Extractable and Non-Extractable Antioxidants Composition in the eBASIS Database: A Key Tool for Dietary Assessment in Human Health and Disease Research
Abstract
1. Introduction
1.1. The Main Feautures of Extractable and Non-Extractable Antioxidants
1.1.1. Chemistry
- (a)
- Polymeric polyphenols (non-extractable proanthocyanidins NEPA), high-molecular-weight proanthocyanidins that are free in the food matrix and proanthocyanidins that are complexed with protein or cell wall polysaccharides [13];
- (b)
- Small phenolic compounds, linked to carbohydrates, mainly polysaccharide constituents of dietary fibre, and to proteins (hydrolysable polyphenols; HPP). These include several classes of bioactive components, e.g., hydrolysable tannins or hydroxycinnamic acids, linked to carbohydrates and proteins via covalent bonds, hydrogen bonds and/or hydrophobic interactions [14,15].
1.1.2. Health Benefits and Role in the Prevention of Chronic Diseases
1.1.3. Occurrence in Foods and Dietary Intake Assessment
1.2. Development of Dedicated Databases on Antioxidant Properties
- Antioxidant measurements—several different assays are performed to evaluate the antioxidant properties of single compounds and foods, these differ in their principles, mechanisms, and experimental conditions, as well as in the way end points are measured [66,67]. For high-quality data, the use of at least two or three assays is strongly recommended [66,68];
- Expression of results—there are different ways of expressing antioxidant results, including kinetic parameters [16].
1.3. eBASIS
2. Materials and Methods
2.1. eBASIS
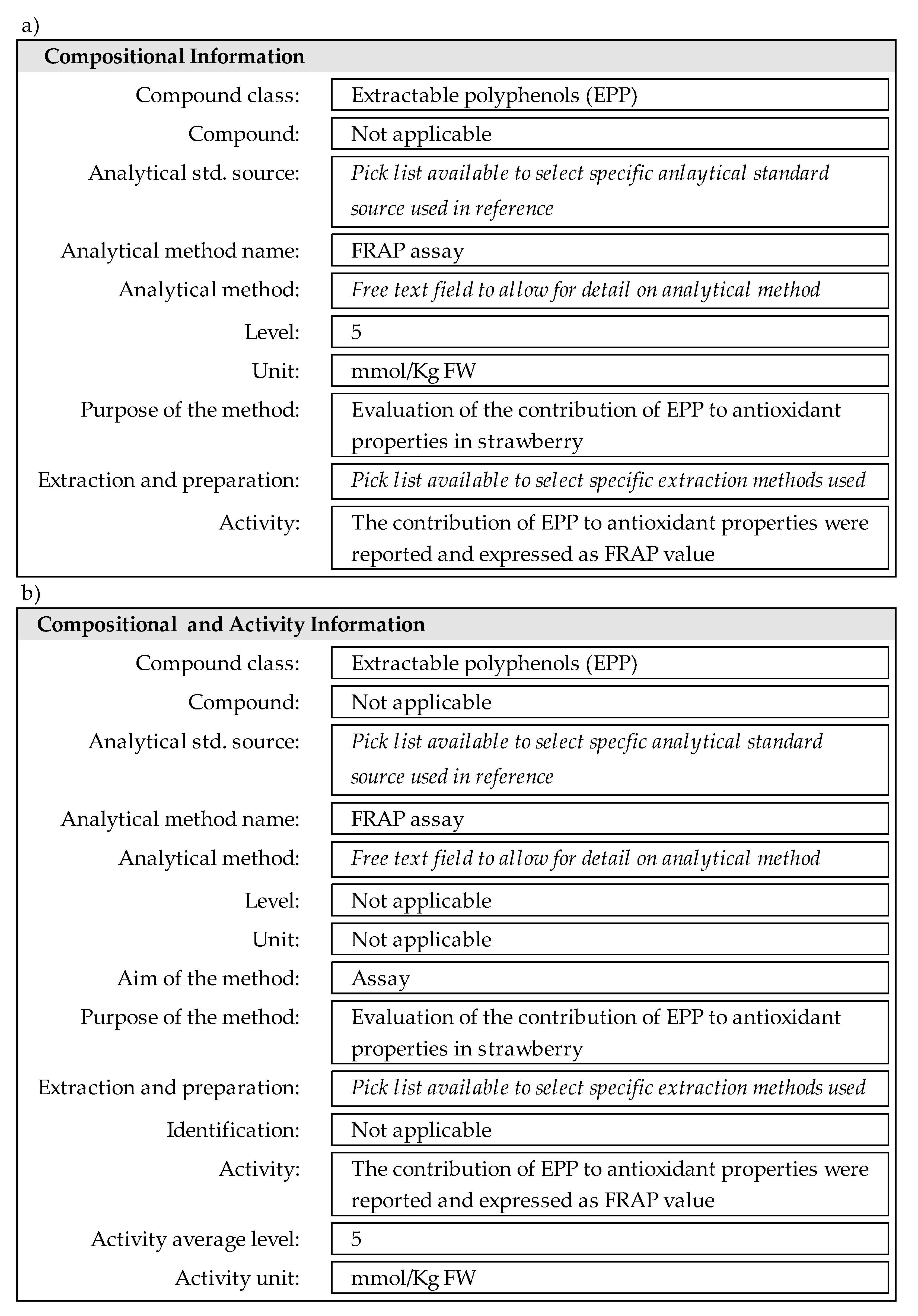
2.2. eBASIS Revisions to Expand the Database for Antioxidants
- Extractable polyphenols (EPP);
- Extractable proanthocyanidins (EPA);
- Non-extractable polyphenols (NEPP);
- Non-extractable proanthocyanidins (NEPA);
- Hydrolysable polyphenols (HPP).
- The content of antioxidant EPP/EPA/NEPP/NEPA/HPP: the data are entered as documented in the original publication, e.g., milligrams of EPP/NEPP/EPA/NEPA/HPP per kilogram of food in fresh or dry weight;
- Antioxidant activity properties evaluated by 2,2 -azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS), 2,2-Diphenyl-1-picrylhydrazyl radical (DPPH), Ferric Reducing Antioxidant Power (FRAP), Oxygen Radical Absorbance Capacity (ORAC) and Folin-Ciocalteu assays: since units will vary for the contribution of EPP/EPA/NEPP/NEPA/HPP, it was essential to add new units to eBASIS. Examples include: ABTS assay data reported as mM trolox per kilogram of food, FRAP assay expressed as mM Fe2SO4 per kilogram of food, and ORAC assay data expressed as mM trolox per kilogram of food.
2.3. Search Criteria and Suitable Papers for Data Entry
3. Results and Discussion
3.1. Test Input Systems for Data Entry
3.2. Producing eBASIS Reports
3.3. Data on Extractable and/or Non-Extractable Compounds, from Peer-Reviewed Literature
4. Discussion
4.1. Data on Extractable and/or Non-Extractable Compounds Exploitation
4.2. Bioactive Composition Data Aggregation for Dietary Assessment
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Durazzo, A.; Lucarini, M. The state of science and innovation of bioactive research and applications, health, and diseases. Front. Nutr. 2019, 6, 178. [Google Scholar] [CrossRef] [PubMed]
- Durazzo, A.; Lucarini, M.; Santini, A. Nutraceuticals in Human Health. Foods. Mar. 2020, 23, 370. [Google Scholar] [CrossRef] [PubMed]
- Daliu, P.; Santini, A.; Novellino, E. A decade of nutraceutical patents: Where are we now in 2018? Expert Opin. Ther. Pat. 2018, 28, 875–882. [Google Scholar] [CrossRef] [PubMed]
- Santini, A.; Tenore, G.C.; Novellino, E. Nutraceuticals: A paradigm of proactive medicine. Eur. J. Pharm. Sci. 2017, 96, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Durazzo, A.; D’Addezio, L.; Camilli, E.; Piccinelli, R.; Turrini, A.; Marletta, L.; Marconi, S.; Lucarini, M.; Lisciani, S.; Gabrielli, P.; et al. From Plant Compounds to Botanicals and Back: A Current Snapshot. Molecules 2018, 23, 1844. [Google Scholar] [CrossRef]
- Santini, A.; Novellino, E. Nutraceuticals: Shedding light on the grey area between pharmaceuticals and food. Expert Rev. Clin. Pharmacol. 2018, 11, 545–547. [Google Scholar] [CrossRef]
- Daliu, P.; Santini, A.; Novellino, E. From pharmaceuticals to nutraceuticals: Bridging disease prevention and management. Expert Rev. Clin. Pharmacol. 2019, 18, 1–7. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M.; Souto, E.B.; Cicala, C.; Caiazzo, E.; Izzo, A.A.; Novellino, E.; Santini, A. Polyphenols: A concise overview on the chemistry, occurrence and human health. Phytot. Res. 2019, 33, 2221–2243. [Google Scholar] [CrossRef]
- Yeung, A.W.K.; Tzvetkov, N.T.; El-Tawil, O.S.; Bungau, S.G.; Abdel-Daim, M.M.; Atanasov, A.G. Antioxidants: Scientific literature landscape analysis. Oxid. Med. Cell. Longev. 2019, 2019, 8278454. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M. A Current shot and re-thinking of antioxidant research strategy. Braz. J. Anal. Chem. 2018, 5, 9–11. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M. Extractable and non-extractable antioxidants. Molecules 2019, 24, 1933. [Google Scholar] [CrossRef]
- Durazzo, A. Extractable and Non-extractable polyphenols: An overview. In Non-Extractable Polyphenols and Carotenoids: Importance in Human Nutrition and Health; Saura-Calixto, F., Pérez-Jiménez, J., Eds.; Royal Societof Chemistry: London, UK, 2018; pp. 1–37. [Google Scholar]
- Huemmer, W.; Schereier, P. Analysis of proanthocyanidins. Mol. Nutr. Food Res. 2008, 52, 1381–1398. [Google Scholar] [CrossRef]
- Saura-Calixto, F. Concept and health-related properties of nonextractable polyphenols: The missing dietary polyphenols. J. Agric. Food Chem. 2012, 60, 11195–11200. [Google Scholar] [CrossRef]
- Pérez-Jiménez, J.; Diaz-Rubio, M.E.; Saura-Calixto, F. Non-extractable polyphenols, a major dietary antioxidant: Occurrence, metabolic fate and health effects. Nutr. Res. Rev. 2013, 26, 118–129. [Google Scholar] [CrossRef]
- Pérez-Jiménez, J.; Arranz, S.; Tabernero, M.; Diaz-Rubio, M.E.; Serrano, J.; Goni, I.; Saura-Calixto, F. Updated methodology to determine antioxidant capacity in plant foods, oils and beverages: Extraction, measurements and expression of results. Food Res. Int. 2008, 41, 274–285. [Google Scholar]
- Pérez-Jiménez, J.; Torres, J.L. Analysis of non-extractable phenolic compounds in foods: The current state of the art. J. Agric. Food Chem. 2011, 59, 12713–12724. [Google Scholar] [CrossRef]
- Cheng, A.; Han, C.; Fang, X.; Sun, J.; Chen, X.; Wan, F. Extractable and non-extractable polyphenols fromblueberries modulate LPS-induced expression of iNOS and COX-2 in RAW264.7 macrophages via the NF-κBsignalling pathway. J. Sci. Food Agric. 2016, 96, 3393–3400. [Google Scholar] [CrossRef] [PubMed]
- Tow, W.W.; Premier, R.; Jing, H.; Ajlouni, S. Antioxidant and antiproliferation effects of extractable and non-extractable polyphenols isolated from apple waste using different extraction methods. J. Food Sci. 2011, 76, T163–T172. [Google Scholar] [CrossRef]
- Han, Y.; Huang, M.; Li, L.; Cai, X.; Gao, Z.; Li, F.; Rakariyatham, K.; Song, M.; Tomé, S.F.; Xiao, H. Non-extractable Polyphenols from Cranberries: Potential Anti-Inflammation and Anti-Colon-Cancer Agents. Food Funct. 2019, 10, 7714–7723. [Google Scholar] [CrossRef]
- Hamauzu, Y.; Suwannachot, J. Non-extractable polyphenols and in vitro bile acid-binding capacity of dried persimmon (Diospyros kaki) fruit. Food Chem. 2019, 293, 127–133. [Google Scholar] [CrossRef]
- Gonzalez-Sarrías, A.; Espín, J.C.; Tomas-Barberan, F.A. Non-extractable polyphenols produce gut microbiota metabolites that persist in circulation and show anti-inflammatory and free radical scavenging effects. Trends Food Sci. Technol. 2017, 69, 281–288. [Google Scholar]
- Durazzo, A. Study approach of antioxidant properties in foods: Update and considerations. Foods 2017, 6, 17. [Google Scholar] [CrossRef]
- Domínguez-Rodríguez, G.; Marina, M.L.; Plaza, M. Strategies for the extraction and analysis of non-extractable polyphenols from plants. J. Chrom. A 2017, 1514, 1–15. [Google Scholar]
- Arranz, S.; Pérez-Jiménez, J.; Saura-Calixto, F. Antioxidant capacity of walnut (Juglans regia L.): Contribution of oil and defatted matter. Eur. Food Res. Technol. 2008, 227, 425–431. [Google Scholar]
- Ebun, O.; Santosh, K. Effect of domestic cooking on the polyphenolic content and antioxidant capacity of plantain (Musa paradisiaca). World J. Dairy Food Sci. 2011, 6, 189–194. [Google Scholar]
- Durazzo, A.; Turfani, V.; Azzini, E.; Maiani, G.; Carcea, M. Phenols, lignans and antioxidant properties of legume and sweet chestnut flours. Food Chem. 2013, 140, 666–671. [Google Scholar]
- Camelo-Méndez, G.A.; Bello-Pérez, L.A. Antioxidant Capacity of Extractable and Non-extractable Polyphenols of Pigmented Maize. Food Biotechnol. 2014, 4, 6–13. [Google Scholar]
- Durazzo, A.; Turfani, V.; Azzini, E.; Maiani, G.; Carcea, M. Antioxidant properties of experimental pastas made with different wholegrain cereals. J. Food Res. 2014, 3, 33–40. [Google Scholar]
- Durazzo, A.; Turfani, V.; Narducci, V.; Azzini, E.; Maiani, G.; Carcea, M. Nutritional characterisation and bioactive components of commercial carobs flours. Food Chem. 2014, 153, 109–113. [Google Scholar]
- Durazzo, A.; Gabrielli, P.; Manzi, P. Qualitative Study of Functional Groups and Antioxidant Properties of Soy-Based Beverages Compared to Cow Milk. Antioxidants 2015, 4, 523–532. [Google Scholar] [CrossRef]
- Zambrano-Moreno, E.L.; Chávez-Jáuregui, R.N.; de Lurdes, P.M.; Wessel-Beaver, L. Phenolic content and antioxidant capacity in organically and conventionally grown eggplant (Solanum melongena) fruits following thermal processing. Food Sci. Technol. 2015, 35, 414–420. [Google Scholar] [CrossRef]
- Durazzo, A.; Casale, G.; Melini, V.; Maiani, G.; Acquistucci, R. Total polyphenol content and antioxidant properties of Solina (Triticum aestivum L.) and derivatives thereof. J. Food Sci. 2016, 28, 221. [Google Scholar]
- Mattera, M.; Durazzo, A.; Nicoli, S.; Di Costanzo, M.G.; Manzi, P. Chemical, nutritional, physical and antioxidant properties of Pecorino d’abruzzo cheese. Ital. J. Food Sci. 2016, 28, 579–597. [Google Scholar]
- Turfani, V.; Narducci, V.; Durazzo, A.; Galli, V.; Carcea, M. Technological, nutritional and functional properties of wheat bread enriched with lentil or carob flours. LWT–Food Sci. Technol. 2017, 78, 361. [Google Scholar] [CrossRef]
- Durazzo, A.; Lisciani, S.; Camilli, E.; Gabrielli, P.; Marconi, S.; Gambelli, L.; Aguzzi, A.; Lucarini, M.; Maiani, G.; Casale, G.; et al. Nutritional composition and antioxidant properties of traditional Italian dishes. Food Chem. 2017, 218, 70–77. [Google Scholar] [CrossRef]
- Olivas-Aguirre, F.J.; González-Aguilar, G.A.; Velderrain-Rodríguez, G.R.; Torres-Moreno, H.; Robles-Zepeda, R.E.; Vázquez-Flores, A.A.; de la Rosa, L.A.; Wall-Medrano, A. Radical scavenging and anti-proliferative capacity of three freeze-dried tropical fruits. Int. J. Food Sci. Technol. 2017, 52, 1699–1709. [Google Scholar] [CrossRef]
- Peng, H.; Li, W.; Li, H.; Deng, Z.; Zhang, B. Extractable and non-extractable bound phenolic compositions and their antioxidant properties in seed coat and cotyledon of black soybean (Glycinemax (L.) Merr). J. Funct. Foods 2017, 32, 296–312. [Google Scholar] [CrossRef]
- Sanz-Pintos, N.; Pérez-Jiménez, J.; Buschmann, A.H.; Vergara-Salinas, J.R.; Pérez-Correa, J.R.; Saura-Calixto, F. Macromolecular antioxidants and dietary fiber in edibles. J. Food Sci. 2017, 82, 289–295. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Wei, X.Q.; Li, M.Y.; Duan, X.W.; Sun, Y.M.; Yang, R.L.; Su, X.D.; Huang, R.M.; Wang, H. Nutritional composition and antioxidant properties of the fruits of a chinese wild passiflora foetida. Molecules 2018, 23, 459. [Google Scholar] [CrossRef]
- Anokwuru, C.; Sigidi, M.; Boukandou, M.; Tshisikhawe, P.; Traore, A.; Potgieter, N. Antioxidant activity and spectroscopic characteristics of extractable and non-extractable phenolics from Terminalia sericea Burch. ex DC. Molecules 2018, 23, 1303. [Google Scholar] [CrossRef]
- Li, Q.; Yang, S.; Li, Y.; Xue, X.; Huang, Y.; Luo, H.; Zhang, Y.; Lu, Z. Comparative evaluation of soluble and insoluble-bound phenolics and antioxidant activity of two Chinese mistletoes. Molecules 2018, 23, 359. [Google Scholar] [CrossRef]
- Esparza-Martínez, F.J.; Miranda-López, R.; Mata-Sánchez, S.M.; Guzmán-Maldonado, S.H. Extractable and non-extractable phenolics and antioxidant capacity of mandarin waste dried at different temperatures. Plant Foods Human Nutr. 2016, 71, 294–300. [Google Scholar] [CrossRef]
- Toh, P.Y.; Leong, F.S.; Chang, S.K.; Khoo, H.E.; Yim, H.S. Optimization of extraction parameters on the antioxidant properties of banana waste. Acta Sci. Pol. Technol. Aliment. 2016, 15, 65–78. [Google Scholar] [CrossRef]
- Lucarini, M.; Durazzo, A.; Romani, A.; Campo, M.; Lombardi-Boccia, G.; Cecchini, F. Bio-based compounds from grape seeds: A biorefinery approach. Molecules 2018, 23, 1888. [Google Scholar] [CrossRef] [PubMed]
- Mushtaq, M.; Sultana, B.; Akram, S.; Anwar, F.; Adnan, A.; Rizvi, S.S.H. Enzyme-assisted supercritical fluid extraction: An alternative and green technology for non-extractable polyphenols. Anal. Bioanal. Chem. 2017, 409, 3645–3655. [Google Scholar] [CrossRef] [PubMed]
- Lucarini, M.; Durazzo, A.; Kiefer, J.; Santini, A.; Lombardi-Boccia, G.; Souto, E.B.; Romani, A.; Lampe, A.; Ferrari Nicoli, S.; Gabrielli, P.; et al. Grape Seeds: Chromatographic profile of fatty acids and phenolic compounds and qualitative analysis by FTIR-ATR spectroscopy. Foods 2019, 9, 10. [Google Scholar] [CrossRef]
- Saura-Calixto, F.; Serrano, J.; Goni, I. Intake and Bioaccessibility of Total Polyphenols in a Whole Diet. Food Chem. 2007, 101, 492–501. [Google Scholar] [CrossRef]
- Faller, A.L.K.; Fialho, E.; Liu, R.H. Cellular Antioxidant Activity of Feijoada Whole Meal Coupled with an in Vitro Digestion. J. Agric. Food. Chem. 2012, 60, 4826–4832. [Google Scholar] [CrossRef]
- Koehnlein, E.A.; Bracht, A.; Nishida, V.S.; Peralta, R.M. Total antioxidant capacity and phenolic content of the Brazilian diet: A real scenario. Int. J. Food Sci. Nutr. 2014, 65, 293–298. [Google Scholar] [CrossRef]
- Pérez-Jiménez, J.; Saura-Calixto, F. Macromolecular antioxidants or non-extractable polyphenols in fruit and vegetables: Intake in four European countries. Food Res. Int. 2015, 74, 315–323. [Google Scholar] [CrossRef]
- Koehnlein, E.A.; Koehnlein, É.M.; Corrêa, R.C.; Nishida, V.S.; Correa, V.G.; Bracht, A.; Peralta, R.M. Analysis of a whole diet in terms of phenolic content and antioxidant capacity: Effects of a simulated gastrointestinal digestion. Int. J. Food Sci. Nutr. 2016, 67, 614–623. [Google Scholar] [CrossRef] [PubMed]
- Pinto, P.; Santo, C.N. Worldwide (poly) phenol intake: Assessment methods and identified gaps. Eur. J. Nutr. 2017, 56, 1393–1408. [Google Scholar] [CrossRef]
- Luthria, D.L. Significance of sample preparation in developing analytical methodologies for accurate estimation of bioactive compounds in functional foods. J. Sci. Food Agric. 2006, 86, 2266–2272. [Google Scholar] [CrossRef]
- Tabart, J.; Kevers, C.; Pincemail, J.; Defraigne, J.O.; Dommes, J. Comparative antioxidant capacities of phenolic compounds measured by various tests. Food Chem. 2009, 113, 1226–1233. [Google Scholar] [CrossRef]
- Apak, R.; Özyürek, M.; Güçlü, K.; Çapanoglu, E. Antioxidant activity/capacity measurement. 1. Classification, physicochemical principles, mechanisms, and electron transfer (ET)-based assays. J. Agric. Food Chem. 2016, 64, 997–1027. [Google Scholar] [CrossRef]
- Apak, R.; Özyürek, M.; Güçlü, K.; Çapanoglu, E. Antioxidant Activity/Capacity Measurement. 2. Hydrogen Atom Transfer (HAT)-Based, Mixed-Mode (Electron Transfer (ET)/HAT), and Lipid Peroxidation Assays. J. Agric. Food Chem. 2016, 64, 1028–1045. [Google Scholar] [CrossRef]
- Apak, A.; Capanoglu, E.; Shahidi, F. Measurement of Antioxidant Activity and Capacity: Recent Trends and Applications; Wiley: New York, NY, USA, 2018; ISBN 978-1-119-13535-7. [Google Scholar]
- Liyana-Pathirana, C.M.; Shahidi, F. Antioxidant properties of commercial soft and hard winter wheat (Triticum aestivum L.) and their milling fractions. J. Sci. Food Agric. 2006, 86, 477–485. [Google Scholar] [CrossRef]
- Celik, S.E.; Ozyürek, M.; Güçlü, K.; Apak, R. Solvent effects on the antioxidant capacity of lipophilic and hydrophilic antioxidants measured by CUPRAC, ABTS/persulphate and FRAP methods. Talanta 2010, 81, 1300–1309. [Google Scholar] [CrossRef]
- Verma, B.; Hucl, P.; Chibbar, R.N. Phenolic acid composition and antioxidant capacity of acid and alkalihydrolysed wheat bran fractions. Food Chem. 2009, 116, 947–954. [Google Scholar] [CrossRef]
- Delgrado-Andrade, C.; Conde-Aguilera, J.A.; Haro, A.; de la Cueva, S.P.; Rufian-Henares, J.A. A combined procedure to evaluate the global antioxidant response of bread. J. Cereal Sci. 2010, 52, 239–246. [Google Scholar] [CrossRef]
- White, B.L.; Howard, L.R.; Prior, R.L. Release of bound procyanidins from cranberry pomace by alkaline hydrolysis. J. Agric. Food Chem. 2010, 58, 7572–7579. [Google Scholar] [CrossRef]
- Navarro-Gonzalez, I.; García-Valverde, V.; García-Alonso, J.; Periago, M.J. Chemical profile, functional and antioxidant properties of tomato peel fiber. Food Res. Int. 2011, 44, 1528–1535. [Google Scholar] [CrossRef]
- Royer, M.; Diouf, P.N.; Stevanovic, T. Polyphenol contents and radical scavenging capacities of red maple (Acer rubrum L.) extracts. Food Chem. Toxicol. 2011, 49, 2180–2218. [Google Scholar] [CrossRef]
- Prior, R.L.; Wu, X.; Schaich, K. Standardized methods for the determination of antioxidant capacity and phenolics in foods and dietary supplements. J. Agric. Food Chem. 2005, 53, 4290–4302. [Google Scholar] [CrossRef]
- Leopoldini, M.; Russo, N.; Toscano, M. The molecular basis of working mechanism of natural polyphenolic antioxidants. Food Chem. 2011, 125, 288–306. [Google Scholar] [CrossRef]
- Schlesier, K.; Harwat, M.; Bohm, V.; Bitsch, R. Assessment of antioxidant activity by using different in vitro methods. Free Radic. Res. 2002, 36, 177–187. [Google Scholar] [CrossRef]
- Carlsen, H.; Halvorsen, B.L.; Holte, K.; Bøhn, S.K.; Dragland, S.; Sampson, L.; Willey, C.; Senoo, H.; Umezono, Y.; Sanada, C.; et al. The total antioxidant content of more than 3100 foods, beverages, spices, herbs and supplements used worldwide. J. Nutr. 2010, 9, 3. [Google Scholar] [CrossRef] [PubMed]
- Haytowitz, D.B.; Bhagwat, S. USDA Database for the Oxygen Radical Absorbance Capacity (ORAC) of Selected Foods, Release Nutrient Data Laboratory Home Page; U.S. Department of Agriculture, Agricultural Research Service: Washington, DC, USA, 2010. Available online: http://www.orac-info-portal.de/download/ORAC_R2.pdf (accessed on 5 November 2020).
- eBASIS—Bioactive Substances in Food Information System. Available online: http://ebasis.eurofir.org/Default.asp (accessed on 8 July 2019).
- Gry, J.; Black, L.; Eriksen, F.D.; Pilegaard, K.; Plumb, J.; Rhodes, M.; Sheehan, D.; Kiely, M.; Kroon, P.A. EuroFIR-BASIS—A combined composition and biological activity database for bioactive compounds in plant-based foods. Trends Food Sci. Technol. 2007, 18, 434–444. [Google Scholar] [CrossRef]
- Kiely, M.; Black, L.J.; Plumb, J.; Kroon, P.A.; Hollman, P.C.; Larsen, J.C.; Speijers, G.J.; Kapsokefalou, M.; Sheehan, D.; Gry, J.; et al. EuroFIR eBASIS: Application for health claims submissions and evaluations. Eur. J. Clin. Nutr. 2010, 64, S101–S107. [Google Scholar] [CrossRef]
- Plumb, J.; Pigat, S.; Bompola, F.; Cushen, M.; Pinchen, H.; Nørby, E.; Astley, S.; Lyons, J.; Kiely, M.; Finglas, P. eBASIS (Bioactive Substances in Food Information Systems) and bioactive intakes: Major updates of the bioactive compound composition and beneficial bio effects database and the development of a probabilistic model to assess intakes in Europe. Nutrients 2017, 9, 320. [Google Scholar] [CrossRef]
- Plumb, J.; Lyons, J.; Nørby, K.; Thomas, M.; Nørby, E.; Poms, R.; Bucchini, L.; Restani, P.; Kiely, M.; Finglas, P. ePlantLIBRA: A composition and biological activity database for bioactive compounds in plant food supplements. Food Chem. 2016, 193, 121–127. [Google Scholar]
- Peterson, J.J.; Dwyer, J.T.; Jacques, P.F.; McCullough, M.L. Improving estimation of flavonoid intake for study of health outcomes. Nutr. Rev. 2015, 73, 553–576. [Google Scholar]
- Bonaccio, M.; Pounis, G.; Cerletti, C.; Donati, M.B.; Iacoviello, L.; de Gaetano, G. On behalf of the MOLI-SANI study investigators. Mediterranean diet, dietary polyphenols and low grade inflammation: Results from the MOLI-SANI study. Br. J. Clin. Pharmacol. 2017, 83, 107–113. [Google Scholar]
- Pigat, S.; Bompola, F.; Cushen, M.; Plumb, J.; Finglas, F. Combining National Food Consumption Survey Data with a Bioactive Database in a Probabilistic Intake Model to Assess Bioactive Intakes in Europe—A Tool Resulting from the European BACCHUS Project. FASEB J. 2017, 31, 648. [Google Scholar]
- Southan, C.; Várkonyi, P.; Muresan, S. Quantitative assessment of the expanding complementarity between public and commercial databases of bioactive compounds. J. Cheminform. 2009, 1, 10. [Google Scholar] [PubMed]
- Hersey, A.; Chambers, J.; Bellis, L.; Bento, A.P.; Gaulton, A.; Overington, J.P. Chemical databases: Curation or integration by user-defined equivalence? Drug Disc. Tod. Technol. 2015, 14, 17–24. [Google Scholar]
- Barabási, A.; Menichetti, G.; Loscalzo, J. The unmapped chemical complexity of our diet. Nat. Food 2020, 1, 33–37. [Google Scholar]
- FNS—Cloud, Food Nutrition Security. Available online: https://www.fns-cloud.eu/ (accessed on 19 September 2020).
- Traka, M.H.; Plumb, J.; Berry, R.; Pinchen, H.; Finglas, P. Maintaining and updating food composition datasets for multiple users and novel technologies: Current challenges from a UK perspective. Nutr. Bull. 2020, 45, 230–240. [Google Scholar]
- Finglas, P.M.; Berry, R.; Astley, S. Assessing and improving the quality of food composition databases for nutrition and health applications in Europe: The contribution of EuroFIR. Adv. Nutr. 2014, 5, 608s–614s. [Google Scholar]


| Reference | Food | Processing |
| Author Title Journal Year, Volume Summary of paper contents | Plant, part, cultivar, maturity, country of origin, region, season, growing conditions, EuroFIR classification, generic food name, plant description | If food is processed, these fields are included state, heat treatment, cooking method, treatment applied, preservation method |
| Sampling | Composition | Quality |
| Sampling year, primary sample unit size, primary sample units, analytical sample size, portion replicates, sample plan, sample handling | Compound class/activity, compound, standard source, analytical method name, analytical method, level, min, max, standard deviation, unit, extraction method | Plant description, processing defined, sample plan, sample handling, compound identification, analytical method, analytical performance |
| Data Points | Number of Food Plants | References | |
|---|---|---|---|
| EPP | 187 | 44 | 23 |
| EPA | 11 | 3 | 2 |
| NEPP | 36 | 5 | 5 |
| NEPA | 88 | 27 | 10 |
| HPP | 115 | 48 | 15 |
| Combined total | 437 | 63 | 27 |
| eBASIS total | 44,300 | 271 | 1300 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Plumb, J.; Durazzo, A.; Lucarini, M.; Camilli, E.; Turrini, A.; Marletta, L.; Finglas, P. Extractable and Non-Extractable Antioxidants Composition in the eBASIS Database: A Key Tool for Dietary Assessment in Human Health and Disease Research. Nutrients 2020, 12, 3405. https://doi.org/10.3390/nu12113405
Plumb J, Durazzo A, Lucarini M, Camilli E, Turrini A, Marletta L, Finglas P. Extractable and Non-Extractable Antioxidants Composition in the eBASIS Database: A Key Tool for Dietary Assessment in Human Health and Disease Research. Nutrients. 2020; 12(11):3405. https://doi.org/10.3390/nu12113405
Chicago/Turabian StylePlumb, Jenny, Alessandra Durazzo, Massimo Lucarini, Emanuela Camilli, Aida Turrini, Luisa Marletta, and Paul Finglas. 2020. "Extractable and Non-Extractable Antioxidants Composition in the eBASIS Database: A Key Tool for Dietary Assessment in Human Health and Disease Research" Nutrients 12, no. 11: 3405. https://doi.org/10.3390/nu12113405
APA StylePlumb, J., Durazzo, A., Lucarini, M., Camilli, E., Turrini, A., Marletta, L., & Finglas, P. (2020). Extractable and Non-Extractable Antioxidants Composition in the eBASIS Database: A Key Tool for Dietary Assessment in Human Health and Disease Research. Nutrients, 12(11), 3405. https://doi.org/10.3390/nu12113405








