1. Introduction
There is an increasing incidence in prostate cancer worldwide, thought to be due to multiple reasons, including that of a western dietary lifestyle [
1,
2,
3]. Studies have been attempted to provide dietary solutions for prostate cancer as reviewed by DiMarco-Crook et al., 2015, including the joint application of selenium with other nutrients [
4]. Meanwhile, there is variable reporting on the benefits of a 200 μg/d selenium supplementation towards reducing prostate cancer risk [
5,
6]. A reduction in cancer incidence including that of prostate cancer by selenium supplementation was shown with 200 µg/d selenized yeast as the supplementation mode over a mean supplementation period of 6.4 years in a cohort of men with a 115 ng/mL baseline median serum Se level in the Nutrition Prevention of Cancer (NPC) study [
5]. An extended NPC study analysis with a mean supplementation period of 7.4 years showed similar but attenuating benefit trends with overall and prostate cancer incidence, while showing an increased risk of some other cancers, [
6] and increased diabetes risk among those with baseline selenium levels above 121.6 ng/mL [
7]. Meanwhile, no benefits were observed by way of prostate cancer reduction when 200 µg/d selenium as selenomethionine was supplemented for 5.5 years in a cohort of men with a median baseline serum selenium level of 135 ng/mL in the Selenium and Vitamin E Cancer Prevention Trial (SELECT) [
8]. Subsequent analyses from this study have recorded increased overall and high-grade prostate cancer hazards ratios in men within the top two quintiles of baseline toe nail selenium levels, when receiving selenium with 400 IU/day of all-racemic alpha-tocopheryl acetate (AT) as well as in the pooled group of men receiving both selenium and AT and selenium alone [
9]. A twelve-year prospective monitoring study of selenium intakes in men initially diagnosed with non-metastatic prostate cancer has shown increased prostate cancer-specific mortality in those consuming over 140 µg/d in the form of selenium containing supplements [
10]. The most recent Cochrane systematic review on selenium for preventing cancer concludes that the risk ratio for prostate cancer incidence or mortality has no strong negative association with selenium exposure particularly among randomized control studies [
11]. However, the authors view on the SELECT study is that sub-group analyses with specific characteristics including baseline selenium exposure and genetic factors would provide further clarification on selenium impacts on cancer prevention. One such SELECT sub-group study is reported by Martinez et al., 2014 [
12], where an association of the NK3 Homeobox 1 (NKX3.1) rs11781886 CC genotype, combined with selenium administration, is shown to increase both overall prostate cancer risk as well as low grade prostate cancer risk. These authors further report that men carrying both the NKX3.1 rs11781886 CC and CT genotypes have increased prostate cancer risk when supplemented with vitamin E.
In New Zealand, prostate cancer patients record lower serum selenium levels (101.2 ± 1.01 ng/mL) at diagnosis compared to New Zealand men without prostate cancer diagnoses (112.9 ± 1.01 ng/mL) [
13], and prostate cancer patients from the USA (135 ± 21 ng/mL and 132 ± 25 ng/mL for European Americans (EA) and African Americans (AA) respectively) [
14]. The levels recorded for New Zealand men with no known diagnoses of prostate cancers [
13] is also lower than the levels recorded for similar men from the USA (140 ± 28 and 134 ± 21 ng/mL for EA and AA respectively) [
14]. Our studies have also shown that optimal levels of serum selenium towards minimizing DNA damage are recorded between 116 and 150 ng/mL which also have genetic influences [
15]. The influence of age and BMI in influencing serum selenium retention was reported by us previously [
16]. We have also shown that a 200 μg/d selenium supplementation benefits by way of DNA damage reduction is achieved dependent on dietary methionine and folate levels and BMI [
16,
17]. Studies including ours have reported selenium supplementation to lower serum prostate-specific antigen (PSA) levels among men with high-risk for prostate cancer and among middle-aged US men [
18,
19]. Our studies have previously identified several single nucleotide polymorphism (SNP) genotypes including those of selenoproteins that increase the risk for prostate cancer incidence in New Zealand [
13,
20].
The
glutathione peroxidase 1 (
GPX1) is a selenoprotein encoding gene located in the chromosomal region 3p21.3 and records rs1050450 C>T SNP that is responsible for a nonsynonymous amino acid change from proline to leucine [
21,
22]. The catalytic activity of GPX is affected by the rs1050450 T allele [
23], while this allele is also associated with several cancers [
13,
24,
25]. The selenoprotein 15 (SEL15) encoding gene is located in chromosomal region 1p31 and records rs5845 C>T SNP [
26,
27]. This rs5845 C>T SNP which relates to the amino acid position 811 is located in a selenocysteine insertion sequence (SECIS)-like structure within the SEL15 protein, and is associated with the rs5859 G>A SNP which relates to the amino acid position 1125 located within the SECIS element [
27]. According to these authors, the latter polymorphism influences selenocysteine incorporation in SEL15. The SEL15 rs5845 and its associated SNP rs5859 are associated with several cancers [
13,
28,
29]. The mitochondrial manganese superoxide dismutase (MnSOD) encoding gene is located in the chromosomal region 6q25.3 and the rs4880 C>T SNP produces a nonsynonymous alanine to valine amino acid change [
30,
31]. This rs4880 polymorphism also has been associated with various cancers [
32,
33]. The aldo-keto reductase 1C3 (AKR1C3) encoding gene is located in the chromosomal region 10p15 and the rs12529 C>G polymorphism produces a nonsynonymous amino acid change from histidine to glutamine [
34]. This rs12529 polymorphism is also associated with various cancers [
20,
35,
36]. The
kallikrein-related peptidase 3 (
KLK3) gene encodes the serine protease PSA and this gene is located in the chromosomal region 19q13 [
37]. The
KLK3 rs17632542 T>C is a nonsynonymous polymorphism with an amino acid change from isoleucine to threonine and has an association with prostate cancer risk as well as with the PSA level [
20,
38].
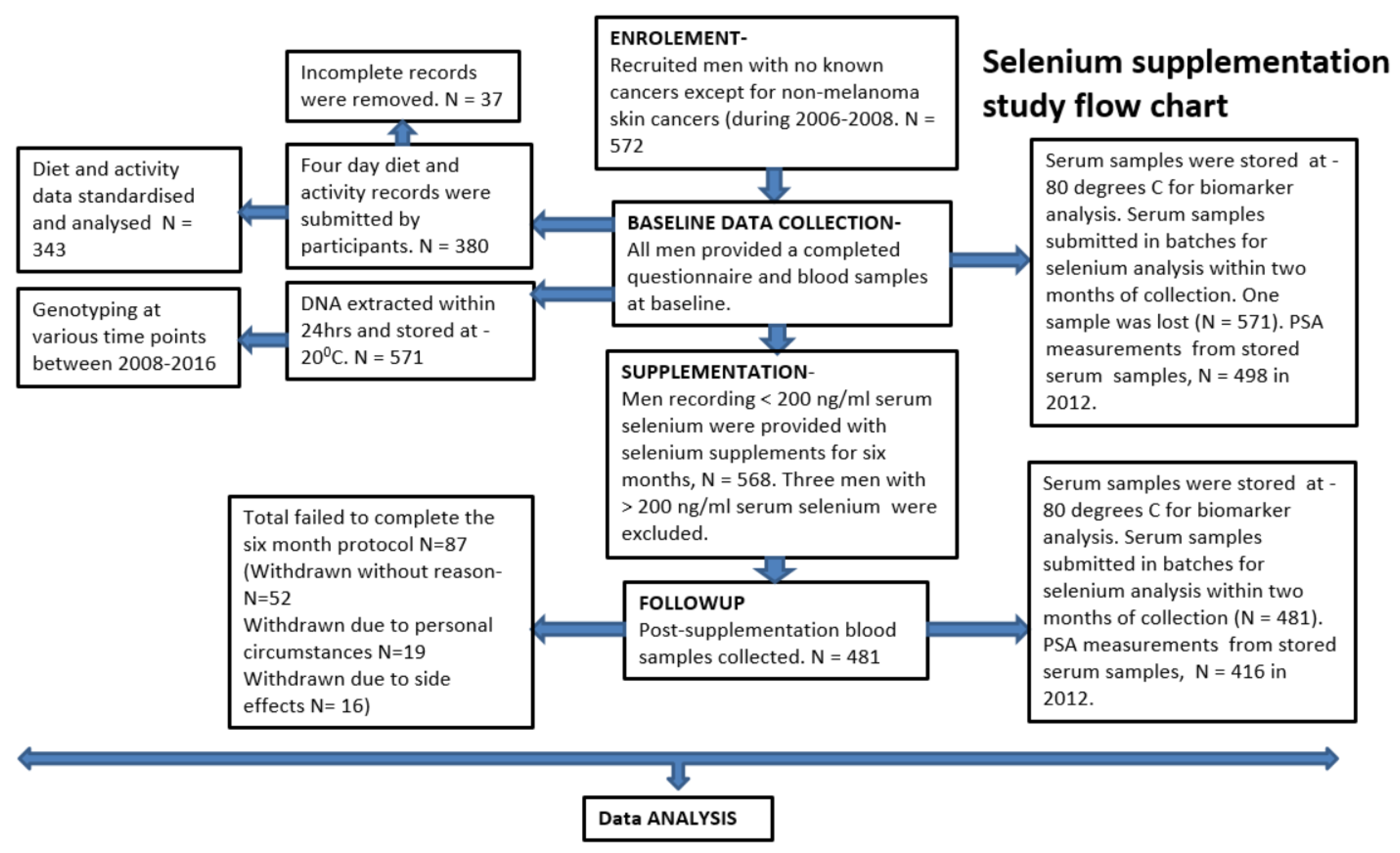
Here, we present an analysis that looked into variable utilization of 200 μg/d selenium in the form of a selenized yeast supplement for six months, and subsequent benefit variation in maintaining prostate glandular epithelium microarchitecture stability, as measured through the surrogate marker serum PSA. This is a stratified analyses based on demographic and lifestyle characteristics, baseline serum selenium levels and a set of genotypes known to associate with prostate cancer risk [
13,
20,
39,
40], as well as a panel of dietary nutrient factors associated with DNA methylation [
41,
42,
43].
4. Discussion
In an era of highest rate of age standardized global incidence of prostate cancer recorded from New Zealand [
52], it is inevitable that we should be looking at every possible source to ease this problem. Restoring general prostate health in New Zealand men could indirectly reduce this long-term public health burden. Meanwhile, New Zealand is known for soil deficiencies in selenium that affects the dietary content of this trace mineral [
53]. Our previous studies with New Zealand men have shown that benefits of 200 μg/d selenium supplementation for six months in preserving DNA integrity is achieved only in stratified groups of men [
16,
45]. We have also shown that DNA integrity benefits vary between serum selenium levels between 116 and 150 ng/mL depending on genetic variation [
15]. The current analysis looked into the utilization of supplemented selenium in changing serum selenium levels and subsequent changes in the stability of prostate glandular epithelium microarchitecture, using PSA as a surrogate biomarker.
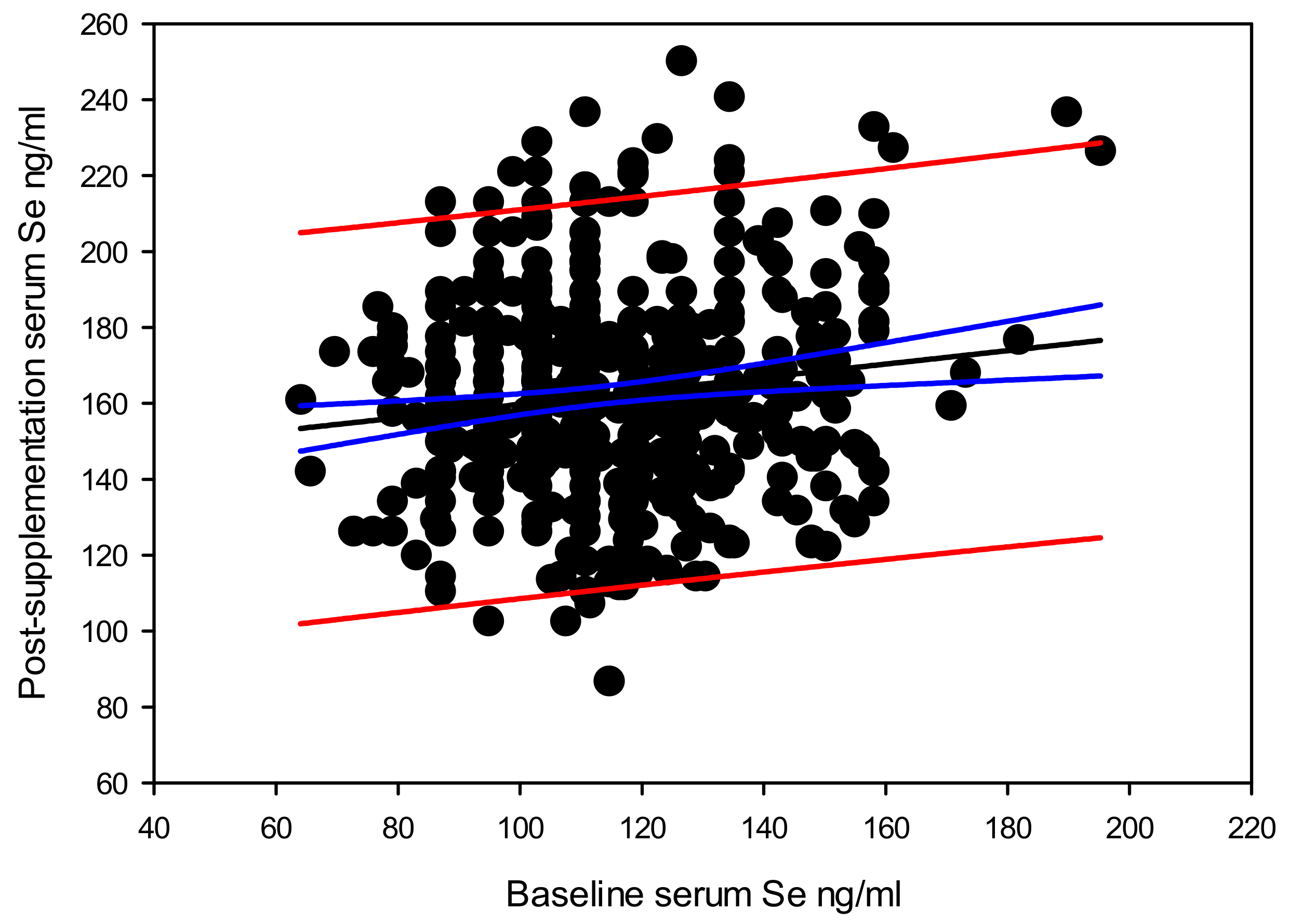
We observed a significant positive dependence of post-supplementation serum selenium level, on the baseline serum selenium level. When stratified by genotypes, the GPX1 rs1050450 TT, AKR1C3 rs12529 CG and SEL15 rs5845 CT showed the best correlation between the baseline and post-supplementation serum selenium levels. This implies that the individual serum selenium carrying capacity could be varying with the genotypes.
Beyond the highest gain in serum selenium by supplementation of 126 ng/mL, the overall gain in serum selenium by supplementation declined at a rate of 0.828 ng/mL with each one ng/mL increase in baseline serum selenium level. This means that part of the supplemented selenium would have passed though different routes in addition to replenishing the serum component. Among such possible selenium channeling routes may include, excretion [
54]; metabolism by gut biota to forms that reduce absorption into circulation [
55]; used up for toxicity modulation [
56]; channeled towards other tissue optimization [
57] including that of the prostate [
58,
59,
60]; or the selenomethionine component in the selenized yeast [
61] getting non-specifically incorporated into a wide range of tissue proteins in place of methionine [
62,
63]. Takata et al., 2009 have shown that serum selenium level is a reflection of the levels in the prostate [
59]. Our previous studies with the current study cohort have shown that selenium supplementation led to no gain in red blood cell
GPX activity, indicating, this has been optimized with the baseline selenium levels; although the thioredoxin reductases activity was upregulated with selenium supplementation [
16,
45]. This is in line with the whole body selenium regulation as discussed by Burk and Hill 2015 [
64].
There was no overall significant correlation between the difference in serum selenium after supplementation and subsequent difference in PSA level, although significant correlations were recorded among, men below the median age of 55 years as well as the group who were never-smokers. Our previous studies have shown that age based PSA increase in men with prostate cancer beyond 69 years could be due to the increase in extra-testicular androgen production from adrenal dehydroepiandrosterone (DHEA)-based androgen precursors [
65] catalyzed by the AKR1C3 enzyme, particularly among men carrying the AKR1C3 rs12529 CG and GG genotypes [
66]. Therefore, our current observation of a significant inverse correlation between the difference in serum selenium and difference in PSA level after selenium supplementation among men below the age of 55 years may indicate that increased selenium level does not support a significant reduction in PSA when the extra-testicular androgen production starts to increase with advancing age. A placebo-controlled study tested against 200µg selenium daily for 8 weeks with women aged 10–40 years with polycystic ovarian syndrome has recorded decreased serum DHEA levels with selenium supplementation [
67]. This further implies that at an age below 55 years, PSA decline by selenium supplementation may happen due to indirect DHEA suppression. Among ever-tobacco smokers, supplemented selenium may get triaged towards seleno-antioxidant enzyme activity [
16] over prostate glandular epithelium stability, restricting prostate health benefits to never-smokers.
When data were stratified by the SNP genotypes, we see a PSA reducing benefit by supplementation only with the
GPX rs1050450 CT and TT genotypes. The EPIC-Heidelberg nested prostate cancer case-control study indicates that men with the variant T allele of the
GPX1 rs1050450 polymorphism record a risk ratio (odds ratio OR) of 0.87 per 10 ng/mL increase in serum selenium [
68]. The authors also showed that the prostate cancer risk ratio was further reduced (OR, 0.64) per 10 ng/mL increase in serum selenium among men with high-grade prostate cancer carrying the rs1050450 variant T allele, while no such risk modifications were observed with increasing serum selenium with the
GPX1 rs1050450 dominant allele C.
PSA is exclusively produced by the glandular prostate epithelium, and is abundant in the seminal fluid [
69]. PSA production is signaled by androgen activated androgen receptors [
65,
67,
70]. According to Prcic and co-workers, PSA reaches serum when the microarchitecture of prostate glandular tissue is disrupted [
71]. According to these authors increased levels in circulation is an indication of prostate disease or trauma in the prostate gland including prostate cancer. Therefore, the decline in PSA with increase in serum selenium among men carrying the
GPX1 rs1050450 T allele in our study is an indirect reflection of prostate health benefits that may parallel prostate cancer risk reduction as recorded by the Steinbrecher et al. study [
68].
In a previous study, an inverse trend in PSA change with increasing baseline serum selenium level was recorded by us for a group of New Zealand men with benign prostate issues but with negative biopsies, and carrying a PSA >4 ng/mL at recruitment [
19]. When these men were supplemented with either a 200 or 400 µg/d selenium as selenized yeast or placebo for six months, two thirds of the cohort showed a reduction in the rate of PSA progression. Zhang et al., 2011 reported that a 3-month supplementation with 200 μg/d selenium (in the form of glycinate) increased plasma and erythrocyte glutathione peroxidase (
GPX) and lowered serum PSA levels [
18]. The Procomb study from Italy, where men with lower urinary tract infections (UTI) randomized to receive either a Serenoa repens plant extract containing 50 µg of selenium and 5 mg of lycopene or a placebo for over one year showed no significant change in PSA between the test and the placebo groups [
72]. It is a possibility that this combination supplement with 50 µg/d of selenium was not sufficient to influence a pharmacological benefit of selenium in PSA reduction in these men with UTI. A six-month 55 µg/d of selenium supplementation with 35 mg/d of lycopene and 600 mg/d of polyphenol supplementation is reported in a group of men diagnosed with isolated high-grade prostatic intraepithelial neoplasia HGPIN on biopsy [
73]. These authors record increased incidence of prostate cancer and markers indicating prostate cancer progression in men receiving the supplements. A six-month supplementation of 100 µg/d selenium, 30 mg/d vitamin E, and 50 mg/d soy isoflavonoids in men diagnosed with HGPIN has resulted in a stable PSA within three months in 4.2% of the supplemented group, while a decrease in PSA level was recorded in 57.8% [
74]. These authors also record that 34%, 48% and 18.3% of these men having either prostate cancer or remaining with HGPIN or no HGPIN or carcinoma respectively after six months. They further recorded that the prostate cancer risk remained at 25% among men having a stable or decreasing PSA due to supplementation (68%) while the risk reaching 52% in the group with increasing PSA after supplementation (32%). Richie et al., 2014 supplemented 200 µg/d selenium as selenomethionine or 200 or 285 µg/d selenized yeast or placebo for nine months in a group of 69 healthy men, in the age range of 20–79 y who were non-smokers and carrying PSA levels (<4 ng/mL) at baseline [
75]. The authors report no change in PSA level compared to the baseline in any of the groups consisting of 15–20 men. Contrary to 110 ng/mL baseline median serum selenium (or 113 ng/mL mean serum selenium) level in our cohort, their cohort recorded a higher mean baseline plasma selenium level ranging from 129 to 139 ng/mL. Our cohort gained a post-supplementation median serum selenium levels ranging from 157 ng/mL among the
KLKrs17632542 CT genotype carriers to 181 ng/mL in
SEL15 rs5845 TT genotype carriers within six months of supplementation, while the Richie et al., (2014) study recorded mean post-supplementation levels above 200 ng/mL within six months for all three types of supplements. Waters and Chiang (2018) discuss the influence of both inadequate as well as excess selenium in prostate health in dogs [
76]. The inverse correlation of serum PSA level with increasing serum selenium level in our cohort with
GPX rs1050450 T alleles could be due to our men with lower baseline serum selenium levels showing a differential benefit compared to men from Ritchie et al. study [
75]. We have previously recorded that tobacco smoking is associated with increased levels of serum PSA levels in New Zealand men [
77]. The impact of tobacco smoking on prostate tissue blood perfusion and vascular injury have been reported in men with benign prostatic hyperplasia [
78]. It is a possibility that increased serum PSA due to tobacco smoking related prostatic glandular epithelium damage could not be circumvented by the selenium supplementation protocol followed by us.
The post-supplementation selenium level showed a significant difference among those carrying the
SEL15 rs5845 genotypes, with the TT genotype showing the highest level. However, this is contrary to the findings in studies with functional gene constructs, where selenium supplementation response was shown superior with the
SEL15 rs5845 C allele [
27]. A US study by Ekoue et al., 2018 have reported that AAs have a higher frequency (34%) of the TT genotype compared to EAs (3.4%) [
60], and this genotype distribution in our New Zealand cohort is similar (3.2%) to the latter group. These authors also record that AAs record a lower level of SEL15 protein in their prostate tissue samples and in the serum compared to the European Americans, and that SEL15 levels are generally lower in prostate cancer tissue compared to adjacent benign tissues. They also record that each ng/mL increase in serum selenium level increases the odds ratio for Gleason sum by 2% in men carrying the
SEL15 rs5845 TT genotype, while in men carrying the CC genotype a five-fold decrease in the odds ratio of higher Gleason sum is recorded. Considering the highest retention of serum selenium by supplementation among men carrying the
SEL15 rs5845 TT genotype in our cohort without an accompanying decline in PSA level, they may be a group that should consider serum selenium adjustments cautiously.
The interaction of a panel of dietary nutrients associated with DNA methylation, shows that intakes above the RDI for zinc, benefitting with a significant reduction in PSA levels with increasing selenium levels due to supplementation. Reduction in PSA with increasing serum selenium levels was more stringent with vitamin B12 levels below the RDI. If the selenium supplementation-related PSA reduction was at least partially a consequence of increasing the activity of the antioxidant seleno enzyme
GPX, it is imperative that nutrient levels of both selenium and zinc be sufficient. The redox action of seleno enzyme
GPX is preceded by prior production of H
2O
2 through the superoxide dismutases. Such superoxide dismutases include the mitochondrial MnSOD and cytosolic Cu/Zn SOD, which catalyze reduction of the superoxide radicals to H
2O
2. Therefore, besides being an important molecule in the DNA methylation process [
41], zinc is also an important trace mineral in the Cu/Zn SOD metaloenzyme, supporting redox cycling of reactive oxygen species. Findings of the 2008/09 New Zealand Adult Nutrition Survey indicates that 39% of men had inadequate intakes of zinc (median daily intake 12.9 mg) while in the category of men >71 years, this inadequacy was common among 89.7% of men [
79]. The New Zealand Adult Nutrition Survey indicates that 32% of the New Zealand males also have inadequate intakes of selenium (67 μg/d that is closer to our current estimate of 65 μg/d). The comparative dietary intake of selenium recorded for US males >20 years was 135.9 μg/d [
80]. The major source of selenium in New Zealand diets comes from breads (15%) followed by fish, and sea food (12%) and poultry (10%) [
79]. An inadequate intake of vitamin B12 recorded in the 2008/09 New Zealand Adult Nutrition Survey [
79] was only 1.3% for men whereas our cohort had 4.4% men with this inadequacy. The above details indicate the importance of dietary nutrient adjustments in achieving optimum prostate glandular epithelial health benefits of selenium as shown through the surrogate serum PSA marker.
Our study implies that it could be beneficial for New Zealand men carrying the
GPX1 rs1050450 T alleles, to get their serum selenium levels adjusted, at least to reach the level between 116 and 150 ng/mL as estimated by us as the optimum level for DNA integrity [
15,
45]. Besides, our data indicate the importance of serum selenium level adjustments in men carrying the
GPX1 rs1050450 T alleles for prostate glandular epithelial architecture stability that can be monitored with serum PSA levels, rather than using a set dose of selenium supplements. This study also highlights the importance of other nutritional requirements such as dietary zinc and vitamin B12 in maximizing selenium related benefits on prostate health.
Among the shortcomings of the current study are the duration of the study being limited to six months, the nutrient measures except serum selenium were assessed based on the four-day diet and activity diaries that may not provide an accurate nutrient profile in blood of the participants. Another shortcoming is the nutrient profiles were analysed only with a limited panel associated with DNA methylation. It will be beneficial to assess a wider range of serum/plasma based nutrient profiles for better understanding of the interactive nature of nutrients with selenium supplementation and PSA outcomes. Our study has also not recorded urinary selenium excretion, which would have further supported our claim on variable supplemented selenium retention levels based on baseline serum selenium level. As prostate cancer has a long aetiology, it is important that men from this New Zealand selenium supplementation study be prospectively monitored for any future prostate cancer recordings, and to understand the interactive genetic, nutrient and serum PSA measures in such prostate cancer outcomes.