Predictors of Early Introduction of Core and Discretionary Foods in Australian Infants—Results from HSHK Birth Cohort Study
Abstract
1. Introduction
- (a)
- to assess the timing of introduction of core and discretionary foods in infants residing in SWS region; and
- (b)
- to ascertain the family and infant-related predictors of early introduction of core and discretionary foods in this population group.
2. Methods
2.1. Sample
2.2. Data Collection
2.3. Outcome Measure
2.4. Potential Predictors of Timing
2.5. Statistical Analyses
2.6. Ethics Approval and Consent to Participate
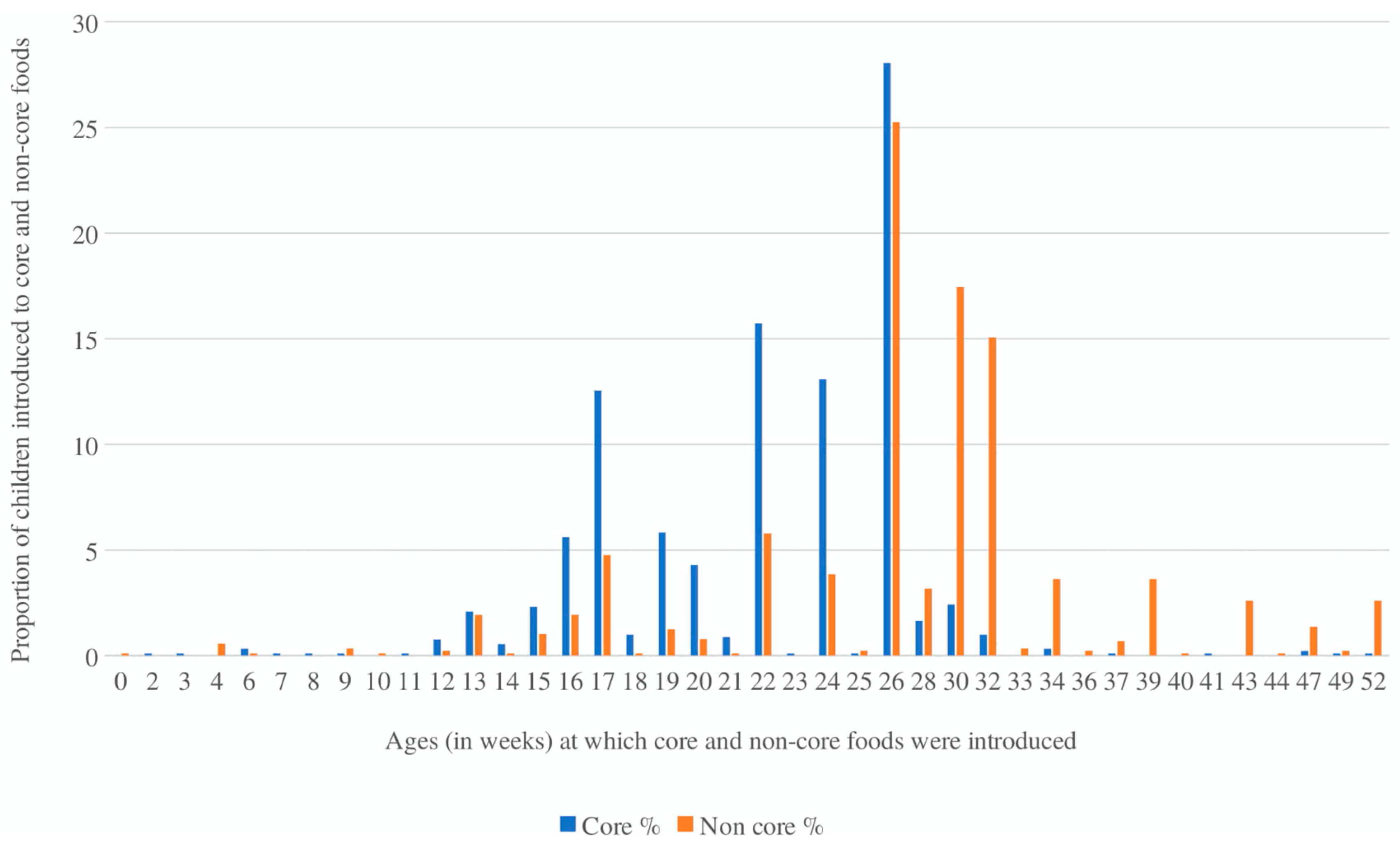
3. Results
3.1. Predictors of Early Introduction of Core Foods
3.2. Predictors of Early Introduction of Discretionary Foods
4. Discussion
4.1. Predictors of Early Introduction of Core Foods
4.2. Predictors of Early Introduction of Discretionary Foods
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Exclusive Breastfeeding for Six Month Best for Babies Everywhere; Director of the Department of Communications, World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Fewtrell, M.; Bronsky, J.; Campoy, C.; Domellöf, M.; Embleton, N.; Mis, N.F.; Hojsak, I.; Hulst, J.M.; Indrio, F.; Lapillonne, A.; et al. Complementary feeding: A position paper by the European Society for Paediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Netting, M.J.; Campbell, D.E.; Koplin, J.J.; Beck, K.M.; McWilliam, V.; Dharmage, S.C.; Tang, M.L.K.; Ponsonby, A.L.; Prescott, S.L.; Vale, S.; et al. An Australian consensus on infant feeding guidelines to prevent food allergy: Outcomes from the Australian Infant Feeding Summit. J. Allergy Clin. Immunol. Pract. 2017, 5, 1617–1624. [Google Scholar] [CrossRef]
- National Health and Medical Research Council. Eat for Health; Infant Feeding Guidelines; Information for Health Workers; Commonwealth of Australia: Canberra, Australia, 2013.
- National Health and Medical Research Council. Infant Feeding Guidelines; Commonwealth of Australia: Canberra, Australia, 2012.
- Kramer, M.S.; Kakuma, R. Optimal duration of exclusive breastfeeding. Cochrane Database Syst. Rev. 2012. [Google Scholar] [CrossRef] [PubMed]
- Huh, S.Y.; Rifas-Shiman, S.L.; Taveras, E.M.; Oken, E.; Gillman, M.W. Timing of solid food introduction and risk of obesity in preschool-aged children. Pediatrics 2011, 127, e544–e551. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.; Binns, C.W.; Lee, A.H. The Early Introduction of Complementary (Solid) Foods: A Prospective Cohort Study of Infants in Chengdu, China. Nutrients 2019, 11, 760. [Google Scholar] [CrossRef]
- Pluymen, L.P.M.; Wijga, A.H.; Gehring, U.; Koppelman, G.H.; Smit, H.A.; van Rossem, L. Early introduction of complementary foods and childhood overweight in breastfed and formula-fed infants in the Netherlands: The PIAMA birth cohort study. Eur. J. Nutr. 2018, 57, 1985–1993. [Google Scholar] [CrossRef] [PubMed]
- Ha, D.H.; Do, L.G.; Spencer, A.J.; William Murray, T.; Golley, R.K.; Rugg-Gunn, A.J.; Levy, S.M.; Scott, J.A. Factors Influencing Early Feeding of Foods and Drinks Containing Free Sugars-A Birth Cohort Study. Int. J. Environ. Res. Public Health 2017, 14, 1270. [Google Scholar] [CrossRef]
- Ho, N.; Olds, T.; Schranz, N.; Maher, C. Secular trends in the prevalence of childhood overweight and obesity across Australian states: A meta-analysis. J. Sci. Med. Sport 2017, 20, 480–488. [Google Scholar] [CrossRef] [PubMed]
- Ha, D.H.; Roberts-Thomson, K.F.; Arrow, P.; Peres, K.G.; Do, L.G. Children’s oral health status in Australia, 2012–2014. In Oral Health of Australian Children: The National Child Oral Health Study 2012–2014; Do, L.G., Spencer, A.J., Eds.; Adelaide University Press: Adelaide, Australia, 2016; pp. 86–152. [Google Scholar]
- De Cosmi, V.; Scaglioni, S.; Agostoni, C. Early Taste Experiences and Later Food Choices. Nutrients 2017, 9, 107. [Google Scholar] [CrossRef]
- Kellett, E.; Smith, A.; Schmerlaib, Y. The Australian Guide to Healthy Eating. Commonwealth Department of Health and Family Services. 1998. Available online: http://www.health.gov.au/internet/main/publishing.nsf/content/E384CFA588B74377CA256F190004059B/$File/fd-cons.pdf (accessed on 10 June 2019).
- Special Supplemental Nutrition Program for Women Infants, and Children. Infant Nutrition and Feeding: A Guide for Use in the WIC and CSF Programs; USDA, Food and Nutrition Service, Special Supplemental Nutrition Program for Women Infants, and Children: Washington, DC, USA, 2009.
- Schrempft, S.; van Jaarsveld, C.H.M.; Fisher, A.; Wardle, J. Family and infant characteristics associated with timing of core and non-core food introduction in early childhood. Eur. J. Clin. Nutr. 2013, 67, 652–657. [Google Scholar] [CrossRef] [PubMed]
- National Health and Medical Research Council. Eat for Health—Australian Dietary Guidelines; National Health and Medical Research Council: Canberra, Australia, 2013.
- Rangan, A.M.; Randall, D.; Hector, D.J.; Gill, T.P.; Webb, K.L. Consumption of ‘extra’foods by Australian children: Types, quantities and contribution to energy and nutrient intakes. Eur. J. Clin. Nutr. 2008, 62, 356. [Google Scholar] [CrossRef] [PubMed]
- Australian Institute of Health and Welfare. Australia’s Health 2012: Australia’s Health Series No.13. Cat No. AUS 156; AIHW, Ed.; AIHW: Canberra, Australia, 2012.
- Centre for Epidemiology and Evidence. 2009–2010 Summary Report from the New South Wales Child Health Survey; NSW Ministry of Health Sydney: Sydney, Australia, 2012. [Google Scholar]
- Mannan, H. Early infant feeding of formula or solid foods and risk of childhood overweight or obesity in a socioeconomically disadvantaged region of Australia: A longitudinal cohort analysis. Int. J. Environ. Res. Public Health 2018, 15, 1685. [Google Scholar] [CrossRef] [PubMed]
- Wen, L.; Rissel, C.; Baur, L.; Hayes, A.; Xu, H.; Whelan, A.; Hua, M.; Shaw, M. Phongsavan P: A 3-arm randomised controlled trial of Communicating Healthy Beginnings Advice by Telephone (CHAT) to mothers with infants to prevent childhood obesity. BMC Public Health 2017, 17, 79. [Google Scholar] [CrossRef] [PubMed]
- NSW Government. South West Sydney: Our Health in-Depth; District SWSLH, Ed.; SWSLH: Sydney, Australia, 2019.
- Arora, A.; Manohar, N.; Hayen, A.; Bhole, S.; Eastwood, J.; Levy, S.; Scott, J.A. Determinants of breastfeeding initiation among mothers in Sydney, Australia: Findings from a birth cohort study. Int. Breastfeed. J. 2017, 12, 39. [Google Scholar] [CrossRef]
- Arora, A.; Scott, J.; Bhole, S.; Do, L.; Schwarz, E.; Blinkhorn, A. Early childhood feeding practices and dental caries in preschool children: A multi-centre birth cohort study. BMC Public Health 2011, 11, 28. [Google Scholar] [CrossRef]
- Ogbo, F.A.; Eastwood, J.; Page, A.; Arora, A.; McKenzie, A.; Jalaludin, B.; Tennant, E.; Miller, E.; Kohlhoff, J.; Noble, J.; et al. Prevalence and determinants of cessation of exclusive breastfeeding in the early postnatal period in Sydney, Australia. Int. Breastfeed. J. 2016, 12, 16. [Google Scholar] [CrossRef]
- Arora, A.; Bedros, D.; Bhole, S.; Do, L.G.; Scott, J.; Blinkhorn, A.; Schwarz, E. Child and family health nurses’ experiences of oral health of preschool children: A qualitative approach. J. Public Health Dent. 2012, 72, 149–155. [Google Scholar] [CrossRef]
- Scott, J.A.; Binns, C.W.; Graham, K.I.; Oddy, W.H. Temporal changes in the determinants of breastfeeding initiation. Birth 2006, 33, 37–45. [Google Scholar] [CrossRef]
- Arora, A.; Manohar, N.; Bedros, D.; Hua, A.P.D.; You, S.Y.H.; Blight, V.; Ajwani, S.; Eastwood, J.; Bhole, S. Lessons learnt in recruiting disadvantaged families to a birth cohort study. BMC Nurs. 2018, 17, 7. [Google Scholar] [CrossRef]
- Scott, J.A.; Binns, C.W.; Aroni, R.A. Breast-feeding in Perth: Recent trends. Aust. N. Z. J. Public Health 1996, 20, 210–211. [Google Scholar] [CrossRef]
- Scott, J.A.; Binns, C.W.; Graham, K.I.; Oddy, W.H. Predictors of the early introduction of solid foods in infants: Results of a cohort study. BMC Pediatr. 2009, 9, 60. [Google Scholar] [CrossRef] [PubMed]
- Rankin, S.J.; Levy, S.M.; Warren, J.J.; Gilmore, J.E.; Broffitt, B. Relative validity of an FFQ for assessing dietary fluoride intakes of infants and young children living in Iowa. Public Health Nutr. 2011, 14, 1229–1236. [Google Scholar] [CrossRef] [PubMed]
- National Health and Medical Research Council. Eat for Health; Educator Guide; Commonwealth of Australia: Canberra, Australia, 2013.
- National Health and Medical Research Council (NHMRC). Dietary Guidelines for Children and Adolescents in Australia Incorporating the Infant Feeding Guidelines for Health Workers; Commonwealth of Australia: Canberra, Australia, 2003.
- Australian Bureau of Statistics. Census of Population and Housing: Socio-Economic Indexes for Areas (SEIFA); Statistics Abo, Ed.; Australian Bureau of Statistics: Canberra, Australia, 2016.
- StataCorp. Stata Statistical Software: Release 15; StataCorp LP: College Station, TX, USA, 2017. [Google Scholar]
- Agostoni, C.; Decsi, T.; Fewtrell, M.; Goulet, O.; Kolacek, S.; Koletzko, B.; Michaelsen, K.F.; Moreno, L.; Puntis, J.; Rigo, J.; et al. Complementary feeding: A commentary by the ESPGHAN Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2008, 46, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Magarey, A.; Kavian, F.; Scott, J.A.; Markow, K.; Daniels, L. Feeding mode of Australian infants in the first 12 months of life: An assessment against national breastfeeding indicators. J. Hum. Lact. 2016, 32, 95–104. [Google Scholar] [CrossRef]
- Amezdroz, E.; Carpenter, L.; O’callaghan, E.; Johnson, S.; Waters, E. Transition from milks to the introduction of solid foods across the first 2 years of life: Findings from an Australian birth cohort study. J. Hum. Nutr. Diet. 2015, 28, 375–383. [Google Scholar] [CrossRef]
- Wang, L.; Van Grieken, A.; Van Der Velde, L.A.; Vlasblom, E.; Beltman, M.; L’Hoir, M.P.; Boere-Boonekamp, M.M.; Raat, H. Factors associated with early introduction of complementary feeding and consumption of non-recommended foods among Dutch infants: The BeeBOFT study. BMC Public Health 2019, 19, 388. [Google Scholar] [CrossRef]
- Hollis, J.; Crozier, S.; Inskip, H.; Cooper, C.; Godfrey, K.; Robinson, S. Age at introduction of solid foods and feeding difficulties in childhood: Findings from the Southampton Women’s Survey. Br. J. Nutr. 2016, 116, 743–750. [Google Scholar] [CrossRef]
- Australian Institute of Health and Welfare. 2010 Australian National Infant Feeding Survey: Indicator Results; Australian Institute of Health and Welfare: Canberra, Australia, 2011.
- NSW Department of Health Centre for Epidemiology and Research. NSW Child Health Survey: 2009–2010 Summary Report; Health NMo, Ed.; Health NMo: North Sydney, Auatralia, 2011.
- Koh, G.A.; Scott, J.A.; Oddy, W.H.; Graham, K.I.; Binns, C.W. Exposure to non-core foods and beverages in the first year of life: Results from a cohort study. Nutr. Diet. 2010, 67, 137–142. [Google Scholar] [CrossRef]
- Boudet-Berquier, J.; Salanave, B.; de Launay, C.; Castetbon, K. Introduction of complementary foods with respect to French guidelines: Description and associated socio-economic factors in a nationwide birth cohort (Epifane survey). Matern. Child Nutr. 2017, 13, e12339-n/a. [Google Scholar] [CrossRef]
- McGowan, L.; Croker, H.; Wardle, J.; Cooke, L.J. Environmental and individual determinants of core and non-core food and drink intake in preschool-aged children in the United Kingdom. Eur. J. Clin. Nutr. 2012, 66, 322. [Google Scholar] [CrossRef]
- Kuo, A.A.; Inkelas, M.; Slusser, W.M.; Maidenberg, M.; Halfon, N. Introduction of solid food to young infants. Matern. Child Health J. 2011, 15, 1185–1194. [Google Scholar] [CrossRef] [PubMed]
- Bolton, K.A.; Kremer, P.; Hesketh, K.D.; Laws, R.; Kuswara, K.; Campbell, K.J. Differences in infant feeding practices between Chinese-born and Australian-born mothers living in Australia: A cross-sectional study. BMC Pediatr. 2018, 18, 209. [Google Scholar] [CrossRef] [PubMed]
- Rebhan, B.; Kohlhuber, M.; Schwegler, U.; Koletzko, B.V.; Fromme, H. Infant feeding practices and associated factors through the first 9 months of life in Bavaria, Germany. J. Pediatr. Gastroenterol. Nutr. 2009, 49, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, L.J.; Tate, A.R.; Dezateux, C.; Group MCSCH. Do early infant feeding practices vary by maternal ethnic group? Public Health Nutr. 2007, 10, 957–964. [Google Scholar] [CrossRef]
- Toh, J.; Yip, G.; Han, W.; Fok, D.; Low, Y.-L.; Lee, Y.; Rebello, S.; Saw, S.-M.; Kwek, K.; Godfrey, K.; et al. Infant Feeding Practices in a Multi-Ethnic Asian Cohort: The GUSTO Study. Nutrients 2016, 8, 293. [Google Scholar] [CrossRef] [PubMed]
- Rossiter, J.C.; Yam, B. Breastfeeding: How could it be enhanced? The perceptions of Vietnamese women in Sydney, Australia. J. Midwifery Women’s Health 2000, 45, 271–276. [Google Scholar] [CrossRef]
- Brodribb, W.; Miller, Y. Introducing solids and water to Australian infants. J. Hum. Lact. 2013, 29, 214–221. [Google Scholar] [CrossRef]
- Tang, L.; Lee, A.H.; Binns, C.W. Predictors of early introduction of complementary feeding: Longitudinal study. Pediatr. Int. 2015, 57, 126–130. [Google Scholar] [CrossRef]
- Young, B.E.; Krebs, N.F. Complementary feeding: Critical considerations to optimize growth, nutrition, and feeding behavior. Curr. Pediatr. Rep. 2013, 1, 247–256. [Google Scholar] [CrossRef]
- Moore, T.; McDonald, M.; McHugh-Dillon, H. Evidence Review: Early Childhood Development and the Social Determinants of Health Inequities; Victorian Health Promotion Foundation: Melbourne, Australia, 2015. [Google Scholar]
- Kant, A.K.; Graubard, B.I. Family income and education were related with 30-year time trends in dietary and meal behaviors of American children and adolescents. J. Nutr. 2013, 143, 690–700. [Google Scholar] [CrossRef]
- Braveman, P.; Barclay, C. Health disparities beginning in childhood: A life-course perspective. Pediatrics 2009, 124 (Suppl. 3), S163–S175. [Google Scholar] [CrossRef] [PubMed]
- Hansen, E.; Tesch, L.; Ayton, J. ‘They’re born to get breastfed’-how fathers view breastfeeding: A mixed method study. BMC Pregnancy Childbirth 2018, 18, 238. [Google Scholar] [CrossRef] [PubMed]
- Walsh, A.D.; Cameron, A.J.; Crawford, D.; Hesketh, K.D.; Campbell, K.J. Dietary associations of fathers and their children between the ages of 20 months and 5 years. Public Health Nutr. 2016, 19, 2033–2039. [Google Scholar] [CrossRef]
- White, B.; Giglia, R.C.; White, J.A.; Dhaliwal, S.; Burns, S.K.; Scott, J.A. Gamifying Breastfeeding for Fathers: Process Evaluation of the Milk Man Mobile App. JMIR Pediatr. Parent. 2019, 2, e12157. [Google Scholar] [CrossRef] [PubMed]
- van Zyl, Z.; Maslin, K.; Dean, T.; Blaauw, R.; Venter, C. The accuracy of dietary recall of infant feeding and food allergen data. J. Hum. Nutr. Diet. 2016, 29, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Gillespie, B.; d’Arcy, H.; Schwartz, K.; Bobo, J.K.; Foxman, B. Recall of age of weaning and other breastfeeding variables. Int. Breastfeed. J. 2006, 1, 4. [Google Scholar] [CrossRef]
- Jain, A.; Bongaarts, J. Breastfeeding: Patterns correlates and fertility effects. Stud. Fam. Plan. 1981, 12, 79–99. [Google Scholar] [CrossRef]
| Characteristics | Core Foods (n = 934) a | Discretionary Foods (n = 900) b | ||
|---|---|---|---|---|
| <17 weeks | ≥17 weeks | <52 weeks | ≥52 weeks | |
| Family Characteristics * | N (%) | N (%) | N (%) | N (%) |
| Maternal education | ||||
| Left school before 12 | 30 (17.9%) | 138 (82.1%) | 157 (99.4%) | 1 (0.6%) |
| Completed school | 34 (17.7%) | 158 (82.3%) | 182 (97.9%) | 4 (2.1%) |
| College/TAFE | 20 (11.8%) | 150 (88.2%) | 157 (95.1%) | 8 (4.9%) |
| University | 28 (6.9%) | 376 (93.1%) | 362 (92.6%) | 29 (7.4%) |
| Maternal marital status | ||||
| Married | 72 (9.8%) | 662 (90.2%) | 672 (94.8%) | 37 (5.2%) |
| Living with partner | 18 (16.4%) | 92 (83.6%) | 101 (96.2%) | 4 (3.8%) |
| Single | 22 (24.4%) | 68 (75.6%) | 85 (98.8%) | 1 (1.2%) |
| Maternal work status at 17-weeks | ||||
| Not working | 93 (11.3%) | 733 (88.7%) | - | - |
| Casual | 5 (20.0%) | 20 (80.0%) | - | - |
| Part-time | 10 (22.7%) | 34 (77.8%) | - | - |
| Full-time | 4 (10.8%) | 33 (89.2%) | - | - |
| Maternal work status at 52-weeks | ||||
| Not working | - | - | 461 (95.6%) | 21 (4.4%) |
| Casual | - | - | 33 (97.1%) | 1 (2.9%) |
| Part-time | - | - | 209 (94.6%) | 12 (5.4%) |
| Full-time | - | - | 111 (94.1%) | 7 (5.9%) |
| Maternal country of birth | ||||
| Australia born | 65 (14.9%) | 372 (85.1%) | 410 (96.5%) | 15 (3.5%) |
| Other English-speaking countries | 16 (26.7%) | 44 (73.3%) | 52 (89.7%) | 6 (10.3%) |
| Non-English-speaking countries | 31 (7.1%) | 406 (92.9%) | 396 (95.0%) | 21 (5.0%) |
| Partner country of birth | ||||
| Australia born | 52 (14.0%) | 319 (86.0%) | 349 (96.7%) | 12 (3.3%) |
| Other English-speaking countries | 9 (15.5%) | 49 (84.5%) | 48 (85.7%) | 8 (14.3%) |
| Non-English-speaking countries | 33 (7.8%) | 389 (92.2%) | 389 (95.3%) | 19 (4.7%) |
| Index for relative socioeconomic advantage and disadvantage | ||||
| Least disadvantaged | 16 (7.2%) | 205 (92.8%) | 201 (93.9%) | 13 (6.1%) |
| Low disadvantaged | 24 (15.0%) | 136 (85.0%) | 141 (90.4%) | 15 (9.6%) |
| Moderately disadvantaged | 7 (23.3%) | 23 (76.7%) | 27 (90.0%) | 3 (10.0%) |
| Highly disadvantaged | 27 (12.3%) | 193 (87.7%) | 204 (97.1%) | 6 (2.9%) |
| Most disadvantaged | 38 (12.5%) | 265 (87.5%) | 285 (98.3%) | 5 (1.7%) |
| Maternal smoking during pregnancy | ||||
| No | 95 (10.8%) | 785 (89.2%) | 812 (95.5%) | 38 (4.5%) |
| Yes | 16 (30.2%) | 37 (69.8%) | 45 (91.8%) | 4 (8.2%) |
| Maternal alcohol consumption during pregnancy | ||||
| No | 95 (11.4%) | 737 (88.6%) | 762 (95.0%) | 40 (5.0%) |
| Yes | 16 (16.0%) | 84 (84.0%) | 94 (97.9%) | 2 (2.1%) |
| Parity | ||||
| Primiparous | 57 (12.3%) | 408 (87.7%) | 421 (94.2%) | 26 (5.8%) |
| Multiparous | 55 (11.7%) | 414 (88.3%) | 437 (96.5%) | 16 (3.5%) |
| Infant characteristics | ||||
| Infant gender | ||||
| Male | 59 (12.4%) | 418 (87.6%) | 433 (93.9%) | 28 (6.1%) |
| Female | 53 (11.6%) | 404 (88.4%) | 425 (96.8%) | 14 (3.2%) |
| Infant gestational age | ||||
| Pre-term | 5 (7.1%) | 65 (92.9%) | 64 (97.0%) | 2 (3.0%) |
| Full-term | 107 (12.4%) | 757 (87.6%) | 794 (95.2%) | 40 (4.8%) |
| Infant birth-weight | ||||
| ≥2500g | 106 (12.0%) | 781 (88.0%) | 816 (95.3%) | 40 (4.7%) |
| <2500g | 6 (12.8%) | 41 (87.2%) | 42 (95.45%) | 2 (4.55%) |
| Method of delivery | ||||
| Vaginal | 79 (12.1%) | 573 (87.9%) | 595 (95.0%) | 31 (5.0%) |
| Caesarean | 32 (11.4%) | 249 (88.6%) | 262 (96.0%) | 11 (4.0%) |
| Infant feeding method at 4-weeks age | ||||
| Only breastfeeding | 52 (9.0%) | 523 (91.0%) | 526 (95.0%) | 28 (5.0%) |
| Only formula feeding | 43 (28.3%) | 109 (71.7%) | 140 (96.6%) | 5 (3.4%) |
| Both breastfeeding and formula feeding | 16 (7.8%) | 189 (92.2%) | 190 (95.5%) | 9 (4.5%) |
| Characteristics | Unadjusted Odds Ratio a | Adjusted Odds Ratio b | ||||
|---|---|---|---|---|---|---|
| OR (95% CI) | p-Value | Overall p-Value | OR (95% CI) | p-Value | Overall p-Value | |
| Family Characteristics ** | ||||||
| Maternal Age | 0.90 (0.87–0.94) | <0.001 | <0.001 | 0.92 (0.88–0.96) | <0.001 | <0.001 |
| Maternal education | ||||||
| Left school before 12 | 1.00 | |||||
| Completed school | 0.98 (0.57–1.70) | 0.990 | 1.24 (0.68–2.25) | 0.472 | ||
| College/TAFE | 0.61 (0.33–1.13) | 0.117 | <0.001 | 0.75 (0.38–1.47) | 0.414 | 0.037 |
| University | 0.34 (0.19–0.59) | <0.001 | 0.53 (0.28–0.99) | 0.047 | ||
| Maternal marital status | ||||||
| Married | 1.00 | Not retained in the final model | ||||
| Living with partner | 1.79 (1.02–3.15) | 0.040 | <0.001 | |||
| Single | 2.97 (1.73–5.09) | <0.001 | ||||
| Maternal work status at 17-weeks | ||||||
| Not working | 1.00 | |||||
| Casual | 1.97 (0.72–5.37) | 0.185 | 2.0 (0.67–5.99) | 0.212 | ||
| Part-time | 2.31 (1.10–4.84) | 0.025 | 0.126 | 3.42 (1.54–7.62) | 0.003 | 0.017 |
| Full-time | 0.95 (0.33–2.75) | 0.933 | 1.25 (0.41–3.80) | 0.688 | ||
| Maternal country of birth | ||||||
| Australia born | 1.00 | |||||
| Other English-speaking countries | 0.31 (0.11–0.85) | 0.023 | 0.099 | 2.21 (1.11–4.40) | 0.023 | <0.001 |
| Non-English-speaking countries | 0.68 (0.35–1.35) | 0.282 | 0.53 (0.32–0.86) | 0.011 | ||
| Partner country of birth | ||||||
| Australia born | 1.00 | Not retained in the final model | ||||
| Other English-speaking countries | 0.20 (0.08–0.53) | 0.001 | 0.010 | |||
| Non-English-speaking countries | 0.70 (0.33–1.47) | 0.351 | ||||
| Index for relative socioeconomic advantage and disadvantage | ||||||
| Least disadvantaged | 1.00 | Not retained in the final model | ||||
| Low disadvantaged | 2.26 (1.15–4.41) | 0.017 | ||||
| Moderately disadvantaged | 3.89 (1.45–10.46) | 0.007 | 0.044 | |||
| Highly disadvantaged | 1.79 (0.93–3.42) | 0.078 | ||||
| Most disadvantaged | 1.83 (0.99–3.38) | 0.051 | ||||
| Maternal smoking during pregnancy | ||||||
| No | 1.00 | |||||
| Yes | 3.57 (0.95–5.70) | <0.001 | <0.001 | Not retained in the final model | ||
| Maternal alcohol consumption during pregnancy | ||||||
| No | 1.00 | Not retained in the final model | ||||
| Yes | 1.47 (0.83–2.62) | 0.184 | 0.198 | |||
| Infant Characteristics ** | ||||||
| Infant gestational age | ||||||
| Pre-term | 1.00 | Not retained in the final model | ||||
| Full-term | 1.83 (0.72–4.66) | 0.201 | 0.166 | |||
| Infant feeding method at 4 weeks of age | ||||||
| Only breastfeeding | 1.00 | |||||
| Only formula feeding | 3.96 (2.52–6.24) | <0.001 | <0.001 | 3.26 (1.99–5.33) | <0.001 | <0.001 |
| Both breastfeeding and formula feeding | 0.85 (0.47–1.52) | 0.59 | 0.91 (0.49–1.69) | 0.787 | ||
| Characteristics | Unadjusted Odds Ratio a | Adjusted Odds Ratio b | ||||
|---|---|---|---|---|---|---|
| OR (95% CI) | p-Value | Overall p-Value | OR (95% CI) | p-Value | Overall p-Value | |
| Family Characteristics ** | ||||||
| Maternal Age | 0.94 (0.89–1.00) | 0.055 | 0.055 | Not retained in the final model | ||
| Maternal education | ||||||
| Left school before 12 | 1.00 | Not retained in the final model | ||||
| Completed school | 0.28 (0.03–2.61) | 0.270 | ||||
| College/TAFE | 0.12 (0.01–1.01) | 0.051 | <0.001 | |||
| University | 0.07 (0.01–0.58) | 0.013 | ||||
| Maternal marital status | ||||||
| Married | 1.00 | Not retained in the final model | ||||
| Living with partner | 1.39 (0.48–3.98) | 0.54 | 0.134 | |||
| Single | 4.68 (0.63–34.54) | 0.13 | ||||
| Maternal country of birth | ||||||
| Australia born | 1.00 | |||||
| Other English-speaking countries | 0.31 (0.11–0.85) | 0.023 | 0.098 | Not retained in the final model | ||
| Non-English-speaking countries | 0.68 (0.35–1.35) | 0.282 | ||||
| Partner country of birth | ||||||
| Australia born | 1.00 | |||||
| Other English-speaking countries | 0.20 (0.08–0.53) | 0.001 | 0.009 | 0.18 (0.07–0.49) | 0.001 | 0.002 |
| Non-English-speaking countries | 0.70 (0.33–1.47) | 0.351 | 0.42 (0.19–0.94) | 0.035 | ||
| Index for relative socioeconomic advantage and disadvantage | ||||||
| Least disadvantaged | 1.00 | |||||
| Low disadvantaged | 0.60 (0.28–1.31) | 0.207 | 0.60 (0.25–1.43) | 0.254 | ||
| Moderately disadvantaged | 0.58 (0.15–1.55) | 0.421 | 0.001 | 0.39 (0.09–1.55) | 0.183 | 0.004 |
| Highly disadvantaged | 2.19 (0.81–5.89) | 0.118 | 2.06 (0.71–5.92) | 0.179 | ||
| Most disadvantaged | 3.68 (1.29–10.50) | 0.015 | 3.72 (1.17–11.78) | 0.025 | ||
| Maternal alcohol consumption during pregnancy | ||||||
| No | 1.00 | Not retained in the final model | ||||
| Yes | 2.46 (0.58–10.3) | 0.218 | 0.159 | |||
| Parity | ||||||
| Primiparous | 1.00 | Not retained in the final model | ||||
| Multiparous | 1.68 (0.89–3.18) | 0.018 | 0.102 | |||
| Infant Characteristics ** | ||||||
| Infant gender | ||||||
| Male | 1.00 | Not retained in the final model | ||||
| Female | 1.96 (1.01–3.78) | 0.044 | 0.038 | |||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manohar, N.; Hayen, A.; Bhole, S.; Arora, A. Predictors of Early Introduction of Core and Discretionary Foods in Australian Infants—Results from HSHK Birth Cohort Study. Nutrients 2020, 12, 258. https://doi.org/10.3390/nu12010258
Manohar N, Hayen A, Bhole S, Arora A. Predictors of Early Introduction of Core and Discretionary Foods in Australian Infants—Results from HSHK Birth Cohort Study. Nutrients. 2020; 12(1):258. https://doi.org/10.3390/nu12010258
Chicago/Turabian StyleManohar, Narendar, Andrew Hayen, Sameer Bhole, and Amit Arora. 2020. "Predictors of Early Introduction of Core and Discretionary Foods in Australian Infants—Results from HSHK Birth Cohort Study" Nutrients 12, no. 1: 258. https://doi.org/10.3390/nu12010258
APA StyleManohar, N., Hayen, A., Bhole, S., & Arora, A. (2020). Predictors of Early Introduction of Core and Discretionary Foods in Australian Infants—Results from HSHK Birth Cohort Study. Nutrients, 12(1), 258. https://doi.org/10.3390/nu12010258






