Nutritional Status, Dietary Intake, and Adherence to the Mediterranean Diet of Children with Celiac Disease on a Gluten-Free Diet: A Case-Control Prospective Study
Abstract
1. Introduction
2. Material and Methods
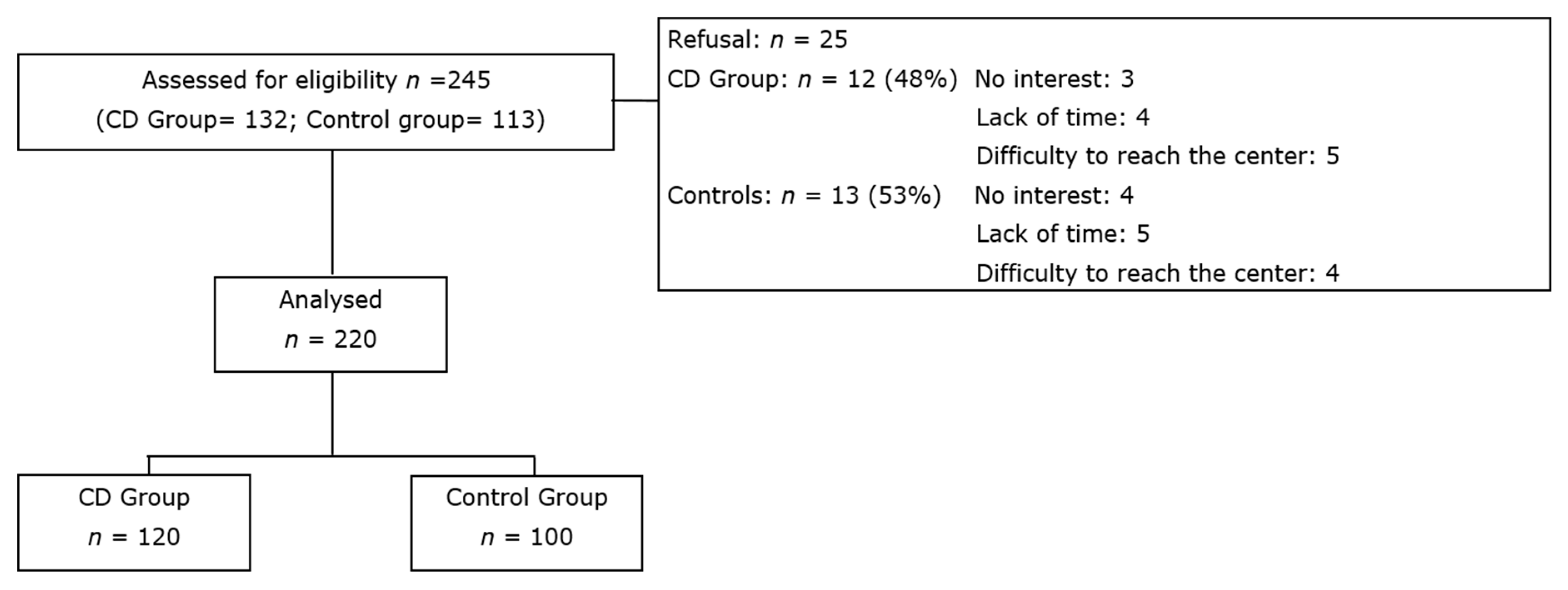
2.1. Study Population
2.2. Anthropometric Measurements
2.3. Physical Activity
2.4. Dietary Assessment
2.5. Statistical Analysis
3. Results
3.1. Study Population
3.2. Anthropometric Results and Energy Expenditure
3.3. Total Energy, Macronutrient Intakes and Adherence to LARN
3.4. Food Group Intake and Adherence to IFP
3.5. Impact of Commercial GF Products Specifically Formulated for CD
3.6. KIDMED Index
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fasano, A.; Catassi, C. Clinical practice. Celiac disease. N. Engl. J. Med. 2012, 367, 2419–2426. [Google Scholar] [CrossRef] [PubMed]
- Lionetti, E.; Catassi, C. New clues in celiac disease epidemiology, pathogenesis, clinical manifestations, and treatment. Int. Rev. Immunol. 2011, 30, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Penagini, F.; Dilillo, D.; Meneghin, F.; Mameli, C.; Fabiano, V.; Zuccotti, G.V. Gluten-free diet in children: An approach to a nutritionally adequate and balanced diet. Nutrients 2013, 5, 4553–4565. [Google Scholar] [CrossRef] [PubMed]
- Snyder, J.; Butzner, J.D.; DeFelice, A.R.; Fasano, A.; Guandalini, S.; Liu, E.; Newton, K.P. Evidence-Informed Expert Recommendations for the Management of Celiac Disease in Children. Pediatrics 2016, 138, e20153147. [Google Scholar] [CrossRef]
- Mariani, P.; Viti, M.G.; Montuori, M.; La Vecchia, A.; Cipolletta, E.; Calvani, L.; Bonamico, M. The gluten-free diet: A nutritional risk factor for adolescents with celiac disease? J. Pediatr. Gastroenterol. Nutr. 1998, 27, 519–523. [Google Scholar] [CrossRef]
- Bardella, M.T.; Fredella, C.; Prampolini, L.; Molteni, N.; Giunta, A.M.; Bianchi, P.A. Body composition and dietary intakes in adult celiac disease patients consuming a strict gluten-free diet. Am. J. Clin. Nutr. 2000, 72, 937–939. [Google Scholar] [CrossRef]
- Hopman, E.G.D.; le Cessie, S.; von Blomberg, B.M.E.; Mearin, M.L. Nutritional management of the gluten-free diet in young people with celiac disease in The Netherlands. J. Pediatr. Gastroenterol. Nutr. 2006, 43, 102–108. [Google Scholar] [CrossRef]
- Ferrara, P.; Cicala, M.; Tiberi, E.; Spadaccio, C.; Marcella, L.; Gatto, A.; Calzolari, P.; Castellucci, G. High fat consumption in children with celiac disease. Acta Gastro Enterol. Belg. 2009, 72, 296–300. [Google Scholar]
- Ohlund, K.; Olsson, C.; Hernell, O.; Ohlund, I. Dietary shortcomings in children on a gluten-free diet. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2010, 23, 294–300. [Google Scholar] [CrossRef]
- Shepherd, S.J.; Gibson, P.R. Nutritional inadequacies of the gluten-free diet in both recently-diagnosed and long-term patients with coeliac disease. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2013, 26, 349–358. [Google Scholar] [CrossRef]
- Zuccotti, G.; Fabiano, V.; Dilillo, D.; Picca, M.; Cravidi, C.; Brambilla, P. Intakes of nutrients in Italian children with celiac disease and the role of commercially available gluten-free products. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2013, 26, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Kautto, E.; Ivarsson, A.; Norström, F.; Högberg, L.; Carlsson, A.; Hörnell, A. Nutrient intake in adolescent girls and boys diagnosed with coeliac disease at an early age is mostly comparable to their non-coeliac contemporaries. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2014, 27, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Babio, N.; Alcázar, M.; Castillejo, G.; Recasens, M.; Martínez-Cerezo, F.; Gutiérrez-Pensado, V.; Masip, G.; Vaqué, C.; Vila-Martí, A.; Torres-Moreno, M.; et al. Patients with Celiac Disease Reported Higher Consumption of Added Sugar and Total Fat Than Healthy Individuals. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Larretxi, I.; Simon, E.; Benjumea, L.; Miranda, J.; Bustamante, M.A.; Lasa, A.; Eizaguirre, F.J.; Churruca, I. Gluten-free-rendered products contribute to imbalanced diets in children and adolescents with celiac disease. Eur. J. Nutr. 2019, 58, 775–783. [Google Scholar] [CrossRef] [PubMed]
- Cornicelli, M.; Saba, M.; Machello, N.; Silano, M.; Neuhold, S. Nutritional composition of gluten-free food versus regular food sold in the Italian market. Dig. Liver Dis. Off. J. Ital. Soc. Gastroenterol. Ital. Assoc. Study Liver 2018, 50, 1305–1308. [Google Scholar] [CrossRef]
- Fry, L.; Madden, A.M.; Fallaize, R. An investigation into the nutritional composition and cost of gluten-free versus regular food products in the UK. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2018, 31, 108–120. [Google Scholar] [CrossRef]
- Husby, S.; Koletzko, S.; Korponay-Szabó, I.R.; Mearin, M.L.; Phillips, A.; Shamir, R.; Troncone, R.; Giersiepen, K.; Branski, D.; Catassi, C.; et al. European Society for Pediatric Gastroenterology, Hepatology, and Nutrition guidelines for the diagnosis of coeliac disease. J. Pediatr. Gastroenterol. Nutr. 2012, 54, 136–160. [Google Scholar] [CrossRef]
- Gatti, S.; Lionetti, E.; Balanzoni, L.; Verma, A.K.; Galeazzi, T.; Gesuita, R.; Scattolo, N.; Cinquetti, M.; Fasano, A.; Catassi, C. Increased prevalence of celiac disease in school-age children in Italy. Clin. Gastroenterol. Hepatol. 2019. [Google Scholar] [CrossRef]
- Banca Dati di Composizione Degli Alimenti per Studi Epidemiologici in Italia. Available online: http://www.bda-ieo.it/ (accessed on 19 December 2019).
- Nutrients and Energy Reference Intake for Italian Population; 4th revision; SINU (Italian Society of Human Nutrition): Milan, Italy, 2014.
- Italian Society of Pediatrics. Italian Food Pyramid; SIP: Rome, Italy, 2017. [Google Scholar]
- Serra-Majem, L.; Ribas, L.; Ngo, J.; Ortega, R.M.; García, A.; Pérez-Rodrigo, C.; Aranceta, J. Food, youth and the Mediterranean diet in Spain. Development of KIDMED, Mediterranean Diet Quality Index in children and adolescents. Public Health Nutr. 2004, 7, 931–935. [Google Scholar] [CrossRef]
- Brambilla, P.; Picca, M.; Dilillo, D.; Meneghin, F.; Cravidi, C.; Tischer, M.C.; Vivaldo, T.; Bedogni, G.; Zuccotti, G.V. Changes of body mass index in celiac children on a gluten-free diet. Nutr. Metab. Cardiovasc. Dis. NMCD 2013, 23, 177–182. [Google Scholar] [CrossRef]
- Barera, G.; Mora, S.; Brambilla, P.; Ricotti, A.; Menni, L.; Beccio, S.; Bianchi, C. Body composition in children with celiac disease and the effects of a gluten-free diet: A prospective case-control study. Am. J. Clin. Nutr. 2000, 72, 71–75. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Brar, P.S.; Lee, A.R.; Green, P.H.R. Body mass index in celiac disease: Beneficial effect of a gluten-free diet. J. Clin. Gastroenterol. 2010, 44, 267–271. [Google Scholar] [CrossRef] [PubMed]
- Radlović, N.; Mladenović, M.; Leković, Z.; Zivanović, D.; Brdar, R.; Radlović, V.; Ristić, D.; Pavlović, M.; Stojsić, Z.; Vuletić, B.; et al. Effect of gluten-free diet on the growth and nutritional status of children with coeliac disease. Srp. Arh. Celok. Lek. 2009, 137, 632–637. [Google Scholar] [CrossRef] [PubMed]
- Kabbani, T.A.; Goldberg, A.; Kelly, C.P.; Pallav, K.; Tariq, S.; Peer, A.; Hansen, J.; Dennis, M.; Leffler, D.A. Body mass index and the risk of obesity in coeliac disease treated with the gluten-free diet. Aliment. Pharmacol. Ther. 2012, 35, 723–729. [Google Scholar] [CrossRef] [PubMed]
- Valletta, E.; Fornaro, M.; Cipolli, M.; Conte, S.; Bissolo, F.; Danchielli, C. Celiac disease and obesity: Need for nutritional follow-up after diagnosis. Eur. J. Clin. Nutr. 2010, 64, 1371–1372. [Google Scholar] [CrossRef] [PubMed]
- Thompson, F.E.; Subar, A.F. Dietary assessment methodology. In Nutrition in the Prevention and Treatment of Disease; Coulston, A.M., Boushey, C.J., Ferruzzi, M.G., Eds.; Elsevier: Amsterdam, The Netherlands; Academic Press: Amsterdam, The Netherlands, 2013; pp. 5–46. [Google Scholar]
| Variable | CD Group (n = 120) | Control Group (n = 100) | p |
|---|---|---|---|
| Age (years) | 0.494 | ||
| Median | 10.5 | 10.1 | |
| First to third quartiles | 8.3–12.2 | 9.4–13.5 | |
| Weight (Kg) | 0.448 | ||
| Median | 31.5 | 32.5 | |
| First to third quartiles | 25.2–42.0 | 28.2–50.0 | |
| Height (cm) | 0.26 | ||
| Median | 1.4 | 1.4 | |
| First to third quartiles | 1.2–1.5 | 1.4–1.5 | |
| Body mass index (Kg/m2) | 0.988 | ||
| Median | 16.8 | 16.0 | |
| First to third quartiles | 15.3–18.9 | 15.2–19.5 | |
| Body mass index class (Kg/m2) | 0.35 | ||
| Underweight-n (%) | 6 (5) | 5 (5) | |
| Adequate weight-n (%) | 78 (65) | 70 (70) | |
| Overweight-n (%) | 24 (20) | 18 (18) | |
| Obese-n (%) | 12 (10) | 7 (7) | |
| Physical activity (hours/week) | 0.729 | ||
| Median | 4 | 4 | |
| First to third quartiles | 3–8 | 4–5 | |
| Sedentary activity (hours/week) | 0.247 | ||
| Median | 2 | 2 | |
| First to third quartiles | 1–2 | 2–3 |
| Group | CD Group (n = 120) | Control group (n = 100) | p | LARN | |
|---|---|---|---|---|---|
| Median (1st; 3rd Quartiles) | Median (1st; 3rd Quartiles) | ||||
| 95% Confidence Interval | 95% Confidence Interval | ||||
| Energy | Kcal | 1819.3 (1589.6; 1997.0) | 1838.2 (1782.2; 1964.6) | 0.225 | |
| 1715.5–1923.0 | 1775.3–1901.1 | ||||
| Total Proteins | grams | 59.4 (53.4; 67.7) | 65.9 (60.7; 72.0) | 0.095 | |
| 55.7–63.0 | 62.0–69.8 | 31–62 (PRI) | |||
| % Energy | 13.4 (11.8; 14.4) | 14.1 (12.4; 14.7) | 0.335 | ||
| 12.7–14.0 | 13.4–14.9 | 10–15% (RI) | |||
| grams/Kg | 1.9 (1.5; 2.6) | 1.9 (1.7; 2.2) | 0.670 | ||
| 1.6–2.2 | 1.7–2.1 | 0.90–0.99 (PRI) | |||
| Total Carbohydrates | grams | 209.7 (184.1; 252.1) | 260.5 (245.4; 298.4) | 0.001 * | |
| 192.5–226.8 | 242.2–278.8 | ||||
| % Energy | 46.9 (42.6; 51.7) | 53 (50.5; 56.8) | 0.001 * | ||
| 44.5–49.2 | 50.9–55.2 | 45–60% (RI) | |||
| Total sugars | grams | 68.1 (49.1; 83.3) | 83.1 (69.7; 95.3) | 0.036 * | |
| 59.4–76.9 | 74.2–91.9 | ||||
| % Energy | 14.5 (10.4; 17.6) | 17.6 (14.2; 19.6) | 0.036 * | ||
| 12.6–16.3 | 15.8–19.4 | <15% (SDT) | |||
| Total fats | grams | 78.1 (63.9; 92.2) | 64.4 (59.5; 74.4) | 0.015 * | |
| 70.8–85.4 | 59.2–69.5 | ||||
| % Energy | 37.5 (32.8; 40.5) | 30.5 (28.7; 32.3) | 0.001 * | ||
| 35.6–39.5 | 29.3–31.8 | 20–35% (RI) | |||
| Saturated fats | grams | 25.3 (20.2; 30.8) | 18.7 (16.5; 21.7) | 0.003 * | |
| 22.5–28.1 | 16.9–20.4 | ||||
| % Energy | 12.8 (10; 14.7) | 8.8 (7.8; 10.4) | 0.001 * | ||
| 11.6–14.0 | 7.9–9.7 | <10% (SDT) | |||
| Total Fiber | grams | 12.6 (10.9; 16.7) | 15 (13.5; 19.1) | 0.015 * | |
| 11.1–14.2 | 13.1–16.9 | ||||
| % Energy | 1.4 (1.1; 1.7) | 1.7 (1.4; 1.9) | 0.067 | ||
| 1.2–1.5 | 1.5–1.9 | At least 1.7% (AI) |
| Food Groups | CD Group (n = 120) | Control Group (n = 100) | p | IFP |
|---|---|---|---|---|
| Median (1; 3 Quartiles) | Median (1; 3 Quartiles) | |||
| IC 95% | IC 95% | |||
| Sugary drinks | 1 (0; 2) | 2 (1; 2) | 0.401 | |
| 0.5–1.5 | 1.5–2.5 | Lowest consumption | ||
| Meat | 2 (1; 3) | 2 (1; 3) | 0.831 | |
| 1.5–2.5 | 1.5–2.5 | 1 | ||
| Processed Meat | 2.5 (2; 4) | 2 (1; 2) | 0.009 * | |
| 2.0–3.0 | 1.5–2.5 | 1 | ||
| Cheese | 1 (0; 3) | 1 (0; 2) | 0.431 | |
| 0.5–2.0 | 0.5–1.5 | 1 | ||
| Fruits | 2.5 (1; 4) | 3 (2; 5) | 0.121 | |
| 1.5–3.0 | 2.0–4.0 | 3–6 | ||
| Milk and yogurt | 3 (1; 3) | 3 (3; 4) | 0.116 | |
| 2.5–3.5 | 2.5–3.5 | 3–6 | ||
| Legumes | 0 (0; 0) | 0 (0; 1) | 0.175 | |
| 0–0 | 0–0.5 | 2 | ||
| Potatoes | 1 (0; 1) | 1 (0; 1) | 0.475 | |
| 0.5–1.0 | 0.5–1.5 | 1 | ||
| Fish | 1 (0; 1) | 1 (0; 2) | 0.121 | |
| 0.5–1.0 | 0.5–1.5 | 1–2 | ||
| Eggs | 0 (0; 1) | 0 (0; 1) | 0.569 | |
| 0.0–0.5 | 0.0–0.5 | 1 | ||
| Vegetables | 2.5 (1; 4) | 3 (2;4) | 0.122 | |
| 1.5–3.0 | 2.5–3.5 | 6–9 | ||
| Total cereals | 15.5 (13; 17) | 15 (14; 18.5) | 0.463 | |
| 14.5–16.5 | 13.5–16.5 | 9–15 | ||
| Maize | 0 (0; 0) | 0 (0; 0) | 0.906 | |
| 0–0 | 0–0 | Not indicated | ||
| Rice | 1 (0; 1) | 1 (0; 1) | 1 | |
| 0.5–1.0 | 0.5–1.5 | Not indicated | ||
| Minor and pseudo-cereals | 0 (0; 0) | 0 (0; 0) | 0.167 | |
| 0–0 | 0–0 | Not indicated | ||
| Salty snaks | 1 (0; 2) | 0 (0; 0.5) | 0.001 * | Lowest consumption |
| 0.5–1.5 | 0–0.5 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lionetti, E.; Antonucci, N.; Marinelli, M.; Bartolomei, B.; Franceschini, E.; Gatti, S.; Catassi, G.N.; Verma, A.K.; Monachesi, C.; Catassi, C. Nutritional Status, Dietary Intake, and Adherence to the Mediterranean Diet of Children with Celiac Disease on a Gluten-Free Diet: A Case-Control Prospective Study. Nutrients 2020, 12, 143. https://doi.org/10.3390/nu12010143
Lionetti E, Antonucci N, Marinelli M, Bartolomei B, Franceschini E, Gatti S, Catassi GN, Verma AK, Monachesi C, Catassi C. Nutritional Status, Dietary Intake, and Adherence to the Mediterranean Diet of Children with Celiac Disease on a Gluten-Free Diet: A Case-Control Prospective Study. Nutrients. 2020; 12(1):143. https://doi.org/10.3390/nu12010143
Chicago/Turabian StyleLionetti, Elena, Niki Antonucci, Michele Marinelli, Beatrice Bartolomei, Elisa Franceschini, Simona Gatti, Giulia Naspi Catassi, Anil K. Verma, Chiara Monachesi, and Carlo Catassi. 2020. "Nutritional Status, Dietary Intake, and Adherence to the Mediterranean Diet of Children with Celiac Disease on a Gluten-Free Diet: A Case-Control Prospective Study" Nutrients 12, no. 1: 143. https://doi.org/10.3390/nu12010143
APA StyleLionetti, E., Antonucci, N., Marinelli, M., Bartolomei, B., Franceschini, E., Gatti, S., Catassi, G. N., Verma, A. K., Monachesi, C., & Catassi, C. (2020). Nutritional Status, Dietary Intake, and Adherence to the Mediterranean Diet of Children with Celiac Disease on a Gluten-Free Diet: A Case-Control Prospective Study. Nutrients, 12(1), 143. https://doi.org/10.3390/nu12010143








