Ketoacid Analogues Supplementation in Chronic Kidney Disease and Future Perspectives
Abstract
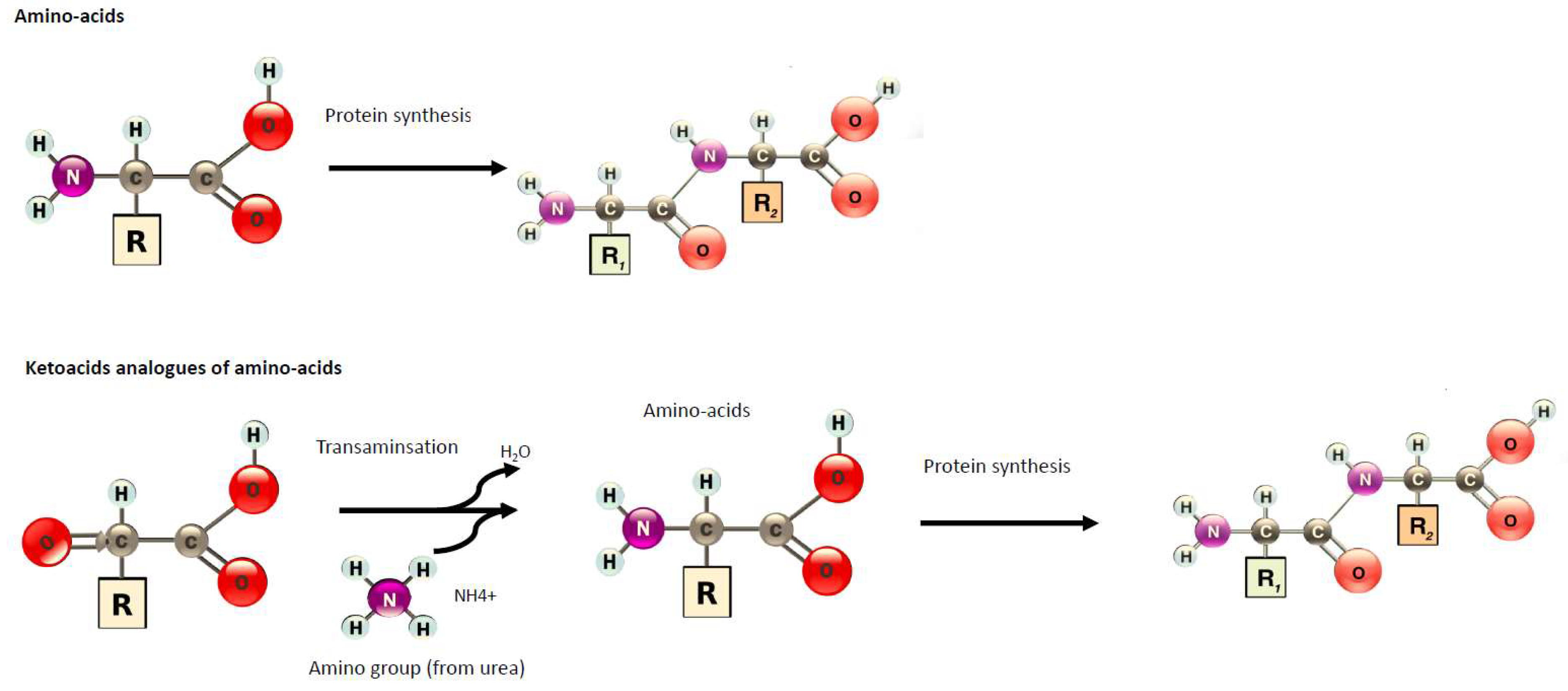
1. Introduction
2. Methods
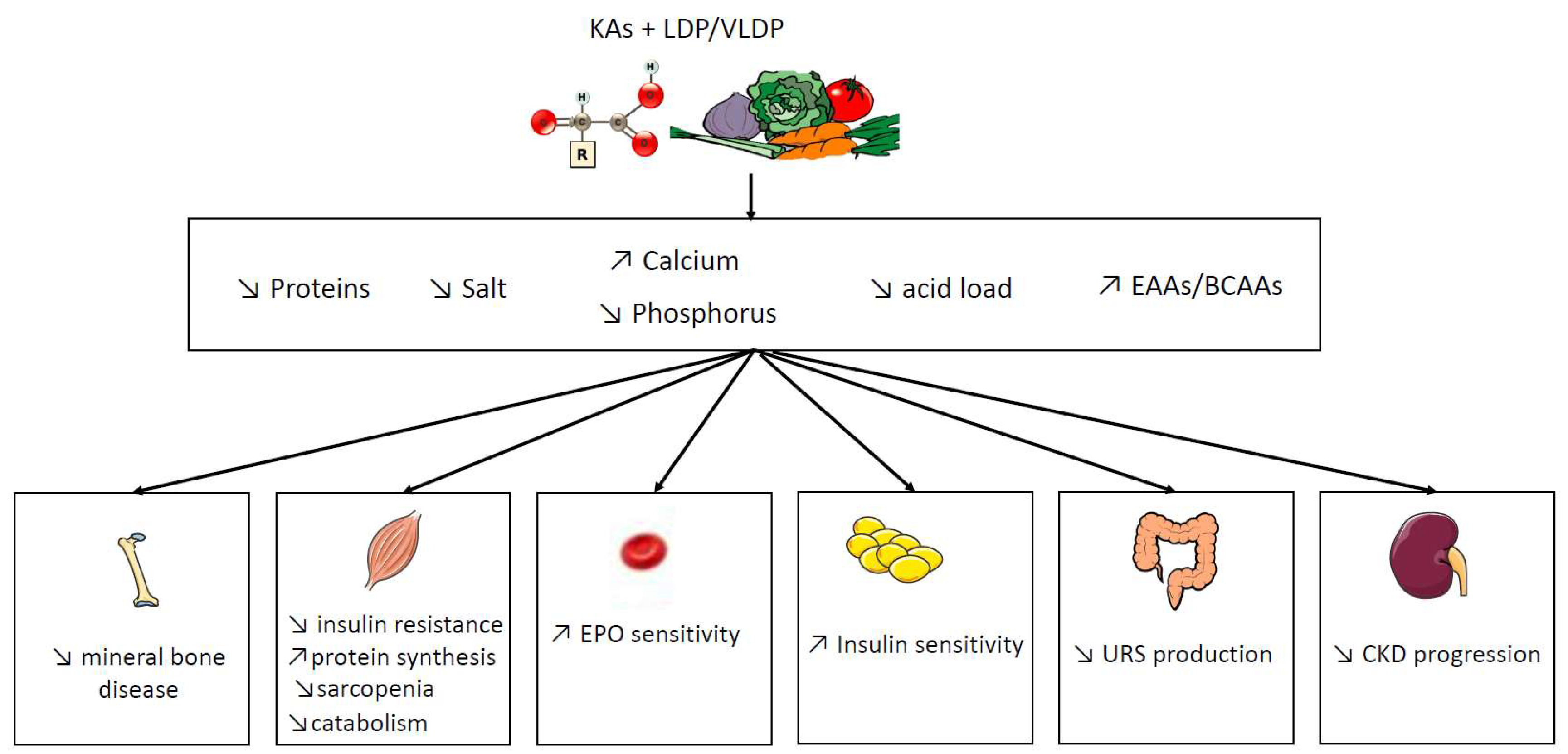
3. Potential Benefit of Ketoacid Analogues
3.1. Anabolic Action of Ketoacid Analogues
3.2. Role of Ketoacid Analogues in Intestinal Metabolism and Uremic Toxins Production
3.3. Other Roles of Ketoacid Analogues
4. Do Ketoacid Analogues Have an Impact on Renal Function and Mortality in CKD?
5. Is a Keto Acid Analogues Supplementation a Modern Treatment?
6. Conclusion
7. Future Directions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Brenner, B.M.; Meyer, T.W.; Hostetter, T.H. Dietary protein intake and the progressive nature of kidney disease: The role of hemodynamically mediated glomerular injury in the pathogenesis of progressive glomerular sclerosis in aging, renal ablation, and intrinsic renal disease. N. Engl. J. Med. 1982, 307, 652–659. [Google Scholar] [PubMed]
- Shah, A.P.; Kalantar-Zadeh, K.; Kopple, J.D. Is there a role for ketoacid supplements in the management of CKD? Am. J. Kidney Dis. 2015, 65, 659–673. [Google Scholar] [CrossRef] [PubMed]
- Milovanova, L.; Fomin, V.; Moiseev, S.; Taranova, M.; Milovanov, Y.; Lysenko Kozlovskaya, L.; Kozlov, V.; Kozevnikova, E.; Milovanova, S.; Lebedeva, M.; et al. Effect of essential amino acid кetoanalogues and protein restriction diet on morphogenetic proteins (FGF-23 and Klotho) in 3b-4 stages chronic кidney disease patients: A randomized pilot study. Clin. Exp. Nephrol. 2018, 22, 1351–1359. [Google Scholar] [CrossRef] [PubMed]
- Teplan, V.; Schück, O.; Racek, J.; Mareckova, O.; Stollova, M.; Hanzal, V.; Malý, J. Reduction of plasma asymmetric dimethylarginine in obese patients with chronic kidney disease after three years of a low-protein diet supplemented with keto-amino acids: A randomized controlled trial. Wien. Klin. Wochenschr. 2008, 120, 478–485. [Google Scholar] [CrossRef] [PubMed]
- Teplan, V.; Schück, O.; Knotek, A.; Hajný, J.; Horácková, M.; Kvapil, M. Czech multicenter study Enhanced metabolic effect of erythropoietin and keto acids in CRF patients on low-protein diet: Czech multicenter study. Am. J. Kidney Dis. 2003, 41, S26–S30. [Google Scholar] [CrossRef] [PubMed]
- Bernhard, J.; Beaufrère, B.; Laville, M.; Fouque, D. Adaptive response to a low-protein diet in predialysis chronic renal failure patients. J. Am. Soc. Nephrol. 2001, 12, 1249–1254. [Google Scholar] [PubMed]
- Hecking, E.; Andrzejewski, L.; Prellwitz, W.; Opferkuch, W.; Müller, D.; Port, F.K. A controlled study of supplementation with essential amino acids and alpha-keto acids in the conservative management of patients with chronic renal failure. Z. Ernahrungswiss. 1982, 21, 299–311. [Google Scholar] [CrossRef]
- Wang, D.; Wei, L.; Yang, Y.; Liu, H. Dietary supplementation with ketoacids protects against CKD-induced oxidative damage and mitochondrial dysfunction in skeletal muscle of 5/6 nephrectomised rats. Skelet Muscle 2018, 8, 18. [Google Scholar] [CrossRef]
- Liu, D.; Wu, M.; Li, L.; Gao, X.; Yang, B.; Mei, S.; Fu, L.; Mei, C. Low-protein diet supplemented with ketoacids delays the progression of diabetic nephropathy by inhibiting oxidative stress in the KKAy mice model. Br. J. Nutr. 2018, 119, 22–29. [Google Scholar] [CrossRef]
- Zhang, J.-Y.; Yin, Y.; Ni, L.; Long, Q.; You, L.; Zhang, Q.; Lin, S.-Y.; Chen, J. Low-protein diet supplemented with ketoacids ameliorates proteinuria in 3/4 nephrectomised rats by directly inhibiting the intrarenal renin-angiotensin system. Br. J. Nutr. 2016, 116, 1491–1501. [Google Scholar] [CrossRef]
- Zhang, Y.; Huang, J.; Yang, M.; Gu, L.; Ji, J.; Wang, L.; Yuan, W. Effect of a low-protein diet supplemented with keto-acids on autophagy and inflammation in 5/6 nephrectomized rats. Biosci. Rep. 2015, 35, e00263. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.-T.; Lu, L.; Shi, Y.; Geng, Z.-B.; Yin, Y.; Wang, M.; Wei, L.-B. Supplementation of ketoacids contributes to the up-regulation of the Wnt7a/Akt/p70S6K pathway and the down-regulation of apoptotic and ubiquitin-proteasome systems in the muscle of 5/6 nephrectomised rats. Br. J. Nutr. 2014, 111, 1536–1548. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Wu, J.; Dong, Z.; Hua, C.; Hu, H.; Mei, C. A low-protein diet supplemented with ketoacids plays a more protective role against oxidative stress of rat kidney tissue with 5/6 nephrectomy than a low-protein diet alone. Br. J. Nutr. 2010, 103, 608–616. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Huang, L.; Grosjean, F.; Esposito, V.; Wu, J.; Fu, L.; Hu, H.; Tan, J.; He, C.; Gray, S.; et al. Low-protein diet supplemented with ketoacids reduces the severity of renal disease in 5/6 nephrectomized rats: A role for KLF15. Kidney Int. 2011, 79, 987. [Google Scholar] [CrossRef] [PubMed]
- Maniar, S.; Beaufils, H.; Laouari, D.; Forget, D.; Kleinknecht, C. Supplemented low-protein diets protect the rat kidney without causing undernutrition. J. Lab. Clin. Med. 1992, 120, 851–860. [Google Scholar] [PubMed]
- Laouari, D.; Jean, G.; Kleinknecht, C.; Broyer, M. Growth, free plasma and muscle amino-acids in uraemic rats fed various low-protein diets. Pediatr. Nephrol. 1991, 5, 318–322. [Google Scholar] [CrossRef]
- Benjelloun, A.S.; Merville, P.; Cambar, J.; Aparicio, M. Effects of a low-protein diet on urinary glycosaminoglycan excretion in adriamycin-treated rats. Nephron 1993, 64, 242–248. [Google Scholar] [CrossRef] [PubMed]
- Barsotti, G.; Moriconi, L.; Cupisti, A.; Dani, L.; Ciardella, F.; Lupetti, S.; Giovannetti, S. Protection of renal function and of nutritional status in uremic rats by means of a low-protein, low-phosphorus supplemented diet. Nephron 1988, 49, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Meisinger, E.; Gretz, N.; Strauch, M. Hyperfiltration due to amino and keto acid supplements of low-protein diets: Influence on proteinuria. Infus. Klin Ernahr 1987, 14 (Suppl. 5), 26–29. [Google Scholar]
- Di Iorio, B.R.; Marzocco, S.; Bellasi, A.; De Simone, E.; Dal Piaz, F.; Rocchetti, M.T.; Cosola, C.; Di Micco, L.; Gesualdo, L. Nutritional therapy reduces protein carbamylation through urea lowering in chronic kidney disease. Nephrol. Dial. Transplant. 2018, 33, 804–813. [Google Scholar] [CrossRef]
- Garneata, L.; Stancu, A.; Dragomir, D.; Stefan, G.; Mircescu, G. Ketoanalogue-Supplemented Vegetarian Very Low-Protein Diet and CKD Progression. J. Am. Soc. Nephrol. 2016, 27, 2164–2176. [Google Scholar] [PubMed]
- Di Iorio, B.; Di Micco, L.; Torraca, S.; Sirico, M.L.; Russo, L.; Pota, A.; Mirenghi, F.; Russo, D. Acute effects of very-low-protein diet on FGF23 levels: A randomized study. Clin. J. Am. Soc. Nephrol. 2012, 7, 581–587. [Google Scholar] [PubMed]
- Marzocco, S.; Dal Piaz, F.; Di Micco, L.; Torraca, S.; Sirico, M.L.; Tartaglia, D.; Autore, G.; Di Iorio, B. Very low protein diet reduces indoxyl sulfate levels in chronic kidney disease. Blood Purif. 2013, 35, 196–201. [Google Scholar] [CrossRef] [PubMed]
- Di Iorio, B.R.; Cucciniello, E.; Martino, R.; Frallicciardi, A.; Tortoriello, R.; Struzziero, G. Acute and persistent antiproteinuric effect of a low-protein diet in chronic kidney disease. G Ital. Nefrol 2009, 26, 608–615. [Google Scholar] [PubMed]
- Menon, V.; Kopple, J.D.; Wang, X.; Beck, G.J.; Collins, A.J.; Kusek, J.W.; Greene, T.; Levey, A.S.; Sarnak, M.J. Effect of a very low-protein diet on outcomes: Long-term follow-up of the Modification of Diet in Renal Disease (MDRD) Study. Am. J. Kidney Dis. 2009, 53, 208–217. [Google Scholar] [CrossRef] [PubMed]
- Mircescu, G.; Gârneaţă, L.; Stancu, S.H.; Căpuşă, C. Effects of a supplemented hypoproteic diet in chronic kidney disease. J. Ren. Nutr. 2007, 17, 179–188. [Google Scholar] [PubMed]
- Gennari, F.J.; Hood, V.L.; Greene, T.; Wang, X.; Levey, A.S. Effect of dietary protein intake on serum total CO2 concentration in chronic kidney disease: Modification of Diet in Renal Disease study findings. Clin. J. Am. Soc. Nephrol. 2006, 1, 52–57. [Google Scholar] [CrossRef]
- Menon, V.; Wang, X.; Greene, T.; Beck, G.J.; Kusek, J.W.; Selhub, J.; Levey, A.S.; Sarnak, M.J. Homocysteine in chronic kidney disease: Effect of low protein diet and repletion with B vitamins. Kidney Int. 2005, 67, 1539–1546. [Google Scholar]
- Feiten, S.F.; Draibe, S.A.; Watanabe, R.; Duenhas, M.R.; Baxmann, A.C.; Nerbass, F.B.; Cuppari, L. Short-term effects of a very-low-protein diet supplemented with ketoacids in nondialyzed chronic kidney disease patients. Eur. J. Clin. Nutr. 2005, 59, 129–136. [Google Scholar] [PubMed]
- Prakash, S.; Pande, D.P.; Sharma, S.; Sharma, D.; Bal, C.S.; Kulkarni, H. Randomized, double-blind, placebo-controlled trial to evaluate efficacy of ketodiet in predialytic chronic renal failure. J. Ren. Nutr. 2004, 14, 89–96. [Google Scholar] [CrossRef]
- Di Iorio, B.R.; Minutolo, R.; De Nicola, L.; Bellizzi, V.; Catapano, F.; Iodice, C.; Rubino, R.; Conte, G. Supplemented very low protein diet ameliorates responsiveness to erythropoietin in chronic renal failure. Kidney Int. 2003, 64, 1822–1828. [Google Scholar] [CrossRef] [PubMed]
- Malvy, D.; Maingourd, C.; Pengloan, J.; Bagros, P.; Nivet, H. Effects of severe protein restriction with ketoanalogues in advanced renal failure. J. Am. Coll. Nutr. 1999, 18, 481–486. [Google Scholar] [CrossRef] [PubMed]
- Kopple, J.D.; Levey, A.S.; Greene, T.; Chumlea, W.C.; Gassman, J.J.; Hollinger, D.L.; Maroni, B.J.; Merrill, D.; Scherch, L.K.; Schulman, G.; et al. Effect of dietary protein restriction on nutritional status in the Modification of Diet in Renal Disease Study. Kidney Int. 1997, 52, 778–791. [Google Scholar] [CrossRef] [PubMed]
- Levey, A.S.; Adler, S.; Caggiula, A.W.; England, B.K.; Greene, T.; Hunsicker, L.G.; Kusek, J.W.; Rogers, N.L.; Teschan, P.E. Effects of dietary protein restriction on the progression of advanced renal disease in the Modification of Diet in Renal Disease Study. Am. J. Kidney Dis. 1996, 27, 652–663. [Google Scholar] [CrossRef]
- Klahr, S.; Levey, A.S.; Beck, G.J.; Caggiula, A.W.; Hunsicker, L.; Kusek, J.W.; Striker, G. The effects of dietary protein restriction and blood-pressure control on the progression of chronic renal disease. Modification of Diet in Renal Disease Study Group. N. Engl. J. Med. 1994, 330, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Coggins, C.H.; Dwyer, J.T.; Greene, T.; Petot, G.; Snetselaar, L.G.; Van Lente, F. Serum lipid changes associated with modified protein diets: Results from the feasibility phase of the Modification of Diet in Renal Disease Study. Am. J. Kidney Dis. 1994, 23, 514–523. [Google Scholar] [CrossRef]
- Lindenau, K.; Abendroth, K.; Kokot, F.; Vetter, K.; Rehse, C.; Fröhling, P.T. Therapeutic effect of keto acids on renal osteodystrophy. A prospective controlled study. Nephron 1990, 55, 133–135. [Google Scholar] [CrossRef] [PubMed]
- Jungers, P.; Chauveau, P.; Ployard, F.; Lebkiri, B.; Ciancioni, C.; Man, N.K. Comparison of ketoacids and low protein diet on advanced chronic renal failure progression. Kidney Int. Suppl. 1987, 22, S67–S71. [Google Scholar] [PubMed]
- Mitch, W.E.; Walser, M.; Sapir, D.G. Nitrogen sparing induced by leucine compared with that induced by its keto analogue, alpha-ketoisocaproate, in fasting obese man. J. Clin. Investig. 1981, 67, 553–562. [Google Scholar] [CrossRef] [PubMed]
- Goodship, T.H.; Mitch, W.E.; Hoerr, R.A.; Wagner, D.A.; Steinman, T.I.; Young, V.R. Adaptation to low-protein diets in renal failure: Leucine turnover and nitrogen balance. J. Am. Soc. Nephrol. 1990, 1, 66–75. [Google Scholar]
- Garibotto, G.; Sofia, A.; Parodi, E.L.; Ansaldo, F.; Bonanni, A.; Picciotto, D.; Signori, A.; Vettore, M.; Tessari, P.; Verzola, D. Effects of Low-Protein, and Supplemented Very Low–Protein Diets, on Muscle Protein Turnover in Patients With CKD. Kidney Int. Rep. 2018, 3, 701–710. [Google Scholar] [CrossRef] [PubMed]
- Walser, M.; Coulter, A.W.; Dighe, S.; Crantz, F.R. The effect of keto-analogues of essential amino acids in severe chronic uremia. J. Clin. Investig. 1973, 52, 678–690. [Google Scholar] [PubMed]
- Tom, K.; Young, V.R.; Chapman, T.; Masud, T.; Akpele, L.; Maroni, B.J. Long-term adaptive responses to dietary protein restriction in chronic renal failure. Am. J. Physiol. 1995, 268, E668–E677. [Google Scholar] [PubMed]
- Koppe, L.; Mafra, D.; Fouque, D. Probiotics and chronic kidney disease. Kidney Int. 2015, 88, 958–966. [Google Scholar] [CrossRef] [PubMed]
- Vaziri, N.D.; Wong, J.; Pahl, M.; Piceno, Y.M.; Yuan, J.; Desantis, T.Z.; Ni, Z.; Nguyen, T.-H.; Andersen, G.L. Chronic kidney disease alters intestinal microbial flora. Kidney Int. 2012, 83, 308–315. [Google Scholar]
- Walser, M.; Jarskog, F.L.; Hill, S.B. Branched-chain-ketoacid metabolism in patients with chronic renal failure. Am. J. Clin. Nutr. 1989, 50, 807–813. [Google Scholar] [CrossRef] [PubMed]
- Koppe, L.; Nyam, E.; Vivot, K.; Manning Fox, J.E.; Dai, X.-Q.; Nguyen, B.N.; Trudel, D.; Attané, C.; Moullé, V.S.; MacDonald, P.E.; et al. Urea impairs β cell glycolysis and insulin secretion in chronic kidney disease. J. Clin. Investig. 2016, 126, 3598–3612. [Google Scholar] [CrossRef]
- Vaziri, N.D.; Yuan, J.; Norris, K. Role of urea in intestinal barrier dysfunction and disruption of epithelial tight junction in chronic kidney disease. Am. J. Nephrol. 2013, 37, 1–6. [Google Scholar] [CrossRef]
- D’Apolito, M.; Du, X.; Zong, H.; Catucci, A.; Maiuri, L.; Trivisano, T.; Pettoello-Mantovani, M.; Campanozzi, A.; Raia, V.; Pessin, J.E.; et al. Urea-induced ROS generation causes insulin resistance in mice with chronic renal failure. J. Clin. Investig. 2009, 120, 203–213. [Google Scholar] [CrossRef]
- Koeth, R.A.; Kalantar-Zadeh, K.; Wang, Z.; Fu, X.; Tang, W.H.W.; Hazen, S.L. Protein carbamylation predicts mortality in ESRD. J. Am. Soc. Nephrol. 2013, 24, 853–861. [Google Scholar] [CrossRef]
- Chauveau, P.; Koppe, L.; Combe, C.; Lasseur, C.; Trolonge, S.; Aparicio, M. Vegetarian diets and chronic kidney disease. Nephrol. Dial. Transplant. 2018, 34, 199–207. [Google Scholar]
- Bellizzi, V.; Di Iorio, B.R.; De Nicola, L.; Minutolo, R.; Zamboli, P.; Trucillo, P.; Catapano, F.; Cristofano, C.; Scalfi, L.; Conte, G.; et al. Very low protein diet supplemented with ketoanalogs improves blood pressure control in chronic kidney disease. Kidney Int. 2007, 71, 245–251. [Google Scholar] [CrossRef] [PubMed]
- de Brito-Ashurst, I.; Varagunam, M.; Raftery, M.J.; Yaqoob, M.M. Bicarbonate supplementation slows progression of CKD and improves nutritional status. J. Am. Soc. Nephrol. 2009, 20, 2075–2084. [Google Scholar] [PubMed]
- Menon, V.; Tighiouart, H.; Vaughn, N.S.; Beck, G.J.; Kusek, J.W.; Collins, A.J.; Greene, T.; Sarnak, M.J. Serum bicarbonate and long-term outcomes in CKD. Am. J. Kidney Dis. 2010, 56, 907–914. [Google Scholar] [PubMed]
- Lorenzo, C.; Nath, S.D.; Hanley, A.J.G.; Abboud, H.E.; Haffner, S.M. Relation of Low Glomerular Filtration Rate to Metabolic Disorders in Individuals without Diabetes and with Normoalbuminuria. Clin. J. Am. Soc. Nephrol. 2008, 3, 783–789. [Google Scholar] [CrossRef] [PubMed]
- Mak, R.H. Insulin and its role in chronic kidney disease. Pediatr. Nephrol. 2008, 23, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Menon, V.; Greene, T.; Pereira, A.A.; Wang, X.; Beck, G.J.; Kusek, J.W.; Collins, A.J.; Levey, A.S.; Sarnak, M.J. Glycosylated Hemoglobin and Mortality in Patients with Nondiabetic Chronic Kidney Disease. JASN 2005, 16, 3411–3417. [Google Scholar] [CrossRef]
- Rigalleau, V.; Blanchetier, V.; Combe, C.; Guillot, C.; Deleris, G.; Aubertin, J.; Aparicio, M.; Gin, H. A low-protein diet improves insulin sensitivity of endogenous glucose production in predialytic uremic patients. Am. J. Clin. Nutr. 1997, 65, 1512–1516. [Google Scholar] [CrossRef][Green Version]
- Rigalleau, V.; Combe, C.; Blanchetier, V.; Aubertin, J.; Aparicio, M.; Gin, H. Low protein diet in uremia: Effects on glucose metabolism and energy production rate. Kidney Int. 1997, 51, 1222–1227. [Google Scholar]
- Fontana, L.; Cummings, N.E.; Arriola Apelo, S.I.; Neuman, J.C.; Kasza, I.; Schmidt, B.A.; Cava, E.; Spelta, F.; Tosti, V.; Syed, F.A.; et al. Decreased Consumption of Branched-Chain Amino Acids Improves Metabolic Health. Cell Rep. 2016, 16, 520–530. [Google Scholar] [CrossRef]
- Hahn, D.; Hodson, E.M.; Fouque, D. Low protein diets for non-diabetic adults with chronic kidney disease. Cochrane Database Syst. Rev. 2018, 10, CD001892. [Google Scholar] [PubMed]
- Li, A.; Lee, H.Y.; Lin, Y.C. The Effect of Ketoanalogues on Chronic Kidney Disease Deterioration: A Meta-Analysis. Nutrients 2019, 26, 957. [Google Scholar]
- Chauveau, P.; Combe, C.; Rigalleau, V.; Vendrely, B.; Aparicio, M. Restricted protein diet is associated with decrease in proteinuria: Consequences on the progression of renal failure. J. Ren. Nutr. 2007, 17, 250–257. [Google Scholar] [PubMed]
- Di Iorio, B.R.; Bellizzi, V.; Bellasi, A.; Torraca, S.; D’Arrigo, G.; Tripepi, G.; Zoccali, C. Phosphate attenuates the anti-proteinuric effect of very low-protein diet in CKD patients. Nephrol. Dial. Transplant. 2013, 28, 632–640. [Google Scholar] [PubMed]
- Bellizzi, V.; Chiodini, P.; Cupisti, A.; Viola, B.F.; Pezzotta, M.; De Nicola, L.; Minutolo, R.; Barsotti, G.; Piccoli, G.B.; Di Iorio, B. Very low-protein diet plus ketoacids in chronic kidney disease and risk of death during end-stage renal disease: A historical cohort controlled study. Nephrol. Dial. Transplant. 2015, 30, 71–77. [Google Scholar] [PubMed]
- Chauveau, P.; Couzi, L.; Vendrely, B.; de Précigout, V.; Combe, C.; Fouque, D.; Aparicio, M. Long-term outcome on renal replacement therapy in patients who previously received a keto acid-supplemented very-low-protein diet. Am. J. Clin. Nutr. 2009, 90, 969–974. [Google Scholar] [CrossRef] [PubMed]
- Koppe, L.; Fouque, D. The Role for Protein Restriction in Addition to Renin-Angiotensin-Aldosterone System Inhibitors in the Management of CKD. Am. J. Kidney Dis. 2018, 73, 248–257. [Google Scholar] [PubMed]
- Jiang, Z.; Zhang, X.; Yang, L.; Li, Z.; Qin, W. Effect of restricted protein diet supplemented with keto analogues in chronic kidney disease: A systematic review and meta-analysis. Int. Urol. Nephrol. 2016, 48, 409–418. [Google Scholar] [CrossRef] [PubMed]
- Chauveau, P.; Barthe, N.; Rigalleau, V.; Ozenne, S.; Castaing, F.; Delclaux, C.; de Précigout, V.; Combe, C.; Aparicio, M. Outcome of nutritional status and body composition of uremic patients on a very low protein diet. Am. J. Kidney Dis. 1999, 34, 500–507. [Google Scholar] [CrossRef]
- Bellizzi, V.; Calella, P.; Hernández, J.N.; González, V.F.; Lira, S.M.; Torraca, S.; Arronte, R.U.; Cirillo, P.; Minutolo, R.; Montúfar Cárdenas, R.A. Safety and effectiveness of low-protein diet supplemented with ketoacids in diabetic patients with chronic kidney disease. BMC Nephrol. 2018, 19, 110. [Google Scholar]
- Aparicio, M.; Chauveau, P.; Combe, C. Low protein diets and outcome of renal patients. J. Nephrol. 2001, 14, 433–439. [Google Scholar] [PubMed]
- Zoccali, C.; Mallamaci, F. Moderator’s view: Low-protein diet in chronic kidney disease: Effectiveness, efficacy and precision nutritional treatments in nephrology. Nephrol. Dial. Transplant. 2018, 33, 387–391. [Google Scholar] [CrossRef] [PubMed]
- Brunori, G.; Viola, B.F.; Parrinello, G.; De Biase, V.; Como, G.; Franco, V.; Garibotto, G.; Zubani, R.; Cancarini, G.C. Efficacy and safety of a very-low-protein diet when postponing dialysis in the elderly: A prospective randomized multicenter controlled study. Am. J. Kidney Dis. 2007, 49, 569–580. [Google Scholar] [CrossRef] [PubMed]
- Misra, M.; Nolph, K. Efficacy and safety of a very-low-protein diet in the elderly: What are the options? Am. J. Kidney Dis. 2008, 51, 530–531. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, G.B.; Leone, F.; Attini, R.; Parisi, S.; Fassio, F.; Deagostini, M.C.; Ferraresi, M.; Clari, R.; Ghiotto, S.; Biolcati, M.; et al. Association of low-protein supplemented diets with fetal growth in pregnant women with CKD. Clin. J. Am. Soc. Nephrol. 2014, 9, 864–873. [Google Scholar] [CrossRef] [PubMed]
- Jiang, N.; Qian, J.; Sun, W.; Lin, A.; Cao, L.; Wang, Q.; Ni, Z.; Wan, Y.; Linholm, B.; Axelsson, J.; et al. Better preservation of residual renal function in peritoneal dialysis patients treated with a low-protein diet supplemented with keto acids: A prospective, randomized trial. Nephrol. Dial. Transplant. 2009, 24, 2551–2558. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-B.; Cheng, B.-C.; Kao, T.-W. A comparison of progression of chronic renal failure: low dose vs. standard dose ketoacids. Kidney Res. Clin. Pract. 2012, 31, A24. [Google Scholar] [CrossRef][Green Version]
- Shimomura, A.; Matsui, I.; Hamano, T.; Ishimoto, T.; Katou, Y.; Takehana, K.; Inoue, K.; Kusunoki, Y.; Mori, D.; Nakano, C.; et al. Dietary l-Lysine Prevents Arterial Calcification in Adenine-Induced Uremic Rats. JASN 2014, 25, 1954–1965. [Google Scholar] [CrossRef] [PubMed]
- Clinical practice guidelines for nutrition in chronic renal failure. K/DOQI, National Kidney Foundation. Am. J. Kidney Dis. 2000, 35, S1–S140. [Google Scholar]
- Cano, N.; Fiaccadori, E.; Tesinsky, P.; Toigo, G.; Druml, W.; DGEM (German Society for Nutritional Medicine); Kuhlmann, M.; Mann, H.; Hörl, W.H.; ESPEN (European Society for Parenteral and Enteral Nutrition). ESPEN Guidelines on Enteral Nutrition: Adult renal failure. Clin. Nutr. 2006, 25, 295–310. [Google Scholar] [CrossRef]
- Campbell, K.L.; Rangan, G.K.; Lopez-Vargas, P.; Tong, A. KHA-CARI Autosomal Dominant Polycystic Kidney Disease Guideline: Diet and Lifestyle Management. Semin. Nephrol. 2015, 35, 572–581. [Google Scholar] [CrossRef] [PubMed]
- Levin, A.; Stevens, P.E. Summary of KDIGO 2012 CKD Guideline: Behind the scenes, need for guidance, and a framework for moving forward. Kidney Int. 2014, 85, 49–61. [Google Scholar] [CrossRef] [PubMed]
- Carrero, J.J.; Stenvinkel, P.; Cuppari, L.; Ikizler, T.A.; Kalantar-Zadeh, K.; Kaysen, G.; Mitch, W.E.; Price, S.R.; Wanner, C.; Wang, A.Y.M.; et al. Etiology of the protein-energy wasting syndrome in chronic kidney disease: A consensus statement from the International Society of Renal Nutrition and Metabolism (ISRNM). J. Ren. Nutr. 2013, 23, 77–90. [Google Scholar] [CrossRef] [PubMed]
- Kalantar-Zadeh, K.; Fouque, D. Nutritional Management of Chronic Kidney Disease. N. Engl. J. Med. 2017, 377, 1765–1776. [Google Scholar] [CrossRef] [PubMed]
- Mennini, F.S.; Russo, S.; Marcellusi, A.; Quintaliani, G.; Fouque, D. Economic effects of treatment of chronic kidney disease with low-protein diet. J. Ren. Nutr. 2014, 24, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Piccoli, G.B.; Nazha, M.; Capizzi, I.; Vigotti, F.N.; Scognamiglio, S.; Consiglio, V.; Mongilardi, E.; Bilocati, M.; Avagnina, P.; Versino, E. Diet as a system: An observational study investigating a multi-choice system of moderately restricted low-protein diets. BMC Nephrol. 2016, 17, 197. [Google Scholar] [CrossRef] [PubMed]
| Component Name | mg/pill |
|---|---|
| Ca-Keto-dl-isoleucine | 67 |
| Ca-Ketoeucine | 101 |
| Ca-Ketophénylalanine | 68 |
| Ca-Ketovaline | 86 |
| Ca-Hydroxy-dl-methionine | 59 |
| l-Lysine monoacetate | 105 |
| l-Threonine | 53 |
| l-Tryptophan | 23 |
| l-Histidine | 38 |
| l-Tyrosine | 30 |
| Study | Models | Diet Intervention | Follow-Up | Results (LPD vs. VLDP/LPD + KAs) |
|---|---|---|---|---|
| Wang et al., 2018 [8] | 5/6 nephrectomy rats | NPD: 22% protein vs. LPD: 6% protein vs. LPD + KAs: 5% protein plus 1% KA | 24 weeks | ↓ muscle atrophy ↑ activities of mitochondrial electron transport chain complexes and mitochondrial respiration, ↓ muscle oxidative damage ↑body weight |
| Liu et al., 2018 [9] | KKAy mice, an early type 2 DN model | NPD: 22% protein vs. LPD: 6% protein vs. LPD + KAs: 5% protein plus 1% KA | 12 weeks | ↓ proteinuria ↓ mesangial proliferation and oxidative stress ↑ serum albumin and body weight No difference in creatinine and GFR |
| Zhang et al., 2016 [10] | 3/4 nephrectomy rats | NPD: 18% protein vs. LPD: 6% protein vs. LPD + KAs: 5% protein plus 1% KA | 12 weeks | ↓ proteinuria ↓ intrarenal RAS activation. ↓ transforming growth factor-β1 in the mesangial cells |
| Zhang et al., 2015 [11] | 5/6 nephrectomy rats | NPD: 11 g/kg/day protein vs. LPD: 3 g/kg/day protein vs. LPD + KAs: 3 g/kg/day protein which including 5% protein plus 1% KA | 24 weeks | ↑ body weight, gastrocnemius muscle mass ↓ autophagy marker in muscle No difference of inflammation markers |
| Wang et al., 2014 [12] | 5/6 nephrectomy rats | NPD: 22% protein vs. LPD: 6% protein vs. LPD + KAs: 5% protein plus 1% KA | 24 weeks | ↑improved protein synthesis and increased related mediators such as phosphorylated Akt in the muscle ↓ protein degradation and proteasome activity in the muscle |
| Gao et al., 2010 [13] | 5/6 Nephrectomy rats | NPD: 22% protein vs. LPD: 6% protein vs. LPD + KAs: 5% protein plus 1% KA | 24 weeks | ↓ proteinuria, glomerular sclerosis, and tubulointerstitial fibrosis ↑renal function ↑ body weight and albumin ↓ lipid and protein oxidative products |
| Gao et al., 2011 [14] | 5/6 Nephrectomy rats | NPD: 22% protein vs. LPD: 6% protein vs. LPD + KAs: 5% protein plus 1% KA | 6 months | ↑ body weight and albumin ↑ Kruppel-like factor-15, a transcription factor shown to reduce fibrosis |
| Maniar et al., 1992 [15] | 5/6 Nephrectomy rats | NPD: 16% casein vs. LPD + EAA: 6% casein + EAA vs. LPD + KAs: 6% casein + KA | 3 months | No difference on body weight No difference on proteinuria vs. LDP + EAA but reduction vs. NPD ↓ creatinemia, proteinuria, glomerular sclerosis, and tubulointerstitial fibrosis vs. NPD but no difference vs. LPD + EAA ↑survival vs. NPD but no difference vs. LPD + EAA |
| Laouari et al., 1991 [16] | 5/6 Nephrectomy rats | NPD: 12% casein vs. LPD + EAAs: 5% casein + EAA vs. LPD + KAs: 5% casein + KA | ↓Appetite and growth No increase in BCAAs | |
| Benjelloun et al., 1993 [17] | Rats with after a single 5 mg/kg intravenous injection of Adriamycin: a model of induces glomerular damage in glomerulonephritis. | NPD: 21% protein vs. LPD + KAs: 6% protein plus KA | 15 days | ↓ proteinuria ↓ glycosaminoglycan excretion and glomerular glycosaminoglycan contents |
| Barsotti et al; 1988 [18] | 5/6 Nephrectomy rats | NPD: 20.5% protein vs. LPD + KAs: 3.3% protein plus 7.5% KA | 3 months | ↑survival ↑ GFR ↓ proteinuria and histological damage of kidney No difference in body weight and albuminuria |
| Meisinger et al., 1987 [19] | 5/6 Nephrectomy rats | LPD: 8% protein vs. LPD + KAs: 8% protein plus KA | 3 months | ↓ proteinuria |
| Study | Design of Study | Diet | Follow-Up | Results | Comments |
|---|---|---|---|---|---|
| Milovanova et al., 2018 [3] | RCT n = 42 in LPD + KA vs. LPD n = 37 Non-diabetic CKD 3B–4 | LPD (0.6 g/kg of body weight/day, comprising 0.3 g of vegetable protein and 0.3 g of animal protein, phosphorus content ≤ 800 mg/day and calories: 34–35 kcal/kg/day) vs. LPD + KA: 0.6 g/kg of body weight/day | 14 months | ↑ eGFR (29.1 L/min/1.73 m2 vs. 26.6) ↓SBP ↑BMI and muscle body mass NO change in albumin levels No change in lipids parameters ↓ phosphate, FGF23, and PTH levels ↑Klotho levels and phosphate binder uses ↑bicarbonates levels | Similar protein intake in both group Long follow up |
| Di Iorio et al., 2018 [20] | RCT, crossover trial CKD stages 3B–4 Group A1: 3 months of FD, 6 months of VLPD + KA, 3 months of FD and 6 months of MD Group B: 3 months of FD, 6 months of MD, 3 months of FD and 6 months of VLPD + KA. n = 30 in each group | FD: proteins 1 g/kg body weight (bw)/day (animal proteins 50–70 g/day, vegetal proteins 15–20 g/day), energy 30–35 kcal/bw/day, calcium (Ca) 1.1–1.3 g/day, phosphorus (P) 1.2–1.5 g/day, sodium (Na) 6 g/day and potassium (K) 2–4 g/day. MD: proteins 0.7–0.8 g/kg bw/day (animal proteins 30–40 g/day, vegetal proteins 40–50 g/day), energy 30–35 kcal/bw/day, Ca 1.1–1.3 g/day, P 1.2–1.5 g/day, Na 2.5–3 g/day and K 2–4 g/day. VLPD + KA: proteins 0.3–0.5 g/kg bw/day (animal proteins 0 g/day, vegetal proteins 30–40 g/day), energy 30–35 kcal/bw/day, Ca 1.1–1.3 g/day, P 0.6–0.8 g/day, Na 6 g/day, K 2–4 g/day plus a mixture of KA | 6 months | ↓ SBP No change in creatinuria ↓proteinuria ↓ phosphate, FGF23, and PTH levels ↑bicarbonates levels ↑Hg levels ↓protein carbamylation | Sodium intake and phosphore intake was reduce in VLDP + KA group |
| Garneata et al., 2016 [21] | RCT CKD stage 4–5, proteinuria < 1 g/24 h n = 207 | LPD = 0.6 g protein/kg per day vs. VLPD + KA = vegetarian diet, 0.3 g protein/kg per day + KA | 15 months | ↓ RRT initiation or a >50% reduction in the initial GFR (13% in KA+LDP vs. 42% in LPD reached the primary composite efficacy point i.e., RRT initiation or a >50% reduction in the initial GFR) ↓CRP ↑bicarbonates levels ↓uric acid ↓ phosphate, FGF23 and PTH levels and phosphate binder uses No difference in proteinuria No difference of death and CV events No difference of albumin, BMI No change in lipids parameters | Long follow up Large effective Only 14% of patients screened was included |
| Di Iorio et al., 2012 [22] | RCT, crossover trial eGFR < 55 and > 20 mL/min/1.73 m2 Group A: VLDP + KA during the first week and LPD during the second week Group B: LPD during the first week and a VLPD + KA during the second week. n = 16 in each group | LPD = 0.6 g protein/kg per day vs. VLPD + KA = 0.3 g protein/kg per day + KA | 1 week | ↓ phosphate (−12%), FGF23 (−33.5) No change on calcium a post hoc of this study, ↓ indoxyl sulfate [23] ↑bicarbonates levels | Short exposition |
| Di Iorio et al., 2009 [24] | RCT, crossover trial eGFR < 55 and > 20 mL/min Group A: VLDP + KA during 6 month and a LPD during 6 month Group B: LPD during 6 month and a VLDP + KA during 6 month. n = 16 in each group 32 patients | LPD = 0.6 g protein/kg per day vs. VLPD + KA = 0.3 g protein/kg per day + KA | 6 months | ↓proteinuria and AGE | Open label Phosphor intake was different and lower in VLDP+ KA |
| Menon et al., 2009 [25] | Post hoc study of MDRD study B CKD stage 4 nondiabetic n = 255 | LPD = 0.6 g protein/kg per day vs. VLPD + KA = 0.3 g protein/kg per day + KA | 10.2 years | No delay progression to kidney failure ↑the risk of death. | Long follow up without intervention -Observance and protein intake was not monitored during the follow up |
| Teplan et al., 2008 [4] | RCT, double-blind placebo CKD stage 4 n = 111 | LDP: 0.6 g protein/kg per day vs. LPD + KA: 0.6 g protein/kg per day + KA | 36 months | ↓ADMA ↓ BMI and visceral body fat in obese patients ↓proteinuria ↓ glycated hemoglobin ↓LDL-cholesterol | Mean BMI was > 30 kg/m2 at the inclusion Long follow up No difference of protein intake Using a placebo |
| Mircescu et al., 2007 [26] | RCT eGFR <30 mL/min/1.73 m2, nondiabetic n = 53 | VLPD + KA =0.3 g/kg vegetable proteins + KA vs. LPD =0.6 g/kg/d) | 48 weeks | ↑bicarbonates levels ↑calcium levels and ↓ phosphate lower percentages of patients in group I required renal replacement therapy initiation (4% vs. 27%). No change of rate of eGFR and proteinuria No change in SBP | Open label |
| Gennari et al., 2006 [27] | Post hoc study of MDRD study RCT CKD stage 4–5 n = 255 | LPD = 0.6 g protein/kg per day vs. VLPD + KA = 0.3 g protein/kg per day + KA | 2,2 years | No significant effect of diet on serum total CO2 was seen | |
| Menon et al., 2005 [28] | Post oc study of MDRD study RCT CKD stage 4–5 n = 255 | LPD = 0.6 g protein/kg per day vs. VLPD + KA = 0.3 g protein/kg per day + KA | 2.2 years | ↓ homocysteinemia by 24% at 1 year | |
| Feiten et al., 2005 [29] | RCT n = 24 eGFR <25 mL/min | VLPD + KA = 0.3 g/kg vegetable proteins + KA vs. LPD = 0.6 g/kg/d | 4 months | ↑bicarbonates levels No change on calcium levels ↓ phosphate and PTH Decrease the progression of renal decline function of rate of eGFR No change in lipid parameters No change in nutritional status (BMI, albumin) | Open label Short time of follow up Significant reduction in dietary phosphorus (529 ± 109 to 373 ± 125 mg/day, p < 0.05) |
| Prakash et al., 2004 [30] | RCT, double-blind placebo eGFR:28 mL/min/1.73 m2 n = 34 | LPD = 0.6 g protein/kg per day + placebo vs. VLPD = 0.3 g protein/kg per day + KA | 9 months | preserve mGFR (−2% in LDP + KA vs. −21% in LPD) No effect on proteinuria No effect of BMI and albumin | Measure of GFR with 99mTc-DTPA The placebo is problematic because protein intake was different between both groups. |
| Teplan et al., 2003 [5] | RCT eGFR: 22–36 mL/min/1.73 m2 n = 186 | LPD 0.6 g protein/kg per day + rhuEPO + KA vs. LPD: 0.6 g protein/kg per day + rhuEPO vs. LPD: 0.6 g protein/kg per day | 3 years | Slower progression of CKD ↓proteinuria ↓LDL-cholesterol No change in SBP ↑albumin ↑ plasmatic leucine levels | Role of rhuEPO unclear Insulin clearance |
| Di Iorio et al., 2003 [31] | RCT eGFR: < or =25 mL/min/1.73 m2 n = 10 in each group | LPD = 0.6 g protein/kg per day vs. VLPD = 0.3 g protein/kg per day + KA | 2 years | No difference on hemoglobin ↓ EPO dose ↓ phosphate and PTH No change in BMI and albumin No difference in the rate of RRT initiation (8 vs. 7) Slower rate of GFR decline (creatinine clearance) ↓SBP and 24 h NA excretion ↓LDL-cholesterol | Very few populations |
| Bernhard et al., 2001 [6] | RCT CKD stage 4–5 n = 6 in each group | LPD = 0.6 g protein/kg per day vs. LPD + KA = 0.6 g protein/kg per day + KA | 3 months | No difference could be attributed to the ketoanalogs total body flux and leucine oxidation No difference on phosphorus, calcium levels No difference on BMI and albumin No difference in renal function and proteinuria No difference on bicarbonatemia | KA is metabolically safe Short follow-up Small effective |
| Malvy et al., 1999 [32] | RCT eGFR<20 mL/min/1.73 m2 n = 50 | LPD:LPD = 0.65 g protein/kg per day + Ca+ vs. VLPD + KA = 0.3 g protein/kg per day + KA | 3 months or time to eGFR < 5 mL/min/1.73 m2 or RRT | No difference on GFR progression ↑calcium levels ↓ phosphate and PTH No difference on lipid parameters | |
| Kopple et al., 1997 [33] | Post hoc study of MDRD study RCT CKD stage 4–5 n = 255 | LPD = 0.6 g protein/kg per day vs. VLPD + KA = 0.3 g protein/kg per day + KA | 2,2 years | No difference of death and first hospitalization ↑ albumin ↓ transferrin, body wt, percent body fat, arm muscle area, and urine creatinine excretion No correlation between nutritional parameters and death or hospitalization ↓ energy intake | |
| Levey et al., 1996 [34] | Post hoc study of MDRD study RCT CKD stage 4–5 n = 255 | LPD = 0.6 g protein/kg per day vs. VLPD + KA = 0.3 g protein/kg per day + KA | 2.2 years | A 0.2 g/kg/d lower achieved total protein intake was associated with a 1.15 mL/min/yr slower mean decline in GFR (p = 0.011), which is equivalent to 29% of the mean GFR decline | Reanalyze of MDRD study by using correlations of protein intake with a rate of decline in GFR and time to renal failure |
| Klahr et al., 1994 Study 2 [35] | RCT CKD stage 4–5 n = 255 | LPD = 0.6 g protein/kg per day vs. VLPD + KA = 0.3 g protein/kg per day + KA | 27 months | Marginally slower eGFR decline (−19% in LPD vs. 12% in VLDP + KA, p 0.067) No significant interactions between blood-pressure interventions and the rate of decline in eGFR No difference on albumin No difference in proteinuria | -Large RCT study -Good adherence of diet -Measured GFR with iothalamate |
| Coggins et al. 1994 [36] | Feasibility phase of the MDRD Study eGFR: 8 to 56 mL/min/1.73 m2 n = 96 25 participants were excluded | LPD = 0.6 g protein/kg per day vs. VLPD + KA = 0.3 g protein/kg per day + KA | 6 months | No difference on lipid parameters | Pilot study |
| Lindenau et al. 1990 [37] | RCT eGFR<15 mL/min/1.73 m2 n = 40 | LPD = 0.6 g protein/kg per day + Ca+ vs. VLPD + KA = 0.4 g protein/kg per day + KA | 12 months | Improvement in osteo-fibrotic as well as in osteo-malacic changes | A calcium supplementation was given in LPD diet as a control for KA |
| Jungers et al. 1987 [38] | RCT CKD stage 5 n = 19 | LPD = 0.6 g protein/kg per day + Ca+ vs. VLPD + KA = 0.4 g protein/kg per day + KA | 12 months | No difference on biochemical or morphometric sign of de-nutrition ↑mean renal survival duration until dialysis | Small and effective |
| Hecking et al., 1982 [7] | RCT Mean eGFR: 10.8 mL/min/1.73 m2 n = 15 | LPD = 0.6 g protein/kg per day + Ca+ vs. LPD + KA = 0.6 g protein/kg per day + KA or EAA or placebo | 3 weeks per periods | ↓ phosphate No difference on GFR and proteinuria No difference on lipids parameters No difference on albumin | Small and effective versus the placebo |
| CKD Stage | NKF/KDOQI Clinical Practice Guidelines for Nutrition in Chronic Renal Failure, 2000 [79] | ESPEN Guidelines on Enteral Nutrition: Adult Renal Failure, 2006 [80] | Australian KHA-CARI Guidelines [81] | KDIGO, 2012 [82] | International Society of Renal Nutrition and Metabolism, 2013 [83] | Review, 2017, NEJM [84] |
|---|---|---|---|---|---|---|
| 3 | 0.60–0.75 g/kg/day of protein | 0.55–0.6 g/kg/day of protein | 0.75–1.0 g/kg/day of protein | <1.3 g/kg/day protein | 0.6 and 0.8 g/kg/day protein, ≥50% of protein of HBV | <1.0 g/kg/day protein (consider 0.6–0.8 if eGFR <45 mL/min/1.73 m2 or rapid progression) |
| 4–5 | 0.60–0.75 g/kg/day of protein | 0.55–0.6 g/kg/day of protein (2/3 HBV) or ~0.3 g/kg/day of protein supplemented with KAs/EAAs (0.1 g/kg/day) | 0.75–1.0 g/kg/day | 0.8 g/kg/day protein | 0.6 and 0.8 g/kg/day protein, ≥50% of protein of HBV | 0.6–0.8 g/kg/day protein, including 50% HBV protein, or <0.6 with addition of EAAs or KAs (0.1 g/kg/day) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koppe, L.; Cassani de Oliveira, M.; Fouque, D. Ketoacid Analogues Supplementation in Chronic Kidney Disease and Future Perspectives. Nutrients 2019, 11, 2071. https://doi.org/10.3390/nu11092071
Koppe L, Cassani de Oliveira M, Fouque D. Ketoacid Analogues Supplementation in Chronic Kidney Disease and Future Perspectives. Nutrients. 2019; 11(9):2071. https://doi.org/10.3390/nu11092071
Chicago/Turabian StyleKoppe, Laetitia, Mariana Cassani de Oliveira, and Denis Fouque. 2019. "Ketoacid Analogues Supplementation in Chronic Kidney Disease and Future Perspectives" Nutrients 11, no. 9: 2071. https://doi.org/10.3390/nu11092071
APA StyleKoppe, L., Cassani de Oliveira, M., & Fouque, D. (2019). Ketoacid Analogues Supplementation in Chronic Kidney Disease and Future Perspectives. Nutrients, 11(9), 2071. https://doi.org/10.3390/nu11092071






