Dietary Patterns and Their Association with Body Composition and Cardiometabolic Markers in Children and Adolescents: Genobox Cohort
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sample
2.2. Body Composition Indicators
2.3. Blood Pressure
2.4. Blood Samples and Biomarkers
2.5. Dietary Assessment
2.6. Covariates
2.7. Statistical Analyses
3. Results
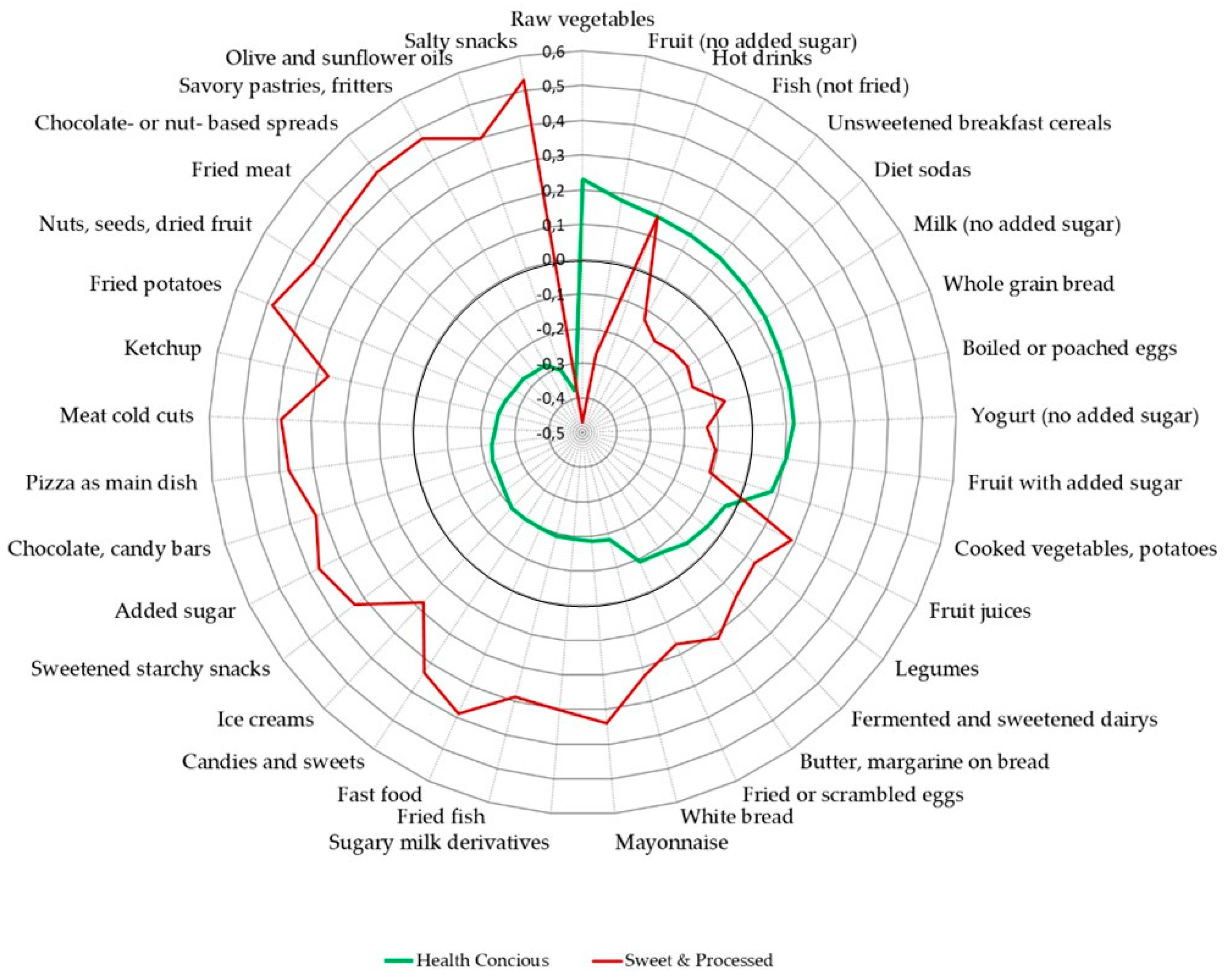
3.1. Dietary Patterns
3.2. Obesity Related Cardiometabolic Risk Indicators and Dietary Patterns
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Obesity and Overweight. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 29 October 2020).
- Perez-Farinos, N.; Lopez-Sobaler, A.M.; Dal Re, M.A.; Villar, C.; Labrado, E.; Robledo, T.; Ortega, R.M. The ALADINO study: A national study of prevalence of overweight and obesity in Spanish children in 2011. Biomed. Res. Int. 2013, 2013, 163687. [Google Scholar] [CrossRef] [PubMed]
- Han, J.C.; Lawlor, D.A.; Kimm, S.Y. Childhood obesity. Lancet 2010, 375, 1737–1748. [Google Scholar] [CrossRef]
- Consultation, W.F.E. Joint WHO/FAO Expert Consultation: Diet, Nutrition and the Prevention of Chronic Diseases; WHO: Geneva, Switzerland, 2003. [Google Scholar]
- GBD-Collaborators. Global, regional, and national comparative risk assessment of 84 behavioural, environmental and occupational, and metabolic risks or clusters of risks, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1345–1422. [Google Scholar] [CrossRef]
- Devlin, U.M.; McNulty, B.A.; Nugent, A.P.; Gibney, M.J. The use of cluster analysis to derive dietary patterns: Methodological considerations, reproducibility, validity and the effect of energy mis-reporting. Proc. Nutr. Soc. 2012, 71, 599–609. [Google Scholar] [CrossRef] [PubMed]
- Newby, P.K.; Tucker, K.L. Empirically derived eating patterns using factor or cluster analysis: A review. Nutr. Rev. 2004, 62, 177–203. [Google Scholar] [CrossRef]
- Wirfalt, E.; Drake, I.; Wallstrom, P. What do review papers conclude about food and dietary patterns? Food Nutr. Res. 2013, 57. [Google Scholar] [CrossRef]
- Smith, A.D.; Emmett, P.M.; Newby, P.K.; Northstone, K. Dietary patterns and changes in body composition in children between 9 and 11 years. Food Nutr. Res. 2014, 58. [Google Scholar] [CrossRef]
- Fernandez-Alvira, J.M.; Bammann, K.; Eiben, G.; Hebestreit, A.; Kourides, Y.A.; Kovacs, E.; Michels, N.; Pala, V.; Reisch, L.; Russo, P.; et al. Prospective associations between dietary patterns and body composition changes in European children: The IDEFICS study. Public Health Nutr. 2017, 20, 3257–3265. [Google Scholar] [CrossRef][Green Version]
- Leech, R.M.; McNaughton, S.A.; Timperio, A. The clustering of diet, physical activity and sedentary behavior in children and adolescents: A review. Int. J. Behav. Nutr. Phys. Act. 2014, 11, 4. [Google Scholar] [CrossRef]
- Cunha, C.M.; Costa, P.R.F.; de Oliveira, L.P.M.; Queiroz, V.A.O.; Pitangueira, J.C.D.; Oliveira, A.M. Dietary patterns and cardiometabolic risk factors among adolescents: Systematic review and meta-analysis. Br. J. Nutr. 2018, 119, 859–879. [Google Scholar] [CrossRef]
- Aranceta, J.; Perez-Rodrigo, C.; Ribas, L.; Serra-Majem, L. Sociodemographic and lifestyle determinants of food patterns in Spanish children and adolescents: The enKid study. Eur. J. Clin. Nutr. 2003, 57 (Suppl. 1), S40–S44. [Google Scholar] [CrossRef]
- Perez-Rodrigo, C.; Gil, A.; Gonzalez-Gross, M.; Ortega, R.M.; Serra-Majem, L.; Varela-Moreiras, G.; Aranceta-Bartrina, J. Clustering of Dietary Patterns, Lifestyles, and Overweight among Spanish Children and Adolescents in the ANIBES Study. Nutrients 2015, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Plaza-Díaz, J.; Molina-Montes, E.; Soto-Méndez, M.J.; Madrigal, C.; Hernández-Ruiz, Á.; Valero, T.; Lara Villoslada, F.; Leis, R.; Martínez de Victoria, E.; Moreno, J.M.; et al. Clustering of Dietary Patterns and Lifestyles Among Spanish Children in the EsNuPI Study. Nutrients 2020, 12, 2536. [Google Scholar] [CrossRef]
- Bodega, P.; Fernández-Alvira, J.M.; Santos-Beneit, G.; de Cos-Gandoy, A.; Fernández-Jiménez, R.; Moreno, L.A.; de Miguel, M.; Carral, V.; Orrit, X.; Carvajal, I.; et al. Dietary Patterns and Cardiovascular Risk Factors in Spanish Adolescents: A Cross-Sectional Analysis of the SI! Program for Health Promotion in Secondary Schools. Nutrients 2019, 11, 2297. [Google Scholar] [CrossRef]
- Stewart, A.; Marfell-Jones, M.; Olds, T.; de Ridder, H. Protocolo Internacional Para la Valoración Antropométrica; Sociedad internacional para el avance de la cineantropometría, Universidad Católica de Murcia, Campus de los Jerónimos: Guadalupe (Murcia), España, Spain, 2011. [Google Scholar]
- Sobradillo, B.; Aguirre, A.; Aresti, U.; Bilbao, A.; Fernández-Ramos, C.; Lizárraga, A.; Lorenzo, H.; Madariaga, L.; Rica, I.; Ruiz, I. Curvas y tablas de crecimiento (estudios longitudinal y transversal). Fundación Faustino Orbegozo Eizaguirre. In Curvas y Tablas de Crecimiento (Estudios Longitudinal y Transversal); Sobradillo, B., Aguirre, A., Aresti, U., Bilbao, A., Fernández-Ramos, C., Lizárraga, A., Lorenzo, H., Madariaga, L., Rica, I.I.R., Eds.; Fundación Faustino Orbegozo Eizaguirre: Madrid, Spain, 2004. [Google Scholar]
- Cole, T.J.; Bellizzi, M.C.; Flegal, K.M.; Dietz, W.H. Establishing a standard definition for child overweight and obesity worldwide: International survey. BMJ 2000, 320, 1240–1243. [Google Scholar] [CrossRef]
- Wells, J.C.; Williams, J.E.; Chomtho, S.; Darch, T.; Grijalva-Eternod, C.; Kennedy, K.; Haroun, D.; Wilson, C.; Cole, T.J.; Fewtrell, M.S. Body-composition reference data for simple and reference techniques and a 4-component model: A new UK reference child. Am. J. Clin. Nutr. 2012, 96, 1316–1326. [Google Scholar] [CrossRef]
- Garcia-Rodriguez, C.E.; Mesa, M.D.; Olza, J.; Vlachava, M.; Kremmyda, L.S.; Diaper, N.D.; Noakes, P.S.; Miles, E.A.; Ramirez-Tortosa, M.C.; Liaset, B.; et al. Does consumption of two portions of salmon per week enhance the antioxidant defense system in pregnant women? Antioxid. Redox Signal. 2012, 16, 1401–1406. [Google Scholar] [CrossRef] [PubMed]
- Aebi, H. Catalase in vitro. Methods Enzymol. 1984, 105, 121–126. [Google Scholar]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef]
- Tojo, R.; Leis, R. Alimentación del niño escolar. In Manual Práctico de Nutrición en Pediatría; Pediatría, A., Ed.; Ergón: Jackson, MS, USA, 2007; pp. 91–106. [Google Scholar]
- UNESCO. International Standard Classification of Education (ISCED); UNESCO: Paris, France, 2011. [Google Scholar]
- Verbestel, V.; De Henauw, S.; Bammann, K.; Barba, G.; Hadjigeorgiou, C.; Eiben, G.; Konstabel, K.; Kovács, E.; Pitsiladis, Y.; Reisch, L.; et al. Are context-specific measures of parental-reported physical activity and sedentary behaviour associated with accelerometer data in 2-9-year-old European children? Public Health Nutr. 2015, 18, 860–868. [Google Scholar] [CrossRef]
- Tanner, J.M.; Whitehouse, R.H. Clinical longitudinal standards for height, weight, height velocity, weight velocity, and stages of puberty. Arch. Dis. Child. 1976, 51, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Leis, R.; Jurado-Castro, J.M.; Llorente-Cantarero, F.J.; Anguita-Ruiz, A.; Iris-Rupérez, A.; Bedoya-Carpente, J.J.; Vázquez-Cobela, R.; Aguilera, C.M.; Bueno, G.; Gil-Campos, M. Cluster Analysis of Physical Activity Patterns, and Relationship with Sedentary Behavior and Healthy Lifestyles in Prepubertal Children: Genobox Cohort. Nutrients 2020, 12, 1288. [Google Scholar] [CrossRef]
- Aranceta-Bartrina, J.; Partearroyo, T.; López-Sobaler, A.M.; Ortega, R.M.; Varela-Moreiras, G.; Serra-Majem, L.; Pérez-Rodrigo, C. Updating the Food-Based Dietary Guidelines for the Spanish Population: The Spanish Society of Community Nutrition (SENC) Proposal. Nutrients 2019, 11, 2675. [Google Scholar] [CrossRef] [PubMed]
- Serra-Majem, L.; Aranceta Bartrina, J.; Perez-Rodrigo, C.; Ribas-Barba, L.; Delgado-Rubio, A. Prevalence and deteminants of obesity in Spanish children and young people. Br. J. Nutr. 2006, 96 (Suppl. 1), S67–S72. [Google Scholar] [CrossRef]
- Aranceta-Bartrina, J.; Perez-Rodrigo, C. Determinants of childhood obesity: ANIBES study. Nutr. Hosp. 2016, 33, 339. [Google Scholar] [CrossRef]
- Samaniego-Vaesken, M.L.; Partearroyo, T.; Ruiz, E.; Aranceta-Bartrina, J.; Gil, A.; Gonzalez-Gross, M.; Ortega, R.M.; Serra-Majem, L.; Varela-Moreiras, G. The Influence of Place of Residence, Gender and Age Influence on Food Group Choices in the Spanish Population: Findings from the ANIBES Study. Nutrients 2018, 10, 392. [Google Scholar] [CrossRef]
- Fernández-Alvira, J.M.; Börnhorst, C.; Bammann, K.; Gwozdz, W.; Krogh, V.; Hebestreit, A.; Barba, G.; Reisch, L.; Eiben, G.; Iglesia, I.; et al. Prospective associations between socio-economic status and dietary patterns in European children: The Identification and Prevention of Dietary- and Lifestyle-induced Health Effects in Children and Infants (IDEFICS) Study. Br. J. Nutr. 2015, 113, 517–525. [Google Scholar] [CrossRef]
- Van der Sluis, M.E.; Lien, N.; Twisk, J.W.; Steenhuis, I.H.; Bere, E.; Klepp, K.I.; Wind, M. Longitudinal associations of energy balance-related behaviours and cross-sectional associations of clusters and body mass index in Norwegian adolescents. Public Health Nutr. 2010, 13, 1716–1721. [Google Scholar] [CrossRef]
- Boone-Heinonen, J.; Gordon-Larsen, P.; Adair, L.S. Obesogenic clusters: Multidimensional adolescent obesity-related behaviors in the U.S. Ann. Behav. Med. 2008, 36, 217–230. [Google Scholar] [CrossRef]
- Patel, C.; Ghanim, H.; Ravishankar, S.; Sia, C.L.; Viswanathan, P.; Mohanty, P.; Dandona, P. Prolonged reactive oxygen species generation and nuclear factor-kappaB activation after a high-fat, high-carbohydrate meal in the obese. J. Clin. Endocrinol. Metab. 2007, 92, 4476–4479. [Google Scholar] [CrossRef]
- Yip, C.S.C.; Chan, W.; Fielding, R. The Associations of Fruit and Vegetable Intakes with Burden of Diseases: A Systematic Review of Meta-Analyses. J. Acad. Nutr. Diet. 2019, 119, 464–481. [Google Scholar] [CrossRef] [PubMed]
- Aguila, Q.; Ramon, M.A.; Matesanz, S.; Vilatimo, R.; Del Moral, I.; Brotons, C.; Ulied, A. Assessment study of the nutritional status, eating habits and physical activity of the schooled population of Centelles, Hostalets de Balenya and Sant Marti de Centelles (ALIN 2014 Study). Endocrinol. Diabetes Nutr. 2017, 64, 138–145. [Google Scholar] [CrossRef]
- Guthold, R.; Stevens, G.A.; Riley, L.M.; Bull, F.C. Global trends in insufficient physical activity among adolescents: A pooled analysis of 298 population-based surveys with 1·6 million participants. Lancet Child Adolesc. Health 2020, 4, 23–35. [Google Scholar] [CrossRef]
- Ortega Anta, R.M.; Lopez-Solaber, A.M.; Perez-Farinos, N. Associated factors of obesity in Spanish representative samples. Nutr. Hosp. 2013, 28 (Suppl. 5), 56–62. [Google Scholar] [CrossRef]
- Fernandez-Alvira, J.M.; De Bourdeaudhuij, I.; Singh, A.S.; Vik, F.N.; Manios, Y.; Kovacs, E.; Jan, N.; Brug, J.; Moreno, L.A. Clustering of energy balance-related behaviors and parental education in European children: The ENERGY-project. Int. J. Behav. Nutr. Phys. Act. 2013, 10, 5. [Google Scholar] [CrossRef]
- Asarian, L.; Geary, N. Sex differences in the physiology of eating. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013, 305, R1215–R1267. [Google Scholar] [CrossRef] [PubMed]
- De Cosmi, V.; Scaglioni, S.; Agostoni, C. Early Taste Experiences and Later Food Choices. Nutrients 2017, 9, 107. [Google Scholar] [CrossRef]
- Huon, G.; Lim, J. The emergence of dieting among female adolescents: Age, body mass index, and seasonal effects. Int. J. Eat. Disord. 2000, 28, 221–225. [Google Scholar] [CrossRef]
- Sichert-Hellert, W.; Beghin, L.; De Henauw, S.; Grammatikaki, E.; Hallstrom, L.; Manios, Y.; Mesana, M.I.; Molnar, D.; Dietrich, S.; Piccinelli, R.; et al. Nutritional knowledge in European adolescents: Results from the HELENA (Healthy Lifestyle in Europe by Nutrition in Adolescence) study. Public Health Nutr. 2011, 14, 2083–2091. [Google Scholar] [CrossRef]
- Villamor, E.; Jansen, E.C. Nutritional Determinants of the Timing of Puberty. Annu. Rev. Public Health 2016, 37, 33–46. [Google Scholar] [CrossRef]
- Bandini, L.G.; Schoeller, D.A.; Cyr, H.N.; Dietz, W.H. Validity of reported energy intake in obese and nonobese adolescents. Am. J. Clin. Nutr. 1990, 52, 421–425. [Google Scholar] [CrossRef]
- Ventura, A.K.; Loken, E.; Mitchell, D.C.; Smiciklas-Wright, H.; Birch, L.L. Understanding reporting bias in the dietary recall data of 11-year-old girls. Obesity 2006, 14, 1073–1084. [Google Scholar] [CrossRef]
- Coon, K.A.; Goldberg, J.; Rogers, B.L.; Tucker, K.L. Relationships between use of television during meals and children’s food consumption patterns. Pediatrics 2001, 107, E7. [Google Scholar] [CrossRef] [PubMed]
- Castells Cuixart, M.; Capdevila Prim, C.; Girbau Sola, T.; Rodriguez Caba, C. Study on feeding behavior in school children aged 11-13 years from Barcelona. Nutr. Hosp. 2006, 21, 517–532. [Google Scholar]
- Varela-Moreiras, G.; Requejo, A.; Ortega, R.; Zamora, S.; Salas, J.; Cabrerizo, L.; Aranceta, J.; Ávila, J.; Murillo, J.; Belmonte, S.; et al. Libro Blanco de la Nutrición en España; Fundación Española de la Nutrición; Lesinguer: Madrid, Spain, 2013. [Google Scholar]
- Taillie, L.S.; Afeiche, M.C.; Eldridge, A.L.; Popkin, B.M. The contribution of at-home and away-from-home food to dietary intake among 2-13-year-old Mexican children. Public Health Nutr. 2017, 20, 2559–2568. [Google Scholar] [CrossRef][Green Version]
- Marcano, M.; Solano, L.; Pontiles, M. Hyperlipidemia and hyperglycemia prevalence in obese children: Increased risk of cardiovascular disease? Nutr. Hosp. 2006, 21, 474–483. [Google Scholar]
- Cruz, M.L.; Bergman, R.N.; Goran, M.I. Unique effect of visceral fat on insulin sensitivity in obese Hispanic children with a family history of type 2 diabetes. Diabetes Care 2002, 25, 1631–1636. [Google Scholar] [CrossRef]
- Weiss, R.; Dufour, S.; Taksali, S.E.; Tamborlane, W.V.; Petersen, K.F.; Bonadonna, R.C.; Boselli, L.; Barbetta, G.; Allen, K.; Rife, F.; et al. Prediabetes in obese youth: A syndrome of impaired glucose tolerance, severe insulin resistance, and altered myocellular and abdominal fat partitioning. Lancet 2003, 362, 951–957. [Google Scholar] [CrossRef]
- Stevens, J.; McClain, J.E.; Truesdale, K.P. Selection of measures in epidemiologic studies of the consequences of obesity. Int. J. Obes. 2008, 32 (Suppl. 3), S60–S66. [Google Scholar] [CrossRef]
- Revenga-Frauca, J.; González-Gil, E.M.; Bueno-Lozano, G.; de Miguel-Etayo, P.; Velasco-Martínez, P.; Rey-López, J.P.; Bueno-Lozano, O.; Moreno, L.A. Abdominal fat and metabolic risk in obese children and adolescents. J. Physiol. Biochem. 2009, 65, 415–420. [Google Scholar] [CrossRef]
- Pérez-Gimeno, G.; Rupérez, A.I.; Vázquez-Cobela, R.; Herráiz-Gastesi, G.; Gil-Campos, M.; Aguilera, C.M.; Moreno, L.A.; Leis Trabazo, M.R.; Bueno-Lozano, G. Energy Dense Salty Food Consumption Frequency Is Associated with Diastolic Hypertension in Spanish Children. Nutrients 2020, 12, 1027. [Google Scholar] [CrossRef]
- Da Costa, L.A.; Badawi, A.; El-Sohemy, A. Nutrigenetics and modulation of oxidative stress. Ann. Nutr. Metab. 2012, 60 (Suppl. 3), 27–36. [Google Scholar] [CrossRef]
- Rupérez, A.I.; Mesa, M.D.; Anguita-Ruiz, A.; González-Gil, E.M.; Vázquez-Cobela, R.; Moreno, L.A.; Gil, Á.; Gil-Campos, M.; Leis, R.; Bueno, G.; et al. Antioxidants and Oxidative Stress in Children: Influence of Puberty and Metabolically Unhealthy Status. Antioxidants 2020, 9, 618. [Google Scholar] [CrossRef]
- González-Gil, E.M.; Tognon, G.; Lissner, L.; Intemann, T.; Pala, V.; Galli, C.; Wolters, M.; Siani, A.; Veidebaum, T.; Michels, N.; et al. Prospective associations between dietary patterns and high sensitivity C-reactive protein in European children: The IDEFICS study. Eur. J. Nutr. 2018, 57, 1397–1407. [Google Scholar] [CrossRef]
- Antonopoulos, A.S.; Tousoulis, D. The molecular mechanisms of obesity paradox. Cardiovasc. Res 2017, 113, 1074–1086. [Google Scholar] [CrossRef]
- Codoñer-Franch, P.; Valls-Bellés, V.; Arilla-Codoñer, A.; Alonso-Iglesias, E. Oxidant mechanisms in childhood obesity: The link between inflammation and oxidative stress. Transl. Res. 2011, 158, 369–384. [Google Scholar] [CrossRef]
- Bulló, M.; Casas-Agustench, P.; Amigó-Correig, P.; Aranceta, J.; Salas-Salvadó, J. Inflammation, obesity and comorbidities: The role of diet. Public Health Nutr. 2007, 10, 1164–1172. [Google Scholar] [CrossRef]
- Galland, L. Diet and inflammation. Nutr. Clin. Pract. 2010, 25, 634–640. [Google Scholar] [CrossRef]
- Calder, P.C.; Ahluwalia, N.; Brouns, F.; Buetler, T.; Clement, K.; Cunningham, K.; Esposito, K.; Jönsson, L.S.; Kolb, H.; Lansink, M.; et al. Dietary factors and low-grade inflammation in relation to overweight and obesity. Br. J. Nutr. 2011, 106 (Suppl. 3), S5–S78. [Google Scholar] [CrossRef]

| All n = 674 (100%) | Normal Weight n = 178 (26.4%) | Overweight and Obesity n = 496 (73.6%) | p | |
|---|---|---|---|---|
| Gender | 0.018 | |||
| Male | 307 (45.5%) | 95 (53.4%) | 212 (42.7%) | |
| Female | 367 (54.5%) | 83 (46.6%) | 284 (57.3%) | |
| Pubertal stage (Tanner) | 0.163 | |||
| Prepubertal | 333 (49.4%) | 96 (53.9%) | 237 (47.8%) | |
| Pubertal | 341 (50.6%) | 82 (46.1%) | 259 (52.2%) | |
| Maternal education level | <0.001 | |||
| Low | 58 (8.9%) | 9 (5.2%) | 49 (10.3%) | |
| Medium | 480 (73.8%) | 116 (66.7%) | 364 (76.5%) | |
| High | 112 (17.2%) | 49 (28.2%) | 63 (13.2%) | |
| Age | 0.273 | |||
| Children (5–11 years) | 439 (65.1%) | 122 (68.5%) | 317 (63.9%) | |
| Adolescents (12–16 years) | 235 (304.9%) | 56 (31.5%) | 179 (36.1%) | |
| Age (years) Mean ± SD | 10.7 (2.5) | 10.5 (2.7) | 10.7 (2.5) | 0.297 |
| BMI | <0.001 | |||
| Normal weight | 178 (26.4%) | 178 (100%) | 0 (0%) | |
| Overweight | 165 (24.5%) | 0 (0%) | 165 (33.3%) | |
| Obesity | 331 (49.1%) | 0 (0%) | 331 (66.7%) | |
| BMI (kg/m2) Mean (SD) | 24.0 (5.6) | 17.3 (2.3) | 26.4 (4.3) | <0.001 |
| BMI Z-score (kg/m2) Mean (SD) | 1.8 (1.7) | −0.3 (0.6) | 2.6 (1.3) | <0.001 |
| Health Conscious n = 403 (59.8%) | Sweet and Processed n = 271 (40.2%) | p | |
|---|---|---|---|
| Gender | 0.102 | ||
| Male | 175 (43.4%) | 132 (48.7%) | |
| Female | 228 (56.6%) | 139 (51.3%) | |
| Pubertal stage (Tanner) | 0.019 | ||
| Prepubertal | 184 (45.7%) | 149 (55.0%) | |
| Pubertal | 219 (54.3%) | 122 (45.0%) | |
| Maternal education level | 0.288 | ||
| Low | 29 (7.5%) | 29 (11.1%) | |
| Medium | 291 (75.0%) | 189 (72.1%) | |
| High | 68 (17.5%) | 44 (16.8%) | |
| Age | 0.004 | ||
| Children (5–11 years) | 245 (60.8%) | 194 (71.6%) | |
| Adolescents (12–16 years) | 158 (39.2%) | 77 (28.4%) | |
| Age (years) Mean (SD) | 10.9 (2.5) | 10.3 (2.5) | 0.002 |
| BMI | <0.001 | ||
| Normal weight | 80 (19.9%) | 98 (36.2%) | |
| Overweight | 117 (29.0%) | 48 (17.7%) | |
| Obesity | 206 (51.1%) | 125 (46.1%) |
| All (n = 674) | Normal Weight (n = 178) | Overweight (n = 165) | Obesity (n = 331) | |||||
|---|---|---|---|---|---|---|---|---|
| Health Conscious | Sweet and Processed | Health Conscious | Sweet and Processed | Health Conscious | Sweet and Processed | Health Conscious | Sweet and Processed | |
| (n = 403) | (n = 271) | (n = 80) | (n = 98) | (n = 117) | (n = 48) | (n = 206) | (n = 125) | |
| Age (years) | 10.9 (2.5) | 10.3 (2.5) ** | 10.8 (2.9) | 10.2 (2.5) | 11.7 (2.1) | 10.2 (2.1) *** | 10.5 (2.5) | 10.4 (2.6) |
| Body composition indicators | ||||||||
| BMI (kg/m2) | 24.6 (5.1) | 23.2 (6.3) | 17.7 (2.6) | 17.0 (2.0) | 23.7 (2.4) | 21.8 (2.3) ** | 27.7 (3.9) | 28.5 (4.4) |
| BMI Z-score (kg/m2) | 2.0 (1.6) | 1.6 (1.9) | −0.21 (0.56) | −0.33 (0.54) | 1.44 (0.49) | 1.22 (0.42) * | 3.11 (1.06) | 3.35 (1.18) |
| Body mass (kg) | 54.6 (18.5) | 49.5 (20.6 | 38.7 (14.0) | 34.3 (11.0) | 54.4 (12.2) | 45.9 (11.9) * | 60.9 (19.2) | 62.8 (20.3) |
| Hip circumference (cm) | 90.2 (13.6) | 83.2 (15.2) ** | 76.5 (11.4) | 72.0 (9.5) | 91.2 (9.0) | 83.5 (8.6) ** | 95.5 (12.8) | 95.3 (13.7) |
| Waist circumference (cm) | 82.3 (15.0) | 77.1 (17.6) | 64.8 (11.8) | 60.8 (5.9) | 81.7 (9.7) | 75.4 (9.4) | 89.5 (12.6) | 91.3 (14.4) |
| Waist to hip index | 0.56 (0.08) | 0.54 (0.10) | 0.85 (0.08) | 0.85 (0.07) | 0.90 (0.08) | 0.90 (0.07) | 0.93 (0.07) | 0.94 (0.09) ** |
| Waist to height index | 83.7 (30.1) | 68.1 (35.7) | 0.45 (0.05) | 0.44 (0.04) | 0.54 (0.05) | 0.52 (0.04) | 0.61 (0.06) | 0.63 (0.06) ** |
| Skinfold sum (mm) | 38.3 (10.0) | 33.9 (9.5) ** | 42.1 (18.7) | 37.2 (17.3) | 88.4 (17.7) | 77.0 (22.1) | 102.4 (18.8) | 103.0 (22.6) |
| FMI Z-score (kg/m2) | 11.6 (5.1) | 10.4 (5.6) | 3.5 (2.7) | 2.7 (1.8) | 6.4 (2.3) | 5.7 (2.4) | 9.7 (4.06) | 9.3 (4.0) |
| FFMI Z-score (kg/m2) | 10.9 (2.5) | 10.3 (2.5) | 8.1(4.2) | 7.2 (3.9) | 11.6 (4.7) | 10.3 (4.1) | 13.6 (4.9) | 14.7(5.2) |
| Cardiometabolic indicators | ||||||||
| Blood pressure | ||||||||
| SBP (mm Hg) ^ | 109 (13) | 108 (14) | 104 (12) | 100 (12) | 108 (12) | 106 (13) | 112 (14) | 121 |
| DBP (mm Hg) ^ | 65 (11) | 66 (10) * | 61 (9) | 63 (10) * | 64 (11) | 63 (8) | 67 (11) | 121 |
| General metabolic biomarkers | ||||||||
| Glucose (mg/dL) | 84 (8) | 86 (8) | 84 (8) | 86 (7) | 85 (8) | 88 (8) | 84 (8) | 85 (8) |
| Insulin (mU/L) | 12.20 (8.42) | 11.52 (9.92) | 8.00 (4.54) | 7.27 (4.87) | 11.11 (7.78) | 10.55 (6.12) | 14.47 (9.18) | 15.4 (12.44) |
| HOMA-IR | 2.57 (1.84) | 2.48 (2.18) | 1.68 (1.00) | 1.56 (1.08) | 2.35 (1.73) | 2.36 (1.53) | 3.05 (2.00) | 3.27 (2.70) |
| TG (mg/dL) | 69 (34) | 69(35) | 57(23) | 54 (23) | 66 (30) | 77 (44) | 76(38) | 77 (34) |
| Cholesterol (mg/dL) | 165 (30) | 161 (28) | 169 (26) | 164 (28) | 166 (33) | 163 (31) | 162 (30) | 159 (26) |
| LDLc (mg/dL) | 97 (26) | 92 (25) | 95 (22) | 87 (26) | 99 (30) | 93 (25) | 97 (26) | 94 (24) |
| HDLc (mg/dL) ^ | 50 (13) | 55 (15) | 59 (13) | 65 (15) | 49 (11) | 54 (14) | 47 (12) | 47 (11) |
| HDLc/LDLc index | 0.61 (0.46) | 0.81 (0.67) | 0.66 (0.22) | 0.94 (0.6) ** | 0.59 (0.43) | 0.78 (0.65) | 0.6 (0.54) | 0.72 (0.73) |
| AST (U/L) | 22 (9) | 23 (7) | 24 (8) | 25 (6) | 20 (6) | 24 (9) * | 22 (10) | 22 (7) |
| ALT (U/L) | 20 (12) | 19 (11) | 17 (9) | 16 (6) | 18 (9) | 21 (21) | 22 (14) | 20 (8) |
| GGT (U/L) | 12 (7) | 13 (7) | 10 (3) | 10 (3) | 11 (5) | 14 (13) * | 14 (9) | 15 (5) |
| Oxidative stress biomarkers | ||||||||
| Carotenes/TG | 1.71 (1.65) | 1.53 (1.25) * | 2.70 (2.32) | 2.55 (1.56) | 1.43 (1.12) | 1.34 (0.81) | 1.38 (1.36) | 1.00 (0.76) |
| Tocopherols/TG | 0.14 (0.07) | 0.15 (0.07) | 0.17 (0.07) | 0.19 (0.07) | 0.13 (0.06) | 0.12 (0.06) | 0.13 (0.08) | 0.13 (0.05) |
| TAC (mM Eq Trolox®) | 2.05 (0.87) | 2.09 (0.91) | 1.88 (0.66) | 1.99 (0.75) | 2.06 (0.85) | 2.19 (1.13) | 2.11 (0.97) | 2.17 (0.96) |
| Catalase (U/g Hb) | 164.68 (103.66) | 163.94 (153.67) ** | 119.65 (71.23) | 136.22 (119.74) | 165.71 (83.01) | 232.91 (283.63) * | 180.90 (118.02) | 162.27 (74.02) |
| Adipokines and biomarkers of inflammation and endothelial damage | ||||||||
| Adiponectin (mg/L) | 14.58 (8.67) | 15.18 (8.31) | 17.27 (11.37) | 17.08 (9.52) | 14.91 (7.86) | 14.51 (8.86) | 13.13 (7.31) | 13.51 (6.05) |
| Leptin (ug/L) | 15.56 (12.96) | 14.09 (15.82) ** | 4.59 (4.89) | 3.95 (4.26) | 12.44 (5.83) | 12.47 (9.57) | 22.47 (14.32) | 25.13 (18.36) |
| Resistin (ug/L) ^ | 20.18 (14.89) | 21.06 (14.47) * | 24.07 (21.24) | 23.85 (17.18) | 18.43 (10.37) | 18.80 (14.73) | 19.4 (13.27) | 19.24 (10.32) |
| TNFα (ng/L) ^ | 2.82 (1.72) | 2.89 (1.58) * | 2.27 (1.20) | 2.4 (1.34) | 2.67 (2.11) | 3.26 (1.95) | 3.15 (1.59) | 3.23 (1.48) |
| MCP-1 (ng/L) | 88.47 (37.69) | 92.81 (38.84) ** | 83.54 (28.28) | 85.21 (39.62) | 88.01 (48.11) | 89.70 (32.51) | 91.01 (34.32) | 102.26 (39.22) * |
| tPAI-1 (ug/L) | 22.9 (14.29) | 25.48 (17.31) *** | 16.31 (12.21) | 17.8 (12.94) | 21.76 (12.64) | 28.85 (17.23) ** | 26.61 (14.93) | 31.92 (18.34) |
| Selectin (ug/L) ^ | 27.34 (14.7) | 30.38 (16.59) | 27.86 (15.66) | 24.51 (11.69) | 22.45 (11.9) | 34.89 (20.32) * | 31.06 (15.25) | 33.6 (17.38) |
| sVCAM-1 (mg/L) ^ | 1.04 (0.32) | 1.14 (0.25) * | 1.07 (0.33) | 1.20 (0.27) | 1.00 (0.27) | 1.15 (0.28) | 1.04 (0.34) | 1.07 (0.22) |
| MPO (ug/L) | 39.99 (83.94) | 32.95 (42.29) * | 42.25 (77.48) | 36.98 (50.22) | 27.96 (29.79) | 29.97 (39.36) | 46.15 (105.63) | 30.18 (33.95) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Latorre-Millán, M.; Rupérez, A.I.; González-Gil, E.M.; Santaliestra-Pasías, A.; Vázquez-Cobela, R.; Gil-Campos, M.; Aguilera, C.M.; Gil, Á.; Moreno, L.A.; Leis, R.; et al. Dietary Patterns and Their Association with Body Composition and Cardiometabolic Markers in Children and Adolescents: Genobox Cohort. Nutrients 2020, 12, 3424. https://doi.org/10.3390/nu12113424
Latorre-Millán M, Rupérez AI, González-Gil EM, Santaliestra-Pasías A, Vázquez-Cobela R, Gil-Campos M, Aguilera CM, Gil Á, Moreno LA, Leis R, et al. Dietary Patterns and Their Association with Body Composition and Cardiometabolic Markers in Children and Adolescents: Genobox Cohort. Nutrients. 2020; 12(11):3424. https://doi.org/10.3390/nu12113424
Chicago/Turabian StyleLatorre-Millán, Miriam, Azahara I. Rupérez, Esther M. González-Gil, Alba Santaliestra-Pasías, Rocío Vázquez-Cobela, Mercedes Gil-Campos, Concepción M. Aguilera, Ángel Gil, Luis A. Moreno, Rosaura Leis, and et al. 2020. "Dietary Patterns and Their Association with Body Composition and Cardiometabolic Markers in Children and Adolescents: Genobox Cohort" Nutrients 12, no. 11: 3424. https://doi.org/10.3390/nu12113424
APA StyleLatorre-Millán, M., Rupérez, A. I., González-Gil, E. M., Santaliestra-Pasías, A., Vázquez-Cobela, R., Gil-Campos, M., Aguilera, C. M., Gil, Á., Moreno, L. A., Leis, R., & Bueno, G. (2020). Dietary Patterns and Their Association with Body Composition and Cardiometabolic Markers in Children and Adolescents: Genobox Cohort. Nutrients, 12(11), 3424. https://doi.org/10.3390/nu12113424









