Anti-Obesity Effects of the Flower of Prunus persica in High-Fat Diet-Induced Obese Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.2. High-Performance Liquid Chromatography (HPLC) Analysis
2.3. Animals and Diets
2.4. Serum Biochemical Analysis
2.5. Real-Time Quantitative PCR Analysis
2.6. Statistical Analysis
3. Results
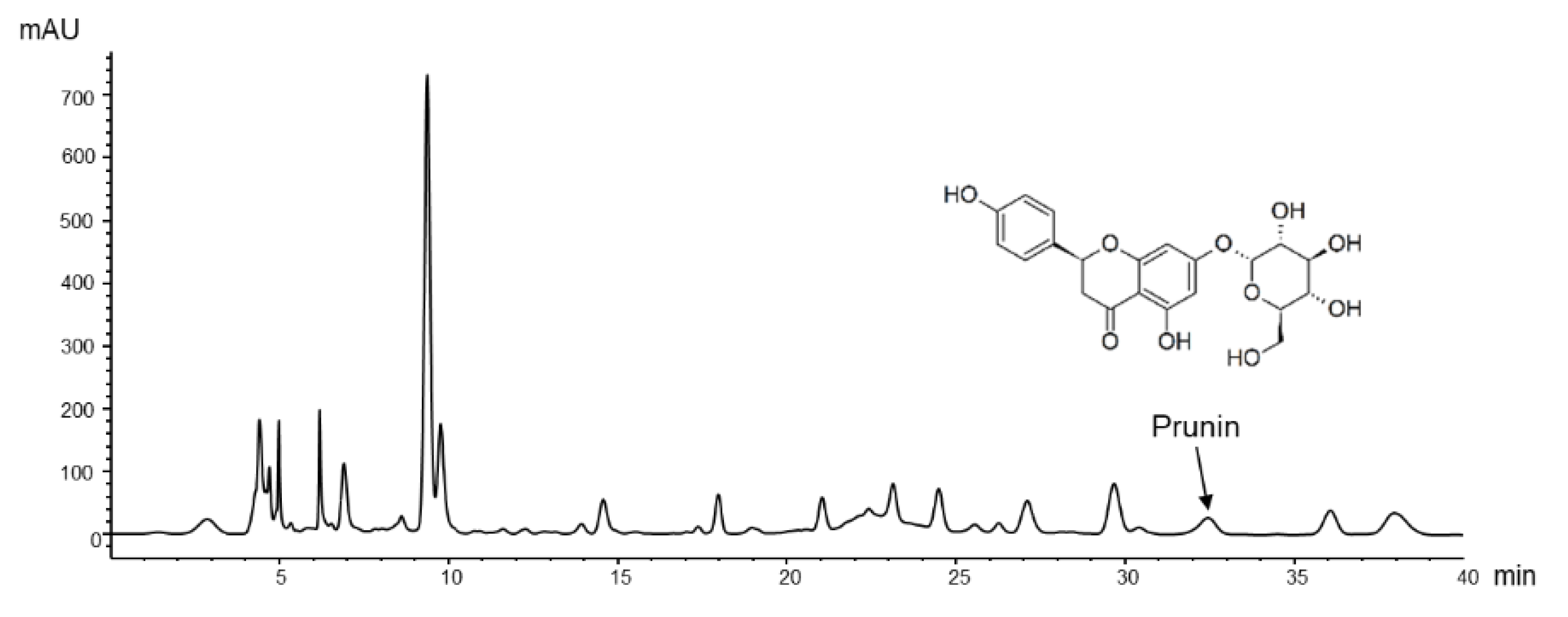
3.1. HPLC Analysis
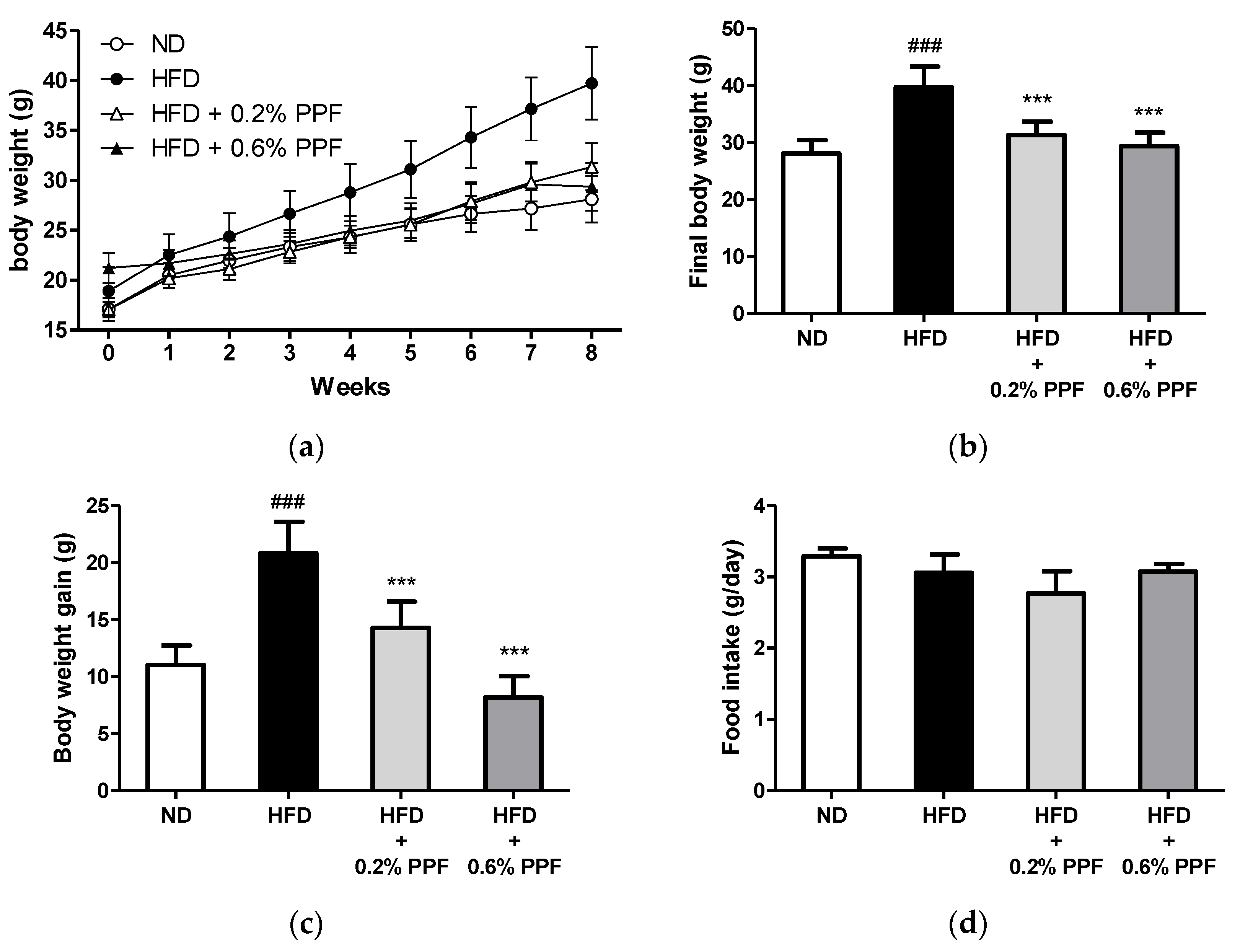
3.2. Effects of P. persica Flower on Body Weight and Food Intake
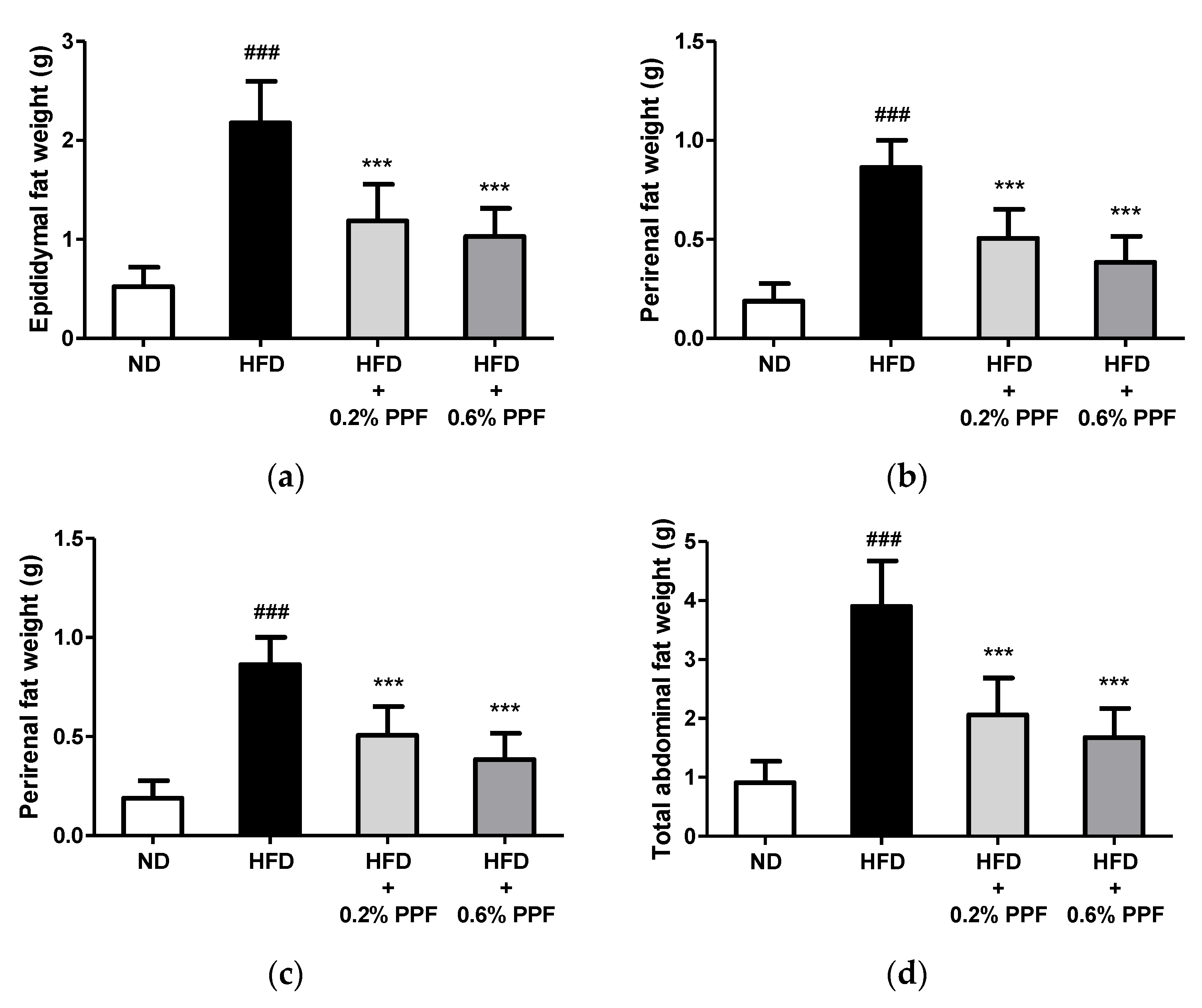
3.3. Effects of P. persica Flower on Abdominal Fat Weights
3.4. Effects of P. persica Flower on Serum Biochemical Profiles
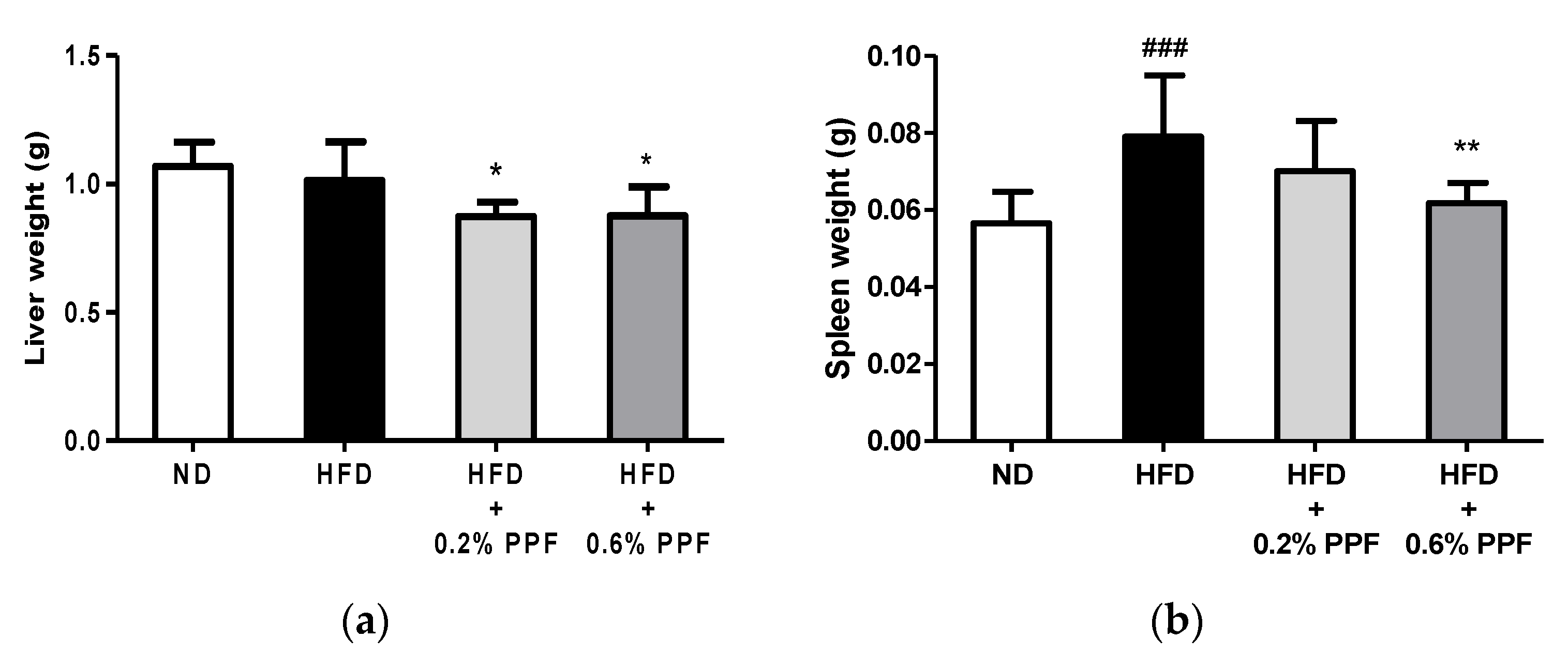
3.5. Effects of P. persica Flower on Liver and Spleen Weights
3.6. Effects of P. persica Flower on Lipogenesis-Related Gene Expression in the Liver
3.7. Effects of P. persica Flower on Fatty Acid Oxidation-Related Gene Expression in the Liver
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Longo, M.; Zatterale, F.; Naderi, J.; Parrillo, L.; Formisano, P.; Raciti, G.A.; Beguinot, F.; Miele, C. Adipose tissue dysfunction as determinant of obesity-associated metabolic complications. Int. J. Mol. Sci. 2019, 20, 2358. [Google Scholar] [CrossRef]
- Montesi, L.; El Ghoch, M.; Brodosi, L.; Calugi, S.; Marchesini, G.; Dalle Grave, R. Long-term weight loss maintenance for obesity: A multidisciplinary approach. Diabetes Metab. Syndr. Obes. 2016, 9, 37–46. [Google Scholar] [PubMed]
- Apovian, C.M.; Aronne, L.J.; Bessesen, D.H.; McDonnell, M.E.; Murad, M.H.; Pagotto, U.; Ryan, D.H.; Still, C.D. Pharmacological management of obesity: An endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2015, 100, 342–362. [Google Scholar] [CrossRef]
- Sun, N.N.; Wu, T.Y.; Chau, C.F. Natural dietary and herbal products in anti-obesity treatment. Molecules 2016, 21, 1351. [Google Scholar] [CrossRef] [PubMed]
- Nowicka, P.; Wojdylo, A.; Laskowski, P. Inhibitory potential against digestive enzymes linked to obesity and type 2 diabetes and content of bioactive compounds in 20 cultivars of the peach fruit grown in Poland. Plant Foods Hum. Nutr. 2018, 73, 314–320. [Google Scholar] [CrossRef]
- Han, W.; Xu, J.D.; Wei, F.X.; Zheng, Y.D.; Ma, J.Z.; Xu, X.D.; Wei, Z.G.; Wang, W.; Zhang, Y.C. Prokinetic activity of Prunus persica (L.) Batsch flowers extract and its possible mechanism of action in rats. BioMed Res. Int. 2015, 2015, 569853. [Google Scholar] [CrossRef]
- Kwak, C.S.; Yang, J. Prevention Effect of Prunus persica Flos extract from reactive oxygen species generation and matrix metalloproteinases production induced by UVB irradiation in human skin cells. Asian J. Beauty Cosmetol. 2016, 14, 179–190. [Google Scholar] [CrossRef]
- Kwak, C.S.; Yang, J.; Shin, C.Y.; Chung, J.H. Topical or oral treatment of peach flower extract attenuates UV-induced epidermal thickening, matrix metalloproteinase-13 expression and pro-inflammatory cytokine production in hairless mice skin. Nutr. Res. Pract. 2018, 12, 29–40. [Google Scholar] [CrossRef]
- Kim, Y.H.; Yang, H.E.; Park, B.K.; Heo, M.Y.; Jo, B.K.; Kim, H.P. The extract of the flowers of Prunus persica, a new cosmetic ingredient, protects against solar ultraviolet-induced skin damage in vivo. J. Cosmet. Sci. 2002, 53, 27–34. [Google Scholar] [PubMed]
- Murata, K.; Takahashi, K.; Nakamura, H.; Itoh, K.; Matsuda, H. Search for skin-whitening agent from Prunus plants and the molecular targets in melanogenesis pathway of active compounds. Nat. Prod. Commun. 2014, 9, 185–188. [Google Scholar] [CrossRef]
- Takagi, S.; Yamaki, M.; Masuda, K.; Kubota, M.; Minami, J. Studies on the purgative drugs. III. On the constituents of the flowers of Prunus persica Batsch. Yakugaku Zasshi 1977, 97, 109–111. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.C.; Jiao, Z.G.; Yang, W.B.; Zhang, C.L.; Liu, H.; Lv, Z.Z. Variation in phenolics, flavanoids, antioxidant and tyrosinase inhibitory activity of peach blossoms at different developmental stages. Molecules 2015, 20, 20460–20472. [Google Scholar] [CrossRef] [PubMed]
- Niu, Y.F.; Shao, Y.; Zhao, X.H.; Wen, H.X.; Tao, Y.D. RP-HPLC determination of flavonoids in several flowers. Zhongguo Zhong Yao Za Zhi 2008, 33, 2102–2104. [Google Scholar] [PubMed]
- Cui, L. Simultaneous determination of three components in peach blossom by HPLC. Chin. Trad. Patent Med. 2017, 39, 216–218. [Google Scholar]
- Cho, A.S.; Jeon, S.M.; Kim, M.J.; Yeo, J.; Seo, K.I.; Choi, M.S.; Lee, M.K. Chlorogenic acid exhibits anti-obesity property and improves lipid metabolism in high-fat diet-induced-obese mice. Food Chem. Toxicol. 2010, 48, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Ohkoshi, E.; Miyazaki, H.; Shindo, K.; Watanabe, H.; Yoshida, A.; Yajima, H. Constituents from the leaves of Nelumbo nucifera stimulate lipolysis in the white adipose tissue of mice. Planta Med. 2007, 73, 1255–1259. [Google Scholar] [CrossRef] [PubMed]
- Torres-Villarreal, D.; Camacho, A.; Castro, H.; Ortiz-Lopez, R.; de la Garza, A.L. Anti-obesity effects of kaempferol by inhibiting adipogenesis and increasing lipolysis in 3T3-L1 cells. J. Physiol. Biochem. 2019, 75, 83–88. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Wang, Y.; Jiang, W.W.; Luo, X.F.; Dai, T.Y.; Peng, L.; Song, S.; Li, L.F.; Tao, L.; Shi, C.Y.; et al. Moringa oleifera leaf petroleum ether extract inhibits lipogenesis by activating the AMPK signaling pathway. Front. Pharmacol. 2018, 9, 1447. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Kim, B.; Oh, M.J.; Yoon, J.; Kim, H.Y.; Lee, K.J.; Lee, J.D.; Choi, K.Y. Persicaria hydropiper (L.) spach and its flavonoid components, isoquercitrin and isorhamnetin, activate the Wnt/β-catenin pathway and inhibit adipocyte differentiation of 3T3-L1 cells. Phytother. Res. 2011, 25, 1629–1635. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Wei, G.; You, Y.; Huang, Y.; Lee, H.J.; Dong, M.; Lin, J.; Hu, T.; Zhang, H.; Zhang, C.; et al. Rutin ameliorates obesity through brown fat activation. FASEB J. 2017, 31, 333–345. [Google Scholar] [CrossRef]
- Shirosaki, M.; Goto, Y.; Hirooka, S.; Masuda, H.; Koyama, T.; Yazawa, K. Peach leaf contains multiflorin A as a potent inhibitor of glucose absorption in the small intestine in mice. Biol. Pharm. Bull. 2012, 35, 1264–1268. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Verma, N.; Amresh, G.; Sahu, P.K.; Mishra, N.; Rao Ch, V.; Singh, A.P. Pharmacological evaluation of hyperin for antihyperglycemic activity and effect on lipid profile in diabetic rats. Indian J. Exp. Biol. 2013, 51, 65–72. [Google Scholar] [PubMed]
- Erez, A.; Lavee, S. Prunin identification, biological activity and quantitative change in comparison to naringenin in dormant peach buds. Plant Physiol. 1969, 44, 342–346. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.; Stubbins, R.E.; Smith, R.R.; Harvey, A.E.; Nunez, N.P. Differential susceptibility to obesity between male, female and ovariectomized female mice. Nutr. J. 2009, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Chusyd, D.E.; Wang, D.; Huffman, D.M.; Nagy, T.R. Relationships between rodent white adipose fat pads and human white adipose fat depots. Front. Nutr. 2016, 3, 10. [Google Scholar] [CrossRef] [PubMed]
- Janochova, K.; Haluzik, M.; Buzga, M. Visceral Fat and Insulin Resistance—What We Know? Biomed. Pap. Med. Fac. Univ. Palacky Olomouc Czech Repub. 2019, 163, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Item, F.; Konrad, D. Visceral fat and metabolic inflammation: The portal theory revisited. Obes. Rev. 2012, 13, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Catalano, K.J.; Stefanovski, D.; Bergman, R.N. Critical role of the mesenteric depot versus other intra-abdominal adipose depots in the development of insulin resistance in young rats. Diabetes 2010, 59, 1416–1423. [Google Scholar] [CrossRef] [PubMed]
- Hoffler, U.; Hobbie, K.; Wilson, R.; Bai, R.; Rahman, A.; Malarkey, D.; Travlos, G.; Ghanayem, B.I. Diet-induced obesity is associated with hyperleptinemia, hyperinsulinemia, hepatic steatosis, and glomerulopathy in C57BL/6J mice. Endocrine 2009, 36, 311–325. [Google Scholar] [CrossRef] [PubMed]
- Eisinger, K.; Liebisch, G.; Schmitz, G.; Aslanidis, C.; Krautbauer, S.; Buechler, C. Lipidomic analysis of serum from high fat diet induced obese mice. Int. J. Mol. Sci. 2014, 15, 2991–3002. [Google Scholar] [CrossRef] [PubMed]
- Nishikawa, S.; Yasoshima, A.; Doi, K.; Nakayama, H.; Uetsuka, K. Involvement of sex, strain and age factors in high fat diet-induced obesity in C57BL/6J and BALB/cA mice. Exp. Anim. 2007, 56, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, A.; Terauchi, Y. Lessons from mouse models of high-fat diet-induced NAFLD. Int. J. Mol. Sci. 2013, 14, 21240–21257. [Google Scholar] [CrossRef] [PubMed]
- Schindhelm, R.K.; Diamant, M.; Dekker, J.M.; Tushuizen, M.E.; Teerlink, T.; Heine, R.J. Alanine aminotransferase as a marker of non-alcoholic fatty liver disease in relation to type 2 diabetes mellitus and cardiovascular disease. Diabetes Metab. Res. Rev. 2006, 22, 437–443. [Google Scholar] [CrossRef] [PubMed]
- Altunkaynak, B.Z.; Ozbek, E.; Altunkaynak, M.E. A stereological and histological analysis of spleen on obese female rats, fed with high fat diet. Saudi Med. J. 2007, 28, 353–357. [Google Scholar] [PubMed]
- Gotoh, K.; Inoue, M.; Masaki, T.; Chiba, S.; Shimasaki, T.; Ando, H.; Fujiwara, K.; Katsuragi, I.; Kakuma, T.; Seike, M.; et al. A novel anti-inflammatory role for spleen-derived interleukin-10 in obesity-induced inflammation in white adipose tissue and liver. Diabetes 2012, 61, 1994–2003. [Google Scholar] [CrossRef] [PubMed]
- Magnuson, A.M.; Regan, D.P.; Fouts, J.K.; Booth, A.D.; Dow, S.W.; Foster, M.T. Diet-induced obesity causes visceral, but not subcutaneous, lymph node hyperplasia via increases in specific immune cell populations. Cell Prolif. 2017, 50, e12365. [Google Scholar] [CrossRef] [PubMed]
- Perla, F.M.; Prelati, M.; Lavorato, M.; Visicchio, D.; Anania, C. The role of lipid and lipoprotein metabolism in non-alcoholic fatty liver disease. Children 2017, 4, 46. [Google Scholar] [CrossRef] [PubMed]
- Oosterveer, M.H.; van Dijk, T.H.; Tietge, U.J.; Boer, T.; Havinga, R.; Stellaard, F.; Groen, A.K.; Kuipers, F.; Reijngoud, D.J. High fat feeding induces hepatic fatty acid elongation in mice. PLoS ONE 2009, 4, e6066. [Google Scholar] [CrossRef]
- Li, H.; Yoo, W.; Park, H.M.; Lim, S.Y.; Shin, D.H.; Kim, S.; Park, H.Y.; Jeong, T.S. Arazyme suppresses hepatic steatosis and steatohepatitis in diet-induced non-alcoholic fatty liver disease-like mouse model. Int. J. Mol. Sci. 2019, 20, 2325. [Google Scholar] [CrossRef] [PubMed]
- Musso, G.; Cassader, M.; Paschetta, E.; Gambino, R. Bioactive lipid species and metabolic pathways in progression and resolution of nonalcoholic steatohepatitis. Gastroenterology 2018, 155, 282–302. [Google Scholar] [CrossRef] [PubMed]
- Soulard, P.; McLaughlin, M.; Stevens, J.; Connolly, B.; Coli, R.; Wang, L.; Moore, J.; Kuo, M.S.; LaMarr, W.A.; Ozbal, C.C.; et al. Development of a high-throughput screening assay for stearoyl-CoA desaturase using rat liver microsomes, deuterium labeled stearoyl-CoA and mass spectrometry. Anal. Chim. Acta 2008, 627, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Ntambi, J.M.; Miyazaki, M.; Stoehr, J.P.; Lan, H.; Kendziorski, C.M.; Yandell, B.S.; Song, Y.; Cohen, P.; Friedman, J.M.; Attie, A.D. Loss of stearoyl-CoA desaturase-1 function protects mice against adiposity. Proc. Natl. Acad. Sci. USA 2002, 99, 11482–11486. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, M.; Dobrzyn, A.; Sampath, H.; Lee, S.H.; Man, W.C.; Chu, K.; Peters, J.M.; Gonzalez, F.J.; Ntambi, J.M. Reduced adiposity and liver steatosis by stearoyl-CoA desaturase deficiency are independent of peroxisome proliferator-activated receptor-alpha. J. Biol. Chem. 2004, 279, 35017–35024. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, L.; Ntambi, J. Stearoyl-CoA desaturase-2 deficiency protects mice against high fat diet-induced adiposity. FASEB J. 2014, 28, 605. [Google Scholar]
- Dorn, C.; Riener, M.O.; Kirovski, G.; Saugspier, M.; Steib, K.; Weiss, T.S.; Gabele, E.; Kristiansen, G.; Hartmann, A.; Hellerbrand, C. Expression of fatty acid synthase in nonalcoholic fatty liver disease. Int. J. Clin. Exp. Pathol. 2010, 3, 505–514. [Google Scholar]
- Delgado, T.C.; Pinheiro, D.; Caldeira, M.; Castro, M.M.; Geraldes, C.F.; Lopez-Larrubia, P.; Cerdan, S.; Jones, J.G. Sources of hepatic triglyceride accumulation during high-fat feeding in the healthy rat. NMR Biomed. 2009, 22, 310–317. [Google Scholar] [CrossRef]
- Pawlak, M.; Lefebvre, P.; Staels, B. Molecular mechanism of PPARα action and its impact on lipid metabolism, inflammation and fibrosis in non-alcoholic fatty liver disease. J. Hepatol. 2015, 62, 720–733. [Google Scholar] [CrossRef]
- Qu, Q.; Zeng, F.; Liu, X.; Wang, Q.J.; Deng, F. Fatty acid oxidation and carnitine palmitoyltransferase I: Emerging therapeutic targets in cancer. Cell Death Dis. 2016, 7, e2226. [Google Scholar] [CrossRef]
- Dobrzyn, P.; Dobrzyn, A.; Miyazaki, M.; Cohen, P.; Asilmaz, E.; Hardie, D.G.; Friedman, J.M.; Ntambi, J.M. Stearoyl-CoA desaturase 1 deficiency increases fatty acid oxidation by activating AMP-activated protein kinase in liver. Proc. Natl. Acad. Sci. USA 2004, 101, 6409–6414. [Google Scholar] [CrossRef]
- Bhandarkar, N.S.; Brown, L.; Panchal, S.K. Chlorogenic acid attenuates high-carbohydrate, high-fat diet-induced cardiovascular, liver, and metabolic changes in rats. Nutr. Res. 2019, 62, 78–88. [Google Scholar] [CrossRef]
- Calderon-Montano, J.M.; Burgos-Moron, E.; Perez-Guerrero, C.; Lopez-Lazaro, M. A review on the dietary flavonoid kaempferol. Mini Rev. Med. Chem. 2011, 11, 298–344. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Yao, J.; Han, C.; Yang, J.; Chaudhry, M.T.; Wang, S.; Liu, H.; Yin, Y. Quercetin, inflammation and immunity. Nutrients 2016, 8, 167. [Google Scholar] [CrossRef] [PubMed]
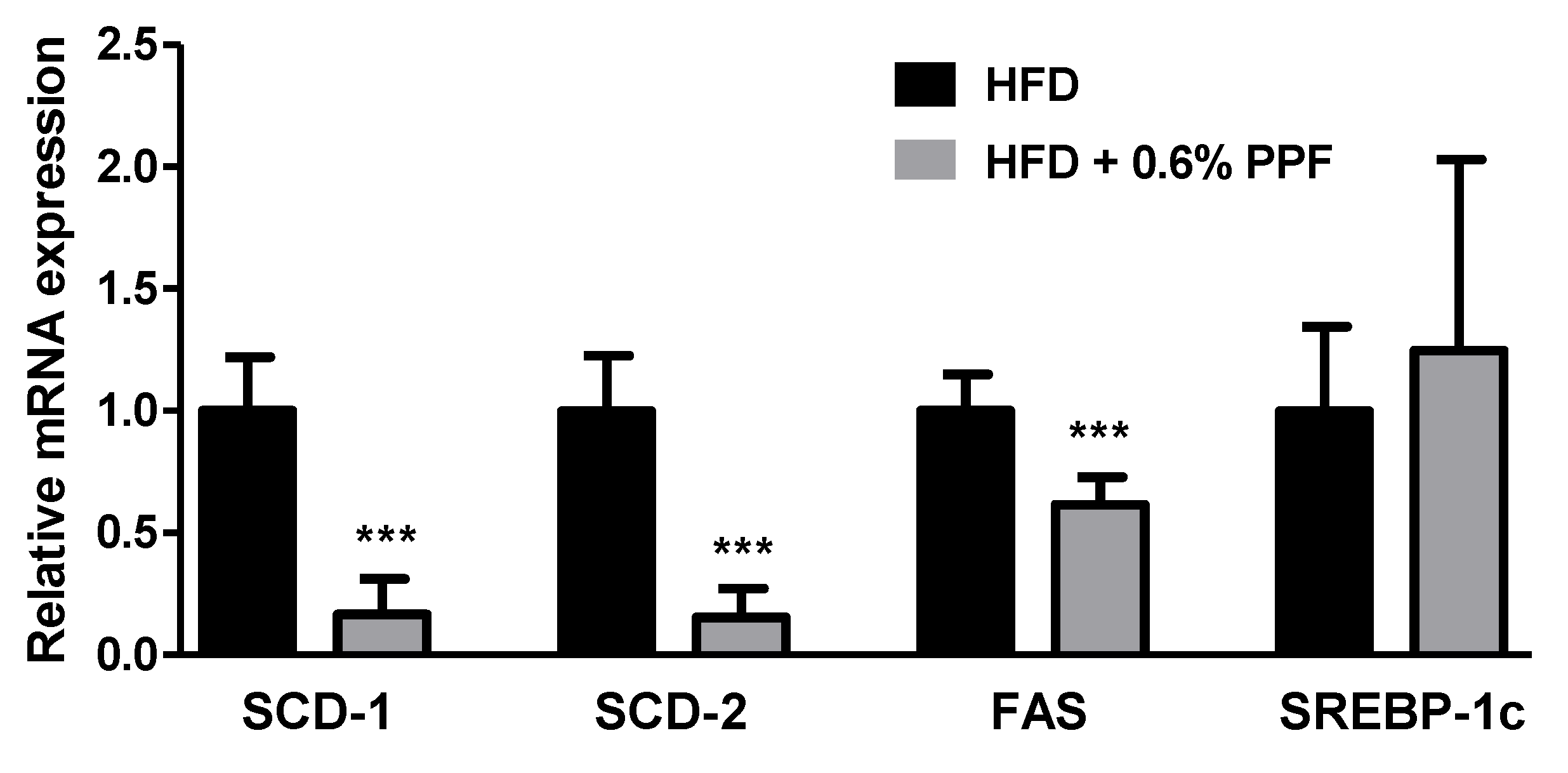
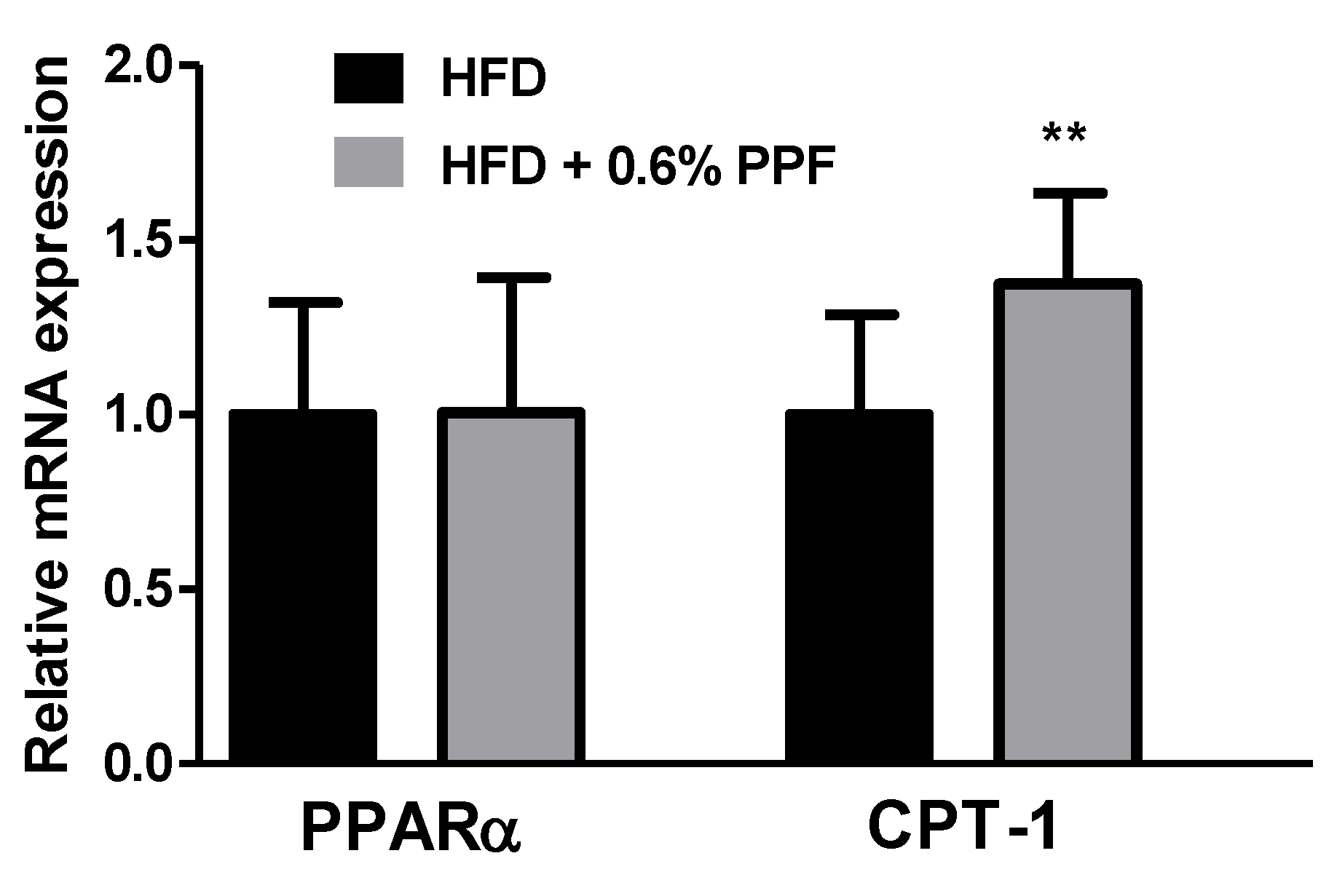
| Groups | Glucose (mg/dL) | TC (mg/dL) | Triglyceride (mg/dL) | ALT (U/L) | AST (U/L) |
|---|---|---|---|---|---|
| ND | 179.3 ± 6.1 1 | 77.5 ± 6.7 | 68.5 ± 13.0 | 38.6 ± 13.6 | 35.6 ± 8.9 |
| HFD | 239.6 ± 27.1 ### | 106.8 ± 18.2 ### | 71.8 ± 16.4 | 61.8 ± 23.1 ### | 61.4 ± 13.8 ### |
| HFD + 0.2% PPF | 178.9 ± 38.8 *** | 105.9 ± 13.8 | 87.0 ± 29.7 | 38.0 ± 8.9 ** | 44.8 ± 8.1 ** |
| HFD + 0.6% PPF | 136.6 ± 31.9 *** | 101.8 ± 17.1 | 84.3 ± 26.7 | 36.2 ± 10.3 *** | 49.0 ± 14.0 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, J.; Kim, Y.-S.; Kim, L.; Park, H.J.; Lee, D.; Kim, H. Anti-Obesity Effects of the Flower of Prunus persica in High-Fat Diet-Induced Obese Mice. Nutrients 2019, 11, 2176. https://doi.org/10.3390/nu11092176
Song J, Kim Y-S, Kim L, Park HJ, Lee D, Kim H. Anti-Obesity Effects of the Flower of Prunus persica in High-Fat Diet-Induced Obese Mice. Nutrients. 2019; 11(9):2176. https://doi.org/10.3390/nu11092176
Chicago/Turabian StyleSong, Jungbin, Young-Sik Kim, Linae Kim, Hyo Jin Park, Donghun Lee, and Hocheol Kim. 2019. "Anti-Obesity Effects of the Flower of Prunus persica in High-Fat Diet-Induced Obese Mice" Nutrients 11, no. 9: 2176. https://doi.org/10.3390/nu11092176
APA StyleSong, J., Kim, Y.-S., Kim, L., Park, H. J., Lee, D., & Kim, H. (2019). Anti-Obesity Effects of the Flower of Prunus persica in High-Fat Diet-Induced Obese Mice. Nutrients, 11(9), 2176. https://doi.org/10.3390/nu11092176






