Relative Contribution of Gestational Weight Gain, Gestational Diabetes, and Maternal Obesity to Neonatal Fat Mass
Abstract
1. Introduction
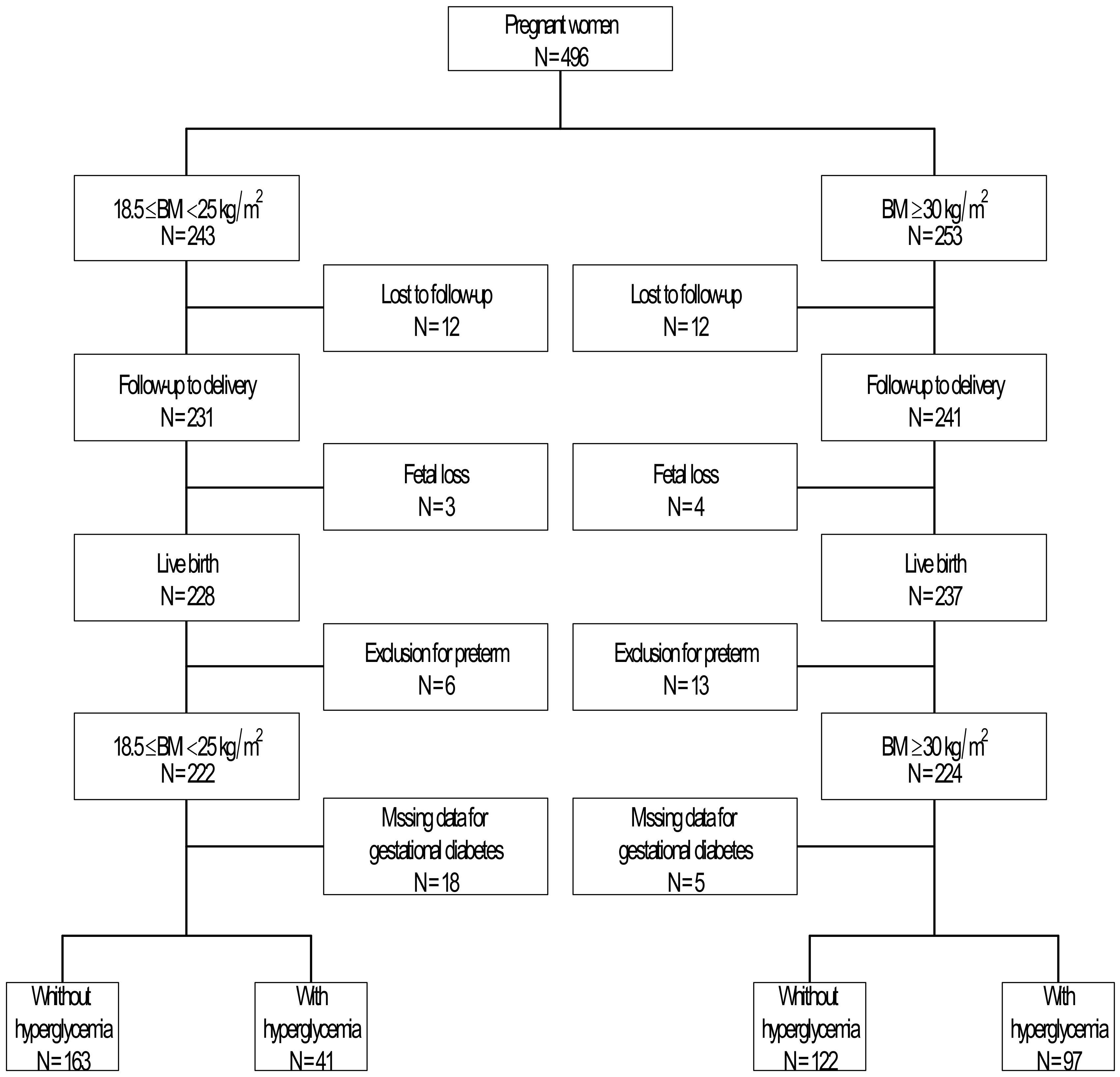
2. Materials and Methods
2.1. Maternal Weight Gain
2.2. Gestational Diabetes
2.3. Neonatal Characteristics
2.4. Biological Assays
2.5. Statistical Analysis
3. Results
3.1. Characteristics of Maternal GWG
3.2. Contribution of Total GWG to Birthweight, Skinfolds, and Leptin
3.3. Contribution of Trimester-Specific GWG on Birthweight, Skinfolds, and Leptin
4. Discussion
4.1. Principal Findings
4.2. Strengths and Limitations
4.3. GWG, Birthweight, and Neonatal Fat Mass
4.4. Interaction between Diabetes and GWG
4.5. The Specific Effect of Maternal Obesity on Fat Mass
4.6. Hypothesized Mechanisms
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Mitanchez, D.; Chavatte-Palmer, P. Review shows that maternal obesity induces serious adverse neonatal effects and is associated with childhood obesity in their offspring. Acta Paediatr. 2018, 107, 1156–1165. [Google Scholar] [CrossRef] [PubMed]
- Mitanchez, D.; Burguet, A.; Simeoni, U. Infants born to mothers with gestational diabetes mellitus: Mild neonatal effects, a long-term threat to global health. J. Pediatr. 2014, 164, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, R.F.; Abell, S.K.; Ranasinha, S.; Misso, M.; Boyle, J.A.; Black, M.H.; Li, N.; Hu, G.; Corrado, F.; Rode, L.; et al. Association of gestational weight gain with maternal and infant outcomes: A systematic review and meta-analysis. JAMA 2017, 317, 2207–2225. [Google Scholar] [CrossRef] [PubMed]
- Chiavaroli, V.; Derraik, J.G.; Hofman, P.L.; Cutfield, W.S. Born large for gestational age: Bigger is not always better. J. Pediatr. 2016, 170, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Chawla, R.; Badon, S.E.; Rangarajan, J.; Reisetter, A.C.; Armstrong, L.L.; Lowe, L.P.; Urbanek, M.; Metzger, B.E.; Hayes, M.G.; Scholtens, D.M.; et al. Genetic risk score for prediction of newborn adiposity and large-for-gestational-age birth. J. Clin. Endocrinol. Metab. 2014, 99, E2377–E2386. [Google Scholar] [CrossRef] [PubMed]
- Catalano, P.M.; McIntyre, H.D.; Cruickshank, J.K.; McCance, D.R.; Dyer, A.R.; Metzger, B.E.; Lowe, L.P.; Trimble, E.R.; Coustan, D.R.; Hadden, D.R.; et al. The hyperglycemia and adverse pregnancy outcome study: Associations of gdm and obesity with pregnancy outcomes. Diabetes Care 2012, 35, 780–786. [Google Scholar] [CrossRef] [PubMed]
- Badon, S.E.; Dyer, A.R.; Josefson, J.L.; Group, H.S.C.R. Gestational weight gain and neonatal adiposity in the hyperglycemia and adverse pregnancy outcome study-north american region. Obesity 2014, 22, 1731–1738. [Google Scholar] [CrossRef]
- Hull, H.R.; Thornton, J.C.; Ji, Y.; Paley, C.; Rosenn, B.; Mathews, P.; Navder, K.; Yu, A.; Dorsey, K.; Gallagher, D. Higher infant body fat with excessive gestational weight gain in overweight women. Am. J. Obstet. Gynecol. 2011, 205, 211.e1–211.e7. [Google Scholar] [CrossRef]
- Gallo, L.A.; Barrett, H.L.; Dekker Nitert, M. Review: Placental transport and metabolism of energy substrates in maternal obesity and diabetes. Placenta 2017, 54, 59–67. [Google Scholar] [CrossRef]
- Mitanchez, D.; Jacqueminet, S.; Nizard, J.; Tanguy, M.L.; Ciangura, C.; Lacorte, J.M.; De Carne, C.; Foix L’Helias, L.; Chavatte-Palmer, P.; Charles, M.A.; et al. Effect of maternal obesity on birthweight and neonatal fat mass: A prospective clinical trial. PLoS ONE 2017, 12, e0181307. [Google Scholar] [CrossRef]
- Institute of Medicine. Weight Gain during Pregnancy: Reexamining the Guidelines; National Academies Press: Washington, DC, USA, 2009. [Google Scholar]
- Metzger, B.E.; Gabbe, S.G.; Persson, B.; Buchanan, T.A.; Catalano, P.A.; Damm, P.; Dyer, A.R.; Leiva, A.; Hod, M.; Kitzmiler, J.L.; et al. International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care 2010, 33, 676–682. [Google Scholar] [CrossRef] [PubMed]
- Schmelzle, H.R.; Fusch, C. Body fat in neonates and young infants: Validation of skinfold thickness versus dual-energy x-ray absorptiometry. Am. J. Clin. Nutr. 2002, 76, 1096–1100. [Google Scholar] [CrossRef] [PubMed]
- Lin, L.I. A concordance correlation coefficient to evaluate reproducibility. Biometrics 1989, 45, 255–268. [Google Scholar] [CrossRef] [PubMed]
- Proust-Lima, C.; Philipps, V.; Liquet, B. Estimation of Extended Mixed Models Using Latent Classes and Latent Processes: The R Package lcmm. J. Stat. Softw. 2017, 78, 1–56. [Google Scholar] [CrossRef]
- Catalano, P.M.; Shankar, K. Obesity and pregnancy: Mechanisms of short term and long term adverse consequences for mother and child. BMJ 2017, 356, j1. [Google Scholar] [CrossRef]
- Tessier, D.R.; Ferraro, Z.M.; Gruslin, A. Role of leptin in pregnancy: Consequences of maternal obesity. Placenta 2013, 34, 205–211. [Google Scholar] [CrossRef]
- Kiel, D.W.; Dodson, E.A.; Artal, R.; Boehmer, T.K.; Leet, T.L. Gestational weight gain and pregnancy outcomes in obese women: How much is enough? Obstet. Gynecol. 2007, 110, 752–758. [Google Scholar] [CrossRef]
- Robillard, P.Y.; Dekker, G.; Boukerrou, M.; Le Moullec, N.; Hulsey, T.C. Relationship between pre-pregnancy maternal bmi and optimal weight gain in singleton pregnancies. Heliyon 2018, 4, e00615. [Google Scholar] [CrossRef]
- Faucher, M.A.; Barger, M.K. Gestational weight gain in obese women by class of obesity and select maternal/newborn outcomes: A systematic review. Women Birth 2015, 28, e70–e79. [Google Scholar] [CrossRef]
- Broskey, N.T.; Wang, P.; Li, N.; Leng, J.; Li, W.; Wang, L.; Gilmore, L.A.; Hu, G.; Redman, L.M. Early pregnancy weight gain exerts the strongest effect on birth weight, posing a critical time to prevent childhood obesity. Obesity 2017, 25, 1569–1576. [Google Scholar] [CrossRef]
- Karachaliou, M.; Georgiou, V.; Roumeliotaki, T.; Chalkiadaki, G.; Daraki, V.; Koinaki, S.; Dermitzaki, E.; Sarri, K.; Vassilaki, M.; Kogevinas, M.; et al. Association of trimester-specific gestational weight gain with fetal growth, offspring obesity, and cardiometabolic traits in early childhood. Am. J. Obstet. Gynecol. 2015, 212, 502.e1–502.e14. [Google Scholar] [CrossRef] [PubMed]
- Gaillard, R.; Durmus, B.; Hofman, A.; Mackenbach, J.P.; Steegers, E.A.; Jaddoe, V.W. Risk factors and outcomes of maternal obesity and excessive weight gain during pregnancy. Obesity 2013, 21, 1046–1055. [Google Scholar] [CrossRef] [PubMed]
- Ruchat, S.M.; Allard, C.; Doyon, M.; Lacroix, M.; Guillemette, L.; Patenaude, J.; Battista, M.C.; Ardilouze, J.L.; Perron, P.; Bouchard, L.; et al. Timing of excessive weight gain during pregnancy modulates newborn anthropometry. J. Obstet. Gynaecol. Can. 2016, 38, 108–117. [Google Scholar] [CrossRef]
- Josefson, J.L.; Simons, H.; Zeiss, D.M.; Metzger, B.E. Excessive gestational weight gain in the first trimester among women with normal glucose tolerance and resulting neonatal adiposity. J. Perinatol. 2016, 36, 1034–1038. [Google Scholar] [CrossRef]
- Davenport, M.H.; Ruchat, S.M.; Giroux, I.; Sopper, M.M.; Mottola, M.F. Timing of excessive pregnancy-related weight gain and offspring adiposity at birth. Obstet. Gynecol. 2013, 122, 255–261. [Google Scholar] [CrossRef]
- Aiken, C.E.M.; Hone, L.; Murphy, H.R.; Meek, C.L. Improving outcomes in gestational diabetes: Does gestational weight gain matter? Diabet. Med. 2019, 36, 167–176. [Google Scholar] [CrossRef]
- Desoye, G.; Nolan, C.J. The fetal glucose steal: An underappreciated phenomenon in diabetic pregnancy. Diabetologia 2016, 59, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- Waters, T.P.; Huston-Presley, L.; Catalano, P.M. Neonatal body composition according to the revised institute of medicine recommendations for maternal weight gain. J. Clin. Endocrinol. Metab. 2012, 97, 3648–3654. [Google Scholar] [CrossRef]
- Martino, J.; Sebert, S.; Segura, M.T.; Garcia-Valdes, L.; Florido, J.; Padilla, M.C.; Marcos, A.; Rueda, R.; McArdle, H.J.; Budge, H.; et al. Maternal body weight and gestational diabetes differentially influence placental and pregnancy outcomes. J. Clin. Endocrinol. Metab. 2016, 101, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Brett, K.E.; Ferraro, Z.M.; Holcik, M.; Adamo, K.B. Placenta nutrient transport-related gene expression: The impact of maternal obesity and excessive gestational weight gain. J. Matern. Fetal Neonatal Med. 2016, 29, 1399–1405. [Google Scholar] [CrossRef]
- Vrachnis, N.; Antonakopoulos, N.; Iliodromiti, Z.; Dafopoulos, K.; Siristatidis, C.; Pappa, K.I.; Deligeoroglou, E.; Vitoratos, N. Impact of maternal diabetes on epigenetic modifications leading to diseases in the offspring. Exp. Diabetes Res. 2012, 2012, 538474. [Google Scholar] [CrossRef]
- Heerwagen, M.J.; Miller, M.R.; Barbour, L.A.; Friedman, J.E. Maternal obesity and fetal metabolic programming: A fertile epigenetic soil. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 299, R711–R722. [Google Scholar] [CrossRef]
- McMillen, I.C.; Edwards, L.J.; Duffield, J.; Muhlhausler, B.S. Regulation of leptin synthesis and secretion before birth: Implications for the early programming of adult obesity. Reproduction 2006, 131, 415–427. [Google Scholar] [CrossRef][Green Version]
- Briffa, J.F.; McAinch, A.J.; Romano, T.; Wlodek, M.E.; Hryciw, D.H. Leptin in pregnancy and development: A contributor to adulthood disease? Am. J. Physiol. Endocrinol. Metab. 2015, 308, E335–E350. [Google Scholar] [CrossRef]
- Mando, C.; Calabrese, S.; Mazzocco, M.I.; Novielli, C.; Anelli, G.M.; Antonazzo, P.; Cetin, I. Sex specific adaptations in placental biometry of overweight and obese women. Placenta 2016, 38, 1–7. [Google Scholar] [CrossRef]
- Leon-Garcia, S.M.; Roeder, H.A.; Nelson, K.K.; Liao, X.; Pizzo, D.P.; Laurent, L.C.; Parast, M.M.; LaCoursiere, D.Y. Maternal obesity and sex-specific differences in placental pathology. Placenta 2016, 38, 33–40. [Google Scholar] [CrossRef]
- Brass, E.; Hanson, E.; O’Tierney-Ginn, P.F. Placental oleic acid uptake is lower in male offspring of obese women. Placenta 2013, 34, 503–509. [Google Scholar] [CrossRef] [PubMed]
- Joshi, A.; Azuma, R.; Akumuo, R.; Goetzl, L.; Pinney, S.E. Gestational diabetes and maternal obesity are associated with sex-specific changes in mirna and target gene expression in the fetus. Int. J. Obes. 2020, 44, 1497–1507. [Google Scholar] [CrossRef]
- Isganaitis, E. Developmental programming of body composition: Update on evidence and mechanisms. Curr. Diabetes Rep. 2019, 19, 60. [Google Scholar] [CrossRef] [PubMed]
- Zou, T.; He, D.; Yu, B.; Yu, J.; Mao, X.; Zheng, P.; He, J.; Huang, Z.; Shu, Y.; Liu, Y.; et al. Moderately increased maternal dietary intake delays foetal skeletal muscle differentiation and maturity in pigs. Eur. J. Nutr. 2016, 55, 1777–1787. [Google Scholar] [CrossRef] [PubMed]
| 18.5 ≤ BMI < 25 kg/m2 N = 204 | BMI ≥ 30 kg/m2 N = 219 | P-Value | |
|---|---|---|---|
| Maternal characteristics | |||
| Age (years) | 31.0 (4.1) | 30.7 (4.7) | NS |
| Primiparity n (%) | 72 (35.0) | 87 (40.0) | NS |
| Gestational age at inclusion (weeks) | 15.3 (2.1) | 15.2 (2.3) | NS |
| BMI before pregnancy (kg/m2) | 21.3 (1.7) | 34.7 (4.6) | <0.0001 |
| Ethnicity n (%) | <0.0001 | ||
| White European | 141 (69.5) | 74 (34.1) | |
| Northern Africa | 17 (8.4) | 50 (23.0) | |
| Sub Saharan Africa | 28 (13.8) | 86 (39.6) | |
| Other | 17 (8.4) | 7 (3.2) | |
| Gestational diabetes n (%) | 41 (20.0) | 97 (44.0) | <0.0001 |
| Total gestational weight gain (kg) | 13.3 (4.3) | 8.2 (7.5) | <0.0001 |
| Relative weight gain (%) | 22.9 (7.5) | 9.1 (7.9) | <0.0001 |
| 1st trimester weight gain | 5.4 (3.1) | 3.0 (5.0) | <0.0001 |
| 2nd trimester weight gain | 3.6 (2.6) | 2.4 (2.7) | |
| 3rd trimester weight gain | 4.4 (2.5) | 2.9 (3.8) | |
| Within recommended range n (%) * | 91 (48.0) | 48 (23.0) | <0.0001 |
| More than recommended n (%) | 49 (26.0) | 103 (50.0) | |
| Less than recommended n (%) | 51 (27.0) | 57 (27.0) | |
| Neonatal characteristics | |||
| Gestational age (weeks) | 39.7 (1.1) | 39.6 (1.1) | NS |
| Boys n (%) | 106 (52) | 99 (45) | NS |
| Birthweight (g) | |||
| Boys | 3466 (427) | 3397 (463) | NS |
| Girls | 3307 (410) | 3396 (405) | NS |
| Sum of skinfolds (mm) | |||
| Boys | 18.2 (3.5) | 18.9 (4.3) | NS |
| Girls | 17.7 (3.0) | 19.9 (3.6) | <0.0001 |
| Cord blood leptin (ng/mL) | |||
| Boys | 8.9 (8.2) | 9.0 (6.5) | NS |
| Girls | 10.6 (8.1) | 15.4 (11.2) | 0.001 |
| Birthweight (g) | Skinfold Thickness (mm) | Cord Leptin (ng/mL) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Model I | Estimate | CI 95% | p | Estimate | CI 95% | p | Estimate | CI 95% | p |
| (Intercept) | 3298.3 | 3180.2–3416.5 | <0.001 | 16.54 | 15.52–17.56 | <0.001 | 6.14 | 3.42–8.87 | <0.001 |
| Gestational weight gain (per kg) | 9.9 | 3.1–16.7 | 0.004 | 0.08 | 0.02–0.14 | 0.009 | 0.12 | −0.04 to 0.27 | NS |
| Gestational diabetes | 26.9 | −66.4 to 120.2 | NS | 0.91 | 0.11–1.72 | 0.027 | 0.95 | −1.18 to 3.07 | NS |
| Obesity | 36.2 | −55.3 to 127.8 | NS | 1.63 | 0.84–2.42 | <0.001 | 2.88 | 0.77–5.00 | 0.008 |
| Newborn sex (girl) | −79.9 | −163.0 to 3.2 | 0.059 | 0.33 | −0.38 to 1.05 | NS | 4.00 | 2.08–5.92 | <0.001 |
| Model II | |||||||||
| (Intercept) | 3205.855 | 2991.2–3420.5 | <0.001 | 16.19 | 14.35–18.03 | <0.001 | 6.40 | 1.46–11.33 | 0.011 |
| Gestational weight gain (per kg) | 17.174 | 1.4–32.9 | 0.032 | 0.10 | −0.03 to 0.23 | NS | 0.09 | −0.26 to 0.45 | NS |
| Gestational diabetes | 86.766 | −76.0 to 249.5 | NS | 0.57 | −0.82 to 1.95 | NS | −2.94 | −6.65 to 0.77 | NS |
| Obesity | 102.848 | −117.5 to 323.2 | NS | 2.39 | 0.51–4.26 | 0.013 | 4.67 | −0.36 to 9.70 | NS |
| Newborn sex (girl) | −77.563 | −235.9 to 80.8 | NS | 0.15 | −1.20 to 1.51 | NS | 5.22 | 1.64–8.79 | 0.004 |
| Gestational weight gain: gestational diabetes | −6.321 | −19.4 to 6.7 | NS | 0.03 | −0.08 to 0.14 | NS | 0.37 | 0.08–0.66 | 0.013 |
| Gestational weight gain: obesity | −5.304 | −21.7 to 11.1 | NS | −0.06 | −0.20 to 0.08 | NS | −0.15 | −0.52 to 0.22 | NS |
| Gestational weight gain: newborn sex (girl) | −0.007 | −12.6 to 12.6 | NS | 0.02 | −0.09 to 0.12 | NS | −0.12 | −0.40 to 0.16 | NS |
| Birthweight (g) | Skinfold Thickness (mm) | Cord Leptin (ng/mL) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Model I | Estimate | CI 95% | p | Estimate | CI 95% | p | Estimate | CI 95% | p |
| (Intercept) | 3375.8 | 3277.4–3474.3 | <0.001 | 16.83 | 16.05–17.61 | <0.001 | 4.33 | 2.10–6.56 | <0.001 |
| 3rd trimester weight gain (per kg) | 13.4 | −0.19 to 27.1 | 0.053 | 0.17 | 0.06–0.28 | 0.002 | 0.64 | 0.33–0.96 | <0.001 |
| Gestational diabetes | 22.1 | −75.5 to 119.7 | NS | 0.78 | −0.00 to 1.56 | 0.051 | 2.09 | −0.03 to 4.21 | 0.053 |
| Obesity | 9.2 | −79.9 to 98.4 | NS | 1.42 | 0.71–2.13 | <0.001 | 3.50 | 1.52–5.47 | 0.001 |
| Newborn sex (girl) | −86.7 | −172.0 to −1.4 | 0.046 | 0.31 | −0.37 to 0.99 | NS | 3.90 | 2.02–5.78 | <0.001 |
| Model II | |||||||||
| (Intercept) | 3389.3 | 3242.5–3536.1 | <0.001 | 17.17 | 16.00–18.33 | <0.001 | 2.52 | −0.76 to 5.80 | 0.132 |
| 3rd trimester weight gain (per kg) | 10.3 | −19.4 to 40.0 | NS | 0.08 | −0.15 to 0.32 | NS | 1.09 | 0.44–1.75 | 0.001 |
| Gestational diabetes | −84.1 | −217.1 to 49.0 | NS | 0.23 | −0.83 to 1.30 | NS | −0.90 | −3.82 to 2.03 | NS |
| Obesity | 76.7 | −72.8 to 226.3 | NS | 1.68 | 0.49–2.87 | 0.006 | 6.91 | 3.59–10.23 | 0.000 |
| Newborn sex (girl) | −94.3 | −221.4 to 32.7 | NS | −0.13 | −1.15 to 0.89 | NS | 5.59 | 2.77–8.41 | 0.000 |
| 3rd trimester weight gain: gestational diabetes | 32.2 | 4.4–60.0 | 0.023 | 0.15 | −0.07 to 0.37 | NS | 0.89 | 0.27–1.51 | 0.005 |
| 3rd trimester weight gain: obesity | −15.0 | −44.7 to 14.8 | NS | −0.05 | −0.29 to 0.18 | NS | −0.82 | −1.48 to −0.15 | 0.016 |
| 3rd trimester weight gain: newborn sex (girl) | −0.1 | −26.1 to 25.8 | NS | 0.11 | −0.10 to 0.31 | NS | −0.52 | −1.10 to 0.06 | NS |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mitanchez, D.; Jacqueminet, S.; Lebbah, S.; Dommergues, M.; Hajage, D.; Ciangura, C. Relative Contribution of Gestational Weight Gain, Gestational Diabetes, and Maternal Obesity to Neonatal Fat Mass. Nutrients 2020, 12, 3434. https://doi.org/10.3390/nu12113434
Mitanchez D, Jacqueminet S, Lebbah S, Dommergues M, Hajage D, Ciangura C. Relative Contribution of Gestational Weight Gain, Gestational Diabetes, and Maternal Obesity to Neonatal Fat Mass. Nutrients. 2020; 12(11):3434. https://doi.org/10.3390/nu12113434
Chicago/Turabian StyleMitanchez, Delphine, Sophie Jacqueminet, Said Lebbah, Marc Dommergues, David Hajage, and Cécile Ciangura. 2020. "Relative Contribution of Gestational Weight Gain, Gestational Diabetes, and Maternal Obesity to Neonatal Fat Mass" Nutrients 12, no. 11: 3434. https://doi.org/10.3390/nu12113434
APA StyleMitanchez, D., Jacqueminet, S., Lebbah, S., Dommergues, M., Hajage, D., & Ciangura, C. (2020). Relative Contribution of Gestational Weight Gain, Gestational Diabetes, and Maternal Obesity to Neonatal Fat Mass. Nutrients, 12(11), 3434. https://doi.org/10.3390/nu12113434





