Resveratrol and the Interaction between Gut Microbiota and Arterial Remodelling
Abstract
1. Introduction
2. Resveratrol and Arterial Remodelling
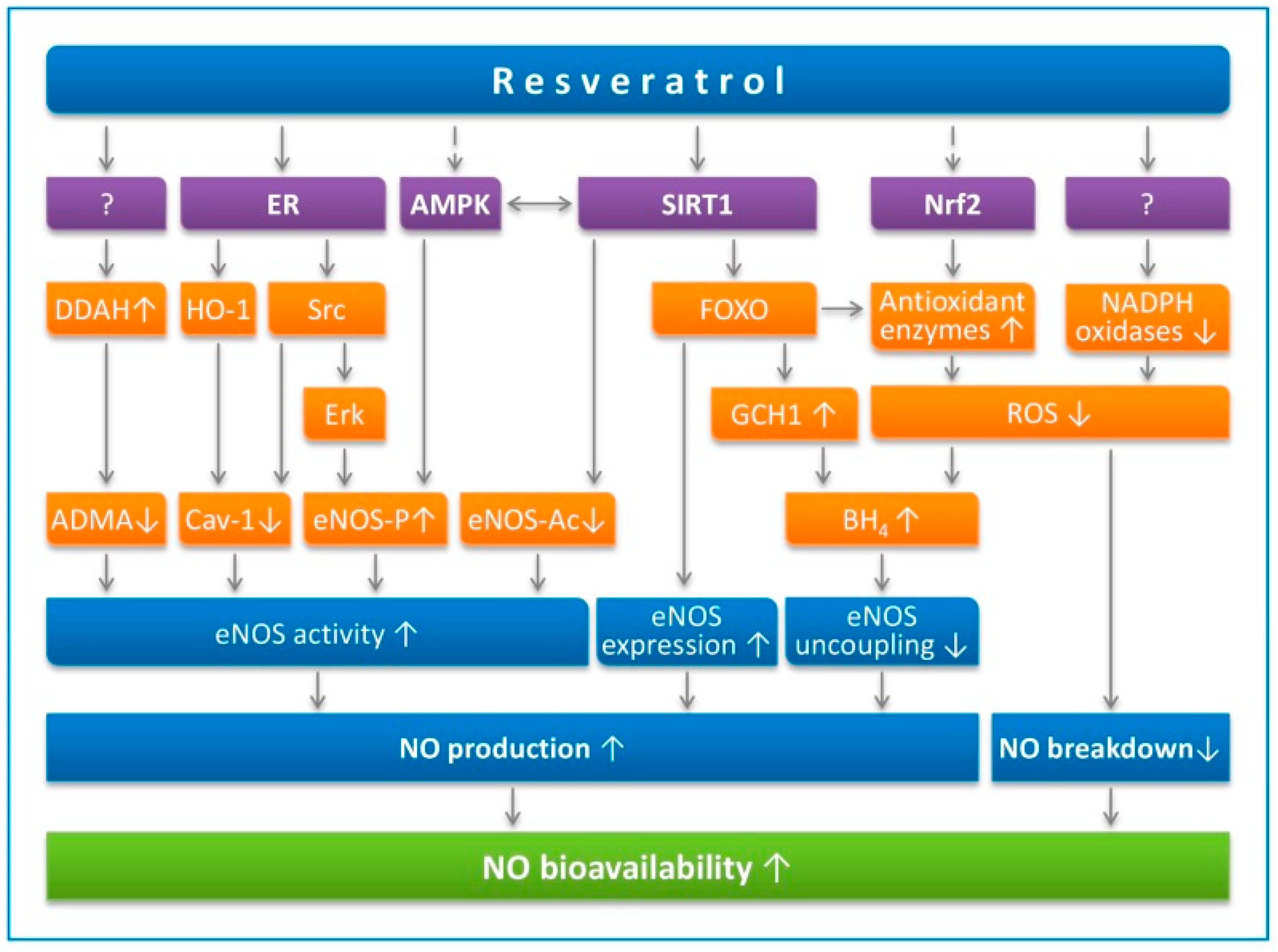
2.1. Improvement of Endothelial Function
2.2. Inhibition of Neointima Formation
2.3. Prevention of Arterial Stiffening
3. Gut Microbiota and Microbiota-Derived Metabolites Modulates Arterial Remodelling
3.1. Gut Microbiota and Arterial Remodelling
3.2. Gut Microbiota Derived Metabolites and Arterial Remodelling
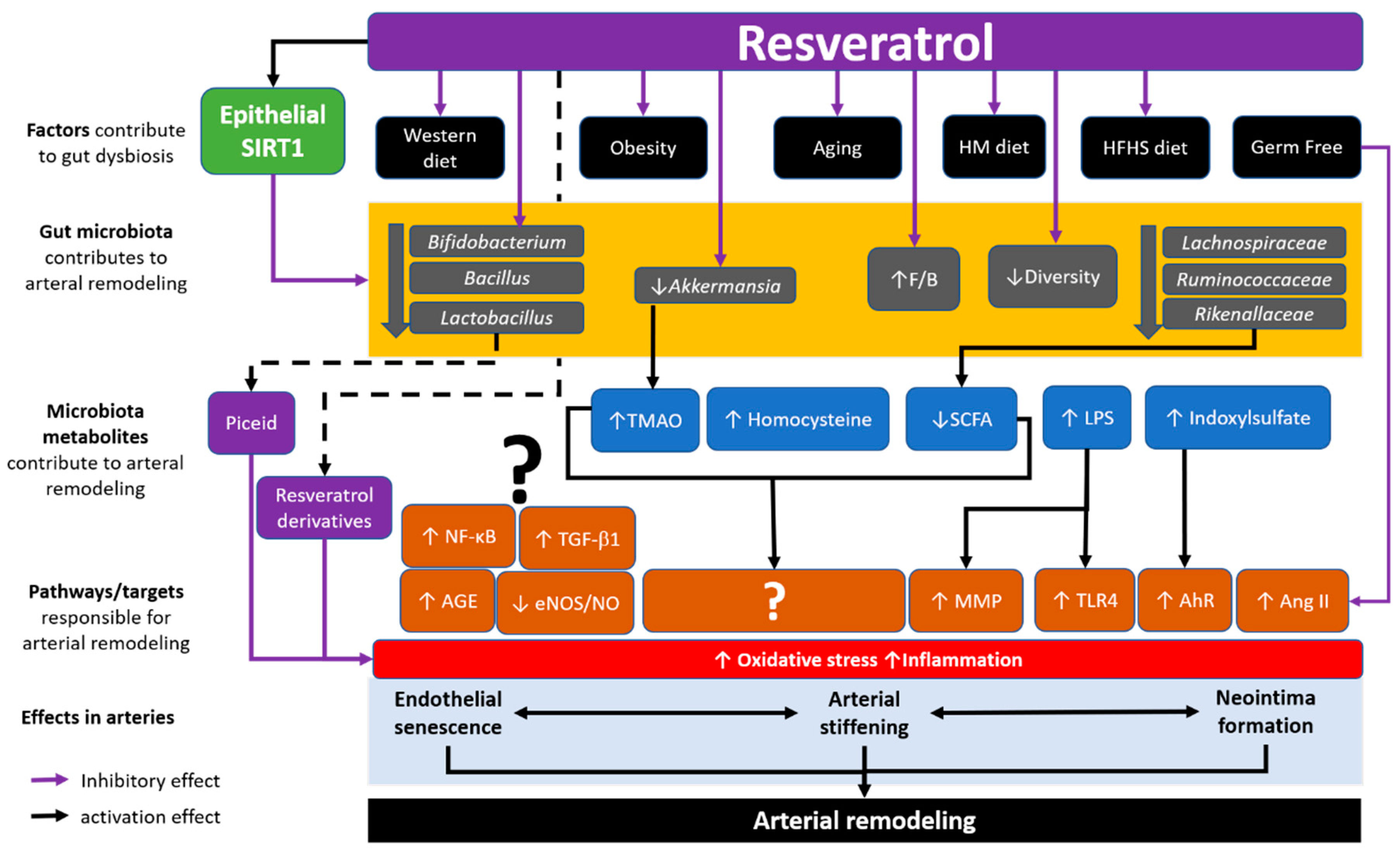
4. The Interaction between Resveratrol, Gut Microbiota and Arterial Remodelling
Summary and Future Directions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organization. Cardiovascular Disease: Global Atlas on Cardiovascular Disease Prevention and Control; WHO: Geneva, Switzerland, 2012. [Google Scholar]
- Laslett, L.J.; Alagona, P.; Clark, B.A.; Drozda, J.P.; Saldivar, F.; Wilson, S.R.; Poe, C.; Hart, M. The worldwide environment of cardiovascular disease: Prevalence, diagnosis, therapy, and policy issues: A report from the American College of Cardiology. J. Am. Coll. Cardiol. 2012, 60, S1–S49. [Google Scholar] [CrossRef] [PubMed]
- Oyama, J.-I.; Node, K. Gut microbiota and hypertension. Hypertens. Res. 2019, 42, 741. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J.; Brands, M.W.; Daniels, S.R.; Karanja, N.; Elmer, P.J.; Sacks, F.M. Dietary approaches to prevent and treat hypertension: A scientific statement from the American Heart Association. Hypertension 2006, 47, 296–308. [Google Scholar] [CrossRef] [PubMed]
- Savoia, C. Vascular Remodeling. In Textbook of Vascular Medicine; Springer: Berlin, Germany, 2019; pp. 193–201. [Google Scholar]
- Man, A.W.; Wang, Y. Age-Associated Arterial Remodelling. EC Cardiol. 2017, 4, 137–164. [Google Scholar]
- Baur, J.A.; Sinclair, D.A. Therapeutic potential of resveratrol: the in vivo evidence. Nat. Rev. Drug Discov. 2006, 5, 493. [Google Scholar] [CrossRef]
- Jang, M.; Cai, L.; Udeani, G.O.; Slowing, K.V.; Thomas, C.F.; Beecher, C.W.; Fong, H.H.; Farnsworth, N.R.; Kinghorn, A.D.; Mehta, R.G. Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science 1997, 275, 218–220. [Google Scholar] [CrossRef]
- Bo, S.; Ciccone, G.; Castiglione, A.; Gambino, R.; De Michieli, F.; Villois, P.; Durazzo, M.; Cavallo-Perin, P.; Cassader, M. Anti-inflammatory and antioxidant effects of resveratrol in healthy smokers a randomized, double-blind, placebo-controlled, cross-over trial. Curr. Med. Chem. 2013, 20, 1323–1331. [Google Scholar] [CrossRef]
- Peluso, I.; Villaño Valencia, D.; Chen, C.-Y.O.; Palmery, M. Antioxidant, Anti-Inflammatory, and Microbial-Modulating Activities of Nutraceuticals and Functional Foods 2018. Oxid. Med. Cell. Longev. 2018, 2018, 2. [Google Scholar] [CrossRef]
- Aguilar-Alonso, P.; Vera-López, O.; Brambila-Colombres, E.; Segura-Badilla, O.; Avalos-López, R.; Lazcano-Hernández, M.; Navarro-Cruz, A. Evaluation of Oxidative Stress in Cardiomyocytes during the Aging Process in Rats Treated with Resveratrol. Oxid. Med. Cell. Longev. 2018, 2018, 9. [Google Scholar] [CrossRef]
- Li, H.; Xia, N.; Förstermann, U. Cardiovascular effects and molecular targets of resveratrol. Nitric Oxide 2012, 26, 102–110. [Google Scholar] [CrossRef]
- Springer, M.; Moco, S. Resveratrol and Its Human Metabolites—Effects on Metabolic Health and Obesity. Nutrients 2019, 11, 143. [Google Scholar] [CrossRef]
- Li, H.; Xia, N.; Hasselwander, S.; Daiber, A. Resveratrol and vascular function. Int. J. Mol. Sci. 2019, 20, 2155. [Google Scholar] [CrossRef]
- Ma, C.; Wang, Y.; Dong, L.; Li, M.; Cai, W. Anti-inflammatory effect of resveratrol through the suppression of NF-κB and JAK/STAT signaling pathways. Acta Biochim. Biophys. Sin. 2015, 47, 207–213. [Google Scholar] [CrossRef]
- Sun, Y.; Li, J.; Xiao, N.; Wang, M.; Kou, J.; Qi, L.; Huang, F.; Liu, B.; Liu, K. Pharmacological activation of AMPK ameliorates perivascular adipose/endothelial dysfunction in a manner interdependent on AMPK and SIRT1. Pharmacol. Res. 2014, 89, 19–28. [Google Scholar] [CrossRef]
- Gutsol, A.A.; Blanco, P.; Samokhina, S.I.; Afanasiev, S.A.; Kennedy, C.R.; Popov, S.V.; Burns, K.D. A novel method for comparison of arterial remodeling in hypertension: Quantification of arterial trees and recognition of remodeling patterns on histological sections. PLoS ONE 2019, 14, e0216734. [Google Scholar] [CrossRef]
- Chaplin, A.; Carpéné, C.; Mercader, J. Resveratrol, metabolic syndrome, and gut microbiota. Nutrients 2018, 10, 1651. [Google Scholar] [CrossRef]
- Jin, M.; Qian, Z.; Yin, J.; Xu, W.; Zhou, X. The role of intestinal microbiota in cardiovascular disease. J. Cell. Mol. Med. 2019, 23, 2343–2350. [Google Scholar] [CrossRef]
- Laurent, S.; Bruno, R.M. Gut microbiome composition, a third player in the inflammation–arterial stiffness relationship. Eur. Heart J. 2018, 39, 2398–2400. [Google Scholar] [CrossRef]
- Menni, C.; Lin, C.; Cecelja, M.; Mangino, M.; Matey-Hernandez, M.L.; Keehn, L.; Mohney, R.P.; Steves, C.J.; Spector, T.D.; Kuo, C.-F. Gut microbial diversity is associated with lower arterial stiffness in women. Eur. Heart J. 2018, 39, 2390–2397. [Google Scholar] [CrossRef]
- Langille, B.L.; O’Donnell, F. Reductions in arterial diameter produced by chronic decreases in blood flow are endothelium-dependent. Science 1986, 231, 405–407. [Google Scholar] [CrossRef]
- Gibbons, G.H.; Dzau, V.J. The emerging concept of vascular remodeling. N. Engl. J. Med. 1994, 330, 1431–1438. [Google Scholar]
- Sandoo, A.; Veldhuijzen van Zanten, J.J.; Metsios, G.S.; Carroll, D.; Kitas, G.D. The endothelium and its role in regulating vascular tone. Open Cardiovasc. Med. J. 2010, 4, 302. [Google Scholar] [CrossRef]
- Avolio, A.; Butlin, M.; Liu, Y.-Y.; Viegas, K.; Avadhanam, B.; Lindesay, G. Regulation of arterial stiffness: Cellular, molecular and neurogenic mechanisms. Artery Res. 2011, 5, 122–127. [Google Scholar] [CrossRef]
- Touyz, R.M.; Tabet, F.; Schiffrin, E.L. Redox-dependent signalling by angiotensin II and vascular remodelling in hypertension. Clin. Exp. Pharmacol. Physiol. 2003, 30, 860–866. [Google Scholar] [CrossRef]
- Wilkinson, I.B.; McEniery, C.M. Arterial stiffness, endothelial function and novel pharmacological approaches. Clin. Exp. Pharmacol. Physiol. 2004, 31, 795–799. [Google Scholar] [CrossRef]
- Joannides, R.; Richard, V.; Haefeli, W.E.; Benoist, A.; Linder, L.; Lüscher, T.F.; Thuillez, C. Role of nitric oxide in the regulation of the mechanical properties of peripheral conduit arteries in humans. Hypertension 1997, 30, 1465–1470. [Google Scholar] [CrossRef]
- Wilkinson, I.B.; MacCallum, H.; Cockcroft, J.R.; Webb, D.J. Inhibition of basal nitric oxide synthesis increases aortic augmentation index and pulse wave velocity in vivo. Br. J. Clin. Pharmacol. 2002, 53, 189–192. [Google Scholar] [CrossRef]
- Latson, T.W.; Hunter, W.C.; Katoh, N.; Sagawa, K. Effect of nitroglycerin on aortic impedance, diameter, and pulse-wave velocity. Circ. Res. 1988, 62, 884–890. [Google Scholar] [CrossRef]
- Kobs, R.W.; Chesler, N.C. The mechanobiology of pulmonary vascular remodeling in the congenital absence of eNOS. Biomech. Model. Mechanobiol. 2006, 5, 217–225. [Google Scholar] [CrossRef]
- Ozaki, M.; Kawashima, S.; Yamashita, T.; Ohashi, Y.; Rikitake, Y.; Inoue, N.; Hirata, K.-I.; Hayashi, Y.; Itoh, H.; Yokoyama, M. Reduced hypoxic pulmonary vascular remodeling by nitric oxide from the endothelium. Hypertension 2001, 37, 322–327. [Google Scholar] [CrossRef]
- Jeremy, J.Y.; Rowe, D.; Emsley, A.M.; Newby, A.C. Nitric oxide and the proliferation of vascular smooth muscle cells. Cardiovasc. Res. 1999, 43, 580–594. [Google Scholar] [CrossRef]
- Moroi, M.; Zhang, L.; Yasuda, T.; Virmani, R.; Gold, H.K.; Fishman, M.C.; Huang, P.L. Interaction of genetic deficiency of endothelial nitric oxide, gender, and pregnancy in vascular response to injury in mice. J. Clin. Investig. 1998, 101, 1225. [Google Scholar] [CrossRef]
- Xia, N.; Daiber, A.; Förstermann, U.; Li, H. Antioxidant effects of resveratrol in the cardiovascular system. Br. J. Pharmacol. 2017, 174, 1633–1646. [Google Scholar] [CrossRef]
- Xia, N.; Förstermann, U.; Li, H. Resveratrol and endothelial nitric oxide. Molecules 2014, 19, 16102–16121. [Google Scholar] [CrossRef] [PubMed]
- Buhrmann, C.; Shayan, P.; Popper, B.; Goel, A.; Shakibaei, M. Sirt1 is required for resveratrol-mediated chemopreventive effects in colorectal cancer cells. Nutrients 2016, 8, 145. [Google Scholar] [CrossRef]
- Dang, W. The controversial world of sirtuins. Drug Discov. Today Technol. 2014, 12, e9–e17. [Google Scholar] [CrossRef]
- Pezzuto, J.M. Resveratrol: Twenty years of growth, development and controversy. Biomol. Ther. 2019, 27, 1. [Google Scholar] [CrossRef]
- Baur, J.A. Resveratrol, sirtuins, and the promise of a DR mimetic. Mech. Ageing Dev. 2010, 131, 261–269. [Google Scholar] [CrossRef]
- Csiszar, A.; Labinskyy, N.; Pinto, J.T.; Ballabh, P.; Zhang, H.; Losonczy, G.; Pearson, K.; De Cabo, R.; Pacher, P.; Zhang, C. Resveratrol induces mitochondrial biogenesis in endothelial cells. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, H13–H20. [Google Scholar] [CrossRef]
- Xia, N.; Strand, S.; Schlufter, F.; Siuda, D.; Reifenberg, G.; Kleinert, H.; Förstermann, U.; Li, H. Role of SIRT1 and FOXO factors in eNOS transcriptional activation by resveratrol. Nitric Oxide 2013, 32, 29–35. [Google Scholar] [CrossRef]
- Cui, X.; Liu, X.; Feng, H.; Zhao, S.; Gao, H. Grape seed proanthocyanidin extracts enhance endothelial nitric oxide synthase expression through 5′-AMP activated protein kinase/Surtuin 1–Krüpple like factor 2 pathway and modulate blood pressure in ouabain induced hypertensive rats. Biol. Pharm. Bull. 2012, 35, 2192–2197. [Google Scholar] [CrossRef]
- Man, A.W.; Bai, B.; Yang, K.; Guo, Y.; Xu, C.; Tse, H.-F.; Han, W.; Bloksgaard, M.; De Mey, J.G.; Vanhoutte, P.M. Endothelial SIRT1 prevents adverse arterial remodeling by facilitating HERC2-mediated degradation of acetylated LKB1. Oncotarget 2016, 7, 39065. [Google Scholar]
- Kedenko, L.; Lamina, C.; Kedenko, I.; Kollerits, B.; Kiesslich, T.; Iglseder, B.; Kronenberg, F.; Paulweber, B. Genetic polymorphisms at SIRT1 and FOXO1 are associated with carotid atherosclerosis in the SAPHIR cohort. BMC Med. Genet. 2014, 15, 112. [Google Scholar] [CrossRef]
- Faulx, M.D.; Wright, A.T.; Hoit, B.D. Detection of endothelial dysfunction with brachial artery ultrasound scanning. Am. Heart J. 2003, 145, 943–951. [Google Scholar] [CrossRef]
- Imamura, H.; Yamaguchi, T.; Nagayama, D.; Saiki, A.; Shirai, K.; Tatsuno, I. Resveratrol ameliorates arterial stiffness assessed by cardio-ankle vascular index in patients with type 2 diabetes mellitus. Int. Heart J. 2017, 58, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.; Howe, P.; Buckley, J.; Coates, A.; Kunz, I.; Berry, N. Acute resveratrol supplementation improves flow-mediated dilatation in overweight/obese individuals with mildly elevated blood pressure. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 851–856. [Google Scholar] [CrossRef]
- Wong, R.H.; Berry, N.M.; Coates, A.M.; Buckley, J.D.; Bryan, J.; Kunz, I.; Howe, P.R. Chronic resveratrol consumption improves brachial flow-mediated dilatation in healthy obese adults. J. Hypertens. 2013, 31, 1819–1827. [Google Scholar] [CrossRef]
- Fujitaka, K.; Otani, H.; Jo, F.; Jo, H.; Nomura, E.; Iwasaki, M.; Nishikawa, M.; Iwasaka, T.; Das, D.K. Modified resveratrol Longevinex improves endothelial function in adults with metabolic syndrome receiving standard treatment. Nutr. Res. 2011, 31, 842–847. [Google Scholar] [CrossRef]
- Thompson, A.M.; Martin, K.A.; Rzucidlo, E.M. Resveratrol induces vascular smooth muscle cell differentiation through stimulation of SirT1 and AMPK. PLoS ONE 2014, 9, e85495. [Google Scholar] [CrossRef]
- Wang, D.; Uhrin, P.; Mocan, A.; Waltenberger, B.; Breuss, J.M.; Tewari, D.; Mihaly-Bison, J.; Huminiecki, Ł.; Starzyński, R.R.; Tzvetkov, N.T. Vascular smooth muscle cell proliferation as a therapeutic target. Part 1: Molecular targets and pathways. Biotechnol. Adv. 2018, 36, 1586–1607. [Google Scholar] [CrossRef]
- Kamiya, A.; Togawa, T. Adaptive regulation of wall shear stress to flow change in the canine carotid artery. Am. J. Physiol. Heart Circ. Physiol. 1980, 239, H14–H21. [Google Scholar] [CrossRef]
- Ong, E.-T.; Hwang, T.-L.; Huang, Y.-L.; Lin, C.-F.; Wu, W.-B. Vitisin B, a resveratrol tetramer, inhibits migration through inhibition of PDGF signaling and enhancement of cell adhesiveness in cultured vascular smooth muscle cells. Toxicol. Appl. Pharmacol. 2011, 256, 198–208. [Google Scholar] [CrossRef] [PubMed]
- Ekshyyan, V.P.; Hebert, V.Y.; Khandelwal, A.; Dugas, T.R. Resveratrol inhibits rat aortic vascular smooth muscle cell proliferation via estrogen receptor dependent nitric oxide production. J. Cardiovasc. Pharmacol. 2007, 50, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Heckenkamp, J.; Lamuraglia, G.M. Intimal hyperplasia, arterial remodeling, and restenosis: An overview. Perspect. Vasc. Surg. Endovasc. Ther. 1999, 11, 71–94. [Google Scholar] [CrossRef]
- Guo, R.; Li, W.; Liu, B.; Li, S.; Zhang, B.; Xu, Y. Resveratrol protects vascular smooth muscle cells against high glucose-induced oxidative stress and cell proliferation in vitro. Med. Sci. Monit. Basic Res. 2014, 20, 82. [Google Scholar] [PubMed]
- Xia, N.; Forstermann, U.; Li, H. Resveratrol as a gene regulator in the vasculature. Curr. Pharm. Biotechnol. 2014, 15, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Hasegawa, Y.; Ishigaki, Y.; Yamada, T.; Gao, J.; Imai, J.; Uno, K.; Kaneko, K.; Ogihara, T.; Shimosawa, T. Importance of endothelial NF-κB signalling in vascular remodelling and aortic aneurysm formation. Cardiovasc. Res. 2012, 97, 106–114. [Google Scholar] [CrossRef]
- Csiszar, A.; Sosnowska, D.; Wang, M.; Lakatta, E.G.; Sonntag, W.E.; Ungvari, Z. Age-associated proinflammatory secretory phenotype in vascular smooth muscle cells from the non-human primate Macaca mulatta: Reversal by resveratrol treatment. J. Gerontol. Ser. A Biomed. Sci. Med. Sci. 2012, 67, 811–820. [Google Scholar] [CrossRef]
- Liu, Y.; Liu, G. Isorhapontigenin and resveratrol suppress oxLDL-induced proliferation and activation of ERK1/2 mitogen-activated protein kinases of bovine aortic smooth muscle cells. Biochem. Pharmacol. 2004, 67, 777–785. [Google Scholar] [CrossRef]
- Li, L.; Zhang, H.-N.; Chen, H.-Z.; Gao, P.; Zhu, L.-H.; Li, H.-L.; Lv, X.; Zhang, Q.-J.; Zhang, R.; Wang, Z. SIRT1 acts as a modulator of neointima formation following vascular injury in mice. Circ. Res. 2011, 108, 1180–1189. [Google Scholar] [CrossRef]
- Wan, Y.Z.; Gao, P.; Zhou, S.; Zhang, Z.Q.; Hao, D.L.; Lian, L.S.; Li, Y.J.; Chen, H.Z.; Liu, D.P. SIRT1-mediated epigenetic downregulation of plasminogen activeator inhibitor-1 prevents vascular endothelial replicative senescence. Aging Cell 2014, 13, 890–899. [Google Scholar] [CrossRef]
- Miyazaki, R.; Ichiki, T.; Hashimoto, T.; Inanaga, K.; Imayama, I.; Sadoshima, J.; Sunagawa, K. SIRT1, a longevity gene, downregulates angiotensin II type 1 receptor expression in vascular smooth muscle cells. Arterioscler. Thromb. Vasc. Biol. 2008, 28, 1263–1269. [Google Scholar] [CrossRef] [PubMed]
- Hossain, E.; Anand-Srivastava, M.B. Resveratrol prevents angiotensin II-induced hypertrophy of vascular smooth muscle cells through the transactivation of growth factor receptors. Can. J. Physiol. Pharmacol. 2017, 95, 945–953. [Google Scholar] [CrossRef] [PubMed]
- Sheen, J.-M.; Yu, H.-R.; Tain, Y.-L.; Tsai, W.-L.; Tiao, M.-M.; Lin, I.-C.; Tsai, C.-C.; Lin, Y.-J.; Huang, L.-T. Combined maternal and postnatal high-fat diet leads to metabolic syndrome and is effectively reversed by resveratrol: A multiple-organ study. Sci. Rep. 2018, 8, 5607. [Google Scholar] [CrossRef] [PubMed]
- Tiao, M.-M.; Lin, Y.-J.; Yu, H.-R.; Sheen, J.-M.; Lin, I.-C.; Lai, Y.-J.; Tain, Y.-L.; Huang, L.-T.; Tsai, C.-C. Resveratrol ameliorates maternal and post-weaning high-fat diet-induced nonalcoholic fatty liver disease via renin-angiotensin system. Lipids Health Dis. 2018, 17, 178. [Google Scholar] [CrossRef]
- Jang, I.; Kim, E.; Lim, J.; Kim, M.; Ban, T.; Yoon, H.; Park, C.; Chang, Y.; Choi, B. Effects of Resveratrol on the Renin-Angiotensin System in the Aging Kidney. Nutrients 2018, 10, 1741. [Google Scholar] [CrossRef]
- Kim, J.W.; Lim, S.C.; Lee, M.Y.; Lee, J.W.; Oh, W.K.; Kim, S.K.; Kang, K.W. Inhibition of neointimal formation by trans-resveratrol: Role of phosphatidyl inositol 3-kinase-dependent Nrf2 activation in heme oxygenase-1 induction. Mol. Nutr. Food Res. 2010, 54, 1497–1505. [Google Scholar] [CrossRef]
- Khandelwal, A.R.; Hebert, V.Y.; Kleinedler, J.J.; Rogers, L.K.; Ullevig, S.L.; Asmis, R.; Shi, R.; Dugas, T.R. Resveratrol and quercetin interact to inhibit neointimal hyperplasia in mice with a carotid injury. J. Nutr. 2012, 142, 1487–1494. [Google Scholar] [CrossRef][Green Version]
- Orozco-Sevilla, V.; Naftalovich, R.; Hoffmann, T.; London, D.; Czernizer, E.; Yang, C.; Dardik, A.; Dardik, H. Epigallocatechin-3-gallate is a potent phytochemical inhibitor of intimal hyperplasia in the wire-injured carotid artery. J. Vasc. Surg. 2013, 58, 1360–1365. [Google Scholar] [CrossRef][Green Version]
- Zhu, Y.; Takayama, T.; Wang, B.; Kent, A.; Zhang, M.; Binder, B.Y.; Urabe, G.; Shi, Y.; DiRenzo, D.; Goel, S.A. Restenosis Inhibition and Re-differentiation of TGFβ/Smad3-activated Smooth Muscle Cells by Resveratrol. Sci. Rep. 2017, 7, 41916. [Google Scholar] [CrossRef]
- Choi, C.I.; Koo, B.H.; Hong, D.; Kwon, H.J.; Hoe, K.L.; Won, M.H.; Kim, Y.M.; Lim, H.K.; Ryoo, S. Resveratrol is an arginase inhibitor contributing to vascular smooth muscle cell vasoconstriction via increasing cytosolic calcium. Mol. Med. Rep. 2019, 19, 3767–3774. [Google Scholar] [CrossRef]
- Dyck, G.J.; Raj, P.; Zieroth, S.; Dyck, J.R.; Ezekowitz, J.A. The Effects of Resveratrol in Patients with Cardiovascular Disease and Heart Failure: A Narrative Review. Int. J. Mol. Sci. 2019, 20, 904. [Google Scholar] [CrossRef] [PubMed]
- Oliver, J.J.; Webb, D.J. Noninvasive assessment of arterial stiffness and risk of atherosclerotic events. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 554–566. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-J.; O’Brien, A.B.; Shrive, N.G.; Parker, K.H.; Tyberg, J.V. Time-domain representation of ventricular-arterial coupling as a windkessel and wave system. Am. J. Physiol. Heart Circ. Physiol. 2003, 284, H1358–H1368. [Google Scholar] [CrossRef] [PubMed]
- Lakatta, E.G. Arterial and cardiac aging: Major shareholders in cardiovascular disease enterprises. Circulation 2003, 107, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Roy, C.S. The elastic properties of the arterial wall. J. Physiol. 1881, 3, 125–159. [Google Scholar] [CrossRef] [PubMed]
- Lacolley, P.; Challande, P.; Osborne-Pellegrin, M.; Regnault, V. Genetics and pathophysiology of arterial stiffness. Cardiovasc. Res. 2008, 81, 637–648. [Google Scholar] [CrossRef]
- Petit, M.; Guihot, A.-L.; Grimaud, L.; Vessieres, E.; Toutain, B.; Menet, M.-C.; Nivet-Antoine, V.; Arnal, J.-F.; Loufrani, L.; Procaccio, V. Resveratrol improved flow-mediated outward arterial remodeling in ovariectomized rats with hypertrophic effect at high dose. PLoS ONE 2016, 11, e0146148. [Google Scholar] [CrossRef]
- Ahmet, I.; Tae, H.-J.; Lakatta, E.G.; Talan, M. Long-term low dose dietary resveratrol supplement reduces cardiovascular structural and functional deterioration in chronic heart failure in rats. Can. J. Physiol. Pharmacol. 2016, 95, 268–274. [Google Scholar] [CrossRef]
- Behbahani, J.; Thandapilly, S.J.; Louis, X.L.; Huang, Y.; Shao, Z.; Kopilas, M.A.; Wojciechowski, P.; Netticadan, T.; Anderson, H.D. Resveratrol and small artery compliance and remodeling in the spontaneously hypertensive rat. Am. J. Hypertens. 2010, 23, 1273–1278. [Google Scholar] [CrossRef]
- Jacob, M.P. Extracellular matrix remodeling and matrix metalloproteinases in the vascular wall during aging and in pathological conditions. Biomed. Pharmacother. 2003, 57, 195–202. [Google Scholar] [CrossRef]
- Zaragoza, C.; Balbín, M.; López-Otín, C.; Lamas, S. Nitric oxide regulates matrix metalloprotease-13 expression and activity in endothelium. Kidney Int. 2002, 61, 804–808. [Google Scholar] [CrossRef] [PubMed]
- Upchurch, G.R.; Ford, J.W.; Weiss, S.J.; Knipp, B.S.; Peterson, D.A.; Thompson, R.W.; Eagleton, M.J.; Broady, A.J.; Proctor, M.C.; Stanley, J.C. Nitric oxide inhibition increases matrix metalloproteinase–9 expression by rat aortic smooth muscle cells in vitro. J. Vasc. Surg. 2001, 34, 76–83. [Google Scholar] [CrossRef]
- Tronc, F.; Mallat, Z.; Lehoux, S.; Wassef, M.; Esposito, B.; Tedgui, A. Role of Matrix Metalloproteinases in Blood Flow–Induced Arterial Enlargement. Arterioscler. Thromb. Vasc. Biol. 2000, 20, e120–e126. [Google Scholar] [CrossRef] [PubMed]
- Lu, P.; Takai, K.; Weaver, V.M.; Werb, Z. Extracellular matrix degradation and remodeling in development and disease. Cold Spring Harb. Perspect. Biol. 2011, 3, a005058. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, S.; Medina, C.; Ledwidge, M.; Radomski, M.W.; Gilmer, J.F. Nitric oxide-matrix metaloproteinase-9 interactions: Biological and pharmacological significance--NO and MMP-9 interactions. Biochim. Biophys. Acta 2014, 1843, 603–617. [Google Scholar] [CrossRef] [PubMed]
- Bai, Y.; Yang, H.; Zhang, G.; Hu, L.; Lei, Y.; Qin, Y.; Yang, Y.; Wang, Q.; Li, R.; Mao, Q. Inhibitory effects of resveratrol on the adhesion, migration and invasion of human bladder cancer cells. Mol. Med. Rep. 2017, 15, 885–889. [Google Scholar] [CrossRef]
- Pandey, A.K.; Bhattacharya, P.; Swet Chand Shukla, S.P.; Patnaik, R. Resveratrol inhibits matrix metalloproteinases to attenuate neuronal damage in cerebral ischemia: A molecular docking study exploring possible neuroprotection. Neural Regen. Res. 2015, 10, 568. [Google Scholar] [CrossRef]
- Farrokhi, E.; Ghatreh-Samani, K.; Salehi-Vanani, N.; Mahmoodi, A. The effect of resveratrol on expression of matrix metalloproteinase 9 and its tissue inhibitors in vascular smooth muscle cells. ARYA Atheroscler. 2018, 14, 157–162. [Google Scholar]
- Gagliano, N.; Moscheni, C.; Torri, C.; Magnani, I.; Bertelli, A.A.; Gioia, M. Effect of resveratrol on matrix metalloproteinase-2 (MMP-2) and Secreted Protein Acidic and Rich in Cysteine (SPARC) on human cultured glioblastoma cells. Biomed. Pharmacother. 2005, 59, 359–364. [Google Scholar] [CrossRef]
- Gweon, E.J.; Kim, S.J. Resveratrol induces MMP-9 and cell migration via the p38 kinase and PI-3K pathways in HT1080 human fibrosarcoma cells. Oncol. Rep. 2013, 29, 826–834. [Google Scholar] [CrossRef]
- Lee, B.; Moon, S.-K. Resveratrol Inhibits TNF-α–Induced Proliferation and Matrix Metalloproteinase Expression in Human Vascular Smooth Muscle Cells. J. Nutr. 2005, 135, 2767–2773. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Kim, S.H.; Monticone, R.E.; Lakatta, E.G. Matrix metalloproteinases promote arterial remodeling in aging, hypertension, and atherosclerosis. Hypertension 2015, 65, 698–703. [Google Scholar] [CrossRef]
- Shi, W.; Zhai, C.; Feng, W.; Wang, J.; Zhu, Y.; Li, S.; Wang, Q.; Zhang, Q.; Yan, X.; Chai, L. Resveratrol inhibits monocrotaline-induced pulmonary arterial remodeling by suppression of SphK1-mediated NF-κB activation. Life Sci. 2018, 210, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Fry, J.L.; Al Sayah, L.; Weisbrod, R.M.; Van Roy, I.; Weng, X.; Cohen, R.A.; Bachschmid, M.M.; Seta, F. Vascular smooth muscle sirtuin-1 protects against diet-induced aortic stiffness. Hypertension 2016, 68, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Kim, B.C.; Wang, M.; Huang, J.; Isak, A.; Bexiga, N.M.; Monticone, R.; Ha, T.; Lakatta, E.G.; An, S.S. TGFβ1 reinforces arterial aging in the vascular smooth muscle cell through a long-range regulation of the cytoskeletal stiffness. Sci. Rep. 2018, 8, 2668. [Google Scholar] [CrossRef] [PubMed]
- Rensen, S.; Doevendans, P.; Van Eys, G. Regulation and characteristics of vascular smooth muscle cell phenotypic diversity. Neth. Heart J. 2007, 15, 100–108. [Google Scholar] [CrossRef]
- Bailey, A.J. Molecular mechanisms of ageing in connective tissues. Mech. Ageing Dev. 2001, 122, 735–755. [Google Scholar] [CrossRef]
- Mizutani, K.; Ikeda, K.; Yamori, Y. Resveratrol inhibits AGEs-induced proliferation and collagen synthesis activity in vascular smooth muscle cells from stroke-prone spontaneously hypertensive rats. Biochem. Biophys. Res. Commun. 2000, 274, 61–67. [Google Scholar] [CrossRef]
- Cardona, F.; Andrés-Lacueva, C.; Tulipani, S.; Tinahones, F.J.; Queipo-Ortuño, M.I. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 2013, 24, 1415–1422. [Google Scholar] [CrossRef]
- Castaner, O.; Goday, A.; Park, Y.-M.; Lee, S.-H.; Magkos, F.; Shiow, S.-A.T.E.; Schröder, H. The gut microbiome profile in obesity: A systematic review. Int. J. Endocrinol. 2018, 2018, 9. [Google Scholar] [CrossRef]
- Qin, J.; Li, R.; Raes, J.; Arumugam, M.; Burgdorf, K.S.; Manichanh, C.; Nielsen, T.; Pons, N.; Levenez, F.; Yamada, T. A human gut microbial gene catalogue established by metagenomic sequencing. Nature 2010, 464, 59. [Google Scholar] [CrossRef] [PubMed]
- Ley, R.E.; Bäckhed, F.; Turnbaugh, P.; Lozupone, C.A.; Knight, R.D.; Gordon, J.I. Obesity alters gut microbial ecology. Proc. Natl. Acad. Sci. USA 2005, 102, 11070–11075. [Google Scholar] [CrossRef] [PubMed]
- Espín, J.C.; González-Sarrías, A.; Tomás-Barberán, F.A. The gut microbiota: A key factor in the therapeutic effects of (poly) phenols. Biochem. Pharmacol. 2017, 139, 82–93. [Google Scholar] [CrossRef] [PubMed]
- Kiouptsi, K.; Reinhardt, C. Contribution of the commensal microbiota to atherosclerosis and arterial thrombosis. Br. J. Pharmacol. 2018, 175, 4439–4449. [Google Scholar] [CrossRef]
- De Bruyne, T.; Steenput, B.; Roth, L.; De Meyer, G.R.Y.; dos Santos, C.N.; Valentová, K.; Dambrova, M.; Hermans, N. Dietary Polyphenols Targeting Arterial Stiffness: Interplay of Contributing Mechanisms and Gut Microbiome-Related Metabolism. Nutrients 2019, 11, 578. [Google Scholar] [CrossRef]
- Brunt, V.E.; Gioscia-Ryan, R.A.; Richey, J.J.; Zigler, M.C.; Cuevas, L.M.; Gonzalez, A.; Vázquez-Baeza, Y.; Battson, M.L.; Smithson, A.T.; Gilley, A.D. Suppression of the gut microbiome ameliorates age-related arterial dysfunction and oxidative stress in mice. J. Physiol. 2019, 597, 2361–2378. [Google Scholar] [CrossRef]
- Battson, M.L.; Lee, D.M.; Li Puma, L.C.; Ecton, K.E.; Thomas, K.N.; Febvre, H.P.; Chicco, A.J.; Weir, T.L.; Gentile, C.L. Gut microbiota regulates cardiac ischemic tolerance and aortic stiffness in obesity. Am. J. Physiol. Heart Circ. Physiol. 2019, 317, H1210–H1220. [Google Scholar] [CrossRef]
- Wun, K.; Theriault, B.R.; Pierre, J.F.; Chen, E.B.; Leone, V.A.; Harris, K.G.; Xiong, L.; Jiang, Q.; Spedale, M.; Eskandari, O.M. Microbiota control acute arterial inflammation and neointimal hyperplasia development after arterial injury. PLoS ONE 2018, 13, e0208426. [Google Scholar] [CrossRef]
- Nirmalkar, K.; Murugesan, S.; Pizano-Zárate, M.; Villalobos-Flores, L.; García-González, C.; Morales-Hernández, R.; Nuñez-Hernández, J.; Hernández-Quiroz, F.; Romero-Figueroa, M.; Hernández-Guerrero, C. Gut Microbiota and Endothelial Dysfunction Markers in Obese Mexican Children and Adolescents. Nutrients 2018, 10, 2009. [Google Scholar] [CrossRef]
- Beale, A.L.; Kaye, D.M.; Marques, F.Z. The role of the gut microbiome in sex differences in arterial pressure. Biol. Sex Differ. 2019, 10, 22. [Google Scholar] [CrossRef]
- Valdés, L.; Cuervo, A.; Salazar, N.; Ruas-Madiedo, P.; Gueimonde, M.; González, S. The relationship between phenolic compounds from diet and microbiota: Impact on human health. Food Funct. 2015, 6, 2424–2439. [Google Scholar] [CrossRef]
- Karbach, S.H.; Schönfelder, T.; Brandão, I.; Wilms, E.; Hörmann, N.; Jäckel, S.; Schüler, R.; Finger, S.; Knorr, M.; Lagrange, J. Gut microbiota promote angiotensin II–induced arterial hypertension and vascular dysfunction. J. Am. Heart Assoc. 2016, 5, e003698. [Google Scholar] [CrossRef] [PubMed]
- Paulos, C.M.; Wrzesinski, C.; Kaiser, A.; Hinrichs, C.S.; Chieppa, M.; Cassard, L.; Palmer, D.C.; Boni, A.; Muranski, P.; Yu, Z. Microbial translocation augments the function of adoptively transferred self/tumor-specific CD8+ T cells via TLR4 signaling. J. Clin. Investig. 2007, 117, 2197–2204. [Google Scholar] [CrossRef] [PubMed]
- Yong, K.; Dogra, G.; Boudville, N.; Chan, D.; Adams, L.; Ching, H.; Lim, E.M.; Lim, W.H. Interleukin-12 is associated with arterial stiffness in healthy individuals. Am. J. Hypertens. 2013, 26, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Callejo, M.; Mondejar-Parreño, G.; Barreira, B.; Izquierdo-Garcia, J.L.; Morales-Cano, D.; Esquivel-Ruiz, S.; Moreno, L.; Cogolludo, Á.; Duarte, J.; Perez-Vizcaino, F. Pulmonary arterial hypertension affects the rat gut microbiome. Sci. Rep. 2018, 8, 9681. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhao, Y. Gut microbiota derived metabolites in cardiovascular health and disease. Protein Cell 2018, 9, 416–431. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; DuGar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.-M. Gut flora metabolism of phosphatidylcholine promotes cardiovascular disease. Nature 2011, 472, 57. [Google Scholar] [CrossRef]
- Kau, A.L.; Ahern, P.P.; Griffin, N.W.; Goodman, A.L.; Gordon, J.I. Human nutrition, the gut microbiome and the immune system. Nature 2011, 474, 327. [Google Scholar] [CrossRef]
- Li, M.; van Esch, B.C.; Henricks, P.A.; Folkerts, G.; Garssen, J. The anti-inflammatory effects of short chain fatty acids on lipopolysaccharide-or tumor necrosis factor α-stimulated endothelial cells via activation of GPR41/43 and inhibition of HDACs. Front. Pharmacol. 2018, 9, 533. [Google Scholar] [CrossRef]
- Natarajan, N.; Hori, D.; Flavahan, S.; Steppan, J.; Flavahan, N.A.; Berkowitz, D.E.; Pluznick, J.L. Microbial short chain fatty acid metabolites lower blood pressure via endothelial G protein-coupled receptor 41. Physiol. Genom. 2016, 48, 826–834. [Google Scholar] [CrossRef]
- Pluznick, J.L.; Protzko, R.J.; Gevorgyan, H.; Peterlin, Z.; Sipos, A.; Han, J.; Brunet, I.; Wan, L.-X.; Rey, F.; Wang, T. Olfactory receptor responding to gut microbiota-derived signals plays a role in renin secretion and blood pressure regulation. Proc. Natl. Acad. Sci. 2013, 110, 4410–4415. [Google Scholar] [CrossRef] [PubMed]
- Ho, K.J.; Xiong, L.; Hubert, N.J.; Nadimpalli, A.; Wun, K.; Chang, E.B.; Kibbe, M.R. Vancomycin treatment and butyrate supplementation modulate gut microbe composition and severity of neointimal hyperplasia after arterial injury. Physiol. Rep. 2015, 3, e12627. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Wang, L.; Bhat, O.M.; Lohner, H.; Li, P.-L. Differential effects of short chain fatty acids on endothelial Nlrp3 inflammasome activation and neointima formation: Antioxidant action of butyrate. Redox Biol. 2018, 16, 21–31. [Google Scholar] [CrossRef] [PubMed]
- Kurilshikov, A.; van den Munckhof, I.C.; Chen, L.; Bonder, M.J.; Schraa, K.; Rutten, J.H.; Riksen, N.P.; de Graaf, J.; Oosting, M.; Sanna, S. Gut Microbial Associations to Plasma Metabolites Linked to Cardiovascular Phenotypes and Risk: A Cross-Sectional Study. Circ. Res. 2019, 124, 1808–1820. [Google Scholar] [CrossRef] [PubMed]
- Huo, Y.; Wu, X.; Ding, J.; Geng, Y.; Qiao, W.; Ge, A.; Guo, C.; Lv, J.; Bao, H.; Fan, W. Vascular Remodeling, Oxidative Stress, and Disrupted PPARγ Expression in Rats of Long-Term Hyperhomocysteinemia with Metabolic Disturbance. PPAR Res. 2018, 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Tyagi, N.; Moshal, K.S.; Sen, U.; Kundu, S.; Mishra, P.K.; Givvimani, S.; Tyagi, S.C. Homocysteine decreases blood flow to the brain due to vascular resistance in carotid artery. Neurochem. Int. 2008, 53, 214–219. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Munjal, C.; Givvimani, S.; Qipshidze, N.; Tyagi, N.; Falcone, J.; Tyagi, S. Mesenteric vascular remodeling in hyperhomocysteinemia. Mol. Cell. Biochem. 2011, 348, 99–108. [Google Scholar] [CrossRef]
- Zhang, J.-X.; Wang, Z.-M.; Zhang, J.-J.; Zhu, L.-L.; Gao, X.-F.; Chen, S.-L. Association of glutathione peroxidase-1 (GPx-1) rs1050450 Pro198Leu and Pro197Leu polymorphisms with cardiovascular risk: A meta-analysis of observational studies. J. Geriatr. Cardiol. JGC 2014, 11, 141. [Google Scholar]
- Hong, M.K.; Park, S.W.; Lee, C.W.; Choi, S.W.; Song, J.M.; Kang, D.H.; Song, J.K.; Kim, J.J.; Park, S.J. Elevated homocysteine levels might be associated with coronary artery remodeling in patients with stable angina: An intravascular ultrasound study. Clin. Cardiol. Int. Index. Peer Rev. J. Adv. Treat. Cardiovasc. Dis. 2002, 25, 225–229. [Google Scholar] [CrossRef]
- Velasquez, M.; Ramezani, A.; Manal, A.; Raj, D. Trimethylamine N-oxide: the good, the bad and the unknown. Toxins 2016, 8, 326. [Google Scholar] [CrossRef]
- Rath, S.; Heidrich, B.; Pieper, D.H.; Vital, M. Uncovering the trimethylamine-producing bacteria of the human gut microbiota. Microbiome 2017, 5, 54. [Google Scholar] [CrossRef] [PubMed]
- Senthong, V.; Wang, Z.; Li, X.S.; Fan, Y.; Wu, Y.; Wilson Tang, W.; Hazen, S.L. Intestinal microbiota-generated metabolite trimethylamine-N-oxide and 5-year mortality risk in stable coronary artery disease: The contributory role of intestinal microbiota in a COURAGE-like patient cohort. J. Am. Heart Assoc. 2016, 5, e002816. [Google Scholar] [CrossRef] [PubMed]
- Randrianarisoa, E.; Lehn-Stefan, A.; Wang, X.; Hoene, M.; Peter, A.; Heinzmann, S.S.; Zhao, X.; Königsrainer, I.; Königsrainer, A.; Balletshofer, B. Relationship of serum trimethylamine N-oxide (TMAO) levels with early atherosclerosis in humans. Sci. Rep. 2016, 6, 26745. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Gregory, J.C.; Org, E.; Buffa, J.A.; Gupta, N.; Wang, Z.; Li, L.; Fu, X.; Wu, Y.; Mehrabian, M. Gut microbial metabolite TMAO enhances platelet hyperreactivity and thrombosis risk. Cell 2016, 165, 111–124. [Google Scholar] [CrossRef]
- Li, T.; Chen, Y.; Gua, C.; Li, X. Elevated circulating trimethylamine N-oxide levels contribute to endothelial dysfunction in aged rats through vascular inflammation and oxidative stress. Front. Physiol. 2017, 8, 350. [Google Scholar] [CrossRef]
- Barreto, F.C.; Barreto, D.V.; Liabeuf, S.; Meert, N.; Glorieux, G.; Temmar, M.; Choukroun, G.; Vanholder, R.; Massy, Z.A.; Group EUTWG. Serum indoxyl sulfate is associated with vascular disease and mortality in chronic kidney disease patients. Clin. J. Am. Soc. Nephrol. 2009, 4, 1551–1558. [Google Scholar] [CrossRef]
- Chitalia, V.C.; Shivanna, S.; Martorell, J.; Balcells, M.; Bosch, I.; Kolandaivelu, K.; Edelman, E.R. Uremic serum and solutes increase post–vascular interventional thrombotic risk through altered stability of smooth muscle cell tissue factor. Circulation 2013, 127, 365–376. [Google Scholar] [CrossRef]
- Eckers, A.; Jakob, S.; Heiss, C.; Haarmann-Stemmann, T.; Goy, C.; Brinkmann, V.; Cortese-Krott, M.M.; Sansone, R.; Esser, C.; Ale-Agha, N. The aryl hydrocarbon receptor promotes aging phenotypes across species. Sci. Rep. 2016, 6, 19618. [Google Scholar] [CrossRef]
- Hollestelle, S.C.; de Vries, M.R.; van Keulen, J.K.; Schoneveld, A.H.; Vink, A.; Strijder, C.F.; van Middelaar, B.J.; Pasterkamp, G.; Quax, P.H.; de Kleijn, D.P. Toll-like receptor 4 is involved in outward arterial remodeling. Circulation 2004, 109, 393–398. [Google Scholar] [CrossRef]
- Li, H.; Xu, H.; Liu, S. Toll-like receptors 4 induces expression of matrix metalloproteinase-9 in human aortic smooth muscle cells. Mol. Biol. Rep. 2011, 38, 1419–1423. [Google Scholar] [CrossRef]
- Ma, J.; Li, H. The role of gut microbiota in atherosclerosis and hypertension. Front. Pharmacol. 2018, 9, 1082. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Xu, H.; Sun, B. Lipopolysaccharide regulates MMP-9 expression through TLR4/NF-κB signaling in human arterial smooth muscle cells. Mol. Med. Rep. 2012, 6, 774–778. [Google Scholar] [CrossRef] [PubMed]
- Tang, P.C.; Qin, L.; Zielonka, J.; Zhou, J.; Matte-Martone, C.; Bergaya, S.; van Rooijen, N.; Shlomchik, W.D.; Min, W.; Sessa, W.C. MyD88-dependent, superoxide-initiated inflammation is necessary for flow-mediated inward remodeling of conduit arteries. J. Exp. Med. 2008, 205, 3159–3171. [Google Scholar] [CrossRef]
- Serrano, M.; Moreno-Navarrete, J.M.; Puig, J.; Moreno, M.; Guerra, E.; Ortega, F.; Xifra, G.; Ricart, W.; Fernández-Real, J.M. Serum lipopolysaccharide-binding protein as a marker of atherosclerosis. Atherosclerosis 2013, 230, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Sakura, T.; Morioka, T.; Shioi, A.; Kakutani, Y.; Miki, Y.; Yamazaki, Y.; Motoyama, K.; Mori, K.; Fukumoto, S.; Shoji, T. Lipopolysaccharide-binding protein is associated with arterial stiffness in patients with type 2 diabetes: A cross-sectional study. Cardiovasc. Diabetol. 2017, 16, 62. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, L.; Youssef, S.; Bhattacharya, S.; Kenealey, J.; Polans, A.S.; van Ginkel, P.R. Resveratrol: Challenges in translation to the clinic—A critical discussion. Clin. Cancer Res. 2010, 16, 5942–5948. [Google Scholar] [CrossRef]
- Anhê, F.F.; Roy, D.; Pilon, G.; Dudonné, S.; Matamoros, S.; Varin, T.V.; Garofalo, C.; Moine, Q.; Desjardins, Y.; Levy, E. A polyphenol-rich cranberry extract protects from diet-induced obesity, insulin resistance and intestinal inflammation in association with increased Akkermansia spp. population in the gut microbiota of mice. Gut 2015, 64, 872–883. [Google Scholar] [CrossRef]
- Etxeberria, U.; Fernández-Quintela, A.; Milagro, F.I.; Aguirre, L.; Martínez, J.A.; Portillo, M.P. Impact of polyphenols and polyphenol-rich dietary sources on gut microbiota composition. J. Agric. Food Chem. 2013, 61, 9517–9533. [Google Scholar] [CrossRef]
- Man, A.W.; Xia, N.; Daiber, A.; Li, H. The roles of gut microbiota and circadian rhythm in the cardiovascular protective effects of polyphenols. Br. J. Pharmacol. 2019. [Google Scholar] [CrossRef]
- Fabris, S.; Momo, F.; Ravagnan, G.; Stevanato, R. Antioxidant properties of resveratrol and piceid on lipid peroxidation in micelles and monolamellar liposomes. Biophys. Chem. 2008, 135, 76–83. [Google Scholar] [CrossRef]
- Wang, H.-L.; Gao, J.-P.; Han, Y.-L.; Xu, X.; Wu, R.; Gao, Y.; Cui, X.-H. Comparative studies of polydatin and resveratrol on mutual transformation and antioxidative effect in vivo. Phytomedicine 2015, 22, 553–559. [Google Scholar] [CrossRef] [PubMed]
- Du, Q.-H.; Peng, C.; Zhang, H. Polydatin: A review of pharmacology and pharmacokinetics. Pharm. Biol. 2013, 51, 1347–1354. [Google Scholar] [CrossRef] [PubMed]
- Cichewicz, R.H.; Kouzi, S.A. Biotransformation of resveratrol to piceid by Bacillus cereus. J. Nat. Prod. 1998, 61, 1313–1314. [Google Scholar] [CrossRef] [PubMed]
- Basholli-Salihu, M.; Schuster, R.; Mulla, D.; Praznik, W.; Viernstein, H.; Mueller, M. Bioconversion of piceid to resveratrol by selected probiotic cell extracts. Bioprocess. Biosyst. Eng. 2016, 39, 1879–1885. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, Z.; Ju, J.; Wang, X.; Qiu, W. Investigation of piceid metabolites in rat by liquid chromatography tandem mass spectrometry. J. Chromatogr. B 2011, 879, 69–74. [Google Scholar] [CrossRef]
- Bode, L.M.; Bunzel, D.; Huch, M.; Cho, G.-S.; Ruhland, D.; Bunzel, M.; Bub, A.; Franz, C.M.; Kulling, S.E. In vivo and in vitro metabolism of trans-resveratrol by human gut microbiota. Am. J. Clin. Nutr. 2013, 97, 295–309. [Google Scholar] [CrossRef]
- Rotches-Ribalta, M.; Andres-Lacueva, C.; Estruch, R.; Escribano, E.; Urpi-Sarda, M. Pharmacokinetics of resveratrol metabolic profile in healthy humans after moderate consumption of red wine and grape extract tablets. Pharm. Res. 2012, 66, 375–382. [Google Scholar] [CrossRef]
- Vogl, S.; Atanasov, A.; Binder, M.; Bulusu, M.; Zehl, M.; Fakhrudin, N.; Heiss, E.; Picker, P.; Wawrosch, C.; Saukel, J. The herbal drug Melampyrum pratense L.(Koch): isolation and identification of its bioactive compounds targeting mediators of inflammation. Evid. Based Complement. Altern. Med. 2013, 2013, 10. [Google Scholar] [CrossRef]
- Most, J.; Penders, J.; Lucchesi, M.; Goossens, G.; Blaak, E. Gut microbiota composition in relation to the metabolic response to 12-week combined polyphenol supplementation in overweight men and women. Eur. J. Clin. Nutr. 2017, 71, 1040. [Google Scholar] [CrossRef]
- Chen, M.-L.; Yi, L.; Zhang, Y.; Zhou, X.; Ran, L.; Yang, J.; Zhu, J.-D.; Zhang, Q.-Y.; Mi, M.-T. Resveratrol attenuates trimethylamine-N-oxide (TMAO)-induced atherosclerosis by regulating TMAO synthesis and bile acid metabolism via remodeling of the gut microbiota. MBio 2016, 7, e02210-15. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, Q.; Ma, W.; Tian, F.; Shen, H.; Zhou, M. A combination of quercetin and resveratrol reduces obesity in high-fat diet-fed rats by modulation of gut microbiota. Food Funct. 2017, 8, 4644–4656. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.; Sun, J.; Xia, S.; Tang, X.; Shi, Y.; Le, G. Effects of resveratrol on gut microbiota and fat storage in a mouse model with high-fat-induced obesity. Food Funct. 2014, 5, 1241–1249. [Google Scholar] [CrossRef] [PubMed]
- Huycke, M.M.; Abrams, V.; Moore, D.R. Enterococcus faecalis produces extracellular superoxide and hydrogen peroxide that damages colonic epithelial cell DNA. Carcinogenesis 2002, 23, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Deng, Q.; Xu, J.; Wang, X.; Hu, C.; Tang, H.; Huang, F. Sinapic acid and resveratrol alleviate oxidative stress with modulation of gut microbiota in high-fat diet-fed rats. Food Res. Int. 2019, 116, 1202–1211. [Google Scholar] [CrossRef]
- Sung, M.M.; Kim, T.T.; Denou, E.; Soltys, C.-L.M.; Hamza, S.M.; Byrne, N.J.; Masson, G.; Park, H.; Wishart, D.S.; Madsen, K.L. Improved glucose homeostasis in obese mice treated with resveratrol is associated with alterations in the gut microbiome. Diabetes 2017, 66, 418–425. [Google Scholar] [CrossRef]
- Wellman, A.S.; Metukuri, M.R.; Kazgan, N.; Xu, X.; Xu, Q.; Ren, N.S.; Czopik, A.; Shanahan, M.T.; Kang, A.; Chen, W. Intestinal epithelial sirtuin 1 regulates intestinal inflammation during aging in mice by altering the intestinal microbiota. Gastroenterology 2017, 153, 772–786. [Google Scholar] [CrossRef]
- Nohr, M.K.; Kroager, T.P.; Sanggaard, K.W.; Knudsen, A.D.; Stensballe, A.; Enghild, J.J.; Olholm, J.; Richelsen, B.; Pedersen, S.B. SILAC-MS Based Characterization of LPS and Resveratrol Induced Changes in Adipocyte Proteomics—Resveratrol as Ameliorating Factor on LPS Induced Changes. PLoS ONE 2016, 11, e0159747. [Google Scholar] [CrossRef]
- Ponzo, V.; Soldati, L.; Bo, S. Resveratrol: A supplementation for men or for mice? J. Transl. Med. 2014, 12, 158. [Google Scholar] [CrossRef]
- Bowey, E.; Adlercreutz, H.; Rowland, I. Metabolism of isoflavones and lignans by the gut microflora: A study in germ-free and human flora associated rats. Food Chem. Toxicol. 2003, 41, 631–636. [Google Scholar] [CrossRef]
- Bai, B.; Vanhoutte, P.M.; Wang, Y. Loss-of-SIRT1 function during vascular ageing: Hyperphosphorylation mediated by cyclin-dependent kinase 5. Trends Cardiovasc. Med. 2014, 24, 81–84. [Google Scholar] [CrossRef]
- Stein, S.; Matter, C.M. Protective roles of SIRT1 in atherosclerosis. Cell Cycle 2011, 10, 640–647. [Google Scholar] [CrossRef] [PubMed]
| Dose and Period | Study Design | Subject Status | Main Findings | Reference |
|---|---|---|---|---|
| 100 mg tablet, oligo-stilbene 27.97 mg/100 mg/day, 12 weeks | 25 volunteers; Double blind, randomized, placebo-controlled | T2D | ↓ systolic BP; ↓ cardio-ankle vascular index | [47] |
| ResVida™; 6 capsules, 30, 90, and 270 mg, single dose | 19 volunteers; Double blind, randomized, placebo-controlled | Overweight/obese/post-menopausal untreated borderline hypertension | ↑ FMD response | [48] |
| ResVida™; 75 mg capsule/day, 6 weeks | 28 obese volunteers; Double blind, randomized, placebo-controlled | Healthy | ↑ FMD response; no effect on BP and arterial compliance | [49] |
| Longevinex, 100 mg/day, 3 months | 34 patients with metabolic syndromes; Double blind, randomized, placebo-controlled | Metabolic syndromes and lifestyle-related disease | ↑ FMD response; no effect on body composition, lipid profile, interleukin-6 (IL-6) and high-sensitive C-reactive protein (hsCRP). | [50] |
| 300 mg (Bioderm Pharmacy) once daily | 24 hypertensive adults; Double blind, cross-over, randomized, placebo-controlled | Hypertension | ↑ FMD response; no effect on Augmentation Index, aortic SBP and peripheral BP | [50] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Man, A.W.C.; Li, H.; Xia, N. Resveratrol and the Interaction between Gut Microbiota and Arterial Remodelling. Nutrients 2020, 12, 119. https://doi.org/10.3390/nu12010119
Man AWC, Li H, Xia N. Resveratrol and the Interaction between Gut Microbiota and Arterial Remodelling. Nutrients. 2020; 12(1):119. https://doi.org/10.3390/nu12010119
Chicago/Turabian StyleMan, Andy W.C., Huige Li, and Ning Xia. 2020. "Resveratrol and the Interaction between Gut Microbiota and Arterial Remodelling" Nutrients 12, no. 1: 119. https://doi.org/10.3390/nu12010119
APA StyleMan, A. W. C., Li, H., & Xia, N. (2020). Resveratrol and the Interaction between Gut Microbiota and Arterial Remodelling. Nutrients, 12(1), 119. https://doi.org/10.3390/nu12010119






