Can Milk Affect Recovery from Simulated Team-Sport Match Play?
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Exercise Protocol
2.4. Nutritional Intervention and Dietary Control
2.5. Outcome Measures
2.5.1. Blood Sampling and Biochemical Analysis
2.5.2. Muscle Soreness and Muscle Tiredness
2.5.3. Symptoms of Stress
2.5.4. Muscle Function
2.6. Data Analysis
3. Results
3.1. Within-Group Effects
3.2. Between-Group Effects
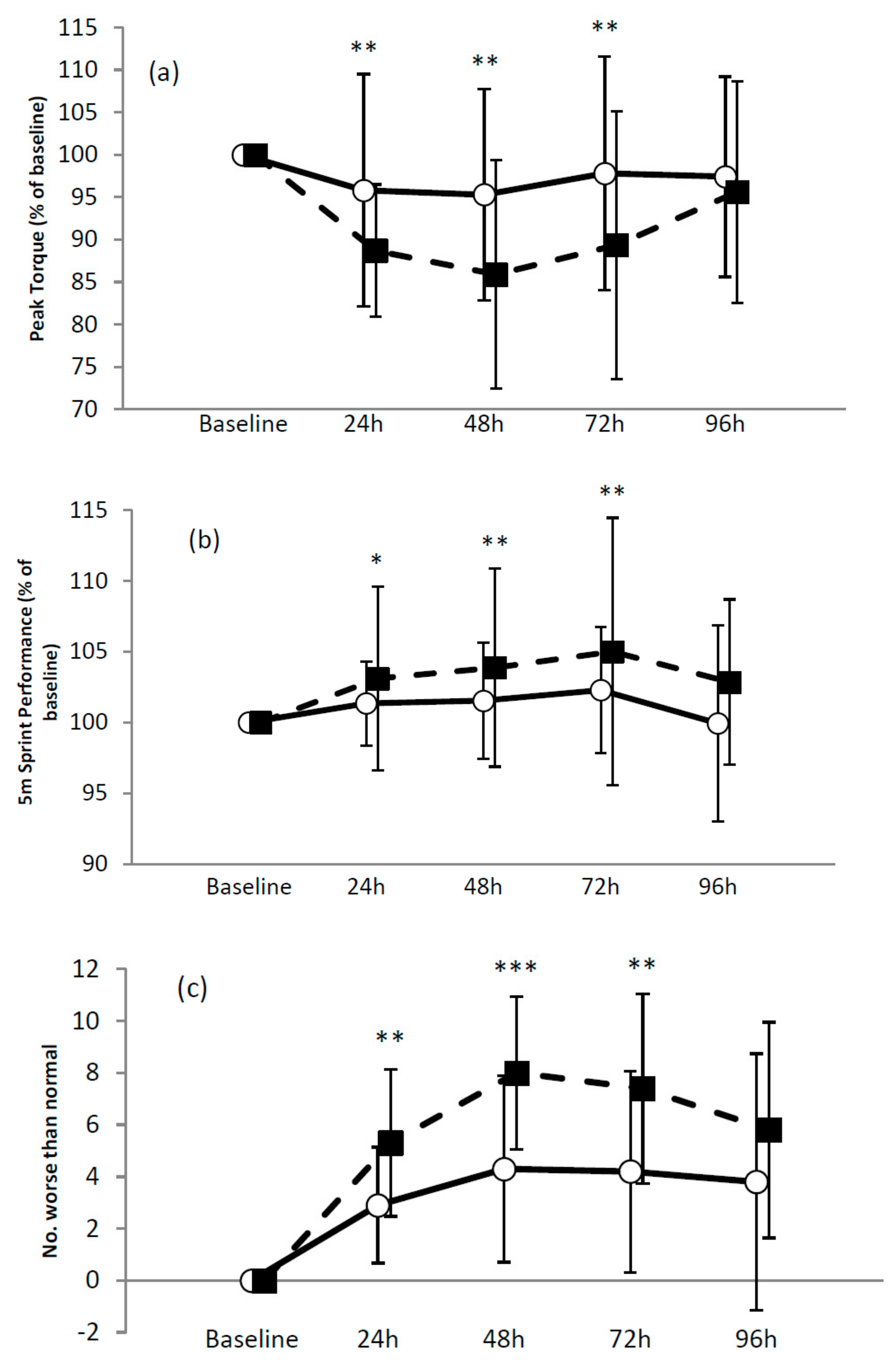
3.2.1. Muscle Function
3.2.2. Muscle Soreness and Tiredness
3.2.3. DALDA
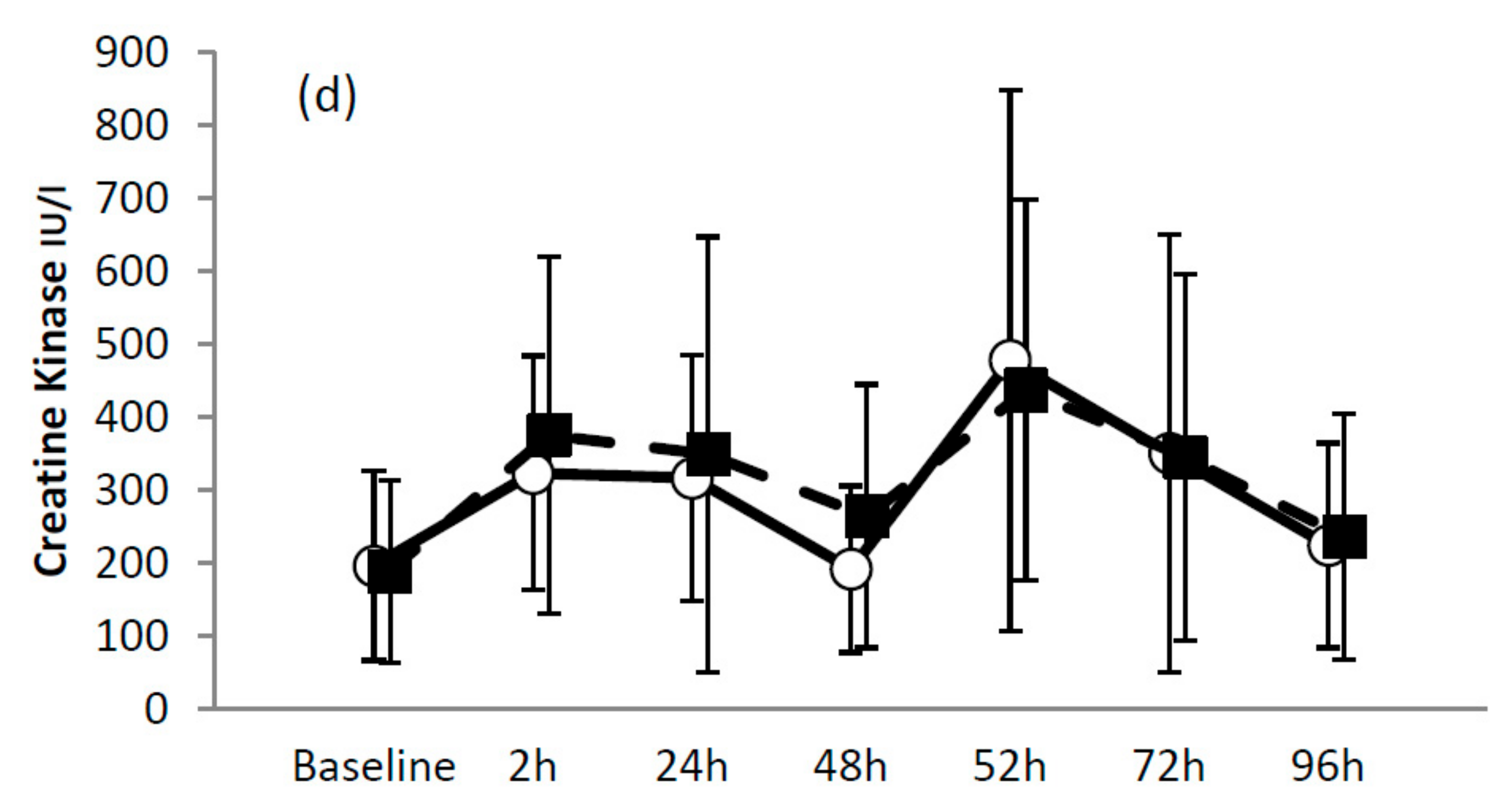
3.2.4. Serum Measures
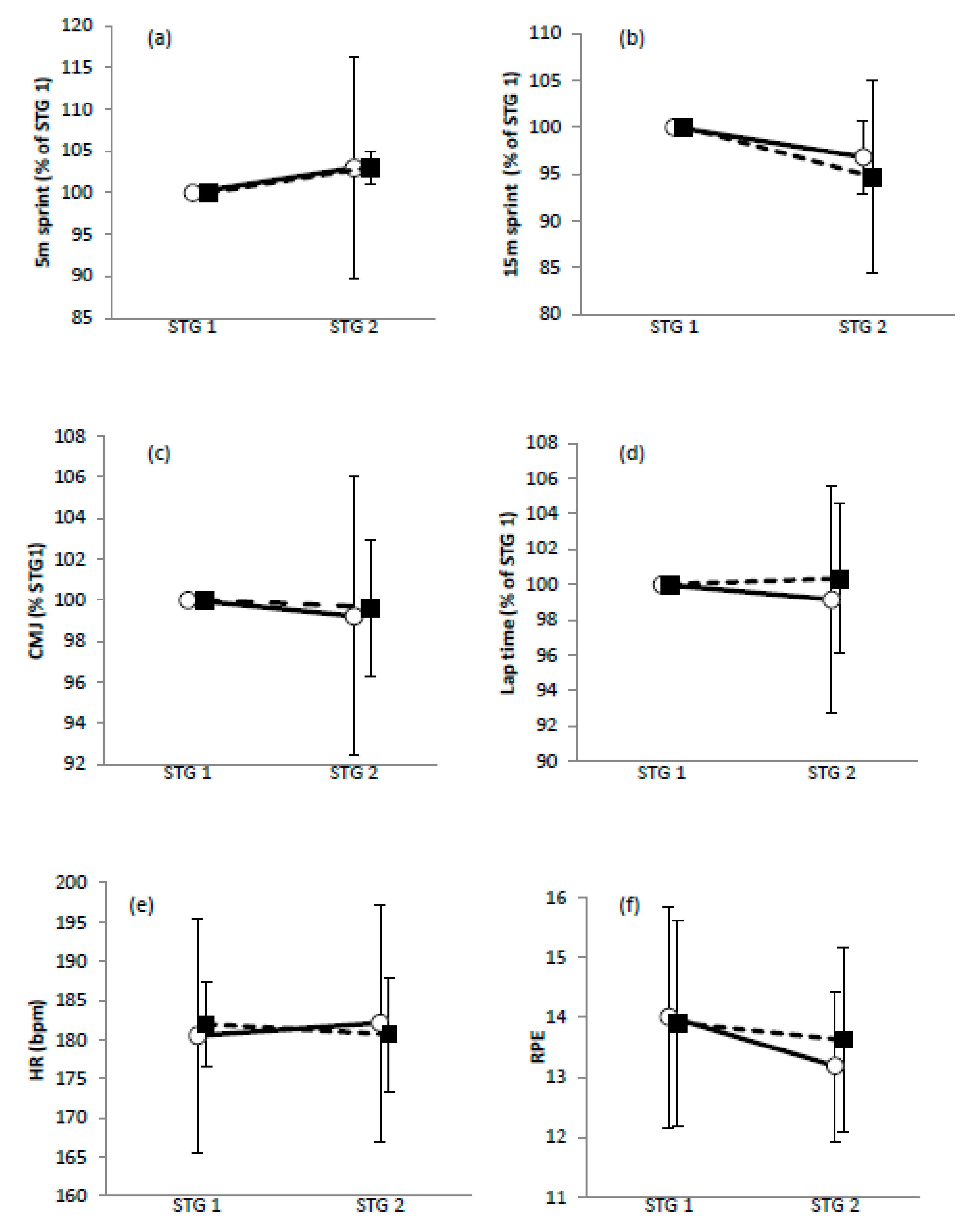
3.3. Within-Group Effects for STG Variables
3.4. Between-Group Effects for STG Variables
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Silva, J.R.; Ascensão, A.; Marques, F.; Seabra, A.; Rebelo, A.; Magalhães, J. Neuromuscular function, hormonal and redox status and muscle damage of professional soccer players after a high level competitive match. Eur. J. Appl. Physiol. 2013, 113, 2193–2201. [Google Scholar] [CrossRef] [PubMed]
- Marin, D.P.; dos Santos, M.; de Cassia, R.; Bolin, A.P.; Guerra, B.A.; Hatanaka, D.; Otton, R. Cytokines and oxidative stress status following a handball game in elite male players. Oxidative Med. Cell. Longev. 2011, 2011. [Google Scholar] [CrossRef] [PubMed]
- Andersson, J.; Raastad, T.; Nilsson, J.; Paulsen, G.; Garthe, I.; Kadi, F. Neuromuscular fatigue and recovery in elite female soccer: Effects of active recovery. Med. Sci. Sports Exerc. 2008, 40, 372–380. [Google Scholar] [CrossRef] [PubMed]
- Gravina, L.; Furiz, F.; Lekue, J.A.; Irazusta, J.; Gil, S.M. Metabolic impact of a soccer match on female players. J. Sports Sci. 2011, 29, 1345–1352. [Google Scholar] [CrossRef] [PubMed]
- Nedelec, M.; McAll, A.; Carling, C.; Legall, F.; Berthoin, S.; Dupont, G. The influence of soccer playing actions on the recovery kinetics after a soccer match. J. Strength Cond. Res. 2014, 28, 1517–1523. [Google Scholar] [CrossRef] [PubMed]
- Fatouros, I.G.; Chatzinikolaou, A.; Douroudos, I.I.; Nikolaidis, M.G.; Kyparos, A.; Margonis, K.; Michailidis, Y.; Vantarakis, A.; Taxildaris, K.; Katrabasas, I.; et al. Time-course of changes in oxidative stress and antioxidant status responses following a soccer game. J. Strength Cond. Res. 2010, 24, 3278–3286. [Google Scholar] [CrossRef] [PubMed]
- Ispirlidis, I.; Fatouros, I.G.; Jamurtas, A.Z.; Nikolaidis, M.G.; Michailidis, I.; Douroudos, I.; Margonis, K.; Chatzinikolaou, A.; Kalistratos, E.; Katrabasas, I.; et al. Time-course of changes in inflammatory markers and performance responses following a soccer game. Clin. J. Sport Med. 2008, 18, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Mohr, M.; Draganidis, D.; Chatzinikolaou, A.; Barbero-Álavarez, J.C.; Castagna, C.; Douroudos, I.; Avloniti, A.; Margeli, A.; Papassotiriou, I.; Flouris, A.D.; et al. Muscle damage, inflammatory, immune and performance responses to three football games in 1 week in competitive male players. Eur. J. Appl. Physiol. 2016, 116, 179–193. [Google Scholar] [CrossRef] [PubMed]
- Cockburn, E.; Stevenson, E.J.; Hayes, P.R.; Robson-Ansley, P.; Howatson, G. Effect of milk-based carbohydrate-protein supplement timing on the attenuation of exercise-induced muscle damage. Appl. Physiol. Nutr. Metab. 2010, 35, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Cockburn, E.; Hayes, P.R.; French, D.N.; Stevenson, E.; St Clair Gibson, A. Acute milk-based protein-CHO supplementation attenuates exercise-induced muscle damage. Appl. Physiol. Nutr. Metab. 2008, 33, 775–783. [Google Scholar] [CrossRef] [PubMed]
- Rankin, P.; Stevenson, E.; Cockburn, E. The effect of milk on the attenuation of exercise-induced muscle damage in males and females. Eur. J. Appl. Physiol. 2015, 115, 1245–1261. [Google Scholar] [CrossRef] [PubMed]
- Rankin, P.; Landy, A.; Stevenson, E.; Cockburn, E. Milk: An effective recovery drink for female athletes. Nutrients 2018, 10, 228. [Google Scholar] [CrossRef] [PubMed]
- Phillips, A.; Hamilton, D.L.; Baar, K. Signals mediating skeletal muscle remodelling by resistance exercise: P13-kinase-independent activation of mTORC1. J. Appl. Physiol. 2011, 110, 561–568. [Google Scholar] [CrossRef] [PubMed]
- Elia, D.; Stadler, K.; Horváth, V.; Jakus, J. Effect of soy- and whey protein-isolate supplemented diet on the redox parameters of trained mice. Eur. J Nutr. 2006, 45, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Kerasioti, E.; Kiskini, A.; Veskoukis, A.; Jamurtas, A.; Tsitsimpikou, C.; Tsatsakis, A.M.; Koutedakis, Y.; Stagos, D.; Kouretas, D.; Vaios, K. Effect of a special carbohydrate-protein cake on oxidative stress markers after exhaustive cycling in humans. Food Chem. Toxicol. 2012, 50, 2805–2810. [Google Scholar] [CrossRef] [PubMed]
- Alcantara, J.M.; Sanchez-Delgado, G.; Martinez-Tellez, B.; Labayen, I.; Ruiz, J.R. Impact of cow’s milk intake on exercise performance and recovery of muscle function: A systematic review. J. Int. Soc. Sports Nutr. 2019, 16, 22. [Google Scholar] [CrossRef]
- Bishop, D.; Spencer, M.; Duffield, R.; Lawrence, S. The validity of a repeated sprint ability test. J. Sci. Med. Sport 2001, 4, 19–29. [Google Scholar] [CrossRef] [PubMed]
- Wolff, S.P. Ferrous ion oxidation in presence of ferric ion indicator xylenol orange for measurement of hypoperoxides. Methods Enzymol. 1994, 233, 183–189. [Google Scholar]
- Nouroo-Zadeh, J.; Tajaddinisarmadi, J.; Wolff, S.P. Measurement of plasma hydroperoxide concentrations by the ferrous oxidation-xylenol orange assay in conjunction with triphenylphosphine. Anal. Biochem. 1994, 220, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Levine, R.L.; Garland, D.; Oliver, C.N.; Amici, A.; Climent, I.; Lenz, A.G.; Ahn, B.W.; Shaltiel, S.; Stadtman, E.R. Determination of carbonyl content in oxidatively modified proteins. Methods Enzymol. 1990, 186, 465–478. [Google Scholar] [CrossRef] [PubMed]
- Rushall, B.S. A tool for measuring stress tolerance in elite athletes. J. Appl. Sport Psychol. 1990, 2, 51–66. [Google Scholar] [CrossRef]
- Peñailillo, L.; Blazevich, A.; Numazawa, H.; Nosaka, K. Rate of force development as a measure of muscle damage. Scand. J. Med. Sci. Sports 2015, 25, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Aagaard, P.; Simonsen, E.B.; Andersen, J.L.; Magnusson, P.; Dyhre-Poulsen, P. Increased rate of force development and neural drive of human skeletal muscle following resistance training. J. Appl. Physiol. 2002, 93, 1318–1326. [Google Scholar] [CrossRef] [PubMed]
- Batterham, A.M.; Hopkins, W.G. Making meaningful inferences about magnitudes. Int. J. Sports Physiol. Perform. 2006, 1, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, W.G. Pre-Post Parallel Groups Trial (Excel Spreadsheet). 2006. Available online: http://www.sportsci.org (accessed on 23 November 2019).
- Hopkins, W.G. P values down but not yet out. In Proceedings of the 8th International Scientific Conference on Kinesiology, Opatiga, Croatia, 10–14 May 2017; Available online: http://www.sportsci.org (accessed on 23 November 2019).
- Nevill, A.; Lane, A. Why self-report ‘Liekert’ scale data should not be log-transformed. J. Sports Sci. 2007, 25, 1–2. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, W.G. A spreadsheet for analysis of straightforward controlled trials. Sportscience 2003, 7, 15. Available online: https://sportsci.org/jour/03/wghtrials.htm (accessed on 23 November 2019).
- Hopkins, W.G. Probabilities of clinical or practical significance. Sportscience 2002, 6. Available online: https://www.sportsci.org/jour/0201/wghprob.htm (accessed on 23 November 2019).
- Hopkins, W.G. Linear models and effect magnitudes for research, clinical and practical applications. Sportscience 2010, 14, 49–58. [Google Scholar]
- Groen, B.B.; Horstman, A.M.; Hamer, H.M.; De Hann, M.; van Kranenburg, J.; Bierau, J.; Poeze, M.; Wodzig, W.K.; Rasmussen, B.B.; van Loon, L.J.C. Post-prandial protein handling: You are what you just ate. PLoS ONE 2015, 10, e0141582. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.R.; Robinson, M.J.; Fry, J.L.; Tang, J.E.; Glover, E.I.; Wilkinson, S.B.; Prior, T.; Tarnopolsky, M.A.; Phillips, S.M. Ingested protein dose-response of muscle and albumin protein synthesis after resistance exercise in young men. Am. J. Clin. Nutr. 2009, 89, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Macnaughton, L.S.; Wardle, S.L.; Witard, O.C.; McGlory, C.; Hamilton, D.L.; Jeromson, S.; Lawrence, C.E.; Wallis, G.A.; Tipton, K.D. The response of muscle protein synthesis following whole-body resistance exercise is greater following 40 g than 20 g of ingested whey protein. Physiol. Rep. 2016, 4, e12893. [Google Scholar] [CrossRef] [PubMed]
- Bishop, N.; Gleeson, M.; Nicholas, C.; Ali, A. Influence of carbohydrate supplementation on plasma cytokine and neutrophil degranulation responses to high intensity intermittent exercise. Int. J. Sport Nutr. Exerc. Metab. 2002, 12, 145–156. [Google Scholar] [CrossRef] [PubMed]
- Andersson, H.; Bohn, S.K.; Raastad, T.; Paulsen, G.; Blomhoff, R.; Kadi, F. Differences in the inflammatory plama cytokine response following two elite female soccer games separated by a 72h recovery. Scand. J. Med. Sci. Sports 2009, 20, 740–747. [Google Scholar] [CrossRef] [PubMed]
- Rankin, P.; Lawlor, M.J.; Hills, F.A.; Bell, P.G.; Stevenson, E.J.; Cockburn, E. The effect of milk on recovery from repeat-sprint cycling in female team-sport athletes. Appl. Physiol. Nutr. Metab. 2018, 43, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Singh, T.K.R.; Guelfi, K.J.; Landers, G.; Dawson, B.; Bishop, D. A comparison of muscle damage, soreness and performance following a simulated contact and non-contact team sport activity circuit. J. Sci. Med. Sport 2011, 14, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Ingram, J.; Dawson, B.; Goodman, C.; Wallman, K.; Beilby, J. Effect of water immersion methods on post-exercise recovery from simulated team sport exercise. J. Sci. Med. Sport 2009, 12, 417–421. [Google Scholar] [CrossRef] [PubMed]
- Duffield, R.; Edge, J.; Merrells, R.; Hawke, E.; Barnes, M.; Simcock, D.; Gill, N. The effects of compression garments on intermittent exercise performance and recovery on consecutive days. Int. J. Sports Physiol. Perform. 2008, 3, 454–468. [Google Scholar] [CrossRef] [PubMed]
- Stupka, N.; Tiidus, P.M. Effects of ovariectomy and estrogen on ischemia-reperfusion in hindlimbs of female rats. J. Appl. Physiol. 2001, 91, 1828–1835. [Google Scholar] [CrossRef] [PubMed]
- Deminice, R.; Rosa, F.T.; Franco, G.S.; Jordao, A.A.; de Freitas, E.C. Effects of creatine supplementation on oxidative stress and inflammatory markers after repeated-sprint exercise in humans. Nutrition 2013, 29, 1127–1132. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.J.; Vasilaki, A.; McArdle, A. Cellular mechanisms underlying oxidative stress in human exercise. Free Radic. Biol. Med. 2016, 98, 13–17. [Google Scholar] [CrossRef] [PubMed]
- McHugh, M.P. Recent advances in the understanding of the repeated bout effect: The protective effect against muscle damage from a single bout of eccentric exercise. Scand. J. Med. Sci. Sports 2003, 13, 88–97. [Google Scholar] [CrossRef] [PubMed]
| Variable | Time Frame | Mean Effect a, ± or ×/÷ 90% CI b | Qualitative Inference c |
|---|---|---|---|
| (ES) | |||
| Peak Torque 60°/s Leg extension | B-24 h | 5.1, ±5.2 | Small (0.26) benefit ** |
| B-48 h | 5.2, ±6.7 | Small (0.28) benefit * | |
| B-72 h | 2.5, ±6.5 | Unclear | |
| B-96 h | 0.3, ±5.6 | Trivial * | |
| Peak Torque 60°/s Leg flexion | B-24 h | 7.4, ±8.6 | Small (0.45) benefit ** |
| B-48 h | 11.4, ±13.6 | Moderate (0.61) benefit ** | |
| B-72 h | 10.5, ±15.2 | Small (0.52) benefit ** | |
| B-96 h | 2.3, ±11.5 | Unclear | |
| Peak Torque 180°/s Leg extension | B-24 h | −1.7, ±5.4 | Trivial * |
| B-48 h | 1.5, ±6.9 | Unclear | |
| B-72 h | 3.0, ±7.1 | Unclear | |
| B-96 h | −0.9, ±6.6 | Trivial * | |
| Peak Torque 180°/s Leg flexion | B-24 h | 2.7, ±7.4 | Unclear |
| B-48 h | 6.6, ±4.6 | Small (0.32) benefit ** | |
| B-72 h | 2.2, ±8.8 | Unclear | |
| B-96 h | 0.9, ±9.4 | Unclear | |
| Rate of Force Development (RFD) (0–200 ms) | B-24 h | 18.8, ±46.6 | Unclear |
| B-48 h | 2.2, ±28.0 | Trivial * | |
| B-72 h | 1.0, ±35.8 | Unclear | |
| B-96 h | 23.9, ±45.6 | Unclear | |
| Countermovement Jump (CMJ) height | B-24 h | 0.4, ±6.8 | Unclear |
| B-48 h | 2.9, ±9.2 | Unclear | |
| B-72 h | 1.5, ±6.8 | Unclear | |
| B-96 h | 1.5, ±8.4 | Unclear | |
| Reactive Strength Index (RSI) | B-24 h | 7.4, ±11.1 | Unclear |
| B-48 h | 1.1, ±10.8 | Unclear | |
| B-72 h | −8.4, ±15.9 | Small (0.47) harm * | |
| B-96 h | −7.8, ±14.3 | Small (0.48) harm * | |
| 5 m sprint | B-24 h | −1.9, ±2.4 | Small (0.40) benefit * |
| B-48 h | −2.5, ±3.2 | Small (0.51) benefit ** | |
| B-72 h | −2.7, ±3.6 | Moderate (0.58) benefit ** | |
| B-96 h | −3.2, ±5.0 | Unclear | |
| 10 m sprint | B-24 h | −0.8, ±2.3 | Trivial ** |
| B-48 h | −2.1, ±2.6 | Small (0.50) benefit ** | |
| B-72 h | −1.3, ±2.6 | Small (0.30) benefit ** | |
| B-96 h | −2.3, ±1.6 | Small (0.53) benefit * | |
| 20 m sprint | B-24 h | 0.1, ±2.1 | Trivial * |
| B-48 h | −0.3, ±2.5 | Unclear | |
| B-72 h | −0.1, ±2.0 | Trivial * | |
| B-96 h | −0.1, ±3.5 | Unclear | |
| Muscle soreness (Squat) | B-24 h | −0.2, ±1.7 | Unclear |
| B-48 h | −1.1, ±1.3 | Unclear | |
| B-72 h | −0.6, ±1.6 | Unclear | |
| B-96 h | −0.5, ±1.5 | Unclear | |
| Muscle soreness (Isokinetic knee extension/flexion) | B-24 h | −0.7, ±0.8 | Trivial benefit * |
| B-48 h | −0.1, ±1.1 | Unclear | |
| B-72 h | −0.5, ±1.1 | Unclear | |
| B-96 h | −0.8, ±1.5 | Unclear | |
| Muscle tiredness | B-24 h | −0.1, ±1.4 | Unclear |
| B-48 h | −1.0, ±1.5 | Unclear | |
| B-72 h | −1.1, ±1.5 | Small benefit ** | |
| B-96 h | −0.5, ±1.4 | Unclear | |
| DALDA Part B | B-24 h | −2.4, ±2.0 | Small benefit ** |
| B-48 h | −3.7, ±2.6 | Small benefit *** | |
| B-72 h | −3.2, ±2.9 | Small benefit ** | |
| B-96 h | −2.0, ±3.6 | Unclear | |
| Creatine Kinase | B-2 h | 1.0, ×/÷1.5 | Unclear |
| B-24 h | 1.0, ×/÷1.8 | Unclear | |
| B-48 h | 0.9, ×/÷ 1.5 | Unclear | |
| B-52 h | 1.1, ×/÷1.6 | Unclear | |
| B-72 h | 1.0, ×/÷1.9 | Unclear | |
| B-96 h | 1.0, ×/÷1.5 | Unclear | |
| hsCRP | B-2 h | 1.3, ×/÷1.5 | Small (0.48) harm ** |
| B-24 h | 1.7, ×/÷1.6 | Moderate (0.76) harm ** | |
| B-48 h | 1.7, ×/÷1.7 | Moderate (0.61) harm ** | |
| B-52 h | 1.0, ×/÷2.1 | Unclear | |
| B-72 h | 1.0, ×/÷2.2 | Unclear | |
| B-96 h | 1.0, ×/÷2.1 | Unclear | |
| Lipid Hydroperoxides (LOOHs) | B-2 h | 1.2, ×/÷1.5 | Unclear |
| B-24 h | 0.9, ×/÷1.2 | Small (0.18) harm ** | |
| B-48 h | 1.1, ×/÷1.3 | Unclear | |
| B-52 h | 1.4, ×/÷1.7 | Unclear | |
| B-72 h | 1.1, ×/÷1.4 | Unclear | |
| B-96 h | 0.9, ×/÷1.4 | Unclear | |
| Protein Carbonyls (PCs) | B-2 h | 0.9, ×/÷2.0 | Unclear |
| B-24 h | 0.9, ×/÷1.5 | Unclear | |
| B-48 h | 0.8, ×/÷1.6 | Unclear | |
| B-52 h | 0.8, ×/÷1.5 | Unclear | |
| B-72 h | 0.9, ×/÷1.3 | Unclear | |
| B-96 h | 0.7, ×/÷1.6 | Unclear |
| Variable | Mean Effect a ± 90% CI b | Qualitative Inference c |
|---|---|---|
| 5 m sprint | −1.5 ± 7.4 | Unclear |
| 15 m sprint | −0.8 ± 3.2 | Unclear |
| CMJ | −0.6 ± 4.4 | Trivial * |
| Lap time | −1.1 ± 3.9 | Unclear |
| HR | −0.3 ± 1.3 | Trivial ** |
| RPE | −0.6 ± 1.0 | Unclear |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rankin, P.; Callanan, D.; O’Brien, K.; Davison, G.; Stevenson, E.J.; Cockburn, E. Can Milk Affect Recovery from Simulated Team-Sport Match Play? Nutrients 2020, 12, 112. https://doi.org/10.3390/nu12010112
Rankin P, Callanan D, O’Brien K, Davison G, Stevenson EJ, Cockburn E. Can Milk Affect Recovery from Simulated Team-Sport Match Play? Nutrients. 2020; 12(1):112. https://doi.org/10.3390/nu12010112
Chicago/Turabian StyleRankin, Paula, Danielle Callanan, Kevin O’Brien, Gareth Davison, Emma J. Stevenson, and Emma Cockburn. 2020. "Can Milk Affect Recovery from Simulated Team-Sport Match Play?" Nutrients 12, no. 1: 112. https://doi.org/10.3390/nu12010112
APA StyleRankin, P., Callanan, D., O’Brien, K., Davison, G., Stevenson, E. J., & Cockburn, E. (2020). Can Milk Affect Recovery from Simulated Team-Sport Match Play? Nutrients, 12(1), 112. https://doi.org/10.3390/nu12010112





