Vitamin D Insufficiency Reduces Grip Strength, Grip Endurance and Increases Frailty in Aged C57Bl/6J Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Serum Analyses
2.3. Dual X-ray Absorptiometry
2.4. Physical Performance Assessments
2.4.1. Rotarod Assessment
2.4.2. Gait Speed Assessment
2.4.3. Open Field Activity
2.4.4. Grip Strength
2.4.5. Inverted Grip Endurance
2.4.6. Treadmill Assessment
2.5. Frailty Determination
2.6. Statistics
3. Results
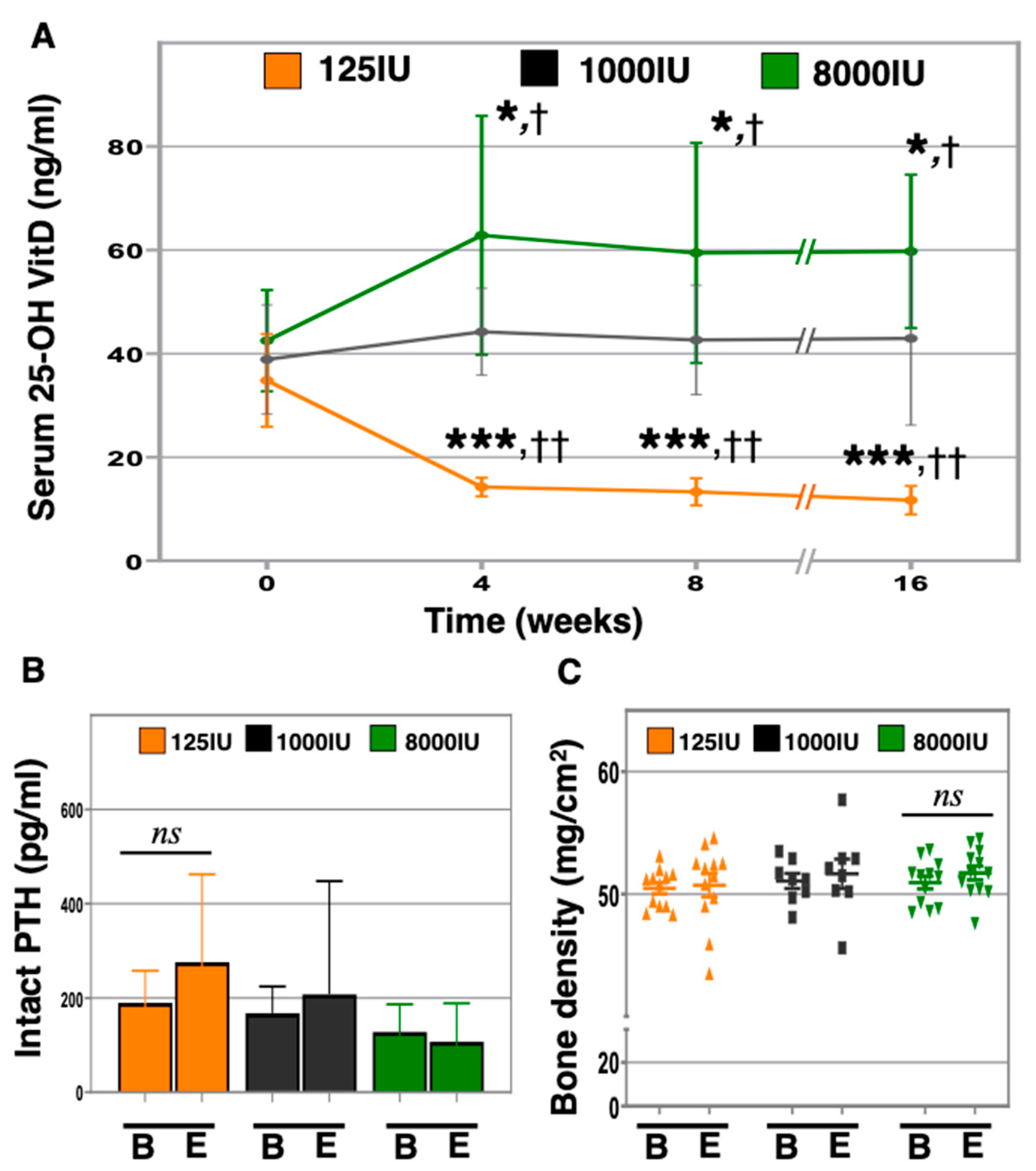
3.1. Changes in Vitamin D Supplementation Rapidly and Consistently Altered 25-OH Vitamin D Serum Levels in Aged Mice, Yet Did Not Affect Serum PTH or Bone Density
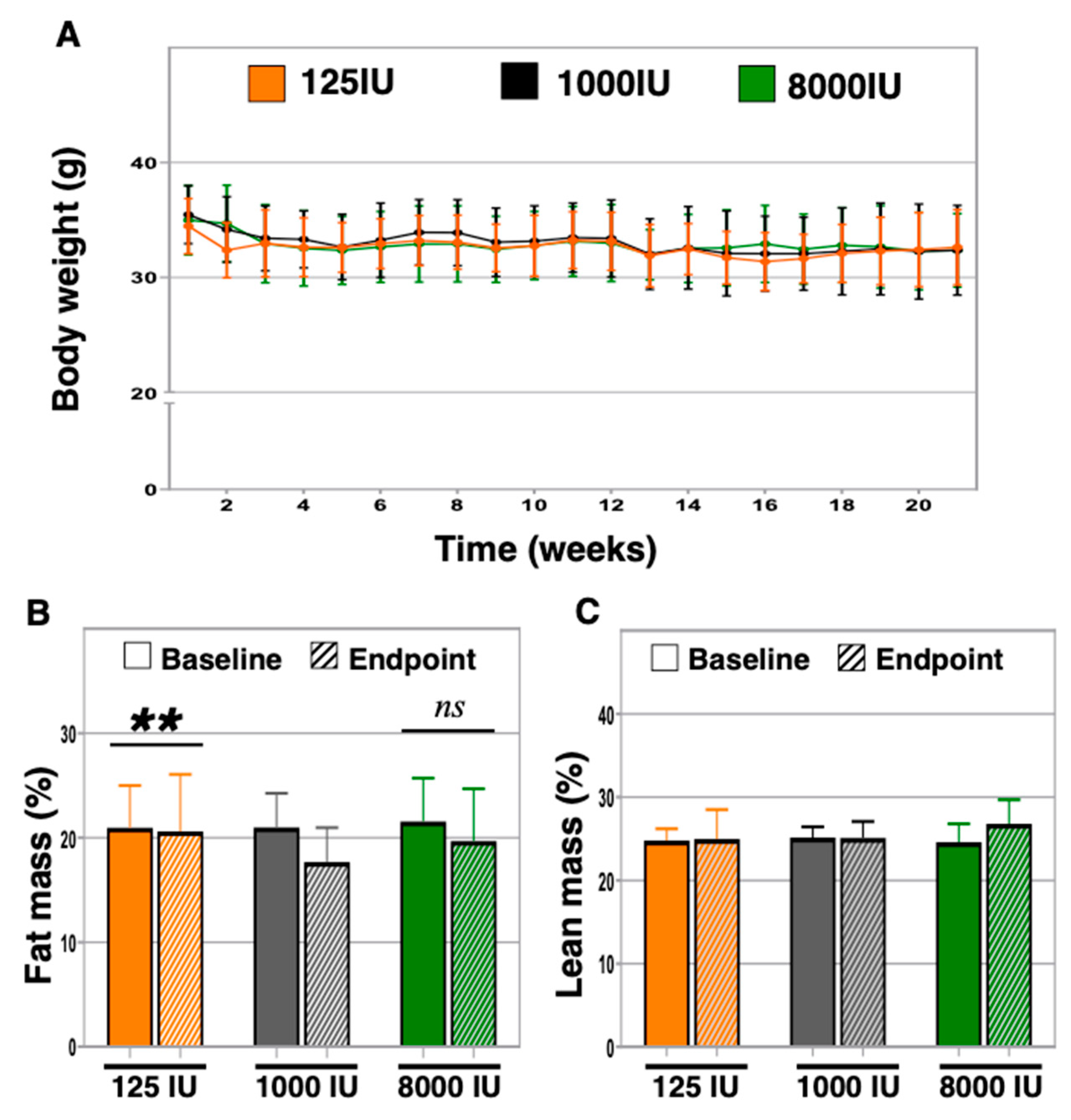
3.2. Serum Levels of 25-OH Vitamin D Did Not Associate with Differences in Body Weight between Groups, but Vitamin D Insufficient Exhibited Increased Body Fat Percentage
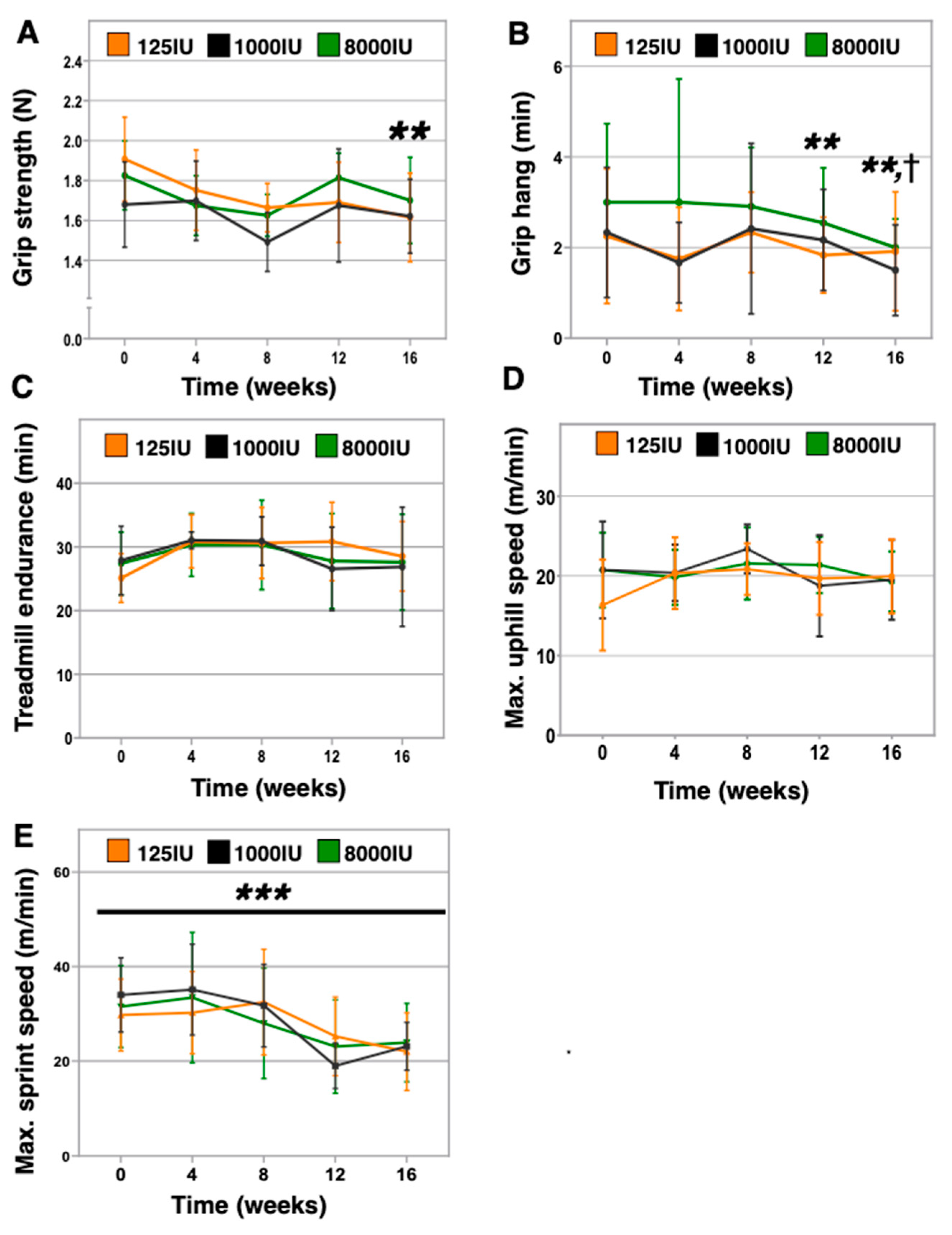
3.3. Vitamin D Insufficient Mice Displayed Lower Grip Strength and Inverted Grip Endurance without Effect on Aerobic or Anaerobic Treadmill Performance
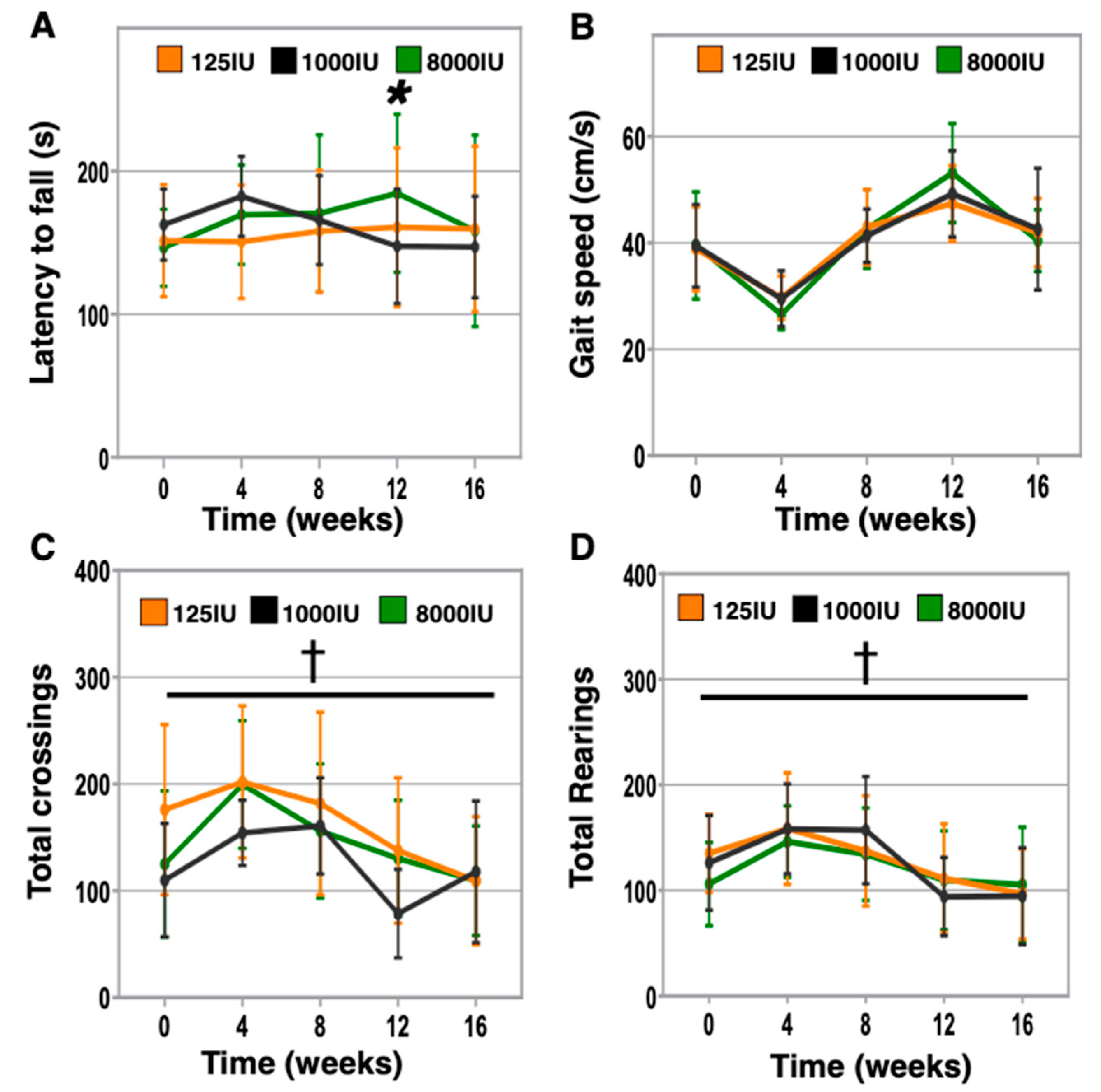
3.4. Vitamin D Hypersufficiency Transiently Improves Balance and Coordination, However Gait Speed and Activity Levels Were Not Affected by Vitamin D Status
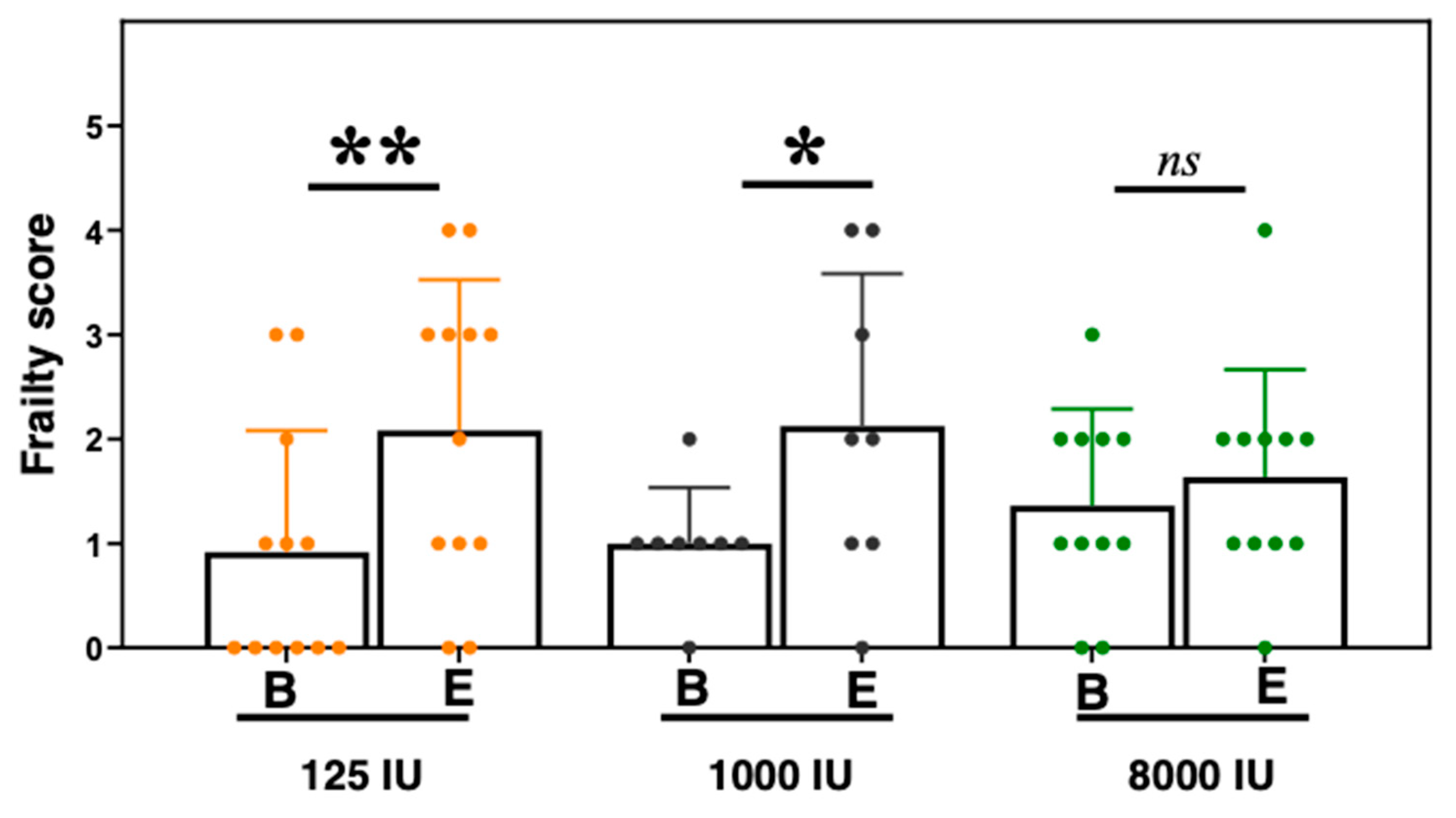
3.5. Frailty Scores Increased in the Vitamin D Insufficient and Sufficient Mice, but Did Not in the Hypersufficient Vitamin D Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Collard, R.M.; Boter, H.; Schoevers, R.A.; Voshaar, R.O. Prevalence of Frailty in Community-Dwelling Older Persons: A Systematic Review. J. Am. Geriatr. Soc. 2012, 60, 1487–1492. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Vaca, J.; de la Rica-Escuin, M.; Silva-Iglesias, M.; Arjonilla-Garcia, M.D.; Varela-Perez, R.; Oliver-Carbonell, J.L.; Abizanda, P. Frailty in INstitutionalized older adults from ALbacete. The FINAL Study: Rationale, design, methodology, prevalence and attributes. Maturitas 2014, 77, 78–84. [Google Scholar] [CrossRef] [PubMed]
- Palacios, C.; Gonzalez, L. Is vitamin D deficiency a major global public health problem? J. Steroid. Biochem. Mol. Biol. 2014, 144, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Halfon, M.; Phan, O.; Teta, D. Vitamin D: A Review on Its Effects on Muscle Strength, the Risk of Fall, and Frailty. Biomed. Res. Int. 2015, 2015, 1–11. [Google Scholar] [CrossRef]
- Boucher, B.J. The problems of vitamin d insufficiency in older people. Aging Dis. 2012, 3, 313–329. [Google Scholar]
- Kammeyer, A.; Luiten, R.M. Oxidation events and skin aging. Ageing Res. Rev. 2015, 21, 16–29. [Google Scholar] [CrossRef]
- Morley, J.E. Frailty and sarcopenia in elderly. Wien Klin Wochenschr. 2016, 128, 439–445. [Google Scholar] [CrossRef]
- Remelli, F.; Vitali, A.; Zurlo, A.; Volpato, S. Vitamin D Deficiency and Sarcopenia in Older Persons. Nutrients 2019, 11, 2861. [Google Scholar] [CrossRef]
- Seldeen, K.L.; Pang, M.; Leiker, M.M.; Bard, J.E.; Rodriguez-Gonzalez, M.; Hernandez, M.; Sheridan, Z.; Nowak, N.; Troen, B.R. Chronic vitamin D insufficiency impairs physical performance in C57BL/6J mice. Aging (Albany N. Y.) 2018, 10, 1338–1355. [Google Scholar] [CrossRef]
- Burne, T.H.; McGrath, J.J.; Eyles, D.W.; Mackay-Sim, A. Behavioural characterization of vitamin D receptor knockout mice. Behav. Brain Res. 2005, 157, 299–308. [Google Scholar] [CrossRef]
- Keisala, T.; Minasyan, A.; Lou, Y.R.; Zou, J.; Kalueff, A.V.; Pyykko, I.; Tuohimaa, P. Premature aging in vitamin D receptor mutant mice. J. Steroid Biochem. Mol. Biol. 2009, 115, 91–97. [Google Scholar] [CrossRef] [PubMed]
- Minasyan, A.; Keisala, T.; Zou, J.; Zhang, Y.; Toppila, E.; Syvala, H.; Lou, Y.R.; Kalueff, A.V.; Pyykko, I.; Tuohimaa, P. Vestibular dysfunction in vitamin D receptor mutant mice. J. Steroid Biochem. Mol. Biol. 2009, 114, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Buchebner, D.; Bartosch, P.; Malmgren, L.; McGuigan, F.E.; Gerdhem, P.; Akesson, K.E. Association Between Vitamin D, Frailty, and Progression of Frailty in Community-Dwelling Older Women. J. Clin. Endocrinol. Metab. 2019, 104, 6139–6147. [Google Scholar] [CrossRef] [PubMed]
- Ju, S.Y.; Lee, J.Y.; Kim, D.H. Low 25-hydroxyvitamin D levels and the risk of frailty syndrome: A systematic review and dose-response meta-analysis. BMC Geriatr. 2018, 18, 206. [Google Scholar] [CrossRef] [PubMed]
- Banga, S.; Heinze-Milne, S.D.; Howlett, S.E. Rodent models of frailty and their application in preclinical research. Mech. Ageing Dev. 2019, 179, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Seldeen, K.L.; Pang, M.; Troen, B.R. Mouse Models of Frailty: An Emerging Field. Curr. Osteoporos. Rep. 2015, 13, 280–286. [Google Scholar] [CrossRef] [PubMed]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef] [PubMed]
- Seldeen, K.L.; Lasky, G.; Leiker, M.M.; Pang, M.; Personius, K.E.; Troen, B.R. High Intensity Interval Training Improves Physical Performance and Frailty in Aged Mice. J. Gerontol. A Biol. Sci. Med. Sci. 2018, 73, 429–437. [Google Scholar] [CrossRef]
- Seldeen, K.L.; Redae, Y.Z.; Thiyagarajan, R.; Berman, R.N.; Leiker, M.M.; Troen, B.R. High intensity interval training improves physical performance in aged female mice: A comparison of mouse frailty assessment tools. Mech. Ageing Dev. 2019, 180, 49–62. [Google Scholar] [CrossRef]
- Justice, J.N.; Carter, C.S.; Beck, H.J.; Gioscia-Ryan, R.A.; McQueen, M.; Enoka, R.M.; Seals, D.R. Battery of behavioral tests in mice that models age-associated changes in human motor function. Age (Dordr.) 2014, 36, 583–592. [Google Scholar] [CrossRef]
- Seldeen, K.L.; Redae, Y.Z.; Thiyagarajan, R.; Berman, R.N.; Leiker, M.M.; Troen, B.R. Short Session High Intensity Interval Training and Treadmill Assessment in Aged Mice. J. Vis. Exp. 2019, e59138. [Google Scholar] [CrossRef]
- Khundmiri, S.J.; Murray, R.D.; Lederer, E. PTH and Vitamin D. Compr. Physiol. 2016, 6, 561–601. [Google Scholar] [PubMed]
- Ekwaru, J.P.; Zwicker, J.D.; Holick, M.F.; Giovannucci, E.; Veugelers, P.J. The importance of body weight for the dose response relationship of oral vitamin D supplementation and serum 25-hydroxyvitamin D in healthy volunteers. PLoS ONE 2014, 9, e111265. [Google Scholar] [CrossRef] [PubMed]
- Khosravi, Z.S.; Kafeshani, M.; Tavasoli, P.; Zadeh, A.H.; Entezari, M.H. Effect of Vitamin D Supplementation on Weight Loss, Glycemic Indices, and Lipid Profile in Obese and Overweight Women: A Clinical Trial Study. Int. J. Prev. Med. 2018, 9, 63. [Google Scholar] [PubMed]
- Dawson-Hughes, B. Vitamin D and muscle function. J. Steroid. Biochem. Mol. Biol. 2017, 173, 313–316. [Google Scholar] [CrossRef]
- Seldeen, K.L.; Pang, M.; Rodriguez-Gonzalez, M.; Hernandez, M.; Sheridan, Z.; Yu, P.; Troen, B.R. A mouse model of vitamin D insufficiency: Is there a relationship between 25(OH) vitamin D levels and obesity? Nutr. Metab. (Lond.) 2017, 14, 26. [Google Scholar] [CrossRef]
- Belenchia, A.M.; Johnson, S.A.; Kieschnick, A.C.; Rosenfeld, C.S.; Peterson, C.A. Time Course of Vitamin D Depletion and Repletion in Reproductive-age Female C57BL/6 Mice. Comp. Med. 2017, 67, 483–490. [Google Scholar]
- Williamson, L.; Hayes, A.; Hanson, E.D.; Pivonka, P.; Sims, N.A.; Gooi, J.H. High dose dietary vitamin D3 increases bone mass and strength in mice. Bone Rep. 2017, 6, 44–50. [Google Scholar] [CrossRef]
- Haslam, A.; Johnson, M.A.; Hausman, D.B.; Cress, M.E.; Houston, D.K.; Davey, A.; Poon, L.W.; Georgia Centenarian, S. Vitamin D status is associated with grip strength in centenarians. J. Nutr. Gerontol. Geriatr. 2014, 33, 35–46. [Google Scholar] [CrossRef]
- Bataille, S.; Landrier, J.F.; Astier, J.; Giaime, P.; Sampol, J.; Sichez, H.; Ollier, J.; Gugliotta, J.; Serveaux, M.; Cohen, J.; et al. The “Dose-Effect” Relationship Between 25-Hydroxyvitamin D and Muscle Strength in Hemodialysis Patients Favors a Normal Threshold of 30 ng/mL for Plasma 25-Hydroxyvitamin D. J. Ren. Nutr. 2016, 26, 45–52. [Google Scholar] [CrossRef]
- Houston, D.K.; Tooze, J.A.; Hausman, D.B.; Johnson, M.A.; Nicklas, B.J.; Miller, M.E.; Neiberg, R.H.; Marsh, A.P.; Newman, A.B.; Blair, S.N.; et al. Change in 25-hydroxyvitamin D and physical performance in older adults. J. Gerontol. A Biol. Sci. Med. Sci. 2011, 66, 430–436. [Google Scholar] [CrossRef] [PubMed]
- Toffanello, E.D.; Perissinotto, E.; Sergi, G.; Zambon, S.; Musacchio, E.; Maggi, S.; Coin, A.; Sartori, L.; Corti, M.C.; Baggio, G.; et al. Vitamin D and physical performance in elderly subjects: The Pro.V.A study. PLoS ONE 2012, 7, e34950. [Google Scholar] [CrossRef]
- Graber, T.G.; Kim, J.H.; Grange, R.W.; McLoon, L.K.; Thompson, L.V. C57BL/6 life span study: Age-related declines in muscle power production and contractile velocity. Age (Dordr.) 2015, 37, 9773. [Google Scholar] [CrossRef] [PubMed]
- Aksoy, M.K.; Altan, L.; Karlibel, I.A. Associations between Handgrip Strength and Vitamin 25(OH)D Levels in Geriatric Patients. Folia Med. (Plovdiv.) 2019, 61, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Scholten, S.D.; Ferley, D.D.; Birger, C.B.; Dowling, C.; Mikkelson, M.; Springer, J.; Lucs, N. Physical performance is not improved with vitamin D repletion: A randomized trial. J. Sports Med. Phys. Fitness 2020, 60, 85–91. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Tanisawa, K.; Zhang, Y.; Ito, T.; Oshima, S.; Higuchi, M.; Cao, Z.B. Effect of Vitamin D Supplementation on Body Composition and Physical Fitness in Healthy Adults: A Double-Blind, Randomized Controlled Trial. Ann. Nutr. Metab. 2019, 75, 231–237. [Google Scholar] [CrossRef] [PubMed]
- Sakai, S.; Suzuki, M.; Tashiro, Y.; Tanaka, K.; Takeda, S.; Aizawa, K.; Hirata, M.; Yogo, K.; Endo, K. Vitamin D receptor signaling enhances locomotive ability in mice. J. Bone Miner. Res. 2015, 30, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Dhaliwal, R.; Aloia, J.F. Effect of Vitamin D on Falls and Physical Performance. Endocrinol. Metab. Clin. N. Am. 2017, 46, 919–933. [Google Scholar] [CrossRef]
- Wu, H.; Pang, Q. The effect of vitamin D and calcium supplementation on falls in older adults: A systematic review and meta-analysis. Orthopade 2017, 46, 729–736. [Google Scholar] [CrossRef]
- Chang, C.I.; Chan, D.C.; Kuo, K.N.; Hsiung, C.A.; Chen, C.Y. Vitamin D insufficiency and frailty syndrome in older adults living in a Northern Taiwan community. Arch. Gerontol. Geriatr. 2010, 50 (Suppl. 1), S17–S21. [Google Scholar] [CrossRef]
- Shardell, M.; Hicks, G.E.; Miller, R.R.; Kritchevsky, S.; Andersen, D.; Bandinelli, S.; Cherubini, A.; Ferrucci, L. Association of low vitamin D levels with the frailty syndrome in men and women. J. Gerontol. A Biol. Sci. Med. Sci. 2009, 64, 69–75. [Google Scholar] [CrossRef] [PubMed]
- Pabst, G.; Zimmermann, A.K.; Huth, C.; Koenig, W.; Ludwig, T.; Zierer, A.; Peters, A.; Thorand, B. Association of low 25-hydroxyvitamin D levels with the frailty syndrome in an aged population: Results from the KORA-age Augsburg study. J. Nutr. Health Aging 2015, 19, 258–264. [Google Scholar] [CrossRef] [PubMed]
- Schottker, B.; Saum, K.U.; Perna, L.; Ordonez-Mena, J.M.; Holleczek, B.; Brenner, H. Is vitamin D deficiency a cause of increased morbidity and mortality at older age or simply an indicator of poor health? Eur. J. Epidemiol. 2014, 29, 199–210. [Google Scholar] [CrossRef] [PubMed]
- Ensrud, K.E.; Ewing, S.K.; Fredman, L.; Hochberg, M.C.; Cauley, J.A.; Hillier, T.A.; Cummings, S.R.; Yaffe, K.; Cawthon, P.M. Study of Osteoporotic Fractures Research G: Circulating 25-hydroxyvitamin D levels and frailty status in older women. J. Clin. Endocrinol. Metab. 2010, 95, 5266–5273. [Google Scholar] [CrossRef]
- Heinze-Milne, S.D.; Banga, S.; Howlett, S.E. Frailty Assessment in Animal Models. Gerontology 2019, 65, 610–619. [Google Scholar] [CrossRef]
- Burt, L.A.; Billington, E.O.; Rose, M.S.; Kremer, R.; Hanley, D.A.; Boyd, S.K. Adverse Effects of High-Dose Vitamin D Supplementation on Volumetric Bone Density Are Greater in Females than Males. J. Bone Miner. Res. 2020. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seldeen, K.L.; Berman, R.N.; Pang, M.; Lasky, G.; Weiss, C.; MacDonald, B.A.; Thiyagarajan, R.; Redae, Y.; Troen, B.R. Vitamin D Insufficiency Reduces Grip Strength, Grip Endurance and Increases Frailty in Aged C57Bl/6J Mice. Nutrients 2020, 12, 3005. https://doi.org/10.3390/nu12103005
Seldeen KL, Berman RN, Pang M, Lasky G, Weiss C, MacDonald BA, Thiyagarajan R, Redae Y, Troen BR. Vitamin D Insufficiency Reduces Grip Strength, Grip Endurance and Increases Frailty in Aged C57Bl/6J Mice. Nutrients. 2020; 12(10):3005. https://doi.org/10.3390/nu12103005
Chicago/Turabian StyleSeldeen, Kenneth Ladd, Reem Nagi Berman, Manhui Pang, Ginger Lasky, Carleara Weiss, Brian Alexander MacDonald, Ramkumar Thiyagarajan, Yonas Redae, and Bruce Robert Troen. 2020. "Vitamin D Insufficiency Reduces Grip Strength, Grip Endurance and Increases Frailty in Aged C57Bl/6J Mice" Nutrients 12, no. 10: 3005. https://doi.org/10.3390/nu12103005
APA StyleSeldeen, K. L., Berman, R. N., Pang, M., Lasky, G., Weiss, C., MacDonald, B. A., Thiyagarajan, R., Redae, Y., & Troen, B. R. (2020). Vitamin D Insufficiency Reduces Grip Strength, Grip Endurance and Increases Frailty in Aged C57Bl/6J Mice. Nutrients, 12(10), 3005. https://doi.org/10.3390/nu12103005





