Expression of Renin-Angiotensin System Components in the Taste Organ of Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. Reverse Transcription-Polymerase Chain Reaction (RT-PCR)
2.3. In Situ Hybridization
2.4. Immunohistochemistry
2.5. Quantitative Densitometric Analysis
2.6. Quantitative PCR (qPCR)
2.7. Statistical Analysis
3. Results
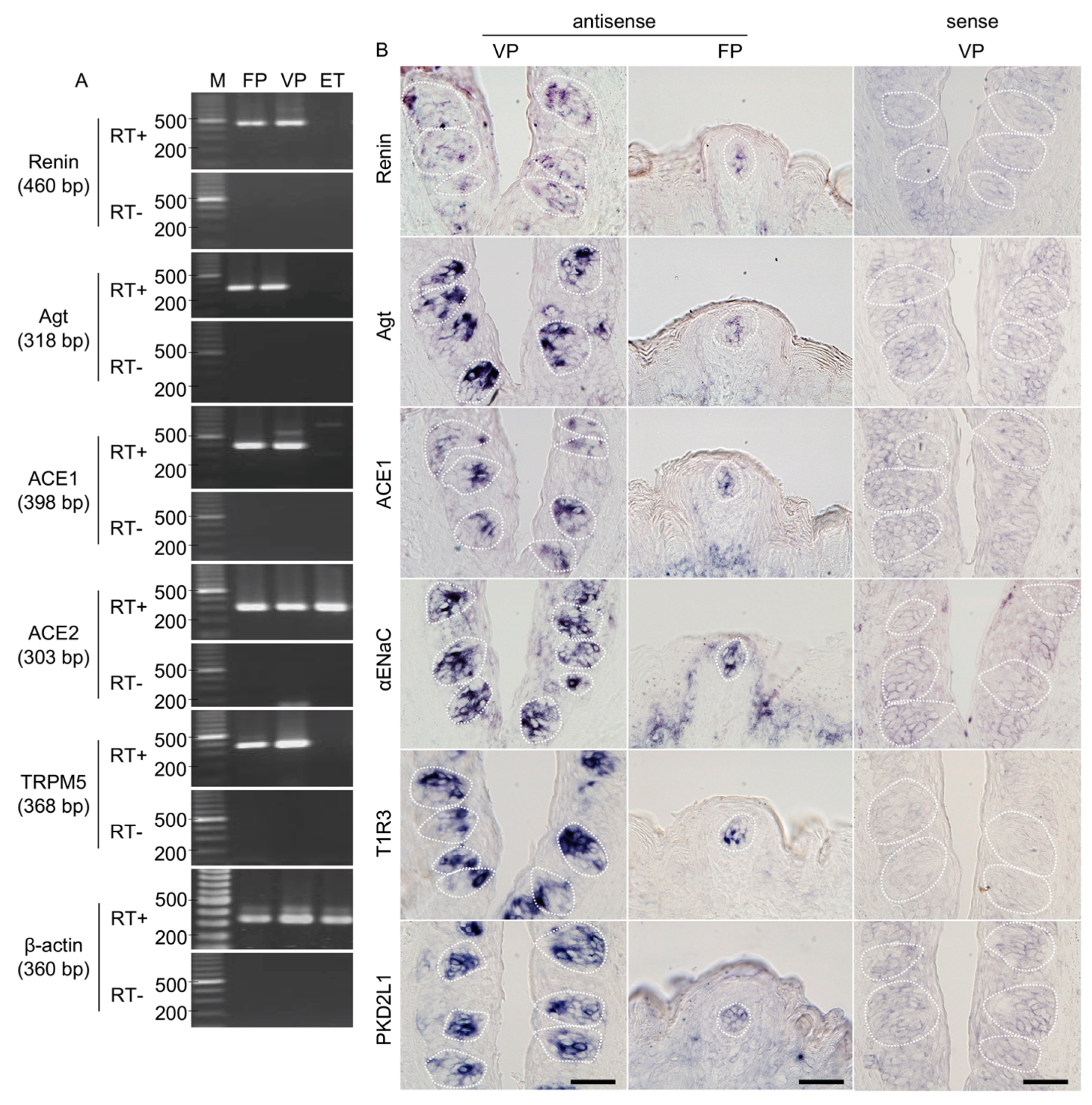
3.1. RT-PCR Reveals Renin-Angiotensin-Related Gene Expression in Mouse Taste Buds
3.2. Renin-Angiotensin-Related Genes Are Localized to a Subset of Taste Bud Cells
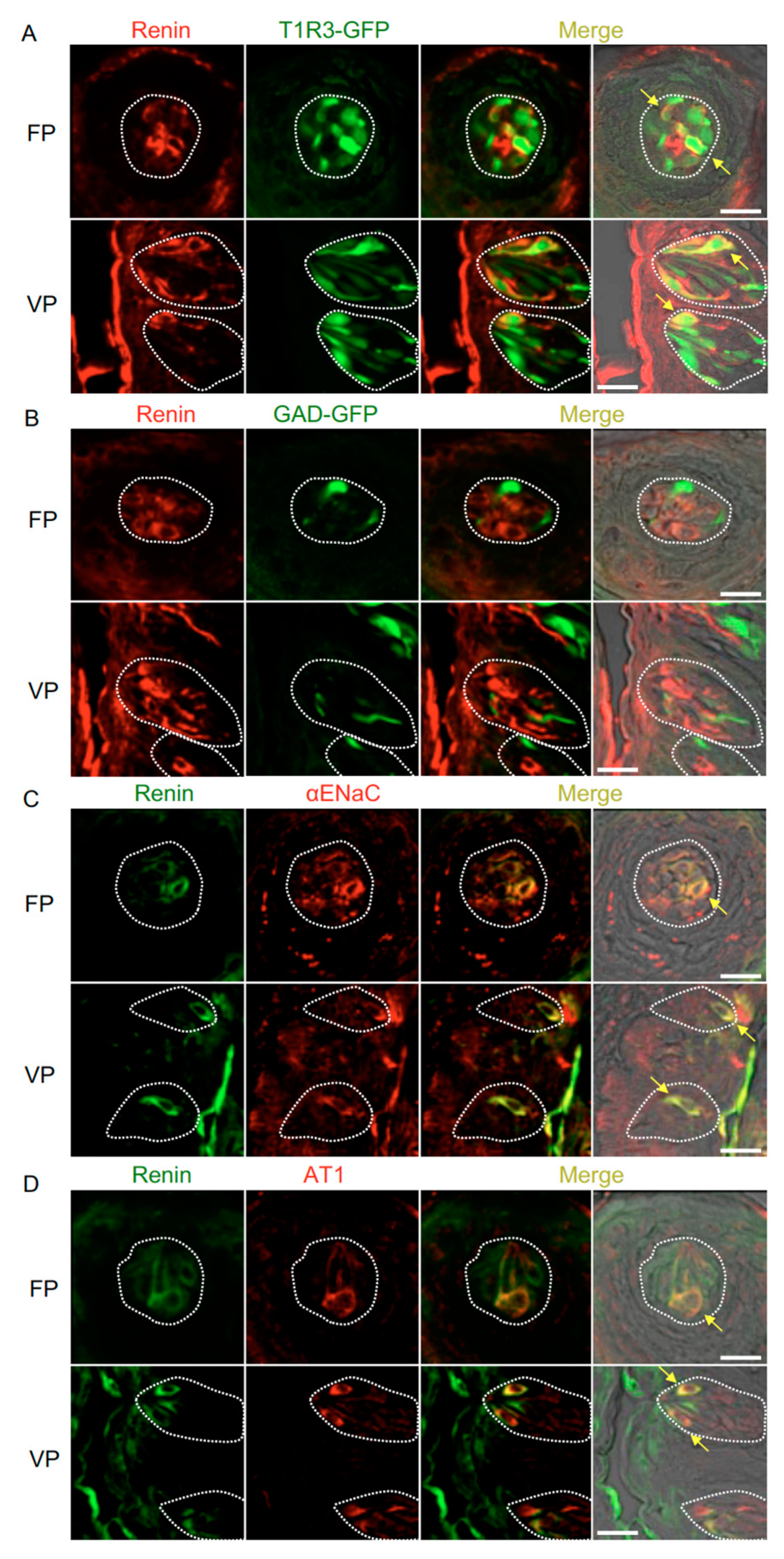
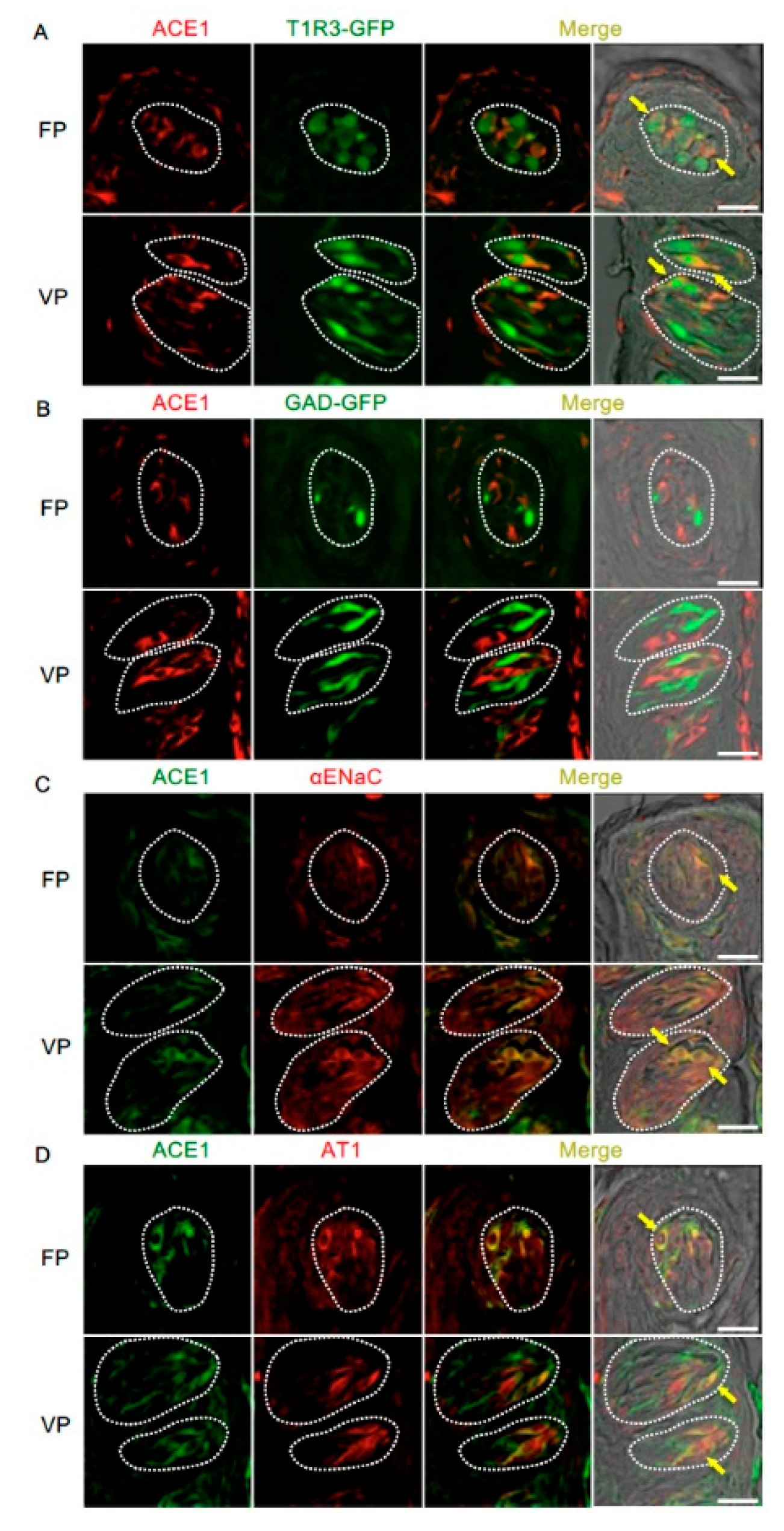
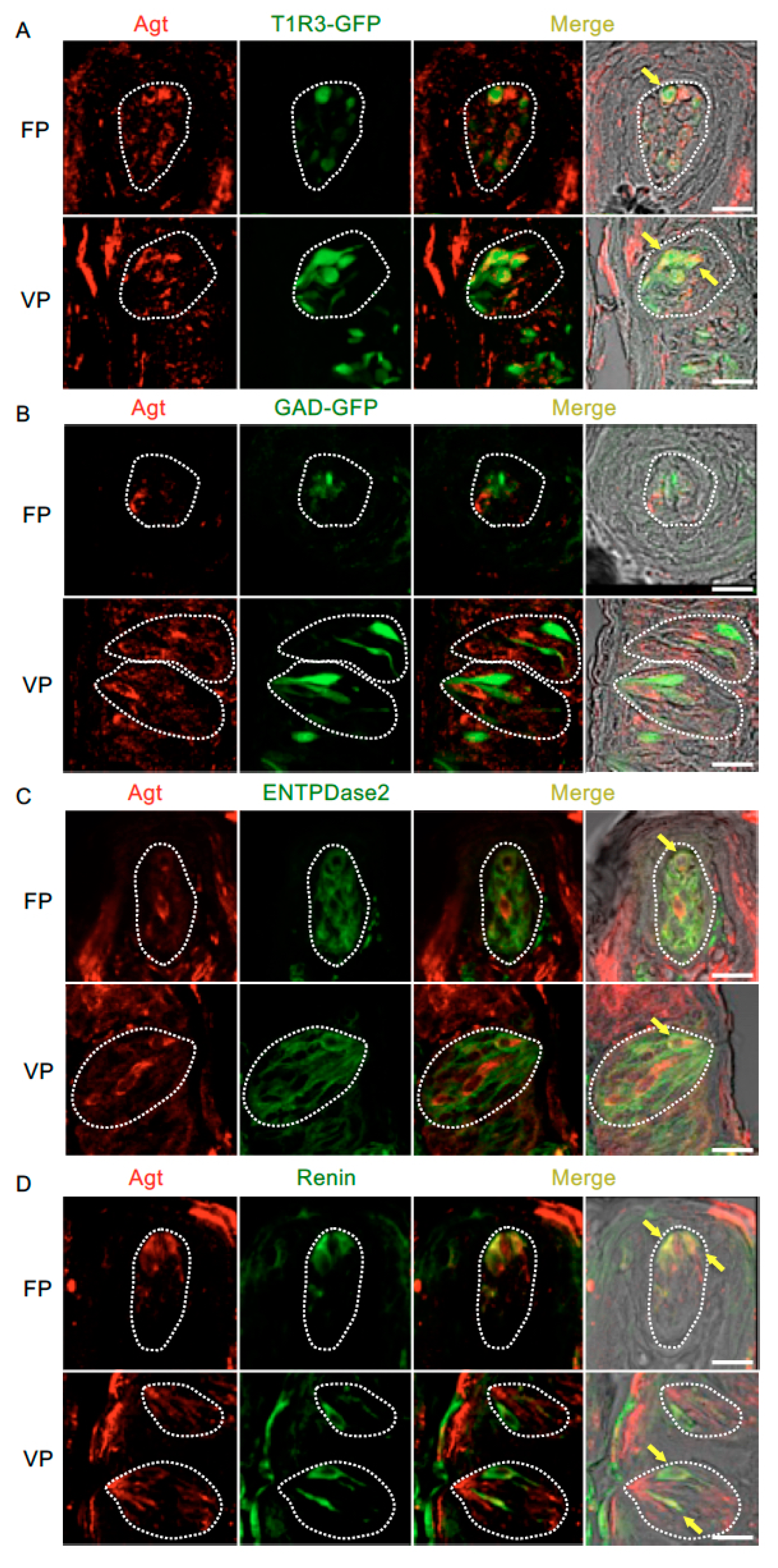
3.3. Renin-Angiotensin-Related Proteins Are Co-Expressed with T1R3 or αENaC in Taste Bud Cells
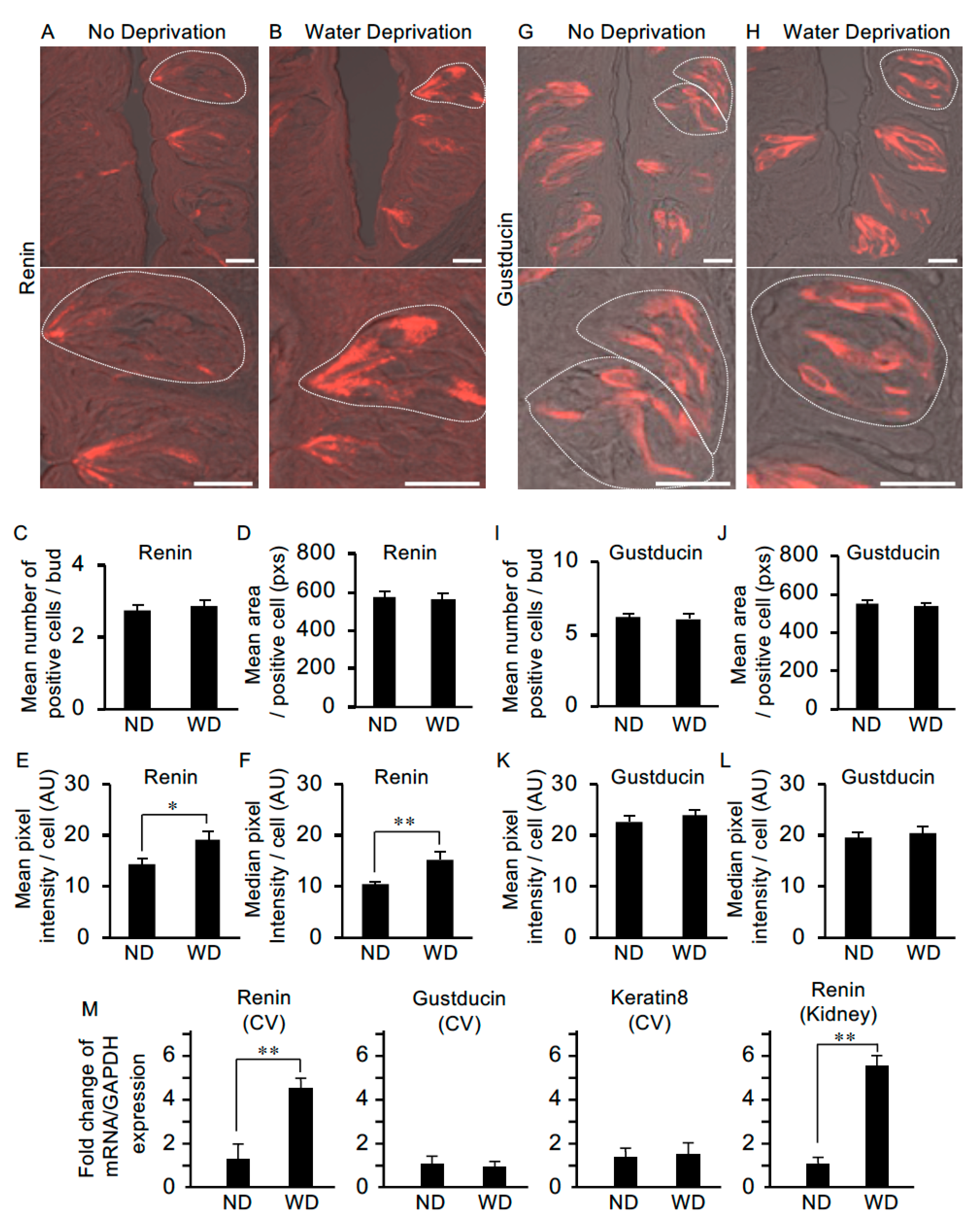
3.4. Renin Expression Is Upregulated in the Taste Buds Cells of Water-Deprived Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fyhrquist, F.; Saijonmaa, O. Renin-angiotensin system revisited. J. Intern. Med. 2008, 264, 224–236. [Google Scholar] [CrossRef] [PubMed]
- Avrith, D.B.; Fitzsimons, J.T. Increased sodium appetite in the rat induced by intracranial administration of components of the renin–angiotensin system. J. Physiol. 1980, 301, 349–364. [Google Scholar] [CrossRef] [PubMed]
- Fitts, D.A.; Thunhorst, R.L. Rapid elicitation of salt appetite by an intravenous infusion of angiotensin II in rats. Am. J. Physiol. 1996, 270, R1092–R1098. [Google Scholar] [CrossRef] [PubMed]
- Shigemura, N.; Iwata, S.; Yasumatsu, K.; Ohkuri, T.; Horio, N.; Sanematsu, K.; Yoshida, R.; Margolskee, R.F.; Ninomiya, Y. Angiotensin II modulates salty and sweet taste sensitivities. J. Neurosci. 2013, 33, 6267–6277. [Google Scholar] [CrossRef] [PubMed]
- Braun-Menéndez, E.; Fasciolo, J.C.; Leloir, L.F.; Munoz, J.M. La sustancia hipertensora de la sangre del riñón isquemido. Rev. Soc. Argent. Biol. 1939, 15, 420–425. [Google Scholar]
- Skeggs, L.T., Jr.; Khan, J.R.; Shumway, N.P. The preparation and function of the hypertensin-converting enzyme. J. Exp. Med. 1956, 103, 295–299. [Google Scholar] [CrossRef] [PubMed]
- De Gasparo, M.; Catt, K.J.; Inagami, T.; Wright, J.W.; Unger, T. International Union of Pharmacology. XXIII. The angiotension II receptors. Pharmacol. Rev. 2000, 52, 415–472. [Google Scholar]
- Donoghue, M.; Hsieh, F.; Baronas, E.; Godbout, K.; Gosselin, M.; Stagliano, N.; Donovan, M.; Woolf, B.; Robinson, K.; Jeyaseelan, R.; et al. A novel angiotensin-converting enzyme-related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1–9. Circ. Res. 2000, 87, E1–E9. [Google Scholar] [CrossRef] [PubMed]
- Fleming, I.; Kohlstedt, K.; Busse, R. The tissue renin-angiotensin system and intracellular signalling. Curr. Opin. Nephrol. Hypertens. 2006, 15, 8–13. [Google Scholar] [CrossRef]
- Paul, M.; Poyan Mehr, A.; Kreutz, R. Physiology of local renin-angiotensin systems. Physiol. Rev. 2006, 86, 747–803. [Google Scholar] [CrossRef]
- Rohrwasser, A.; Morgan, T.; Dillon, H.F.; Zhao, L.; Callaway, C.W.; Hillas, E.; Zhang, S.; Cheng, T.; Inagami, T.; Ward, K.; et al. Elements of a paracrine tubular renin-angiotensin system along the entire nephron. Hypertension 1999, 34, 1265–1274. [Google Scholar] [CrossRef] [PubMed]
- Navar, L.G.; Prieto, M.C.; Satou, R.; Kobori, H. Intrarenal angiotensin II and its contribution to the genesis of chronic hypertension. Curr. Opin. Pharmacol. 2011, 11, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Dostal, D.E.; Baker, K.M. The cardiac renin-angiotensin system: Conceptual, or a regulator of cardiac function? Circ. Res. 1999, 85, 643–650. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Yong, Q.C.; Thomas, C.M.; Baker, K.M. Intracardiac intracellular angiotensin system in diabetes. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2012, 302, R510–R517. [Google Scholar] [CrossRef] [PubMed]
- Haller, H.; Lindschau, C.; Erdmann, B.; Quass, P.; Luft, F.C. Effects of intracellular angiotensin II in vascular smooth muscle cells. Circ. Res. 1996, 79, 765–772. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.P.; Le, B.; Khode, R.; Baker, K.M.; Kumar, R. Intracellular angiotensin II production in diabetic rats is correlated with cardiomyocyte apoptosis, oxidative stress, and cardiac fibrosis. Diabetes 2008, 57, 3297–3306. [Google Scholar] [CrossRef] [PubMed]
- Damak, S.; Mosinger, B.; Margolskee, R.F. Transsynaptic transport of wheat germ agglutinin expressed in a subset of type II taste cells of transgenic mice. BMC Neurosci. 2008, 9, 96. [Google Scholar] [CrossRef] [PubMed]
- Tamamaki, N.; Yanagawa, Y.; Tomioka, R.; Miyazaki, J.; Obata, K.; Kaneko, T. Green fluorescent protein expression and colocalization with calretinin, parvalbumin, and somatostatin in the GAD67-GFP knock-in mouse. J. Comp. Neurol. 2003, 467, 60–79. [Google Scholar] [CrossRef] [PubMed]
- Kawai, K.; Sugimoto, K.; Nakashima, K.; Miura, H.; Ninomiya, Y. Leptin as a modulator of sweet taste sensitivities in mice. Proc. Natl. Acad. Sci. USA 2000, 97, 11044–11049. [Google Scholar] [CrossRef]
- Yoshida, R.; Ohkuri, T.; Jyotaki, M.; Yasuo, T.; Horio, N.; Yasumatsu, K.; Sanematsu, K.; Shigemura, N.; Yamamoto, T.; Margolskee, R.F.; et al. Endocannabinoids selectively enhance sweet taste. Proc. Natl. Acad. Sci. USA 2010, 107, 935–939. [Google Scholar] [CrossRef]
- Shigemura, N.; Ohta, R.; Kusakabe, Y.; Miura, H.; Hino, A.; Koyano, K.; Nakashima, K.; Ninomiya, Y. Leptin modulates behavioral responses to sweet substances by influencing peripheral taste structures. Endocrinology 2004, 145, 839–847. [Google Scholar] [CrossRef]
- Shigemura, N.; Miura, H.; Kusakabe, Y.; Hino, A.; Ninomiya, Y. Expression of leptin receptor (Ob-R) isoforms and signal transducers and activators of transcription (STATs) mRNAs in the mouse taste buds. Arch. Histol. Cytol. 2003, 66, 253–260. [Google Scholar] [CrossRef]
- Shigemura, N.; Islam, A.A.; Sadamitsu, C.; Yoshida, R.; Yasumatsu, K.; Ninomiya, Y. Expression of amiloride-sensitive epithelial sodium channels in mouse taste cells after chorda tympani nerve crush. Chem. Senses 2005, 30, 531–538. [Google Scholar] [CrossRef]
- Shigemura, N.; Ohkuri, T.; Sadamitsu, C.; Yasumatsu, K.; Yoshida, R.; Beauchamp, G.K.; Bachmanov, A.A.; Ninomiya, Y. Amiloride-sensitive NaCl taste responses are associated with genetic variation of ENaC alpha-subunit in mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R66–R75. [Google Scholar] [CrossRef]
- Wong, G.T.; Gannon, K.S.; Margolskee, R.F. Transduction of bitter and sweet taste by gustducin. Nature 1996, 381, 796–800. [Google Scholar] [CrossRef] [PubMed]
- Mattila, J.T.; Ojo, O.O.; Kepka-Lenhart, D.; Marino, S.; Kim, J.H.; Eum, S.Y.; Via, L.E.; Barry, C.E., 3rd; Klein, E.; Kirschner, D.E.; et al. Microenvironments in tuberculous granulomas are delineated by distinct populations of macrophage subsets and expression of nitric oxide synthase and arginase isoforms. J. Immunol. 2013, 191, 773–784. [Google Scholar] [CrossRef] [PubMed]
- Pérez, C.A.; Huang, L.; Rong, M.; Kozak, J.A.; Preuss, A.K.; Zhang, H.; Max, M.; Margolskee, R.F. A transient receptor potential channel expressed in taste receptor cells. Nat. Neurosci. 2002, 5, 1169–1176. [Google Scholar] [CrossRef]
- Zhang, Y.; Hoon, M.A.; Chandrashekar, J.; Mueller, K.L.; Cook, B.; Wu, D.; Zuker, C.S.; Ryba, N.J. Coding of sweet, bitter, and umami tastes: Different receptor cells sharing similar signaling pathways. Cell 2003, 112, 293–301. [Google Scholar] [CrossRef]
- Talavera, K.; Yasumatsu, K.; Voets, T.; Droogmans, G.; Shigemura, N.; Ninomiya, Y.; Margolskee, R.F.; Nilius, B. Heat activation of TRPM5 underlies thermal sensitivity of sweet taste. Nature 2005, 438, 1022–1025. [Google Scholar] [CrossRef]
- Damak, S.; Rong, M.; Yasumatsu, K.; Kokrashvili, Z.; Pérez, C.A.; Shigemura, N.; Yoshida, R.; Mosinger, B., Jr.; Glendinning, J.I.; Ninomiya, Y.; et al. Trpm5 null mice respond to bitter, sweet, and umami compounds. Chem. Senses 2006, 31, 253–264. [Google Scholar] [CrossRef] [PubMed]
- Chandrashekar, J.; Kuhn, C.; Oka, Y.; Yarmolinsky, D.A.; Hummler, E.; Ryba, N.J.; Zuker, C.S. The cells and peripheral representation of sodium taste in mice. Nature 2010, 464, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Nelson, G.; Hoon, M.A.; Chandrashekar, J.; Zhang, Y.; Ryba, N.J.; Zuker, C.S. Mammalian sweet taste receptors. Cell 2001, 106, 381–390. [Google Scholar] [CrossRef]
- Li, X.; Staszewski, L.; Xu, H.; Durick, K.; Zoller, M.; Adler, E. Human receptors for sweet and umami taste. Proc. Natl. Acad. Sci. USA 2002, 99, 4692–4696. [Google Scholar] [CrossRef] [PubMed]
- Ishimaru, Y.; Inada, H.; Kubota, M.; Zhuang, H.; Tominaga, M.; Matsunami, H. Transient receptor potential family members PKD1L3 and PKD2L1 form a candidate sour taste receptor. Proc. Natl. Acad. Sci. USA 2006, 103, 12569–12574. [Google Scholar] [CrossRef] [PubMed]
- Huang, A.L.; Chen, X.; Hoon, M.A.; Chandrashekar, J.; Guo, W.; Tränkner, D.; Ryba, N.J.; Zuker, C.S. The cells and logic for mammalian sour taste detection. Nature 2006, 442, 934–938. [Google Scholar] [CrossRef] [PubMed]
- Vandenbeuch, A.; Anderson, C.B.; Parnes, J.; Enjyoji, K.; Robson, S.C.; Finger, T.E.; Kinnamon, S.C. Role of the ectonucleotidase NTPDase2 in taste bud function. Proc. Natl. Acad. Sci. USA 2013, 110, 14789–14794. [Google Scholar] [CrossRef] [PubMed]
- Komlosi, P.; Fuson, A.L.; Fintha, A.; Peti-Peterdi, J.; Rosivall, L.; Warnock, D.G.; Bell, P.D. Angiotensin I conversion to angiotensin II stimulates cortical collecting duct sodium transport. Hypertension 2003, 42, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Chandrashekar, J.; Hoon, M.A.; Ryba, N.J.; Zuker, C.S. The receptors and cells for mammalian taste. Nature 2006, 444, 288–294. [Google Scholar] [CrossRef]
- Chandrashekar, J.; Mueller, K.L.; Hoon, M.A.; Adler, E.; Feng, L.; Guo, W.; Zuker, C.S.; Ryba, N.J. T2Rs function as bitter taste receptors. Cell 2000, 100, 703–711. [Google Scholar] [CrossRef]
- Hisatsune, C.; Yasumatsu, K.; Takahashi-Iwanaga, H.; Ogawa, N.; Kuroda, Y.; Yoshida, R.; Ninomiya, Y.; Mikoshiba, K. Abnormal taste perception in mice lacking the type 3 inositol 1,4,5-trisphosphate receptor. J. Biol. Chem. 2007, 282, 37225–37231. [Google Scholar] [CrossRef]
- Heck, G.L.; Mierson, S.; DeSimone, J.A. Salt taste transduction occurs through an amiloride-sensitive sodium transport pathway. Science 1984, 223, 403–405. [Google Scholar] [CrossRef] [PubMed]
- McCaughey, S.A.; Scott, T.R. Rapid induction of sodium appetite modifies taste-evoked activity in the rat nucleus of the solitary tract. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2000, 279, R1121–R1131. [Google Scholar] [CrossRef] [PubMed]
- Ferraris, R.P.; Diamond, J.M. Crypt/villus site of substrate-dependent regulation of mouse intestinal glucose transporters. Proc. Natl. Acad. Sci. USA 1993, 90, 5868–5872. [Google Scholar] [CrossRef]
- Bell, P.D.; Lapointe, J.Y.; Peti-Peterdi, J. Macula densa cell signaling. Annu. Rev. Physiol. 2003, 65, 481–500. [Google Scholar] [CrossRef] [PubMed]
- Skøtt, O.; Briggs, J.P. Direct demonstration of macula densa-mediated renin secretion. Science 1987, 237, 1618–1620. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, J.N.; Weihprecht, H.; Schnermann, J.; Skøtt, O.; Briggs, J.P. Renin release from isolated juxtaglomerular apparatus depends on macula densa chloride transport. Am. J. Physiol. 1991, 260, F486–F493. [Google Scholar] [CrossRef] [PubMed]
- He, X.R.; Greenberg, S.G.; Briggs, J.P.; Schnermann, J. Effects of furosemide and verapamil on the NaCl dependency of macula densa-mediated renin secretion. Hypertension 1995, 26, 137–142. [Google Scholar] [CrossRef]
- Peti-Peterdi, J.; Chambrey, R.; Bebok, Z.; Biemesderfer, D.; St John, P.L.; Abrahamson, D.R.; Warnock, D.G.; Bell, P.D. Macula densa Na(+)/H(+) exchange activities mediated by apical NHE2 and basolateral NHE4 isoforms. Am. J. Physiol. Renal Physiol. 2000, 278, F452–F463. [Google Scholar] [CrossRef]
- Hanner, F.; Chambrey, R.; Bourgeois, S.; Meer, E.; Mucsi, I.; Rosivall, L.; Shull, G.E.; Lorenz, J.N.; Eladari, D.; Peti-Peterdi, J. Increased renal renin content in mice lacking the Na+/H+ exchanger NHE2. Am. J. Physiol. Renal Physiol. 2008, 294, F937–F944. [Google Scholar] [CrossRef]
- Vinnikova, A.K.; Alam, R.I.; Malik, S.A.; Ereso, G.L.; Feldman, G.M.; McCarty, J.M.; Knepper, M.A.; Heck, G.L.; DeSimone, J.A.; Lyall, V. Na+-H+ exchange activity in taste receptor cells. J. Neurophysiol. 2004, 91, 1297–1313. [Google Scholar] [CrossRef]
- Prieto-Carrasquero, M.C.; Harrison-Bernard, L.M.; Kobori, H.; Ozawa, Y.; Hering-Smith, K.S.; Hamm, L.L.; Navar, L.G. Enhancement of collecting duct renin in angiotensin II-dependent hypertensive rats. Hypertension 2004, 44, 223–229. [Google Scholar] [CrossRef]
- Crowley, S.D.; Gurley, S.B.; Herrera, M.J.; Ruiz, P.; Griffiths, R.; Kumar, A.P.; Kim, H.S.; Smithies, O.; Le, T.H.; Coffman, T.M. Angiotensin II causes hypertension and cardiac hypertrophy through its receptors in the kidney. Proc. Natl. Acad. Sci. USA 2006, 103, 17985–17990. [Google Scholar] [CrossRef] [PubMed]
- Ramkumar, N.; Ying, J.; Stuart, D.; Kohan, D.E. Overexpression of renin in the collecting duct causes elevated blood pressure. Am. J. Hypertens. 2013, 26, 965–972. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, T.; Fujii, A.; Saito, N.; Kondo, H.; Ohuchi, A. Alteration of amiloride-sensitive salt taste nerve responses in aldosterone/NaCl-induced hypertensive rats. Neurosci. Res. 2016, 108, 60–66. [Google Scholar] [CrossRef] [PubMed]
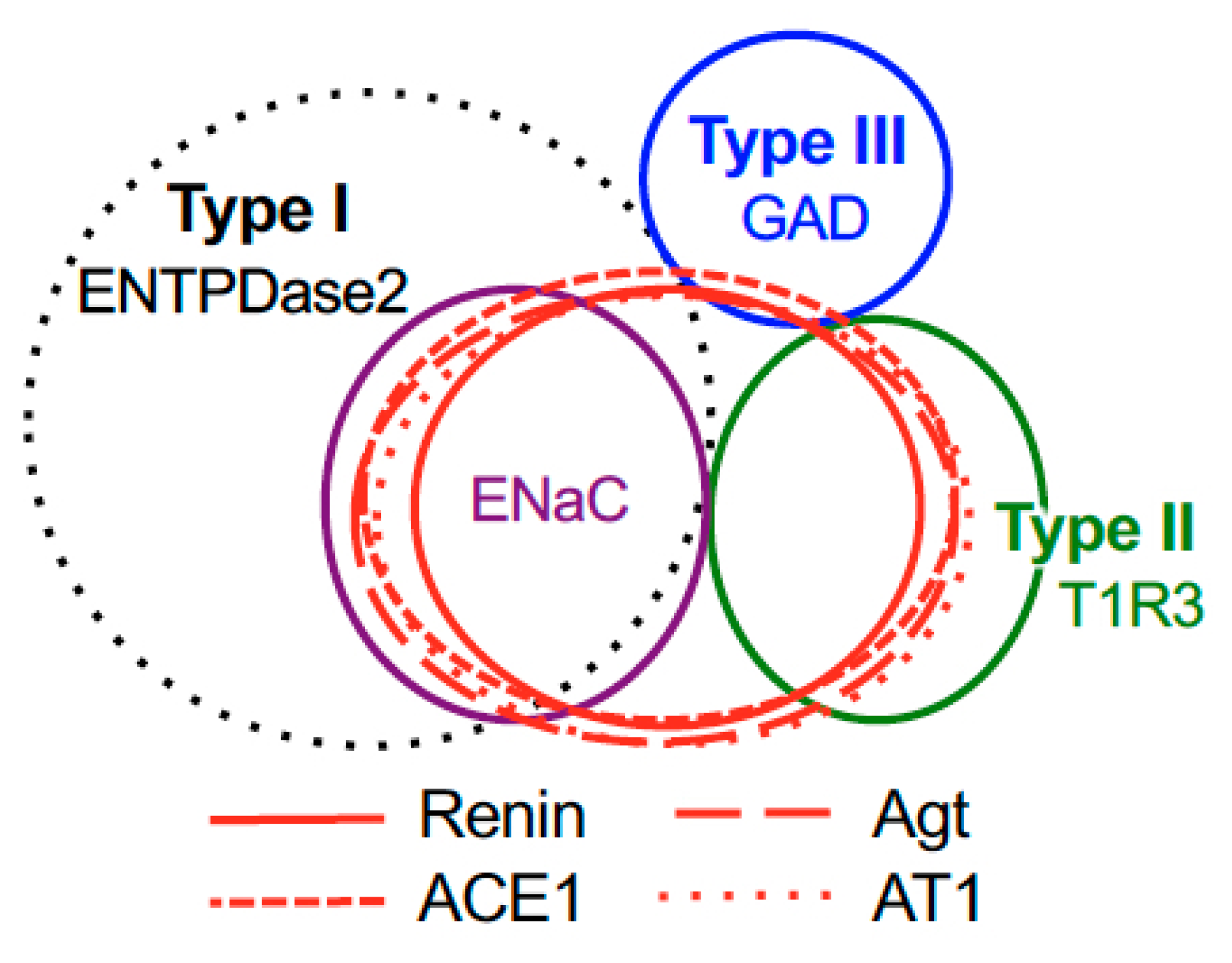
| FP | VP | FP | VP | ||||||
|---|---|---|---|---|---|---|---|---|---|
| T1R3/Renin | 54.7% | (29/53, n = 12) | 49.2% | (31/63, n = 17) | Renin/T1R3 | 39.7% | (29/73, n = 12) | 36.0% | (31/86, n = 17) |
| GAD/Renin | 0% | (0/33, n = 8) | 0% | (0/42, n = 11) | Renin/GAD | 0% | (0/13, n = 8) | 0% | (0/33, n = 11) |
| ENaC/Renin | 83.9% | (26/31, n = 10) | 88.1% | (37/42, n = 11) | Renin/ENaC | 92.9% | (26/28, n = 10) | 86.0% | (37/43, n = 11) |
| AT1/Renin | 72.0% | (18/25, n = 9) | 80.0% | (32/40, n = 11) | Renin/AT1 | 72.0% | (18/25, n = 9) | 91.4% | (32/35, n = 11) |
| FP | VP | FP | VP | ||||||
|---|---|---|---|---|---|---|---|---|---|
| T1R3/ACE | 70.0% | (35/50, n = 11) | 57.5% | (42/73, n = 20) | ACE/T1R3 | 58.3% | (35/60, n = 11) | 46.2% | (42/91, n = 20) |
| GAD/ACE | 0% | (0/38, n = 11) | 2.7% | (2/75, n = 16) | ACE/GAD | 0% | (0/15, n = 11) | 4.3% | (2/47, n = 16) |
| ENaC/ACE | 71.9% | (23/32, n = 7) | 91.3% | (42/46, n = 10) | ACE/ENaC | 85.2% | (23/27, n = 7) | 93.3% | (42/45, n = 10) |
| AT1/ACE | 81.3% | (26/32, n = 7) | 79.7% | (63/79, n = 13) | ACE/AT1 | 96.3% | (26/27, n = 7) | 98.4% | (63/64, n = 13) |
| FP | VP | FP | VP | ||||||
|---|---|---|---|---|---|---|---|---|---|
| T1R3/Agt | 73.0% | (27/37, n = 7) | 70.7% | (41/58, n = 14) | Agt/T1R3 | 60.0% | (27/45, n = 7) | 37.6% | (41/109, n = 14) |
| GAD/Agt | 0% | (0/10, n = 4) | 0% | (0/56, n = 17) | Agt/GAD | 0% | (0/8, n = 4) | 0% | (0/68, n = 17) |
| ENTPD2/Agt | 37.5% | (9/24, n = 9) | 34.1% | (15/44, n = 14) | Agt/ENTPD2 | 8.7% | (9/103, n = 9) | 7.9% | (15/189, n = 14) |
| Renin/Agt | 75.8% | (25/33, n = 9) | 70.6% | (24/34, n = 9) | Agt/Renin | 92.6% | (25/27, n = 9) | 85.7% | (24/28, n = 9) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shigemura, N.; Takai, S.; Hirose, F.; Yoshida, R.; Sanematsu, K.; Ninomiya, Y. Expression of Renin-Angiotensin System Components in the Taste Organ of Mice. Nutrients 2019, 11, 2251. https://doi.org/10.3390/nu11092251
Shigemura N, Takai S, Hirose F, Yoshida R, Sanematsu K, Ninomiya Y. Expression of Renin-Angiotensin System Components in the Taste Organ of Mice. Nutrients. 2019; 11(9):2251. https://doi.org/10.3390/nu11092251
Chicago/Turabian StyleShigemura, Noriatsu, Shingo Takai, Fumie Hirose, Ryusuke Yoshida, Keisuke Sanematsu, and Yuzo Ninomiya. 2019. "Expression of Renin-Angiotensin System Components in the Taste Organ of Mice" Nutrients 11, no. 9: 2251. https://doi.org/10.3390/nu11092251
APA StyleShigemura, N., Takai, S., Hirose, F., Yoshida, R., Sanematsu, K., & Ninomiya, Y. (2019). Expression of Renin-Angiotensin System Components in the Taste Organ of Mice. Nutrients, 11(9), 2251. https://doi.org/10.3390/nu11092251





