Protective Effect of α-Linolenic Acid on Non-Alcoholic Hepatic Steatosis and Interleukin-6 and -10 in Wistar Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Material and Diets
2.2. Profile of Fatty Acids and Indices of Nutritional Quality
2.3. Animals and Experimental Design
2.4. Serum Markers
2.5. Assays of Ectonucleotidase Triphosphate Diphosphohydrolase (E-NTPDase) and Ecto-5′-Nucleotidase (E-5′-NT)
2.6. Enzyme-Linked Immunosorbent Assay (ELISA) for Inflammatory Factors
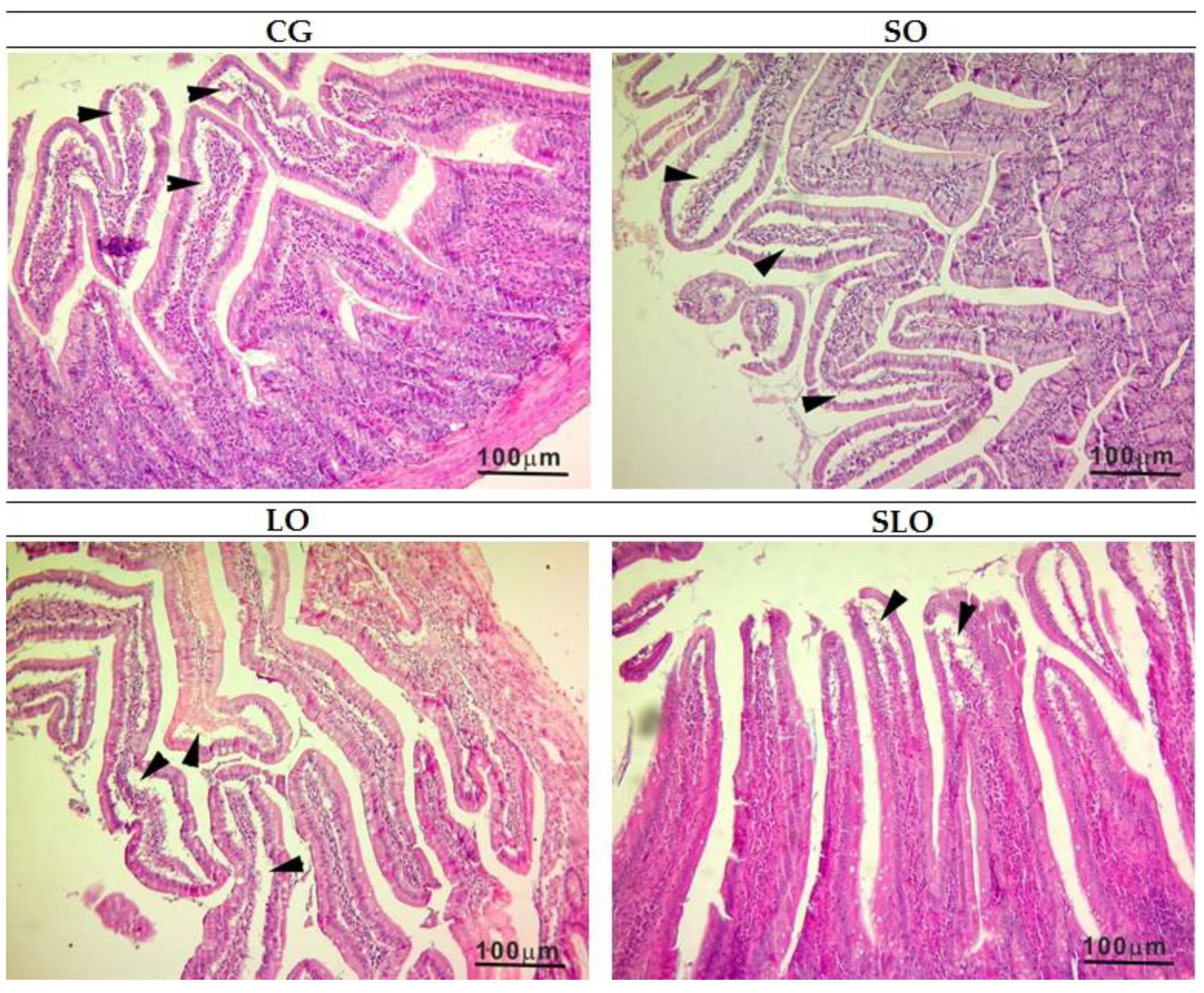
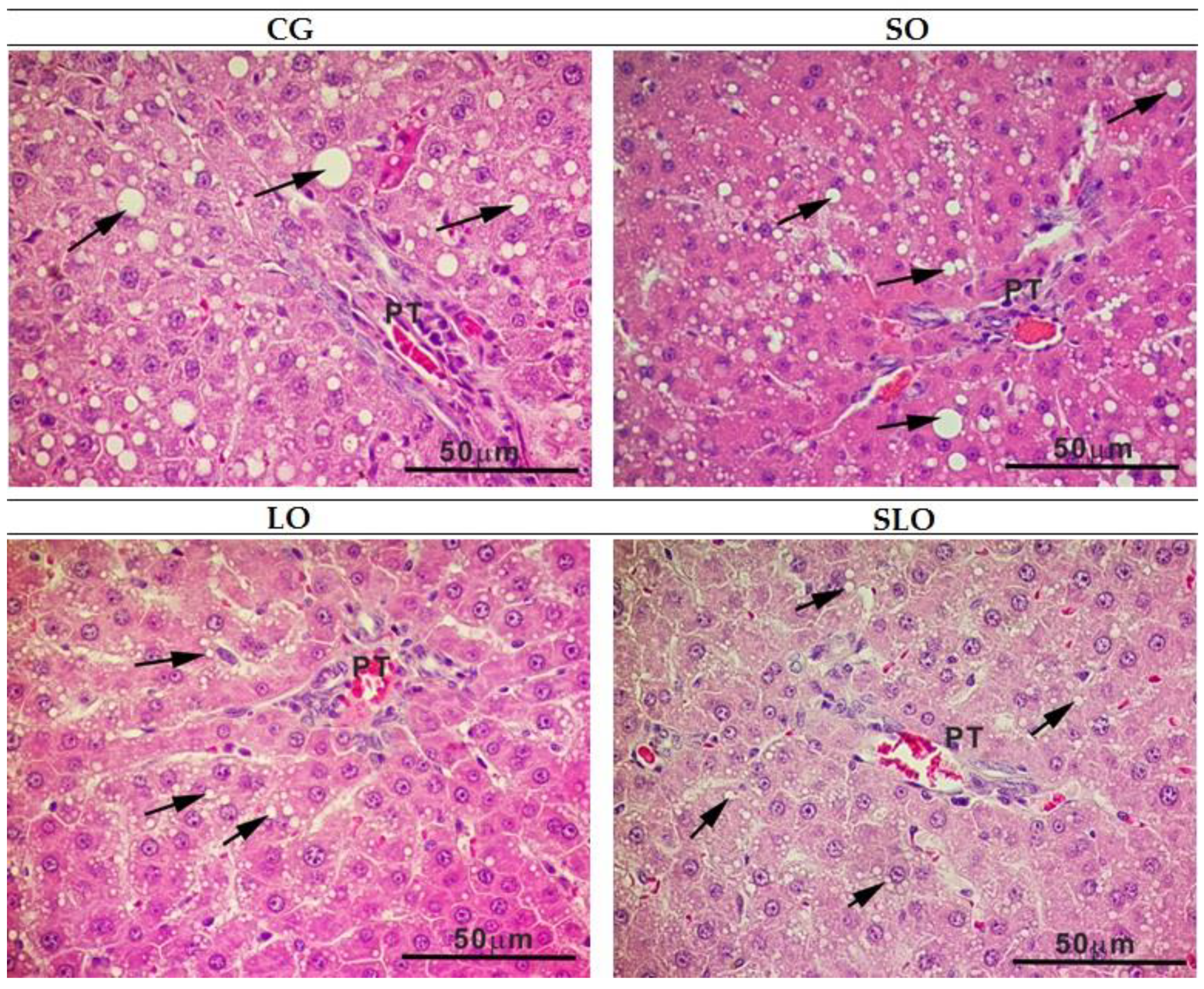
2.7. Histological Analysis
2.8. Statistical Analyses
3. Results
3.1. Profile of Fatty Acids and Indices of Nutritional Quality
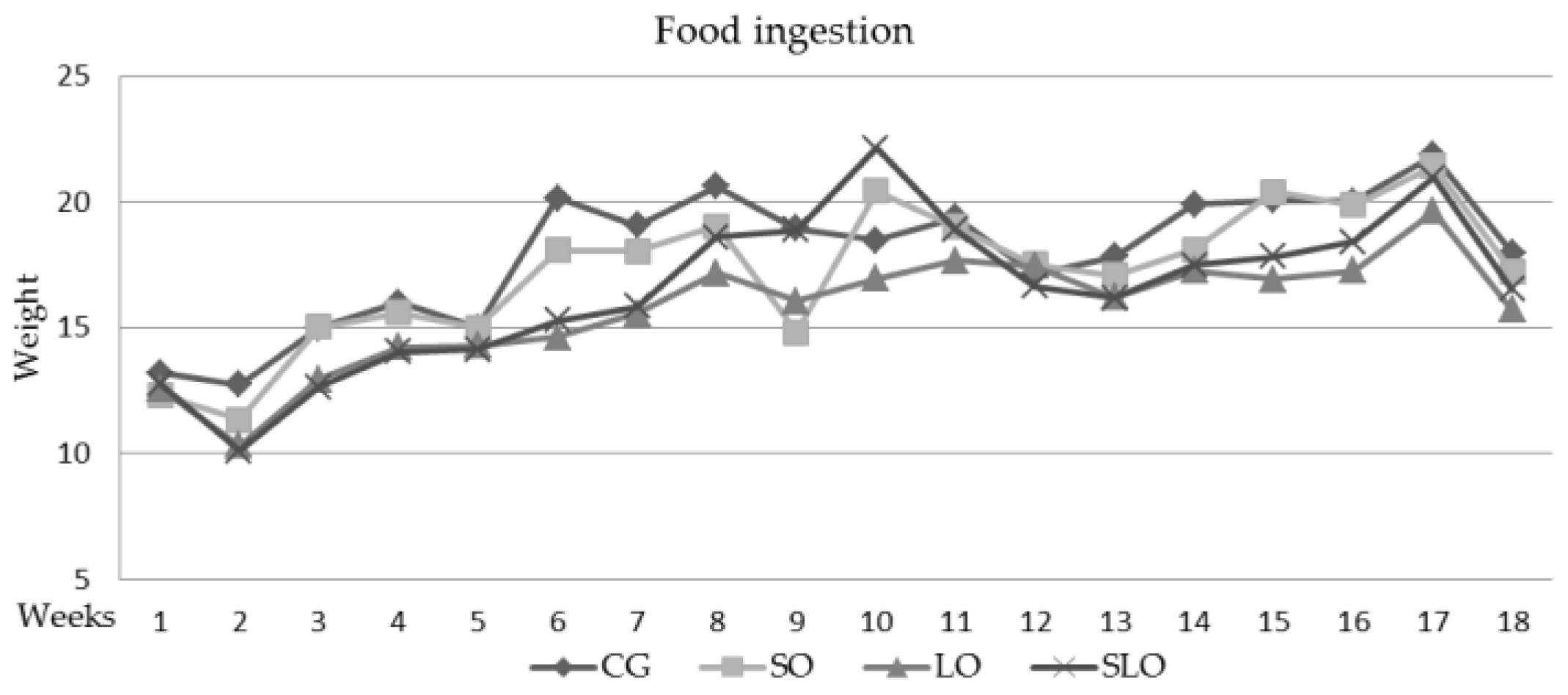
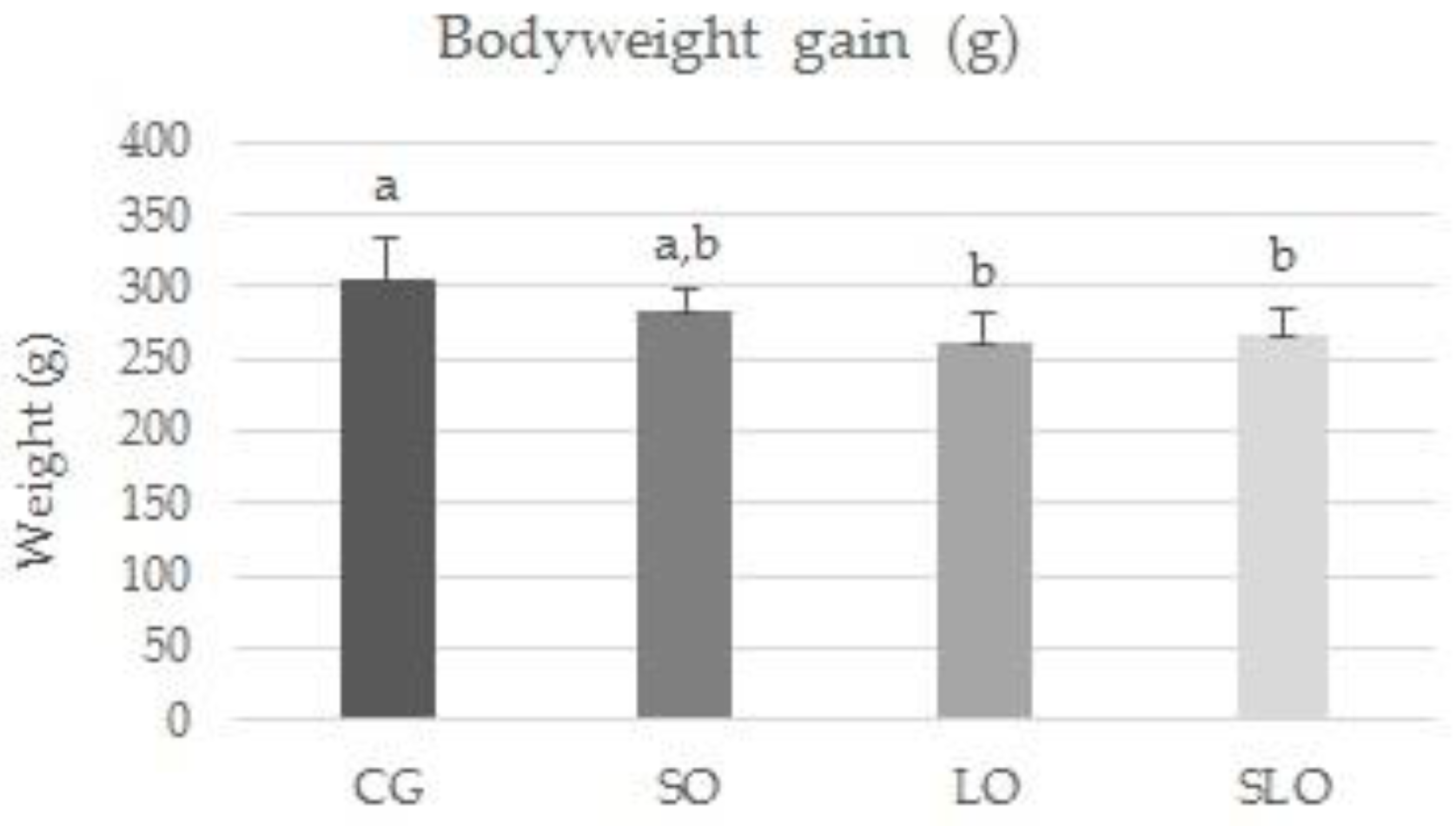
3.2. Ingestion, Weight Gain, Weight of Liver, and Visceral Fats
3.3. Biochemical Parameters
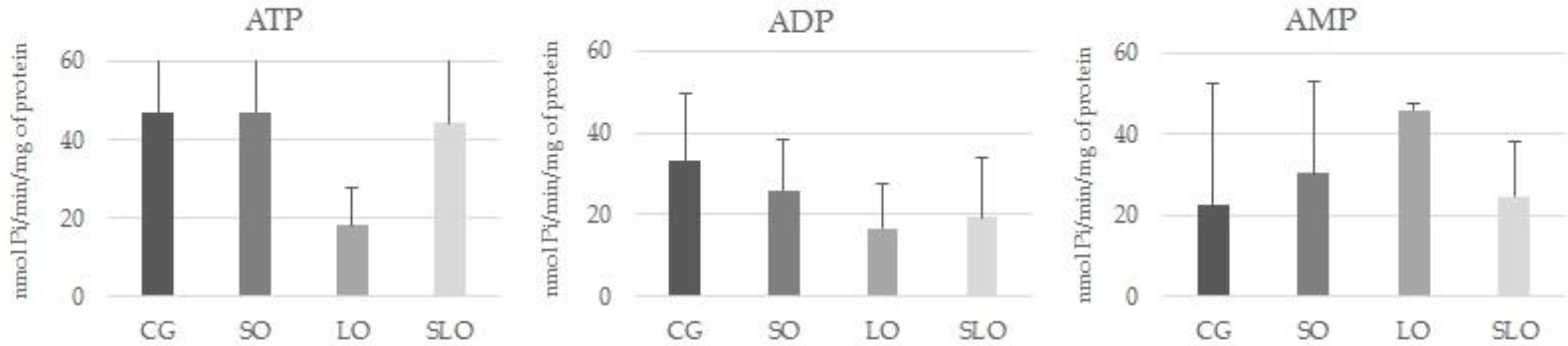
3.4. E-NTPDase and E-5′-NT Assays
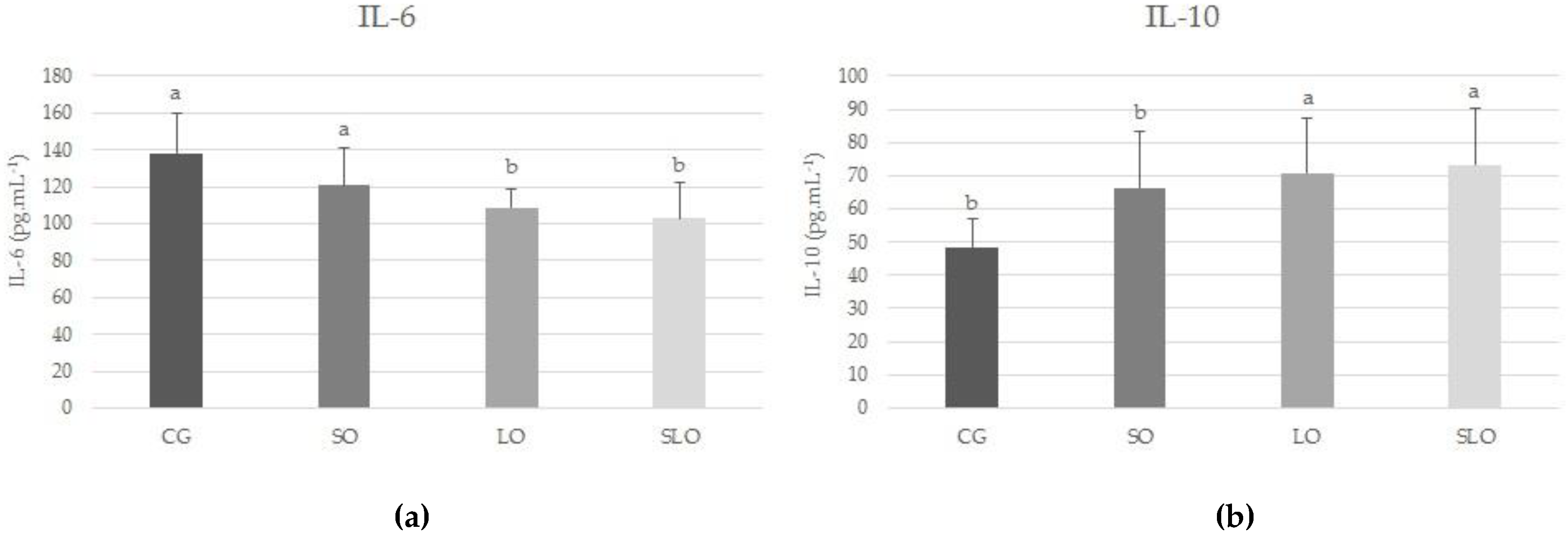
3.5. IL-6 and IL-10
3.6. Histopathology of Liver and Intestine
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Myles, I.A. Fast food fever: Reviewing the impacts of the Western diet on immunity. Nutr. J. 2014, 13, 61. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, P.S.; Inada, A.C.; Marcelino, G.; Cardozo, C.M.L.; Freitas, K.C.; Guimarães, R.C.A.; Castro, A.P.; Nascimento, V.A.; Hiane, P.A. Fatty Acids Consumption: The Role Metabolic Aspects Involved in Obesity and Its Associated Disorders. Nutrients 2017, 9, 1158. [Google Scholar] [CrossRef]
- Simopoulos, A.P. An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. WHO Regional Office for Europe: European Food and Nutrition Action Plan 2015–2020. 2014. Available online: www.euro.who.int/__data/assets/pdf_file/0003/294474/European-Food-Nutrition-Action-Plan-20152020-en.pdf?Ua=1 (accessed on 21 September 2019).
- Fritsche, K.L. The Science of Fatty Acids and Inflammation. Adv. Nutr. 2015, 6, 293S–301S. [Google Scholar] [CrossRef] [PubMed]
- Stolarczyk, E. Adipose tissue inflammation in obesity: A metabolic or immune response? Curr. Opin. Pharmacol. 2017, 37, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Jeyapal, S.; Kona, S.R.; Mullapudi, S.V.; Putcha, U.K.; Gurumurthy, P.; Ibrahim, A. Substitution of linoleic acid with α-linolenic acid or long chain n-3 polyunsaturated fatty acid prevents Western diet induced nonalcoholic steatohepatitis. Sci. Rep. 2018, 8, 10953. [Google Scholar] [CrossRef] [PubMed]
- Lucas, C.; Lucas, G.; Lucas, N.; Krzowska-Firych, J.; Tomasiewicz, K. A systematic review of the present and future of non-alcoholic fatty liver disease. Clin. Exp. Hepatol. 2018, 4, 165–174. [Google Scholar] [CrossRef]
- Minihane, A.M.; Vinoy, S.; Russell, W.R.; Baka, A.; Roche, H.M.; Tuohy, K.M.; Teeling, J.L.; Blaak, E.E.; Fenech, M.; Vauzour, D.; et al. Low-grade inflammation, diet composition and health: Current research evidence and its translation. Br. J. Nutr. 2015, 114, 999–1012. [Google Scholar] [CrossRef]
- Calder, P.C.; Ahluwalia, N.; Brouns, F.; Buetler, T.; Clement, K.; Cunningham, K.; Esposito, K.; Jönsson, L.S.; Kolb, H.; Lansink, M.; et al. Dietary factors and low-grade inflammation in relation to overweight and obesity. Br. J. Nutr. 2011, 106, S1–S78. [Google Scholar] [CrossRef]
- Xuguang, H.; Aofei, T.; Tao, L.; Longyan, Z.; Weijian, B.; Jiao, G. Hesperidin ameliorates insulin resistance by regulating the IRS1-GLUT2 pathway via TLR4 in hepg2 cells. Phytother. Res. 2019, 33, 1697–1705. [Google Scholar] [CrossRef]
- Medeiros, N.I.; Mattos, R.T.; Menezes, C.A.; Fares, R.C.G.; Talvani, A.; Dutra, W.O.; Rios-Santos, F.; Correa-Oliveira, R.; Gomes, J.A.S. IL-10 and TGF-beta unbalanced levels in neutrophils contribute to increase inflammatory cytokine expression in childhood obesity. Eur. J. Nutr. 2017, 57, 2421–2430. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, J.; Donoso, W.; Díaz, N.; Albornoz, M.E.; Huilcaman, R.; Moraes, E.; Moore-Carrasco, R. High fat diet induces adhesion of platelets to endothelium in two models of dyslipidemia. J. Obes. 2014, 2014, 591270. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, P.S.; Candido, C.J.; Jaques, J.A.; Nunes, Â.A.; Caires, A.R.; Michels, F.S.; Almeida, J.A.; Filiú, W.F.; Hiane, P.A.; Nascimento, V.A.; et al. Oxidative stability of sesame and flaxseed oils and their effects on morphometric and biochemical parameters in an animal model. J. Sci. Food Agric. 2016, 97, 3359–3364. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 fatty acids and inflammatory processes: From molecules to man. Biochem. Soc. Trans. 2017, 45, 1105–1115. [Google Scholar] [CrossRef]
- Tan, C.; Voss, U.; Svensson, S.; Erlinge, D.; Olde, B. High glucose and free fatty acids induce beta cell apoptosis via autocrine effects of ADP acting on the P2Y 13 receptor. Purinergic Signal. 2013, 9, 67–79. [Google Scholar] [CrossRef]
- Hernández-Rodas, M.C.; Valenzuela, R.; Echeverría, F.; Rincón-Cervera, M.Á.; Espinosa, A.; Illesca, P.; Videla, L.A. Supplementation with Docosahexaenoic Acid and Extra Virgin Olive Oil Prevents Liver Steatosis Induced by a High-Fat Diet in Mice through PPAR-α and Nrf2 Upregulation with Concomitant SREBP-1c and NF-kb Downregulation. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Deng, X.; Dong, Q.; Bridges, D.; Raghow, R.; Park, E.A.; Elam, M.B. Docosahexaenoic acid inhibits proteolytic processing of sterol regulatory element-binding protein-1c (SREBP-1c) via activation of AMP-activated kinase. Biochim. Biophys. Acta 2015, 1851, 1521–1529. [Google Scholar] [CrossRef]
- Figueiredo, P.S.; Guimarães, R.C.A.; Freitas, K.C.; Hiane, P.A. Physiological and Analytical Approach to Vegetable Oils; Lambert Academic Publishing: Saarbrücken, Germany, 2018. [Google Scholar]
- Burdge, G.C.; Calder, P.C. Conversion of alpha-linolenic acid to longer-chain polyunsaturated fatty acids in human adults. Reprod. Nutr. Dev. 2005, 45, 581–597. [Google Scholar] [CrossRef]
- Xu, J.; Rong, S.; Gao, H.; Chen, C.; Yang, W.; Deng, Q.; Huang, Q.; Xiao, L.; Huang, F. A Combination of Flaxseed Oil and Astaxanthin Improves Hepatic Lipid Accumulation and Reduces Oxidative Stress in High Fat-Diet Fed Rats. Nutrients 2017, 9, 271. [Google Scholar] [CrossRef]
- Moazzami, A.A.; Haese, S.L.; Kamal-Eldin, A. Lignan contents in sesame seeds and products. Eur. J. Lipid Sci. Technol. 2007, 109, 1022–1027. [Google Scholar] [CrossRef]
- Aslam, F.; Iqbal, S.; Nasir, M.; Anjum, A.A. White Sesame Seed Oil Mitigates Blood Glucose Level, Reduces Oxidative Stress, and Improves Biomarkers of Hepatic and Renal Function in Participants with Type 2 Diabetes Mellitus. J. Am. Coll. Nutr. 2019, 38, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Ismail, M.; Hasan, H.; El-Orfali, Y.; Ismail, H.; Khawaja, G. Anti-Inflammatory, Antioxidative, and Hepatoprotective Effects of Trans Δ9-Tetrahydrocannabinol/Sesame Oil on Adjuvant-Induced Arthritis in Rats. Evid. Based Complement. Alternat. Med. 2018, 2018. [Google Scholar] [CrossRef] [PubMed]
- Reeves, P.G.; Nielsen, F.H.; Fahey, G.C., Jr. AIN-93 purified diets for laboratory rodents: Final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J. Nutr. 1993, 123, 1939–1951. [Google Scholar] [CrossRef]
- Maia, E.L.; Rodriguez-Amaya, D.B. Avaliação de um método simples e econômico para a metilação de ácidos graxos com lipídios de diversas espécies de peixes. Rev. Inst. Adolfo Lutz 1993, 53, 27–35. [Google Scholar]
- Hartman, L.; Lago, R.C.A. Rapid preparation of fatty acid methyl esters from lipids. Lab. Pract. 1973, 22, 475–494. [Google Scholar] [PubMed]
- Lepage, G.; Roy, C.C. Direct transesterification of all classes of lipids in a one-step reaction. J. Lipid Res. 1986, 27, 114–120. [Google Scholar] [PubMed]
- Ulbricht, T.L.V.; Southgate, D.A.T. Coronary heart disease: Seven dietary factors. Lancet 1991, 338, 985–992. [Google Scholar] [CrossRef]
- Santos-Silva, J.; Bessa, R.J.B.; Santos-Silva, F. Effect of genotype, feeding system and slaughter weight on the quality of light lambs: Fatty and composition of meat. Livest. Prod. Sci. 2002, 77, 187–194. [Google Scholar] [CrossRef]
- Hagen, J.H.; Hagen, P.B. An enzimatic method for the estimation of glicerol in blood. J. Biochem. Physiol. 1962, 40, 1129–1139. [Google Scholar] [CrossRef]
- Flegg, H.M. An investigation of the determination of serum cholesterol by na enzymatic method. Clin. Biochem. 1973, 10, 79–84. [Google Scholar] [CrossRef]
- Carey, R.N.; Felbruegge, C.; Westgard, J.O. Evaluation of the adaptation of the glucose oxidase/peroxidase-3-methyl-2-benzothiazoline hydrazone-N, Ndimethylaniline procedure to the technicon SMA 12/60 and comparation with other automed methods for glucose. Clin. Chem. 1974, 20, 595–602. [Google Scholar] [PubMed]
- Lunkes, G.I.; Lunkes, D.S.; Morschb, V.M.; Mazzantib, C.M.; Morschb, A.L.B.; Mironb, V.R.; Schetinger, M.R.C. Ntpdase and 5′-nucleotidase activities in rats with alloxan-induced diabetes. Diabetes Res. Clin. Pract. 2004, 65, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Jaques, J.A.S.; Ruchel, J.B.; Schlemmer, K.B.; Pimentel, V.C.; Bagatini, M.; do Carmo Gonçalves Souza, V.; Moretto, M.B.; Morsch, V.M.; Schetinger, M.R.; Leal, D.B. Effects of curcumin on the activities of the enzymes that hydrolyse adenine nucleotides in platelets from cigarette smoke-exposed rats. Cell Biochem. Funct. 2011, 29, 630–635. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.M.; Delfert, D.; Junger, K.D. A direct colorimetric assay for Ca2+-stimulated atpase activity. Anal. Biochem. 1986, 157, 375–380. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 57, 375–380. [Google Scholar]
- Erben, U.; Loddenkemper, C.; Doerfel, K.; Spieckermann, S.; Haller, D.; Heimesaat, M.M.; Zeitz, M.; Siegmund, B.; Kühl, A.A. A guide to histomorphological evaluation of intestinal inflammation in mouse models. Int. J. Clin. Exp. Pathol. 2014, 7, 4557–4576. [Google Scholar]
- Pokorska-S’piewak, M.; Kowalik-Mikolajewska, B.; Aniszewska, M.; Pluta, M.; Marczynska, M. Novel serum biomarkers modified by the body mass index z-score for the detection of liver fibrosis and steatosis in children with chronic hepatitis C. BMC Infect. Dis. 2017, 17, 361. [Google Scholar] [CrossRef]
- Aguila, M.B.; Pinheiro, A.R.; Parente, L.B.; Mandarim-de-Lacerda, C.A. Dietary effect of different high-fat diet on rat liver stereology. Liver Int. 2003, 23, 363–370. [Google Scholar] [CrossRef]
- Calder, P.C. Very long chain omega-3 (n-3) fatty acids and human health. Eur. J. Lipid Sci. Technol. 2014, 116, 1280–1300. [Google Scholar] [CrossRef]
- Rossano, R.; Caggiano, M.A.; Mastrangelo, L.; Di Lauro, R.; Ungaro, N.; Ettorre, M.; Riccio, P. Proteins, fatty acids and nutritional value in the muscle of the fish speciesmora moro (Risso, 1810). Mol. Nutr. Food Res. 2005, 49, 926–931. [Google Scholar] [CrossRef]
- Semwogerere, F.; Neethling, J.; Muchenje, V.; Hoffman, L.C. Meat quality, fatty acid profile, and sensory attributes of spent laying hens fed expeller press canola meal or a conventional diet. Poult. Sci. 2019, 98, 3557–3570. [Google Scholar] [CrossRef] [PubMed]
- Tenyang, N.; Ponka, R.; Tiencheu, B.; Djikeng, F.T.; Azmeera, T.; Karuna, M.S.L.; Prasad, R.B.N.; Womeni, H.M. Effects of boiling and roasting on proximate composition, lipid oxidation, fatty acid profile and mineral content of two sesame varieties commercialized and consumed in Far-North Region of Cameroon. Food Chem. 2017, 221, 1308–1316. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, R.C.A.; Macedo, M.L.R.; Munhoz, C.L.; Filiu, W.; Viana, L.H.; Nozaki, V.T.; Hiane, P.A. Sesame and flaxseed oil: Nutritional quality and effects on serum lipids and glucose in rats. Food Sci. Technol. 2013, 33, 209–217. [Google Scholar] [CrossRef]
- Bersch-Ferreira, A.C.; Sampaio, G.R.; Gehringer, M.O.; Torres, E.A.F.S.; Ross-Fernandes, M.B.; da Silva, J.T.; Torreglosa, C.R.; Kovacs, C.; Alves, R.; Magnoni, C.D.; et al. Association between plasma fatty acids and inflammatory markers in patients with and without insulin resistance and in secondary prevention of cardiovascular disease, a cross-sectional study. Nutr. J. 2018, 17, 26. [Google Scholar] [CrossRef]
- Sousa, A.B.B.; Junior, O.O.S.; Visentainer, J.V.; Almeida, N.M. Total lipid nutritional quality of the adipose tissue from the orbital cavity in Nile tilapia from continental aquaculture. Acta Sci. Anim. Sci. 2017, 39, 335–341. [Google Scholar] [CrossRef][Green Version]
- Devarajan, S.; Singh, R.; Chatterjee, B.; Zhang, B.; Ali, A. A blend of sesame oil and rice bran oil lowers blood pressure and improves the lipid profile in mild-to-moderate hypertensive patients. J. Clin. Lipidol. 2016, 10, 339–349. [Google Scholar] [CrossRef]
- Kandeil, M.A.; Hashem, R.M.; Mahmoud, M.O.; Hetta, M.H.; Tohamy, M.A. Zingiber officinale extract and omega-3 fatty acids ameliorate endoplasmic reticulum stress in a nonalcoholic fatty liver rat model. J. Food Biochem. 2019. [Google Scholar] [CrossRef]
- Wang, X.-H.; Li, C.-Y.; Muhammad, I.; Zhang, X.-Y. Fatty acid composition in serum correlates with that in the liver and non-alcoholic fatty liver disease activity scores in mice fed a high-fat diet. Environ. Toxicol. Pharmacol. 2016, 44, 140–150. [Google Scholar] [CrossRef]
- Monteiro, P.F.; Morganti, R.P.; Delbin, M.A.; Calixto, M.C.; Lopes-Pires, M.E.; Marcondes, S.; Zanesco, A.; Antunes, E. Platelet hyperaggregability in high-fat fed rats: A role for intraplatelet reactive-oxygen species production. Cardiovasc. Diabetol. 2012, 11, 5. [Google Scholar] [CrossRef]
- Duan, Y.; Zeng, L.; Zheng, C.; Song, B.; Li, F.; Kong, X.; Xu, K. Inflammatory links between high fat diets and diseases. Front. Immunol. 2018, 9, 2649. [Google Scholar] [CrossRef]
- Ali, F.F.; Rifaai, R.A. Preventive effect of omega-3 fatty acids in a rat model of stress-induced liver injury. J. Cell. Physiol. 2018, 234, 11960–11968. [Google Scholar] [CrossRef]
- Gonçalves, N.B.; Bannitz, R.F.; Silva, B.R.; Becari, D.D.; Poloni, C.; Gomes, P.M.; Foss, M.C.; Foss-Freitas, M.C. α-Linolenic acid prevents hepatic steatosis and improves glucose tolerance in mice fed a high-fat diet. Clinics 2018, 73, e150. [Google Scholar] [CrossRef] [PubMed]
- Musso, G.; Gambino, R.; Cassader, M. Recent insights into hepatic lipid metabolism in non-alcoholic fatty liver disease (NAFLD). Prog. Lipid Res. 2009, 48, 1–26. [Google Scholar] [CrossRef] [PubMed]
- Clarke, S.D. Polyunsaturated fatty acid regulation of gene transcription: A molecular mechanism to improve the metabolic syndrome. J. Nutr. 2001, 131, 1129–1132. [Google Scholar] [CrossRef] [PubMed]
- Mustonen, A.M.; Kärjä, V.; Kilpiö, M.; Tammi, R.; Tammi, M.; Rouvinen-Watt, K.; Halonen, T.; Nieminen, P. Manifestations of fasting-induced fatty liver and rapid recovery from steatosis in voles fed lard or flaxseed oil lipids. Nutrients 2013, 5, 4211–4230. [Google Scholar] [CrossRef] [PubMed]
- Soleimani, A.; Taghizadeh, M.; Bahmani, F.; Badroj, N.; Asemi, Z. Metabolic response to omega-3 fatty acid supplementation in patients with diabetic nephropathy: A randomized, double-blind, placebo-controlled trial. Clin. Nutr. 2017, 36, 79–84. [Google Scholar] [CrossRef] [PubMed]
| Fatty Acid (%) | CG | SO | LO | SLO |
|---|---|---|---|---|
| Capric, C10:0 | 0.15 ± 0.021 | - | - | - |
| Lauric, C12:0 | 0.30 ± 0.04 | - | 0.12 ± 0.01 | - |
| Myristic, C14:0 | 1.53 ± 0.15 | 0.24 ± 0.01 | 0.24 ± 0.01 | 0.26 ± 0.03 |
| Palmitic, C16:0 | 23.12 ± 0.96 | 13.03 ± 0.05 | 6.20 ± 0.12 | 9.78 ± 0.50 |
| Palmitoleic, C16:1 | 1.86 ± 0.09 | 0.24 ± 0.00 | 0.11 ± 0.00 | 0.27 ± 0.02 |
| Heptadecanoic, C17:0 | 0.38 ± 0.01 | - | - | - |
| Stearic, C18:0 | 11.32 ± 0.04 | 3.31 ± 0.03 | 3.21 ± 0.03 | 3.44 ± 0.07 |
| Elaidic acid (trans), C18:1 | 0.19 ± 0.04 | - | - | - |
| Oleic, C18:1 (n- 9) | 35.53 ± 0.49 | 32.61 ± 0.08 | 16.32 ± 0.05 | 25.92 ± 0.17 |
| Linoleic, C18:2 (n-6) | 16.37 ± 0.12 | 43.89 ± 0.18 | 15.68 ± 0.01 | 30.74 ± 0.02 |
| α-Linolenic, C18:3 (n-3) | 1.07 ± 0.05 | 1.08 ± 0.00 | 51.89 ± 0.14 | 23.38 ± 0.11 |
| Arachidic, C20:0 | 0.24 ± 0.01 | 0.64 ± 0.15 | 0.10 ± 0.00 | 0.35 ± 0.02 |
| Cis-11-eicosenic, C20:1 | 0.80 ± 0.04 | 0.25 ± 0.01 | 0.16 ± 0.04 | 0.21 ± 0.01 |
| Behenic, C22:0 | 0.14 ± 0.02 | 0.13 ± 0.00 | 0.13 ± 0.01 | 0.22 ± 0.05 |
| Erucic, C22:1 | 0.27 ± 0.01 | - | - | - |
| SFA | 37.18 | 17.35 | 10.00 | 14.04 |
| MUFA | 38.65 | 33.29 | 16.85 | 26.40 |
| PUFA | 17.44 | 44.96 | 67.57 | 54.12 |
| n-6/n-3 ratio | 15.29 | 40.64 | 0.30 | 1.31 |
| Index | CG | SO | LO | SLO |
|---|---|---|---|---|
| Atherogenic index (AI) | 0.53 ± 0.03 a | 0.18 ± 0.00 b | 0.09 ± 0.00 c | 0.013 ± 0.1 d |
| Thrombogenic index (TI) | 1.17 ± 0.04 a | 0.40 ± 0.00 b | 0.06 ± 0.00 b | 0.14 ± 0.01 b |
| Hypocholesterolemic: hypercholesterolemic (HH) | 2.15 ± 0.12 d | 5.85 ± 0.03 c | 13.03 ± 0.03 a | 7.99 ± 0.44 b |
| Parameter | CG | SO | LO | SLO |
|---|---|---|---|---|
| Liver | 10.29 ± 1.18 a | 10.03 ± 0.67 a | 8.68 ± 0.72 b | 8.71 ± 0.72 b |
| Epididymal | 8.57 ± 1.91 a | 6.24 ± 1.58 b | 4.52 ± 1.03 b | 4.68 ± 1.24 b |
| Mesenteric | 5.51 ± 1.41 a | 4.02 ± 0.94 b | 3.55 ± 0.9 b | 3.02 ± 0.63 b |
| Retroperitoneal | 8.35 ± 2.08 a | 5.96 ± 1.58 b | 4.54 ± 0.96 b | 4.93 ± 1.78 b |
| Parameter | CG | SO | LO | SLO |
|---|---|---|---|---|
| AST (UI/L | 117.38 ± 14.0 a | 103.00 ± 19.54 a | 91.14 ± 9.33 b | 90.0 ± 7.93 b |
| ALT (UI/L) | 64.9 ± 12.2 a | 73.51 ± 7.4 a | 71.0 ± 16.7 a | 60.98 ± 10.4 a |
| Score | ||||
|---|---|---|---|---|
| Organ | CG | SO | LO | SLO |
| Level of liver steatosis | 6 a | 6 a | 4 b | 6 a |
| Evaluation of the intestine vilosities | 4 a | 4 a | 4 a | 4 a |
| Parameter | CG | SO | LO | SLO |
|---|---|---|---|---|
| % steatosis | 39.91 ± 3.7 a | 53.61 ± 1.5 a | 18.97 ± 2.10 b | 29.72 ± 3.8 a,b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jordão Candido, C.; Silva Figueiredo, P.; Del Ciampo Silva, R.; Candeloro Portugal, L.; Augusto dos Santos Jaques, J.; Alves de Almeida, J.; de Barros Penteado, B.; Albuquerque Dias, D.; Marcelino, G.; Pott, A.; et al. Protective Effect of α-Linolenic Acid on Non-Alcoholic Hepatic Steatosis and Interleukin-6 and -10 in Wistar Rats. Nutrients 2020, 12, 9. https://doi.org/10.3390/nu12010009
Jordão Candido C, Silva Figueiredo P, Del Ciampo Silva R, Candeloro Portugal L, Augusto dos Santos Jaques J, Alves de Almeida J, de Barros Penteado B, Albuquerque Dias D, Marcelino G, Pott A, et al. Protective Effect of α-Linolenic Acid on Non-Alcoholic Hepatic Steatosis and Interleukin-6 and -10 in Wistar Rats. Nutrients. 2020; 12(1):9. https://doi.org/10.3390/nu12010009
Chicago/Turabian StyleJordão Candido, Camila, Priscila Silva Figueiredo, Rafael Del Ciampo Silva, Luciane Candeloro Portugal, Jeandre Augusto dos Santos Jaques, Jeeser Alves de Almeida, Bruna de Barros Penteado, Dhébora Albuquerque Dias, Gabriela Marcelino, Arnildo Pott, and et al. 2020. "Protective Effect of α-Linolenic Acid on Non-Alcoholic Hepatic Steatosis and Interleukin-6 and -10 in Wistar Rats" Nutrients 12, no. 1: 9. https://doi.org/10.3390/nu12010009
APA StyleJordão Candido, C., Silva Figueiredo, P., Del Ciampo Silva, R., Candeloro Portugal, L., Augusto dos Santos Jaques, J., Alves de Almeida, J., de Barros Penteado, B., Albuquerque Dias, D., Marcelino, G., Pott, A., Avellaneda Guimarães, R. d. C., & Aiko Hiane, P. (2020). Protective Effect of α-Linolenic Acid on Non-Alcoholic Hepatic Steatosis and Interleukin-6 and -10 in Wistar Rats. Nutrients, 12(1), 9. https://doi.org/10.3390/nu12010009






