The Emerging Role of Vitamin C in the Prevention and Treatment of COVID-19
Abstract
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Schorah, C.J.; Downing, C.; Piripitsi, A.; Gallivan, L.; Al-Hazaa, A.H.; Sanderson, M.J.; Bodenham, A. Total vitamin C, ascorbic acid, and dehydroascorbic acid concentrations in plasma of critically ill patients. Am. J. Clin. Nutr. 1996, 63, 760–765. [Google Scholar] [CrossRef] [PubMed]
- Galley, H.F.; Davies, M.J.; Webster, N.R. Ascorbyl radical formation in patients with sepsis: Effect of ascorbate loading. Free Radic. Biol. Med. 1996, 20, 139–143. [Google Scholar] [CrossRef]
- Borrelli, E.; Roux-Lombard, P.; Grau, G.E.; Girardin, E.; Ricou, B.; Dayer, J.; Suter, P.M. Plasma concentrations of cytokines, their soluble receptors, and antioxidant vitamins can predict the development of multiple organ failure in patients at risk. Crit. Care Med. 1996, 24, 392–397. [Google Scholar] [CrossRef] [PubMed]
- Long, C.L.; Maull, K.I.; Krishnan, R.S.; Laws, H.L.; Geiger, J.W.; Borghesi, L.; Franks, W.; Lawson, T.C.; Sauberlich, H.E. Ascorbic acid dynamics in the seriously ill and injured. J. Surg. Res. 2003, 109, 144–148. [Google Scholar] [CrossRef]
- de Grooth, H.J.; Manubulu-Choo, W.P.; Zandvliet, A.S.; Spoelstra-de Man, A.M.E.; Girbes, A.R.; Swart, E.L.; Oudemans-van Straaten, H.M. Vitamin-C pharmacokinetics in critically ill patients: A randomized trial of four intravenous regimens. Chest 2018, 153, 1368–1377. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Rosengrave, P.C.; Bayer, S.; Chambers, S.; Mehrtens, J.; Shaw, G.M. Hypovitaminosis C and vitamin C deficiency in critically ill patients despite recommended enteral and parenteral intakes. Crit. Care 2017, 21, 300. [Google Scholar] [CrossRef]
- Fowler, A.A.; Syed, A.A.; Knowlson, S.; Sculthorpe, R.; Farthing, D.; DeWilde, C.; Farthing, C.A.; Larus, T.L.; Martin, E.; Brophy, D.F.; et al. Phase I safety trial of intravenous ascorbic acid in patients with severe sepsis. J. Transl. Med. 2014, 12, 32. [Google Scholar] [CrossRef] [PubMed]
- Fowler, A.A., 3rd; Truwit, J.D.; Hite, R.D.; Morris, P.E.; DeWilde, C.; Priday, A.; Fisher, B.; Thacker, L.R., 2nd ; Natarajan, R.; Brophy, D.F.; et al. Effect of vitamin C infusion on organ failure and biomarkers of inflammation and vascular injury in patients with sepsis and severe acute respiratory failure: The CITRIS-ALI randomized clinical trial. JAMA 2019, 322, 1261–1270. [Google Scholar] [CrossRef]
- Hunt, C.; Chakravorty, N.K.; Annan, G.; Habibzadeh, N.; Schorah, C.J. The clinical effects of vitamin C supplementation in elderly hospitalised patients with acute respiratory infections. Int. J. Vitam. Nutr. Res. 1994, 64, 212–219. [Google Scholar]
- Mochalkin, N.I. Ascorbic acid in the complex therapy of acute pneumonia. (English translation: http://www.mv.helsinki.fi/home/hemila/T5.pdf). Voen. Med. Zhurnal 1970, 9, 17–21. [Google Scholar]
- Carr, A.C. Vitamin C in pneumonia and sepsis. In Vitamin C: New Biochemical and Functional Insights. Oxidative Stress and Disease; Chen, Q., Vissers, M., Eds.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2020; pp. 115–135. [Google Scholar]
- Hemilä, H.; Chalker, E. Reanalysis of the effect of vitamin C on mortality in the CITRIS-ALI trial: Important findings dismissed in the trial report. Front. Med. 2020, 7, 590853. [Google Scholar] [CrossRef]
- Kashiouris, M.G.; L’Heureux, M.; Cable, C.A.; Fisher, B.J.; Leichtle, S.W.; Fowler, A.A. The emerging role of vitamin C as a treatment for sepsis. Nutrients 2020, 12, 292. [Google Scholar] [CrossRef] [PubMed]
- Wiersinga, W.J.; Rhodes, A.; Cheng, A.C.; Peacock, S.J.; Prescott, H.C. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19): A Review. JAMA 2020, 324, 782–793. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Melo, D.; Nilsson-Payant, B.E.; Liu, W.C.; Uhl, S.; Hoagland, D.; Møller, R.; Jordan, T.X.; Oishi, K.; Panis, M.; Sachs, D.; et al. Imbalanced host response to SARS-CoV-2 drives development of COVID-19. Cell 2020, 181, 1036–1045. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Bastard, P.; Liu, Z.; Le Pen, J.; Moncada-Velez, M.; Chen, J.; Ogishi, M.; Sabli, I.K.D.; Hodeib, S.; Korol, C.; et al. Inborn errors of type I IFN immunity in patients with life-threatening COVID-19. Science 2020, 370, eabd4570. [Google Scholar] [CrossRef]
- Bastard, P.; Rosen, L.B.; Zhang, Q.; Michailidis, E.; Hoffmann, H.H.; Zhang, Y.; Dorgham, K.; Philippot, Q.; Rosain, J.; Béziat, V.; et al. Auto-antibodies against type I IFNs in patients with life-threatening COVID-19. Science 2020, 370, eabd4585. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, H.; Bae, S.; Choi, J.; Lim, S.Y.; Lee, N.; Kong, J.M.; Hwang, Y.I.; Kang, J.S.; Lee, W.J. Vitamin C is an essential factor on the anti-viral immune responses through the production of interferon-α/β at the initial stage of influenza A virus (H3N2) infection. Immune Netw. 2013, 13, 70–74. [Google Scholar] [CrossRef]
- Geber, W.F.; Lefkowitz, S.S.; Hung, C.Y. Effect of ascorbic acid, sodium salicylate, and caffeine on the serum interferon level in response to viral infection. Pharmacology 1975, 13, 228–233. [Google Scholar] [CrossRef]
- Zhang, J.; Rao, X.; Li, Y.; Zhu, Y.; Liu, F.; Guo, G.; Luo, G.; Meng, Z.; De Backer, D.; Xiang, H.; et al. High-dose vitamin C infusion for the treatment of critically ill COVID-19. Res. Sq. 2020. [Google Scholar] [CrossRef]
- José, R.J.; Williams, A.; Manuel, A.; Brown, J.S.; Chambers, R.C. Targeting coagulation activation in severe COVID-19 pneumonia: Lessons from bacterial pneumonia and sepsis. Eur. Respir. Rev. 2020, 29, 200240. [Google Scholar] [CrossRef]
- Tyml, K. Vitamin C and microvascular dysfunction in systemic inflammation. Antioxidants 2017, 6, 49. [Google Scholar] [CrossRef] [PubMed]
- Hiedra, R.; Lo, K.B.; Elbashabsheh, M.; Gul, F.; Wright, R.M.; Albano, J.; Azmaiparashvili, Z.; Patarroyo Aponte, G. The use of IV vitamin C for patients with COVID-19: A case series. Expert Rev. Anti Infect. Ther. 2020, 1–3. [Google Scholar] [CrossRef]
- Middleton, E.A.; He, X.Y.; Denorme, F.; Campbell, R.A.; Ng, D.; Salvatore, S.P.; Mostyka, M.; Baxter-Stoltzfus, A.; Borczuk, A.C.; Loda, M.; et al. Neutrophil extracellular traps contribute to immunothrombosis in COVID-19 acute respiratory distress syndrome. Blood 2020, 136, 1169–1179. [Google Scholar] [CrossRef]
- Skendros, P.; Mitsios, A.; Chrysanthopoulou, A.; Mastellos, D.C.; Metallidis, S.; Rafailidis, P.; Ntinopoulou, M.; Sertaridou, E.; Tsironidou, V.; Tsigalou, C.; et al. Complement and tissue factor-enriched neutrophil extracellular traps are key drivers in COVID-19 immunothrombosis. J. Clin. Investig. 2020. [Google Scholar] [CrossRef]
- Mohammed, B.M.; Fisher, B.J.; Kraskauskas, D.; Farkas, D.; Brophy, D.F.; Fowler, A.A.; Natarajan, R. Vitamin C: A novel regulator of neutrophil extracellular trap formation. Nutrients 2013, 5, 3131–3151. [Google Scholar] [CrossRef]
- Qiao, X.; Fisher, B.; Kashiouris, M.G.; Truwit, J.D.; Hite, R.D.; Morris, P.E.; Martin, G.S.; Fowler, A.A. Effects of high dose intravenous vitamin C (IVC) on plasma cell-free DNA levels in patients with sepsis-associated ARDS. Am. J. Respir. Crit. Care Med. 2019, 201, A2100. [Google Scholar]
- Schönrich, G.; Raftery, M.J.; Samstag, Y. Devilishly radical NETwork in COVID-19: Oxidative stress, neutrophil extracellular traps (NETs), and T cell suppression. Adv. Biol. Regul. 2020, 77, 100741. [Google Scholar] [CrossRef] [PubMed]
- Laforge, M.; Elbim, C.; Frère, C.; Hémadi, M.; Massaad, C.; Nuss, P.; Benoliel, J.J.; Becker, C. Tissue damage from neutrophil-induced oxidative stress in COVID-19. Nat. Rev. Immunol. 2020, 20, 515–516. [Google Scholar] [CrossRef]
- Carr, A.C.; Spencer, E.; Mackle, D.; Hunt, A.; Judd, H.; Mehrtens, J.; Parker, K.; Stockwell, Z.; Gale, C.; Beaumont, M.; et al. The effect of conservative oxygen therapy on systemic biomarkers of oxidative stress in critically ill patients. Free Radic. Biol. Med. 2020. under consideration. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Spencer, E.; Dixon, L.; Chambers, S.T. Patients with community acquired pneumonia exhibit depleted vitamin C status and elevated oxidative stress. Nutrients 2020, 12, 1318. [Google Scholar] [CrossRef] [PubMed]
- Galley, H.F.; Howdle, P.D.; Walker, B.E.; Webster, N.R. The effects of intravenous antioxidants in patients with septic shock. Free Radic. Biol. Med. 1997, 23, 768–774. [Google Scholar] [CrossRef]
- Sawyer, M.A.J.; Mike, J.J.; Chavin, K.; Marino, P.L. Antioxidant therapy and survival in ARDS. Crit. Care Med. 1989, 17, S153. [Google Scholar]
- World Health Organization. A Coordinated Global Research Roadmap: 2019 Novel Coronavirus; World Health Organization: Geneva, Switzerland, 2020. [Google Scholar]
- Hemila, H.; Chalker, E. Vitamin C may reduce the duration of mechanical ventilation in critically ill patients: A meta-regression analysis. J. Intensive Care 2020, 8, 15. [Google Scholar] [CrossRef] [PubMed]
- Hemila, H.; Chalker, E. Vitamin C can shorten the length of stay in the ICU: A meta-analysis. Nutrients 2019, 11, 708. [Google Scholar] [CrossRef] [PubMed]
- Siow, W.T.; Liew, M.F.; Shrestha, B.R.; Muchtar, F.; See, K.C. Managing COVID-19 in resource-limited settings: Critical care considerations. Crit. Care 2020, 24, 167. [Google Scholar] [CrossRef]
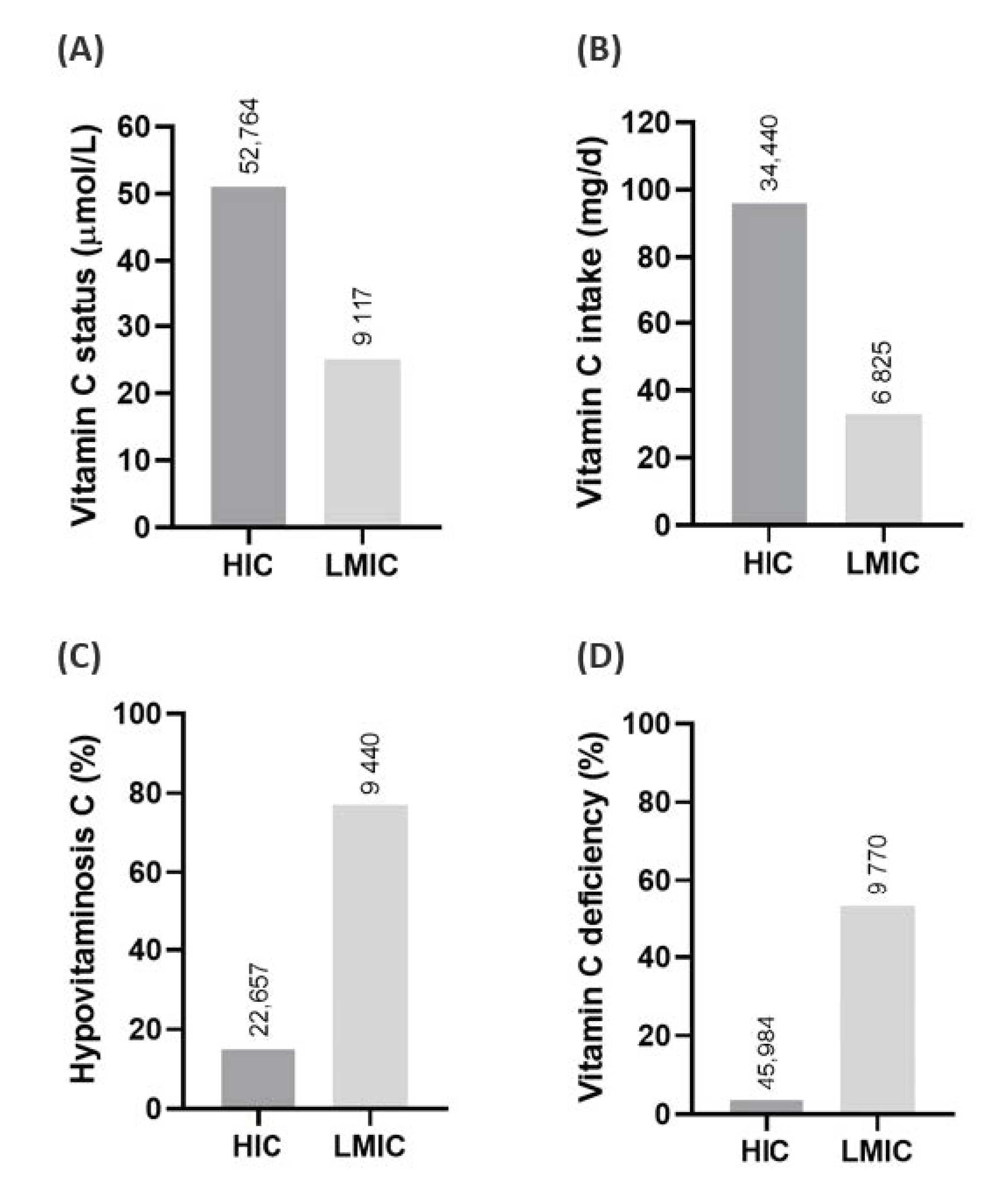
- Rowe, S.; Carr, A.C. Global vitamin C status and prevalence of deficiency: A cause for concern? Nutrients 2020, 12, 2008. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Rowe, S. Factors affecting vitamin C status and prevalence of deficiency: A global health perspective. Nutrients 2020, 12, 1963. [Google Scholar] [CrossRef]
- Centres for Disease Control and Prevention. Assessing Risk Factors for Severe COVID-19 Illness 2020. Available online: https://www.cdc.gov/coronavirus/2019-ncov/covid-data/investigations-discovery/assessing-risk-factors.html (accessed on 5 September 2020).
- Carr, A.C. Micronutrient status of COVID-19 patients: A critical consideration. Crit. Care 2020, 24, 349. [Google Scholar] [CrossRef] [PubMed]
- Lykkesfeldt, J. On the effect of vitamin C intake on human health: How to (mis)interprete the clinical evidence. Redox Biol. 2020, 34, 101532. [Google Scholar] [CrossRef]
- Arvinte, C.; Singh, M.; Marik, P.E. Serum levels of vitamin C and vitamin D in a cohort of critically ill COVID-19 patients of a north American community hospital intensive care unit in may 2020. A pilot study. Med. Drug Discov. 2020, 8, 100064. [Google Scholar] [CrossRef]
- Chiscano-Camón, L.; Ruiz-Rodriguez, J.C.; Ruiz-Sanmartin, A.; Roca, O.; Ferrer, R. Vitamin C levels in patients with SARS-CoV-2-associated acute respiratory distress syndrome. Crit. Care 2020, 24, 522. [Google Scholar] [CrossRef] [PubMed]
- Pullar, J.M.; Bayer, S.; Carr, A.C. Appropriate handling, processing and analysis of blood samples is essential to avoid oxidation of vitamin C to dehydroascorbic acid. Antioxidants 2018, 7, 29. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Zhu, Y.; Zhang, J.; Li, Y.; Peng, Z. Intravenous high-dose vitamin C for the treatment of severe COVID-19: Study protocol for a multicentre randomised controlled trial. BMJ Open 2020, 10, e039519. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H.; Suonsyrjä, T. Vitamin C for preventing atrial fibrillation in high risk patients: A systematic review and meta-analysis. BMC Cardiovasc. Disord. 2017, 17, 49. [Google Scholar] [CrossRef]
| Country Study ID | Title | Participants | Intervention | Primary Outcome(s) |
|---|---|---|---|---|
| Canada NCT04401150 | Lessening Organ Dysfunction with VITamin C—COVID-19 (LOVIT-COVID) | 800 hospitalized patients with COVID-19 | 50 mg/kg/6 h IVC for 96 h vs. placebo | Death or persistent organ dysfunction |
| Italy NCT04323514 | Use of Ascorbic Acid in Patients With COVID-19 | 500 patients with COVID-19 pneumonia | 10 g/d IVC for 72 h uncontrolled | In-hospital mortality |
| USA NCT04344184 | Early Infusion of Vitamin C for Treatment of Novel COVID-19 Acute Lung Injury (EVICT-CORONA-ALI) | 200 patients with COVID-19 acute lung injury | 100 mg/kg/8 h IVC for 96 h vs. placebo | Number of ventilator-free days |
| USA NCT04363216 | Pharmacologic Ascorbic Acid as an Activator of Lymphocyte Signaling for COVID-19 Treatment | 66 patients with COVID-19 | 0.3–0.9 g/kg/d IVC for 6 days vs. control | Clinical Improvement |
| China NCT04264533 | Vitamin C Infusion for the Treatment of Severe 2019-nCoV Infected Pneumonia | 140 patients with COVID-19 pneumonia | 12 g/12 h IVC for 7 days vs. placebo | Ventilator-free days |
| China ChiCTR-2000032400 | The efficacy and safety of high dose IVC in the treatment of novel coronavirus pneumonia (COVID-19) | 120 patients with COVID-19 pneumonia | 100 mg/kg/d IVC for up to 7 days vs. placebo | CRP, ESR, existence of SIRS |
| Iran IRCT2020-0411047025N1 | Evaluation of effectiveness of IVC in Patients with COVID-19 Referred to Imam Khomeini Hospital | 110 patients with COVID-19 | 1.5 g/6 h IVC for up to 5 days vs. control | Improvement of SPO2 |
| Iran IRCT2019-0917044805N2 | Effects of High-dose Vitamin C on Treatment, Clinical Symptoms and Laboratory Signs of Iranian COVID-19 Patients | 60 patients with COVID-19 | 12 g/d IVC for 4 days vs. placebo | Time to clinical improvement |
| Iran IRCT2020-0516047468N1 | Interventional study of IVC in definitive patients with COVID-19 and its effect on changes in lung CT scan and clinical and laboratory symptoms of patients | 50 patients with COVID-19 | 2 g/6 h IVC for 5 days vs. control | The amount of lung involvement in a CT scan |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Carr, A.C.; Rowe, S. The Emerging Role of Vitamin C in the Prevention and Treatment of COVID-19. Nutrients 2020, 12, 3286. https://doi.org/10.3390/nu12113286
Carr AC, Rowe S. The Emerging Role of Vitamin C in the Prevention and Treatment of COVID-19. Nutrients. 2020; 12(11):3286. https://doi.org/10.3390/nu12113286
Chicago/Turabian StyleCarr, Anitra C., and Sam Rowe. 2020. "The Emerging Role of Vitamin C in the Prevention and Treatment of COVID-19" Nutrients 12, no. 11: 3286. https://doi.org/10.3390/nu12113286
APA StyleCarr, A. C., & Rowe, S. (2020). The Emerging Role of Vitamin C in the Prevention and Treatment of COVID-19. Nutrients, 12(11), 3286. https://doi.org/10.3390/nu12113286






