Association of Blood Mercury Level with the Risk of Depression According to Fish Intake Level in the General Korean Population: Findings from the Korean National Health and Nutrition Examination Survey (KNHANES) 2008–2013
Abstract
1. Introduction
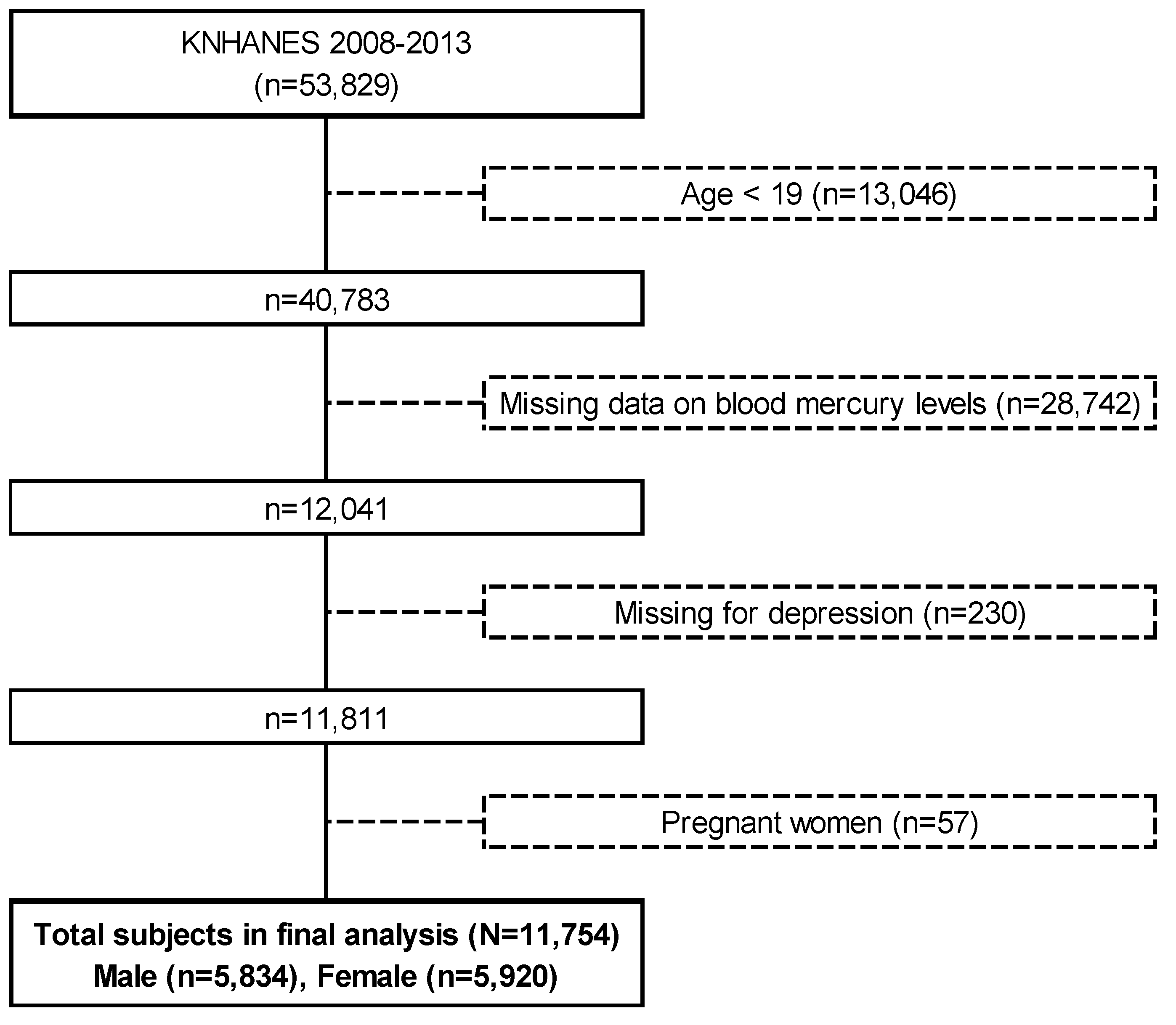
2. Materials and Methods
2.1. Study Design and Population
2.2. Depression Assessment
2.3. Measurements
2.3.1. Determination of Mercury Levels in Blood
2.3.2. Dietary Fish Intake Assessment
2.3.3. Other Covariates
2.4. Statistical Analysis
3. Results
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Kim, C.W.; Kim, Y.W.; Chae, C.H.; Son, J.S.; Koh, J.C.; Kim, D.S. The Effects of the Frequency of Fish Consumption on the Blood Mercury Levels in Koreans. Korean J. Occup. Environ. Med. 2010, 22, 114–121. [Google Scholar] [CrossRef]
- Ng, T.H.; Mossey, J.M.; Lee, B.K. Total blood mercury levels and depression among adults in the United States: National Health and Nutrition Examination Survey 2005-2008. PLoS ONE 2013, 8, e79339. [Google Scholar] [CrossRef] [PubMed]
- Sanfeliu, C.; Sebastia, J.; Cristòfol, R.; Rodríguez-Farré, E. Neurotoxicity of organomercurial compounds. Neurotox. Res. 2003, 5, 283–305. [Google Scholar] [CrossRef] [PubMed]
- Houston, M.C. The role of mercury and cadmium heavy metals in vascular disease, hypertension, coronary heart disease, and myocardial infarction. Altern. Ther. Health Med. 2007, 13, 128–133. [Google Scholar]
- Harada, M. Minamata disease: Methylmercury poisoning in Japan caused by environmental pollution. Crit. Rev. Toxicol. 1995, 25, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Bakir, F.; Damluji, S.F.; Amin-Zaki, L.; Murtadha, M.; Khalidi, A.; Al-Rawi, N.Y.; Tikriti, S.; Dahahir, H.I.; Clarkson, T.W.; Smith, J.C.; et al. Methylmercury poisoining in Iraq. Science 1973, 181, 230–241. [Google Scholar] [CrossRef]
- Brenna, J.T.; Salem, N.; Sinclair, A.J.; Cunnane, S.C. International Society for the Study of Fatty Acids and Lipids. α-Linolenic acid supplementation and conversion to n-3 long-chain polyunsaturated fatty acids in humans. Prostaglandins Leukot. Essent. Fat. Acids 2009, 80, 85–91. [Google Scholar] [CrossRef]
- Matsuoka, Y.J.; Sawada, N.; Mimura, M.; Shikimoto, R.; Nozaki, S.; Hamazaki, K.; Uchitomi, Y.; Tsugane, S. Dietary fish, n-3 polyunsaturated fatty acid consumption, and depression risk in Japan: A population-based prospective cohort study. Transl. Psychiatry 2007, 7, e1242. [Google Scholar] [CrossRef]
- Yoshikawa, E.; Nishi, D.; Matsuoka, Y. Fish consumption and resilience to depression in Japanese company workers: A cross-sectional study. Lipids Health Dis. 2018, 14. [Google Scholar] [CrossRef]
- Burdge, G. Alpha-linolenic acid metabolism in men and women: Nutritional and biological implications. Curr. Opin. Clin. Nutr. Metab. Care 2004, 7, 137–144. [Google Scholar] [CrossRef]
- Fernandes, A.C.; Medeiros, C.O.; Bernardo, G.L.; Ebone, M.V.; Di Pietro, P.F.; Assis, M.A.; Vasconcelos, F.D. Benefits and risks of fish consumption for the human health. Rev. Nutr. 2012, 25, 283–295. [Google Scholar] [CrossRef]
- Riemer, S.; Maes, M.; Christophe, A.; Rief, W. Lowered omega-3 PUFAs are related to major depression, but not to somatization syndrome. J. Affect. Disord. 2010, 123, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Levant, B.; Ozias, M.K.; Carlson, S.E. Specific Brain Regions of Female Rats Are Differentially Depleted of Docosahexaenoic Acid by Reproductive Activity and an (n-3) Fatty Acid-Deficient Diet. J. Nutr. 2007, 137, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Timonen, M.; Horrobin, D.; Jokelainen, J.; Laitinen, J.; Herva, A.; Räsänen, P. Fish consumption and depression: The Northern Finland 1966 birth cohort study. J. Affect. Disord. 2004, 82, 447–452. [Google Scholar] [CrossRef]
- Jang, C.W.; Kim, S.H.; Choi, J.D. The Influence of Fish Consumption on Umbilical Cord Blood Mercury Level in Pregnant Women in the City of Tongyeong, Korea. J. Food Hyg. Saf. 2012, 27, 74–80. [Google Scholar] [CrossRef]
- Nielsen, S.J.; Kit, B.K.; Aoki, Y.; Ogden, C.L. Seafood consumption and blood mercury concentrations in adults aged ≥20 y, 2007–2010. Am. J. Clin. Nutr. 2014, 99, 1066–1070. [Google Scholar] [CrossRef]
- Birch, R.J.; Bigler, J.; Rogers, J.W.; Zhuang, Y.; Clickner, R.P. Trends in blood mercury concentrations and fish consumption among U.S. women of reproductive age, NHANES, 1999–2010. Environ. Res. 2014, 133, 431–438. [Google Scholar] [CrossRef]
- Castro-González, M.I.; Mendez-Armenta, M. Heavy metals: Implications associated to fish consumption. Environ. Toxicol. Pharmacol. 2008, 26, 263–271. [Google Scholar] [CrossRef]
- Kim, N.S.; Lee, B.K. Blood Total Mercury and Fish Consumption in the Korean General Population in KNHANES III, 2005. Sci. Total Environ. 2010, 408, 4841–4847. [Google Scholar] [CrossRef]
- Maghazaji, H.I. Psychatric aspects of methylmercury poisoning. J. Neurol. Neurosurg. Psychiatry 1974, 37, 954–958. [Google Scholar] [CrossRef]
- Yorifuji, T.; Tsuda, T.; Inoue, S.; Takao, S.; Harada, M. Long term exposure to methylmercury and psychatric symptoms in residents of Minimata, Japan. Environ. Int. 2011, 37, 907–913. [Google Scholar] [CrossRef] [PubMed]
- Kweon, S.; Kim, Y.; Jang, M.J.; Kim, Y.; Kim, K.; Choi, S.; Chun, C.; Khang, Y.H.; Oh, K. Data Resource Profile: The Korea National Health and Nutrition Examination Survey (KNHANES). Int. J. Epidemiol. 2014, 43, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.E.; Joo, S.H.; Hahn, C.; Kim, D.J.; Kim, T.S. Association between Alcohol Consumption and Stress Perception, Depressed Mood, and Suicidal Ideation: The 2010–2015 KNHANES. Psychiatry Investig. 2019, 16, 386–396. [Google Scholar] [CrossRef] [PubMed]
- Gigantesco, A.; Morosini, P. Development, reliability and factor analysis of a self-administered questionnaire which originates from the World Health Organization’s Composite International Diagnostic Interview-Short Form (CIDI-SF) for assessing mental disorders. Clin. Pract. Epidemiol. Ment. Health 2008, 4, 8. [Google Scholar] [CrossRef]
- Park, Y.; Kim, S.H.; Lim, Y.T.; Ha, Y.C.; Chang, J.S.; Kim, I.S.; Min, Y.K.; Chung, H.Y. Validation of a New Food Frequency Questionnaire for Assessment of Calcium and Vitamin D Intake in Korean Women. J. Bone Metab. 2013, 20, 67–74. [Google Scholar] [CrossRef]
- Hagströmer, M.; Oja, P.; Sjöström, M. The international physical activity questionnaire (IPAQ): A study of concurrent and construct validity. Public Health Nutr. 2006, 9, 755–762. [Google Scholar] [CrossRef]
- The International Physical Activity Questionnaire(IPAQ). Guidelines for Data Processing and Analysis of the International Physical Activity Questionnaire (Ipaq)–Short and Long Forms; IPAQ: Stockholm, Sweden, 2005. [Google Scholar]
- Desquilbet, L.; Mariotti, F. Dose–response analyses using restricted cubic spline functions in public health research. Stat. Med. 2010, 29, 1037–1057. [Google Scholar] [CrossRef]
- Maximino, C.; Araujo, J.; Leão, L.K.; Grisolia, A.B.; Oliveira, K.R.; Lima, M.G.; Batista Ede, J.; Crespo-López, M.E.; Gouveia, A., Jr.; Herculano, A.M. Possible role of serotoninergic system in the neurobehavioral impairment induced by acute methylmercury exposure in zebrafish (Danio rerio). Neurotoxicol. Teratol. 2011, 33, 727–734. [Google Scholar] [CrossRef]
- Landrigan, P.J. Occupational and Community Exposures to Toxic Metals: Lead, Cadmium, Mercury and Arsenic. West J. Med. 1982, 137, 531–539. [Google Scholar]
- He, K.; Song, Y.; Daviglus, M.L.; Liu, K.; Van Horn, L.; Dyer, A.R.; Goldbourt, U.; Greenland, P. Fish Consumption and Incidence of Stroke: A Meta-Analysis of Cohort Studies. Stroke 2004, 35, 1538–1542. [Google Scholar] [CrossRef]
- Morris, M.C.; Evans, D.A.; Tangney, C.C.; Bienias, J.L.; Wilson, R.S. Fish Consumption and Cognitive Decline with Age in a Large Community Study. Arch. Neurol. 2005, 62, 1849–1853. [Google Scholar] [CrossRef] [PubMed]
- Rice, K.M.; Walker, E.M.; Wu, M.; Gillette, C.; Blough, E.R. Environmental Mercury and Its Toxic Effects. J. Prev. Med. Public Health 2014, 47, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Choi, A.L.; Cordier, S.; Weihe, P.; Grandjean, P. Negative Confounding in the Evaluation of Toxicity: The Case of Methylmercury in Fish and Seafood. Crit. Rev. Toxicol. 2008, 38, 877–893. [Google Scholar] [CrossRef] [PubMed]
- Mahaffey, K.R. Fish and shellfish as dietary sources of methylmercury and the ω-3 fatty acids, eicosahexaenoic acid and docosahexaenoic acid: Risks and benefits. Environ. Res. 2004, 95, 414–428. [Google Scholar] [CrossRef] [PubMed]
- Jo, E.M.; Kim, B.G.; Kim, Y.M.; Yu, S.D.; You, C.H.; Kim, J.Y.; Hong, Y.S. Blood Mercury Concentration and Related Factors in an Urban Coastal Area in Korea. J. Prev. Med. Public Health 2010, 43, 377–386. [Google Scholar] [CrossRef]
- Ilmiawati, C.; Yoshida, T.; Itoh, T.; Nakagi, Y.; Saijo, Y.; Sugioka, Y.; Sakamoto, M.; Ikegami, A.; Ogawa, M.; Kayama, F. Biomonitoring of mercury, cadmium, and lead exposure in Japanese children: A cross-sectional study. Environ. Health Prev. Med. 2015, 20, 18–27. [Google Scholar] [CrossRef]
- Karouna-Renier, N.K.; Ranga, R.K.; Lanza, J.J.; Rivers, S.D.; Wilson, P.A.; Hodges, D.K.; Levine, K.E.; Ross, G.T. Mercury levels and fish consumption practices in women of child-bearing age in the Florida Panhandle. Environ. Res. 2008, 108, 320–326. [Google Scholar] [CrossRef]
- Sakamoto, M.; Kaneoka, T.; Murata, K.; Nakai, K.; Satoh, H.; Akagi, H. Correlations between mercury concentrations in umbilical cord tissue and other biomarkers of fetal exposure to methylmercury in the Japanese population. Environ. Res. 2007, 103, 106–111. [Google Scholar] [CrossRef]
- You, C.H.; Kim, B.G.; Jo, E.M.; Kim, G.Y.; Yu, B.C.; Hong, M.G.; Kim, D.S.; Hong, Y.S. The relationship between the fish consumption and blood total/methyl-mercury concentration of costal area in Korea. Neurotoxicology 2012, 33, 676–682. [Google Scholar] [CrossRef]
- McKelvey, W.; Gwynn, R.C.; Jeffery, N.; Kass, D.; Thorpe, L.E.; Garg, R.K.; Palmer, C.D.; Parsons, P.J. A Biomonitoring Study of Lead, Cadmium, and Mercury in the Blood of New York City Adults. Environ. Health Perspect. 2007, 115, 1435–1441. [Google Scholar] [CrossRef]
- Hightower, J.M.; Moore, D. Mercury levels in high-end consumers of fish. Environ. Health Perspect. 2003, 111, 604–608. [Google Scholar] [CrossRef] [PubMed]
- Batáriová, A.; Spevácková, V.; Benes, B.; Cejchanová, M.; Smíd, J.; Cerná, M. Blood and urine levels of Pb, Cd and Hg in the general population of the Czech Republic and proposed reference values. Int. J. Hyg. Environ. Health 2006, 209, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Mirsaeidi, M.; Sadikot, R.T. Gender susceptibility to mycobacterial infections in patients with non-CF bronchiectasis. Int. J. Mycobact. 2015, 4, 92–96. [Google Scholar] [CrossRef] [PubMed]
- Hightower, J.M.; Hare, A.O.; Hernandez, G.T. Blood Mercury Reporting in NHANES: Identifying Asian, Pacific Islander, Native American, and Multiracial Groups. Environ. Health Perspect. 2006, 114, 173–175. [Google Scholar] [CrossRef] [PubMed]
- Eom, S.Y.; Lee, Y.S.; Lee, S.G.; Seo, M.N.; Choi, B.S.; Kim, Y.D.; Lim, J.A.; Hwang, M.S.; Kwon, H.J.; Kim, Y.M.; et al. Lead, Mercury, and Cadmium Exposure in the Korean General Population. J. Korean Med. Sci. 2018, 33. [Google Scholar] [CrossRef]
- Becker, K.; Kaus, S.; Krause, C.; Lepom, P.; Schulz, C.; Seiwert, M.; Seifert, B. German Environmental Survey 1998 (GerES III): Environmental pollutants in blood of the German population. Int. J. Hyg. Environ. Health 2002, 205, 297–308. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention (CDC). Fourth National Report on Human Exposure to Environmental Chemicals; CDC: Atlanta, GA, USA, 2010. [Google Scholar]
- Health Canada. Third Report on Human Biomonitoring of Environmental Chemicals in Canada: Results of the Canadian Health Measures Survey Cycle 3 (2012–2013); Health Canada: Ottawa, ON, Canada, 2015.
- Lee, S.; Yoon, J.H.; Won, J.U.; Lee, W.; Lee, J.H.; Seok, H.; Kim, Y.K.; Kim, C.N.; Roh, J. The Association Between Blood Mercury Levels and Risk for Overweight in a General Adult Population: Results from the Korean National Health and Nutrition Examination Survey. Biol. Trace Elem. Res. 2016, 171, 251–261. [Google Scholar] [CrossRef]
- Moon, S.S. Association between Blood Mercury Level and Visceral Adiposity in Adults. Diabetes Metab. J. 2017, 41, 96–98. [Google Scholar] [CrossRef]
- Moon, S.S. Additive effect of heavy metals on metabolic syndrome in the Korean population: The Korea National Health and Nutrition Examination Survey (KNHANES) 2009-2010. Endocrine 2014, 46, 263–271. [Google Scholar] [CrossRef]
- You, C.H.; Kim, B.G.; Kim, J.M.; Yu, S.D.; Kim, Y.M.; Kim, R.B.; Hong, Y.S. Relationship Between Blood Mercury Concentration and Waist-to-Hip Ratio in Elderly Korean Individuals Living in Coastal Areas. J. Prev. Med. Public Health 2011, 44, 218–225. [Google Scholar] [CrossRef]
- Knobeloch, L.; Anderson, H.A.; Imm, P.; Peters, D.; Smith, A. Fish consumption, advisory awareness, and hair mercury levels among women of childbearing age. Environ. Res. 2005, 97, 220–227. [Google Scholar] [CrossRef]
- Ye, S.; Shin, J.; Lee, J.; Jung, E.M.; Lee, J.; Yun, E.; Kim, Y.; Oh, Y.; Ha, E.H. Systematic Review of Heavy Metal Concentrations in Fish and Shellfish in Korea. Ewha Med. J. 2018, 41, 1–7. [Google Scholar] [CrossRef]
- Kothari, S.; Kruse, D.; Karimi, R.; Silbernagel, S.; Gursoy, N.; Jaber, R.; Roppelt, H.; Awan, R.; Gold, A.; Meliker, J.R. High mercury seafood consumption associated with fatigue at specialty medical clinics in Long Island, New York. Prev. Med. Rep. 2015, 2, 798–802. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Je, Y. Fish consumption and depression in Korean adults: The Korea National Health and Nutrition Examination Survey, 2013–2015. Eur. J. Clin. Nutr. 2018, 72, 1142–1149. [Google Scholar] [CrossRef] [PubMed]
- Strøm, M.; Mortensen, E.L.; Halldorsson, T.I.; Thorsdottir, I.; Olsen, S.F. Fish and long-chain n-3 polyunsaturated fatty acid intakes during pregnancy and risk of postpartum depression: A prospective study based on a large national birth cohort. Am. J. Clin. Nutr. 2009, 90, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.C.; Guo, Y.; Zhou, M.M.; Xue, C.H.; Chang, Y.G.; Zhang, T.T.; Wang, Y.M. Comparative studies of DHA-enriched phosphatidylcholine and recombination of DHA-ethyl ester with egg phosphatidylcholine on ameliorating memory and cognitive deficiency in SAMP8 mice. Food Funct. 2019, 10, 938–950. [Google Scholar] [CrossRef]
- Tanskanen, A.; Hibbeln, J.R.; Tuomilehto, J.; Uutela, A.; Haukkala, A.; Viinamäki, H.; Lehtonen, J.; Vartiainen, E. Fish Consumption and Depressive Symptoms in the General Population in Finland. Psychiatr. Serv. 2001, 52, 529–531. [Google Scholar] [CrossRef]
| Characteristics | Quintiles of Blood Mercury a | p-Value b | Total Subjects | ||||
|---|---|---|---|---|---|---|---|
| Q1 | Q2 | Q3 | Q4 | Q5 | (n = 11,754) | ||
| (n = 2350) | (n = 2351) | (n = 2352) | (n = 2350) | (n = 2351) | |||
| Mercury (range) | 0.34–2.73 | 2.04–3.89 | 2.74–5.24 | 3.62–7.55 | 5.01–168 | ||
| Age (years) | 43.7 ± 0.52 c | 43.5 ± 0.43 | 44.3 ± 0.38 | 45.8 ± 0.36 | 48.5 ± 0.36 | <0.0001 | 45.2 ± 0.17 |
| Gender | |||||||
| Male | 49.6 d | 49.6 | 49.7 | 49.6 | 49.6 | 0.86 | 49.6 |
| Female | 50.4 | 50.4 | 50.3 | 50.4 | 50.4 | 50.4 | |
| BMI (kg/m2) | 23.0 ± 0.08 | 23.6 ± 0.09 | 23.6 ± 0.08 | 24.0 ± 0.08 | 24.5 ± 0.09 | <0.0001 | 23.7 ± 0.04 |
| Underweight (<18.5) | 6.41 | 5.24 | 4.55 | 3.84 | 1.71 | <0.0001 | 4.35 |
| Normal (18.5–22.9) | 48.1 | 42.7 | 42.1 | 36.1 | 31.4 | 40.1 | |
| Overweight (23–24.9) | 20.8 | 23.9 | 23.4 | 25.0 | 25.7 | 23.8 | |
| Obesity (≥25) | 24.7 | 28.2 | 30.0 | 35.2 | 41.2 | 31.8 | |
| Marital status | |||||||
| Single | 36.1 | 26.2 | 20.6 | 16.0 | 10.3 | <0.0001 | 21.8 |
| Married | 63.9 | 73.8 | 79.4 | 84.0 | 89.7 | 78.2 | |
| Smoking status | |||||||
| Never | 58.9 | 55.7 | 54.4 | 51.8 | 52.1 | 0.02 | 54.6 |
| Former | 17.8 | 19.1 | 19.8 | 21.5 | 22.1 | 20.0 | |
| Current | 23.3 | 25.2 | 25.8 | 26.7 | 25.8 | 25.4 | |
| Alcohol intake e | |||||||
| Non-drinker | 28.2 | 22.9 | 22.5 | 21.2 | 19.5 | <0.0001 | 22.9 |
| Once a month or under | 32.5 | 30.6 | 29.5 | 27.9 | 25.1 | 29.1 | |
| More than twice a month | 31.2 | 35.9 | 35.4 | 36.2 | 36.0 | 34.9 | |
| Heavy | 8.10 | 10.6 | 12.6 | 14.7 | 19.4 | 13.1 | |
| Household income (1000 Korean won) f | |||||||
| Low (~750) | 30.0 | 27.9 | 25.5 | 23.1 | 21.1 | <0.0001 | 25.5 |
| Low-intermediate (750~1500) | 26.6 | 25.7 | 24.7 | 24.3 | 24.1 | 25.1 | |
| Upper-intermediate (1500~2460) | 24.0 | 24.9 | 25.1 | 25.7 | 23.5 | 24.6 | |
| High (2460~) | 19.4 | 21.5 | 24.7 | 26.9 | 31.3 | 24.8 | |
| Physical activity (MET-h/d) | 35.6 ± 62.0 | 41.1 ± 62.3 | 40.7 ± 66.4 | 44.4 ± 70.1 | 45.2 ± 68.9 | 0.03 | 41.1 ± 66.1 |
| Total energy intake (kcal/day) | 1943 ± 23.7 | 2005 ± 24.3 | 2017 ± 23.5 | 2012 ± 25.1 | 2026 ± 24.4 | 0.09 | 2000 ± 10.6 |
| Total fish intake (freq/wk) g | 4.37 ± 0.13 | 5.28 ± 0.13 | 5.91 ± 0.12 | 6.05 ± 0.13 | 6.57 ± 0.15 | <0.0001 | 5.58 ± 0.06 |
| Lowest quintile1 | 30.4 | 22.6 | 17.3 | 15.8 | 14.5 | <0.0001 | 20.0 |
| Quintile2 | 22.5 | 21.0 | 20.6 | 18.6 | 17.9 | 20.1 | |
| Quintile3 | 18.0 | 18.7 | 21.4 | 21.7 | 19.6 | 19.9 | |
| Quintile4 | 16.2 | 20.1 | 19.9 | 21.4 | 22.0 | 20.0 | |
| Highest quintile5 | 12.9 | 17.6 | 20.8 | 22.5 | 26.0 | 20.0 | |
| White fish (freq/wk) | 0.51 ± 0.02 | 0.67 ± 0.03 | 0.79 ± 0.03 | 0.87 ± 0.03 | 1.04 ± 0.04 | <0.0001 | 0.76 ± 0.01 |
| Fatty fish (freq/wk) | 2.16 ± 0.07 | 2.44 ± 0.07 | 2.82 ± 0.08 | 2.75 ± 0.07 | 2.99 ± 0.08 | <0.0001 | 2.61 ± 0.03 |
| Other fish (freq/wk) | 0.96 ± 0.04 | 1.05 ± 0.04 | 1.12 ± 0.04 | 1.05 ± 0.03 | 1.01 ± 0.04 | 0.06 | 1.04 ± 0.02 |
| Shellfish (freq/wk) | 1.32 ± 0.06 | 1.66 ± 0.07 | 1.60 ± 0.06 | 1.75 ± 0.06 | 1.97 ± 0.07 | <0.0001 | 1.58 ± 1.91 |
| Variables | Quintiles of Blood Mercury a | p-Trend b | Continuous of Blood Mercury | ||||
|---|---|---|---|---|---|---|---|
| Q1 | Q2 | Q3 | Q4 | Q5 | |||
| Total (n = 11754) | 2350 | 2351 | 2352 | 2350 | 2351 | ||
| Blood mercury (Mean ± SE) | 1.79 ± 0.01 c | 2.83 ± 0.01 | 3.82 ± 0.02 | 5.22 ± 0.03 | 9.80 ± 0.18 | ||
| Depression cases (n = 342) | 63 (2.68) d | 60 (2.55) | 66 (2.81) | 75 (3.20) | 78 (3.32) | 0.163 | |
| Multivariate Model 1 e | Ref | 1.09 (0.73–1.63) | 1.05 (0.66–1.66) | 1.18 (0.76–1.82) | 1.41 (0.91–2.18) | 0.167 | 1.03 (1.02–1.05) |
| Multivariate Model 2 f | Ref | 1.32 (0.81–2.16) | 1.32 (0.83–2.10) | 1.40 (0.89–2.21) | 1.77 (1.12–2.78) | 0.027 | 1.03 (1.02–1.05) |
| Multivariate Model 3 g | Ref | 1.31 (0.80–2.15) | 1.33 (0.84–2.11) | 1.39 (0.88–2.20) | 1.76 (1.12–2.76) | 0.03 | 1.03 (1.02–1.05) |
| Male (n = 5834) | 1166 | 1167 | 1168 | 1166 | 1167 | ||
| Blood mercury (Mean ± SE) | 2.01 ± 0.02 | 3.29 ± 0.01 | 4.53 ± 0.01 | 6.24 ± 0.02 | 12.1 ± 0.30 | ||
| Depression cases (n = 86) | 18 (1.55) | 14 (1.20) | 21 (1.80) | 19 (1.64) | 14 (1.20) | 0.584 | |
| Multivariate Model 1 | Ref | 0.80 (0.32–2.02) | 1.52 (0.66–3.54) | 1.13 (0.48–2.63) | 0.94 (0.39–2.26) | 0.794 | 1.03 (1.02–1.05) |
| Multivariate Model 2 | Ref | 0.72 (0.26–2.00) | 1.66 (0.71–3.86) | 1.24 (0.52–2.98) | 0.95 (0.37–2.44) | 0.624 | 1.03 (1.02–1.05) |
| Multivariate Model 3 | Ref | 0.72 (0.26–1.98) | 1.65 (0.71–3.86) | 1.23 (0.51–2.97) | 0.95 (0.38–2.40) | 0.629 | 1.03 (1.02–1.05) |
| Female (n = 5920) | 1184 | 1184 | 1184 | 1184 | 1184 | ||
| Blood mercury (Mean ± SE) | 1.56 ± 0.01 | 2.38 ± 0.01 | 3.15 ± 0.01 | 4.23 ± 0.01 | 7.57 ± 0.11 | ||
| Depression cases (n = 256) | 45 (3.80) | 46 (3.89) | 45 (3.81) | 56 (4.73) | 64 (5.41) | 0.211 | |
| Multivariate Model 1 | Ref | 1.19 (0.76–1.86) | 0.93 (0.53–1.61) | 1.20 (0.72–1.99) | 1.54 (0.93–2.56) | 0.168 | 1.03 (0.97–1.10) |
| Multivariate Model 2 | Ref | 1.56 (0.89–2.75) | 1.24 (0.71–2.16) | 1.49 (0.88–2.52) | 2.07 (1.22–3.51) | 0.031 | 1.04 (1.00–1.09) |
| Multivariate Model 3 | Ref | 1.55 (0.88–2.73) | 1.24 (0.71–2.17) | 1.47 (0.87–2.49) | 2.05 (1.20–3.48) | 0.036 | 1.04 (1.00–1.09) |
| Variables | Quintiles of Blood Mercury a | p-Trend b | Continuous of Blood Mercury | ||||
|---|---|---|---|---|---|---|---|
| Q1 | Q2 | Q3 | Q4 | Q5 | |||
| Male (n = 5834) | 1166 | 1167 | 1168 | 1166 | 1167 | ||
| Depression cases (n = 86) | 18 (1.55) c | 14 (1.20) | 21 (1.80) | 19 (1.64) | 14 (1.20) | 0.584 | |
| Total fish tertile 1 (0.00–2.97, n = 1482) d | |||||||
| Age adjusted e | Ref | 0.26 (0.06–1.12) | 0.46 (0.14–1.54) | 0.32 (0.09–1.11) | 0.26 (0.05–1.25) | 0.059 | 0.81 (0.65–1.02) |
| Multivariate adjusted f | Ref | 0.35 (0.08–1.55) | 0.74 (0.21–2.59) | 0.53 (0.14–2.05) | 0.51 (0.11–2.47) | 0.356 | 0.91 (0.76–1.08) |
| Total fish tertile 2 (2.98–6.64, n = 1479) | |||||||
| Age adjusted | Ref | 3.72 (0.36–38.5) | 6.75 (0.67–68.1) | 7.39 (0.84–64.9) | 2.12 (0.21–21.9) | 0.364 | 0.98 (0.92–1.04) |
| Multivariate adjusted | Ref | 3.86 (0.37–40.9) | 7.64 (0.77–75.4) | 8.10 (0.90–72.7) | 2.53 (0.23–27.8) | 0.268 | 1.01 (0.93–1.05) |
| Total fish tertile 3 (6.65–76.2, n = 1480) | |||||||
| Age adjusted | Ref | 1.17 (0.15–9.03) | 3.57 (0.65–19.7) | 0.95 (0.15–6.18) | 2.25 (0.39–13.1) | 0.526 | 1.04 (1.02–1.05) |
| Multivariate adjusted | Ref | 1.28 (0.15–10.8) | 4.10 (0.71–23.6) | 1.19 (0.18–7.81) | 2.91 (0.45–18.9) | 0.325 | 1.04 (1.03–1.06) |
| Female (n = 5920) | 1184 | 1184 | 1184 | 1184 | 1184 | ||
| Depression cases (n = 256) | 45 (3.80) | 46 (3.89) | 45 (3.81) | 56 (4.73) | 64 (5.41) | 0.211 | |
| Total fish tertile 1 (0.00–3.03, n = 1703) | |||||||
| Age adjusted | Ref | 2.94 (1.14–7.60) | 1.78 (0.69–4.55) | 2.74 (1.07–7.01) | 3.39 (1.25–9.15) | 0.042 | 1.06 (0.98–1.15) |
| Multivariate adjusted | Ref | 3.15 (1.21–8.22) | 1.90 (0.74–4.86) | 2.92 (1.13–7.57) | 4.00 (1.51–10.6) | 0.015 | 1.07 (1.00–1.15) |
| Total fish tertile 2 (3.04–6.62, n = 1696) | |||||||
| Age adjusted | Ref | 0.85 (0.33–2.21) | 0.53 (0.19–1.51) | 0.87 (0.36–2.14) | 1.07 (0.45–2.53) | 0.851 | 0.96 (0.87–1.06) |
| Multivariate adjusted | Ref | 0.95 (0.38–2.41) | 0.68 (0.25–1.87) | 0.98 (0.40–2.43) | 1.37 (0.56–3.34) | 0.553 | 0.99 (0.90–1.09) |
| Total fish tertile 3 (6.63–43.2, n = 1702) | |||||||
| Age adjusted | Ref | 1.14 (0.35–3.74) | 1.20 (0.41–3.50) | 1.00 (0.36–2.80) | 1.40 (0.51–3.86) | 0.640 | 1.04 (0.95–1.13) |
| Multivariate adjusted | Ref | 1.07 (0.33–3.48) | 1.25 (0.44–3.56) | 1.08 (0.39–3.02) | 1.55 (0.56–4.31) | 0.422 | 1.05 (0.97–1.14) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, K.W.; Sreeja, S.R.; Kwon, M.; Yu, Y.L.; Kim, M.K. Association of Blood Mercury Level with the Risk of Depression According to Fish Intake Level in the General Korean Population: Findings from the Korean National Health and Nutrition Examination Survey (KNHANES) 2008–2013. Nutrients 2020, 12, 189. https://doi.org/10.3390/nu12010189
Kim KW, Sreeja SR, Kwon M, Yu YL, Kim MK. Association of Blood Mercury Level with the Risk of Depression According to Fish Intake Level in the General Korean Population: Findings from the Korean National Health and Nutrition Examination Survey (KNHANES) 2008–2013. Nutrients. 2020; 12(1):189. https://doi.org/10.3390/nu12010189
Chicago/Turabian StyleKim, Kyung Won, Sundara Raj Sreeja, Minji Kwon, Ye Lee Yu, and Mi Kyung Kim. 2020. "Association of Blood Mercury Level with the Risk of Depression According to Fish Intake Level in the General Korean Population: Findings from the Korean National Health and Nutrition Examination Survey (KNHANES) 2008–2013" Nutrients 12, no. 1: 189. https://doi.org/10.3390/nu12010189
APA StyleKim, K. W., Sreeja, S. R., Kwon, M., Yu, Y. L., & Kim, M. K. (2020). Association of Blood Mercury Level with the Risk of Depression According to Fish Intake Level in the General Korean Population: Findings from the Korean National Health and Nutrition Examination Survey (KNHANES) 2008–2013. Nutrients, 12(1), 189. https://doi.org/10.3390/nu12010189






