Plant Bioactives and the Prevention of Prostate Cancer: Evidence from Human Studies
Abstract
1. Introduction
2. Sulfur-Containing Bioactives from Cruciferous and Alliaceous Vegetables and Prostate Cancer
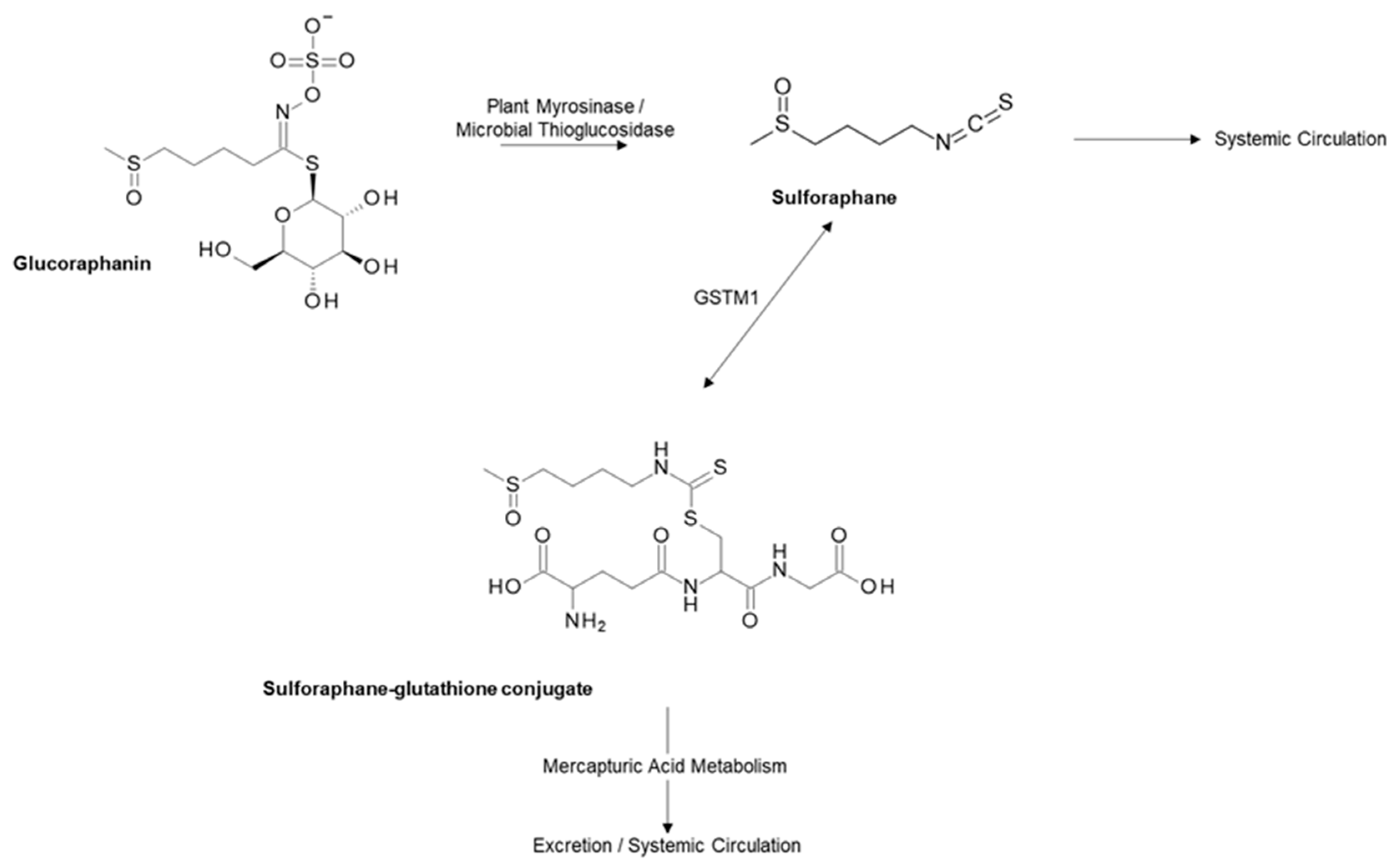
2.1. Glucoraphanin and Sulforaphane from Cruciferous Vegetables
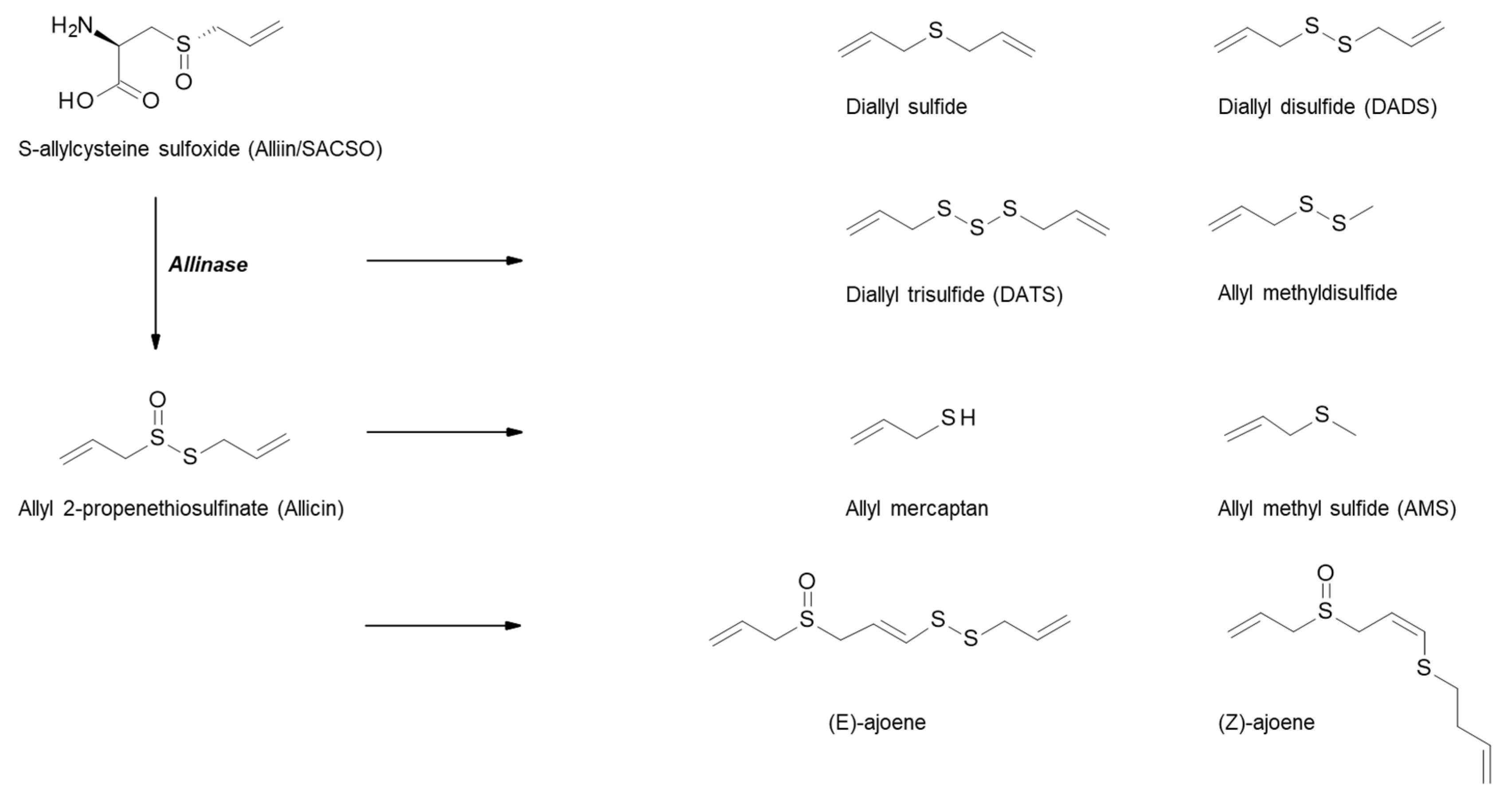
2.2. S-Alk(en)yl-L-Cysteine Sulfoxides (SACSOs) from Alliaceous Vegetables
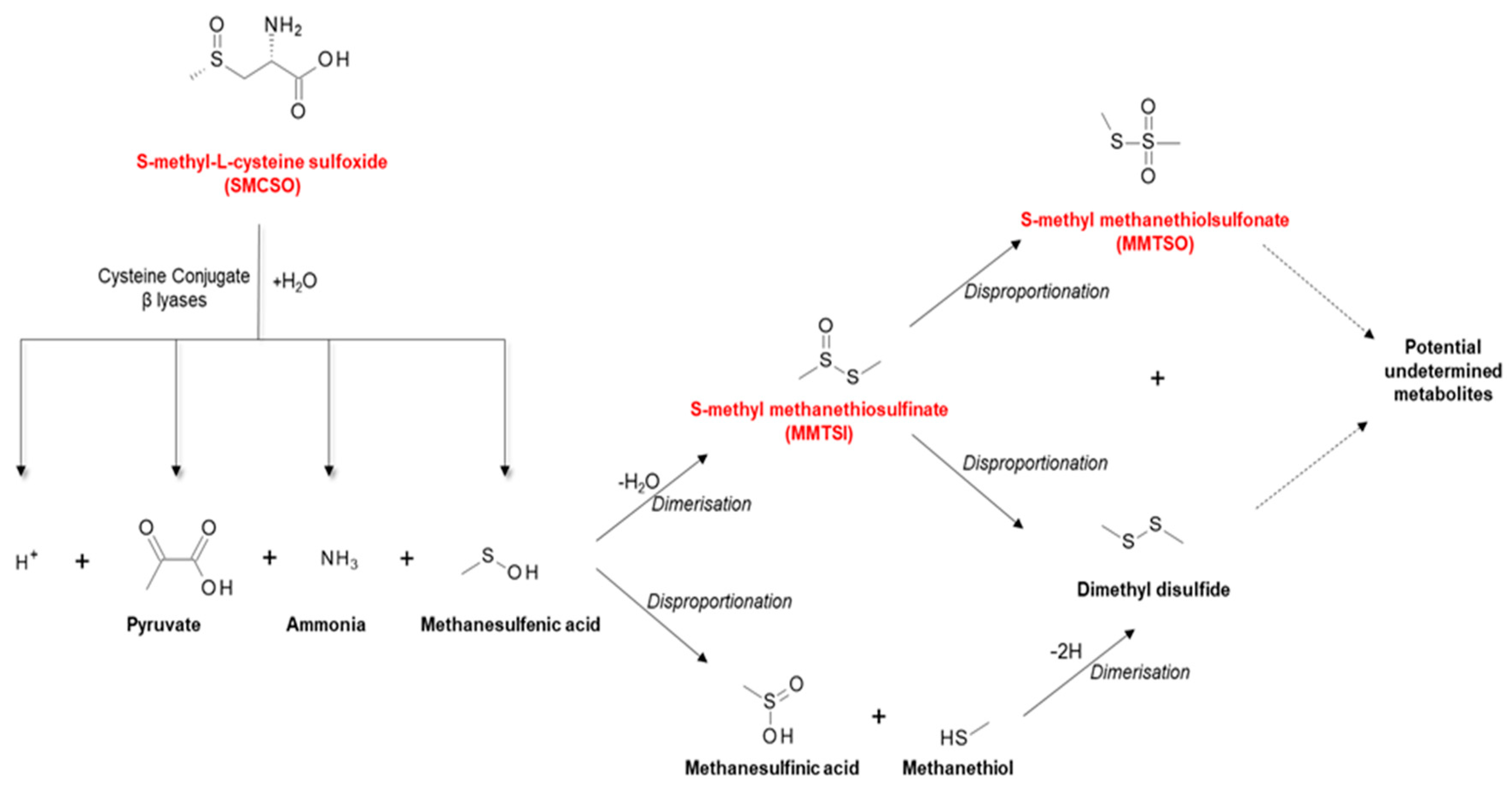
2.3. S-Methyl-L-cysteine Sulfoxide (SMCSO) from Cruciferous and Alliaceous Vegetables
3. Carotenoids and Prostate Cancer
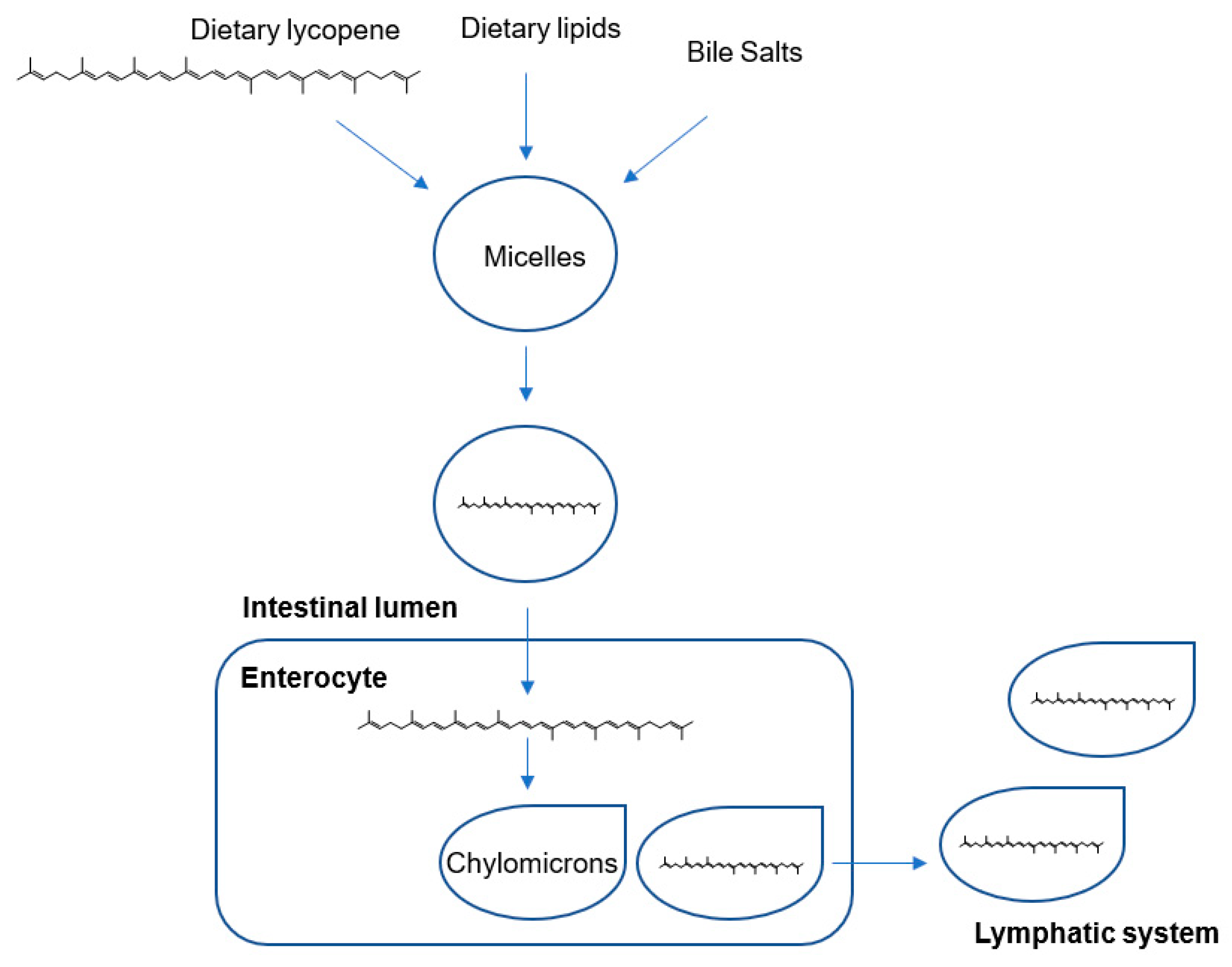
Lycopene from Tomatoes
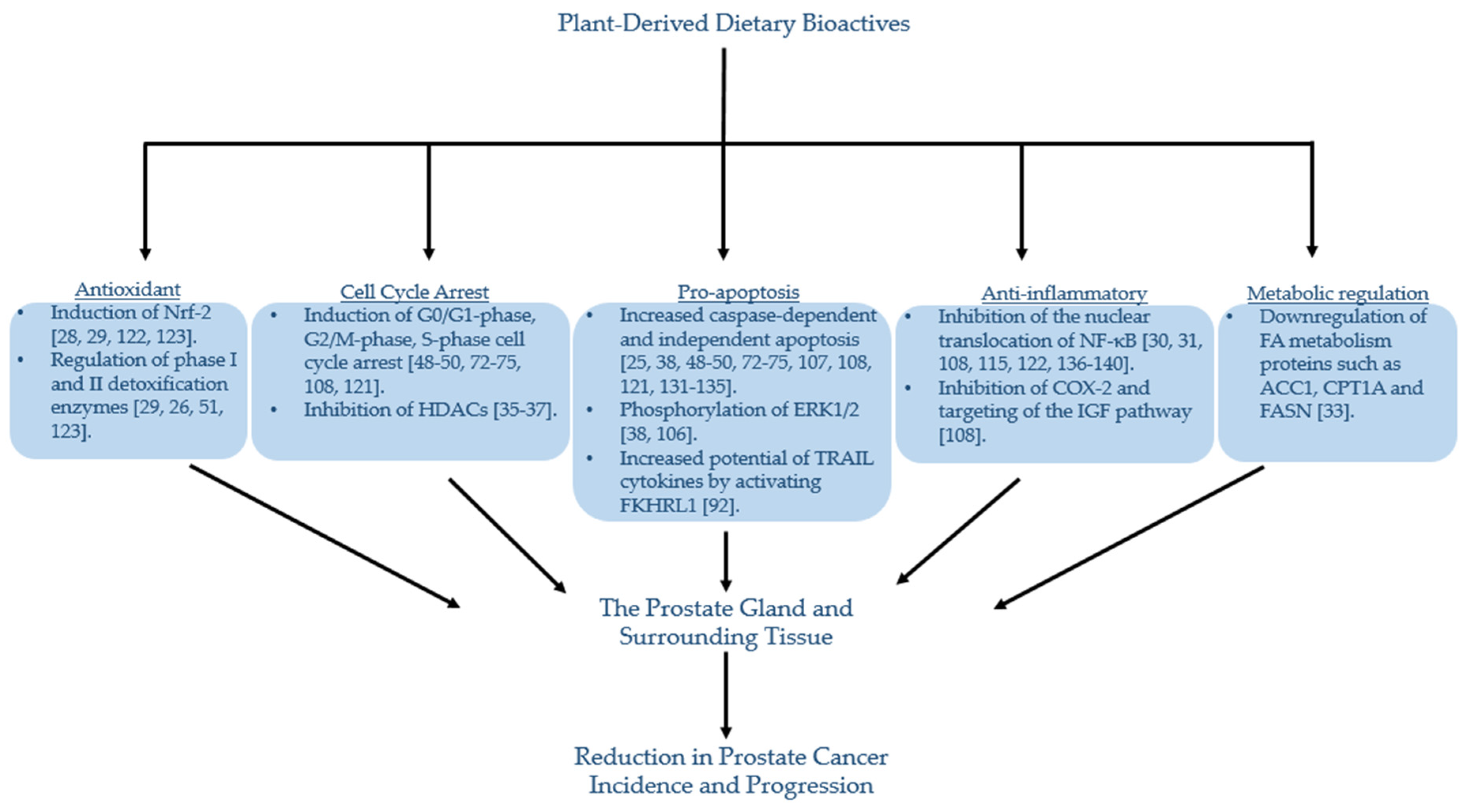
4. Polyphenols and Prostate Cancer
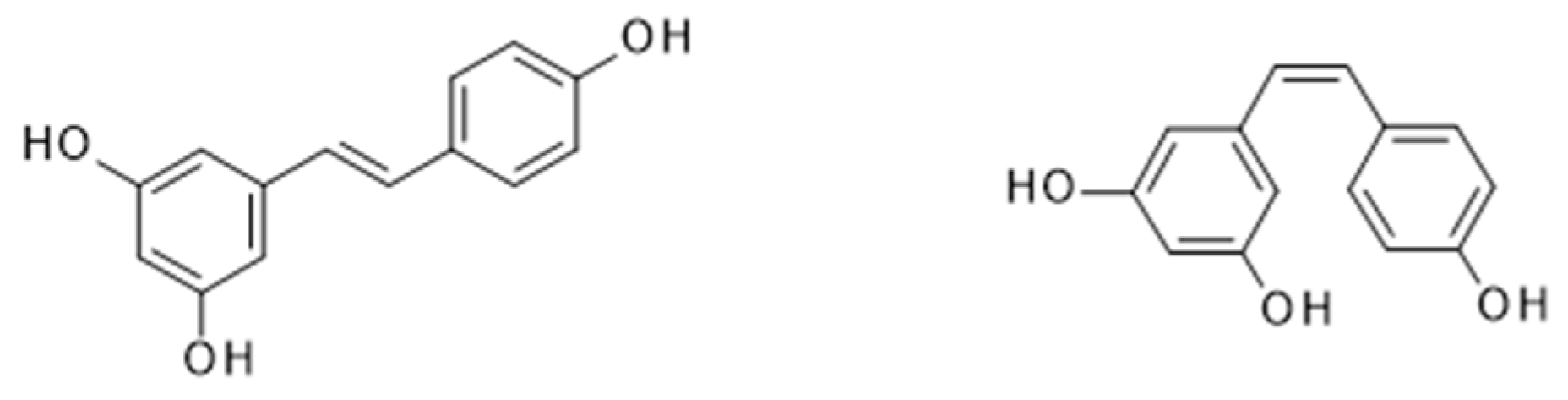
4.1. Resveratrol from Wine
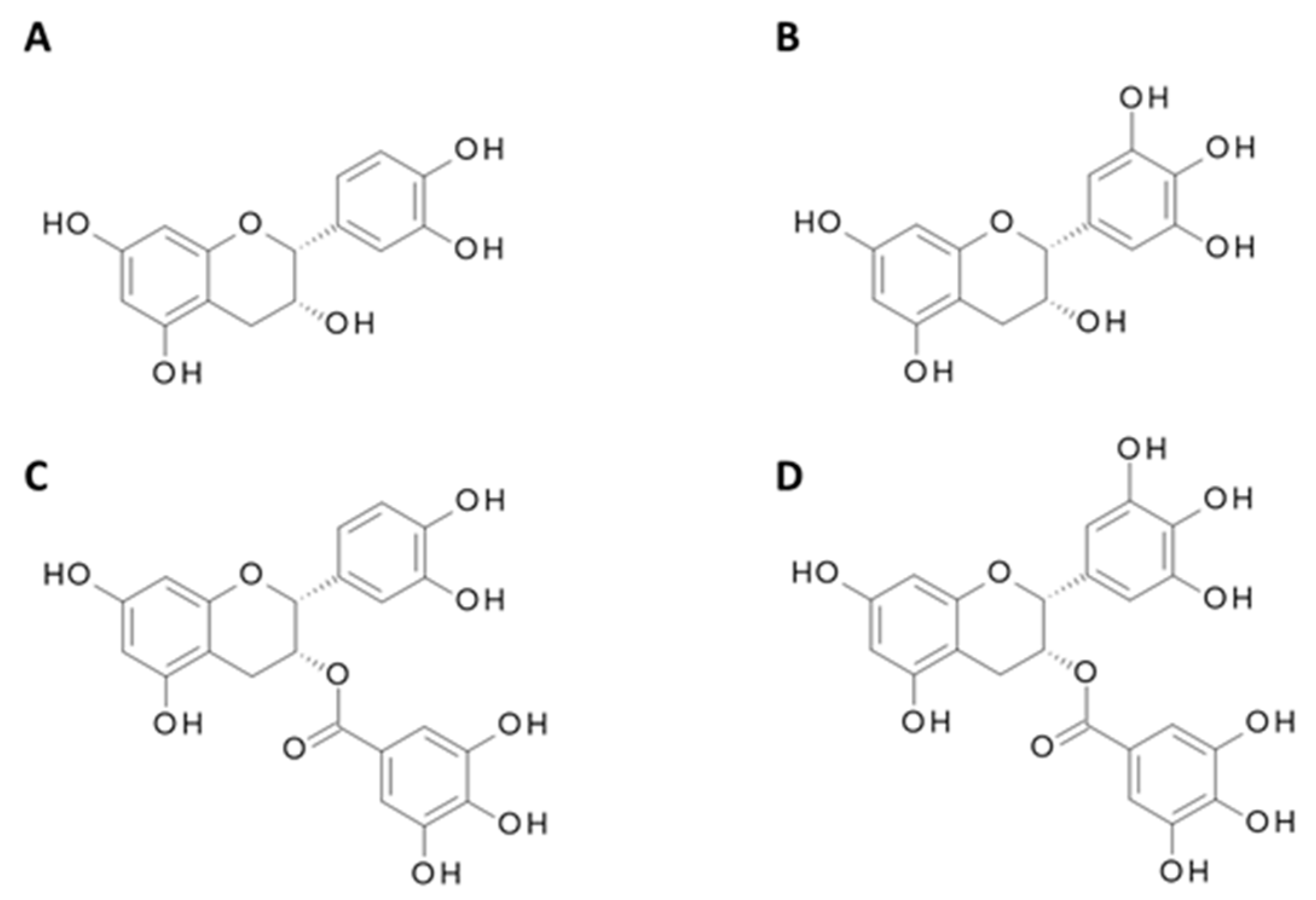
4.2. Catechins from Green Tea
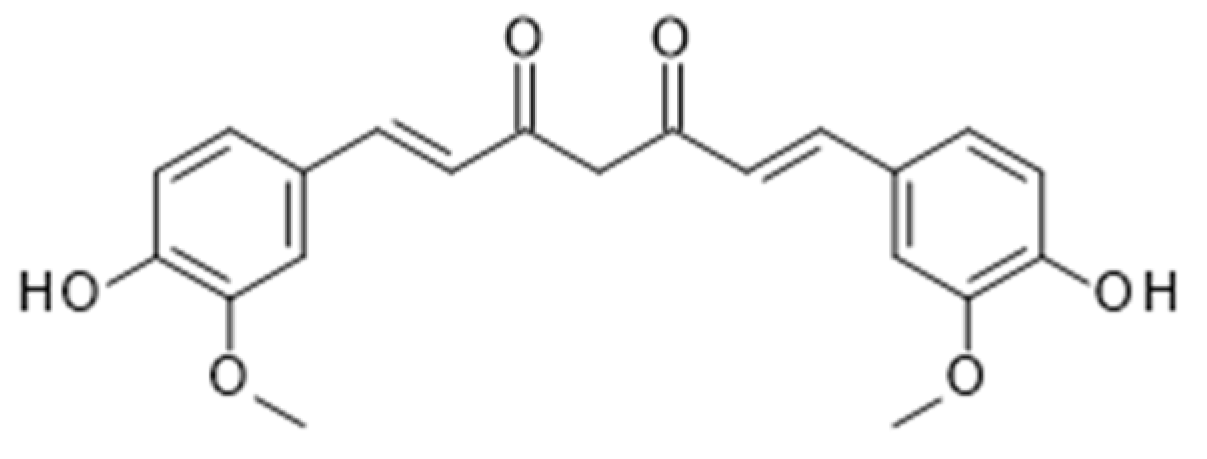
4.3. Curcumin from Turmeric
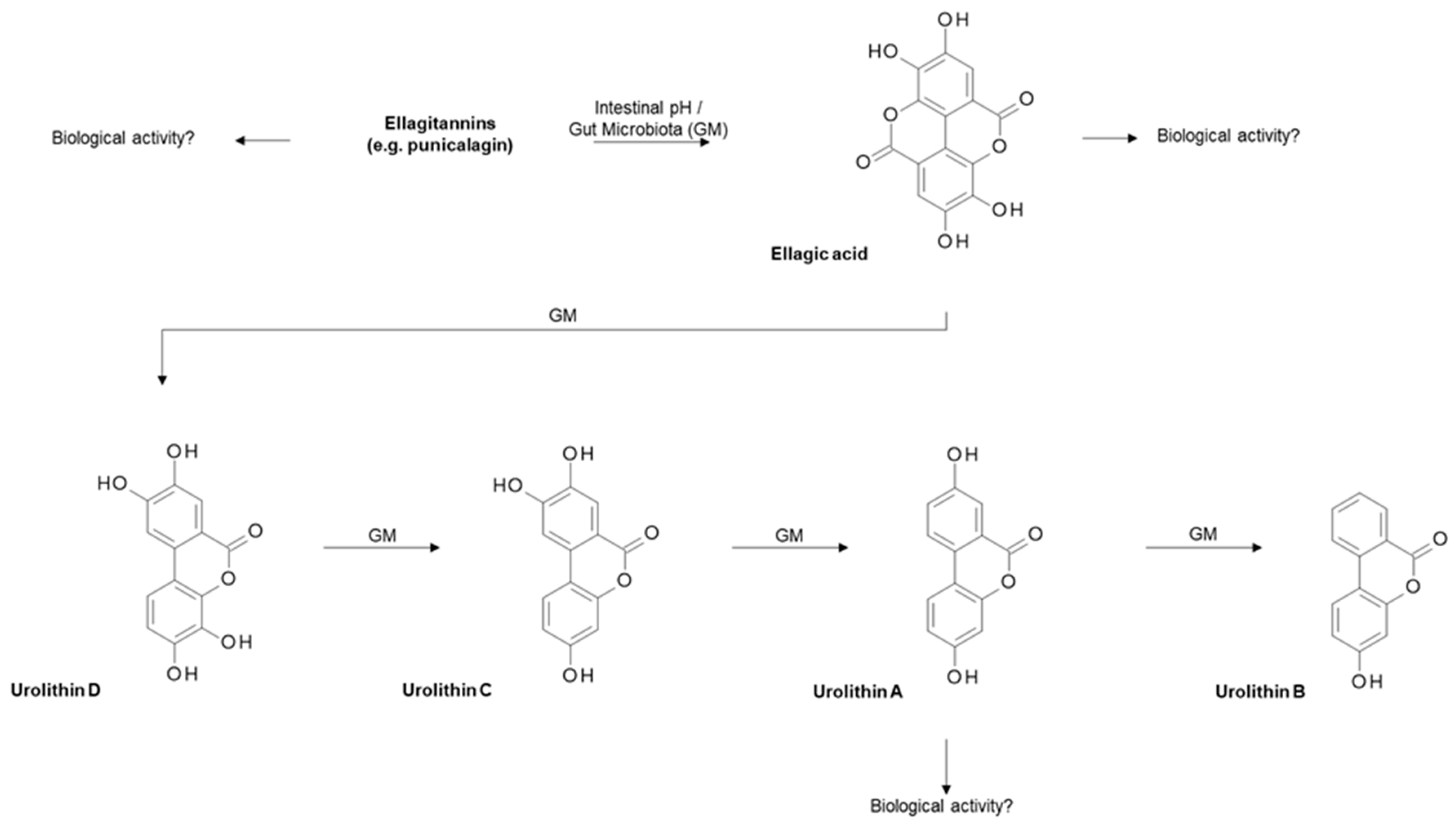
4.4. Ellagitannins from Pomegranate
5. Exposure of Dietary Bioactives to the Prostate
6. Future Perspectives
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ACC1 | acetyl-CoA carboxylase 1 |
| ADT | androgen deprivation treatment |
| ASAP | atypical small acinar proliferation |
| AR | androgen receptor |
| BCG | Bacillus Calmette-Guerin |
| Bcl-2 | B-cell lymphoma 2 |
| Bcl-xL | B-cell lymphoma extra-large |
| COX-2 | cyclooxygenase-2 |
| CBP | cAMP response element-binding protein |
| CPT1A | carnitine palmitoyltransferase 1A |
| CVD | cardiovascular disease |
| CXCR4 | chemokine receptor type 4 |
| DAS | diallyl sulfide |
| DADS | diallyl disulfide |
| DATS | diallyl trisulfide |
| DHEA | dehydroepiandrosterone |
| eBASIS | Bioactive Substances in Food and Information System |
| EC | epicatechin |
| ECG | epicatechin-3-gallate |
| EGC | epigallocatechin |
| EGCG | epigallocatechin-3-gallate ERK1/2: extracellular signal-regulated protein kinase 1 and 2 |
| FA | fatty acid |
| FASN | fatty acid synthase |
| FOXO | forkhead family of transcription factors |
| GRN | glucoraphanin |
| GM | gut microbiota |
| GSL | glucosinolate |
| GST | glutathione-S-transferase |
| GSTM1 | Glutathione S-Transferase Mu 1 |
| HDAC | histone deacetylases |
| HGPIN | high-grade prostatic intraepithelial neoplasia |
| HLH | helix-loop-helix |
| IAD | intermittent androgen deprivation |
| IARC | International Agency for Research on Cancer |
| Id1 | inhibitor of DNA binding 1 |
| IGF | insulin-like growth factor |
| Keap1 | kelch-like ECH-associated protein |
| ITC | isothiocyanate; MDM2: mouse double minute 2 homolog |
| MEK | mitogen-activated protein kinase kinase |
| MMPs | matrix metalloproteinases |
| MMTSI | S-methyl methanethiosulfinate |
| MMTSO | S-methyl methanethiolsulfinate |
| MTA1 | metastasis-associated protein 1 |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| NF-κB | nuclear factor-kappa B |
| Nrf-2 | nuclear factor erythroid 2-related factor 2 |
| OSC | organosulfur compounds |
| PCa | prostate cancer |
| PI3K | phosphoinositide 3-kinase |
| PKB/Akt | protein kinase B |
| PSA | prostate specific antigen |
| RCT | randomised controlled trial |
| ROS | reactive oxygen species |
| SACSO | S-alk(en)yl-L-cysteine sulfoxide |
| SMCSO | S-methyl-L- cysteine sulfoxide |
| SCMCSO | S-carboxymethyl-L-cysteine sulfoxide; SFN: sulforaphane |
| SFN-Cys | sulforaphane-cysteine |
| SFN-NAC | sulforaphane-N-acetyl-cysteine |
| TRAIL | TNF-related apoptosis inducing ligand |
| TRAMP | transgenic adenocarcinoma of the mouse prostate |
| TPB | template-guided transperineal prostate biopsy |
| VEGF | vascular endothelial growth factor |
| VEGFR2 | vascular endothelial growth factor receptor 2 |
References
- Skerrett, P.J.; Willett, W.C. Essentials of healthy eating: A guide. J. Midwifery Womens Health 2010, 55, 492–501. [Google Scholar] [CrossRef] [PubMed]
- Nagai, H.; Kim, Y.H. Cancer prevention from the perspective of global cancer burden patterns. J. Thorac. Dis. 2017, 9, 448–451. [Google Scholar] [CrossRef] [PubMed]
- Aghajanpour, M.; Nazer, M.R.; Obeidavi, Z.; Akbari, M.; Ezati, P.; Kor, N.M. Functional foods and their role in cancer prevention and health promotion: A comprehensive review. Am. J. Cancer Res. 2017, 7, 740–769. [Google Scholar]
- World Health Organisation (WHO), International Agency for Research on Cancer. Available online: https://www.iarc.fr/ (accessed on 6 September 2018).
- Cancer Research UK. Available online: http://www.cancerresearchuk.org/healthprofessional/cancer-statistics/statistics-by-cancer-type/prostate-cancer#heading-Zero (accessed on 14 May 2019).
- Reynard, J.; Brewster, S.; Biers, S. Oxford Handbook of Urology, 3rd ed.; Oxford University Press: Oxford, UK, 2013. [Google Scholar]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, 359–386. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Colli, J.L.; Colli, A. International comparisons of prostate cancer mortality rates with dietary practices and sunlight levels. Urol. Oncol. 2006, 24, 184–194. [Google Scholar] [CrossRef] [PubMed]
- Hsing, A.W.; Tsao, L.; Devesa, S.S. International trends and patterns of prostate cancer incidence and mortality. Int. J. Cancer 2000, 85, 60–67. [Google Scholar] [CrossRef]
- Ambrosini, G.L.; Fritschi, L.; de Klerk, N.H.; Mackerras, D.; Leavy, J. Dietary patterns identified using factor analysis and prostate cancer risk: A case control study in Western Australia. Ann. Epidemiol. 2008, 18, 364–370. [Google Scholar] [CrossRef] [PubMed]
- Wolk, A. Diet, lifestyle and risk of prostate cancer. Acta Oncol. (Stockh. Swed.) 2005, 44, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Deneo-Pellegrini, H.; De Stefani, E.; Ronco, A.; Mendilaharsu, M. Foods, nutrients and prostate cancer: A case-control study in Uruguay. Br. J. Cancer 1999, 80, 591–597. [Google Scholar] [CrossRef] [PubMed]
- Augustsson, K.; Michaud, D.S.; Rimm, E.B.; Leitzmann, M.F.; Stampfer, M.J.; Willett, W.C.; Giovannucci, E. A prospective study of intake of fish and marine fatty acids and prostate cancer. Cancer Epidemiol. Biomark. Prev. A Publ. Am. Assoc. Cancer Res. Cosponsored Am. Soc. Prev. Oncol. 2003, 12, 64–67. [Google Scholar]
- Capurso, C.; Vendemiale, G. The Mediterranean Diet Reduces the Risk and Mortality of the Prostate Cancer: A Narrative Review. Front. Nutr. 2017, 4. [Google Scholar] [CrossRef] [PubMed]
- Castello, A.; Boldo, E.; Amiano, P.; Castano-Vinyals, G.; Aragones, N.; Gomez-Acebo, I.; Peiro, R.; Jimenez-Moleon, J.J.; Alguacil, J.; Tardon, A.; et al. Mediterranean Dietary Pattern is Associated with Low Risk of Aggressive Prostate Cancer: MCC-Spain Study. J. Urol. 2018, 199, 430–437. [Google Scholar] [CrossRef] [PubMed]
- Watson, W.; Beaver, G.M.; Williams, L.E.; Dashwood, D.H.; Ho, R.E. Phytochemicals from cruciferous vegetables, epigenetics, and prostate cancer prevention. AAPS J. 2013, 15, 951–961. [Google Scholar] [CrossRef] [PubMed]
- Pratheeshkumar, P.; Son, Y.O.; Korangath, P.; Manu, K.A.; Siveen, K.S. Phytochemicals in Cancer Prevention and Therapy. BioMed Res. Int. 2015, 2015, 2. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Fokou, P.V.T.; Yamthe, L.R.T.; Tali, B.T.; Adetunji, C.O.; Rahavian, A.; Mudau, F.N.; Martorell, M.; Setzer, W.N.; Rodrigues, C.F.; et al. Phytochemicals in Prostate Cancer: From Bioactive Molecules to Upcoming Therapeutic Agents. Nutrients 2019, 11, 1483. [Google Scholar] [CrossRef] [PubMed]
- Iqbal, J.; Abbasi, B.A.; Mahmood, T.; Kanwal, S.; Ali, B.; Shah, S.A.; Khalil, A.T. Plant-derived anticancer agents: A green anticancer approach. Asian Pac. J. Trop. Biomed. 2017, 7, 1129–1150. [Google Scholar] [CrossRef]
- Thomas, R.; Williams, M.; Sharma, H.; Chaudry, A.; Bellamy, P. A double-blind, placebo-controlled randomised trial evaluating the effect of a polyphenol-rich whole food supplement on PSA progression in men with prostate cancer—The UK NCRN Pomi-T study. Prostate Cancer Prostatic Dis. 2014, 17, 180. [Google Scholar] [CrossRef]
- Cooper, C.S.; Eeles, R.; Wedge, D.C.; Van Loo, P.; Gundem, G.; Alexandrov, L.B.; Kremeyer, B.; Butler, A.; Lynch, A.G.; Camacho, N.; et al. Analysis of the genetic phylogeny of multifocal prostate cancer identifies multiple independent clonal expansions in neoplastic and morphologically normal prostate tissue. Nat. Genet. 2015, 47, 367–372. [Google Scholar] [CrossRef]
- Traka, M.H.; Melchini, A.; Coode-Bate, J.; Al Kadhi, O.; Saha, S.; Defernez, M.; Troncoso-Rey, P.; Kibblewhite, H.; O’Neill, C.M.; Bernuzzi, F.; et al. Transcriptional changes in prostate of men on active surveillance after a 12-mo glucoraphanin-rich broccoli intervention-results from the Effect of Sulforaphane on prostate CAncer PrEvention (ESCAPE) randomized controlled trial. Am. J. Clin. Nutr. 2019, 109, 1133–1144. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, T.A.; Fahey, J.W.; Wade, K.L.; Stephenson, K.K.; Talalay, P. Chemoprotective Glucosinolates and Isothiocyanates of Broccoli Sprouts. Metabo. Excretion Humans 2001, 10, 501–508. [Google Scholar]
- Traka, M. Health Benefits of Glucosinolates; Advances in Botanical Research; Elsevier Ltd.: Amsterdam, The Netherlands, 2016; Volume 80. [Google Scholar] [CrossRef]
- Traka, M.; Mithen, R. Glucosinolates, isothiocyanates and human health. Phytochem. Rev. 2009, 8, 269–282. [Google Scholar] [CrossRef]
- Dinkova-Kostova, A. Chemoprotection Against Cancer by Isothiocyanates: A Focus on the Animal Models and the Protective Mechanisms. Top Curr Chem. 2013, 329, 179–201. [Google Scholar] [CrossRef] [PubMed]
- Sturm, C.; Wagner, A.E. Brassica-Derived Plant Bioactives as Modulators of Chemopreventive and Inflammatory Signaling Pathways. Int. J. Mol. Sci. 2017, 18, 1890. [Google Scholar] [CrossRef]
- Jaramillo, M.C.; Zhang, D.D. The emerging role of the Nrf2-Keap1 signaling pathway in cancer. Genes Dev. 2013, 27, 2179–2191. [Google Scholar] [CrossRef]
- Soundararajan, P.; Kim, J.S. Anti-Carcinogenic Glucosinolates in Cruciferous Vegetables and Their Antagonistic Effects on Prevention of Cancers. Mol. (Basel Switz.) 2018, 23, 2983. [Google Scholar] [CrossRef]
- Heiss, E.; Herhaus, C.; Klimo, K.; Bartsch, H.; Gerhauser, C. Nuclear factor kappa B is a molecular target for sulforaphane-mediated anti-inflammatory mechanisms. J. Biol. Chem. 2001, 276, 32008–32015. [Google Scholar] [CrossRef]
- Singh, S.V.; Warin, R.; Xiao, D.; Powolny, A.A.; Stan, S.D.; Arlotti, J.A.; Zeng, Y.; Hahm, E.R.; Marynowski, S.W.; Bommareddy, A.; et al. Sulforaphane inhibits prostate carcinogenesis and pulmonary metastasis in TRAMP mice in association with increased cytotoxicity of natural killer cells. Cancer Res. 2009, 69, 2117–2125. [Google Scholar] [CrossRef]
- Singh, K.B.; Kim, S.H.; Hahm, E.R.; Pore, S.K.; Jacobs, B.L.; Singh, S.V. Prostate cancer chemoprevention by sulforaphane in a preclinical mouse model is associated with inhibition of fatty acid metabolism. Carcinogenesis 2018, 39, 826–837. [Google Scholar] [CrossRef]
- Ho, E.; Clarke, J.D.; Dashwood, R.H. Dietary sulforaphane, a histone deacetylase inhibitor for cancer prevention. J. Nutr. 2009, 139, 2393–2396. [Google Scholar] [CrossRef]
- Ferreira, P.M.P.; Rodrigues, L.; de Alencar Carnib, L.P.; de Lima Sousa, P.V.; Nolasco Lugo, L.M.; Nunes, N.M.F.; do Nascimento Silva, J.; da Silva Araujo, L.; de Macedo Goncalves Frota, K. Cruciferous Vegetables as Antioxidative, Chemopreventive and Antineoplasic Functional Foods: Preclinical and Clinical Evidences of Sulforaphane Against Prostate Cancers. Curr. Pharm. Des. 2018, 24, 4779–4793. [Google Scholar] [CrossRef]
- Gibbs, A.; Schwartzman, J.; Deng, V.; Alumkal, J. Sulforaphane destabilizes the androgen receptor in prostate cancer cells by inactivating histone deacetylase 6. Proc. Natl. Acad. Sci. USA 2009, 106, 16663–16668. [Google Scholar] [CrossRef]
- Beaver, L.M.; Lhr, C.V.; Clarke, J.D.; Glasser, S.T.; Watson, G.W.; Wong, C.P.; Zhang, Z.; Williams, D.E.; Dashwood, R.H.; Shannon, J.; et al. Broccoli Sprouts Delay Prostate Cancer Formation and Decrease Prostate Cancer Severity with a Concurrent Decrease in HDAC3 Protein Expression in Transgenic Adenocarcinoma of the Mouse Prostate (TRAMP) Mice. Curr. Dev. Nutr. 2018, 2, nzy002. [Google Scholar] [CrossRef]
- Zhou, Y.; Yang, G.; Tian, H.; Hu, Y.; Wu, S.; Geng, Y.; Lin, K.; Wu, W. Sulforaphane metabolites cause apoptosis via microtubule disruption in cancer. Endocr. Relat. Cancer 2018, 25, 255–268. [Google Scholar] [CrossRef]
- Liu, B.; Mao, Q.; Cao, M.; Xie, L. Cruciferous vegetables intake and risk of prostate cancer: A meta-analysis. Int. J. Urol. Off. J. Jpn. Urol. Assoc. 2012, 19, 134–141. [Google Scholar] [CrossRef]
- Richman, E.L.; Carroll, P.R.; Chan, J.M. Vegetable and fruit intake after diagnosis and risk of prostate cancer progression. Int. J. Cancer 2012, 131, 201–210. [Google Scholar] [CrossRef]
- Cohen, J.H.; Kristal, A.R.; Stanford, J.L. Fruit and vegetable intakes and prostate cancer risk. J. Natl. Cancer Inst. 2000, 92, 61–68. [Google Scholar] [CrossRef]
- Jain, M.G.; Hislop, G.T.; Howe, G.R.; Ghadirian, P. Plant foods, antioxidants, and prostate cancer risk: Findings from case-control studies in Canada. Nutr. Cancer 1999, 34, 173–184. [Google Scholar] [CrossRef]
- Kirsh, V.A.; Peters, U.; Mayne, S.T.; Subar, A.F.; Chatterjee, N.; Johnson, C.C.; Hayes, R.B. Prospective study of fruit and vegetable intake and risk of prostate cancer. J. Natl. Cancer Inst. 2007, 99, 1200–1209. [Google Scholar] [CrossRef]
- Zhang, Z.; Garzotto, M.; Davis, E.W., 2nd; Mori, M.; Stoller, W.A.; Farris, P.E.; Wong, C.P.; Beaver, L.M.; Thomas, G.V.; Williams, D.E.; et al. Sulforaphane Bioavailability and Chemopreventive Activity in Men Presenting for Biopsy of the Prostate Gland: A Randomized Controlled Trial. Nutr. Cancer 2019. [Google Scholar] [CrossRef]
- Traka, M.H.; Saha, S.; Huseby, S.; Kopriva, S.; Walley, P.G.; Barker, G.C.; Moore, J.; Mero, G.; van den Bosch, F.; Constant, H.; et al. Genetic regulation of glucoraphanin accumulation in Beneforte broccoli. New Phytol. 2013, 198, 1085–1095. [Google Scholar] [CrossRef]
- Lawson, L.D.; Wang, Z.J. Allicin and Allicin-Derived Garlic Compounds Increase Breath Acetone through Allyl Methyl Sulfide: Use in Measuring Allicin Bioavailability. J. Agric. Food Chem. 2005, 53, 1974–1983. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Arunkumar, A.; Vijayababu, M.R.; Kanagaraj, P.; Balasubramanian, K.; Aruldhas, M.M.; Arunakaran, J. Growth suppressing effect of garlic compound diallyl disulfide on prostate cancer cell line (PC-3) in vitro. Biol. Pharm. Bull. 2005, 28, 740–743. [Google Scholar] [CrossRef]
- Herman-Antosiewicz, A.; Stan, S.D.; Hahm, E.R.; Xiao, D.; Singh, S.V. Activation of a novel ataxia-telangiectasia mutated and Rad3 related/checkpoint kinase 1-dependent prometaphase checkpoint in cancer cells by diallyl trisulfide, a promising cancer chemopreventive constituent of processed garlic. Mol.Cancer Ther. 2007, 6, 1249–1261. [Google Scholar] [CrossRef]
- Shukla, Y.; Kalra, N. Cancer chemoprevention with garlic and its constituents. Cancer Lett. 2007, 247, 167–181. [Google Scholar] [CrossRef]
- Wattenberg, L.W. Inhibition of Carcinogenesis by minor Anutrient Constituents of the Diet. Proc. Nutr. Soc. 1990, 49, 173–183. [Google Scholar] [CrossRef]
- Pinto, J.; Qiao, C.; Xing, J.P.; Suffoletto, B.B.; Schubert, K.S.; Rivlin, R.F.; Huryk, R.; Bacich, D.; Heston, W. Alterations of Prostate Biomarker Expression and Testosterone Utilization in Human LNCaP Prostatic Carcinoma Cells by Garlic-Derived S-Allylmercaptocysteine. Prostate 2000, 45, 304–314. [Google Scholar] [CrossRef]
- Hsing, A.W.; Chokkalingam, A.P.; Gao, Y.T.; Madigan, M.P.; Deng, J.; Gridley, G.; Fraumeni, J.F., Jr. Allium Vegetables and Risk of Prostate Cancer: A Population-Based Study. JNCI J. Natl. Cancer Inst. 2002, 94, 1648–1651. [Google Scholar] [CrossRef]
- Zhou, X.F.; Ding, Z.S.; Liu, N.B. Allium vegetables and risk of prostate cancer: Evidence from 132,192 subjects. Asian Pac. J. Cancer Prev APJCP 2013, 14, 4131–4134. [Google Scholar] [CrossRef]
- Setiawan, V.W.; Yu, G.P.; Lu, Q.Y.; Lu, M.L.; Yu, S.Z.; Mu, L.; Zhang, J.G.; Kurtz, R.C.; Cai, L.; Hsieh, C.C.; et al. Allium vegetables and stomach cancer risk in China. Asian Pac.J. Cancer Prev. APJCP 2005, 6, 387–395. [Google Scholar]
- Edmands, W.M.B.; Gooderham, N.J.; Holmes, E.; Mitchell, S.C. S-Methyl-l-cysteine sulphoxide: The Cinderella phytochemical? Toxicol. Res. 2013, 2, 11–22. [Google Scholar] [CrossRef]
- Coode-Bate, J.S.T.; Melchini, A.; Saha, S.; Needs, P.; Dainty, J.R.; Maicha, J.B.; Beasy, G.; Traka, G.H.; Mills, R.D.; Ball, R.Y.; et al. Accumulation of Dietary S-Methyl Cysteine Sulfoxide in Human Prostate Tissue; WILEY-VCH Verlag GmbH Co. KGaA: Weinheim, Germany, 2019. [Google Scholar] [CrossRef]
- Waring, R.H.; Harris, R.M.; Steventon, G.B.; Mitchell, S.C. Degradation to sulphate of S-methyl-L-cysteine sulphoxide and S-carboxymethyl-L-cysteine sulphoxide in man. Drug Metab. Drug Interact. 2003, 19, 241–255. [Google Scholar] [CrossRef]
- Mangels, A.R.; Holden, J.M.; Beecher, G.R.; Forman, M.R.; Lanza, E. Carotenoid content of fruits and vegetables: An evaluation of analytic data. J. Am. Diet. Assoc. 1993, 93, 284–296. [Google Scholar] [CrossRef]
- Zechmeister, L. Cis-trans Isomerization and Stereochemistry of Carotenoids and Diphenyl-polyenes. Chem. Rev. 1944, 34, 267–344. [Google Scholar] [CrossRef]
- Clinton, S.K. Lycopene: Chemistry, biology, and implications for human health and disease. Nutr. Rev. 1998, 56, 35–51. [Google Scholar] [CrossRef]
- Holzapfel, N.P.; Holzapfel, B.M.; Champ, S.; Feldthusen, J.; Clements, J.; Hutmacher, D.W. The potential role of lycopene for the prevention and therapy of prostate cancer: From molecular mechanisms to clinical evidence. Int. J. Mol. Sci. 2013, 14, 14620–14646. [Google Scholar] [CrossRef]
- Hart, D.J.; Scott, K.J. Development and evaluation of an HPLC method for the analysis of carotenoids in foods, and the measurement of the carotenoid content of vegetables and fruits commonly consumed in the UK. Food Chem. 1995, 54, 101–111. [Google Scholar] [CrossRef]
- Tonucci, L.H.; Holden, J.M.; Beecher, G.R.; Khachik, F.; Davis, C.S.; Mulokozi, G. Carotenoid Content of Thermally Processed Tomato-Based Food Products. J. Agric. Food Chem. 1995, 43, 579–586. [Google Scholar] [CrossRef]
- Story, E.N.; Kopec, R.E.; Schwartz, S.J.; Harris, G.K. An update on the health effects of tomato lycopene. Annu. Rev. Food Sci. Technol. 2010, 1, 189–210. [Google Scholar] [CrossRef]
- Rao, A.V.; Ray, M.R.; Rao, L.G. Lycopene. Adv. Food Nutr. Res. 2006, 51, 99–164. [Google Scholar] [CrossRef]
- Sanadi, R.M.; Doshi, M.; Ambulgekar, J.R.; Khambatta, X. Lycopene: It’s Role in Health and Disease. Int. J. Pharm. Sci. Res. 2012, 3, 4578–4582. [Google Scholar]
- Moran, N.E.; Erdman, J.W.J.; Clinton, S.K. Complex interactions between dietary and genetic factors impact lycopene metabolism and distribution. Arch. Biochem. Biophys. 2013, 539, 171–180. [Google Scholar] [CrossRef]
- Kaplan, L.A.; Lau, J.M.; Stein, E.A. Carotenoid composition, concentrations, and relationships in various human organs. Clin. Physiol. Biochem. 1990, 8, 1–10. [Google Scholar]
- Durairajanayagam, D.; Agarwal, A.; Ong, C.; Prashast, P. Lycopene and male infertility. Asian J. Androl. 2014, 16, 420–425. [Google Scholar] [CrossRef]
- Agarwal, S.; Rao, A.V. Tomato lycopene and its role in human health and chronic diseases. CMAJ Can. Med. Assoc. J. 2000, 163, 739–744. [Google Scholar]
- Hwang, E.S.; Bowen, P.E. Cell cycle arrest and induction of apoptosis by lycopene in LNCaP human prostate cancer cells. J. Med. Food 2004, 7, 284–289. [Google Scholar] [CrossRef]
- Ivanov, N.I.; Cowell, S.P.; Brown, P.; Rennie, P.S.; Guns, E.S.; Cox, M.E. Lycopene differentially induces quiescence and apoptosis in androgen-responsive and -independent prostate cancer cell lines. Clin. Nutr. (Edinb. Scotl.) 2007, 26, 252–263. [Google Scholar] [CrossRef]
- Tang, L.; Jin, T.; Zeng, X.; Wang, J.S. Lycopene inhibits the growth of human androgen-independent prostate cancer cells in vitro and in BALB/c nude mice. J. Nutr. 2005, 135, 287–290. [Google Scholar] [CrossRef] [PubMed]
- Hantz, H.L.; Young, L.F.; Martin, K.R. Physiologically attainable concentrations of lycopene induce mitochondrial apoptosis in LNCaP human prostate cancer cells. Exp. Biol. Med. (Maywood N.J.) 2005, 230, 171–179. [Google Scholar] [CrossRef] [PubMed]
- Applegate, C.C.; Rowles, J.L., 3rd; Erdman, J.W., Jr. Can Lycopene Impact the Androgen Axis in Prostate Cancer?: A Systematic Review of Cell Culture and Animal Studies. Nutrients 2019, 11, 633. [Google Scholar] [CrossRef] [PubMed]
- Rowles, J.M.; Ranard, K.; Smith, J.; An, R.; Erdman, J.J. Increased dietary and circulating lycopene are associated with reduced prostate cancer risk: A systematic review and meta-analysis. Prostate Cancer Prostatic Dis. 2017, 20, 361. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cui, R.; Xiao, Y.; Fang, J.; Xu, Q. Effect of Carotene and Lycopene on the Risk of Prostate Cancer: A Systematic Review and Dose-Response Meta-Analysis of Observational Studies. PLoS ONE 2015, 10, e0137427. [Google Scholar] [CrossRef]
- Van Hoang, D.; Pham, N.M.; Lee, A.H.; Tran, D.N.; Binns, C.W. Dietary Carotenoid Intakes and Prostate Cancer Risk: A Case-Control Study from Vietnam. Nutrients 2018, 10, 70. [Google Scholar] [CrossRef] [PubMed]
- Key, T.J.; Appleby, P.N.; Travis, R.C.; Albanes, D.; Alberg, A.J.; Barricarte, A.; Black, A.; Boeing, H.; Bueno-de-Mesquita, H.B.; Chan, J.M.; et al. Carotenoids, retinol, tocopherols, and prostate cancer risk: Pooled analysis of 15 studies. Am. J. Clin. Nutr. 2015, 102, 1142–1157. [Google Scholar] [CrossRef] [PubMed]
- Weaver, C.M.; Miller, J.W. Challenges in conducting clinical nutrition research. Nutr. Rev. 2017, 75, 491–499. [Google Scholar] [CrossRef]
- Giovannucci, E.; Rimm, E.B.; Liu, Y.; Stampfer, M.J.; Willett, W.C. A prospective study of tomato products, lycopene, and prostate cancer risk. J. Natl. Cancer Inst. 2002, 94, 391–398. [Google Scholar] [CrossRef]
- Kim, H.S.; Bowen, P.; Chen, L.; Duncan, C.; Ghosh, L.; Sharifi, R.; Christov, K. Effects of tomato sauce consumption on apoptotic cell death in prostate benign hyperplasia and carcinoma. Nutr. Cancer 2003, 47, 40–47. [Google Scholar] [CrossRef]
- Morgia, G.; Voce, S.; Palmieri, F.; Gentile, M.; Iapicca, G.; Giannantoni, A.; Blefari, F.; Carini, M.; Vespasiani, G.; Santelli, G.; et al. Association between selenium and lycopene supplementation and incidence of prostate cancer: Results from the post-hoc analysis of the procomb trial. Phytomed. Int. J. Phytother. Phytopharmacol. 2017, 34, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Dybkowska, E.; Sadowska, A.; Swiderski, F.; Rakowska, R.; Wysocka, K. The occurrence of resveratrol in foodstuffs and its potential for supporting cancer prevention and treatment. A review. Rocz. Panstw. Zakl. Hig. 2018, 69, 5–14. [Google Scholar] [PubMed]
- Fernandez-Marin, M.; Mateos, R.; Garcia-Parrilla, M.; Puertas, B.; Cantos-Villar, E. Bioactive compounds in wine: Resveratrol, hydroxytyrosol and melatonin: A review. Food Chem. 2012, 130, 797–813. [Google Scholar] [CrossRef]
- Mikstacka, R.; Ignatowicz, E. Chemopreventive and chemotherapeutic effect of trans-resveratrol and its analogues in cancer. Pol. Merkur. Lek. Organ Pol. Tow. Lek. 2010, 28, 496–500. [Google Scholar]
- Hurst, W.J.; Glinski, J.A.; Miller, K.B.; Apgar, J.; Davey, M.H.; Stuart, D.A. Survey of the trans-resveratrol and trans-piceid content of cocoa-containing and chocolate products. J. Agric. Food Chem. 2008, 56, 8374–8378. [Google Scholar] [CrossRef] [PubMed]
- Patel, K.R.; Brown, V.A.; Jones, D.J.; Britton, R.G.; Hemingway, D.; Miller, A.S.; West, K.P.; Booth, T.D.; Perloff, M.; Crowell, J.A.; et al. Clinical pharmacology of resveratrol and its metabolites in colorectal cancer patients. Cancer Res. 2010, 70, 7392–7399. [Google Scholar] [CrossRef] [PubMed]
- Singh, C.K.; Ndiaye, M.A.; Ahmad, N. Resveratrol and cancer: Challenges for clinical translation. Biochim. Biophys. Acta 2015, 1852, 1178–1185. [Google Scholar] [CrossRef]
- Almeida, L.; Vaz-da-Silva, M.; Falcao, A.; Soares, E.; Costa, R.; Loureiro, A.I.; Fernandes-Lopes, C.; Rocha, J.F.; Nunes, T.; Wright, L.; et al. Pharmacokinetic and safety profile of trans-resveratrol in a rising multiple-dose study in healthy volunteers. Mol. Nutr. Food Res. 2009, 53, 7–15. [Google Scholar] [CrossRef]
- Ganapathy, S.; Chen, Q.; Singh, K.P.; Shankar, S.; Srivastava, R.K. Resveratrol Enhances Antitumor Activity of TRAIL in Prostate Cancer Xenografts through Activation of FOXO Transcription Factor. PLoS ONE 2011, 5, e15627. [Google Scholar] [CrossRef]
- Kumar, A.; Dhar, S.; Rimando, A.M.; Lage, J.M.; Lewin, J.R.; Zhang, X.; Levenson, A.S. Epigenetic potential of resveratrol and analogs in preclinical models of prostate cancer. Ann. N. Y. Acad. Sci. 2015, 1348, 1–9. [Google Scholar] [CrossRef]
- Kampa, M.; Hatzoglou, A.; Notas, G.; Damianaki, A.; Bakogeorgou, E.; Gemetzi, C.; Kouroumalis, E.; Martin, P.M.; Castanas, E. Wine antioxidant polyphenols inhibit the proliferation of human prostate cancer cell lines. Nutr. Cancer 2000, 37, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Harper, C.E.; Patel, B.B.; Wang, J.; Arabshahi, A.; Eltoum, I.A.; Lamartiniere, C.A. Resveratrol suppresses prostate cancer progression in transgenic mice. Carcinogenesis 2007, 28, 1946–1953. [Google Scholar] [CrossRef] [PubMed]
- Jang, Y.G.; Go, R.E.; Hwang, K.A.; Choi, K.C. Resveratrol inhibits DHT-induced progression of prostate cancer cell line through interfering with the AR and CXCR4 pathway. J. Steroid Biochem. Mol. Biol. 2019, 192, 105406. [Google Scholar] [CrossRef] [PubMed]
- Benitez, D.A.; Pozo-Guisado, E.; Clementi, M.; Castellón, E.; Fernandez-Salguero, P.M. Non-genomic action of resveratrol on androgen and oestrogen receptors in prostate cancer: Modulation of the phosphoinositide 3-kinase pathway. Br. J. Cancer 2007, 96, 1595–1604. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Stockwell, T.; Roemer, A.; Chikritzhs, T. Is alcohol consumption a risk factor for prostate cancer? A systematic review and meta-analysis. BMC Cancer 2016, 16, 845. [Google Scholar] [CrossRef] [PubMed]
- Vartolomei, M.D.; Kimura, S.; Ferro, M.; Foerster, B.; Abufaraj, M.; Briganti, A.; Karakiewicz, P.I.; Shariat, S.F. The impact of moderate wine consumption on the risk of developing prostate cancer. Clin. Epidemiol. 2018, 10, 431–444. [Google Scholar] [CrossRef] [PubMed]
- Sutcliffe, S.; Giovannucci, E.; Leitzmann, M.F.; Rimm, E.B.; Stampfer, M.J.; Willett, W.C.; Platz, E.A. A prospective cohort study of red wine consumption and risk of prostate cancer. Int. J. Cancer 2007, 120, 1529–1535. [Google Scholar] [CrossRef] [PubMed]
- Kjaer, T.N.; Ornstrup, M.J.; Poulsen, M.M.; Jorgensen, J.O.; Hougaard, D.M.; Cohen, A.S.; Neghabat, S.; Richelsen, B.; Pedersen, S.B. Resveratrol reduces the levels of circulating androgen precursors but has no effect on, testosterone, dihydrotestosterone, PSA levels or prostate volume. A 4-month randomised trial in middle-aged men. Prostate 2015, 75, 1255–1263. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Wang, H.; Li, G.X.; Yang, Z.; Guan, F.; Jin, H. Cancer prevention by tea: Evidence from laboratory studies. Pharmacol. Res. 2011, 64, 113–122. [Google Scholar] [CrossRef]
- Yang, C.S.; Wang, X.; Lu, G.; Picinich, S.C. Cancer prevention by tea: Animal studies, molecular mechanisms and human relevance. Nat. Rev. Cancer 2009, 9, 429–439. [Google Scholar] [CrossRef]
- Kumar, N.B.; Dickinson, S.I.; Schell, M.J.; Manley, B.J.; Poch, M.A.; Pow-Sang, J. Green tea extract for prevention of prostate cancer progression in patients on active surveillance. Oncotarget 2018, 9, 37798–37806. [Google Scholar] [CrossRef]
- Chuu, C.P.; Chen, R.Y.; Kokontis, J.M.; Hiipakka, R.A.; Liao, S. Suppression of androgen receptor signaling and prostate specific antigen expression by (-)-epigallocatechin-3-gallate in different progression stages of LNCaP prostate cancer cells. Cancer Lett. 2009, 275, 86–92. [Google Scholar] [CrossRef]
- Albrecht, D.S.; Clubbs, E.A.; Ferruzzi, M.; Bomser, J.A. Epigallocatechin-3-gallate (EGCG) inhibits PC-3 prostate cancer cell proliferation via MEK-independent ERK1/2 activation. Chem. Biol. Interac. 2008, 171, 89–95. [Google Scholar] [CrossRef]
- Rocha, S.; Generalov, R.; Pereira Mdo, C.; Peres, I.; Juzenas, P.; Coelho, M.A. Epigallocatechin gallate-loaded polysaccharide nanoparticles for prostate cancer chemoprevention. Nanomed. (Lond. Engl.) 2011, 6, 79–87. [Google Scholar] [CrossRef]
- Johnson, J.J.; Bailey, H.H.; Mukhtar, H. Green tea polyphenols for prostate cancer chemoprevention: A translational perspective. Phytomed. Int. J. Phytother. Phytopharmacol. 2010, 17, 3–13. [Google Scholar] [CrossRef]
- Guo, Y.; Zhi, F.; Chen, P.; Zhao, K.; Xiang, H.; Mao, Q.; Wang, X.; Zhang, X. Green tea and the risk of prostate cancer: A systematic review and meta-analysis. Med. (Baltim.) 2017, 96, e6426. [Google Scholar] [CrossRef]
- Lin, Y.W.; Hu, Z.H.; Wang, X.; Mao, Q.Q.; Qin, J.; Zheng, X.Y.; Xie, L.P. Tea consumption and prostate cancer: An updated meta-analysis. World J. Surg. Oncol. 2014, 12, 38. [Google Scholar] [CrossRef]
- Fei, X.; Shen, Y.; Li, X.; Guo, H. The association of tea consumption and the risk and progression of prostate cancer: A meta-analysis. Int. J. Clin. Exp. Med. 2014, 7, 3881–3891. [Google Scholar]
- Jian, L.; Xie, L.P.; Lee, A.H.; Binns, C.W. Protective effect of green tea against prostate cancer: A case-control study in southeast China. Int. J. Cancer 2004, 108, 130–135. [Google Scholar] [CrossRef]
- Bettuzzi, S.; Brausi, M.; Rizzi, F.; Castagnetti, G.; Peracchia, G.; Corti, A. Chemoprevention of human prostate cancer by oral administration of green tea catechins in volunteers with high-grade prostate intraepithelial neoplasia: A preliminary report from a one-year proof-of-principle study. Cancer Res. 2006, 66, 1234–1240. [Google Scholar] [CrossRef]
- Kumar, N.B.; Pow-Sang, J.; Egan, K.M.; Spiess, P.E.; Dickinson, S.; Salup, R.; Helal, M.; McLarty, J.; Williams, C.R.; Schreiber, F.; et al. Randomized, Placebo-Controlled Trial of Green Tea Catechins for Prostate Cancer Prevention. Cancer Prev. Res. (Phila.) 2015, 8, 879–887. [Google Scholar] [CrossRef]
- Henning, S.M.; Wang, P.; Said, J.W.; Huang, M.; Grogan, T.; Elashoff, D.; Carpenter, C.L.; Heber, D.; Aronson, W.J. Randomized clinical trial of brewed green and black tea in men with prostate cancer prior to prostatectomy. Prostate 2015, 75, 550–559. [Google Scholar] [CrossRef]
- Hewlings, S.J.; Kalman, D.S. Curcumin: A Review of Its’ Effects on Human Health. Foods 2017, 6, 92. [Google Scholar] [CrossRef]
- Pan, M.H.; Huang, T.M.; Lin, J.K. Biotransformation of curcumin through reduction and glucuronidation in mice. Drug Metab. Dispos. Biol. Fate Chem. 1999, 27, 486–494. [Google Scholar]
- Prasad, S.; Tyagi, A.K.; Aggarwal, B.B. Recent developments in delivery, bioavailability, absorption and metabolism of curcumin: The golden pigment from golden spice. Cancer Res. Treat. 2014, 46, 2–18. [Google Scholar] [CrossRef]
- Shoba, G.; Joy, D.; Joseph, T.; Majeed, M.; Rajendran, R.; Srinivas, P.S. Influence of piperine on the pharmacokinetics of curcumin in animals and human volunteers. Planta Med. 1998, 64, 353–356. [Google Scholar] [CrossRef]
- Yu, X.L.; Jing, T.; Zhao, H.; Li, P.J.; Xu, W.H.; Shang, F.F. Curcumin inhibits expression of inhibitor of DNA binding 1 in PC3 cells and xenografts. Asian Pac. J. Cancer Prev. APJCP 2014, 15, 1465–1470. [Google Scholar] [CrossRef]
- Korang-Yeboah, M.; Patel, D.; Morton, D.; Sharma, P.; Gorantla, Y.; Joshi, J.; Nagappan, P.; Pallaniappan, R.; Chaudhary, J. Intra-tumoral delivery of functional ID4 protein via PCL/maltodextrin nano-particle inhibits prostate cancer growth. Oncotarget 2016, 7, 68072–68085. [Google Scholar] [CrossRef]
- Khurana, N.; Sikka, S.C. Targeting Crosstalk between Nrf-2, NF-κB and Androgen Receptor Signaling in Prostate Cancer. Cancers (Basel) 2018, 10, 352. [Google Scholar] [CrossRef]
- Li, W.; Su, Z.Y.; Guo, Y.; Zhang, C.; Wu, R.; Gao, L.; Zheng, X.; Du, Z.Y.; Zhang, K.; Kong, A.N. Curcumin Derivative Epigenetically Reactivates Nrf2 Antioxidative Stress Signaling in Mouse Prostate Cancer TRAMP C1 Cells. Chem. Res. Toxicol. 2018, 31, 88–96. [Google Scholar] [CrossRef]
- Nakamura, K.; Yasunaga, Y.; Segawa, T.; Ko, D.; Moul, J.W.; Srivastava, S.; Rhim, J.S. Curcumin down-regulates AR gene expression and activation in prostate cancer cell lines. Int. J. Oncol. 2002, 21, 825–830. [Google Scholar] [CrossRef]
- Shah, S.; Prasad, S.; Knudsen, K.E. Targeting pioneering factor and hormone receptor cooperative pathways to suppress tumor progression. Cancer Res. 2012, 72, 1248–1259. [Google Scholar] [CrossRef]
- Choi, Y.H.; Han, D.H.; Kim, S.W.; Kim, M.J.; Sung, H.H.; Jeon, H.G.; Jeong, B.C.; Seo, S.I.; Jeon, S.S.; Lee, H.M.; et al. A randomized, double-blind, placebo-controlled trial to evaluate the role of curcumin in prostate cancer patients with intermittent androgen deprivation. Prostate 2019, 79, 614–621. [Google Scholar] [CrossRef]
- Hejazi, J.; Rastmanesh, R.; Taleban, F.A.; Molana, S.H.; Hejazi, E.; Ehtejab, G.; Hara, N. Effect of Curcumin Supplementation During Radiotherapy on Oxidative Status of Patients with Prostate Cancer: A Double Blinded, Randomized, Placebo-Controlled Study. Nutr. Cancer 2016, 68, 77–85. [Google Scholar] [CrossRef]
- Selma, M.V.; Beltran, D.; Luna, M.C.; Romo-Vaquero, M.; Garcia-Villalba, R.; Mira, A.; Espin, J.C.; Tomas-Barberan, F.A. Isolation of Human Intestinal Bacteria Capable of Producing the Bioactive Metabolite Isourolithin A from Ellagic Acid. Front. Microbiol. 2017, 8, 1521. [Google Scholar] [CrossRef]
- Bassiri-Jahromi, S. Punica granatum (Pomegranate) activity in health promotion and cancer prevention. Oncol. Rev. 2018, 12, 345. [Google Scholar] [CrossRef]
- Adaramoye, O.; Erguen, B.; Nitzsche, B.; Hopfner, M.; Jung, K.; Rabien, A. Punicalagin, a polyphenol from pomegranate fruit, induces growth inhibition and apoptosis in human PC-3 and LNCaP cells. Chem. Biol. Interac. 2017, 274, 100–106. [Google Scholar] [CrossRef]
- Mohammed Saleem, Y.I.; Albassam, H.; Selim, M. Urolithin A induces prostate cancer cell death in p53-dependent and in p53-independent manner. Eur. J. Nutr. 2019, 1–2. [Google Scholar] [CrossRef]
- Shi, D.; Gu, W. Dual Roles of MDM2 in the Regulation of p53: Ubiquitination Dependent and Ubiquitination Independent Mechanisms of MDM2 Repression of p53 Activity. Genes Cancer 2012, 3, 240–248. [Google Scholar] [CrossRef]
- Deng, Y.; Li, Y.; Yang, F.; Zeng, A.; Yang, S.; Luo, Y.; Zhang, Y.; Xie, Y.; Ye, T.; Xia, Y.; et al. The extract from Punica granatum (pomegranate) peel induces apoptosis and impairs metastasis in prostate cancer cells. Biomed. Pharmacother. 2017, 93, 976–984. [Google Scholar] [CrossRef]
- Deng, Y.L.; Li, Y.L.; Zheng, T.T.; Hu, M.X.; Ye, T.H.; Xie, Y.M.; Yin, W.Y. The Extract from Punica Granatum (Pomegranate) Leaves Promotes Apoptosis and Impairs Metastasis in Prostate Cancer Cells. Sichuan Da Xue Xue Bao Yi Xue Ban 2018, 49, 8–12. [Google Scholar]
- Landete, J.M. Ellagitannins, ellagic acid and their derived metabolites: A review about source, metabolism, functions and health. Food Res. Int. 2011, 44, 1155. [Google Scholar] [CrossRef]
- Paller, C.J.; Pantuck, A.; Carducci, M.A. A review of pomegranate in prostate cancer. Prostate Cancer Prostatic Dis. 2017, 20, 265–270. [Google Scholar] [CrossRef]
- Domingo-Domenech, J.; Mellado, B.; Ferrer, B.; Truan, D.; Codony-Servat, J.; Sauleda, S.; Alcover, J.; Campo, E.; Gascon, P.; Rovira, A.; et al. Activation of nuclear factor-kappaB in human prostate carcinogenesis and association to biochemical relapse. Br. J. Cancer 2005, 93, 1285–1294. [Google Scholar] [CrossRef]
- Fradet, V.; Lessard, L.; Begin, L.R.; Karakiewicz, P.; Masson, A.M.; Saad, F. Nuclear factor-kappaB nuclear localization is predictive of biochemical recurrence in patients with positive margin prostate cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2004, 10, 8460–8464. [Google Scholar] [CrossRef]
- Klein, K.A.; Reiter, R.E.; Redula, J.; Moradi, H.; Zhu, X.L.; Brothman, A.R.; Lamb, D.J.; Marcelli, M.; Belldegrun, A.; Witte, O.N.; et al. Progression of metastatic human prostate cancer to androgen independence in immunodeficient SCID mice. Nat. Med. 1997, 3, 402–408. [Google Scholar] [CrossRef]
- Rettig, M.B.; Heber, D.; An, J.; Seeram, N.P.; Rao, J.Y.; Liu, H.; Klatte, T.; Belldegrun, A.; Moro, A.; Henning, S.M.; et al. Pomegranate extract inhibits androgen-independent prostate cancer growth through a nuclear factor-kappaB-dependent mechanism. Mol. Cancer Therapeut. 2008, 7, 2662–2671. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, S.; Iqbal, S.; Chen, Z.; Wang, X.; Wang, Y.A.; Liu, D.; Bai, K.; Ritenour, C.; Kucuk, O.; et al. Pomegranate extract inhibits the bone metastatic growth of human prostate cancer cells and enhances the in vivo efficacy of docetaxel chemotherapy. Prostate 2014, 74, 497–508. [Google Scholar] [CrossRef]
- Wang, L.; Martins-Green, M. Pomegranate and its components as alternative treatment for prostate cancer. Int. J. Mol. Sci. 2014, 15, 14949–14966. [Google Scholar] [CrossRef]
- Wang, D.; Ozen, C.; Abu-Reidah, I.M.; Chigurupati, S.; Patra, J.K.; Horbanczuk, J.O.; Jozwik, A.; Tzvetkov, N.T.; Uhrin, P.; Atanasov, A.G. Vasculoprotective Effects of Pomegranate (Punica Granatum L.). Front. Pharmacol. 2018, 9, 544. [Google Scholar] [CrossRef]
- Pantuck, A.J.; Leppert, J.T.; Zomorodian, N.; Aronson, W.; Hong, J.; Barnard, R.J.; Seeram, N.; Liker, H.; Wang, H.; Elashoff, R.; et al. Phase II study of pomegranate juice for men with rising prostate-specific antigen following surgery or radiation for prostate cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2006, 12, 4018–4026. [Google Scholar] [CrossRef]
- Paller, C.J.; Ye, X.; Wozniak, P.J.; Gillespie, B.K.; Sieber, P.R.; Greengold, R.H.; Stockton, B.R.; Hertzman, B.L.; Efros, M.D.; Roper, R.P.; et al. A randomized phase II study of pomegranate extract for men with rising PSA following initial therapy for localized prostate cancer. Prostate Cancer Prostatic Dis. 2013, 16, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Stenner-Liewen, F.; Liewen, H.; Cathomas, R.; Renner, C.; Petrausch, U.; Sulser, T.; Spanaus, K.; Seifert, H.H.; Strebel, R.T.; Knuth, A.; et al. Daily Pomegranate Intake Has No Impact on PSA Levels in Patients with Advanced Prostate Cancer—Results of a Phase IIb Randomized Controlled Trial. J. Cancer 2013, 4, 597–605. [Google Scholar] [CrossRef] [PubMed]
- Freedland, S.J.; Carducci, M.; Kroeger, N.; Partin, A.; Rao, J.Y.; Jin, Y.; Kerkoutian, S.; Wu, H.; Li, Y.; Creel, P.; et al. A double-blind, randomized, neoadjuvant study of the tissue effects of POMx pills in men with prostate cancer before radical prostatectomy. Cancer Prev. Res. (Phila.) 2013, 6, 1120–1127. [Google Scholar] [CrossRef] [PubMed]
- Sivapalan, T.; Melchini, A.; Saha, S.; Needs, P.W.; Traka, M.H.; Tapp, H.; Dainty, J.R.; Mithen, R.F. Bioavailability of Glucoraphanin and Sulforaphane from High-Glucoraphanin Broccoli. Mol. Nut. Food Res. 2018, 62, e1700911. [Google Scholar] [CrossRef] [PubMed]
- Gasper, A.V.; Al-Janobi, A.; Smith, J.A.; Bacon, J.R.; Fortun, P.; Atherton, C.; Taylor, M.A.; Hawkey, C.J.; Barrett, D.A.; Mithen, R.F. Glutathione S-transferase M1 polymorphism and metabolism of sulforaphane from standard and high-glucosinolate broccoli. Am. J. Clin. Nutr. 2005, 82, 1283–1291. [Google Scholar] [CrossRef] [PubMed]
- Doble, A..; Walker, M.M.; Harris, J.R.; Taylor-Robinson, D.; Witherow, R.O. Intraprostatic antibody deposition in chronic abacterial prostatitis. Br. J. Urol. 1990, 65, 598–605. [Google Scholar] [CrossRef] [PubMed]
- Kirby, R.S.; Lowe, D.; Bultitude, M.I.; Shuttleworth, K.E.D. Intra-prostatic Urinary Reflux: An Aetiological Factor in Abacterial Prostatitis. Br. J. Urol. 1982, 54, 729–731. [Google Scholar] [CrossRef] [PubMed]
- Balasar, M.; Doğan, M.; Kandemir, A.; Taskapu, H.H.; Cicekci, F.; Toy, H.; Gurbuz, R. Investigation of granulomatous prostatitis incidence following intravesical BCG therapy. Int. J. Clin. Exp. Med. 2014, 7, 1554–1557. [Google Scholar]
- Terris, M.K.; Macy, M.; Freiha, F.S. Transrectal Ultrasound Appearance of Prostatic Granulomas Secondary to Bacillus Calmette-Guerin Instillation. J. Urol. 1997, 158, 126–127. [Google Scholar] [CrossRef]
- Butel, R.; Ball, R. The distribution of BCG prostatitis: A clue for pathogenetic processes? Prostate 2018, 78, 1134–1139. [Google Scholar] [CrossRef]
- Blacklock, N.J. Anatomical factors in Prostatitis. Br. J. Urol. 1974, 46, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Nickel, J.C. Textbook of Prostatitis; Taylor and Francis, Isis Medical Media Ltd.: Oxford, UK, 1999. [Google Scholar]
- Fraser, M.; Sabelnykova, V.Y.; Yamaguchi, T.N.; Heisler, L.E.; Livingstone, J.; Huang, V.; Shiah, Y.J.; Yousif, F.; Lin, X.; Masella, A.P.; et al. Genomic hallmarks of localized, non-indolent prostate cancer. Nature 2017, 541, 359–364. [Google Scholar] [CrossRef] [PubMed]
- Abida, W.; Cyrta, J.; Heller, G.; Prandi, D.; Armenia, J.; Coleman, I.; Cieslik, M.; Benelli, M.; Robinson, D.; Van Allen, E.M.; et al. Genomic correlates of clinical outcome in advanced prostate cancer. Proc. Natl. Acad. Sci. USA 2019, 116, 11428–11436. [Google Scholar] [CrossRef] [PubMed]
- Quigley, D.A.; Dang, H.X.; Zhao, S.G.; Lloyd, P.; Aggarwal, R.; Alumkal, J.J.; Foye, A.; Kothari, V.; Perry, M.D.; Bailey, A.M.; et al. Genomic Hallmarks and Structural Variation in Metastatic Prostate Cancer. Cell 2018, 174, 758–769.e759. [Google Scholar] [CrossRef]
- Cavarretta, I.; Ferrarese, R.; Cazzaniga, W.; Saita, D.; Lucianò, R.; Ceresola, E.R.; Locatelli, I.; Visconti, L.; Lavorgna, G.; Briganti, A.; et al. The Microbiome of the Prostate Tumor Microenvironment. Eur. Urol. 2017, 72, 625–631. [Google Scholar] [CrossRef]
- Banerjee, S.; Alwine, J.C.; Wei, Z.; Tian, T.; Shih, N.; Sperling, C.; Guzzo, T.; Feldman, M.D.; Robertson, E.S. Microbiome signatures in prostate cancer. Carcinogenesis 2019. [Google Scholar] [CrossRef]
- Dennis, L.K.; Lynch, C.F.; Torner, J.C. Epidemiologic association between prostatitis and prostate cancer. Urology 2002, 60, 78–83. [Google Scholar] [CrossRef]
- Alfano, M.; Canducci, F.; Nebuloni, M.; Clementi, M.; Montorsi, F.; Salonia, A. The interplay of extracellular matrix and microbiome in urothelial bladder cancer. Nat. Rev. Urol. 2015, 13, 77. [Google Scholar] [CrossRef]
- Simons, B.W.; Durham, N.M.; Bruno, T.C.; Grosso, J.F.; Schaeffer, A.J.; Ross, A.E.; Hurley, P.J.; Berman, D.M.; Drake, C.G.; Thumbikat, P.; et al. A human prostatic bacterial isolate alters the prostatic microenvironment and accelerates prostate cancer progression. J. Pathol. 2015, 235, 478–489. [Google Scholar] [CrossRef]
- Sfanos, K.S.; De Marzo, A.M. Prostate cancer and inflammation: The evidence. Histopathology 2012, 60, 199–215. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority (EFSA). Available online: http://www.efsa.europa.eu/en/topics/topic/nutrition (accessed on 8 August 2019).
- Lucey, A.; Heneghan, C.; Kiely, M.E. Guidance for the design and implementation of human dietary intervention studies for health claim submissions. Nutr. Bull. 2016, 41, 378–394. [Google Scholar] [CrossRef]
- Pressman, P.; Clemens, R.A.; Hayes, A.W. Bioavailability of micronutrients obtained from supplements and food: A survey and case study of the polyphenols. Toxicol. Res. Appl. 2017, 1, 2397847317696366. [Google Scholar] [CrossRef]
- Lawson, L.D.; Hunsaker, S.M. Allicin Bioavailability and Bioequivalence from Garlic Supplements and Garlic Foods. Nutrients 2018, 10, 812. [Google Scholar] [CrossRef]
- Mithen, R.; Faulkner, K.; Magrath, R.; Rose, P.; Williamson, G.; Marquez, J. Development of isothiocyanate-enriched broccoli, and its enhanced ability to induce phase 2 detoxification enzymes in mammalian cells. Theor. Appl. Genet. 2003, 106, 727–734. [Google Scholar] [CrossRef] [PubMed]
- United States Deparment of Agriculture Food Composition Databases (USDA). Available online: https://ndb.nal.usda.gov/ndb/ (accessed on 14 June 2019).
- Phenol-Explorer: Database on Polyphenol Content in Foods. Available online: http://phenol-explorer.eu/ (accessed on 14 June 2019).
- Bioactive Substances in Food Information System (eBASIS). Available online: http://ebasis.eurofir.org/ (accessed on 14 June 2019).
- Plumb, J.; Lyons, J.; Norby, K.; Thomas, M.; Norby, E.; Poms, R.; Bucchini, L.; Restani, P.; Kiely, M.; Finglas, P. ePlantLIBRA: A composition and biological activity database for bioactive compounds in plant food supplements. Food Chem. 2016, 193, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Kiely, M.; Black, L.J.; Plumb, J.; Kroon, P.A.; Hollman, P.C.; Larsen, J.C.; Speijers, G.J.; Kapsokefalou, M.; Sheehan, D.; Gry, J.; et al. EuroFIR eBASIS: Application for health claims submissions and evaluations. Eur. J. Clin. Nutr. 2010, 64, S101. [Google Scholar] [CrossRef] [PubMed]
- Plumb, J.; Pigat, S.; Bompola, F.; Cushen, M.; Pinchen, H.; Nørby, E.; Astley, S.; Lyons, J.; Kiely, M.; Finglas, P. eBASIS (Bioactive Substances in Food Information Systems) and Bioactive Intakes: Major Updates of the Bioactive Compound Composition and Beneficial Bioeffects Database and the Development of a Probabilistic Model to Assess Intakes in Europe. Nutrients 2017, 9, 320. [Google Scholar] [CrossRef] [PubMed]
- Francini, E.; Petrioli, R.; Rossi, G.; Laera, L.; Roviello, G. PSA response rate as a surrogate marker for median overall survival in docetaxel-based first-line treatments for patients with metastatic castration-resistant prostate cancer: An analysis of 22 trials. Tumour Biol. J. Int. Soc. Oncodev. Biol. Med. 2014, 35, 10601–10607. [Google Scholar] [CrossRef]
- Hayes, J.H.; Barry, M.J. Screening for prostate cancer with the prostate-specific antigen test: A review of current evidence. Jama 2014, 311, 1143–1149. [Google Scholar] [CrossRef]
- Eggener, S.E.; Cifu, A.S.; Nabhan, C. Prostate Cancer ScreeningProstate Cancer ScreeningProstate Cancer Screening. Jama 2015, 314, 825–826. [Google Scholar] [CrossRef]
- D’Amico, A.V.; Moul, J.; Carroll, P.R.; Sun, L.; Lubeck, D.; Chen, M.H. Cancer-specific mortality after surgery or radiation for patients with clinically localized prostate cancer managed during the prostate-specific antigen era. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2003, 21, 2163–2172. [Google Scholar] [CrossRef]
| Author, Year | Study Type | Patient Cohort/Intervention | Analysis |
|---|---|---|---|
| Glucoraphanin and Sulforaphane from Cruciferous Vegetables | |||
| Liu et al. 2012 [39] | Meta-analysis 6 case-control 7 cohort studies | 10 studies USA/Canada, 1 Asia, 2 Europe. | RR = 0.90; 95% CI 0.85–0.96 for overall cruciferous vegetable intake RR = 0.79; 95% CI 0.69–0.89 for case control studies |
| Richman et al. 2012 [40] | Prospective study n = 1560 | USA PCa registry. Biopsy-verified localised PCa. Clinical survey and FFQ at baseline and every 6 months | 59% reduced risk of PCa progression for highest vs. lowest intake of cruciferous vegetables. HR: 0.41, 95% CI 0.22–0.76 (p = 0.003) |
| Zhang et al. 2019 [44] | 2-arm parallel randomised double-blinded intervention trial n = 98 | USA cohort. Intervention for 4-6 weeks prior to prostate biopsy procedure | Accumulation of urine and plasma SFN ITCs and individual SFN metabolites. 40 differentially expressed genes correlated with treatment Downregulation of AMACR and ARLNC1 genes No significant difference in HDAC activity or prostate tissue biomarkers |
| Traka et al. 2019 [45] | 3-arm parallel randomised double-blinded 12-month intervention trial n = 49 | UK cohort. Prostate biopsies at the start and end of 12-month intervention | Dose-dependent attenuation of gene expression and associated oncogenic pathways |
| SACSO from Alliaceous Vegetables | |||
| Hsing et al. 2002 [53] | Population based study 238 case subjects 471 control subjects | Shanghai, China. Cases: histologically confirmed PCa. In-person interviews and FFQ. | Highest allium intake (>10.0 g/day) OR = 0.51, 95% CI 0.34–0.76 p < 0.001 Garlic (OR = 0.47, 95% CI 0.31–0.71; p < 0.001) |
| Zhou et al. 2013 [54] | Systematic literature review 6 case-control 3 prospective cohort studies | 3 studies Europe, 3 studies USA, 2 studies Asia, 1 Australia. Interview or self-administered FFQ | OR = 0.82 95% CI 0.70–0.97 for allium intake OR = 0.77 95% CI 0.64–0.91 for garlic intake |
| Lycopene from Tomatoes | |||
| Rowles et al. 2017 [77] | Systematic literature review 43 case control studies | 32 studies N. America, 6 Europe, 2 Australia, 2 Asia (China and Singapore), 1 S. America. FFQs and 1 x interview | RR = 0.88, 95% CI 0.78−0.98, p = 0.017 (for localised PCa risk) RR = 0.88, 95% CI 0.79−0.98, p = 0.019 for circulating lycopene concentrations (and localised PCa risk) Dose-response seen. No effect for risk of advanced PCa. |
| Wang et al. 2015 [78] | Systematic review and dose-response meta-analysis 10 cohort 11 nested case-control 13 case-control studies | 22 studies N. America, 7 studies Europe, 2 Australia, 2 Asia, 1 S. America | RR = 0.86, 95% CI 0.75–0.98 (localised PCa risk) RR 0.81, 95% CI 0.69–0.96 for blood lycopene levels. Dose-response seen No effect for risk of advanced PCa |
| Van Hoang et al. 2018 [79] | Case-control study n = 652 | Vietnamese cohort. Cases (244) with localised PCa, and PSA 4 ng/mL. Face-to-face interviews using a structured semi-quantitative validated FFQ | OR = 0.46 95% CI 0.27–0.77 for highest lycopene intake |
| Key et al. 2015 [80] | Pooled Analysis of 15 studies 15 case-control studies n = 29780 | 6 studies Europe, 6 studies US, 1 study Afro-Caribbean, 1 Australia, 1 Mixed-cohort (Australia and Europe) | No association between intake and overall risk of PCa. OR 0.65 95% CI: 0.46, 0.91; p = 0.032 for risk of advanced stage PCa and aggressive disease |
| Giovannucci et al. 2002 [82] | Prospective Study n = 47365 | US male health professional cohort – ‘Health Professionals Follow-Up Study’ (HPFS) | RR = 0.84 CI 0.73–0.96 p = 0.03 for total intake RR 0.77 95% CI 0.66–0.90 p < 0.001 for high tomato sauce intake RR = 0.65 95% CI 0.42–0.99 for high tomato sauce intake and extra-prostatic cancers |
| Kim et al. 2003 [83] | Double-blinded 2-arm randomised control trial n = 32 | US cohort. Tomato sauce intervention vs. no intervention for 3 weeks prior to prostatectomy | Increased abundance of apoptotic cells (from 0.84 +/- 0.13% to 2.76 +/- 0.58% p = 0.0003) and degree of apoptotic cell death (from 0.84 +/- 0.13% to 1.17 +/- 0.19% p = 0.028) in resected tumour areas |
| Resveratrol from Wine | |||
| Vartolomei et al. 2018 [99] | Meta-analysis 17 studies n = 611169 Meta-analysis of moderate wine intake: 14 studies n = 455413 6 cohort 8 case control studies | 4 studies Canada, 4 studies Europe, 4 studies USA, 2 studies Australia | No increased risk of PCa 0.98 95% CI 0.92–1.05, p = 0.57 (for all wine) Increased risk of PCa with moderate intake of white wine (RR 1.26 95% CI 1.10–1.43 p = 0.001) Decreased risk of PCa with moderate intake of red wine RR 0.88 95% CI 0.78–0.999 p = 0.047 (risk reduction of 12%) |
| Catechins from Green Tea | |||
| Guo et al. 2017 [109] | Systematic review and meta-analysis: 7 observational: 4 cohort 3 case control 3 randomised controlled trials | 6 studies Asia (incl. 1 Singapore, 4 Japan, 1 China), 2 Europe, 1 N. America, 1 Africa | RR 0.75 95% CI 0.53–1.07 for highest versus lowest category of green tea intake Dose response with each 1 cup/day increase of green tea 0.954 (95% CI 0.903–1.009) p = 0.08 after removal of heterogeneity and undertaking sensitivity analysis RR of 0.38 (95% CI 0.16–0.86, p = 0.02) - Patients with high-grade prostatic intraepithelial neoplasia (HGPIN) or atypical small acinar proliferation (ASAP) |
| Curcumin from Turmeric | |||
| Choi et al. 2019 [126] | Randomised double-blind, placebo-controlled trial n = 82 | S. Korean cohort. Curcumin capsule intervention or placebo from the beginning of ADT withdrawal. | PSA progression: 10.3% (treatment) vs 30.2% (control) p = 0.0259 |
| Hejazi et al. 2016 [127] | Randomised double-blind, placebo-controlled trial n = 40 | Iranian cohort. Curcumin capsule intervention or placebo during external-beam radiotherapy. | Increase in plasma total antioxidant capacity (TAC) significantly higher in the treatment arm (p < 0.001) Reduction in activity of superoxide of superoxide dismutase (SOD) in the treatment arm (p = 0.026) |
| Ellagitannins from Pomegranate | |||
| Pantuck et al. 2006 [144] | Phase II two-stage clinical trial n = 46 | US cohort. PSA 0.2–5ng/mL documented as rising. 8 ounces of pomegranate juice daily (570 mg total polyphenol gallic acid) until disease progression | Increase in mean PSA doubling time significantly increased with treatment: 15 months at baseline to 54 months post-treatment (p < 0.001) |
| Paller et al. 2013 [145] | Randomised phase II study n = 104 (n = 92 for analysis) | US cohort (multi-centre). Rising PSA without evidence of metastasis. 1g vs. 3g pomegranate extract capsules daily for up to 18 months. | Increase in PSA doubling time with treatment: 11.9 months at baseline to 18.5 months post-treatment (p < 0.001) |
| Stenner-Liewen et al. 2013 [146] | Randomised placebo-controlled trial n = 109 (advanced PCa) | Swiss cohort. PCa with PSA 5ng/mL. 500mL pomegranate juice vs. placebo beverage daily for 4 weeks, then all 250mL POM juice for 4 weeks. PSA measured at defined timepoints. | No significant difference in PSA levels at 28 days (p = 0.11) |
| Freedland et al. 2013 [147] | Randomised double-blind placebo-controlled trial. n = 69 | US cohort. Pomegranate extract capsules or placebo for up to 4 weeks prior to prostatectomy | Urolithin A accumulation (p = 0.031) No effect on oxidative stress / proliferation / progression / PSA levels |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Livingstone, T.L.; Beasy, G.; Mills, R.D.; Plumb, J.; Needs, P.W.; Mithen, R.; Traka, M.H. Plant Bioactives and the Prevention of Prostate Cancer: Evidence from Human Studies. Nutrients 2019, 11, 2245. https://doi.org/10.3390/nu11092245
Livingstone TL, Beasy G, Mills RD, Plumb J, Needs PW, Mithen R, Traka MH. Plant Bioactives and the Prevention of Prostate Cancer: Evidence from Human Studies. Nutrients. 2019; 11(9):2245. https://doi.org/10.3390/nu11092245
Chicago/Turabian StyleLivingstone, Tracey L., Gemma Beasy, Robert D. Mills, Jenny Plumb, Paul W. Needs, Richard Mithen, and Maria H. Traka. 2019. "Plant Bioactives and the Prevention of Prostate Cancer: Evidence from Human Studies" Nutrients 11, no. 9: 2245. https://doi.org/10.3390/nu11092245
APA StyleLivingstone, T. L., Beasy, G., Mills, R. D., Plumb, J., Needs, P. W., Mithen, R., & Traka, M. H. (2019). Plant Bioactives and the Prevention of Prostate Cancer: Evidence from Human Studies. Nutrients, 11(9), 2245. https://doi.org/10.3390/nu11092245





