Fermentation of Milk into Yoghurt and Cheese Leads to Contrasting Lipid and Glyceride Profiles
Abstract
1. Introduction
2. Materials and Methods
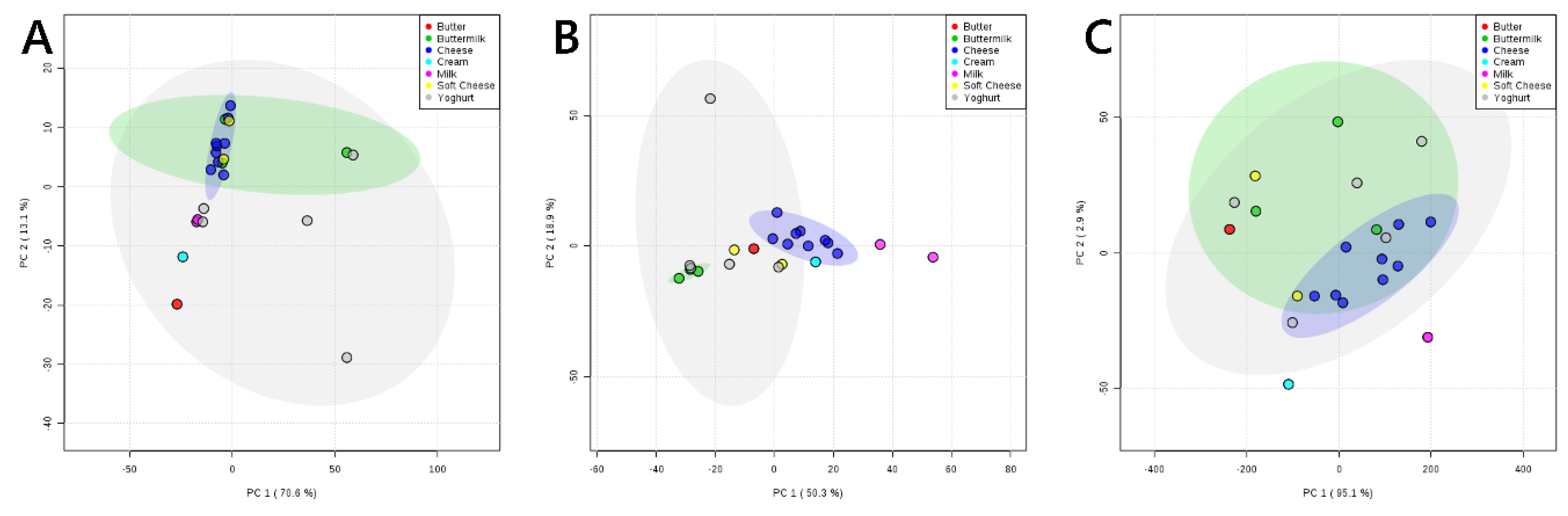
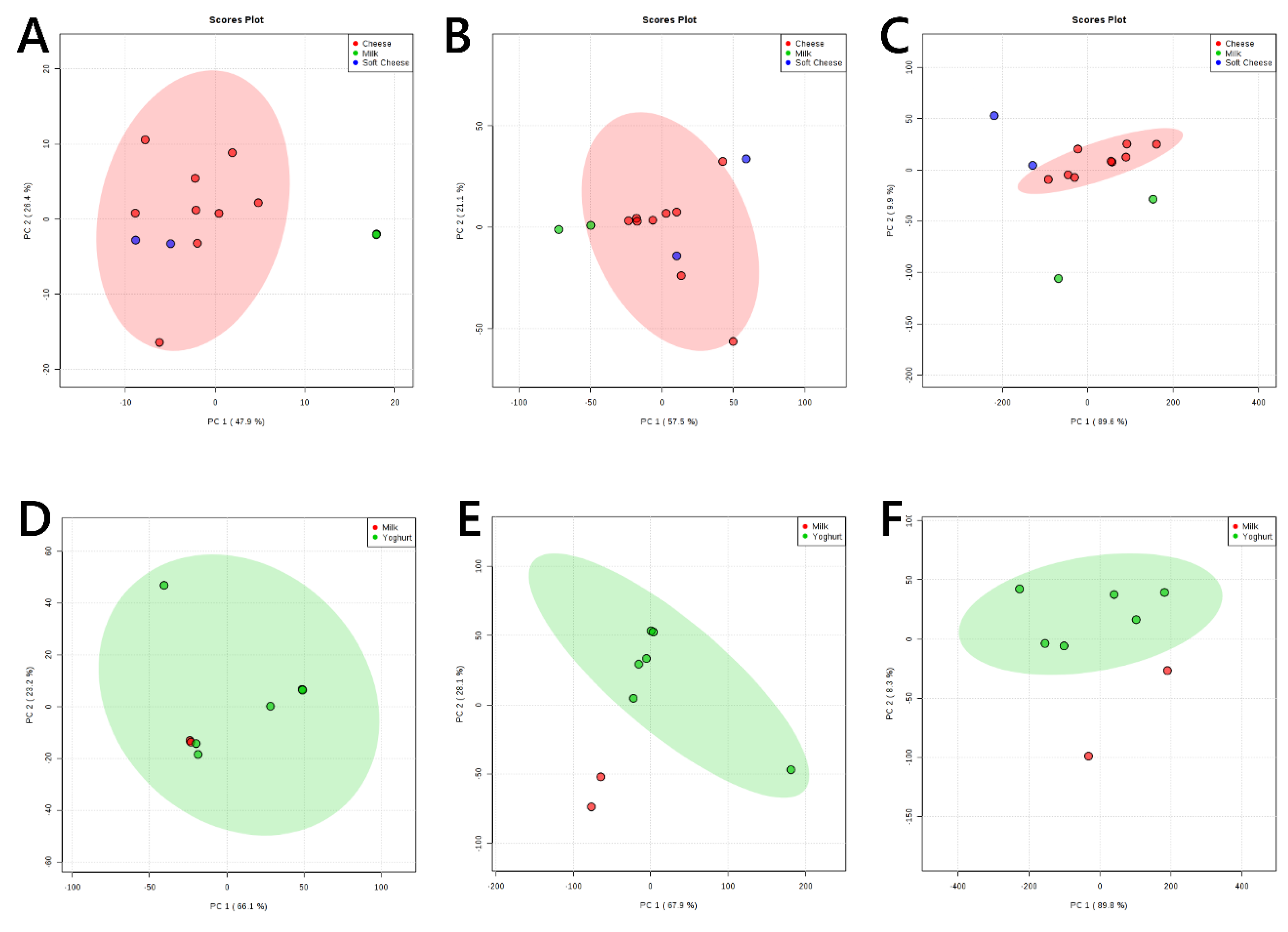
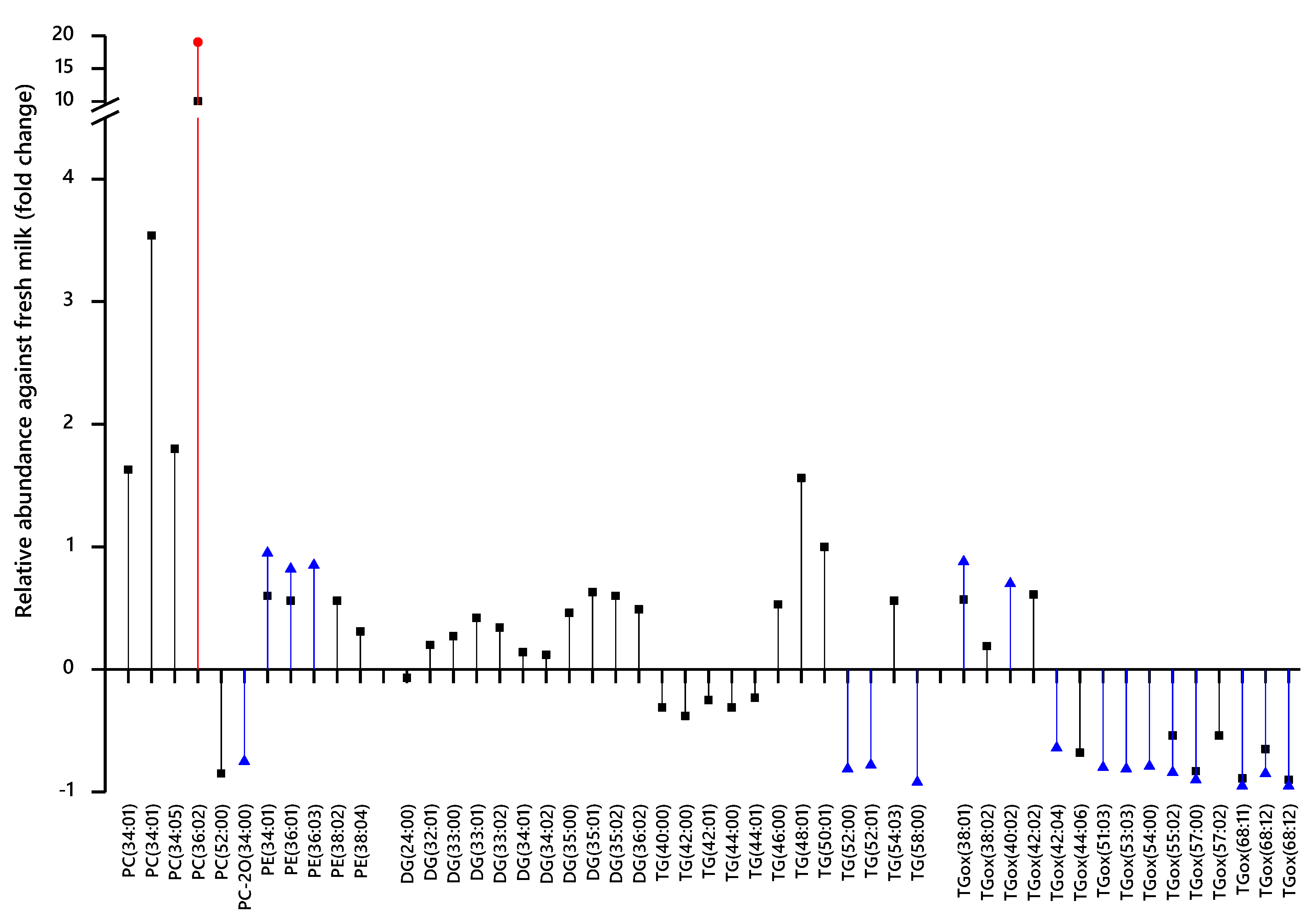
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kalergis, M.; Leung Yinko, S.S.L.; Nedelcu, R. Dairy products and prevention of type 2 diabetes: Implications for research and practice. Front. Endocrinol. 2013, 4, 90. [Google Scholar] [CrossRef] [PubMed]
- Díaz-López, A.; Bulló, M.; Martínez-González, M.A.; Corella, D.; Estruch, R.; Fitó, M.; Gómez-Gracia, E.; Fiol, M.; García de la Corte, F.J.; Ros, E.; et al. Dairy product consumption and risk of type 2 diabetes in an elderly spanish mediterranean population at high cardiovascular risk. Eur. J. Nutr. 2016, 55, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Gijsbers, L.; Ding, E.L.; Malik, V.S.; de Goede, J.; Geleijnse, J.M.; Soedamah-Muthu, S.S. Consumption of dairy foods and diabetes incidence: A dose-response meta-analysis of observational studies. Am. J. Clin. Nutr. 2016, 103, 1111–1124. [Google Scholar] [CrossRef] [PubMed]
- Lordan, R.; Tsoupras, A.; Mitra, B.; Zabetakis, I. Dairy fats and cardiovascular disease: Do we really need to be concerned? Foods 2018, 7, 29. [Google Scholar] [CrossRef] [PubMed]
- Ebringer, L.; Ferenčík, M.; Krajčovič, J. Beneficial health effects of milk and fermented dairy products—Review. Folia Microbiol. 2008, 53, 378–394. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.A.; Marette, A. Novel perspectives on fermented milks and cardiometabolic health with a focus on type 2 diabetes. Nutr. Rev. 2018, 76, 16–28. [Google Scholar] [CrossRef]
- Chen, M.; Sun, Q.; Giovannucci, E.; Mozaffarian, D.; Manson, J.E.; Willett, W.C.; Hu, F.B. Dairy consumption and risk of type 2 diabetes: 3 cohorts of us adults and an updated meta-analysis. BMC Med. 2014, 12, 215. [Google Scholar] [CrossRef]
- Chen, M.; Li, Y.; Sun, Q.; Pan, A.; Manson, J.E.; Rexrode, K.M.; Willett, W.C.; Rimm, E.B.; Hu, F.B. Dairy fat and risk of cardiovascular disease in 3 cohorts of us adults. Am. J. Clin. Nutr. 2016, 104, 1209–1217. [Google Scholar] [CrossRef]
- Nagaya, T.; Yoshida, H.; Hayashi, T.; Takahashi, H.; Kawai, M.; Matsuda, Y. Serum lipid profile in relation to milk consumption in a japanese population. J. Am. Coll. Nutr. 1996, 15, 625–629. [Google Scholar] [CrossRef]
- Xiao, J.Z.; Kondo, S.; Takahashi, N.; Miyaji, K.; Oshida, K.; Hiramatsu, A.; Iwatsuki, K.; Kokubo, S.; Hosono, A. Effects of milk products fermented by bifidobacterium longum on blood lipids in rats and healthy adult male volunteers. J. Dairy Sci. 2003, 86, 2452–2461. [Google Scholar] [CrossRef]
- Kießling, G.; Schneider, J.; Jahreis, G. Long-term consumption of fermented dairy products over 6 months increases hdl cholesterol. Eur. J. Clin. Nutr. 2002, 56, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Pereira, D.I.A.; Gibson, G.R. Effects of consumption of probiotics and prebiotics on serum lipid levels in humans. Crit. Rev. Biochem. Mol. Biol. 2002, 37, 259–281. [Google Scholar] [CrossRef] [PubMed]
- McNamara, D.J.; Lowell, A.E.; Sabb, J.E. Effect of yogurt intake on plasma lipid and lipoprotein levels in normolipidemic males. Atherosclerosis 1989, 79, 167–171. [Google Scholar] [CrossRef]
- Fernandes, C.F.; Shahani, K.M.; Amer, M.A. Therapeutic role of dietary lactobacilli and lactobacillic fermented dairy products. FEMS Microbiol. Rev. 1987, 3, 343–356. [Google Scholar] [CrossRef]
- Mena-Sánchez, G.; Babio, N.; Martínez-González, M.Á.; Corella, D.; Schröder, H.; Vioque, J.; Romaguera, D.; Martínez, J.A.; Lopez-Miranda, J.; Estruch, R.; et al. Fermented dairy products, diet quality, and cardio–metabolic profile of a mediterranean cohort at high cardiovascular risk. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 1002–1011. [Google Scholar] [CrossRef] [PubMed]
- Nestel, P.J. Effects of dairy fats within different foods on plasma lipids. J. Am. Coll. Nutr. 2008, 27, 735S–740S. [Google Scholar] [CrossRef] [PubMed]
- Johansson, I.; Nilsson, L.M.; Esberg, A.; Jansson, J.-H.; Winkvist, A. Dairy intake revisited—Associations between dairy intake and lifestyle related cardio-metabolic risk factors in a high milk consuming population. Nutr. J. 2018, 17, 110. [Google Scholar] [CrossRef]
- Jenkins, B.J.; Seyssel, K.; Chiu, S.; Pan, P.-H.; Lin, S.-Y.; Stanley, E.; Ament, Z.; West, J.A.; Summerhill, K.; Griffin, J.L.; et al. Odd chain fatty acids; new insights of the relationship between the gut microbiota, dietary intake, biosynthesis and glucose intolerance. Sci. Rep. 2017, 7, 44845. [Google Scholar] [CrossRef]
- Yakoob Mohammad, Y.; Shi, P.; Willett Walter, C.; Rexrode Kathryn, M.; Campos, H.; Orav, E.J.; Hu Frank, B.; Mozaffarian, D. Circulating biomarkers of dairy fat and risk of incident diabetes mellitus among men and women in the united states in two large prospective cohorts. Circulation 2016, 133, 1645–1654. [Google Scholar] [CrossRef]
- Jenkins, B.; Aoun, M.; Feillet-Coudray, C.; Coudray, C.; Ronis, M.; Koulman, A. The dietary total-fat content affects the in vivo circulating c15:0 and c17:0 fatty acid levels independently. Nutrients 2018, 10, 1646. [Google Scholar] [CrossRef]
- Norris, L.E.; Collene, A.L.; Asp, M.L.; Hsu, J.C.; Liu, L.-F.; Richardson, J.R.; Li, D.; Bell, D.; Osei, K.; Jackson, R.D.; et al. Comparison of dietary conjugated linoleic acid with safflower oil on body composition in obese postmenopausal women with type 2 diabetes mellitus. Am. J. Clin. Nutr. 2009, 90, 468–476. [Google Scholar] [CrossRef] [PubMed]
- Shadman, Z.; Taleban, F.A.; Saadat, N.; Hedayati, M. Effect of conjugated linoleic acid and vitamin e on glycemic control, body composition, and inflammatory markers in overweight type2 diabetics. J. Diabetes Metab. Disord. 2013, 12, 42. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Risérus, U.; Arner, P.; Brismar, K.; Vessby, B. Treatment with dietary trans-10, cis-12 conjugated linoleic acid causes isomer-specific insulin resistance in obese men with the metabolic syndrome. Diabetes Care 2002, 25, 1516–1521. [Google Scholar] [CrossRef] [PubMed]
- Rubin, D.; Herrmann, J.; Much, D.; Pfeuffer, M.; Laue, C.; Winkler, P.; Helwig, U.; Bell, D.; Auinger, A.; Darabaneanu, S.; et al. Influence of different cla isomers on insulin resistance and adipocytokines in pre-diabetic, middle-aged men with pparγ2 pro12ala polymorphism. Genes Nutr. 2012, 7, 499–509. [Google Scholar] [CrossRef][Green Version]
- Moloney, F.; Yeow, T.-P.; Mullen, A.; Nolan, J.J.; Roche, H.M. Conjugated linoleic acid supplementation, insulin sensitivity, and lipoprotein metabolism in patients with type 2 diabetes mellitus. Am. J. Clin. Nutr. 2004, 80, 887–895. [Google Scholar] [CrossRef]
- Barrett, E.; Ross, R.P.; Fitzgerald, G.F.; Stanton, C. Rapid screening method for analyzing the conjugated linoleic acid production capabilities of bacterial cultures. Appl. Environ. Microbiol. 2007, 73, 2333–2337. [Google Scholar] [CrossRef]
- Van Nieuwenhove, C.P.; Oliszewski, R.; González, S.N.; Pérez Chaia, A.B. Conjugated linoleic acid conversion by dairy bacteria cultured in mrs broth and buffalo milk. Lett. Appl. Microbiol. 2007, 44, 467–474. [Google Scholar] [CrossRef]
- McIntosh, F.M.; Shingfield, K.J.; Devillard, E.; Russell, W.R.; Wallace, R.J. Mechanism of conjugated linoleic acid and vaccenic acid formation in human faecal suspensions and pure cultures of intestinal bacteria. Microbiology 2009, 155, 285–294. [Google Scholar] [CrossRef]
- Furse, S.; Billing, G.; Snowden, S.G.; Smith, J.; Goldberg, G.; Koulman, A. Relationship between the lipid composition of maternal plasma and infant plasma through breast milk. Sci. Rep. 2019. [Google Scholar]
- Torres, A.G.; Ney, J.G.; Meneses, F.; Trugo, N.M.F. Polyunsaturated fatty acids and conjugated linoleic acid isomers in breast milk are associated with plasma non-esterified and erythrocyte membrane fatty acid composition in lactating women. Br. J. Nutr. 2006, 95, 517–524. [Google Scholar] [CrossRef]
- Prentice, P.; Koulman, A.; Matthews, L.; Acerini, C.L.; Ong, K.K.; Dunger, D.B. Lipidomic analyses, breast- and formula-feeding, and growth in infants. J. Pediatrics 2015, 166, 276–281. [Google Scholar] [CrossRef] [PubMed]
- Harshfield, E.L.; Koulman, A.; Ziemek, D.; Marney, L.; Fauman, E.B.; Paul, D.S.; Stacey, D.; Rasheed, A.; Lee, J.-J.; Shah, N.; et al. An unbiased lipid phenotyping approach to study the genetic determinants of lipids and their association with coronary heart disease risk factors. J. Proteome Res. 2019, 18, 2397–2410. [Google Scholar] [CrossRef] [PubMed]
- Furse, S.; Koulman, A. The lipid and glyceride profiles of infant formula differ by manufacturer, region and date sold. Nutrients 2019, 11, 1122. [Google Scholar] [CrossRef] [PubMed]
- Chong, J.; Soufan, O.; Li, C.; Caraus, I.; Li, S.; Bourque, G.; Wishart, D.S.; Xia, J. Metaboanalyst 4.0: Towards more transparent and integrative metabolomics analysis. Nucleic Acids Res. 2018, 46, W486–W494. [Google Scholar] [CrossRef] [PubMed]
- Furse, S.; Jakubec, M.; Rise, F.; Williams, H.E.; Rees, C.E.D.; Halskau, O. Evidence that listeria innocua modulates its membrane’s stored curvature elastic stress, but not fluidity, through the cell cycle. Sci. Rep. 2017, 7, 8012. [Google Scholar] [CrossRef] [PubMed]
- Furse, S.; Wienk, H.; Boelens, R.; de Kroon, A.I.P.M.; Killian, J.A. E. Coli MG1655 modulates its phospholipid composition through the cell cycle. FEBS Lett. 2015, 589, 2726–2730. [Google Scholar] [CrossRef] [PubMed]
- Nunes, J.C.; Torres, A.G. Fatty acid and cla composition of brazilian dairy products, and contribution to daily intake of cla. J. Food Compos. Anal. 2010, 23, 782–789. [Google Scholar] [CrossRef]
- Koulman, A.; Furse, S.; Baumert, M.; Goldberg, G.; Bluck, L. Rapid profiling of triglycerides human breast milk using lesa-ftms reveals new vlcfas and differences within individuals. Rapid Commun. Mass Spectrom. 2019. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Furse, S.; Torres, A.G.; Koulman, A. Fermentation of Milk into Yoghurt and Cheese Leads to Contrasting Lipid and Glyceride Profiles. Nutrients 2019, 11, 2178. https://doi.org/10.3390/nu11092178
Furse S, Torres AG, Koulman A. Fermentation of Milk into Yoghurt and Cheese Leads to Contrasting Lipid and Glyceride Profiles. Nutrients. 2019; 11(9):2178. https://doi.org/10.3390/nu11092178
Chicago/Turabian StyleFurse, Samuel, Alexandre G. Torres, and Albert Koulman. 2019. "Fermentation of Milk into Yoghurt and Cheese Leads to Contrasting Lipid and Glyceride Profiles" Nutrients 11, no. 9: 2178. https://doi.org/10.3390/nu11092178
APA StyleFurse, S., Torres, A. G., & Koulman, A. (2019). Fermentation of Milk into Yoghurt and Cheese Leads to Contrasting Lipid and Glyceride Profiles. Nutrients, 11(9), 2178. https://doi.org/10.3390/nu11092178






