Determinants and Suitability of Carotenoid Reflection Score as a Measure of Carotenoid Status
Abstract
1. Introduction
2. Materials and Methods
2.1. Setting and Participants
2.2. Measurement of Carotenoid-Containing Food Intake
2.3. Measurement of Skin Carotenoid Concentration
2.4. Statistical Analysis
3. Results
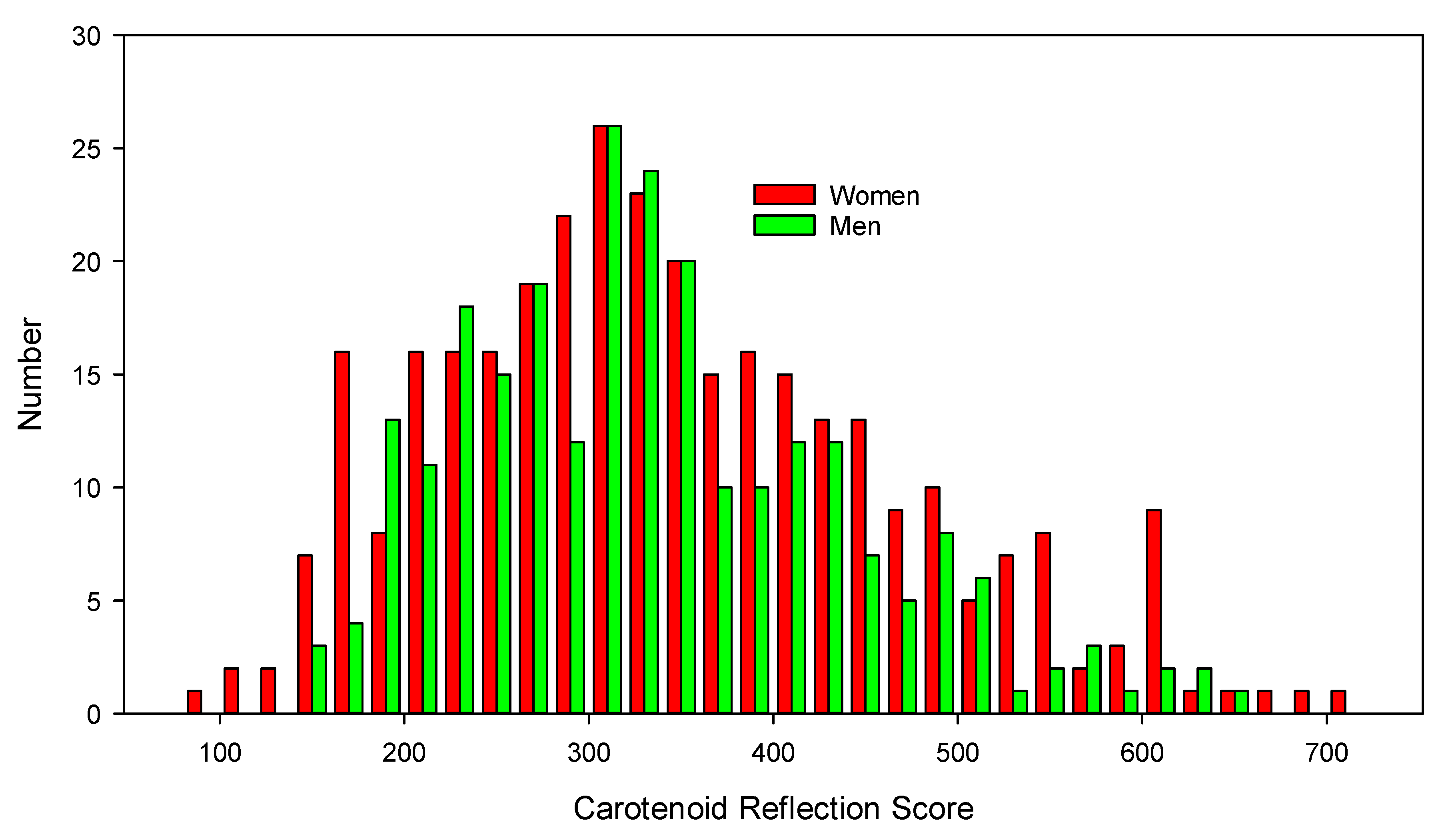
3.1. Participant Characteristics and Carotenoid Reflection Scores
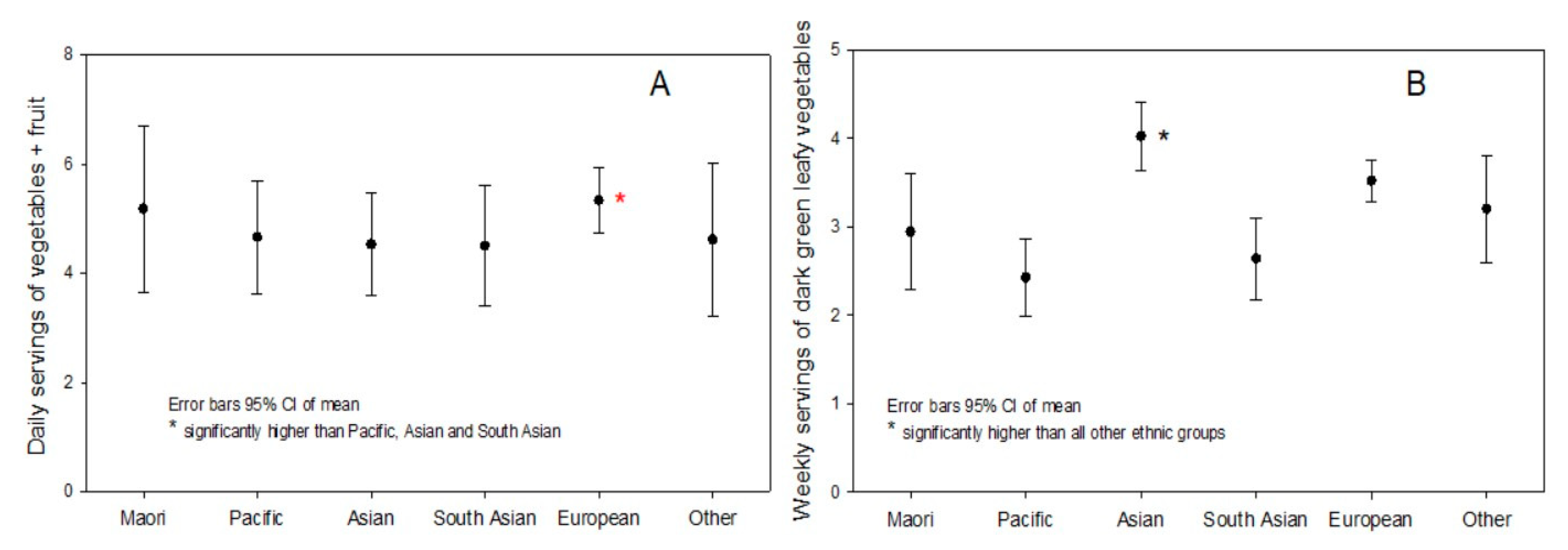
3.2. Carotenoid Food Intake
3.3. Associations between Carotenoid Reflection Score and Determinants
3.4. Predictors of Carotenoid Reflection Score
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Aune, D.; Giovannucci, E.; Boffetta, P.; Fadnes, L.T.; Keum, N.; Norat, T.; Greenwood, D.C.; Riboli, E.; Vatten, L.J.; Tonstad, S. Fruit and vegetable intake and the risk of cardiovascular disease, total cancer and all-cause mortality-a systematic review and dose-response meta-analysis of prospective studies. Int. J. Epidemiol. 2017, 46, 1029–1056. [Google Scholar] [CrossRef] [PubMed]
- Food and Agricultural Organisation of the United Nations. Food-based Dietary Guidelines. Available online: http://www.fao.org/nutrition/nutrition-education/food-dietary-guidelines/en/ (accessed on 14 September 2019).
- Naska, A.; Lagiou, A.; Lagiou, P. Dietary assessment methods in epidemiological research: Current state of the art and future prospects. F1000Reserch 2017, 6, 926. [Google Scholar] [CrossRef] [PubMed]
- Shashirekha, M.N.; Mallikarjuna, S.E.; Rajarathnam, S. Status of bioactive compounds in foods, with focus on fruits and vegetables. Crit. Rev. Food Sci. Nutr. 2015, 55, 1324–1339. [Google Scholar] [CrossRef] [PubMed]
- Moran, N.E.; Mohn, E.S.; Hason, N.; Erdman, J.W., Jr.; Johnson, E.J. Intrinsic and Extrinsic Factors Impacting Absorption, Metabolism, and Health Effects of Dietary Carotenoids. Adv. Nutr. 2018, 9, 465–492. [Google Scholar] [CrossRef] [PubMed]
- Elvira-Torales, L.I.; Garcia-Alonso, J.; Periago-Caston, M.J. Nutritional Importance of Carotenoids and Their Effect on Liver Health: A Review. Antioxidants 2019, 8, 229. [Google Scholar] [CrossRef] [PubMed]
- New Zealand Institute for Plant and Food Research Limited; Ministry of Health (New Zealand). New Zealand Food Composition Database. Available online: https://www.foodcomposition.co.nz/ (accessed on 7 November 2019).
- Ministry of Health. New Zealand Health Survey 2018/19. Available online: https://minhealthnz.shinyapps.io/nz-health-survey-2017-18-annual-data-explorer (accessed on 19 November 2019).
- Bationo, J.F.; Zeba, A.N.; Abbeddou, S.; Coulibaly, N.D.; Sombier, O.O.; Sheftel, J.; Bassole, I.H.N.; Barro, N.; Ouedraogo, J.B.; Tanumihardjo, S.A. Serum Carotenoids Reveal Poor Fruit and Vegetable Intake among Schoolchildren in Burkina Faso. Nutrients 2018, 10, 1422. [Google Scholar] [CrossRef] [PubMed]
- Couillard, C.; Lemieux, S.; Vohl, M.C.; Couture, P.; Lamarche, B. Carotenoids as biomarkers of fruit and vegetable intake in men and women. Br. J. Nutr. 2016, 116, 1206–1215. [Google Scholar] [CrossRef] [PubMed]
- Campbell, D.R.; Gross, M.D.; Martini, M.C.; Grandits, G.A.; Slavin, J.L.; Potter, J.D. Plasma carotenoids as biomarkers of vegetable and fruit intake. Cancer Epidemiol. Biomark. Prev. 1994, 3, 493–500. [Google Scholar]
- Ashton, L.M.; Pezdirc, K.B.; Hutchesson, M.J.; Rollo, M.E.; Collins, C.E. Is Skin Coloration Measured by Reflectance Spectroscopy Related to Intake of Nutrient-Dense Foods? A Cross-Sectional Evaluation in Australian Young Adults. Nutrients 2017, 10, 11. [Google Scholar] [CrossRef] [PubMed]
- Pezdirc, K.; Hutchesson, M.J.; Williams, R.L.; Rollo, M.E.; Burrows, T.L.; Wood, L.G.; Oldmeadow, C.; Collins, C.E. Consuming High-Carotenoid Fruit and Vegetables Influences Skin Yellowness and Plasma Carotenoids in Young Women: A Single-Blind Randomized Crossover Trial. J. Acad. Nutr. Diet. 2016, 116, 1257–1265. [Google Scholar] [CrossRef] [PubMed]
- Ermakov, I.V.; Ermakova, M.; Sharifzadeh, M.; Gorusupudi, A.; Farnsworth, K.; Bernstein, P.S.; Stookey, J.; Evans, J.; Arana, T.; Tao-Lew, L.; et al. Optical assessment of skin carotenoid status as a biomarker of vegetable and fruit intake. Arch. Biochem. Biophys. 2018, 646, 46–54. [Google Scholar] [CrossRef] [PubMed]
- Obana, A.; Gohto, Y.; Gellermann, W.; Ermakov, I.V.; Sasano, H.; Seto, T.; Bernstein, P.S. Skin Carotenoid Index in a large Japanese population sample. Sci. Rep. 2019, 9, 9318. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health. Content Guide 2017/18: New Zealand Health Survey; Ministry of Health: Wellington, New Zealand, 2018. [Google Scholar]
- Priyadarshani, A.M. A review on factors influencing bioaccessibility and bioefficacy of carotenoids. Crit. Rev. Food Sci. Nutr. 2017, 57, 1710–1717. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C.; Albers, R.; Antoine, J.M.; Blum, S.; Bourdet-Sicard, R.; Ferns, G.A.; Folkerts, G.; Friedmann, P.S.; Frost, G.S.; Guarner, F.; et al. Inflammatory disease processes and interactions with nutrition. Br. J. Nutr. 2009, 101 (Suppl. 1), 1–45. [Google Scholar] [CrossRef] [PubMed]
- Bonet, M.L.; Canas, J.A.; Ribot, J.; Palou, A. Carotenoids in adipose tissue biology and obesity. In Carotenoids in Nature; Strange, C., Ed.; Springer: Berlin, Germany, 2016; Volume 79, pp. 377–414. [Google Scholar]
- Devi, A.; Rush, E.; Harper, M.; Venn, B. Vitamin B12 Status of Various Ethnic Groups Living in New Zealand: An Analysis of the Adult Nutrition Survey 2008/2009. Nutrients 2018, 10, 181. [Google Scholar] [CrossRef] [PubMed]
- Jilcott Pitts, S.B.; Jahns, L.; Wu, Q.; Moran, N.E.; Bell, R.A.; Truesdale, K.P.; Laska, M.N. A non-invasive assessment of skin carotenoid status through reflection spectroscopy is a feasible, reliable and potentially valid measure of fruit and vegetable consumption in a diverse community sample. Public Health Nutr. 2018, 21, 1664–1670. [Google Scholar] [CrossRef] [PubMed]
- New Zealand Horticulture; Plant and Food Research. Fresh Facts. Available online: https://www.freshfacts.co.nz/files/freshfacts-2018.pdf (accessed on 19 November 2019).
| Carotenoid Reflection Scores | |||||
|---|---|---|---|---|---|
| Characteristic | N (Women, Men) | All | Women | Men | Difference Women − Men (95% CI) |
| All | 571 (324, 247) | 342 ± 116 | 345 ± 124 | 336 ± 105 | 10 (−9, 29) |
| Age ≤ 40 years | 334 (201, 133) | 333 ± 119 | 338 ± 122 | 325 ± 114 | 13 (−13, 39) |
| >40 years | 237 (125, 122) | 354 ± 111 1 | 358 ± 125 | 349 ± 93 | 9 (−20, 37) |
| Ethnicity | |||||
| Māori | 38 (25, 13) | 315 ± 110 | 293 ± 117 | 360 ± 83 | −66 (−140, 8) |
| Pacific | 84 (42, 42) | 257 ± 85 2 | 247 ± 84 | 267 ± 87 | −20 (−58, 17) |
| Asian | 99 (62, 37) | 388 ± 115 | 400 ± 119 | 367 ± 105 | 32 (−14, 80) |
| South Asian | 72 (37, 35) | 341 ± 113 | 340 ± 110 | 343 ± 118 | −3 (−58, 50) |
| European | 234 (137, 97) | 352 ± 111 | 359 ± 115 | 341 ± 112 | 17 (−11, 46) |
| Other | 44 (23, 21) | 367 ± 126 | 367 ± 157 | 367 ± 84 | 0 (−78, 78) |
| Food Frequency | All (Range) | Women (Range) | Men (Range) | Difference (95% CI) | p1 |
|---|---|---|---|---|---|
| Vegetables/day | 2.8 ± 1.4 (0,7) | 3.0 ± 1.6 (0,7) | 2.5 ± 1.3 (0,6) | 0.5 (0.3, 0.8) | <0.001 |
| Fruit/day | 2.1 ± 1.4 (0,7) | 2.2 ± 1.4 (0,7) | 2.0 ± 1.3 (0,7) | 0.2 (−0.1, 0.4) | 0.181 |
| Fruit + vegetables/day | 4.9 ± 2.4 (0,14) | 5.2 ± 2.5 (0,14) | 4.6 ± 2.2 (0,11) | 0.7 (0.3, 1.0) | 0.001 |
| Dark green leafy vegetables/week | 3.2 ± 2.8 (0,7) | 3.5 ± 2.0 (0,7) | 3.0 ± 2.0 (0,7) | 0.5 (0.2, 0.8) | 0.003 |
| Carrots/pumpkin/week | 2.7 ± 1.8 (0,7) | 2.7 ± 1.7 (0,7) | 2.6 ± 1.8 (0,7) | 0.0 (−0.3, 3.0) | 0.770 |
| Eggs/week | 2.6 ± 1.8 (0,7) | 2.4 ± 2.4 (0,7) | 2.8 ± 2.5 (0,7) | −0.4 (−0.8, 0.0) | 0.078 |
| Dairy/week | 4.6 ± 2.2 (0,7) | 4.5 ± 2.3 (0,7) | 4.7 ± 2.2 (0,7) | −0.2 (−0.6, 0.2) | 0.345 |
| Pearson Correlation Coefficient, r (95% CI) | |||
|---|---|---|---|
| Determinant | All | Women | Men |
| Age | 0.146 (0.065, 0.225) *** | 0.164 (0.056, 0.268) ** | 0.130 (0.005, 0.251) |
| Weight | −0.312 (−0.384, −0.236) *** | −0.397 (−0.485, −0.301) *** | −0.251 (−0.364, −0.130) *** |
| Height | −0.078 (−0.159, 0.004) | −0.078 (−0.185, 0.031) | −0.067 (−0.190, 0.058) |
| BMI | −0.338 (−0.409, −0.263) *** | −0.393 (−0.481, −0.297) *** | −0.248 (−0.362, −0.127) *** |
| Vegetables/day | 0.231 (0.152, 0.307) *** | 0.273 (0.169, 0.371) *** | 0.142 (0.017, 0.262) * |
| Fruits/day | 0.169 (0.088, 0.248) *** | 0.089 (−0.020, 0.196) | 0.303 (0.185, 0.412) *** |
| Vegetables plus fruit/ day | 0.243 (0.164, 0.319) *** | 0.227 (0.121, 0.328) *** | 0.265 (0.145, 0.377) *** |
| Dark green leafy veg/week | 0.242 (0.187, 0.340) *** | 0.266 (0.162, 0.364) *** | 0.199 (0.076, 0.316) ** |
| Carrots/pumpkin/week | 0.203 (0.123, 0.280) *** | 0.138 (0.029, 0.243) | 0.302 (0.184, 0.411) *** |
| Eggs/week | 0.067 (−0.015, 0.148) | 0.054 (−0.055, 0.162) | 0.096 (−0.029, 0.218) |
| Dairy/week | 0.055 (−0.027, 0.136) | 0.024 (−0.085, 0.133) | 0.112 (−0.013, 0.234) |
| Independent Variable | Regression Coefficient, β | SE, β | aR2 (P) |
|---|---|---|---|
| Constant | 439.660 | 24.940 | (<0.001) |
| BMI, kg/m2 | −5.440 | 0.878 | 0.109 (<0.001) |
| Dark green leafy vegetables/week | 6.670 | 2.504 | 0.138 (<0.001) |
| Carrots/pumpkin/week | 9.380 | 2.738 | 0.154 (0.001) |
| Ethnic group b | 16.981 | 7.029 | 0.162 (0.016) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rush, E.; Amoah, I.; Diep, T.; Jalili-Moghaddam, S. Determinants and Suitability of Carotenoid Reflection Score as a Measure of Carotenoid Status. Nutrients 2020, 12, 113. https://doi.org/10.3390/nu12010113
Rush E, Amoah I, Diep T, Jalili-Moghaddam S. Determinants and Suitability of Carotenoid Reflection Score as a Measure of Carotenoid Status. Nutrients. 2020; 12(1):113. https://doi.org/10.3390/nu12010113
Chicago/Turabian StyleRush, Elaine, Isaac Amoah, Tung Diep, and Shabnam Jalili-Moghaddam. 2020. "Determinants and Suitability of Carotenoid Reflection Score as a Measure of Carotenoid Status" Nutrients 12, no. 1: 113. https://doi.org/10.3390/nu12010113
APA StyleRush, E., Amoah, I., Diep, T., & Jalili-Moghaddam, S. (2020). Determinants and Suitability of Carotenoid Reflection Score as a Measure of Carotenoid Status. Nutrients, 12(1), 113. https://doi.org/10.3390/nu12010113







