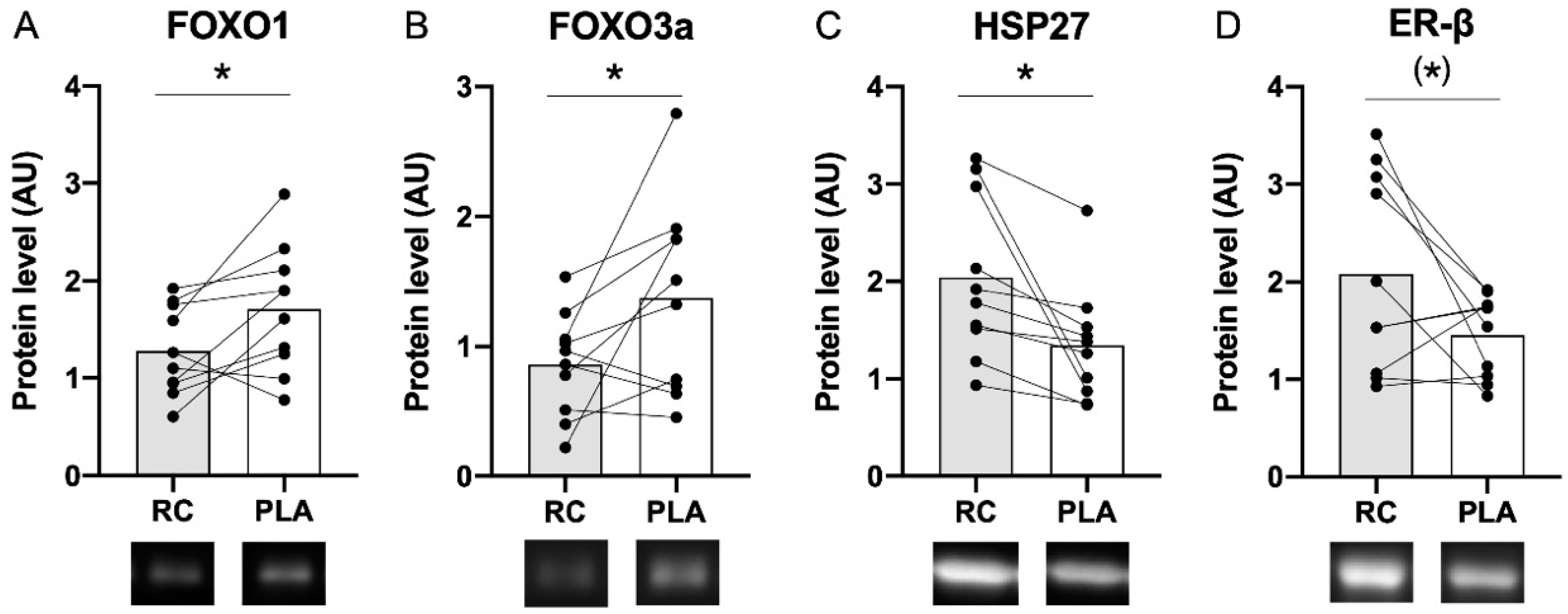
3.1. Muscle Protein Content Following the RC/PLA Intervention Periods
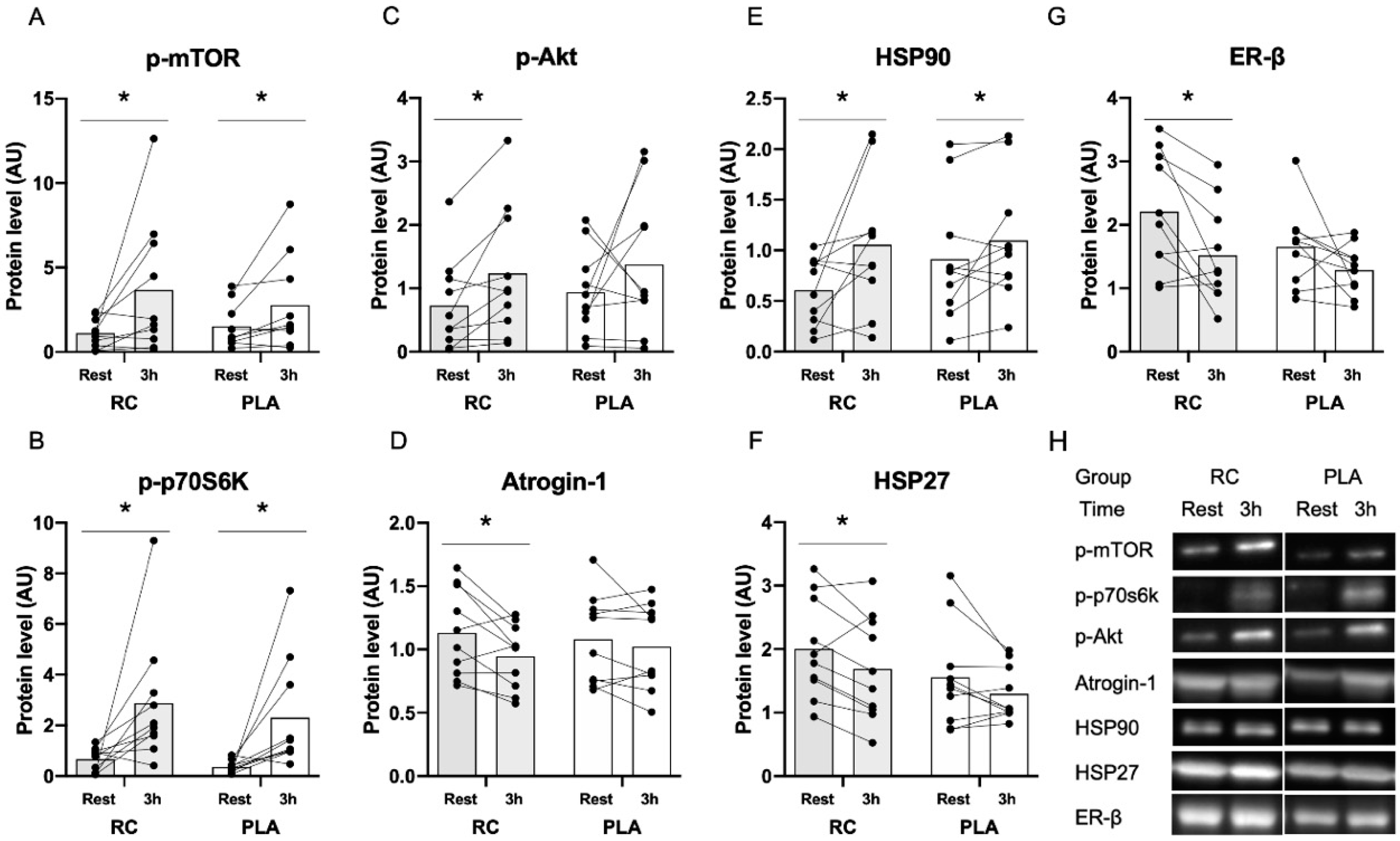
3.2. Muscle Protein Expression in Response to Exercise Combined with Protein Supplementation
3.3. Changes in Muscle Strength and RFD Following the RC/PLA Intervention Periods
4. Discussion
4.1. Influence of RC Extract on Markers of Protein Degradation
4.2. Influence of RC Extract on Heat Shock Proteins
4.3. Changes in Protein Expression in Response to Resistance Exercise Combined with Protein Supplementation
4.4. Changes in Muscle Strength and RFD
4.5. Future Directions
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hirschfeld, H.P.; Kinsella, R.; Duque, G. Osteosarcopenia: Where bone, muscle, and fat collide. Osteoporos. Int. 2017, 28, 2781–2790. [Google Scholar] [CrossRef] [PubMed]
- Tsekoura, M.; Kastrinis, A.; Katsoulaki, M.; Billis, E.; Gliatis, J. Sarcopenia and Its Impact on Quality of Life. Adv. Exp. Med. Biol. 2017, 987, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Maltais, M.L.; Desroches, J.; Dionne, I.J. Changes in muscle mass and strength after menopause. J. Musculoskelet. Neuron. Interact. 2009, 9, 186–197. [Google Scholar]
- McLeod, M.; Breen, L.; Hamilton, D.L.; Philp, A. Live strong and prosper: The importance of skeletal muscle strength for healthy ageing. Biogerontology 2016, 17, 497–510. [Google Scholar] [CrossRef] [PubMed]
- Roth, S.M.; Ferrell, R.F.; Hurley, B.F. Strength training for the prevention and treatment of sarcopenia. J. Nutr. Health Aging 2000, 4, 143–155. [Google Scholar] [PubMed]
- Smith, G.I.; Villareal, D.T.; Sinacore, D.R.; Shah, K.; Mittendorfer, B. Muscle protein synthesis response to exercise training in obese, older men and women. Med. Sci. Sports Exerc. 2012, 44, 1259–1266. [Google Scholar] [CrossRef]
- Hansen, M.; Skovgaard, D.; Reitelseder, S.; Holm, L.; Langbjerg, H.; Kjaer, M. Effects of estrogen replacement and lower androgen status on skeletal muscle collagen and myofibrillar protein synthesis in postmenopausal women. J. Gerontol. A Biol. Sci. Med. Sci. 2012, 67, 1005–1013. [Google Scholar] [CrossRef]
- Bamman, M.M.; Hill, V.J.; Adams, G.R.; Haddad, F.; Wetzstein, C.J.; Gower, B.A.; Ahmed, A.; Hunter, G.R. Gender differences in resistance-training-induced myofiber hypertrophy among older adults. J. Gerontol. A Biol. Sci. Med. Sci. 2003, 58, 108–116. [Google Scholar] [CrossRef]
- Greising, S.M.; Baltgalvis, K.A.; Lowe, D.A.; Warren, G.L. Hormone therapy and skeletal muscle strength: A meta-analysis. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2009, 64, 1071–1081. [Google Scholar] [CrossRef]
- Dam, T.V.; Dalgaard, L.B.; Ringgaard, S.; Johansen, F.T.; Bengtsen, M.B.; Mose, M.; Lauritsen, K.M.; Ørtenblad, N.; Gravholt, C.H.; Hansen, M. Transdermal estrogen therapy improves gains in skeletal muscle mass after 12-1 weeks of resistance training in early postmenopausal women. Front. Physiol. 2020. Under review. [Google Scholar]
- Collaborative Group on Epidemiological Studies of Ovarian Cancer; Beral, V.; Gaitskell, K.; Hermon, C.; Moser, K.; Reeves, G.; Peto, R. Menopausal hormone use and ovarian cancer risk: Individual participant meta-analysis of 52 epidemiological studies. Lancet 2015, 385, 1835–1842. [Google Scholar] [CrossRef]
- Rossouw, J.E.; Anderson, G.L.; Prentice, R.L.; LaCroix, A.Z.; Kooperberg, C.; Stefanick, M.L.; Jackson, R.D.; Beresford, S.A.; Howard, B.V.; Johnson, K.C.; et al. Risks and benefits of estrogen plus progestin in healthy postmenopausal women: Principal results From the Women’s Health Initiative randomized controlled trial. JAMA 2002, 288, 321–333. [Google Scholar] [PubMed]
- Type and timing of menopausal hormone therapy and breast cancer risk: Individual participant meta-analysis of the worldwide epidemiological evidence. Lancet 2019, 394, 1159–1168. [CrossRef]
- Shi, L.F.; Wu, Y.; Li, C.Y. Hormone therapy and risk of ovarian cancer in postmenopausal women: A systematic review and meta-analysis. Menopause 2016, 23, 417–424. [Google Scholar] [CrossRef]
- Gencel, V.B.; Benjamin, M.M.; Bahou, S.N.; Khalil, R.A. Vascular effects of phytoestrogens and alternative menopausal hormone therapy in cardiovascular disease. Mini Rev. Med. Chem. 2012, 12, 149–174. [Google Scholar] [CrossRef]
- Jung, W.; Yu, O.; Lau, S.M.; O’Keefe, D.P.; Odell, J.; Fader, G.; McGonigle, B. Identification and expression of isoflavone synthase, the key enzyme for biosynthesis of isoflavones in legumes. Nat. Biotechnol. 2000, 18, 208–212. [Google Scholar] [CrossRef]
- Pfitscher, A.; Reiter, E.; Jungbauer, A. Receptor binding and transactivation activities of red clover isoflavones and their metabolites. J. Steroid Biochem. Mol. Biol. 2008, 112, 87–94. [Google Scholar] [CrossRef]
- Nagata, C.; Mizoue, T.; Tanaka, K.; Tsuji, I.; Tamakoshi, A.; Matsuo, K.; Wakai, K.; Inoue, M.; Tsugane, S.; Sasazuki, S. Soy intake and breast cancer risk: An evaluation based on a systematic review of epidemiologic evidence among the Japanese population. Jpn. J. Clin. Oncol. 2014, 44, 282–295. [Google Scholar] [CrossRef]
- Zhao, T.T.; Jin, F.; Li, J.G.; Xu, Y.Y.; Dong, H.T.; Liu, Q.; Xing, P.; Zhu, G.L.; Xu, H.; Miao, Z.F. Dietary isoflavones or isoflavone-rich food intake and breast cancer risk: A meta-analysis of prospective cohort studies. Clin. Nutr. 2017. [Google Scholar] [CrossRef]
- Morimoto, Y.; Maskarinec, G.; Park, S.Y.; Ettienne, R.; Matsuno, R.K.; Long, C.; Steffen, A.D.; Henderson, B.E.; Kolonel, L.N.; Le Marchand, L.; et al. Dietary isoflavone intake is not statistically significantly associated with breast cancer risk in the Multiethnic Cohort. Br. J. Nutr. 2014, 112, 976–983. [Google Scholar] [CrossRef]
- Wiik, A.; Ekman, M.; Johansson, O.; Jansson, E.; Esbjornsson, M. Expression of both oestrogen receptor alpha and beta in human skeletal muscle tissue. Histochem. Cell Biol. 2009, 131, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Velders, M.; Schleipen, B.; Fritzemeier, K.H.; Zierau, O.; Diel, P. Selective estrogen receptor-beta activation stimulates skeletal muscle growth and regeneration. FASEB J. 2012, 26, 1909–1920. [Google Scholar] [CrossRef] [PubMed]
- Tabata, S.; Aizawa, M.; Kinoshita, M.; Ito, Y.; Kawamura, Y.; Takebe, M.; Pan, W.; Sakuma, K. The influence of isoflavone for denervation-induced muscle atrophy. Eur. J. Nutr. 2019, 58, 291–300. [Google Scholar] [CrossRef] [PubMed]
- Aoyama, S.; Jia, H.; Nakazawa, K.; Yamamura, J.; Saito, K.; Kato, H. Dietary Genistein Prevents Denervation-Induced Muscle Atrophy in Male Rodents via Effects on Estrogen Receptor-alpha. J. Nutr. 2016, 146, 1147–1154. [Google Scholar] [CrossRef] [PubMed]
- Ogawa, M.; Kitano, T.; Kawata, N.; Sugihira, T.; Kitakaze, T.; Harada, N.; Yamaji, R. Daidzein down-regulates ubiquitin-specific protease 19 expression through estrogen receptor beta and increases skeletal muscle mass in young female mice. J. Nutr. Biochem. 2017, 49, 63–70. [Google Scholar] [CrossRef]
- Zheng, W.; Hengevoss, J.; Soukup, S.T.; Kulling, S.E.; Xie, M.; Diel, P. An isoflavone enriched diet increases skeletal muscle adaptation in response to physical activity in ovariectomized rats. Mol. Nutr. Food Res. 2017, 61, 1600843. [Google Scholar] [CrossRef]
- Aubertin-Leheudre, M.; Lord, C.; Khalil, A.; Dionne, I.J. Six months of isoflavone supplement increases fat-free mass in obese-sarcopenic postmenopausal women: A randomized double-blind controlled trial. Eur. J. Clin. Nutr. 2007, 61, 1442–1444. [Google Scholar] [CrossRef]
- Lebon, J.; Riesco, E.; Tessier, D.; Dionne, I.J. Additive effects of isoflavones and exercise training on inflammatory cytokines and body composition in overweight and obese postmenopausal women: A randomized controlled trial. Menopause 2014, 21, 869–875. [Google Scholar] [CrossRef]
- Choquette, S.; Riesco, E.; Cormier, E.; Dion, T.; Aubertin-Leheudre, M.; Dionne, I.J. Effects of soya isoflavones and exercise on body composition and clinical risk factors of cardiovascular diseases in overweight postmenopausal women: A 6-month double-blind controlled trial. Br. J. Nutr. 2011, 105, 1199–1209. [Google Scholar] [CrossRef]
- Choquette, S.; Dion, T.; Brochu, M.; Dionne, I.J. Soy isoflavones and exercise to improve physical capacity in postmenopausal women. Climacteric 2013, 16, 70–77. [Google Scholar] [CrossRef]
- Orsatti, F.L.; Nahas, E.A.; Nahas-Neto, J.; Maesta, N.; Orsatti, C.L.; Fernandes, C.E. Effects of resistance training and soy isoflavone on body composition in postmenopausal women. Obstet. Gynecol. Int. 2010, 2010, 156037. [Google Scholar] [CrossRef]
- Beck, V.; Unterrieder, E.; Krenn, L.; Kubelka, W.; Jungbauer, A. Comparison of hormonal activity (estrogen, androgen and progestin) of standardized plant extracts for large scale use in hormone replacement therapy. J. Steroid Biochem. Mol. Biol. 2003, 84, 259–268. [Google Scholar] [CrossRef]
- Izumi, T.; Piskula, M.K.; Osawa, S.; Obata, A.; Tobe, K.; Saito, M.; Kataoka, S.; Kubota, Y.; Kikuchi, M. Soy isoflavone aglycones are absorbed faster and in higher amounts than their glucosides in humans. J. Nutr. 2000, 130, 1695–1699. [Google Scholar] [CrossRef] [PubMed]
- Lambert, M.N.T.; Thybo, C.B.; Lykkeboe, S.; Rasmussen, L.M.; Frette, X.; Christensen, L.P.; Jeppesen, P.B. Combined bioavailable isoflavones and probiotics improve bone status and estrogen metabolism in postmenopausal osteopenic women: A randomized controlled trial. Am. J. Clin. Nutr. 2017, 106, 909–920. [Google Scholar] [CrossRef] [PubMed]
- Thorup, A.C.; Lambert, M.N.; Kahr, H.S.; Bjerre, M.; Jeppesen, P.B. Intake of Novel Red Clover Supplementation for 12 Weeks Improves Bone Status in Healthy Menopausal Women. Evid. Based Complement. Altern. Med. 2015, 2015, 689138. [Google Scholar] [CrossRef]
- Park, Y.M.; Pereira, R.I.; Erickson, C.B.; Swibas, T.A.; Kang, C.; Van Pelt, R.E. Time since menopause and skeletal muscle estrogen receptors, PGC-1alpha, and AMPK. Menopause 2017, 24, 815–823. [Google Scholar] [CrossRef]
- Brzycki, M. Strength testing: Predicting a one-rep max from reps-to-fatigue. J. Health 1993, 64, 88–90. [Google Scholar]
- Oxfeldt, M.; Dalgaard, L.B.; Jørgensen, E.B.; Dalgaard, E.B.; Johansen, F.T.; Ørtenblad, N.; Hansen, M. Molecular markers of skeletal muscle hypertrophy following 10 weeks of resistance training in oral contraceptive users and non-users. J. Appl. Physiol. 2020. [Google Scholar] [CrossRef]
- Boland, R.; Vasconsuelo, A.; Milanesi, L.; Ronda, A.C.; de Boland, A.R. 17beta-estradiol signaling in skeletal muscle cells and its relationship to apoptosis. Steroids 2008, 73, 859–863. [Google Scholar] [CrossRef]
- Velders, M.; Diel, P. How sex hormones promote skeletal muscle regeneration. Sports Med. 2013, 43, 1089–1100. [Google Scholar] [CrossRef]
- Serra, M.C.; Beavers, K.M.; Beavers, D.P.; Willoughby, D.S. Effects of 28 days of dairy or soy ingestion on skeletal markers of inflammation and proteolysis in post-menopausal women. Nutr. Health 2012, 21, 117–130. [Google Scholar] [CrossRef] [PubMed]
- Beavers, K.M.; Serra, M.C.; Beavers, D.P.; Cooke, M.B.; Willoughby, D.S. Soy and the exercise-induced inflammatory response in postmenopausal women. Appl. Physiol. Nutr. Metab. Physiol. Appl. Nutr. Metab. 2010, 35, 261–269. [Google Scholar] [CrossRef]
- Tang, J.E.; Moore, D.R.; Kujbida, G.W.; Tarnopolsky, M.A.; Phillips, S.M. Ingestion of whey hydrolysate, casein, or soy protein isolate: Effects on mixed muscle protein synthesis at rest and following resistance exercise in young men. J. Appl. Physiol. 2009, 107, 987–992. [Google Scholar] [CrossRef] [PubMed]
- Seko, D.; Fujita, R.; Kitajima, Y.; Nakamura, K.; Imai, Y.; Ono, Y. Estrogen Receptor beta Controls Muscle Growth and Regeneration in Young Female Mice. Stem Cell Rep. 2020, 15, 577–586. [Google Scholar] [CrossRef]
- Noble, E.G.; Milne, K.J.; Melling, C.W. Heat shock proteins and exercise: A primer. Appl. Physiol. Nutr. Metab. Physiol. Appl. Nutr. Metab. 2008, 33, 1050–1065. [Google Scholar] [CrossRef]
- Lee, C.E.; McArdle, A.; Griffiths, R.D. The role of hormones, cytokines and heat shock proteins during age-related muscle loss. Clin. Nutr. 2007, 26, 524–534. [Google Scholar] [CrossRef]
- Paroo, Z.; Dipchand, E.S.; Noble, E.G. Estrogen attenuates postexercise HSP70 expression in skeletal muscle. Am. J. Physiol. Cell Physiol. 2002, 282, C245–C251. [Google Scholar] [CrossRef]
- Vasconsuelo, A.; Milanesi, L.; Boland, R. Participation of HSP27 in the antiapoptotic action of 17beta-estradiol in skeletal muscle cells. Cell Stress Chaperones 2010, 15, 183–192. [Google Scholar] [CrossRef]
- Bombardier, E.; Vigna, C.; Iqbal, S.; Tiidus, P.M.; Tupling, A.R. Effects of ovarian sex hormones and downhill running on fiber-type-specific HSP70 expression in rat soleus. J. Appl. Physiol. 2009, 106, 2009–2015. [Google Scholar] [CrossRef]
- Bombardier, E.; Vigna, C.; Bloemberg, D.; Quadrilatero, J.; Tiidus, P.M.; Tupling, A.R. The role of estrogen receptor-α in estrogen-mediated regulation of basal and exercise-induced Hsp70 and Hsp27 expression in rat soleus. Can. J. Physiol. Pharmacol. 2013, 91, 823–829. [Google Scholar] [CrossRef]
- Li, B.Y.; Zhao, Y.; Li, X.Q.; Yi, X.Z.; Wang, Y.M. Effect of estrogen on heat shock protein 70 expression in rat masseter muscle. Hua Xi Kou Qiang Yi Xue Za Zhi 2008, 26, 560–562. [Google Scholar]
- Joanisse, S.; Lim, C.; McKendry, J.; McLeod, J.C.; Stokes, T.; Phillips, S.M. Recent advances in understanding resistance exercise training-induced skeletal muscle hypertrophy in humans. F1000Research 2020, 9. [Google Scholar] [CrossRef]
- Hodson, N.; West, D.W.D.; Philp, A.; Burd, N.A.; Moore, D.R. Molecular regulation of human skeletal muscle protein synthesis in response to exercise and nutrients: A compass for overcoming age-related anabolic resistance. Am. J. Physiol. Cell Physiol. 2019, 317, C1061–C1078. [Google Scholar] [CrossRef] [PubMed]
- Spiering, B.A.; Kraemer, W.J.; Anderson, J.M.; Armstrong, L.E.; Nindl, B.C.; Volek, J.S.; Maresh, C.M. Resistance exercise biology: Manipulation of resistance exercise programme variables determines the responses of cellular and molecular signalling pathways. Sports Med. 2008, 38, 527–540. [Google Scholar] [CrossRef] [PubMed]
- Foletta, V.C.; White, L.J.; Larsen, A.E.; Leger, B.; Russell, A.P. The role and regulation of MAFbx/atrogin-1 and MuRF1 in skeletal muscle atrophy. Pflug. Arch. 2011, 461, 325–335. [Google Scholar] [CrossRef]
- Lowe, D.A.; Baltgalvis, K.A.; Greising, S.M. Mechanisms behind estrogen’s beneficial effect on muscle strength in females. Exerc. Sport Sci. Rev. 2010, 38, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Moran, A.L.; Warren, G.L.; Lowe, D.A. Removal of ovarian hormones from mature mice detrimentally affects muscle contractile function and myosin structural distribution. J. Appl. Physiol. 2006, 100, 548–559. [Google Scholar] [CrossRef] [PubMed]
- Moran, A.L.; Nelson, S.A.; Landisch, R.M.; Warren, G.L.; Lowe, D.A. Estradiol replacement reverses ovariectomy-induced muscle contractile and myosin dysfunction in mature female mice. J. Appl. Physiol. 2007, 102, 1387–1393. [Google Scholar] [CrossRef]
- Lambert, M.N.T.; Hu, L.M.; Jeppesen, P.B. A systematic review and meta-analysis of the effects of isoflavone formulations against estrogen-deficient bone resorption in peri- and postmenopausal women. Am. J. Clin. Nutr. 2017, 106, 801–811. [Google Scholar] [CrossRef]
- Chen, M.N.; Lin, C.C.; Liu, C.F. Efficacy of phytoestrogens for menopausal symptoms: A meta-analysis and systematic review. Climacteric 2015, 18, 260–269. [Google Scholar] [CrossRef]
- Panel, E.A. Risk assessment for peri- and post-menopausal women taking food supplements containing isolated isoflavones. EFSA J. 2015, 13, 3496. [Google Scholar] [CrossRef]
- Yamori, Y. Food factors for atherosclerosis prevention: Asian perspective derived from analyses of worldwide dietary biomarkers. Exp. Clin. Cardiol. 2006, 11, 94–98. [Google Scholar] [PubMed]
- Moro, T.; Brightwell, C.R.; Phalen, D.E.; McKenna, C.F.; Lane, S.J.; Porter, C.; Volpi, E.; Rasmussen, B.B.; Fry, C.S. Low skeletal muscle capillarization limits muscle adaptation to resistance exercise training in older adults. Exp. Gerontol. 2019, 127, 110723. [Google Scholar] [CrossRef] [PubMed]
- Snijders, T.; Nederveen, J.P.; Joanisse, S.; Leenders, M.; Verdijk, L.B.; van Loon, L.J.; Parise, G. Muscle fibre capillarization is a critical factor in muscle fibre hypertrophy during resistance exercise training in older men. J. Cachexia Sarcopenia Muscle 2017, 8, 267–276. [Google Scholar] [CrossRef] [PubMed]
| Subjects (n = 10) | |
|---|---|
| Age (y) | 54 ± 4 |
| Height (cm) | 168 ± 6 |
| Weight (kg) | 70 ± 8 |
| FM (kg) | 26 ± 7 |
| FFM (kg) | 42 ± 4 |
| Physical activity (min/week) | 113 ± 55 |
| Steps (Steps/day) | 8550 ± 1892 |
| Time since last bleeding (months) | 18 ± 11 |
| Antibody | Physiological Process | Manufacturer | Catalog No. | Dilution | Blocking Agent | Secondary Antibodies | Dilution |
|---|---|---|---|---|---|---|---|
| P-Akt Ser473 | Promotes cell survival by inhibiting apoptosis when phosphorylated | Cell Sig. | 9271 | 1:1000 | 5% BSA, TBST | Anti-rabbit IgG, 7074S | 1:2000 |
| P-mTOR Ser2448 | Promotes cell growth when phosphorylated | Cell Sig. | 2971 | 1:1000 | 5% BSA, TBST | Anti-rabbit IgG, 7074S | 1:5000 |
| mTOR | Regulates cell growth | Cell Sig. | 2972 | 1:1000 | 5% BSA, TBST | Anti-rabbit igG, 7074S | 1:5000 |
| P-p70S6K | Promotes cell growth, when phosphorylated | Cell Sig. | 9234 | 1:500 | 5% BSA, TBST | Anti-rabbit IgG, 7074S | 1:5000 |
| P-P38 MAPK Thr180/182 | A transducer of stress stimuli | Cell Sig. | 9211 | 1:1000 | 5% BSA, TBST | Anti-rabbit IgG, 7074S | 1:5000 |
| P-FOXO3 Ser253 | Promotes cell cycle arrest and apoptosis when dephosphorylated | Cell Sig. | 9466 | 1:1000 | 5% BSA, TBST | Anti-rabbit IgG, 7074S | 1:2500 |
| FOXO3a | Promotes cell cycle arrest and apoptosis | Cell Sig. | 2497 | 1:1000 | 5% BSA, TBST | Anti-rabbit IgG, 7074S | 1:2000 |
| FOXO1 | Promotes cell cycle arrest and apoptosis | Cell Sig. | 2880 | 1:1000 | 5% Milk, TBST | Anti-rabbit IgG, 7074S | 1:2000 |
| MURF1 | Promotes muscle cell protein degradation | ECM Biosciences. | 3401 | 1:1000 | 0.3% i-block, PBST | Anti-rabbit IgG, 7074S | 1:2000 |
| Atrogin-1 | Promotes muscle cell protein degradation | Abcam | ab168372 | 1:1000 | 5% Milk, TBST | Anti-rabbit IgG, 7074S | 1:2500 |
| HSP27 | Promotes cellular resistance | Abcam | ab109376 | 1:1500 | 5% Milk, TBST | Anti-rabbit IgG, 7074S | 1:10,000 |
| HSP70 | Promotes cellular homeostasis | Abcam | ab181606 | 1:1000 | 5% Milk, TBST | Anti-rabbit IgG, 7074S | 1:5000 |
| HSP90 | Promotes cellular homeostasis | Abcam | ab203126 | 1:1000 | 5% Milk, TBST | Anti-rabbit IgG, 7074S | 1:5000 |
| Myogenin | Promotes early myogenic differentiation | Milipore | 3876 | 1:1000 | 5% Milk, TBST | Goat anti-mouse IgG | 1:5000 |
| MyoD | Promotes late myogenic differentiation | Abcam | ab126726 | 1:1000 | 5% BSA, TBST | Anti-rabbit IgG, 7074S | 1:5000 |
| ER-β | A receptor for estrogenic compounds | Abcam | ab3576 | 1:500 | 5% Milk, TBST | Anti-rabbit IgG, 7074S | 1:5000 |
| RC | PLA | ||||
|---|---|---|---|---|---|
| Pre | Post | Pre | Post | Interaction | |
| Grip strength (kg) | 32.3 ± 7.6 | 32.2 ± 7.4 | 30.8 ± 6.3 | 32.4 ± 7.7 | 0.14 |
| Elbow flexor strength (Nm) | 39.0 ± 5.6 | 38.3 ± 7.5 | 37.4 ± 4.3 | 36.9 ± 4.6 | 0.93 |
| RFD 0–50 ms (Nm) | 5.6 ± 3.4 | 5.9 ± 4.4 | 7.0 ± 3.2 | 6.9 ± 3.7 | 0.80 |
| RFD 0–100 ms (Nm) | 11.1 ± 6.4 | 11.1 ± 6.0 | 12.7 ± 5.2 | 13.1 ± 5.1 | 0.83 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oxfeldt, M.; Dalgaard, L.B.; Risikesan, J.; Johansen, F.T.; Hansen, M. Influence of Fermented Red Clover Extract on Skeletal Muscle in Early Postmenopausal Women: A Double-Blinded Cross-Over Study. Nutrients 2020, 12, 3587. https://doi.org/10.3390/nu12113587
Oxfeldt M, Dalgaard LB, Risikesan J, Johansen FT, Hansen M. Influence of Fermented Red Clover Extract on Skeletal Muscle in Early Postmenopausal Women: A Double-Blinded Cross-Over Study. Nutrients. 2020; 12(11):3587. https://doi.org/10.3390/nu12113587
Chicago/Turabian StyleOxfeldt, Mikkel, Line Barner Dalgaard, Jeyanthini Risikesan, Frank Ted Johansen, and Mette Hansen. 2020. "Influence of Fermented Red Clover Extract on Skeletal Muscle in Early Postmenopausal Women: A Double-Blinded Cross-Over Study" Nutrients 12, no. 11: 3587. https://doi.org/10.3390/nu12113587
APA StyleOxfeldt, M., Dalgaard, L. B., Risikesan, J., Johansen, F. T., & Hansen, M. (2020). Influence of Fermented Red Clover Extract on Skeletal Muscle in Early Postmenopausal Women: A Double-Blinded Cross-Over Study. Nutrients, 12(11), 3587. https://doi.org/10.3390/nu12113587





