Skeletal Muscle Loss during Multikinase Inhibitors Therapy: Molecular Pathways, Clinical Implications, and Nutritional Challenges
Abstract
1. Introduction
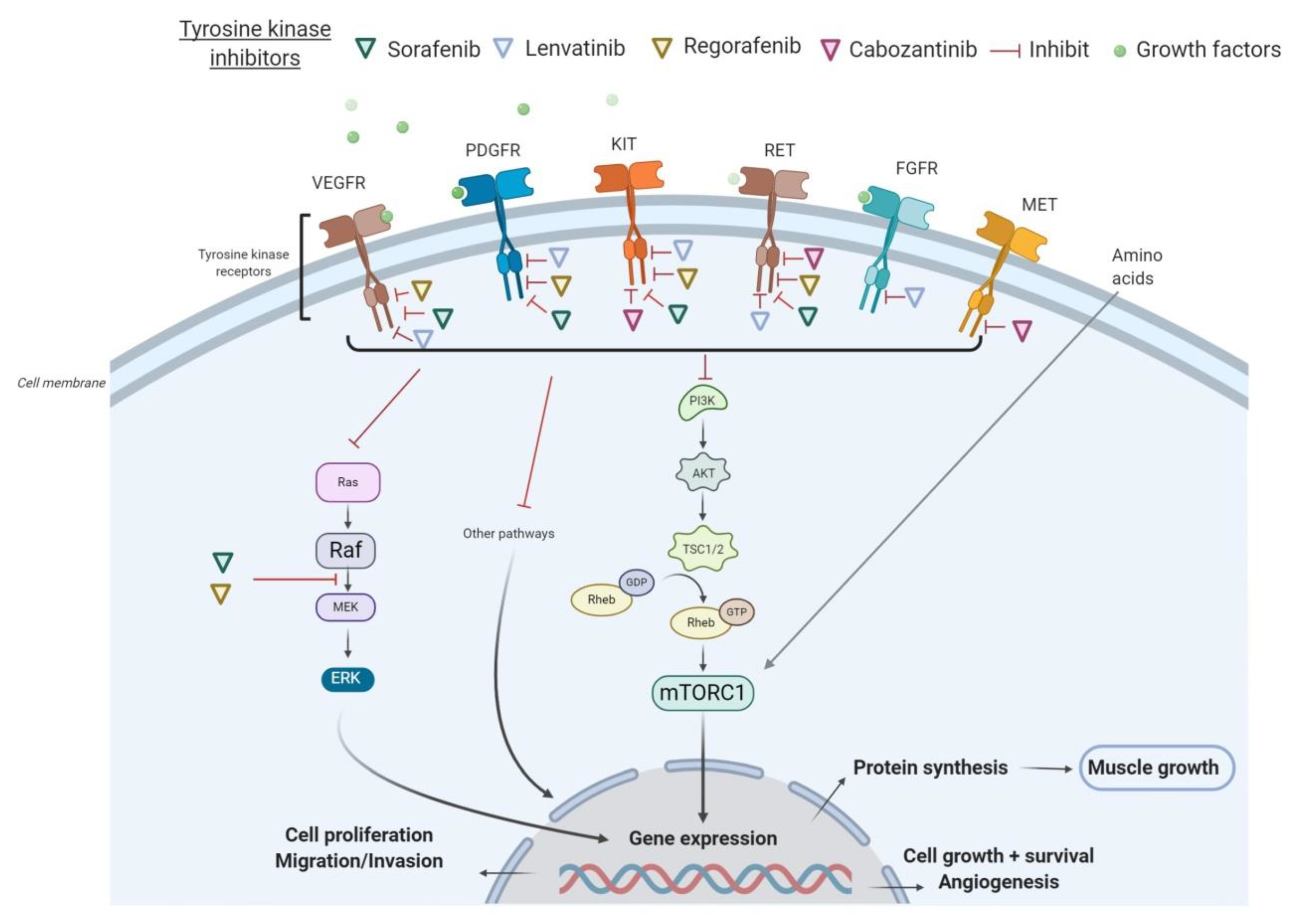
2. Molecular Pathways of TKI Involved in Muscle Wasting
3. Impact of TKI Treatment on Muscle Wasting
3.1. Sorafenib
3.2. Lenvatinib
3.3. Sunitinib
3.4. Regorafenib
3.5. Pazopanib
3.6. Axitinib
3.7. Vandetanib
4. Implications of Muscle Wasting in Clinical Outcomes in Patients Treated with TKI
4.1. RCC
4.2. HCC
4.3. CRC
4.4. Thyroid Cancer
5. Nutritional Challenges
5.1. Early Assessment and Monitoring of Nutritional Status During Treatment
- −
- Appendicular skeletal muscle index (ASMI), determined by dual-energy x-ray absorptiometry (men < 7.26 kg/m2; women < 5.45 kg/m2);
- −
- SMI, determined from oncological CT imaging (men < 55 cm2/m2; women < 39 cm2/m2);
- −
- Whole body fat-free mass index without bone determined by bioelectrical impedance (men < 14.6 kg/m2; women < 11.4 kg/m2).
5.2. Personalized Nutritional Support
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Dagher, R.; Cohen, M.; Williams, G.; Rothmann, M.; Gobburu, J.; Robbie, G.; Rahman, A.; Chen, G.; Staten, A.; Griebel, D.; et al. Approval summary: Imatinib mesylate in the treatment of metastatic and/or unresectable malignant gastrointestinal stromal tumors. Clin. Cancer Res. 2002, 8, 3034–3038. [Google Scholar] [PubMed]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Coppin, C. Sunitinib for advanced renal cell cancer. Biology 2008, 2, 97–105. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mir, O.; Coriat, R.; Blanchet, B.; Durand, J.P.; Boudou-Rouquette, P.; Michels, J.; Ropert, S.; Vidal, M.; Pol, S.; Chaussade, S.; et al. Sarcopenia predicts early dose-limiting toxicities and pharmacokinetics of sorafenib in patients with hepatocellular carcinoma. PLoS ONE 2012, 7, e37563. [Google Scholar] [CrossRef]
- Rinninella, E.; Cintoni, M.; Raoul, P.; Pozzo, C.; Strippoli, A.; Bria, E.; Tortora, G.; Gasbarrini, A.; Mele, M.C. Muscle mass, assessed at diagnosis by L3-CT scan as a prognostic marker of clinical outcomes in patients with gastric cancer: A systematic review and meta-analysis. Clin. Nutr. 2020, 39, 2045–2054. [Google Scholar] [CrossRef]
- Rinninella, E.; Fagotti, A.; Cintoni, M.; Raoul, P.; Scaletta, G.; Scambia, G.; Gasbarrini, A.; Mele, M.C. Skeletal muscle mass as a prognostic indicator of outcomes in ovarian cancer: A systematic review and meta-analysis. Int. J. Gynecol. Cancer 2020, 30, 654–663. [Google Scholar] [CrossRef]
- Shachar, S.S.; Williams, G.R.; Muss, H.B.; Nishijima, T.F. Prognostic value of sarcopenia in adults with solid tumours: A meta-analysis and systematic review. Eur. J. Cancer 2016, 57, 58–67. [Google Scholar] [CrossRef]
- Glass, D.J. Pi3 kinase regulation of skeletal muscle hypertrophy and atrophy. Curr. Top. Microbiol. Immunol. 2010, 346, 267–278. [Google Scholar]
- Schiaffino, S.; Mammucari, C. Regulation of skeletal muscle growth by the IGF1- Akt/PKB pathway: Insights from genetic models. Skelet. Muscle. 2011, 1, 4. [Google Scholar] [CrossRef]
- Adegoke, O.A.; Abdullahi, A.; Tavajohi-Fini, P. mTORC1 and the regulation of skeletal muscle anabolism and mass. Appl. Physiol. Nutr. Metab. 2012, 37, 395–406. [Google Scholar] [CrossRef]
- Arora, A.; Scholar, E.M. Role of tyrosine kinase inhibitors in cancer therapy. J. Pharm. Exp. 2005, 315, 971–979. [Google Scholar] [CrossRef] [PubMed]
- Pawson, T. Regulation and targets of receptor tyrosine kinases. Eur. J. Cancer 2002, 38, S3–S10. [Google Scholar] [CrossRef]
- Shibuya, M. Vascular endothelial growth factor (VEGFR) and its receptor (VEGFR) signaling in angiogenesis: A crucial target for anti- and pro-angiogenic therapies. Genes Cancer 2011, 2, 1097–1105. [Google Scholar] [CrossRef] [PubMed]
- Mendel, D.B.; Laird, A.D.; Smolich, B.D.; Blake, R.A.; Liang, C.; Hannah, A.L.; Shaheen, R.M.; Ellis, L.M.; Weitman, S.; Shawver, L.K.; et al. Development of SU5416, a selective small molecule inhibitor of VEGF receptor tyrosine kinase activity, as an anti-angiogenesis agent. Anticancer Drug Des. 2000, 15, 29–41. [Google Scholar] [PubMed]
- Gotink, K.J.; Verheul, H.M. Anti-angiogenic tyrosine kinase inhibitors: What is their mechanism of action? Angiogenesis 2010, 13, 1–14. [Google Scholar] [CrossRef]
- Nordby, Y.; Richardsen, E.; Rakaee, M.; Ness, N.; Donnem, T.; Patel, H.R.; Busund, L.T.; Bremnes, R.M.; Andersen, S. High expression of PDGFR-β in prostate cancer stroma is independently associated with clinical and biochemical prostate cancer recurrence. Sci. Rep. 2017, 7, 43378. [Google Scholar] [CrossRef] [PubMed]
- Wieduwilt, M.J.; Moasser, M.M. The epidermal growth factor receptor family: Biology driving targeted therapeutics. Cell Mol. Life Sci. 2008, 65, 1566–1584. [Google Scholar] [CrossRef]
- Sigismund, S.; Avanzato, D.; Lanzetti, L. Emerging functions of the EGFR in cancer. Mol. Oncol. 2018, 12, 3–20. [Google Scholar] [CrossRef]
- Du, Z.; Lovly, C.M. Mechanisms of receptor tyrosine kinase activation in cancer. Mol. Cancer 2018, 17, 58. [Google Scholar] [CrossRef]
- Xu, F.; Na, L.; Li, Y.; Chen, L. Roles of the PI3K/AKT/mTOR signalling pathways in neurodegenerative diseases and tumours. Cell Biosci. 2020, 10, 54. [Google Scholar] [CrossRef]
- Bodine, S.C.; Stitt, T.N.; Gonzalez, M.; Kline, W.O.; Stover, G.L.; Bauerlein, R.; Zlotchenko, E.; Scrimgeour, A.; Lawrence, J.C.; Glass, D.J.; et al. Akt/mTOR pathway is a crucial regulator of skeletal muscle hypertrophy and can prevent muscle atrophy in vivo. Nat. Cell Biol. 2001, 3, 1014–1019. [Google Scholar] [CrossRef] [PubMed]
- Edinger, A.L.; Thompson, C.B. Akt maintains cell size and survival by increasing mTOR-dependent nutrient uptake. Mol. Biol. Cell. 2002, 13, 2276–2288. [Google Scholar] [CrossRef]
- Adnane, L.; Trail, P.A.; Taylor, I.; Wilhelm, S.M. Sorafenib (BAY 43-9006, Nexavar), a dual-action inhibitor that targets RAF/MEK/ERK pathway in tumor cells and tyrosine kinases VEGFR/PDGFR in tumor vasculature. Methods Enzym. 2006, 407, 597–612. [Google Scholar]
- Wilhelm, S.M.; Dumas, J.; Adnane, L.; Lynch, M.; Carter, C.A.; Schütz, G.; Thierauch, K.H.; Zopf, D. Regorafenib (BAY 73-4506): A new oral multikinase inhibitor of angiogenic, stromal and oncogenic receptor tyrosine kinases with potent preclinical antitumor activity. Int. J. Cancer 2011, 129, 245–255. [Google Scholar] [CrossRef] [PubMed]
- Gyawali, B.; Shimokata, T.; Honda, K.; Kondoh, C.; Hayashi, N.; Yoshino, Y.; Sassa, N.; Nakano, Y.; Gotoh, M.; Ando, Y. Muscle wasting associated with the long-term use of mTOR inhibitors. Mol. Clin. Oncol. 2016, 5, 641–646. [Google Scholar] [CrossRef]
- Chéry, L.; Borregales, L.D.; Fellman, B.; Urbauer, D.L.; Garg, N.; Parker, N.; Katz, M.H.G.; Wood, C.G.; Karam, J.A. The Effects of Neoadjuvant Axitinib on Anthropometric Parameters in Patients with Locally Advanced Non-metastatic Renal Cell Carcinoma. Urology 2017, 108, 114–121. [Google Scholar] [CrossRef]
- Rinninella, E.; Cintoni, M.; Raoul, P.; Mele, M.C.; De Gaetano, A.M.; Marini, M.G.; Mora, V.; Gasbarrini, A. Minimal impact of lenvatinib (Lenvima®) on muscle mass in advanced hepatocellular carcinoma and implications for treatment duration. Two cases from the REFLECT study. Eur. Rev. Med. Pharm. Sci. 2019, 23, 10132–10138. [Google Scholar]
- Hiraoka, A.; Kumada, T.; Kariyama, K.; Takaguchi, K.; Atsukawa, M.; Itobayashi, E.; Tsuji, K.; Tajiri, K.; Hirooka, M.; Shimada, N.; et al. Real-life Practice Experts for HCC (RELPEC) Study Group, HCC 48 Group (hepatocellular carcinoma experts from 48 clinics in Japan). Clinical features of lenvatinib for unresectable hepatocellular carcinoma in real-world conditions: Multicenter analysis. Cancer Med. 2019, 8, 137–146. [Google Scholar] [CrossRef]
- Huemer, F.; Schlintl, V.; Hecht, S.; Hackl, H.; Melchardt, T.; Rinnerthaler, G.; Greil, R.; Weiss, L. Regorafenib Is Associated With Increased Skeletal Muscle Loss Compared to TAS-102 in Metastatic Colorectal Cancer. Clin. Colorectal Cancer 2019, 18, 159–166. [Google Scholar] [CrossRef]
- Hacioglu, M.B.; Kostek, O.; Kurt, N.; Kucukarda, A.; Gokyer, A.; Ustabasioglu, F.E.; Karatas, F.; Tuncbilek, N.; Uzunoglu, S.; Bilici, A.; et al. Comparison of skeletal muscle mass loss in patients with metastatic colorectal cancer treated with regorafenib or TAS-102. J. BUON 2019, 24, 2198–2204. [Google Scholar]
- Antoun, S.; Baracos, V.E.; Birdsell, L.; Escudier, B.; Sawyer, M.B. Low body mass index and sarcopenia associated with dose-limiting toxicity of sorafenib in patients with renal cell carcinoma. Ann. Oncol. 2010, 21, 1594–1598. [Google Scholar] [CrossRef]
- Huillard, O.; Jouinot, A.; Tlemsani, C.; Brose, M.S.; Arrondeau, J.; Meinhardt, G.; Fellous, M.; De Sanctis, Y.; Schlumberger, M.; Goldwasser, F. Body Composition in Patients with Radioactive Iodine-Refractory, Advanced Differentiated Thyroid Cancer Treated with Sorafenib or Placebo: A Retrospective Analysis of the Phase III DECISION Trial. Thyroid 2019, 29, 1820–1827. [Google Scholar] [CrossRef] [PubMed]
- Uchikawa, S.; Kawaoka, T.; Namba, M.; Kodama, K.; Ohya, K.; Morio, K.; Nakahara, T.; Murakami, E.; Tsuge, M.; Hiramatsu, A.; et al. Skeletal muscle loss during tyrosine kinase inhibitor treatment for advanced hepatocellular carcinoma patients. Liver Cancer 2020, 9, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Gu, W.; Wu, J.; Liu, X.; Zhang, H.; Shi, G.; Zhu, Y.; Ye, D. Early skeletal muscle loss during target therapy is a prognostic biomarker in metastatic renal cell carcinoma patients. Sci. Rep. 2017, 7, 7587. [Google Scholar] [CrossRef] [PubMed]
- Köstek, O.; Yılmaz, E.; Hacıoğlu, M.B.; Demircan, N.C.; Gökyer, A.; Uzunoğlu, S.; Tunçbilek, N.; Çiçin, İ.; Erdoğan, B. Changes in skeletal muscle area and lean body mass during pazopanib vs sunitinib therapy for metastatic renal cancer. Cancer Chemother. Pharm. 2019, 83, 735–742. [Google Scholar] [CrossRef] [PubMed]
- Massicotte, M.H.; Borget, I.; Broutin, S.; Baracos, V.E.; Leboulleux, S.; Baudin, E.; Paci, A.; Deroussent, A.; Schlumberger, M.; Antoun, S. Body composition variation and impact of low skeletal muscle mass in patients with advanced medullary thyroid carcinoma treated with vandetanib: Results from a placebo-controlled study. J. Clin. Endocrinol. Metab. 2013, 98, 2401–2408. [Google Scholar] [CrossRef] [PubMed]
- Allegato I Riassunto Delle Caratteristiche del Prodotto. Available online: https://www.ema.europa.eu/en/documents/product-information/nexavar-epar-product-information_it.pdf (accessed on 21 January 2020).
- Wilhelm, S.; Carter, C.; Lynch, M.; Lowinger, T.; Dumas, J.; Smith, R.A.; Schwartz, B.; Simantov, R.; Kelley, S. Discovery and development of sorafenib: A multikinase inhibitor for treating cancer. Nat. Rev. Drug Discov. 2006, 5, 835–844. [Google Scholar] [CrossRef]
- Li, L.; Zhao, G.D.; Shi, Z.; Qi, L.L.; Zhou, L.Y.; Fu, Z.X. The Ras/Raf/MEK/ERK signaling pathway and its role in the occurrence and development of HCC. Oncol. Lett. 2016, 12, 3045–3050. [Google Scholar] [CrossRef]
- Chiang, I.T.; Liu, Y.C.; Wang, W.H.; Hsu, F.T.; Chen, H.W.; Lin, W.J.; Chang, W.Y.; Hwang, J.J. Sorafenib inhibits TPA-induced MMP-9 and VEGF expression via suppression of ERK/NF-κB pathway in hepatocellular carcinoma cells. Vivo 2012, 26, 671–681. [Google Scholar]
- Damrauer, J.S.; Stadler, M.E.; Acharyya, S.; Baldwin, A.S.; Couch, M.E.; Guttridge, D.C. Chemotherapy-induced muscle wasting: Association with NF-κB and cancer cachexia. Eur. J. Transl. Myol. 2018, 28, 7590. [Google Scholar] [CrossRef]
- Zschäbitz, S.; Grüllich, C. Lenvatinib: A Tyrosine Kinase Inhibitor of VEGFR 1-3, FGFR 1-4, PDGFRα, KIT and RE. Recent Results Cancer Res. 2018, 211, 187–198. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.ema.europa.eu/en/documents/product-information/lenvima-epar-productinformation_it.pdf (accessed on 21 January 2020).
- Motzer, R.J.; Rini, B.I.; Bukowski, R.M.; Curti, B.D.; George, D.J.; Hudes, G.R.; Redman, B.G.; Margolin, K.A.; Merchan, J.R.; Wilding, G.; et al. Sunitinib in patients with metastatic renal cell carcinoma. JAMA 2006, 295, 2516–2524. [Google Scholar] [CrossRef] [PubMed]
- Bruix, J.; Qin, S.; Merle, P.; Granito, A.; Huang, Y.H.; Bodoky, G.; Pracht, M.; Yokosuka, O.; Rosmorduc, O.; Breder, V.; et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): A randomised, double-blind, placebo-controlled, Phase 3 trial. Lancet 2017, 389, 56–66. [Google Scholar] [CrossRef]
- Huot, J.R.; Essex, A.L.; Gutierrez, M.; Barreto, R.; Wang, M.; Waning, D.L.; Plotkin, L.I.; Bonetto, A. Chronic Treatment with Multi-Kinase Inhibitors Causes Differential Toxicities on Skeletal and Cardiac Muscles. Cancers 2019, 11, E571. [Google Scholar] [CrossRef] [PubMed]
- Allegato I Riassunto Delle Caratteristiche del Prodotto. Available online: https://www.ema.europa.eu/en/documents/product-information/votrient-epar-product-information_it.pdf (accessed on 6 October 2020).
- Schutz, F.A.; Choueiri, T.K.; Sternberg, C.N. Pazopanib: Clinicaldevelopment of a potent anti-angiogenic drug. Crit Rev. Oncol. Hematol. 2011, 77, 163–171. [Google Scholar] [CrossRef]
- Schmidinger, M.; Wittes, J. First-line treatment of metastatic renal cell carcinoma after COMPARZ and PISCES. Curr. Opin. Urol. 2015, 25, 395–401. [Google Scholar] [CrossRef]
- Rugo, H.S.; Herbst, R.S.; Liu, G.; Park, J.W.; Kies, M.S.; Steinfeldt, H.M.; Pithavala, Y.K.; Reich, S.D.; Freddo, J.L.; Wilding, G. Phase I trial of the oral antiangiogenesis agent AG-013736 in patients with advanced solid tumors: Pharmacokinetic and clinical results. J. Clin. Oncol. 2005, 23, 5474–5483. [Google Scholar] [CrossRef]
- Available online: https://www.ema.europa.eu/en/documents/product-information/inlyta-epar-productinformation_it.pdf (accessed on 21 January 2020).
- Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/022465s-010S-012lbl.pdf (accessed on 21 January 2020).
- Belizário, J.E.; Fontes-Oliveira, C.C.; Borges, J.P.; Kashiabara, J.A.; Vannier, E. Skeletal muscle wasting and renewal: A pivotal role of myokine IL-6. Springerplus 2016, 5, 619. [Google Scholar] [CrossRef]
- Huillard, O.; Mir, O.; Peyromaure, M.; Tlemsani, C.; Giroux, J.; Boudou-Rouquette, P.; Ropert, S.; Delongchamps, N.B.; Zerbib, M.; Goldwasser, F. Sarcopenia and body mass index predict sunitinib-induced early dose-limiting toxicities in renal cancer patients. Br. J. Cancer. 2013, 108, 1034–1041. [Google Scholar] [CrossRef]
- Cushen, S.J.; Power, D.G.; Teo, M.Y.; MacEneaney, P.; Maher, M.M.; McDermott, R.; O’Sullivan, K.; Ryan, A.M. Body Composition by Computed Tomography as a Predictor of Toxicity in Patients With Renal Cell Carcinoma Treated With Sunitinib. Am. J. Clin. Oncol. 2017, 40, 47–52. [Google Scholar] [CrossRef]
- Sharma, P.; Zargar-Shoshtari, K.; Caracciolo, J.T.; Fishman, M.; Poch, M.A.; Pow-Sang, J.; Sexton, W.J.; Spiess, P.E. Sarcopenia as a predictor of overall survival after cytoreductive nephrectomy for metastatic renal cell carcinoma. Urol. Oncol. 2015, 33, 339.e17–e23. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, H.; Nakanishi, Y.; Kataoka, M.; Tobisu, K.; Koga, F. Prognostic Significance of Sarcopenia in Patients with Metastatic Renal Cell Carcinoma. J. Urol. 2016, 195, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Antonelli, G.; Gigante, E.; Iavarone, M.; Begini, P.; Sangiovanni, A.; Iannicelli, E.; Biondetti, P.; Pellicelli, A.M.; Miglioresi, L.; Marchetti, P.; et al. Sarcopenia is associated with reduced survival in patients with advanced hepatocellular carcinoma undergoing sorafenib treatment. United Eur. Gastroenterol. J. 2018, 6, 1039–1048. [Google Scholar] [CrossRef] [PubMed]
- Uojima, H.; Chuma, M.; Tanaka, Y.; Hidaka, H.; Nakazawa, T.; Iwabuchi, S.; Kobayashi, S.; Hattori, N.; Ogushi, K. Morimoto, M.; et al. Skeletal muscle mass influences tolerability and prognosis in hepatocellular carcinoma patients treated with lenvatinib. Liver Cancer 2019, 9, 193–206. [Google Scholar] [CrossRef] [PubMed]
- Begini, P.; Gigante, E.; Antonelli, G.; Carbonetti, F.; Iannicelli, E.; Anania, G.; Imperatrice, B.; Pellicelli, A.M.; Fave, G.D.; Marignani, M. Sarcopenia predicts reduced survival in patients with hepatocellular carcinoma at first diagnosis. Ann. Hepatol. 2017, 16, 107–114. [Google Scholar] [CrossRef]
- Molfino, A.; Johnson, S.; Medici, V. The Challenges of Nutritional Assessment in Cirrhosis. Curr. Nutr. Rep. 2017, 6, 274–280. [Google Scholar] [CrossRef]
- Silva, M.; Gomes, S.; Peixoto, A.; Torres-Ramalho, P.; Cardoso, H.; Azevedo, R.; Cunha, C.; Macedo, G. Nutrition in Chronic Liver Disease. GE Port. J. Gastroenterol. 2015, 22, 268–276. [Google Scholar] [CrossRef]
- Thoresen, L.; Frykholm, G.; Lydersen, S.; Ulveland, H.; Baracos, V.; Prado, C.M.; Birdsell, L.; Falkmer, U. Nutritional status, cachexia and survival in patients with advanced colorectal carcinoma. Different assessment criteria for nutritional status provide unequal results. Clin. Nutr. 2013, 32, 65–72. [Google Scholar] [CrossRef]
- Blauwhoff-Buskermolen, S.; Versteeg, K.S.; de van der Schueren, M.A.; den Braver, N.R.; Berkhof, J.; Langius, J.A.; Verheul, H.M. Loss of Muscle Mass During Chemotherapy Is Predictive for Poor Survival of Patients With Metastatic Colorectal Cancer. J. Clin. Oncol. 2016, 34, 1339–1344. [Google Scholar] [CrossRef]
- Kurk, S.; Peeters, P.; Stellato, R.; Dorresteijn, B.; de Jong, P.; Jourdan, M.; Creemers, G.J.; Erdkamp, F.; de Jongh, F.; Kint, P.; et al. Skeletal muscle mass loss and dose-limiting toxicities in metastatic colorectal cancer patients. J. Cachexia Sarcopenia Muscle. 2019, 10, 803–813. [Google Scholar] [CrossRef]
- Gökyer, A.; Küçükarda, A.; Köstek, O.; Hacıoğlu, M.B.; Sunal, B.S.; Demircan, N.C.; Uzunoğlu, S.; Solak, S.; İşsever, K.; Çiçin, I.; et al. Relation between sarcopenia and dose-limiting toxicity in patients with metastatic colorectal cancer who received regorafenib. Clin. Transl. Oncol. 2019, 21, 1518–1523. [Google Scholar] [CrossRef] [PubMed]
- Boirie, Y.; Gachon, P.; Beaufrere, B. Splanchnic and whole-body leucine kinetics in young and elderly men. Am. J. Clin. Nutr. 1997, 65, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef] [PubMed]
- Arends, J.; Baracos, V.; Bertz, H.; Bozzetti, F.; Calder, P.C.; Deutz, N.E.P.; Erickson, N.; Laviano, A.; Lisanti, M.P.; Lobo, D.N.; et al. ESPEN expert group recommendations for action against cancer-related malnutrition. Clin. Nutr. 2017, 36, 1187–1196. [Google Scholar] [CrossRef]
- Bauer, J.; Morley, J.E.; Schols, A.M.W.J.; Ferrucci, L.; Cruz-Jentoft, A.J.; Dent, E.; Baracos, V.E.; Crawford, J.A.; Doehner, W.; Heymsfield, S.B.; et al. Sarcopenia: A Time for Action. An SCWD Position Paper. J. Cachexia Sarcopenia Muscle 2019, 10, 956–961. [Google Scholar] [CrossRef]
- Rinninella, E.; Cintoni, M.; De Lorenzo, A.; Anselmi, G.; Gagliardi, L.; Addolorato, G.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. May nutritional status worsen during hospital stay? A sub-group analysis from a cross-sectional study. Intern. Emerg. Med. 2019, 14, 51–57. [Google Scholar] [CrossRef]
- Arends, J.; Bachmann, P.; Baracos, V.; Barthelemy, N.; Bertz, H.; Bozzetti, F.; Fearon, K.; Hütterer, E.; Isenring, E.; Kaasa, S.; et al. ESPEN guidelines on nutrition in cancer patients. Clin. Nutr. 2017, 36, 11–48. [Google Scholar] [CrossRef]
- Mourtzakis, M.; Prado, C.M.; Lieffers, J.R.; Reiman, T.; McCargar, L.J.; Baracos, V.E. A practical and precise approach to quantification of body composition in cancer patients using computed tomography images acquired during routine care. Appl. Physiol. Nutr. Metab. 2008, 33, 997–1006. [Google Scholar] [CrossRef]
- Baracos, V.E. Psoas as a sentinel muscle for sarcopenia: A flawed premise. J. Cachexia Sarcopenia Muscle 2017, 8, 527–528. [Google Scholar] [CrossRef]
- Rutten, I.J.G.; Ubachs, J.; Kruitwagen, R.F.P.M.; Beets-Tan, R.G.H.; Olde Damink, S.W.M.; Van Gorp, T. Psoas muscle area is not representative of total skeletal muscle area in the assessment of sarcopenia in ovarian cancer. J. Cachexia Sarcopenia Muscle 2017, 8, 630–638. [Google Scholar] [CrossRef]
- Schuetz, P.; Fehr, R.; Baechli, V.; Geiser, M.; Deiss, M.; Gomes, F.; Kutz, A.; Tribolet, P.; Bregenzer, T.; Braun, N.; et al. Individualised nutritional support in medical inpatients at nutritional risk: A randomised clinical trial. Lancet 2019, 393, 2312–2321. [Google Scholar] [CrossRef]
- Kaegi-Braun, N.; Tribolet, P.; Gomes, F.; Fehr, R.; Baechli, V.; Geiser, M.; Deiss, M.; Kutz, A.; Bregenzer, T.; Hoess, C.; et al. Six-month outcomes after individualized nutritional support during the hospital stay in medical patients at nutritional risk: Secondary analysis of a prospective randomized trial. Clin. Nutr. 2020. S0261-5614(20)30435-0. [Google Scholar] [CrossRef] [PubMed]
- Rinninella, E.; Persiani, R.; D’Ugo, D.; Pennestrì, F.; Cicchetti, A.; Di Brino, E.; Cintoni, M.; Miggiano, G.A.D.; Gasbarrini, A.; Mele, M.C. NutriCatt protocol in the Enhanced Recovery After Surgery (ERAS) program for colorectal surgery: The nutritional support improves clinical and cost-effectiveness outcomes. Nutrition 2018, 50, 74–81. [Google Scholar] [CrossRef] [PubMed]
- Ardito, F.; Lai, Q.; Rinninella, E.; Mimmo, A.; Vellone, M.; Panettieri, E.; Adducci, E.; Cintoni, M.; Mele, M.C.; Gasbarrini, A.; et al. The impact of personalized nutritional support on postoperative outcome within the enhanced recovery after surgery (ERAS) program for liver resections: Results from the NutriCatt protocol. Updates Surg. 2020. [Google Scholar] [CrossRef]
- Higuera-Pulgar, I.; Ribed, A.; Carrascal-Fabian, M.L.; Romero-Jiménez, R.M.; Velasco-Gimeno, C.; Bretón-Lesmes, I.; Camblor-Álvarez, M.; Cuerda-Compes, C.; García-Peris, P. Evolution of nutritional status and survival in patients with cancer on tyrosine kinase inhibitors treatment. Endocrinol. Diabetes Nutr. 2019, 66, 472–479. [Google Scholar] [CrossRef] [PubMed]
- Antoun, S.; Raynard, B. Muscle protein anabolism in advanced cancer patients: Response to protein and amino acids support, and to physical activity. Ann. Oncol. 2018, 29, ii10–ii17. [Google Scholar] [CrossRef]
- Buse, M.G. In vivo effects of branched chain amino acids on muscle protein synthesis in fasted rats. Horm. Metab. Res. 1981, 13, 502–505. [Google Scholar] [CrossRef]
- Garlick, P.J.; Grant, I. Amino acid infusion increases the sensitivity of muscle protein synthesis in vivo to insulin. Effect of branched chain amino acids. Biochem. J. 1988, 254, 579–584. [Google Scholar] [CrossRef]
- Wolfe, R.R. Branched-chain amino acids and muscle protein synthesis in humans: Myth or reality? J. Int. Soc. Sports Nutr. 2017, 14, 30. [Google Scholar] [CrossRef]
- Nair, K.S.; Schwartz, R.G.; Welle, S. Leucine as a regulator of whole body and skeletal muscle protein metabolism in humans. Am. J. Physiol. 1992, 263, E928–E934. [Google Scholar] [CrossRef]
- Tayek, J.A.; Bistrian, B.R.; Hehir, D.J.; Martin, R.; Moldawer, L.L.; Blackburn, G.L. Improved protein kinetics and albumin synthesis by branched chain amino acid-enriched total parenteral nutrition in cancer cachexia. A prospective randomized crossover trial. Cancer 1986, 58, 147–157. [Google Scholar] [CrossRef]
- Hunter, D.C.; Weintraub, M.; Blackburn, G.L.; Bistrian, B.R. Branched chain amino acids as the protein component of parenteral nutrition in cancer cachexia. Br. J. Surg. 1989, 76, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Takeda, H.; Nishikawa, H.; Iguchi, E.; Ohara, Y.; Sakamoto, A.; Saito, S.; Nishijima, N.; Nasu, A.; Komekado, H.; Kita, R.; et al. Effect of treatment with branched-chain amino acids during sorafenib therapy for unresectable hepatocellular carcinoma. Hepatol. Res. 2014, 44, 302–312. [Google Scholar] [CrossRef] [PubMed]
- Lam, V.W.; Poon, R.T. Role of branched-chain amino acids in management of cirrhosis and hepatocellular carcinoma. Hepatol. Res. 2008, 38, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Takeshita, S.; Ichikawa, T.; Nakao, K.; Miyaaki, H.; Shibata, H.; Matsuzaki, T.; Muraoka, T.; Honda, T.; Otani, M.; Akiyama, M.; et al. A snack enriched with oral branched-chain amino acids prevents a fall in albumin in patients with liver cirrhosis undergoing chemoembolization for hepatocellular carcinoma. Nutr. Res. 2009, 29, 89–93. [Google Scholar] [CrossRef]
- Morihara, D.; Iwata, K.; Hanano, T.; Kunimoto, H.; Kuno, S.; Fukunaga, A.; Yotsumoto, K.; Takata, K.; Tanaka, T.; Sakurai, K.; et al. Late-evening snack with branched-chain amino acids improves liver function after radiofrequency ablation for hepatocellular carcinoma. Hepatol. Res. 2012, 42, 658–667. [Google Scholar] [CrossRef]
- Ishikawa, T.; Michitaka, I.; Kamimura, H.; Higuchi, K.; Kubota, T.; Seki, K.; Ohta, H.; Yoshida, T.; Kamimura, T. Oral branched-chain amino acids administration improves impaired liver dysfunction after radiofrequency ablation therapy for hepatocellular carcinoma. Hepatogastroenterology 2009, 56, 1491–1495. [Google Scholar]
- Kimball, S.R.; Jefferson, L.S. Amino acids as regulators of gene expression. Nutr. Metab. 2004, 17. [Google Scholar] [CrossRef]
- Nair, K.S.; Short, K.R. Hormonal and signaling role of branched-chain amino acids. J. Nutr. 2005, 135, 1547S–1552S. [Google Scholar] [CrossRef]
- Rinninella, E.; Cerrito, L.; Spinelli, I.; Cintoni, M.; Mele, M.C.; Pompili, M.; Gasbarrini, A. Chemotherapy for hepatocellular carcinoma: Current evidence and future perspectives. J. Clin. Transl. Hepatol. 2017, 5, 235–248. [Google Scholar] [CrossRef]
- May, P.E.; Barber, A.; D’Olimpio, J.T.; Hourihane, A.; Abumrad, N.N. Reversal of cancer-related wasting using oral supplementation with a combination of beta-hydroxy-beta-methylbutyrate, arginine, and glutamine. Am. J. Surg. 2002, 183, 471–479. [Google Scholar] [CrossRef]
| TKI Treatment | First Author, Year of Publication, Country | Study Design | Cancer Type | Sample Size | Gender (M/F) | Age (Years) Median (IQR) or Mean ± SD | % Sarcopenic * Patients at Baseline | Method of Muscle Mass Assessment | Outcomes | Comparison | Mean ± SD or Median or % | p-Value |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Axitinib | Chéry, 2017, USA [26] | R | Advanced non-metastatic RCC | 24 | 18/5 | 60 (51.5–66) | 30.4 | L3 SMI (cm2/m2) calculated from CT scan | Median (IQR) | Baseline vs. 12 weeks after starting treatment | −2.9 (1.7–6.2) | <0.001 |
| Lenvatinib | Rinninella, 2019, Italy [27] | CR | Advanced HCC | 2 | 2/0 | 61 case 1 83 case 2 | NR | L3 SMA (cm2) measured from CT scan | % SMA changes | Baseline vs. 24 months after starting treatment | Case 1: −2.13 Case 2: −10.83 | NA |
| Lenvatinib | Hiraoka, 2019, Japan [28] | R | Advanced HCC | 51 | 59/18 | 72.0 ± 8.9 | NR | PI (cm2/m2) calculated from CT scan | Mean difference ± SD | Baseline vs. 4 weeks after starting treatment | −0.210 ± 0.315 | NR |
| Baseline vs. 12 weeks after starting treatment | −0.275 ± 0.372 | NR | ||||||||||
| Regorafenib | Huemer, 2019, Austria [29] | R | Metastatic colorectal cancer | 22 | 11/11 | 59 (42–74) | 54 | L3 SMI (cm2/m2) measured from CT scan | Mean difference | Baseline vs. after initiating treatment | −2.75 ± NR | <0.0001 |
| Regorafenib | Bekir, 2020, Turkey [30] | R | Metastatic colorectal cancer | 36 | 18/18 | 62 (52–69) | NR | L3 SMA (cm2) calculated from CT scan | Median (IQR) | Baseline vs. after initiating treatment | −7.8 (−13.9; −4.8) | 0.001 |
| Sorafenib | Antoun, 2010, France [31] | RCT | Advanced RCC | 80 | 60/20 | 59.8 (38–78) | 52.5 | L3 SMA (cm2) calculated from CT scan | % SMA changes | Baseline vs. 6 months after starting treatment | −4.9 | <0.01 |
| % SMA changes | Baseline vs. 12 months after starting treatment | −8 | <0.01 | |||||||||
| Mean difference ± SD | Placebo vs. sorafenib groups at month-6 | −3.1 ± 1.3 vs. −7.4 ± 1.7 | 0.02 | |||||||||
| Sorafenib | Huillard, 2019, France [32] | R | Advanced differentiated thyroid cancer | 365 | NR | 63 (24–82) | 49.4 | LBM (kg) estimated from L3 SMI calculated from CT scan | Mean difference | Placebo vs. sorafenib groups at month-6 after starting treatment | −0.1 vs. −3.0 | <0.0001 |
| Sorafenib or lenvatinib | Uchikawa, 2020, Japan [33] | R | Advanced HCC | 67 (49/18) | 56/11 | 70 (20–87) | NR | L3 SMI (cm2/m2) calculated from CT scan | Median | Before TKI treatment vs. 1–3 months afterward | 45.3 (before TKI treatment) 42.1 (after treatment) | ≤0.01(1) 0.025(2) |
| Sunitinib or Sorafenib or others | Gu, 2017, China [34] | R | Metastatic RCC | 101 (30/45/26) | 65/36 | 59.6 ± 12.8 | 35.6 | L3 SMI (cm2/m2) | Mean difference | Baseline vs. 4 months after starting treatment | −1.7 ± NR | NR |
| Skeletal muscle density (HU) was calculated from CT scan. | Mean difference | Baseline vs. 4 months after starting treatment | −1.9 ± NR | R | ||||||||
| Sunitinib or Pazopanib | Köstek, 2019, Turkey [35] | R | Metastatic RCC | 36 (18/18) | 25/11 | 60 (49–68) | NR | LBM (kg) estimated from L3 SMA calculated from CT scan | Median change (IQR) | Baseline vs. 4 months after starting sunitinib treatment | −5.6 (−1.2; −10.1) | 0.02 |
| Baseline vs. 4 months after starting pazopanib treatment | −0.3 (−4.1; −1.0) | NS | ||||||||||
| Vandetanib | Massicotte, 2013, France [36] | Controlled trial | Advanced medullary thyroid carcinoma | 23 | 16/7 | 51 (27–69) | NR | L3 SMI (cm2/m2) calculated from CT scan | Mean difference ± SD | Placebo vs. vandetanib group at month-3 after starting treatment | −1.0 ± 2.0 vs. 1.3 ± 2.1 | 0.009 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rinninella, E.; Cintoni, M.; Raoul, P.; Pozzo, C.; Strippoli, A.; Ponziani, F.R.; Pompili, M.; Bria, E.; Tortora, G.; Gasbarrini, A.; et al. Skeletal Muscle Loss during Multikinase Inhibitors Therapy: Molecular Pathways, Clinical Implications, and Nutritional Challenges. Nutrients 2020, 12, 3101. https://doi.org/10.3390/nu12103101
Rinninella E, Cintoni M, Raoul P, Pozzo C, Strippoli A, Ponziani FR, Pompili M, Bria E, Tortora G, Gasbarrini A, et al. Skeletal Muscle Loss during Multikinase Inhibitors Therapy: Molecular Pathways, Clinical Implications, and Nutritional Challenges. Nutrients. 2020; 12(10):3101. https://doi.org/10.3390/nu12103101
Chicago/Turabian StyleRinninella, Emanuele, Marco Cintoni, Pauline Raoul, Carmelo Pozzo, Antonia Strippoli, Francesca Romana Ponziani, Maurizio Pompili, Emilio Bria, Giampaolo Tortora, Antonio Gasbarrini, and et al. 2020. "Skeletal Muscle Loss during Multikinase Inhibitors Therapy: Molecular Pathways, Clinical Implications, and Nutritional Challenges" Nutrients 12, no. 10: 3101. https://doi.org/10.3390/nu12103101
APA StyleRinninella, E., Cintoni, M., Raoul, P., Pozzo, C., Strippoli, A., Ponziani, F. R., Pompili, M., Bria, E., Tortora, G., Gasbarrini, A., & Mele, M. C. (2020). Skeletal Muscle Loss during Multikinase Inhibitors Therapy: Molecular Pathways, Clinical Implications, and Nutritional Challenges. Nutrients, 12(10), 3101. https://doi.org/10.3390/nu12103101









