Role of Dietary Intake and Serum 25(OH)D on the Effects of a Multicomponent Exercise Program on Bone Mass and Structure of Frail and Pre-Frail Older Adults
Abstract
1. Introduction
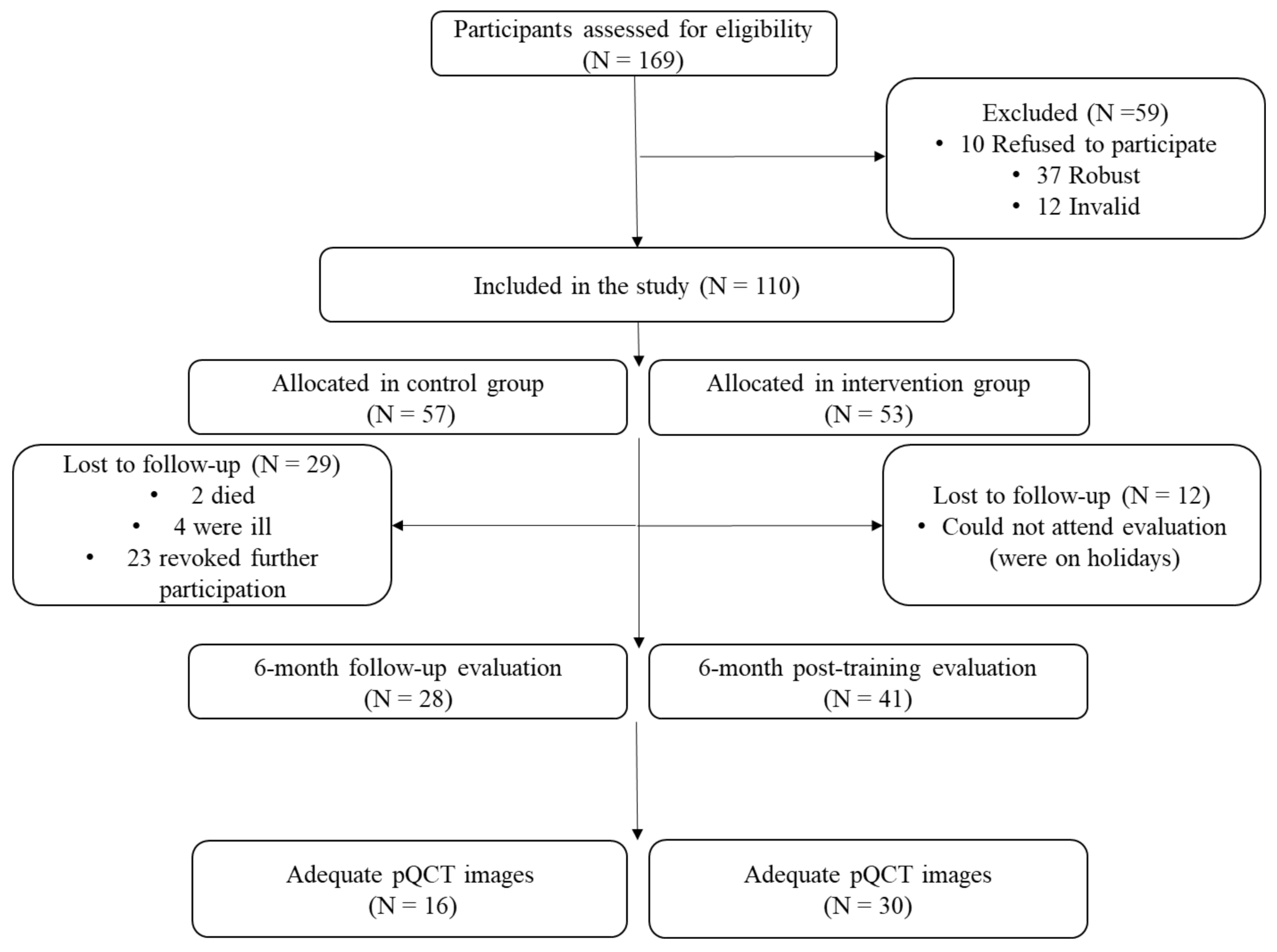
2. Materials and Methods
2.1. Study Design and Participants
2.2. Ethics Statement
2.3. The EXERNET Elder 3.0 Multicomponent Exercise Program
2.4. Peripheral Quantitative Computed Tomography (pQCT)
2.5. Dietary Intake
2.6. Blood Samples and Serum 25(OH)D
2.7. Statistical Analysis
3. Results
3.1. Effects of the MCT Program on Bone Mass and Structure
3.2. Dietary Intake Descriptive Variables and Their Contribution to Changes in Bone Mass and Structure during the 6-Month Intervention Period
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. World Report on Ageing And Health; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Rodriguez-Mañas, L.; Fried, L.P. Frailty in the clinical scenario. Lancet 2015, 385, e7–e9. [Google Scholar] [CrossRef]
- Bouaziz, W.; Lang, P.O.; Schmitt, E.; Kaltenbach, G.; Geny, B.; Vogel, T. Health benefits of multicomponent training programmes in seniors: A systematic review. Int. J. Clin. Pract. 2016, 70, 520–536. [Google Scholar] [CrossRef] [PubMed]
- Klein, B.E.K.; Klein, R.; Knudtson, M.D.; Lee, K.E. Frailty, morbidity and survival. Arch. Gerontol. Geriatr. 2005, 41, 141–149. [Google Scholar] [CrossRef] [PubMed]
- Angulo, J.; El Assar, M.; Bustos, A.Á.; Rodríguez-Mañas, L. Physical activity and exercise: Strategies to manage frailty. Redox Biol. 2020, 101513. [Google Scholar] [CrossRef]
- Farr, J.N.; Khosla, S. Skeletal changes through the lifespan—From growth to senescence. Nat. Rev. Endocrinol. 2015, 11, 513–521. [Google Scholar] [CrossRef]
- Siris, E.S.; Adler, R.; Bilezikian, J.; Bolognese, M.; Dawson-Hughes, B.; Favus, M.J.; Harris, S.T.; Jan de Beur, S.M.; Khosla, S.; Lane, N.E.; et al. The clinical diagnosis of osteoporosis: A position statement from the National Bone Health Alliance Working Group. Osteoporos. Int. 2014, 25, 1439–1443. [Google Scholar] [CrossRef]
- Li, G.; Thabane, L.; Papaioannou, A.; Ioannidis, G.; Levine, M.A.H.; Adachi, J.D. An overview of osteoporosis and frailty in the elderly. BMC Musculoskelet. Disord. 2017, 18, 46. [Google Scholar] [CrossRef]
- Santos, L.; Elliott-Sale, K.J.; Sale, C. Exercise and bone health across the lifespan. Biogerontology 2017, 18, 931–946. [Google Scholar] [CrossRef]
- Gómez-Cabello, A.; Ara, I.; González-Agüero, A.; Casajús, J.A.; Vicente-Rodríguez, G. Effects of Training on Bone Mass in Older Adults. Sports Med. 2012, 42, 301–325. [Google Scholar] [CrossRef]
- Cauley, J.A.; Giangregorio, L. Physical activity and skeletal health in adults. Lancet Diabetes Endocrinol. 2020, 8, 150–162. [Google Scholar] [CrossRef]
- Marín-Cascales, E.; Alcaraz, P.E.; Ramos-Campo, D.J.; Rubio-Arias, J.A. Effects of multicomponent training on lean and bone mass in postmenopausal and older women: A systematic review. Menopause 2018, 25, 346–356. [Google Scholar] [CrossRef] [PubMed]
- Weaver, C.M.; Gordon, C.M.; Janz, K.F.; Kalkwarf, H.J.; Lappe, J.M.; Lewis, R.; O’Karma, M.; Wallace, T.C.; Zemel, B.S. The National Osteoporosis Foundation’s position statement on peak bone mass development and lifestyle factors: A systematic review and implementation recommendations. Osteoporos. Int. 2016, 27, 1281–1386. [Google Scholar] [CrossRef] [PubMed]
- Halfon, M.; Phan, O.; Teta, D. Vitamin D: A review on its effects on muscle strength, the risk of fall, and frailty. BioMed Res. Int. 2015, 2015, 953241. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Chen, W.; Li, D.; Yin, X.; Zhang, X.; Olsen, N.; Zheng, S.G. Vitamin D and Chronic Diseases. Aging Dis. 2017, 8, 346–353. [Google Scholar] [CrossRef]
- Gonzalez-Molero, I.; Morcillo, S.; Valdes, S.; Perez-Valero, V.; Botas, P.; Delgado, E.; Hernandez, D.; Olveira, G.; Rojo, G.; Gutierrez-Repiso, C.; et al. Vitamin D deficiency in Spain: A population-based cohort study. Eur. J. Clin. Nutr. 2011, 65, 321–328. [Google Scholar] [CrossRef]
- Olmos, J.M.; Hernandez, J.L.; Garcia-Velasco, P.; Martinez, J.; Llorca, J.; Gonzalez-Macias, J. Serum 25-hydroxyvitamin D, parathyroid hormone, calcium intake, and bone mineral density in Spanish adults. Osteoporos. Int. 2016, 27, 105–113. [Google Scholar] [CrossRef]
- Palacios, C. The role of nutrients in bone health, from A to Z. Crit. Rev. Food Sci. Nutr. 2006, 46, 621–628. [Google Scholar] [CrossRef]
- Treacy, D.; Hassett, L. The Short Physical Performance Battery. J. Physiother. 2018, 64, 61. [Google Scholar] [CrossRef]
- Lopez-Rodriguez, C.; Laguna, M.; Gomez-Cabello, A.; Gusi, N.; Espino, L.; Villa, G.; Pedrero-Chamizo, R.; Casajus, J.A.; Ara, I.; Aznar, S. Validation of the self-report EXERNET questionnaire for measuring physical activity and sedentary behavior in elderly. Arch. Gerontol. Geriatr. 2017, 69, 156–161. [Google Scholar] [CrossRef]
- Lawton, M.P.; Brody, E.M. Assessment of Older People: Self-Maintaining and Instrumental Activities of Daily Living1. Gerontologist 1969, 9, 179–186. [Google Scholar] [CrossRef]
- Mahoney, F.I. Functional evaluation: The Barthel index. Md. State Med. J. 1965, 14, 61–65. [Google Scholar] [PubMed]
- Guigoz, Y.; Vellas, B.; Garry, P.J. Assessing the nutritional status of the elderly: The Mini Nutritional Assessment as part of the geriatric evaluation. Nutr. Rev. 1996, 54, S59–S65. [Google Scholar] [CrossRef] [PubMed]
- Schroder, H.; Fito, M.; Estruch, R.; Martinez-Gonzalez, M.A.; Corella, D.; Salas-Salvado, J.; Lamuela-Raventos, R.; Ros, E.; Salaverria, I.; Fiol, M.; et al. A short screener is valid for assessing Mediterranean diet adherence among older Spanish men and women. J. Nutr. 2011, 141, 1140–1145. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Ballart, J.D.; Pinol, J.L.; Zazpe, I.; Corella, D.; Carrasco, P.; Toledo, E.; Perez-Bauer, M.; Martinez-Gonzalez, M.A.; Salas-Salvado, J.; Martin-Moreno, J.M. Relative validity of a semi-quantitative food-frequency questionnaire in an elderly Mediterranean population of Spain. Br. J. Nutr. 2010, 103, 1808–1816. [Google Scholar] [CrossRef]
- World Medical Association. World Medical Association Declaration of Helsinki: Ethical principles for medical research involving human subjects. JAMA 2013, 310, 2191–2194. [Google Scholar] [CrossRef]
- Fernández-García, Á.I.; Gómez-Cabello, A.; Moradell, A.; Navarrete-Villanueva, D.; Pérez-Gómez, J.; Ara, I.; González-Gross, M.; Subías-Perié, J.; Muniz-Pardos, B.; Jose, A.; et al. How to improve the functional capacity of frail and pre-frail elderly people? Health, nutritional status and exercise intervention. The EXERNET-Elder 3.0 project. Sustainability 2020, 12, 6246. [Google Scholar]
- Stagi, S.; Cavalli, L.; Cavalli, T.; de Martino, M.; Brandi, M.L. Peripheral quantitative computed tomography (pQCT) for the assessment of bone strength in most of bone affecting conditions in developmental age: A review. Ital. J. Pediatr. 2016, 42, 88. [Google Scholar] [CrossRef]
- Gonzalez-Aguero, A.; Vicente-Rodriguez, G.; Gomez-Cabello, A.; Casajus, J.A. Cortical and trabecular bone at the radius and tibia in male and female adolescents with Down syndrome: A peripheral quantitative computed tomography (pQCT) study. Osteoporos. Int. 2013, 24, 1035–1044. [Google Scholar] [CrossRef]
- Blew, R.M.; Lee, V.R.; Farr, J.N.; Schiferl, D.J.; Going, S.B. Standardizing evaluation of pQCT image quality in the presence of subject movement: Qualitative versus quantitative assessment. Calcif. Tissue Int. 2014, 94, 202–211. [Google Scholar] [CrossRef]
- Martin-Moreno, J.M.; Boyle, P.; Gorgojo, L.; Maisonneuve, P.; Fernandez-Rodriguez, J.C.; Salvini, S.; Willett, W.C. Development and validation of a food frequency questionnaire in Spain. Int. J. Epidemiol. 1993, 22, 512–519. [Google Scholar] [CrossRef]
- Moreiras, O.; Carbajal, A.; Cabrera, L.; Cuadrado, C. Tablas de Composición de los Alimentos; GUÍA de Prácticas; Ediciones Pirámide: Madrid, Spain, 2015. [Google Scholar]
- Mataix, J. Tabla de Composición de Alimentos, 5th ed.; Universidad de Granada: Granada, Spain, 2009. [Google Scholar]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Academic press: Cambridge, MA, USA, 2013; ISBN 1483276481. [Google Scholar]
- Chen, H.; Zhou, X.; Fujita, H.; Onozuka, M.; Kubo, K.-Y. Age-Related Changes in Trabecular and Cortical Bone Microstructure. Int. J. Endocrinol. 2013, 2013, 213234. [Google Scholar] [CrossRef] [PubMed]
- Tolomio, S.; Ermolao, A.; Lalli, A.; Zaccaria, M. The Effect of a Multicomponent Dual-Modality Exercise Program Targeting Osteoporosis on Bone Health Status and Physical Function Capacity of Postmenopausal Women. J. Women Aging 2010, 22, 241–254. [Google Scholar] [CrossRef] [PubMed]
- Karinkanta, S.; Heinonen, A.; Sievänen, H.; Uusi-Rasi, K.; Pasanen, M.; Ojala, K.; Fogelholm, M.; Kannus, P. A multi-component exercise regimen to prevent functional decline and bone fragility in home-dwelling elderly women: Randomized, controlled trial. Osteoporos. Int. 2007, 18, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Marín-Cascales, E.; Alcaraz, P.E.; Rubio-Arias, J.A. Effects of 24 Weeks of Whole Body Vibration Versus Multicomponent Training on Muscle Strength and Body Composition in Postmenopausal Women: A Randomized Controlled Trial. Rejuvenation Res. 2017, 20, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Vicente-Rodríguez, G.; Ezquerra, J.; Mesana, M.I.; Fernández-Alvira, J.M.; Rey-López, J.P.; Casajus, J.A.; Moreno, L.A. Independent and combined effect of nutrition and exercise on bone mass development. J. Bone Miner. Metab. 2008, 26, 416–424. [Google Scholar] [CrossRef]
- Julián-Almárcegui, C.; Gómez-Cabello, A.; Huybrechts, I.; González-Agüero, A.; Kaufman, J.M.; Casajús, J.A.; Vicente-Rodríguez, G. Combined effects of interaction between physical activity and nutrition on bone health in children and adolescents: A systematic review. Nutr. Rev. 2015, 73, 127–139. [Google Scholar] [CrossRef]
- Laskou, F.; Dennison, E. Interaction of Nutrition and Exercise on Bone and Muscle. Eur. Endocrinol. 2019, 15, 11–12. [Google Scholar] [CrossRef]
- Klein, W.M.P.; Jacobsen, P.B.; Helzlsouer, K.J. Alcohol and cancer risk: Clinical and research implications. JAMA 2020, 323, 23–24. [Google Scholar] [CrossRef]
- Luo, Z.; Liu, Y.; Liu, Y.; Chen, H.; Shi, S.; Liu, Y. Cellular and molecular mechanisms of alcohol-induced osteopenia. Cell. Mol. Life Sci. CMLS 2017, 74, 4443–4453. [Google Scholar] [CrossRef]
- Järvinen, R.; Tuppurainen, M.; Erkkilä, A.T.; Penttinen, P.; Kärkkäinen, M.; Salovaara, K.; Jurvelin, J.S.; Kröger, H. Associations of dietary polyunsaturated fatty acids with bone mineral density in elderly women. Eur. J. Clin. Nutr. 2012, 66, 496–503. [Google Scholar] [CrossRef]
- Virtanen, J.K.; Mozaffarian, D.; Willett, W.C.; Feskanich, D. Dietary intake of polyunsaturated fatty acids and risk of hip fracture in men and women. Osteoporos. Int. 2012, 23, 2615–2624. [Google Scholar] [CrossRef] [PubMed]
- Navarro-Valverde, C.; Caballero-Villarraso, J.; Mata-Granados, J.M.; Casado-Díaz, A.; Sosa-Henríquez, M.; Malouf-Sierra, J.; Nogués-Solán, X.; Rodríguez-Mañas, L.; Cortés-Gil, X.; Delgadillo-Duarte, J.; et al. High Serum Retinol as a Relevant Contributor to Low Bone Mineral Density in Postmenopausal Osteoporotic Women. Calcif. Tissue Int. 2018, 102, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Tanumihardjo, S.A. Vitamin A and bone health: The balancing act. J. Clin. Densitom. Off. J. Int. Soc. Clin. Densitom. 2013, 16, 414–419. [Google Scholar] [CrossRef] [PubMed]
- de Jonge, E.A.L.; Kiefte-de Jong, J.C.; Campos-Obando, N.; Booij, L.; Franco, O.H.; Hofman, A.; Uitterlinden, A.G.; Rivadeneira, F.; Zillikens, M.C. Dietary vitamin A intake and bone health in the elderly: The Rotterdam Study. Eur. J. Clin. Nutr. 2015, 69, 1360–1368. [Google Scholar] [CrossRef]
- Uusi-Rasi, K.; Patil, R.; Karinkanta, S.; Kannus, P.; Tokola, K.; Lamberg-Allardt, C.; Sievänen, H. Exercise and Vitamin D in Fall Prevention Among Older Women: A Randomized Clinical Trial. JAMA Intern. Med. 2015, 175, 703–711. [Google Scholar] [CrossRef]
- Uusi-Rasi, K.; Patil, R.; Karinkanta, S.; Kannus, P.; Tokola, K.; Lamberg-Allardt, C.; Sievänen, H. A 2-Year Follow-Up After a 2-Year RCT with Vitamin D and Exercise: Effects on Falls, Injurious Falls and Physical Functioning Among Older Women. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2017, 72, 1239–1245. [Google Scholar] [CrossRef]
- Bruyère, O.; Cavalier, E.; Buckinx, F.; Reginster, J.-Y. Relevance of vitamin D in the pathogenesis and therapy of frailty. Curr. Opin. Clin. Nutr. Metab. Care 2017, 20, 26–29. [Google Scholar] [CrossRef]
| CON (n = 16) | TRAIN (n = 30) | p-Value | |
|---|---|---|---|
| Age and Anthropometrics | |||
| Mean ± SD | Mean ± SD | ||
| Age (years) | 79.1 ± 5.1 | 81.3 ± 5.0 | 0.162 |
| Height (cm) | 153.1 ± 7.2 | 159.3 ± 8.1 | 0.037 |
| Weight (kg) | 67.0 ± 7.2 | 73.8 ± 11.3 | 0.046 |
| BMI (kg/m2) | 29.2 ± 4.3 | 29.3 ± 3.6 | 0.952 |
| Walking hours per day | 2.1 ± 1.5 | 1.0 ± 0.8 | 0.014 |
| Sedentary hours per day | 5.9 ± 2.8 | 6.1 ± 2.6 | 0.907 |
| Sex | n (%) | n (%) | |
| Women | 14 (87.5) | 22 (73.3) | 0.267 |
| Men | 2 (12.5) | 8 (26.7) | |
| SPPB | |||
| Frail | 1 (6.3) | 5 (16.7) | 0.312 |
| Pre-frail | 15 (93.7) | 25 (83.3) | |
| Health related habits | |||
| Smoke | |||
| Yes | 2 (12.5) | 0 (0.0) | 0.220 |
| No | 14 (87.5) | 30 (100.0) | |
| Nutrition related questionnaires | |||
| ADM | |||
| Yes | 11 (68.7) | 23 (76.7) | 0.827 |
| No | 5 (31.3) | 7 (23.3) | |
| MNA | |||
| At risk of malnourishment | 1 (6.3) | 6 (20.0) | 0.291 |
| Normal nutritional status | 15 (93.7) | 24 (80.0) | |
| Dependence questionnaires | |||
| Barthel Index | |||
| Mildly dependendent | 7 (43.7) | 9 (30.0) | 0.348 |
| Independent | 9 (56.3) | 21 (70.0) | |
| IADL | |||
| Moderate dependence | 3 (18.2) | 4 (13.3) | 0.220 |
| Mildly dependendent | 1 (6.3) | 9 (30.0) | |
| Independent | 12 (7.5) | 17 (56.7) | |
| CON | TRAIN | ||||||
|---|---|---|---|---|---|---|---|
| Pre-Training | Post-Training | F | Pre-Training | Post-Training | F | GxT Interaction ηp2 | |
| Tt.BMC 4% (g) | 2.53 ± 0.37 | 2.48 ± 0.37 | 12.297 * | 2.84 ± 0.35 ♦ | 2.80 ± 0.35 ♦ | 5.599 * | 0.046 |
| Tt.BMD 4% (mg/cm3) | 233.50 ± 40.24 | 232.68 ± 41.66 | 0.149 | 250.78 ± 39.81 | 251.37 ± 41.19 | 0.151 | 0.007 |
| Tb.BMD 4% (mg/cm3) | 194.29 ± 39.46 | 190.84 ± 40.52 | 8.435 * | 205.21 ± 39.04 | 202.96 ± 40.09 | 6.875 * | 0.015 |
| Tt.Ar 4% (mm2) | 6.99 ± 0.03 | 6.97 ± 0.03 | 2.060 | 7.03 ± 0.02 | 7.01 ± 0.02 | 2.271 | 0.002 |
| Tt.BMC 38% (g) | 2.98 ± 0.33 | 2.96 ± 0.33 | 4.278 * | 3.14 ± 0.32 | 3.13 ± 0.32 | 1.932 | 0.017 |
| Tt.BMD 38% (mg/cm3) | 796.27 ± 99.97 | 789.78 ± 100.66 | 10.110 * | 810.45 ± 98.66 | 805.93 ± 99.34 | 8.644 * | 0.015 |
| Ct.BMD 38% (mg/cm3) | 1128.50 ± 47.80 | 1124.15 ± 47.82 | 6.665 * | 1144.58 ± 47.35 | 1139.61 ± 47.38 | 12.518 * | 0.001 |
| Ct.Th 38% (mm) | 4.28 ± 0.63 | 4.25 ± 0.63 | 3.289 | 4.47 ± 0.62 | 4.47 ± 0.62 | 0.117 | 0.039 |
| Tot.Ar 38% (mm2) | 5.92 ± 0.02 | 5.93 ± 0.02 | 1.418 | 5.95 ± 0.02 | 5.95 ± 0.02 | 0.120 | 0.014 |
| Fracture Load X 38% (N) | 5385.55 ± 771.14 | 5290.65 ± 855.09 | 0.335 | 5665.81 ± 762.35 | 5552.71 ± 845.33 | 2.088 | 0.003 |
| SSIp 38% (mm3) | 1385.07 ± 190.08 | 1374.73 ± 195.66 | 0.736 | 1478.21 ± 187.59 | 1476.46 ± 193.14 | 0.006 | 0.014 |
| CON | TRAIN | p-Value | |
|---|---|---|---|
| Tt.BMC 4% (g) | −2.4 ± 3.2 | −1.0 ± 1.9 | 0.089 |
| Tt.BMD 4% (mg/cm3) | −0.3 ± 3.8 | 0.1 ± 2.1 | 0.799 |
| Tb.BMD 4% (mg/cm3) | −1.9 ± 2.4 | −1.2 ± 2.4 | 0.360 |
| Tot.Ar 4% (mm2) | −1.9 ± 6.1 | −0.9 ± 3.0 | 0.496 |
| Tt.BMC 38% (g) | −0.6 ± 0.9 | −0.3 ± 1.3 | 0.572 |
| Tt.BMD 38% (mg/cm3) | −0.9 ± 0.7 | −0.3 ± 0.5 | 0.006 |
| Ct.BMD 38% (mg/cm3) | −0.5 ± 0.6 | −0.3 ± 0.5 | 0.463 |
| Ct.Th 38% (mm) | −0.7 ± 0.8 | 0.2 ± 1.1 | 0.012 |
| Tot.Ar 38% (mm2) | 0.4 ± 0.9 | 0.0 ± 1.1 | 0.302 |
| Fracture Load X 38% (N) | −1.3 ± 5.9 | −2.2 ± 6.8 | 0.731 |
| SSIp 38% (mm3) | −0.1 ± 2.3 | −0.1 ± 3.9 | 0.999 |
| CON | TRAIN | p-Value | ||
|---|---|---|---|---|
| Protein (g/kg) | 1.38 ± 0.28 | 1.42 ± 0.40 | 0.743 | |
| Alcohol (g) | 1.10 ± 1.64 | 1.16 ± 1.87 | 0.744 | |
| PUFA (g) | 15.94 ± 7.11 | 20.23 ± 9.38 | 0.134 | |
| Vitamins | RDI (F/M) | |||
| Vit A (µg) | 800/1000 | 1480.26 ± 765.08 | 1583.69 ± 504.71 | 0.601 |
| Vit D (µg) | 20 | 5.65 ± 3.40 | 5.95 ± 3.15 | 0.774 |
| Minerals | ||||
| Ca (mg) | 1200 | 994.86 ± 266.71 | 1176.68 ± 370.07 | 0.102 |
| P (mg) | 700 | 1709.05 ± 393.83 | 1898.33 ± 444.44 | 0.177 |
| 25(OH)D | Normal Values | |||
| Vit D (ng/dL) | >30 | 24.17 ± 3.28 | 27.32 ± 10.71 | 0.392 |
| Tt.BMC 4% (g) | Tt.BMD 4% (mg/cm3) | Tb.BMD 4% (mg/cm3) | Tt.Ar 4% (mm2) | Tt.BMC 38% (g) | Tt.BMD 38% (mg/cm3) | Ct.BMD 38% (mg/cm3) | Ct.Th 38% (mm) | Tt.Ar 38% (mm2) | Fracture Load X 38% (N) | SSIp 38% (mm3) | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Overall r2 b | 0.176 | 0.041 | 0.031 | 0.047 | 0.052 | 0.062 | 0.229 | 0.042 | 0.057 | 0.138 | 0.195 | |
| Calcium (mg) | Change r2 | 0.000 | 0.014 | 0.002 | 0.004 | 0.004 | 0.011 | 0.027 | 0.005 | 0.013 | 0.001 | 0.067 |
| Standardized β | 0.008 | 0.123 | 0.052 | 0.711 | −0.062 | 0.110 | 0.170 | −0.073 | −0.120 | −0.033 | −0.269 | |
| Vitamin D (µg) | Change r2 | 0.079 | 0.036 | 0.085 | 0.063 | 0.005 | 0.001 | 0.008 | 0.009 | 0.004 | 0.009 | 0.000 |
| Standardized β | 0.288 | −0.193 | 0.298 | 0.256 | −0.070 | 0.023 | 0.090 | −0.100 | −0.062 | 0.096 | −0.020 | |
| Vitamin A (µg) | Change r2 | 0.035 | 0.092 | 0.065 | 0.079 | 0.016 | 0.174 | 0.158 | 0.001 | 0.011 | 0.051 | 0.003 |
| Standardized β | 0.189 | −0.306 | 0.256 | 0.283 | −0.131 | −0.425 | −0.405 | −0.030 | 0.109 | 0.227 | −0.052 | |
| Phosphorus (mg) | Change r2 | 0.000 | 0.003 | 0.024 | 0.000 | 0.021 | 0.001 | 0.021 | 0.045 | 0.022 | 0.001 | 0.036 |
| Standardized β | 0.019 | 0.055 | 0.164 | −0.010 | −0.150 | 0.026 | 0.150 | −0.220 | −0.155 | 0.033 | −0.197 | |
| Protein (g/kg) | Change r2 | 0.008 | 0.042 | 0.003 | 0.006 | 0.072 | 0.001 | 0.019 | 0.077 | 0.060 | 0.033 | 0.057 |
| Standardized β | 0.087 | 0.205 | −0.050 | 0.079 | −0.269 | −0.025 | 0.134 | −0.278 | −0.252 | −0.188 | −0.241 | |
| PUFA (g) | Change r2 | 0.003 | 0.097 | 0.005 | 0.091 | 0.085 | 0.028 | 0.001 | 0.062 | 0.061 | 0.021 | 0.002 |
| Standardized β | −0.055 | 0.339 | 0.074 | −0.236 | 0.317 | 0.181 | −0.042 | 0.270 | 0.268 | −0.156 | 0.045 | |
| Alcohol (g) | Change r2 | 0.016 | 0.016 | 0.021 | .026 | 0.033 | 0.104 | 0.024 | 0.030 | 0.000 | 0.149 | 0.002 |
| Standardized β | −0.137 | 0.137 | −0.156 | −0.176 | 0.207 | 0.368 | 0.177 | 0.194 | 0.014 | −0.466 | −0.054 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moradell, A.; Navarrete-Villanueva, D.; Fernández-García, Á.I.; Marín-Puyalto, J.; Gómez-Bruton, A.; Pedrero-Chamizo, R.; Pérez-Gómez, J.; Ara, I.; Casajus, J.A.; Gómez-Cabello, A.; et al. Role of Dietary Intake and Serum 25(OH)D on the Effects of a Multicomponent Exercise Program on Bone Mass and Structure of Frail and Pre-Frail Older Adults. Nutrients 2020, 12, 3016. https://doi.org/10.3390/nu12103016
Moradell A, Navarrete-Villanueva D, Fernández-García ÁI, Marín-Puyalto J, Gómez-Bruton A, Pedrero-Chamizo R, Pérez-Gómez J, Ara I, Casajus JA, Gómez-Cabello A, et al. Role of Dietary Intake and Serum 25(OH)D on the Effects of a Multicomponent Exercise Program on Bone Mass and Structure of Frail and Pre-Frail Older Adults. Nutrients. 2020; 12(10):3016. https://doi.org/10.3390/nu12103016
Chicago/Turabian StyleMoradell, Ana, David Navarrete-Villanueva, Ángel I. Fernández-García, Jorge Marín-Puyalto, Alejandro Gómez-Bruton, Raquel Pedrero-Chamizo, Jorge Pérez-Gómez, Ignacio Ara, Jose A. Casajus, Alba Gómez-Cabello, and et al. 2020. "Role of Dietary Intake and Serum 25(OH)D on the Effects of a Multicomponent Exercise Program on Bone Mass and Structure of Frail and Pre-Frail Older Adults" Nutrients 12, no. 10: 3016. https://doi.org/10.3390/nu12103016
APA StyleMoradell, A., Navarrete-Villanueva, D., Fernández-García, Á. I., Marín-Puyalto, J., Gómez-Bruton, A., Pedrero-Chamizo, R., Pérez-Gómez, J., Ara, I., Casajus, J. A., Gómez-Cabello, A., & Vicente-Rodríguez, G. (2020). Role of Dietary Intake and Serum 25(OH)D on the Effects of a Multicomponent Exercise Program on Bone Mass and Structure of Frail and Pre-Frail Older Adults. Nutrients, 12(10), 3016. https://doi.org/10.3390/nu12103016










