The Dietary Management of Patients with Irritable Bowel Syndrome: A Narrative Review of the Existing and Emerging Evidence
Abstract
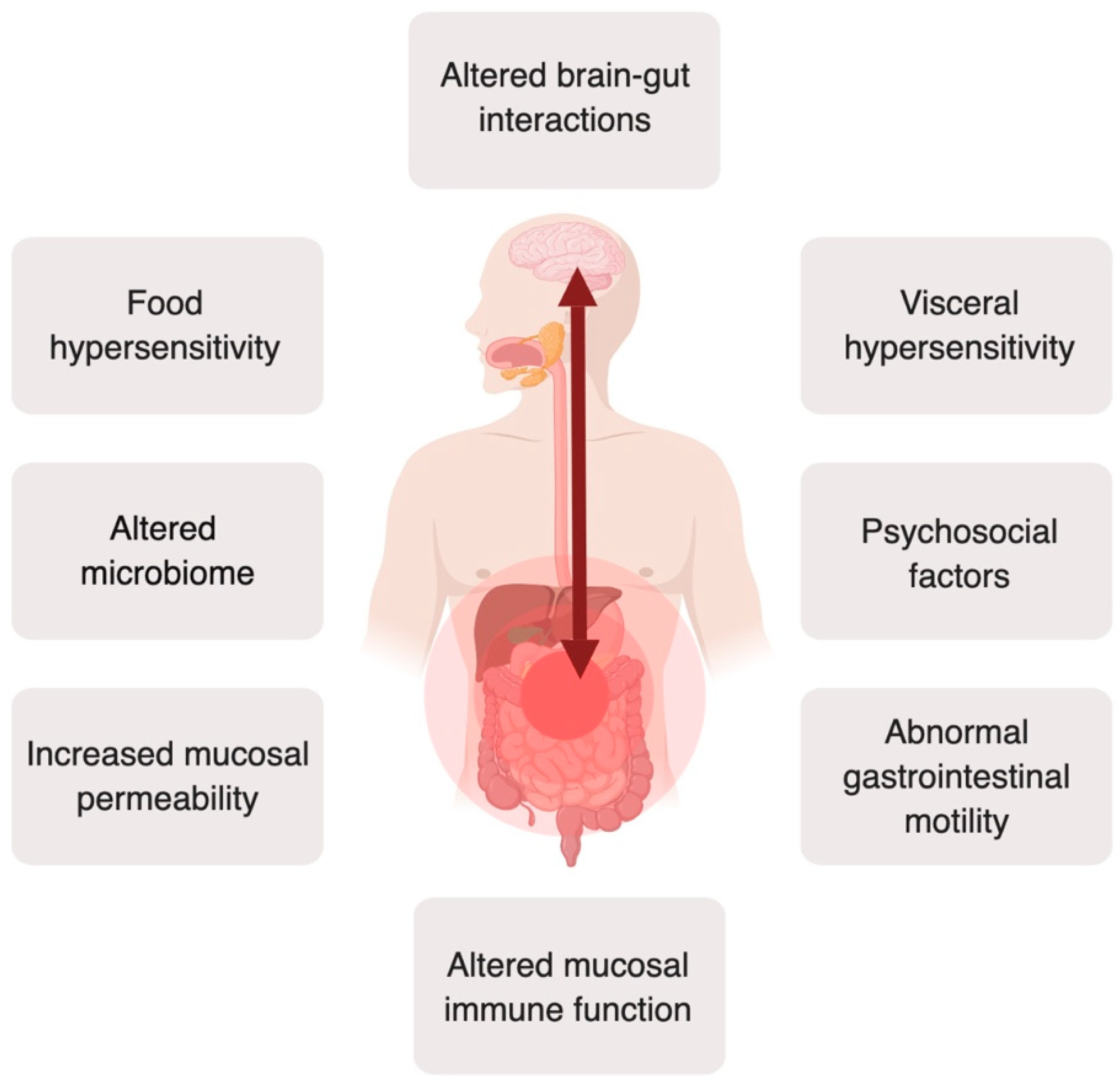
1. Introduction
2. Methodology
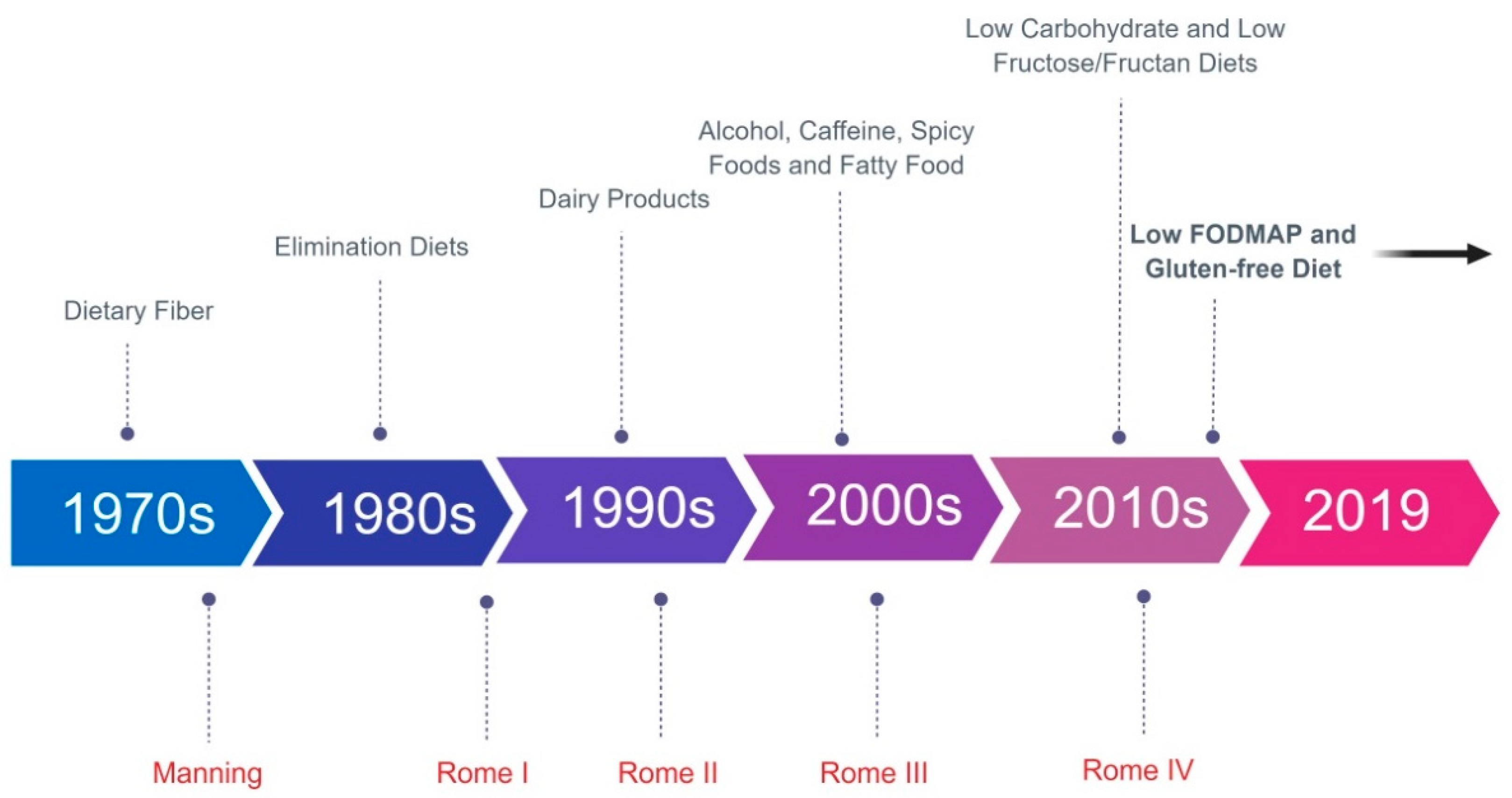
3. History of Dietary Management of IBS: The Bran Era and Exclusion Diets
4. IgG Elimination Diet
5. Dietary Fiber
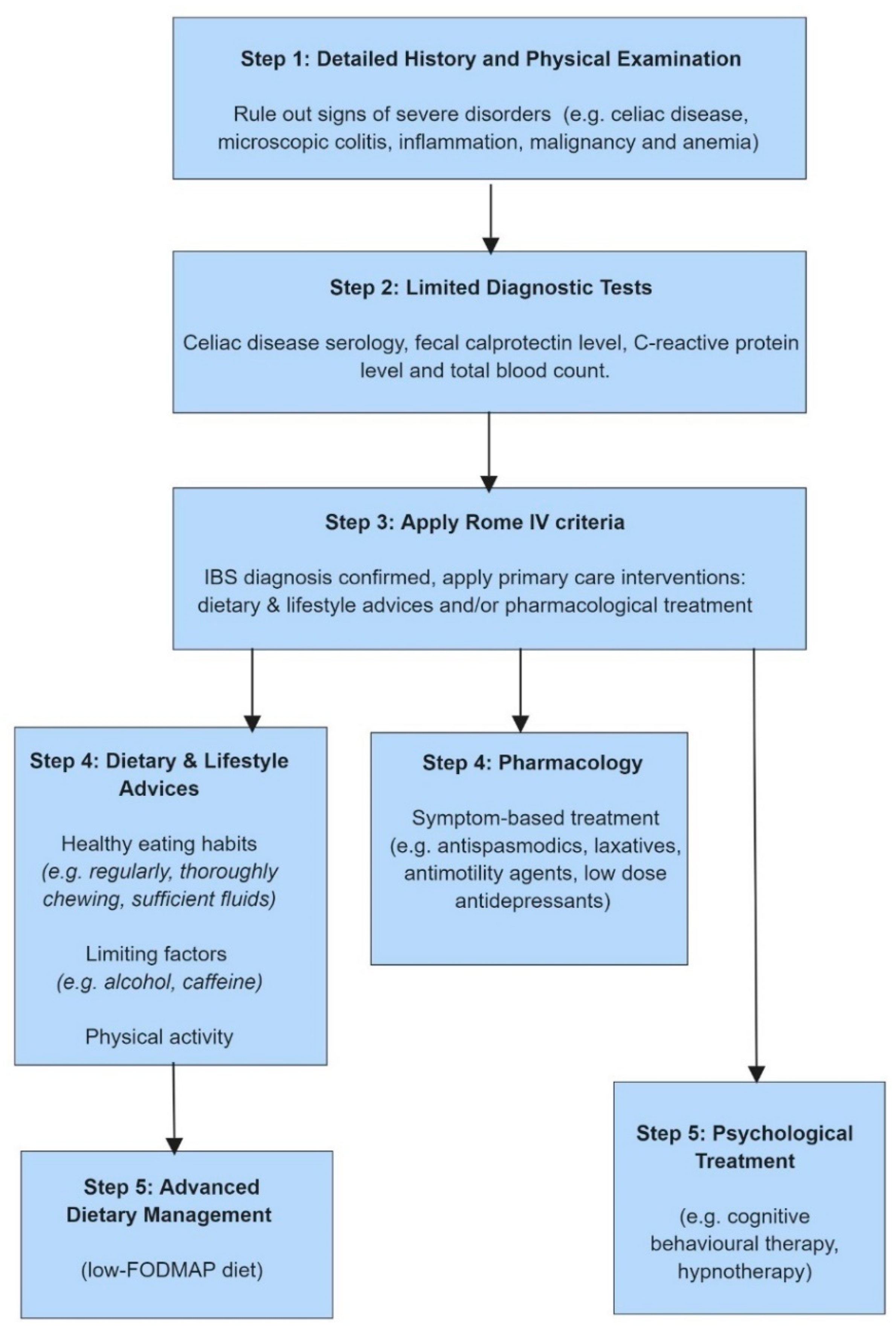
6. The NICE Guidelines
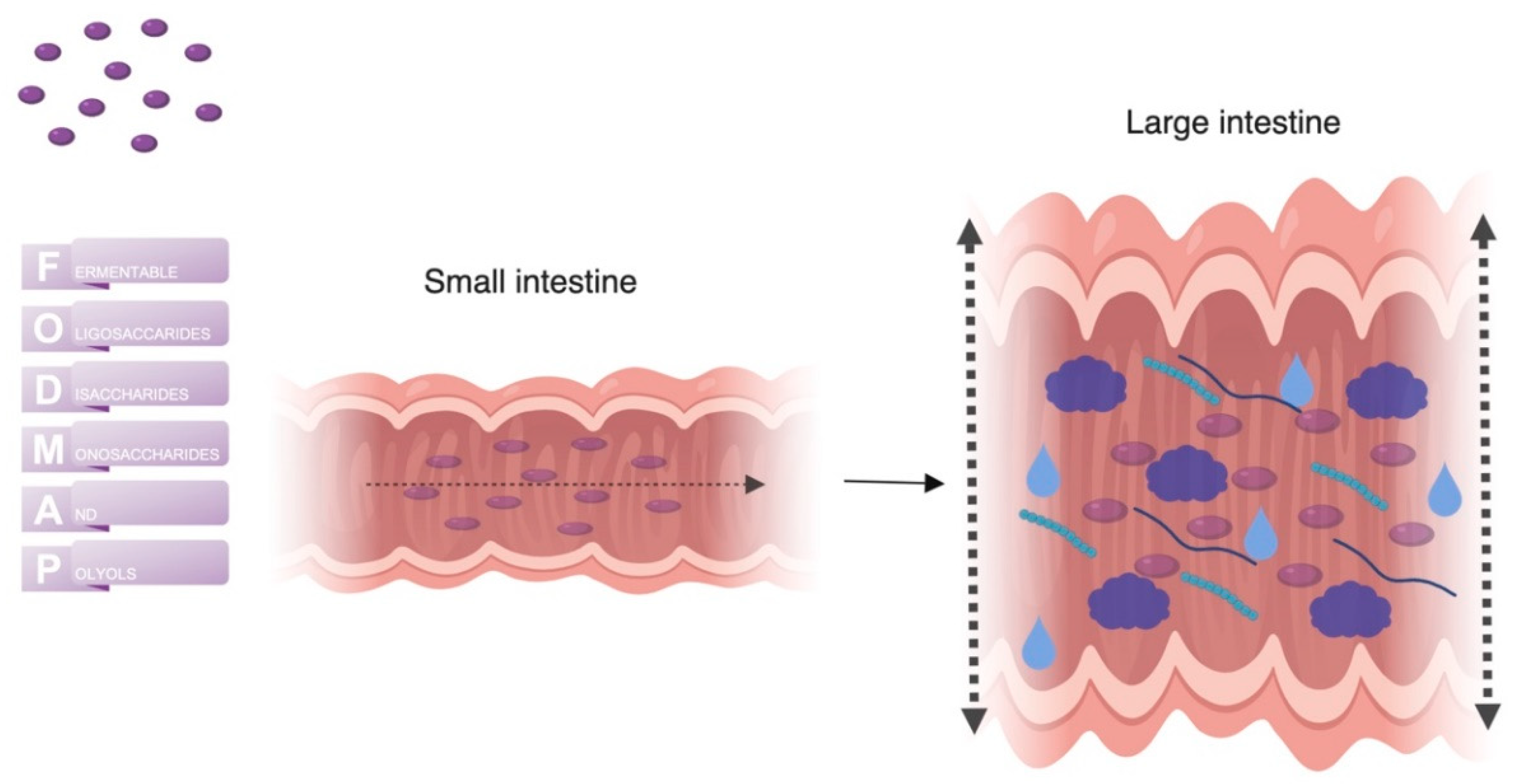
7. FODMAPs
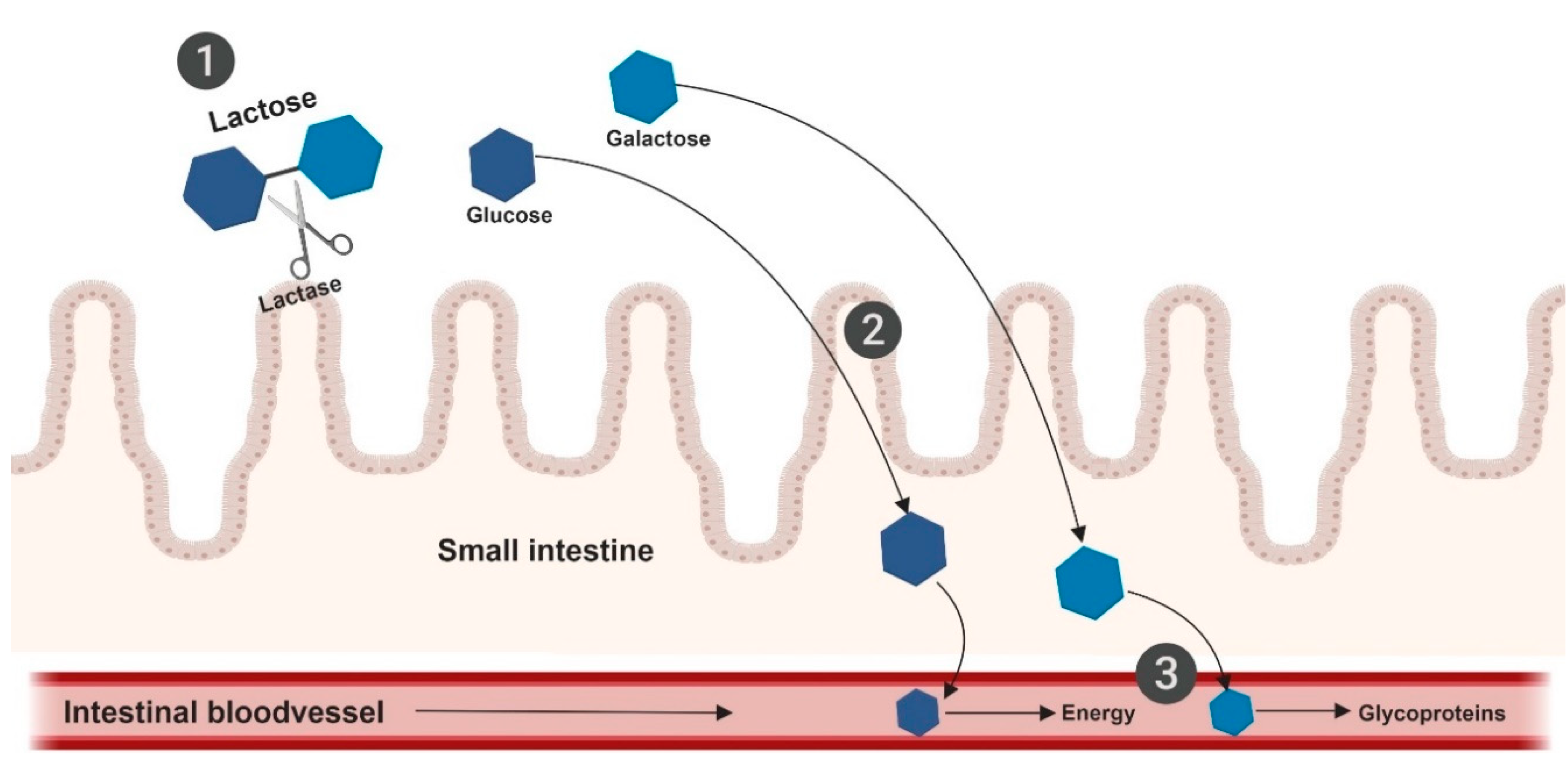
8. Lactose
9. Low-Fructose/Fructan Diet
10. Low-Carbohydrate Diet
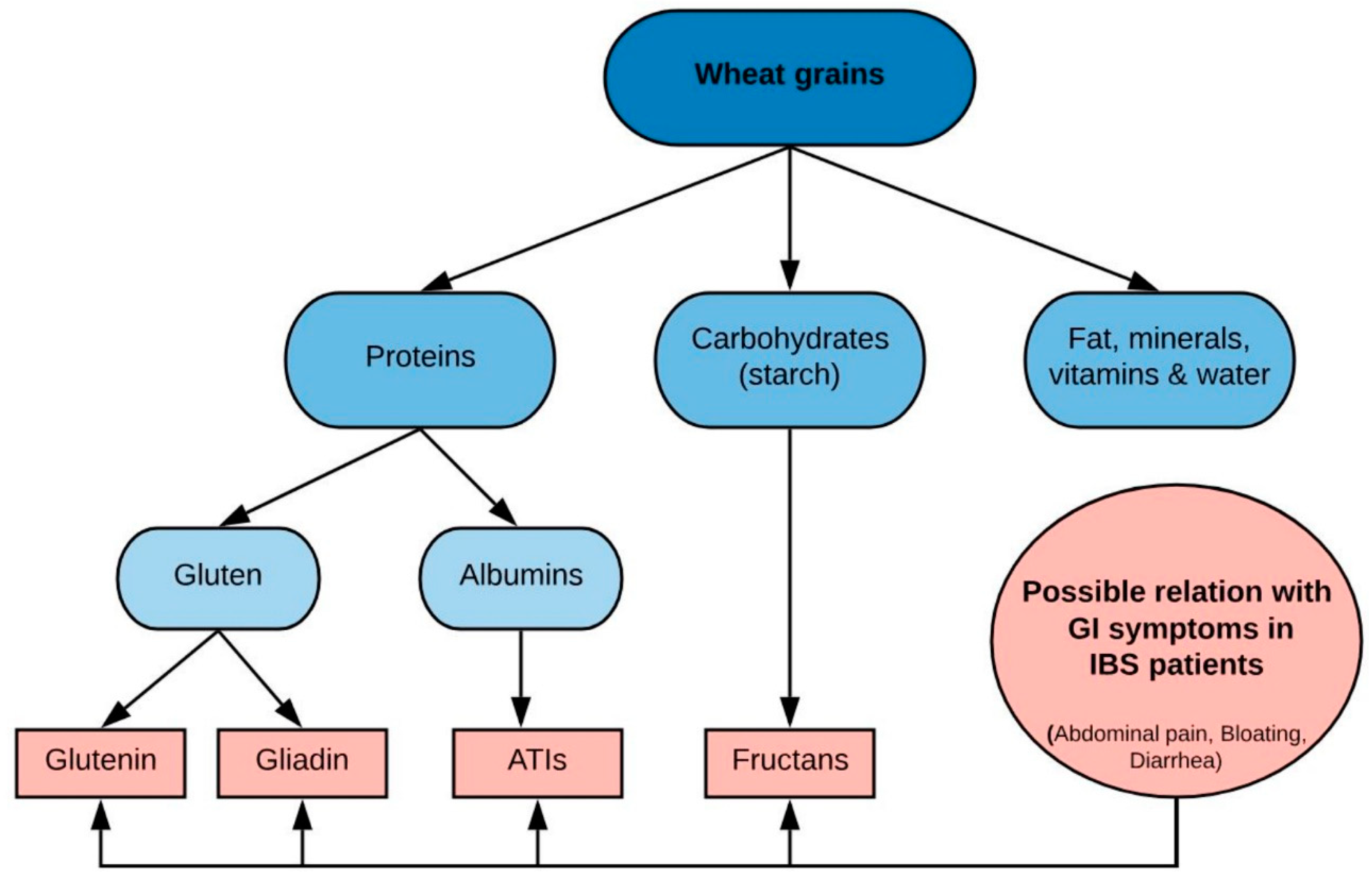
11. Gluten
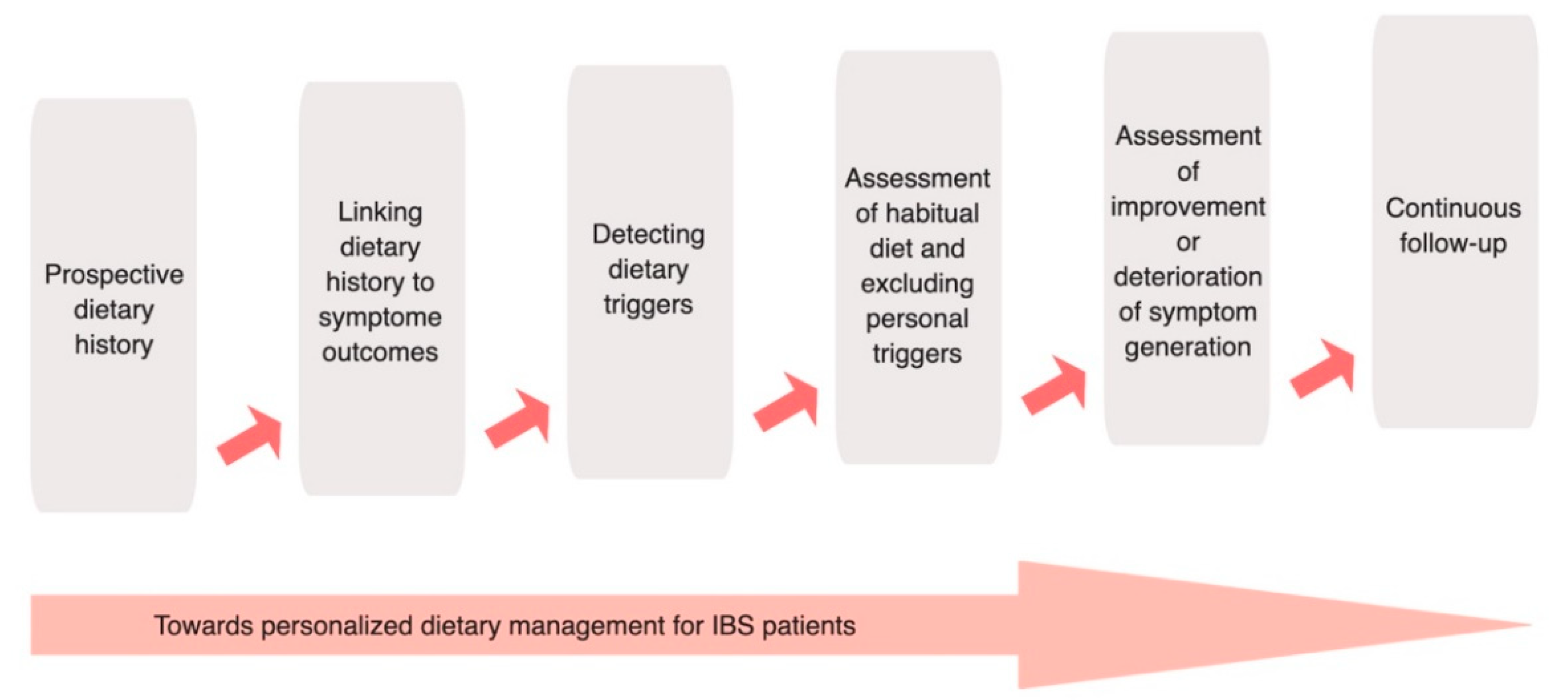
12. Future Directions and Recommendations
Author Contributions
Funding
Conflicts of Interest
References
- Lovell, R.; Ford, A. Global prevalence of and risk factors for irritable bowel syndrome: A meta-analysis. Clin. Gastroenterol. Hepatol. 2012, 10, 712–721. [Google Scholar] [CrossRef] [PubMed]
- Sperber, A.D.; Dumitrascu, D.; Fucudo, S.; Gerson, C.; Ghoshal, U.C.; Gwee, K.A.; Hungin, A.P.S.; Kang, J.Y.; Minhu, C.; Schmulson, M.; et al. The global prevalence of IBS in adults remains elusive due to the heterogeneity of studies: A Rome Foundation working team literature review. Gut 2017, 66, 1075–1082. [Google Scholar] [CrossRef] [PubMed]
- Lacy, B.E.; Mearin, F.; Chang, L.; Chey, W.D.; Lembo, A.J.; Simrén, M.; Spiller, R. Bowel disorders. Gastroenterology 2016, 150, 1393–1407. [Google Scholar] [CrossRef]
- Longstreth, G.F.; Thompson, W.G.; Chey, W.D.; Houghton, L.A.; Mearin, F.; Spiller, R.C. Functional Bowel Disorders. Gastroenterology 2006, 130, 1480–1491. [Google Scholar] [CrossRef]
- Enck, P.; Aziz, Q.; Barbara, G.; Farmer, A.D.; Fukudo, S.; Mayer, E.A.; Niesler, B.; Quigley, E.M.; Rajilić-Stojanović, M.; Schemann, M.; et al. Irritable bowel syndrome. Nat. Rev. Dis. Prim. 2016, 2, 16014. [Google Scholar] [CrossRef] [PubMed]
- Tillisch, K.; Labus, J.S. Advances in imaging the BrainGut axis: Functional gastrointestinal disorders. Gastroenterology 2011, 140, 407–411. [Google Scholar] [CrossRef]
- Barbara, G.; Zecchi, L.; Barbaro, R.; Cremon, C.; Bellacosa, L.; Marcellini, M.; De Giorgio, R.; Corinaldesi, R.; Stanghellini, G. Mucosal permeability and immune activation as potential therapeutic targets of probiotics in irritable bowel syndrome. J. Clin. Gastroenterol. 2012, 46, S52–S55. [Google Scholar] [CrossRef]
- Tap, J.; Derrien, M.; Törnblom, H.; Brazeilles, R.; Cools-Portier, S.; Doré, J.; Störsrud, S.; Le Nevé, B.; Öhman, L.; Simrén, M. Identification of an Intestinal Microbiota Signature Associated With Severity of Irritable Bowel Syndrome. Gastroenterology 2017, 152, 111–123. [Google Scholar] [CrossRef]
- Posserud, I.; Strid, H.; Störsrud, S.; Törnblom, H.; Svenson, U.; Tack, J.; Van Oudenhove, L.; Simrén, M. Symptom pattern following a meal challenge test in patients with irritable bowel syndrome and healthy controls. United Eur. Gastroenterol. J. 2013, 1, 358–367. [Google Scholar] [CrossRef]
- Öhman, L.; Simrén, M. New insights into the pathogenesis and pathophysiology of irritable bowel syndrome. Dig. Liver Dis. 2007, 39, 201–215. [Google Scholar] [CrossRef]
- Simrén, M.; Öhman, L. Pathogenesis of IBS: Role of inflammation, immunity and neuroimmune interactions. Nat. Rev. Gastroenterol. Hepatol. 2010, 7, 163–173. [Google Scholar] [CrossRef]
- Simrén, M.; Törnblom, H.; Palsson, O.S.; Whitehead, W. Management of the multiple symptoms of irritable bowel syndrome. Lancet Gastroenterol. Hepatol. 2017, 2, 112–122. [Google Scholar] [CrossRef]
- Böhn, L.; Störsrud, S.; Törnblom, H.; Bengtsson, U.; Simrén, M. Self-reported food-related gastrointestinal symptoms in IBS are common and associated with more severe symptoms and reduced quality of life. Am. J. Gastroenterol. 2013, 108, 634–641. [Google Scholar] [CrossRef] [PubMed]
- Simrén, M.; Månsson, A.; Langkilde, A.M.; Svedlund, J.; Abrahamsson, H.; Bengtsson, U.; Björnsson, E.S. Food-related gastrointestinal symptoms in the irritable bowel syndrome. Digestion 2001, 63, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Le Nevé, B.; Brazeilles, R.; Derrien, M.; Tap, J.; Guyonnet, D.; Öhman, L.; Törnblom, H.; Simrén, M. Lactulose Challenge Determines Visceral Sensitivity and Severity of Symptoms in Patients With Irritable Bowel Syndrome. Clin. Gastroenterol. Hepatol. 2016, 14, e1–e3. [Google Scholar] [CrossRef]
- Lenhart, A.; Ferch, C.; Shaw, M.; Chey, W.D. Use of Dietary Management in Irritable Bowel Syndrome: Results of a Survey of over 1500 U.S. Gastroenterologists. J. Neurogastroenterol. Motil. 2018, 24, 437–451. [Google Scholar] [CrossRef]
- Monsbakken, K.W.; Vandvik, P.O.; Farup, P.G. Perceived food intolerance in subjects with irritable bowel syndrome-Etiology, prevalence and consequences. Eur. J. Clin. Nutr. 2006, 60, 667–672. [Google Scholar] [CrossRef]
- Jones, V.A.; McLaughlan, P.; Shorthouse, M.; Workman, E.; Hunter, J.O. Food intolerance: A major factor in the pathogenesis of irritable bowel syndrome. Lancet 1982, 2, 1115–1117. [Google Scholar] [CrossRef]
- Bentley, S.J.; Pearson, D.J.; Rix, K.J. Food hypersensitivity in irritable bowel syndrome. Lancet 1983, 2, 295–297. [Google Scholar] [CrossRef]
- Suri, Y.P. The bran wagon. Lancet 1987, 2, 42–43. [Google Scholar] [CrossRef]
- Manning, A.P.; Harvey, R.F.; Heaton, K.W.; Uglow, P. Wheat fibre and irritable bowel syndrome. A Controlled Trial. Lancet 1977, 2, 417–418. [Google Scholar] [CrossRef]
- Lambert, J.P.; Brunt, P.W.; Mowat, N.A.; Khin, C.C.; Lai, C.K.; Morrison, V.; Dickerson, J.W.; Eastwood, M.A. The value of prescribed high-fiber diets for the treatment of the irritable-bowel-syndrome. Eur. J. Clin. Nutr. 1991, 45, 601–609. [Google Scholar] [PubMed]
- Arffmann, S.; Andersen, J.R.; Hegnhøj, J.; Schaffalitzky de Muckadell, O.B.; Mogensen, N.B.; Krag, E. The effect of coarse wheat bran in the irritable bowel syndrome. A double-blind Cross-over study. Scand. J. Gastroenterol. 1985, 20, 295–298. [Google Scholar] [CrossRef] [PubMed]
- Lewis, M.J.V.; Whorwell, P.J. Bran: May irritate irritable bowel. Nutrition 1998, 14, 470–471. [Google Scholar] [PubMed]
- Nanda, R.; James, R.; Smith, H.; Dudley, C.R.; Jewell, D.P. Food intolerances and irritable bowel syndrome. Gut 1989, 30, 1099–1104. [Google Scholar] [CrossRef] [PubMed]
- McKee, A.M.; Prior, A.; Whorwell, P.J. Exclusion diets in irritable bowel syndrome: Are they worthwhile? J. Clin. Gastroenterol. 1987, 9, 526–528. [Google Scholar] [CrossRef] [PubMed]
- Zwetchkenbaum, B.; Burakoff, R. The irritable bowel syndrome and food hypersensitivity. Ann. Allergy 1988, 61, 47–49. [Google Scholar]
- Friedman, G. Diet and the irritable bowel syndrome. Gastroenterol. Clin. N. Am. 1991, 20, 313–324. [Google Scholar]
- Burden, S. Dietary treatment of irritable bowel syndrome: Current evidence and guidelines for future practice. J. Hum. Nutr. Diet. 2001, 14, 231–241. [Google Scholar] [CrossRef]
- Atkinson, W.; Sheldon, T.A.; Shaath, N.; Whorwell, P.J. Food elimination based on IgG antibodies in irritable bowel syndrome: A randomised controlled trial. Gut 2004, 53, 1459–1464. [Google Scholar] [CrossRef]
- Zar, S.; Mincher, L.; Benson, M.J.; Kumar, D. Food-specific IgG4 antibody-guided exclusion diet improves symptoms and rectal compliance in irritable bowel syndrome. Scand. J. Gastroenterol. 2005, 40, 800–807. [Google Scholar] [CrossRef] [PubMed]
- Drisko, J.; Bischoff, B.; Hall, M.; McCallum, R. Treating Irritable Bowel Syndrome with a Food Elimination Diet Followed by Food Challenge and Probiotics. J. Am. Coll. Nutr. 2006, 25, 514–522. [Google Scholar] [CrossRef] [PubMed]
- Müller, M.; Canfora, E.E.; Blaak, E.E. Gastrointestinal Transit Time, Glucose Homeostasis and Metabolic Health: Modulation by Dietary Fibers. Nutrients 2018, 10, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Francis, C.; Whorwell, P. Bran and irritable bowel syndrome: Time for reappraisal. Lancet 1994, 344, 39–40. [Google Scholar] [CrossRef]
- Bijkerk, C.J.; Muris, J.W.M.; Erus, J.A.K.; Hoes, A.W.; De Wit, N.J. Systematic review: The role of different types of fibre in the treatment of irritable bowel syndrome. Aliment. Pharmacol. Ther. 2004, 19, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Bijkerk, C.J.; De Wit, N.; Muris, J.W.M.; Whorwell, P.J.; Knottnerus, J.A.; Hoes, A.W. Soluble or insoluble fibre in irritable bowel syndrome in primary care? Randomised placebo controlled trial. BMJ 2009, 339, 613–615. [Google Scholar] [CrossRef] [PubMed]
- Moayyedi, P.; Quigley, E.M.; Lacy, B.E.; Lembo, A.J.; Saito, Y.A.; Schiller, L.R.; Soffer, E.E.; Spiegel, B.M.; Ford, A.C. The effect of fiber supplementation on irritable bowel syndrome: A systematic review and meta-analysis. Am. J. Gastroenterol. 2014, 109, 1367–1374. [Google Scholar] [CrossRef]
- McCleary, B.V.; De Vries, J.; Rader, J.; Cohen, G.; Prosky, L.; Mugford, D.C.; Champ, M.; Okuma, K. Determination of Insoluble, Soluble, and Total Dietary Fiber (CODEX Definition) by Enzymatic-Gravimetric Method and Liquid Chromatography: Collaborative Study. J. AOAC Int. 2010, 93, 221–233. [Google Scholar] [CrossRef]
- De Vries, J.; Birkett, A.; Hulshof, T.; Verbeke, K.; Gibes, K. Effects of Cereal, Fruit and Vegetable Fibers on Human Fecal Weight and Transit Time: A Comprehensive Review of Intervention Trials. Nutrients 2016, 8, 130. [Google Scholar] [CrossRef]
- Hookway, C.; Buckner, S.; Crosland, P.; Longson, D. Irritable bowel syndrome in adults in primary care: Summary of updated NICE guidance. BMJ 2015, 350, 710. [Google Scholar] [CrossRef]
- Boekema, P.J.; Samsom, M.; Van Berge Henegouwen, G.P.; Smout, A.J.P.M. Coffee and Gastrointestinal Function: Facts and Fiction. A Review. Scand. J. Gastroenterol. 1999, 34, 35–39. [Google Scholar] [CrossRef]
- Bagyánszki, M.; Krecsmarik, M.; De Winter, B.Y. Chronic alcohol consumption affects gastrointestinal motility and reduces the proportion of neuronal NOS-immunoreactive myenteric neurons in the murine jejunum. Anat. Rec. 2010, 293, 1536–1542. [Google Scholar] [CrossRef] [PubMed]
- Bhat, Y.M.; Bielefeldt, K. Capsaicin receptor (TRPV1) and non-erosive reflux disease. Eur. J. Gastroenterol. Hepatol. 2006, 18, 263–270. [Google Scholar] [CrossRef]
- Simrén, M.; Abrahamsson, H.; Björnsson, E.S. An exaggerated sensory component of the gastrocolonic response in patients with irritable bowel syndrome. Gut 2001, 48, 20–27. [Google Scholar] [CrossRef]
- Whelan, K.; Martin, L.D.; Staudacher, H.M.; Lomer, M.C.E. The low FODMAP diet in the management of irritable bowel syndrome: An evidence-based review of FODMAP restriction, reintroduction and personalisation in clinical practice. J. Hum. Nutr. Diet. 2018, 31, 239–255. [Google Scholar] [CrossRef] [PubMed]
- Ong, D.K.; Mitchell, S.B.; Barrett, J.S.; Shepherd, S.J.; Irving, P.M.; Biesiekierski, J.R.; Smith, S.; Gibson, P.R.; Muir, J.G. Manipulation of dietary short chain carbohydrates alters the pattern of gas production and genesis of symptoms in irritable bowel syndrome. J. Gastroenterol. Hepatol. 2010, 25, 1366–1373. [Google Scholar] [CrossRef]
- Barrett, J.S.; Gearry, R.B.; Muir, J.G.; Irving, P.M.; Rose, R.; Rosella, O.; Haines, M.L.; Shepherd, S.J.; Gibson, P.R. Dietary poorly absorbed, short-chain carbohydrates increase delivery of water and fermentable substrates to the proximal colon. Aliment. Pharmacol. Ther. 2010, 31, 874–882. [Google Scholar] [CrossRef]
- Shepherd, S.J.; Lomer, M.C.E.; Gibson, P.R. Short-chain carbohydrates and functional gastrointestinal disorders. Am. J. Gastroenterol. 2013, 108, 707–717. [Google Scholar] [CrossRef]
- Murray, K.; Wilkinson-Smith, V.; Hoad, C.; Costigan, C.; Cox, E.; Lam, C.; Marciani, L.; Gowland, P.; Spiller, R.C. Differential effects of FODMAPs (Fermentable Oligo-, Di-, Mono-Saccharides and Polyols) on small and large intestinal contents in healthy subjects shown by MRI. Am. J. Gastroenterol. 2014, 109, 110–119. [Google Scholar] [CrossRef]
- Halmos, E.P.; Power, V.A.; Shepherd, S.J.; Gibson, P.R.; Muir, J.G. A diet low in FODMAPs reduces symptoms of irritable bowel syndrome. Gastroenterology 2014, 146, 67–75. [Google Scholar] [CrossRef]
- Böhn, L.; Störsrud, S.; Liljebo, T.; Collin, L.; Lindfors, P.; Törnblom, H.; Simrén, M. Diet Low in FODMAPs Reduces Symptoms of Irritable Bowel Syndrome as Well as Traditional Dietary Advice: A Randomized Controlled Trial. Gastroenterology 2015, 149, 1399–1407. [Google Scholar] [CrossRef] [PubMed]
- O’Keeffe, M. Long-term impact of the low-FODMAP diet on gastrointestinal symptoms, dietary intake, patient acceptability, and healthcare utilization in irritable bowel syndrome. Neurogastroenterol. Motil. 2018, 30. [Google Scholar] [CrossRef] [PubMed]
- Eswaran, S.L.; Chey, W.D.; Han-Markey, T.; Ball, S.; Jackson, K. A Randomized Controlled Trial Comparing the Low FODMAP Diet vs. Modified NICE Guidelines in US Adults with IBS-D. Am. J. Gastroenterol. 2016, 111, 1824–1832. [Google Scholar] [CrossRef] [PubMed]
- Eswaran, S.L.; Chey, W.D.; Jackson, K.; Pillai, S.; Chey, S.W.; Han-Markey, T. A Diet Low in Fermentable Oligo-, Di-, and Monosaccharides and Polyols Improves Quality of Life and Reduces Activity Impairment in Patients With Irritable Bowel Syndrome and Diarrhea. Clin. Gastroenterol. Hepatol. 2017, 15, 1890–1899. [Google Scholar] [CrossRef] [PubMed]
- Dionne, J.; Ford, A.C.; Yuan, Y.; Chey, W.D.; Lacy, B.E.; Saito, Y.A.; Quigley, E.M.M.; Moayyedi, P.A. Systematic Review and Meta-Analysis Evaluating the Efficacy of a Gluten-Free Diet and a Low FODMAPs Diet in Treating Symptoms of Irritable Bowel Syndrome. Am. J. Gastroenterol. 2018, 113, 1290–1300. [Google Scholar] [CrossRef] [PubMed]
- Halmos, E.P.; Christophersen, C.T.; Bird, A.R.; Shepherd, S.J.; Gibson, P.R.; Muir, J.G. Diets that differ in their FODMAP content alter the colonic luminal microenvironment. Gut 2015, 64, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, H.M.; Lomer, M.C.E.; Farquharson, F.M.; Louis, P.; Fava, F.; Franciosi, E.; Scholz, M.; Tuohy, K.M.; Lindsay, J.O.; Irving, P.M.; et al. A Diet Low in FODMAPs Reduces Symptoms in Patients With Irritable Bowel Syndrome and A Probiotic Restores Bifidobacterium Species: A Randomized Controlled Trial. Gastroenterology 2017, 153, 936–947. [Google Scholar] [CrossRef]
- Shepherd, S.J.; Gibson, P.R. Fructose Malabsorption and Symptoms of Irritable Bowel Syndrome: Guidelines for Effective Dietary Management. J. Am. Diet. Assoc. 2006, 106, 1631–1639. [Google Scholar] [CrossRef]
- Staudacher, H.M.; Whelan, K.; Irving, P.M.; Lomer, M.C.E. Comparison of symptom response following advice for a diet low in fermentable carbohydrates (FODMAPs) versus standard dietary advice in patients with irritable bowel syndrome. J. Hum. Nutr. Diet. 2011, 24, 487–495. [Google Scholar] [CrossRef]
- Staudacher, H.M.; Lomer, M.C.; Anderson, J.L.; Barrett, J.S.; Muir, J.G.; Irving, P.M.; Whelan, K.; Anderson, J.L. Fermentable Carbohydrate Restriction Reduces Luminal Bifidobacteria and Gastrointestinal Symptoms in Patients with Irritable Bowel Syndrome. J. Nutr. 2012, 142, 1510–1518. [Google Scholar] [CrossRef]
- De Roest, R.H.; Dobbs, B.R.; Chapman, B.A.; Batman, B.; O’Brien, L.A.; Leeper, J.A.; Hebblethwaite, C.R.; Gearry, R.B. The low FODMAP diet improves gastrointestinal symptoms in patients with irritable bowel syndrome: A prospective study. Int. J. Clin. Pract. 2013, 67, 895–903. [Google Scholar] [CrossRef] [PubMed]
- Hustoft, T.N.; Hausken, T.; Ystad, S.O.; Valeur, J.; Brokstad, K.; Hatlebakk, J.G.; Lied, G.A. Effects of varying dietary content of fermentable short-chain carbohydrates on symptoms, fecal microenvironment, and cytokine profiles in patients with irritable bowel syndrome. Neurogastroenterol. Motil. 2017, 29, e12969. [Google Scholar] [CrossRef] [PubMed]
- McIntosh, K.; Reed, D.E.; Schneider, T.; Dang, F.; Keshteli, A.H.; De Palma, G.; Madsen, K.; Bercik, P.; Vanner, S. FODMAPs alter symptoms and the metabolome of patients with IBS: A randomised controlled trial. Gut 2017, 66, 1241–1251. [Google Scholar] [CrossRef] [PubMed]
- Fassio, F.; Facioni, M.S.; Guagnini, F. Lactose Maldigestion, Malabsorption, and Intolerance: A Comprehensive Review with a Focus on Current Management and Future Perspectives. Nutrients 2018, 10, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Szilagyi, A.; Ishayek, N. Lactose Intolerance, Dairy Avoidance, and Treatment Options. Nutrients 2018, 10, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Vesa, T.; Seppo, L.; Marteau, P.; Sahi, T.; Korpela, R. Role of irritable bowel syndrome in subjective lactose intolerance. Am. J. Clin. Nutr. 1998, 67, 710–715. [Google Scholar] [CrossRef] [PubMed]
- Böhmer, C.; Tuynman, H. The clinical relevance of lactose malabsorption in irritable bowel syndrome. Eur. J. Gastroenterol. Hepatol. 1996, 8, 1013–1016. [Google Scholar] [CrossRef]
- Böhmer, C.; Tuynman, H. The effect of a lactose-restricted diet in patients with a positive lactose tolerance test, earlier diagnosed as irritable bowel syndrome: A 5-year follow-up study. Eur. J. Gastroenterol. Hepatol. 2001, 13, 941–944. [Google Scholar] [CrossRef]
- Bozzani, A.; Penagini, R.; Velio, P.; Camboni, G.; Corbellini, A.; Quatrini, M.; Conte, D.; Bianchi, P.A. Lactose Malabsorption and Intolerance in Italians Clinical Implications. Dig. Dis. Sci. 1986, 31, 1313–1316. [Google Scholar] [CrossRef]
- Parker, T.J.; Woolner, J.T.; Prevost, A.T.; Tuffnell, Q.; Shorthouse, M.; Hunter, J.O. Irritable bowel syndrome: Is the search for lactose intolerance justified? Eur. J. Gastroenterol. Hepatol. 2001, 13, 219–225. [Google Scholar] [CrossRef]
- McKenzie, Y.A.; Bowyer, R.K.; Leach, H.; Gulia, P.; Horobin, J.; O’Sullivan, N.A.; Pettitt, C.; Reeves, L.B.; Seamark, L.; Wiliams, M.; et al. British Dietetic Association systematic review and evidence-based practice guidelines for the dietary management of irritable bowel syndrome in adults (2016 update). J. Hum. Nutr. Diet. 2016, 29, 549–575. [Google Scholar] [CrossRef] [PubMed]
- Marriott, B.P.; Cole, N.; Lee, E. National Estimates of Dietary Fructose Intake Increased from 1977 to 2004 in the United States. J. Nutr. 2009, 139, 1228S–1235S. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.K.; Kraft, N.; Zimmerman, B.; Jackson, M.; Rao, S.S.C. Fructose intolerance in IBS and utility of fructose-restricted diet. J. Clin. Gastroenterol. 2008, 42, 233–238. [Google Scholar] [CrossRef] [PubMed]
- Brouns, F. Overweight and diabetes prevention: Is a low-carbohydrate-high-fat diet recommendable? Eur. J. Nutr. 2018, 57, 1301–1312. [Google Scholar] [CrossRef] [PubMed]
- Noakes, T.D.; Windt, J. Evidence that supports the prescription of low-carbohydrate high-fat diets: A narrative review. Br. J. Sports Med. 2017, 51, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Giugliano, D.; Maiorino, M.I.; Bellastella, G.; Esposito, K. More sugar? No, thank you! The elusive nature of low carbohydrate diets. Endocrine 2018, 61, 383–387. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.K.; Borer, K.; Lin, P.J. Low-Carbohydrate-High-Fat Diet: Can it Help Exercise Performance? J. Hum. Kinet. 2017, 56, 81–92. [Google Scholar] [CrossRef]
- Austin, G.L.; Dalton, C.B.; Hu, Y.; Morris, C.B.; Hankins, J.; Weinland, S.R.; Westman, E.C.; Yancy, W.S., Jr.; Drossman, D.A. A Very Low-Carbohydrate Diet Improves Symptoms and Quality of Life in Diarrhea-Predominant Irritable Bowel Syndrome. Clin. Gastroenterol. Hepatol. 2009, 7, 706–708. [Google Scholar] [CrossRef]
- Biesiekierski, J.R. What is gluten? J. Gastroenterol. Hepatol. 2017, 32, 78–81. [Google Scholar] [CrossRef]
- Biesiekierski, J.R.; Rosella, O.; Rose, R.; Liels, K.; Barrett, J.S.; Shepherd, S.J.; Gibson, P.R.; Muir, J.G. Quantification of fructans, galacto-oligosacharides and other short-chain carbohydrates in processed grains and cereals. J. Hum. Nutr. Diet. 2011, 24, 154–176. [Google Scholar] [CrossRef]
- Tovoli, F. Clinical and diagnostic aspects of gluten related disorders. World J. Clin. Cases 2015, 3, 275. [Google Scholar] [CrossRef]
- Makharia, A.; Catassi, C.; Makharia, G.K. The Overlap between Irritable Bowel Syndrome and Non-Celiac Gluten Sensitivity: A Clinical Dilemma. Nutrients 2015, 7, 10417–10426. [Google Scholar] [CrossRef] [PubMed]
- Ludvigsson, J.F.; Leffler, D.A.; Bai, J.C.; Biagi, F.; Fasano, A.; Green, P.H.R.; Hadjivassiliou, M.; Kaukinen, K.; Kelly, C.P.; Leonard, J.N.; et al. The Oslo definitions for coeliac disease and related terms. Gut 2013, 62, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Dale, H.F.; Biesiekierski, J.R.; Lied, G.A. Non-coeliac gluten sensitivity and the spectrum of gluten-related disorders: An updated overview. Nutr. Res. Rev. 2018, 16, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Castillejo, G.; Cellier, C.; Cristofori, F.; De Magistris, L. Diagnosis of Non-Celiac Gluten Sensitivity (NCGS): The Salerno Experts Criteria. Nutrients 2015, 7, 4966–4977. [Google Scholar] [CrossRef]
- Biesiekierski, J.R.; Peters, S.L.; Newnham, E.D.; Rosella, O.; Muir, J.G.; Gibson, P.R. No effects of gluten in patients with self-reported non-celiac gluten sensitivity after dietary reduction of fermentable, poorly absorbed, short-chain carbohydrates. Gastroenterology 2013, 145, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Molina-Infante, J.; Carroccio, A. Suspected Nonceliac Gluten Sensitivity Confirmed in Few Patients After Gluten Challenge in Double-Blind, Placebo-Controlled Trials. Clin. Gastroenterol. Hepatol. 2017, 15, 339–348. [Google Scholar] [CrossRef] [PubMed]
- Skodje, G.I.; Sarna, V.K.; Minelle, I.H.; Rolfsen, K.L.; Muir, J.G.; Gibson, P.R.; Veieröd, M.B.; Henriksen, C.; Lundin, K.E.A. Fructan, Rather Than Gluten, Induces Symptoms in Patients With Self-Reported Non-Celiac Gluten Sensitivity. Gastroenterology 2018, 154, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Dale, H.F.; Hatlebakk, J.G.; Hovdenak, N.; Ystad, S.O.; Lied, G.A. The effect of a controlled gluten challenge in a group of patients with suspected non-coeliac gluten sensitivity: A randomized, double-blind placebo-controlled challenge. Neurogastroenterol. Motil. 2018, 30, 1–10. [Google Scholar] [CrossRef]
- Picarelli, A.; Borghini, R.; Tola, M.; Di Marino, M.; Urciuoli, C. Intestinal, Systemic, and Oral Gluten-related Alterations in Patient with Nonceliac Gluten Sensitivity. J. Clin. Gastroenterol. 2016, 50, 849–858. [Google Scholar] [CrossRef]
- Aziz, I.; Trott, N.; Briggs, R.; North, J.R.; Hadjivassiliou, M.; Sanders, D.S. Efficacy of a Gluten-Free Diet in Subjects With Irritable Bowel Syndrome-Diarrhea Unaware of Their HLA-DQ2/8 Genotype. Clin. Gastroenterol. Hepatol. 2016, 14, 696–703. [Google Scholar] [CrossRef] [PubMed]
- Elli, L.; Tomba, C.; Branchi, F.; Roncoroni, L.; Lombardo, V.; Bardella, M.T.; Ferretti, F.; Conte, D.; Valiante, F.; Fini, L.; et al. Evidence for the Presence of Non-Celiac Gluten Sensitivity in Patients with Functional Gastrointestinal Symptoms: Results from a Multicenter Randomized Placebo-Controlled Gluten Challenge. Nutrients 2016, 8, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Shahbazkhani, B.; Sadeghi, A.; Malekzadeh, R.; Khatavi, F.; Etemadi, M.; Kalantri, E.; Rostami-Nejad, M.; Rostami, K. Non-celiac gluten sensitivity has narrowed the spectrum of irritable bowel syndrome: A double-blind randomized placebo-controlled trial. Nutrients 2015, 7, 4542–4554. [Google Scholar] [CrossRef] [PubMed]
- Di Sabatino, A.; Volta, U.; Salvatore, C.; Biancheri, P.; Caio, G.; De Giorgio, R.; Di Stefano, M.; Corazza, G.R. Small Amounts of Gluten in Subjects With Suspected Nonceliac Gluten Sensitivity: A Randomized, Double-Blind, Placebo-Controlled, Cross-Over Trial. Clin. Gastroenterol. Hepatol. 2015, 13, 1604–1612. [Google Scholar] [CrossRef] [PubMed]
- Peters, S.L.; Biesiekierski, J.R.; Yelland, G.W.; Muir, J.G.; Gibson, P.R. Randomised clinical trial: Gluten may cause depression in subjects with non-coeliac gluten sensitivity–An exploratory clinical study. Aliment Pharmacol. Ther. 2014, 39, 1104–1112. [Google Scholar] [CrossRef] [PubMed]
- Vazquez-Roque, M.I.; Camilleri, M.; Smyrk, T.; Murray, J.A.; Marietta, E.; O’Neill, J.; Carlson, P.; Lamsam, J.; Janzow, D.; Eckert, D.; et al. A controlled trial of gluten-free diet in patients with irritable bowel syndrome-diarrhea: Effects on bowel frequency and intestinal function. Gastroenterology 2013, 144, 903–911. [Google Scholar] [CrossRef] [PubMed]
- Carroccio, A.; Mansueto, P.; Iacono, G.; Soresi, M. Non-Celiac Wheat Sensitivity Diagnosed by Double-Blind Placebo-Controlled Challenge: Exploring a New Clinical Entity. Am. J. Gastroenterol. 2012, 107, 1898–1906. [Google Scholar] [CrossRef]
- Biesiekierski, J.R.; Newnham, E.D.; Irving, P.M.; Barrett, J.S.; Haines, M.; Doecke, J.D.; Shepherd, S.J.; Muir, J.G.; Gibson, P.R. Gluten Causes gastrointestinal symptoms in subjects without celiac disease: A double-blind randomized placebo-controlled trial. Am. J. Gastroenterol. 2011, 106, 508–514. [Google Scholar] [CrossRef]
- Yao, C.K.; Gibson, P.R.; Shepherd, S.J. Design of clinical trials evaluating dietary interventions in patients with functional gastrointestinal disorders. Am. J. Gastroenterol. 2013, 108, 748–758. [Google Scholar] [CrossRef]
- Ford, A.C.; Moayyedi, P. Meta-analysis: Factors affecting placebo response rate in the irritable bowel syndrome. Aliment. Pharmacol. Ther. 2010, 32, 144–158. [Google Scholar] [CrossRef]
- Irvine, E.J.; Tack, J.; Crowell, M.D.; Gwee, K.A.; Ke, M.; Schmulson, M.J.; Whitehead, W.E.; Spiegel, B. Design of Treatment Trials for Functional Gastrointestinal Disorders. Gastroenterology 2016, 150, 1469–1480. [Google Scholar] [CrossRef] [PubMed]
- Bennet, S.M.P.; Böhn, L.; Störsrud, S.; Liljebo, T.; Collin, L.; Lindfors, P.; Törnblom, H.; Öhman, L.; Simrén, M. Multivariate modelling of faecal bacterial profiles of patients with IBS predicts responsiveness to a diet low in FODMAPs. Gut 2018, 67, 872–881. [Google Scholar] [CrossRef] [PubMed]
| Study (Year) Country | Design, Population (n) | Interventions | Main Findings |
|---|---|---|---|
| Shepherd et al. (2006) Australia [58] | Single-center study, IBS patients with fructose malabsorption (n = 62) | Diet avoiding free fructose and short-chain fructans, limitation of the total dietary fructose load, encouragement of foods with balanced amount of fructose/glucose, 40 months | Seventy-four percent responded positively regarding overall abdominal symptoms. This positive response was better in the adherent group compared to the non-adherent group. |
| Ong et al. (2010) Australia [46] | Single-center RCT, IBS patients and HV (n = 30) | Low FODMAP diet vs. high FODMAP diet for 2 days with 7-day wash-out period | Higher levels of breath hydrogen were found in HV and IBS patients on a high FODMAP diet. Patient following the high FODMAP diet had more GI symptoms and lethargy. HV receiving the high FODMAP diet only reported more flatulence. |
| Staudacher et al. (2011) UK [59] | Single-center Clinical Observational study, IBS patients (n = 82) | Low FODMAP vs. standard dietary advice for IBS patients (based on the NICE guidelines) | Seventy-six percent of the patients on a low-FODMAP diet were satisfied with their symptom response compared to 54% receiving the standard dietary advice. Eighty-two percent reported improvement in bloating with low-FODMAP vs 49% following the NICE guidelines. For 85% and 87% of patients following the low-FODMAP diet abdominal pain and flatulence improved respectively compared to 61% and 50% on the standard diet. |
| Staudacher et al. (2012) Australia [60] | Single-center RCT, IBS patients (n = 41) | Low FODMAP diet vs. habitual diet for 4 weeks | Lower intake of fermentable carbohydrates, and lower proportions/concentrations of bifidobacteria was noted in the intervention group compared to the group following their habitual diet. Sixty-eight percent of the patients in the intervention group reported adequate symptom control compared to 23% of the patients with habitual food intake. |
| de Roest et al. (2013) New Zealand [61] | Single-center study, IBS patients (n = 90) | Low FODMAP diet, mean of 15.7 months follow-up | At follow-up, patients reported improvement in abdominal pain, bloating, flatulence and diarrhea. Patients with fructose intolerance experienced an even greater improvement. |
| Halmos et al. (2014) Australia [50] | Single-center RCT, cross-over, IBS patients (n = 30) and healthy controls (n = 8) | Low FODMAP vs. typical Australian diet for 21 days with a washout period of at least 21 days | Patients on the low FODMAP diet reported improvement of their global IBS symptoms. Abdominal pain, bloating, and passing flatus were significantly better in the low FODMAP group. In most patients, the greatest improvement in symptoms occurred during the first week. Symptoms were minimal and unaltered by either diet among controls. |
| Böhn et al. (2015) Sweden [51] | Multicenter RCT, IBS patients (n = 75) | Low FODMAP diet vs. a traditional IBS diet (based on the NICE guidelines) for 4 weeks | During the intervention, the severity of IBS symptoms was reduced in both groups. At the end of the intervention, 50% of the patients on a low-FODMAP diet had a reduction in IBS severity scores (≥50) compared with baseline vs 46% of the patients following the traditional IBS diet. |
| Eswaran et al. (2016) US [53,54] | Single-center RCT, IBS-D patients (n = 92) | Low FODMAP diet vs. a modified diet based on the NICE guidelines (mNICE) for 4 weeks | Fifty-two percent of the low FODMAP vs. 41% of the mNICE group reported adequate relief of their IBS-D symptoms, which was not significant. The low FODMAP diet led to significantly greater improvement in individual IBS symptoms, particularly pain and bloating, and quality of life compared with the mNICE diet. |
| Hustoft et al. (2017) Norway [62] | Single-center RCT, IBS-D and IBS-M patients (n = 20) | Low FODMAP diet for 3 weeks & afterwards randomization to a FODMAP supplement or maltodextrin (placebo) for 10 days with a wash-out period of 3 weeks | Patients receiving the placebo compared to the FODMAP supplement reported a significant relief of symptoms, 80% compared to 30% respectively. After following the low FODMAP diet, alterations in inflammatory cytokines, microbiota profile and SCFAs were detected. |
| Staudacher et al. (2017) UK [57] | Two-center RCT, IBS patients (n = 104) | Low FODMAP diet vs. sham diet (restriction of similar amount of foods, but maintaining the FODMAP content in the diet) with randomization to a multi-strain probiotics vs. placebo for 4 weeks | The low FODMAP diet was associated with an adequate relief of symptoms and a significant reduction of symptom scores compared to placebo, 57% compared to 38% respectively. Co-administration of the probiotic increased the number of Bifidobacterium species compared to placebo. |
| McIntosh et al. (2017) Canada [63] | Single-center RCT, IBS patients (n = 37) | Low FODMAP diet vs. high FODMAP diet for 3 weeks | Patients with a low FODMAP intake had a significant improvement in symptom scores and had changes in their metabolome compared to patient following the high FODMAP diet. FODMAPs modulated the microbiota and histamine levels in a subset of patients. |
| Study (Year) Country | Design, Population (n) | Interventions | Main Findings |
|---|---|---|---|
| Dale et al. (2018) Norway [89] | RDBPC, cross-over trial NCGS patients on a GFD (n = 20) | GFD vs. GCD, 4 challenges (2 gluten, 2 placebo) 4 days per intervention, 3 days washout. (muffins with gluten 11 g/day vs. gluten-free muffins) | No significant differences in symptom severity between gluten and placebo challenges. High symptom scores during all challenges. |
| Skodje et al. (2018) Norway [88] | RDBPC, cross-over trial self-reported NCGS patients on GFD >6 months (n = 59) | GFD (placebo) vs. GCD (5.7 g/day) vs. Fructans (2.1 g/day), 1 week per intervention, 1 week washout. (concealed muesli bars). | Significant differences in GI symptoms between all interventions. Fructans: overall GI symptoms and bloating significantly higher than gluten. |
| Picarelli et al. (2016) Italy [90] | RDBPC trial, NCGS patients (n = 26) | GFD vs. GCD, 1 day. (croissant with 10 g of gluten vs. gluten-free croissant) | No significant difference in overall symptom severity between gluten and placebo challenge. |
| Aziz et al. (2016) UK [91] | Open label, IBS-D patients (n = 41) | GFD, 6 weeks (information and advice GFD by dietician) | Decrease of symptoms in >70% of patients, significant after 2 weeks, similar results in HLA-DQ positive and negative |
| Elli et al. (2016) Italy [92] | RDBPC, cross-over trial, IBS patients with NCGS (n = 98) | GFD vs. GCD, 1 week per intervention, 1 week washout (gastro-soluble capsules with 5.6 g/day gluten powder or placebo). Run-in period of 3 weeks GFD. | 14% of patients that responded to gluten withdrawal had symptomatic relapse during gluten challenge. |
| Shahbazkhani et al. (2015) Iran [93] | DB RCT, IBS patients (n = 148) | GFD vs. GCD, 6 weeks (packages with 52 g/day gluten powder, or rice starch as placebo). Run-in period of 6 weeks. | Significant improvement in overall symptom severity GFD (83.8%) vs. GCD (25.7%). |
| Di Sabatino et al. (2015) Italy [94] | RDBPC, cross-over trial, suspected NCGS patients (n = 61) | GFD vs. GCD, 1 week per intervention, 1 week washout. (gastro-soluble capsules with 4.4 g/day gluten vs. rice starch) | Significant increase in overall symptom severity during gluten compared to placebo. Abdominal bloating, pain and (extra)-intestinal symptoms significantly more severe during gluten-period. |
| Peters et al. (2014) Australia [95] | RDBPC, cross-over trial NCGS patients (n = 22) | Gluten (16 g/day) vs. Whey (16 g/day) vs. Placebo, 3 days per intervention, at least 3 days washout. (provided meals with 16 g/day whey protein vs. placebo) | No significant differences in GI symptoms between interventions. Significant more feelings of depression due to short-term exposure to gluten. |
| Vazquez-Roque et al. (2013) USA [96] | RCT, IBS-D patients (n = 45) | GFD vs. GCD, 4 weeks (standardized meals provided by metabolic kitchen, with or without gluten) | Significant increase in stool frequency GCD vs. GFD. Greater difference in HLA-DQ positive patients. |
| Biesiekierski et al. (2013) Australia [86] | (1) RDBPC, cross-over trial, IBS patients with NCGS (n = 40) (2) Rechallenge, IBS patients with NCGS (n = 22) | (1) High gluten (16 g/day) vs. Low gluten (2 g/day) vs. Whey (16 g/day), 1 week per intervention. (2) Gluten (16 g/day) vs. Whey (16 g/day) vs. Placebo (no additional protein), 3 days. Run-in period of 2 weeks, GFD and low FODMAP diet | Symptom improvement in all patients during run-in period (low FODMAP, gluten-free). Symptom deterioration in all groups, no specific gluten dose response. |
| Carroccio et al. (2012) Italy [97] | RDBPC, cross-over trial, suspected NCGS patients (n = 920) | Wheat (20 g/day) vs. Xylose (placebo), 2 weeks per intervention, at least 1 week washout. (gastro-soluble capsules). Elimination diet of 4 weeks prior to challenge. | Symptom improvement of at least 30% in wheat-free period (Salerno experts’ criteria): NCGS diagnosis was confirmed in 30% (n = 276) of subjects. |
| Biesiekierski et al. (2011) Australia [98] | RCT, IBS patients (n = 39) | GFD vs. GCD, 6 weeks (Muffin and bread with or without gluten, 16 g/day) | GCD baseline vs. 1 week: significant increase in overall symptom severity, as well as bloating, abdominal pain, tiredness, dissatisfaction with stool. GCD vs. GFD, 6 weeks: significant increase in severity of abdominal pain, tiredness and dissatisfaction with stool. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Algera, J.; Colomier, E.; Simrén, M. The Dietary Management of Patients with Irritable Bowel Syndrome: A Narrative Review of the Existing and Emerging Evidence. Nutrients 2019, 11, 2162. https://doi.org/10.3390/nu11092162
Algera J, Colomier E, Simrén M. The Dietary Management of Patients with Irritable Bowel Syndrome: A Narrative Review of the Existing and Emerging Evidence. Nutrients. 2019; 11(9):2162. https://doi.org/10.3390/nu11092162
Chicago/Turabian StyleAlgera, Joost, Esther Colomier, and Magnus Simrén. 2019. "The Dietary Management of Patients with Irritable Bowel Syndrome: A Narrative Review of the Existing and Emerging Evidence" Nutrients 11, no. 9: 2162. https://doi.org/10.3390/nu11092162
APA StyleAlgera, J., Colomier, E., & Simrén, M. (2019). The Dietary Management of Patients with Irritable Bowel Syndrome: A Narrative Review of the Existing and Emerging Evidence. Nutrients, 11(9), 2162. https://doi.org/10.3390/nu11092162





