Mildly Pasteurized Whey Protein Promotes Gut Tolerance in Immature Piglets Compared with Extensively Heated Whey Protein
Abstract
1. Introduction
2. Materials and Methods
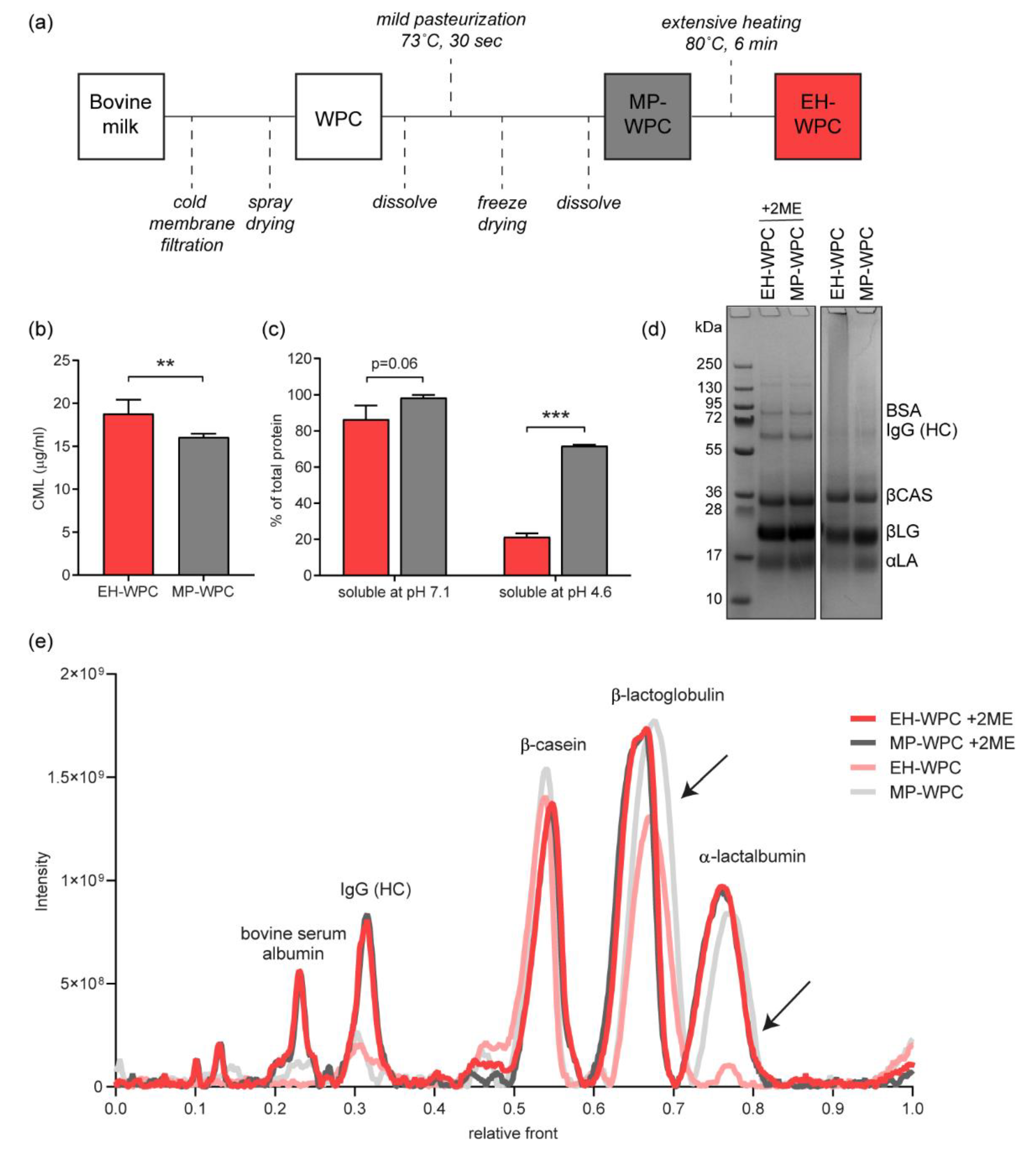
2.1. Whey Protein Concentrate (WPC)
2.2. Protein Profile Analysis
2.3. Piglet Study
- (1)
- EH-WPC group.
- (2)
- MP-WPC group.
2.4. Feeding Tolerance in Piglets
2.5. Galactose, Lactose and Lactulose/Mannitol Test
2.6. Gastro-Intestinal (GI) Transit by X-ray Analysis
2.7. Sample Collection
2.8. Analysis of Digesta
2.9. Enzyme Activity Assay
2.10. Bacterial Staining
2.11. In Vitro Digestion
2.12. Statistical Analysis
3. Results
3.1. Characterization of WPCs: Impact of Thermal Processing on Whey Protein Nativity, Carboxymethyllysine (CML) Formation and Protein Aggregation
3.2. Piglet Study: Clinical Symptoms and Adverse Events
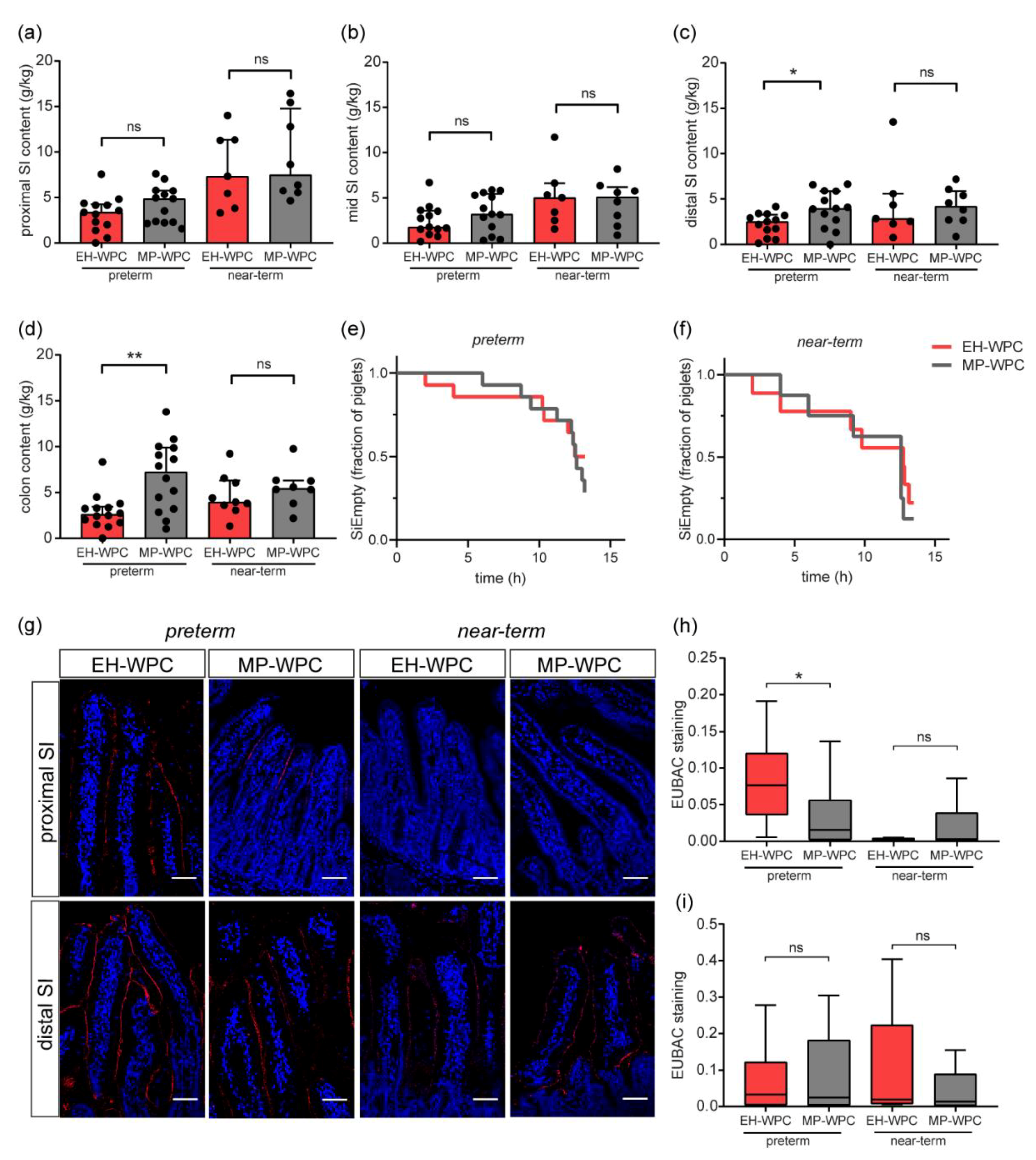
3.3. Piglet Study: GI Tolerance
3.4. Piglet Study: Digestive and Absorptive Capacity and Intestinal Permeability
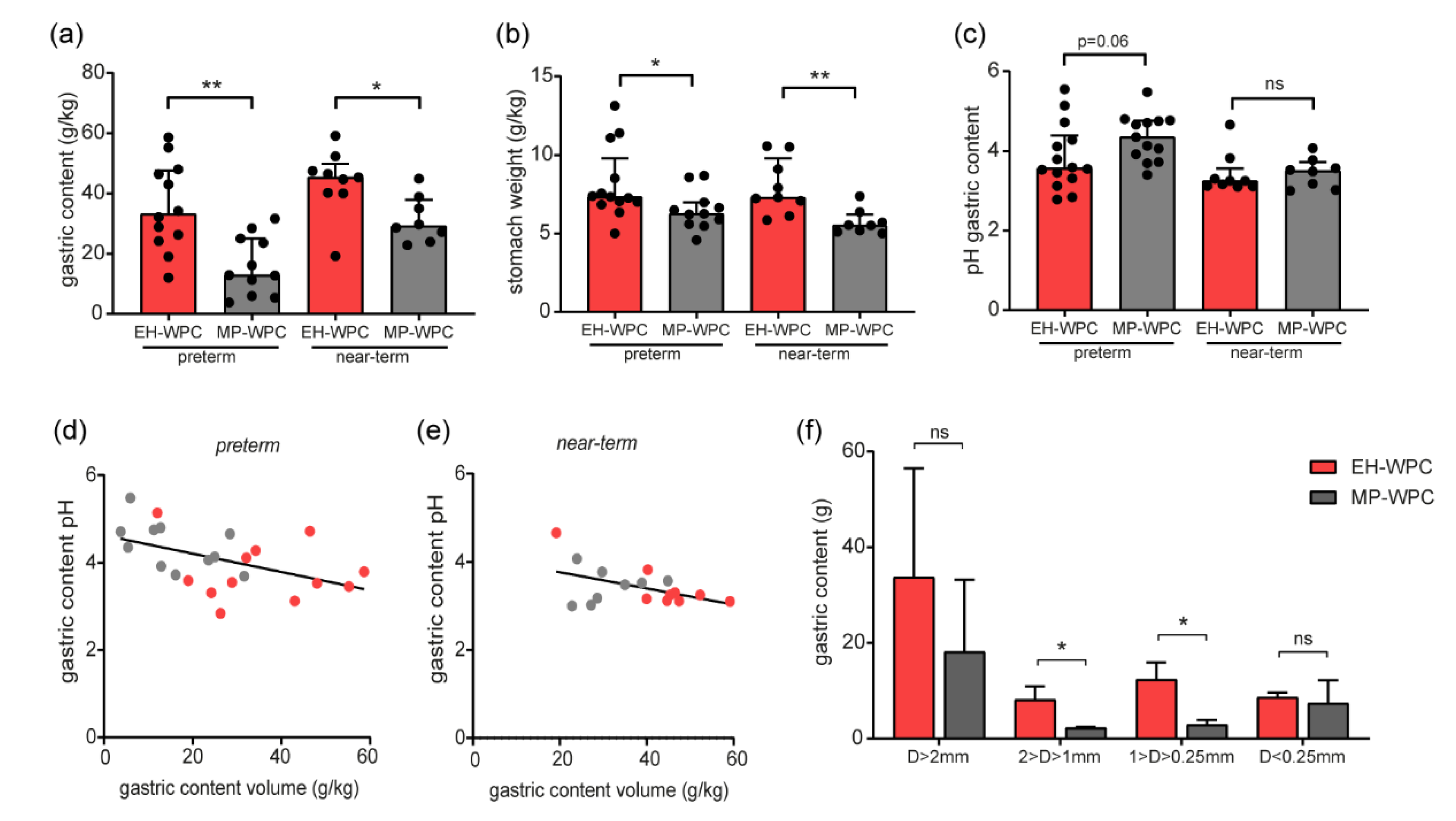
3.5. Gastric Content: pH and Formation of Coagulates In Vivo
3.6. GI Transit and Mucosal Bacterial Adherence
3.7. In Vitro Digestion and Coagulation
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| WPC | whey protein concentrate |
| MP-WPC | mildly pasteurized WPC |
| EH-WPC | extensively heated WPC |
| EN | enteral nutrition |
| PN | parenteral nutrition |
| SI | small intestine |
| 2ME | 2-mercaptoethanol |
| ALP | alkaline phosphatase |
| BSA | bovine serum albumin |
| BCA | bicinchoninic acid |
| IgG (HC) | immunoglobulin G (heavy chain) |
| βCAS | β-casein |
| βLG | β-lactoglobulin |
| αLA | α-lactalbumin |
| CML | Carboxymethyllysine |
| SIM model | Semi-dynamic digestion model |
References
- Horta, B.L.; Bahl, R.; Martinés, J.C.; Victora, C.G. Evidence on the Long-Term Effects of Breastfeeding: Systematic Review and Meta-Analyses; World Health Organization: Geneva, Switzerland, 2007.
- Reisinger, K.W.; de Vaan, L.; Kramer, B.W.; Wolfs, T.G.; van Heurn, L.W.; Derikx, J.P. Breast-feeding improves gut maturation compared with formula feeding in preterm babies. J. Pediatric Gastroenterol. Nutr. 2014, 59, 720–724. [Google Scholar] [CrossRef]
- Eidelman, A.I.; Schanler, R.J. Breastfeeding and the use of human milk. Pediatrics 2012, 115, 496–506. [Google Scholar]
- Victora, C.G.; Bahl, R.; Barros, A.J.; Franca, G.V.; Horton, S.; Krasevec, J.; Murch, S.; Sankar, M.J.; Walker, N.; Rollins, N.C. Breastfeeding in the 21st century: Epidemiology, mechanisms, and lifelong effect. Lancet 2016, 387, 475–490. [Google Scholar] [CrossRef]
- Madureira, A.R.; Pereira, C.I.; Gomes, A.M.; Pintado, M.E.; Malcata, F.X. Bovine whey proteins–Overview on their main biological properties. Food Res. Int. 2007, 40, 1197–1211. [Google Scholar] [CrossRef]
- Haschke, F.; Haiden, N.; Thakkar, S.K. Nutritive and Bioactive Proteins in Breastmilk. Ann. Nutr. Metab. 2016, 69 (Suppl. S2), 17–26. [Google Scholar] [CrossRef]
- Gila-Diaz, A.; Arribas, S.M.; Algara, A.; Martin-Cabrejas, M.A.; Lopez de Pablo, A.L.; Saenz de Pipaon, M.; Ramiro-Cortijo, D. A Review of Bioactive Factors in Human Breastmilk: A Focus on Prematurity. Nutrients 2019, 11, 1307. [Google Scholar] [CrossRef]
- Joyce, A.M.; Brodkorb, A.; Kelly, A.L.; O’Mahony, J.A. Separation of the effects of denaturation and aggregation on whey-casein protein interactions during the manufacture of a model infant formula. Dairy Sci. Technol. 2017, 96, 787–806. [Google Scholar] [CrossRef]
- De Wit, J.N. Nutritional and functional characteristics of whey proteins in food products. J. Dairy Sci. 1998, 81, 597–608. [Google Scholar] [CrossRef]
- Lonnerdal, B. Nutritional and physiologic significance of human milk proteins. Am. J. Clin. Nutr. 2003, 77, 1537s–1543s. [Google Scholar] [CrossRef] [PubMed]
- Martin, C.R.; Ling, P.R.; Blackburn, G.L. Review of Infant Feeding: Key Features of Breast Milk and Infant Formula. Nutrients 2016, 8, 279. [Google Scholar] [CrossRef] [PubMed]
- Alarcon, P.A.; Tressler, R.L.; Mulvaney, A.; Lam, W.; Comer, G.M. Gastrointestinal tolerance of a new infant milk formula in healthy babies: An international study conducted in 17 countries. Nutrition 2002, 18, 484–489. [Google Scholar] [CrossRef]
- Fanaro, S. Feeding intolerance in the preterm infant. Early Hum. Dev. 2013, 89 (Suppl. 2), S13–S20. [Google Scholar] [CrossRef]
- Van Lieshout, G.A.A.; Lambers, T.T.; Bragt, M.C.E.; Hettinga, K.A. How processing may affect milk protein digestion and overall physiological outcomes: A systematic review. Crit. Rev. Food Sci. Nutr. 2019, 1–24. [Google Scholar] [CrossRef]
- Pischetsrieder, M.; Henle, T. Glycation products in infant formulas: Chemical, analytical and physiological aspects. Amino Acids 2012, 42, 1111–1118. [Google Scholar] [CrossRef]
- McCarthy, N.A.; Wijayanti, H.B.; Crowley, S.V.; O’Mahony, J.A.; Fenelon, M.A. Pilot-scale ceramic membrane filtration of skim milk for the production of a protein base ingredient for use in infant milk formula. Int. Dairy J. 2017, 73, 57–62. [Google Scholar] [CrossRef]
- Miller, E.R.; Ullrey, D.E. The pig as a model for human nutrition. Annu. Rev. Nutr. 1987, 7, 361–382. [Google Scholar] [CrossRef]
- Alizadeh, A.; Akbari, P.; Difilippo, E.; Schols, H.A.; Ulfman, L.H.; Schoterman, M.H.; Garssen, J.; Fink-Gremmels, J.; Braber, S. The piglet as a model for studying dietary components in infant diets: Effects of galacto-oligosaccharides on intestinal functions. Br. J. Nutr. 2016, 115, 605–618. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Nguyen, D.N.; Obelitz-Ryom, K.; Andersen, A.D.; Thymann, T.; Chatterton, D.E.W.; Purup, S.; Heckmann, A.B.; Bering, S.B.; Sangild, P.T. Bioactive Whey Protein Concentrate and Lactose Stimulate Gut Function in Formula-fed Preterm Pigs. J. Pediatric Gastroenterol. Nutr. 2018, 66, 128–134. [Google Scholar] [CrossRef]
- Rasmussen, S.O.; Martin, L.; Ostergaard, M.V.; Rudloff, S.; Li, Y.; Roggenbuck, M.; Bering, S.B.; Sangild, P.T. Bovine colostrum improves neonatal growth, digestive function, and gut immunity relative to donor human milk and infant formula in preterm pigs. Am. J. Physiol. Gastrointest. Liver Physiol. 2016, 311, G480–G491. [Google Scholar] [CrossRef]
- Kappel, S.S.; Sangild, P.T.; Hilsted, L.; Hartmann, B.; Thymann, T.; Aunsholt, L. Gastric Residual to Predict Necrotizing Enterocolitis in Preterm Piglets as Models for Infants. JPEN J. Parenter. Enter. Nutr. 2020. [Google Scholar] [CrossRef]
- Ren, S.; Hui, Y.; Obelitz-Ryom, K.; Brandt, A.B.; Kot, W.; Nielsen, D.S.; Thymann, T.; Sangild, P.T.; Nguyen, D.N. Neonatal gut and immune maturation is determined more by postnatal age than by postconceptional age in moderately preterm pigs. Am. J. Physiol. Gastrointest. Liver Physiol. 2018, 315, G855–G867. [Google Scholar] [CrossRef] [PubMed]
- Delahaije, R.J.; Gruppen, H.; van Eijk-van Boxtel, E.L.; Cornacchia, L.; Wierenga, P.A. Controlling the Ratio between Native-Like, Non-Native-Like, and Aggregated beta-Lactoglobulin after Heat Treatment. J. Agric. Food Chem. 2016, 64, 4362–4370. [Google Scholar] [CrossRef]
- Chibnall, A.C.; Rees, M.W.; Williams, E.F. The total nitrogen content of egg albumin and other proteins. Biochem. J. 1943, 37, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Welch-Jernigan, R.J.; Abrahamse, E.; Stoll, B.; Smith, O.; Wierenga, P.A.; van de Heijning, B.J.M.; Renes, I.B.; Burrin, D.G. Postprandial Amino Acid Kinetics of Milk Protein Mixtures are Affected by Composition, But Not Denaturation, in Neonatal Piglets. Curr. Dev. Nutr. 2019, 3, nzy102. [Google Scholar] [CrossRef]
- Jovanovic, S.; Barac, M.; Macej, O.; Vucic, T.; Lacnjevac, C. SDS-PAGE analysis of soluble proteins in reconstituted milk exposed to different heat treatments. Sensors 2007, 7, 371–383. [Google Scholar] [CrossRef]
- Smith, P.K.; Krohn, R.I.; Hermanson, G.T.; Mallia, A.K.; Gartner, F.H.; Provenzano, M.D.; Fujimoto, E.K.; Goeke, N.M.; Olson, B.J.; Klenk, D.C. Measurement of protein using bicinchoninic acid. Anal. Biochem. 1985, 150, 76–85. [Google Scholar] [CrossRef]
- Navis, M.; Muncan, V.; Sangild, P.T.; Moller Willumsen, L.; Koelink, P.J.; Wildenberg, M.E.; Abrahamse, E.; Thymann, T.; van Elburg, R.M.; Renes, I.B. Beneficial Effect of Mildly Pasteurized Whey Protein on Intestinal Integrity and Innate Defense in Preterm and Near-Term Piglets. Nutrients 2020, 12, 1125. [Google Scholar] [CrossRef]
- Sangild, P.T.; Thymann, T.; Schmidt, M.; Stoll, B.; Burrin, D.G.; Buddington, R.K. Invited review: The preterm pig as a model in pediatric gastroenterology. J. Anim. Sci. 2013, 91, 4713–4729. [Google Scholar] [CrossRef]
- Cilieborg, M.S.; Boye, M.; Thymann, T.; Jensen, B.B.; Sangild, P.T. Diet-dependent effects of minimal enteral nutrition on intestinal function and necrotizing enterocolitis in preterm pigs. JPEN. J. Parenter. Enter. Nutr. 2011, 35, 32–42. [Google Scholar] [CrossRef]
- Thymann, T.; Burrin, D.G.; Tappenden, K.A.; Bjornvad, C.R.; Jensen, S.K.; Sangild, P.T. Formula-feeding reduces lactose digestive capacity in neonatal pigs. Br. J. Nutr. 2006, 95, 1075–1081. [Google Scholar] [CrossRef]
- Chen, W.; Sun, J.; Kappel, S.S.; Gormsen, M.; Sangild, P.T.; Aunsholt, L. Gut transit time, using radiological contrast imaging, to predict early signs of necrotizing enterocolitis. Pediatric Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Sangild, P.T.; Sjostrom, H.; Noren, O.; Fowden, A.L.; Silver, M. The prenatal development and glucocorticoid control of brush-border hydrolases in the pig small intestine. Pediatric Res. 1995, 37, 207–212. [Google Scholar] [CrossRef]
- Navis, M.; Martins Garcia, T.; Renes, I.B.; Vermeulen, J.L.; Meisner, S.; Wildenberg, M.E.; van den Brink, G.R.; van Elburg, R.M.; Muncan, V. Mouse fetal intestinal organoids: New model to study epithelial maturation from suckling to weaning. EMBO Rep. 2019, 20. [Google Scholar] [CrossRef]
- Van den Braak, C.C.; Klebach, M.; Abrahamse, E.; Minor, M.; Hofman, Z.; Knol, J.; Ludwig, T. A novel protein mixture containing vegetable proteins renders enteral nutrition products non-coagulating after in vitro gastric digestion. Clin. Nutr. 2013, 32, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Bourlieu, C.; Ménard, O.; Bouzerzour, K.; Mandalari, G.; Macierzanka, A.; Mackie, A.R.; Dupont, D. Specificity of infant digestive conditions: Some clues for developing relevant in vitro models. Crit. Rev. Food Sci. Nutr. 2014, 54, 1427–1457. [Google Scholar] [CrossRef]
- Havea, P.; Singh, H.; Creamer, L.K. Characterization of heat-induced aggregates of beta-lactoglobulin, alpha-lactalbumin and bovine serum albumin in a whey protein concentrate environment. J. Dairy Res. 2001, 68, 483–497. [Google Scholar] [CrossRef]
- Butler, J.E. Bovine immunoglobulins: A review. J. Dairy Sci. 1969, 52, 1895–1909. [Google Scholar] [CrossRef]
- Abiramalatha, T.; Thanigainathan, S.; Ninan, B. Routine monitoring of gastric residual for prevention of necrotising enterocolitis in preterm infants. Cochrane Database Syst. Rev. 2019, 7, Cd012937. [Google Scholar] [CrossRef] [PubMed]
- Shen, R.L.; Thymann, T.; Ostergaard, M.V.; Stoy, A.C.; Krych, L.; Nielsen, D.S.; Lauridsen, C.; Hartmann, B.; Holst, J.J.; Burrin, D.G.; et al. Early gradual feeding with bovine colostrum improves gut function and NEC resistance relative to infant formula in preterm pigs. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 309, G310–G323. [Google Scholar] [CrossRef]
- Everaert, N.; Van Cruchten, S.; Weström, B.; Bailey, M.; Van Ginneken, C.; Thymann, T.; Pieper, R. A review on early gut maturation and colonization in pigs, including biological and dietary factors affecting gut homeostasis. Anim. Feed Sci. Technol. 2017, 233, 89–103. [Google Scholar] [CrossRef]
- Wijayanti, H.B.; Bansal, N.; Deeth, H.C. Stability of whey proteins during thermal processing: A review. Compr. Rev. Food Sci. Food Saf. 2014, 13, 1235–1251. [Google Scholar] [CrossRef]
- Boirie, Y.; Dangin, M.; Gachon, P.; Vasson, M.P.; Maubois, J.L.; Beaufrère, B. Slow and fast dietary proteins differently modulate postprandial protein accretion. Proc. Natl. Acad. Sci. USA 1997, 94, 14930–14935. [Google Scholar] [CrossRef]
- Tari, N.R.; Fan, M.Z.; Archbold, T.; Kristo, E.; Guri, A.; Arranz, E.; Corredig, M. Effect of milk protein composition of a model infant formula on the physicochemical properties of in vivo gastric digestates. J. Dairy Sci. 2018, 101, 2851–2861. [Google Scholar] [CrossRef]
- Grotzinger, U.; Bergegardh, S.; Olbe, L. Effect of fundic distension on gastric acid secretion in man. Gut 1977, 18, 105–110. [Google Scholar] [CrossRef]
- Tunick, M.H.; Ren, D.X.; Van Hekken, D.L.; Bonnaillie, L.; Paul, M.; Kwoczak, R.; Tomasula, P.M. Effect of heat and homogenization on in vitro digestion of milk. J. Dairy Sci. 2016, 99, 4124–4139. [Google Scholar] [CrossRef] [PubMed]
- Thymann, T.; Moller, H.K.; Stoll, B.; Stoy, A.C.; Buddington, R.K.; Bering, S.B.; Jensen, B.B.; Olutoye, O.O.; Siggers, R.H.; Molbak, L.; et al. Carbohydrate maldigestion induces necrotizing enterocolitis in preterm pigs. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 297, G1115–G1125. [Google Scholar] [CrossRef]
- Call, L.; Stoll, B.; Oosterloo, B.; Ajami, N.; Sheikh, F.; Wittke, A.; Waworuntu, R.; Berg, B.; Petrosino, J.; Olutoye, O.; et al. Metabolomic signatures distinguish the impact of formula carbohydrates on disease outcome in a preterm piglet model of NEC. Microbiome 2018, 6, 111. [Google Scholar] [CrossRef] [PubMed]
- Windey, K.; De Preter, V.; Verbeke, K. Relevance of protein fermentation to gut health. Mol. Nutr. Food Res. 2012, 56, 184–196. [Google Scholar] [CrossRef]
- Van Haver, E.R.; Sangild, P.T.; Oste, M.; Siggers, J.L.; Weyns, A.L.; Van Ginneken, C.J. Diet-dependent mucosal colonization and interleukin-1beta responses in preterm pigs susceptible to necrotizing enterocolitis. J. Pediatric Gastroenterol. Nutr. 2009, 49, 90–98. [Google Scholar] [CrossRef]
- Grzeskowiak, L.; Martinez-Vallespin, B.; Dadi, T.H.; Radloff, J.; Amasheh, S.; Heinsen, F.A.; Franke, A.; Reinert, K.; Vahjen, W.; Zentek, J.; et al. Formula Feeding Predisposes Neonatal Piglets to Clostridium difficile Gut Infection. J. Infect. Dis. 2018, 217, 1442–1452. [Google Scholar] [CrossRef]


| % of Total Protein | EH-WPC | MP-WPC |
|---|---|---|
| β-lactoglobulin | 39% | 37% |
| α-lactalbumin | 20% | 23% |
| β-casein | 28% | 24% |
| others | 13% | 16% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Navis, M.; Schwebel, L.; Soendergaard Kappel, S.; Muncan, V.; Sangild, P.T.; Abrahamse, E.; Aunsholt, L.; Thymann, T.; van Elburg, R.M.; Renes, I.B. Mildly Pasteurized Whey Protein Promotes Gut Tolerance in Immature Piglets Compared with Extensively Heated Whey Protein. Nutrients 2020, 12, 3391. https://doi.org/10.3390/nu12113391
Navis M, Schwebel L, Soendergaard Kappel S, Muncan V, Sangild PT, Abrahamse E, Aunsholt L, Thymann T, van Elburg RM, Renes IB. Mildly Pasteurized Whey Protein Promotes Gut Tolerance in Immature Piglets Compared with Extensively Heated Whey Protein. Nutrients. 2020; 12(11):3391. https://doi.org/10.3390/nu12113391
Chicago/Turabian StyleNavis, Marit, Lauriane Schwebel, Susanne Soendergaard Kappel, Vanesa Muncan, Per Torp Sangild, Evan Abrahamse, Lise Aunsholt, Thomas Thymann, Ruurd M. van Elburg, and Ingrid B. Renes. 2020. "Mildly Pasteurized Whey Protein Promotes Gut Tolerance in Immature Piglets Compared with Extensively Heated Whey Protein" Nutrients 12, no. 11: 3391. https://doi.org/10.3390/nu12113391
APA StyleNavis, M., Schwebel, L., Soendergaard Kappel, S., Muncan, V., Sangild, P. T., Abrahamse, E., Aunsholt, L., Thymann, T., van Elburg, R. M., & Renes, I. B. (2020). Mildly Pasteurized Whey Protein Promotes Gut Tolerance in Immature Piglets Compared with Extensively Heated Whey Protein. Nutrients, 12(11), 3391. https://doi.org/10.3390/nu12113391






