Cancer and Mediterranean Diet: A Review
Abstract
1. Introduction
2. Methodology
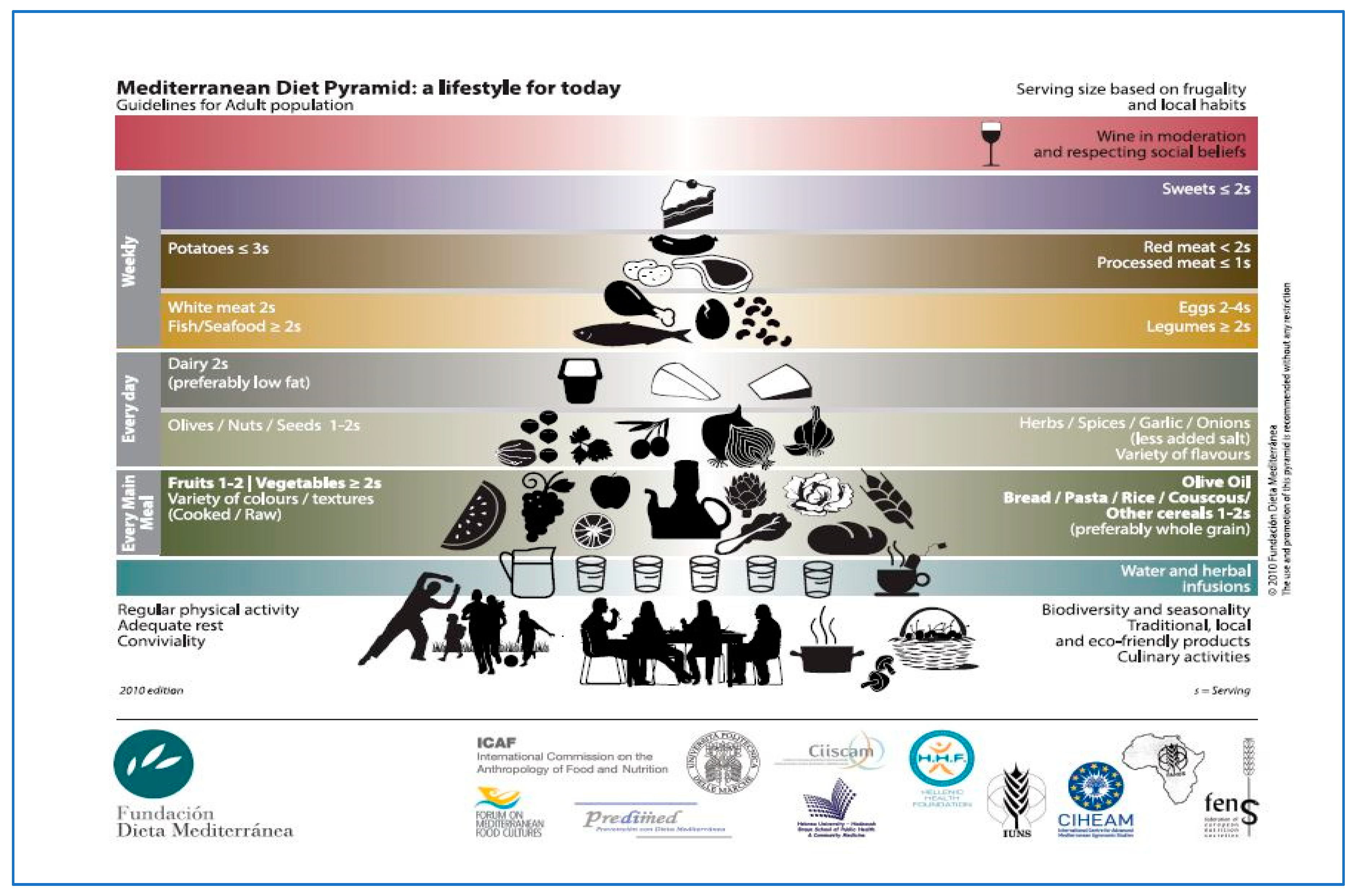
3. Mediterranean Diet
3.1. Mediterranean Diet: Open Issues
3.2. Scores for Measuring the MD Adherence
3.3. Beneficial Effects of Mediterranean Diet in the Cancer Prevention
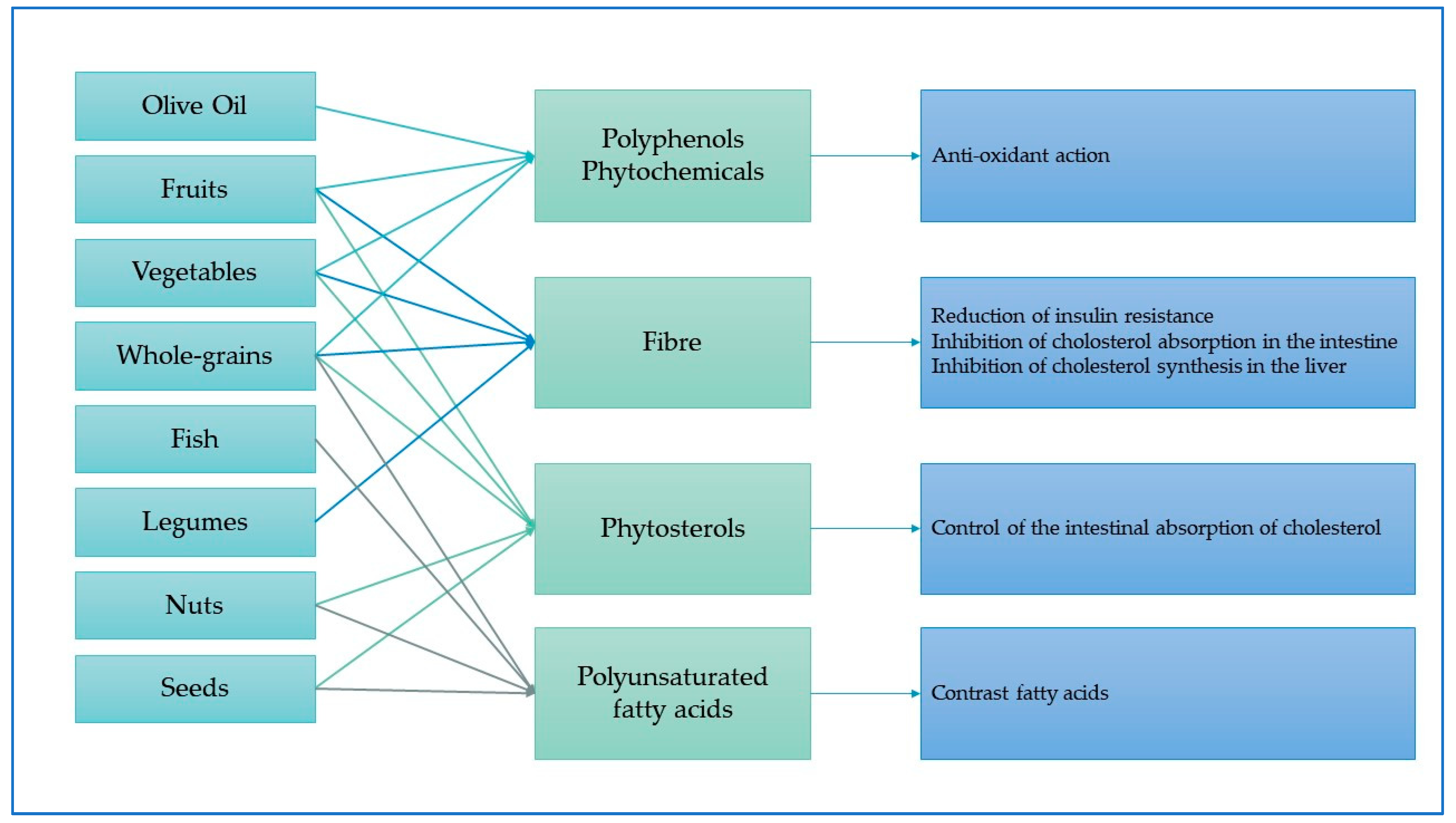
3.4. Mechanisms between Mediterranean Diet Foods and Types of Cancers
- -
- lowering of lipid and modulating of their effects;
- -
- anti-inflammatory, anti-oxidative and anti-aggregating effects;
- -
- modulation of cancer-prone mediators (hormones or growth factors);
- -
- reduction, through the changes in amino acid content, of stimulation of hormones or other extra- and intracellular transmitting pathways involved in cancer;
- -
- changes in gut microbiota thanks to a positive and modified production of bacterial metabolites.
4. Impact of Mediterranean Diet on Cancer
4.1. Breast Cancer
4.2. Colorectal Cancer (CRC)
4.3. Prostate Cancer (PCa)
4.4. Gastric Cancer (GC)
4.5. Bladder Cancer
4.6. Malignant Tumors of the Female Reproductive System
4.7. Head-Neck Cancer (HNC)
4.8. Biliary Tract Cancer (BTC), Pancreatic Tumors
4.9. Lung Cancer
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
| Typical Foods | Elements | Function | Cancer |
|---|---|---|---|
| Fruits & Vegetables | Antioxidants and micronutrients (carotenoids, vitamin C, vitamin E, selenium, dietary fiber, dithiolthiones, glucosinates, polyphenols, protease inhibitors, allium compounds, plant sterols, and limonene) | Anti-tumorigenic effect | Less risk of: -Epithelial cancer -Digestive tract cancer -Breast cancer -Female genital tract cancer -Urinary tract cancer |
| Fish | Long-chain omega-3 fatty acids docosahexaenoic acid and eicosapentaenoic acid | Reducing tumor cell growth Modulation of transcription factor activity and signal transduction Alteration of oestrogen metabolism | Less risk of: -Liver cancer -Colorectal cancer |
| Heterocyclic amines and polycyclic aromatic hydrocarbons may be formed when fish is cooked on a grill or barbecue | Production of mutagenic chemicals | High risk of stomach cancer | |
| Olive oil | Polyphenols (oleuropein and hydroxytyrosol) | Antioxidant activity, anti-inflammatory and anti-mutagenic effects | Less risk of: -breast cancer -ovarian cancer -upper aero-digestive tract cancer -colorectal cancer |
| Oleic acid, poly unsaturated fatty acids (PUFA), low n-6 PUFA/n-3 PUFA ratio | Chemoprotective effect | ||
| Meat | Heterocyclic amines and polycyclic aromatic hydrocarbons formed when meat is cooked at high temperatures | Carcinogens | High risk of: -colorectal cancer -nasopharynx cancer -ung cancer -pancreatic cancer -bladder cancer -esophagus cancer (squamous cell carcinoma) -stomach (no-cardia) cancer |
| Haem iron, present in high level | Promotion of tumorigenesis by stimulating the endogenous formation of carcinogenic N-nitroso compounds | ||
| High-temperature cooking of red and processed meats may enhance production of advanced glycation endproducts (AGEs). | Produce several cancer-promoting effects | High risk of pancreatic cancer | |
| Consumption of meat may lead to insulin resistance and hyperinsulinemia, promoting growth of cancer cells | Promoting growth of cancer cells | ||
| Whole grains | Provide various nutrients: vitamin E, selenium, copper, zinc and bioactive non-nutrient compounds (lignans, phytoestrogens, and phenolic compounds), and dietary fiber | Anti-carcinogenic properties, as anti-oxidative activity Reduce insulin resistance | Less risk of: -colorectum cancer -upper aero-digestive tract -stomach cancer -breast cancer -ovarian cancer -kidney cancer |
| Aflatoxin (mycotoxin produced by molds of the Aspergillus species) | High mutation load in TP3 | High risk of liver cancer | |
| Dairy Products | Calcium, lactic acid-producing bacteria, vitamin D, linoleic acids, lactoferrin, | Inhibit tumor development | Less risk of: -breast cancer (pre-menopausal and post-menopausal women) -colorectal cancer |
| High level of calcium | Downregulating the formation of the biologically active form of vitamin D → increasing cellular proliferation | Higher risk of prostate cancer | |
| Red Wine | Phytoalexin presents in grape skin | Antioxidant and cancer chemo preventive agent → inhibiting tumor initiation, promotion and progression | Controversial results about impact |
| Resveratrol and quercetin | Modulating cell cycle-regulating proteins Inducing apoptosis in multiple carcinoma cell lines Anti-inflammatory, growth → inhibiting activity and immunomodulation properties |
References
- World Health Organization. Cancer. Available online: www.who.int/news-room/fact-sheets/detail/cancer (accessed on 30 June 2019).
- World Cancer Research Fund. Worldwide Cancer Data. Available online: https://www.wcfr.org/dietandcancer/cancer-trend/worldwide-cancer-data (accessed on 30 June 2019).
- Barchitta, M.; Maugeri, A.; Quattrocchi, A.; Agrifoglio, O.; Scalisi, A.; Agodi, A. The Association of Dietary Patterns with High-Risk Human Papillomavirus Infection and Cervical Cancer: A Cross-Sectional Study in Italy. Nutrients 2018, 10, 469. [Google Scholar] [CrossRef] [PubMed]
- Ricceri, F.; Giraudo, M.T.; Fasanelli, F.; Milanese, D.; Sciannameo, V.; Fiorini, L.; Sacerdote, C. Diet and Endometrial Cancer: A Focus on the Role of Fruit and Vegetable Intake, Mediterranean Diet and Dietary Inflammatory Index in the Endometrial Cancer Risk. BMC Cancer 2017, 17, 757. [Google Scholar] [CrossRef] [PubMed]
- Hodge, A.M.; Bassett, J.K.; Shivappa, N.; Hebert, J.R.; English, D.R.; Giles, G.G.; Severi, G. Dietary Inflammatory Index, Mediterranean Diet Score, and Lung Cancer: A Prospective Study. Cancer Causes Control 2016, 27, 907–917. [Google Scholar] [CrossRef] [PubMed]
- Bodén, S.; Myte, R.; Wennberg, M.; Harlid, S.; Johansson, I.; Shivappa, N.; Hébert, J.R.; Van Guelpen, B.; Nilsson, L.M. The Inflammatory Potential of Diet in Determining Cancer Risk; A Prospective Investigation of Two Dietary Pattern Scores. PLoS ONE 2019, 14, e0214551. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G. Adherence to Mediterranean Diet and Risk of Cancer: An Updated Systematic Review and Meta-Analysis of Observational Studies. Cancer Med. 2015, 4, 1933–1947. [Google Scholar] [CrossRef] [PubMed]
- Panunzio, M.; Caporizzi, R.; Cela, E.P.; Antoniciello, A.; Di Martino, V.; Ferguson, L.R. Promotion of the Mediterranean Diet Incancer Long-Survivors by Means of the Med-Food Anticancer Program: A Pilot Study. Ann. Ig. 2019, 31, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Ndlovu, T.; van Jaarsveld, F.; Caleb, O.J. French and Mediterranean-Style Diets: Contradictions, Misconceptions and Scientific Facts-A Review. Food Res. Int. 2019, 116, 840–858. [Google Scholar] [CrossRef] [PubMed]
- Donaldson, M.S. Nutrition and Cancer: A Review of the Evidence for an Anti-Cancer Diet. Nutr. J. 2004, 3, 19. [Google Scholar] [CrossRef] [PubMed]
- Vineis, P.; Wild, C.P. Global Cancer Patterns: Causes and Prevention. Lancet 2014, 383, 549–557. [Google Scholar] [CrossRef]
- Solans, M.; Castelló, A.; Benavente, Y.; Marcos-Gragera, R.; Amiano, P.; Gracia-Lavedan, E.; Costas, L.; Robles, C.; Gonzalez-Barca, E.; de la Banda, E.; et al. Adherence to the Western, Prudent, and Mediterranean Dietary Patterns and Chronic Lymphocytic Leukemia in the MCC-Spain Study. Haematologica 2018, 103, 1881–1888. [Google Scholar] [CrossRef] [PubMed]
- Fliss-Isakov, N.; Kariv, R.; Webb, M.; Ivancovsky, D.; Margalit, D.; Zelber-Sagi, S. Mediterranean Dietary Components Are Inversely Associated with Advanced Colorectal Polyps: A Case-Control Study. World J. Gastroenterol. 2018, 24, 2617–2627. [Google Scholar] [CrossRef] [PubMed]
- Torres Stone, R.A.; Waring, M.E.; Cutrona, S.L.; Kiefe, C.I.; Allison, J.; Doubeni, C.A. The Association of Dietary Quality with Colorectal Cancer among Normal Weight, Overweight and Obese Men and Women: A Prospective Longitudinal Study in the USA. BMJ Open 2017, 7, e015619. [Google Scholar] [CrossRef] [PubMed]
- Bamia, C.; Lagiou, P.; Buckland, G.; Grioni, S.; Agnoli, C.; Taylor, A.J.; Dahm, C.C.; Overvad, K.; Olsen, A.; Tjønneland, A.; et al. Mediterranean Diet and Colorectal Cancer Risk: Results from a European Cohort. Eur. J. Epidemiol. 2013, 28, 317–328. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, B.; Bastos, J.; Lunet, N. Dietary Patterns and Colorectal Cancer: A Case-Control Study from Portugal. Eur. J. Cancer Prev. 2011. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Garrett, W.S.; Chan, A.T. Nutrients, Foods, and Colorectal Cancer Prevention. Gastroenterology 2015. [Google Scholar] [CrossRef] [PubMed]
- Gann, P.H. Risk Factors for Prostate Cancer. Rev. Urol. 2002, 4, S3–S10. [Google Scholar] [PubMed]
- Pelser, C.; Mondul, A.M.; Hollenbeck, A.R.; Park, Y. Dietary Fat, Fatty Acids, and Risk of Prostate Cancer in the NIH-AARP Diet and Health Study. Cancer Epidemiol. Prev. Biomark. 2013, 22, 697–707. [Google Scholar] [CrossRef]
- Aune, D.; Navarro Rosenblatt, D.A.; Chan, D.S.M.; Vieira, A.R.; Vieira, R.; Greenwood, D.C.; Vatten, L.J.; Norat, T. Dairy Products, Calcium, and Prostate Cancer Risk: A Systematic Review and Meta-Analysis of Cohort Studies. Am. J. Clin. Nutr. 2014, 101, 87–117. [Google Scholar] [CrossRef]
- Cheung, E.; Wadhera, P.; Dorff, T.; Pinski, J. Diet and Prostate Cancer Risk Reduction. Expert Rev. Anticancer Ther. 2008, 8, 43–50. [Google Scholar] [CrossRef]
- Hardin, J.; Cheng, I.; Witte, J.S. Impact of Consumption of Vegetable, Fruit, Grain, and High Glycemic Index Foods on Aggressive Prostate Cancer Risk. Nutr. Cancer 2011, 63, 860–872. [Google Scholar] [CrossRef]
- Wei, M.Y.; Giovannucci, E.L. Lycopene, Tomato Products, and Prostate Cancer Incidence: A Review and Reassessment in the PSA Screening Era. J. Oncol. 2012. [Google Scholar] [CrossRef] [PubMed]
- Lăcătușu, C.M.; Grigorescu, E.D.; Floria, M.; Onofriescu, A.; Mihai, B.M. The Mediterranean Diet: From an Environment-Driven Food Culture to an Emerging Medical Prescription. Int. J. Environ. Res. Public Health 2019, 16, 942. [Google Scholar] [CrossRef] [PubMed]
- Buckland, G.; González, C.A.; Agudo, A.; Vilardell, M.; Berenguer, A.; Amiano, P.; Ardanaz, E.; Arriola, L.; Barricarte, A.; Basterretxea, M.; et al. Adherence to the Mediterranean Diet and Risk of Coronary Heart Disease in the Spanish EPIC Cohort Study. Am. J. Epidemiol. 2009. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Critselis, E. Mediterranean Diet and Longevity. Eur. J. Cancer Prev. 2004, 13, 453–456. [Google Scholar] [CrossRef] [PubMed]
- Martínez-González, M.Á.; Hershey, M.S.; Zazpe, I.; Trichopoulou, A. Transferability of the Mediterranean Diet to Non-Mediterranean Countries. What Is and What Is Not the Mediterranean Diet. Nutrients 2017, 9, 1226. [Google Scholar] [CrossRef] [PubMed]
- Dinu, M.; Pagliai, G.; Casini, A.; Sofi, F. Mediterranean Diet and Multiple Health Outcomes: An Umbrella Review of Meta-Analyses of Observational Studies and Randomised Trials. Eur. J. Clin. Nutr. 2018, 72, 30–43. [Google Scholar] [CrossRef] [PubMed]
- UNESCO. Representative List of the Intangible Cultural Heritage of Humanity Representative List of the Intangible Cultural Heritage of Humanity; UNESCO: Paris, France, 2013. [Google Scholar]
- Keys, A.; Menotti, A.; Karvonen, M.J.; Aravanis, C.; Blackburn, H.; Buzina, R.; Djordjevic, B.S.; Dontas, A.S.; Fidanza, F.; Keys, M.H.; et al. The diet and 15-year death rate in the seven countries study. Am. J. Epidemiol. 1986, 124, 903–915. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Gonzalez, M.A.; Bes-Rastrollo, M.; Serra-Majem, L.; Lairon, D.; Estruch, R.; Trichopoulou, A. Mediterranean Food Pattern and the Primary Prevention of Chronic Disease: Recent Developments. Nutr. Rev. 2009, 67, S111–S116. [Google Scholar] [CrossRef] [PubMed]
- European Union. Regolamento (CEE). Available online: http://data.europa.eu/eli/reg/1991/2568/2015-01-01 (accessed on 30 June 2019).
- Panagiotakos, D.B.; Pitsavos, C.; Chrysohoou, C.; Stefanadis, C.; Toutouzas, P. Risk Stratification of Coronary Heart Disease in Greece: Final Results from the CARDIO2000 Epidemiological Study. Prev. Med. 2002. [Google Scholar] [CrossRef]
- Dilis, V.; Katsoulis, M.; Lagiou, P.; Trichopoulos, D.; Naska, A.; Trichopoulou, A. Mediterranean Diet and CHD: The Greek European Prospective Investigation into Cancer and Nutrition Cohort. Br. J. Nutr. 2012, 108, 699–709. [Google Scholar] [CrossRef] [PubMed]
- Rizzello, F.; Spisni, E.; Giovanardi, E.; Imbesi, V.; Salice, M.; Alvisi, P.; Valerii, M.C.; Gionchetti, P. Implications of the Westernized Diet in the Onset and Progression of IBD. Nutrients 2019, 11, 1033. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Lagiou, P. Healthy Traditional Mediterranean Diet: An Expression of Culture, History, and Lifestyle. Nutr. Rev. 2009. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F. The Mediterranean Diet Revisited: Evidence of Its Effectiveness Grows. Curr. Opin. Cardiol. 2009, 24, 442–446. [Google Scholar] [CrossRef] [PubMed]
- Prabhakaran, D.; Khandelwal, S.; Martínez-González, M.A.; Tong, T.Y.; Forouhi, N.G.; Trichopoulou, A.; Mozaffarian, D.; de Lorgeril, M. Definitions and Potential Health Benefits of the Mediterranean Diet: Views from Experts around the World. BMC Med. 2014, 12, 112. [Google Scholar] [CrossRef]
- Pérez-Martínez, P.; Mikhailidis, D.P.; Athyros, V.G.; Bullo, M.; Couture, P.; Covas, M.I.; de Koning, L.; Delgado-Lista, J.; Díaz-López, A.; Drevon, C.A.; et al. Lifestyle Recommendations for the Prevention and Management of Metabolic Syndrome: An International Panel Recommendation. Nutr. Rev. 2017, 75, 307–326. [Google Scholar] [CrossRef] [PubMed]
- Pilis, W.; Stec, K.; Zych, M.; Pilis, A. Health Benefits and Risk Associated with Adopting a Vegetarian Diet. Rocz. Panstw. Zakl. Hig. 2014, 65, 9–14. [Google Scholar]
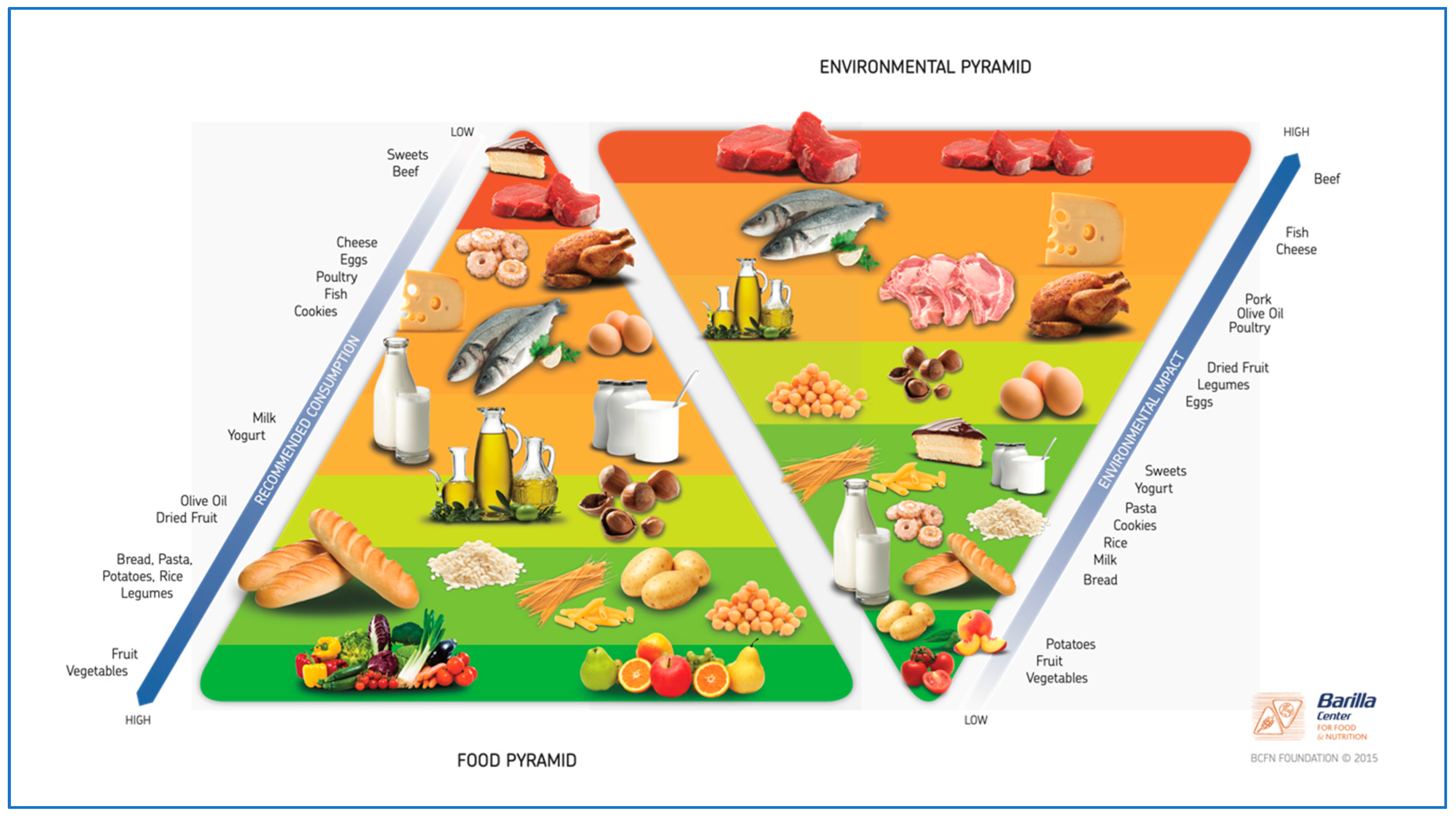
- Dernini, S.; Berry, E.M. Mediterranean Diet: From a Healthy Diet to a Sustainable Dietary Pattern. Front. Nutr. 2015, 2, 15. [Google Scholar] [CrossRef] [PubMed]
- Alsaffar, A.A. Sustainable Diets: The Interaction between Food Industry, Nutrition, Health and the Environment. Food Sci. Technol. Int. 2016, 22, 102–111. [Google Scholar] [CrossRef]
- Bach-Faig, A.; Berry, E.M.; Lairon, D.; Reguant, J.; Trichopoulou, A.; Dernini, S.; Medina, F.X.; Battino, M.; Belahsen, R.; Miranda, G.; et al. Mediterranean Diet Pyramid Today. Science and Cultural Updates. Public Health Nutr. 2011, 14, 2274–2284. [Google Scholar] [CrossRef]
- Bonaccio, M.; Iacoviello, L.; De Gaetano, G. The Mediterranean Diet: The Reasons for a Success. Thromb. Res. 2012, 129, 401–404. [Google Scholar] [CrossRef]
- Ciati, R.; Ruini, L. Double Pyramid: Healthy Food for People, Sustainable Food for the Planet. In Sustainable Diets and Biodiversity Directions and Solutions for Policy, Research and Action; Barilla Center for Food & Nutrition: Parma, ITALY, 2012. [Google Scholar]
- Affret, A.; Severi, G.; Dow, C.; Rey, G.; Delpierre, C.; Boutron-Ruault, M.C.; Clavel-Chapelon, F.; Fagherazzi, G. Socio-Economic Factors Associated with a Healthy Diet: Results from the E3N Study. Public Health Nutr. 2017, 20, 1574–1583. [Google Scholar] [CrossRef] [PubMed]
- Burlingame, B.; Dernini, S. Sustainable Diets and Biodiversity: Directions and Solutions for Policy, Research and Action. Available online: http://www.fao.org/3/a-i3004e.pdf (accessed on 30 June 2019).
- Drewnowski, A.; Specter, S.E. Poverty and Obesity: The Role of Energy Density and Energy Costs. Am. J. Clin. Nutr. 2004, 79, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Darmon, N.; Drewnowski, A. Does Social Class Predict Diet Quality? Am. J. Clin. Nutr. 2008, 87, 1107–1117. [Google Scholar] [CrossRef] [PubMed]
- Seconda, L.; Baudry, J.; Allès, B.; Hamza, O.; Boizot-Szantai, C.; Soler, L.G.; Galan, P.; Hercberg, S.; Lairon, D.; Kesse-Guyot, E. Assessment of the Sustainability of the Mediterranean Diet Combined with Organic Food Consumption: An Individual Behaviour Approach. Nutrients 2017, 9, 61. [Google Scholar] [CrossRef] [PubMed]
- Davis, C.; Bryan, J.; Hodgson, J.; Murphy, K. Definition of the Mediterranean Diet: A Literature Review. Nutrients 2015, 7, 9139–9153. [Google Scholar] [CrossRef] [PubMed]
- Gerber, M.; Hoffman, R. The Mediterranean Diet: Health, Science and Society. Br. J. Nutr. 2015, 113, S4–S10. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean Diet and Survival in a Greek Population. N. Engl. J. Med. 2003. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Abbate, R.; Gensini, G.F.; Casini, A. Accruing Evidence on Benefits of Adherence to the Mediterranean Diet on Health: An Updated Systematic Review and Meta-Analysis. Am. J. Clin. Nutr. 2010. [Google Scholar] [CrossRef]
- Knoops, K.T.B.; De Groot, L.C.P.G.M.; Kromhout, D.; Perrin, A.E.; Moreiras-Varela, O.; Menotti, A.; Van Staveren, W.A. Mediterranean Diet, Lifestyle Factors, and 10-Year Mortality in Elderly European Men and Women: The HALE Project. J. Am. Med. Assoc. 2004. [Google Scholar] [CrossRef]
- Buckland, G.; Agudo, A.; Travier, N.; María Huerta, J.; Cirera, L.; Tormo, M.J.; Navarro, C.; Dolores Chirlaque, M.; Moreno-Iribas, C.; Ardanaz, E.; et al. Adherence to the Mediterranean Diet Reduces Mortality in the Spanish Cohort of the European Prospective Investigation into Cancer and Nutrition (EPIC-Spain). Br. J. Nutr. 2011. [Google Scholar] [CrossRef]
- Schwedhelm, C.; Boeing, H.; Hoffmann, G.; Aleksandrova, K.; Schwingshackl, L. Effect of Diet on Mortality and Cancer Recurrence among Cancer Survivors: A Systematic Review and Meta-Analysis of Cohort Studies. Nutr. Rev. 2016, 74, 737–748. [Google Scholar] [CrossRef] [PubMed]
- Naska, A.; Trichopoulou, A. Back to the Future: The Mediterranean Diet Paradigm. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 216–219. [Google Scholar] [CrossRef] [PubMed]
- De Lorenzo, A.; Alberti, A.; Andreoli, A.; Iacopino, L.; Serrano, P.; Perriello, G. Food Habits in a Southern Italian Town (Nicotera) in 1960 and 1996: Still a Reference Italian Mediterranean Diet? Diabetes Nutr. Metab. 2001, 14, 121–125. [Google Scholar] [PubMed]
- Dernini, S. The Erosion and the Renaissance of the Mediterranean Diet: A Sustainable Cultural Resource. Quad. Mediterrània 2011, 16, 75–82. [Google Scholar]
- Bonaccio, M.; Di Castelnuovo, A.; Costanzo, S.; Persichillo, M.; De Curtis, A.; Donati, M.B.; De Gaetano, G.; Iacoviello, L. Adherence to the Traditional Mediterranean Diet and Mortality in Subjects with Diabetes. Prospective Results from the MOLI-SANI Study. Eur. J. Prev. Cardiol. 2016, 23, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Hauner, H.; Hauner, D. The Impact of Nutrition on the Development and Prognosis of Breast Cancer. Breast Care 2010, 5, 377–381. [Google Scholar] [CrossRef]
- Russo, G.I.; Campisi, D.; Di Mauro, M.; Regis, F.; Reale, G.; Marranzano, M.; Ragusa, R.; Solinas, T.; Madonia, M.; Cimino, S.; et al. Dietary Consumption of Phenolic Acids and Prostate Cancer: A Case-Control Study in Sicily, Southern Italy. Molecules 2017, 22, 2159. [Google Scholar] [CrossRef]
- Turati, F.; Bravi, F.; Polesel, J.; Bosetti, C.; Negri, E.; Garavello, W.; Taborelli, M.; Serraino, D.; Libra, M.; Montella, M.; et al. Adherence to the Mediterranean Diet and Nasopharyngeal Cancer Risk in Italy. Cancer Causes Control 2017, 28, 89–95. [Google Scholar] [CrossRef]
- Ciancarelli, M.G.; Massimo, C.; Amicis, D.; Ciancarelli, I. Mediterranean Diet and Health Promotion: Evidence and Current Concerns. Med. Res. Arch. 2017. [Google Scholar] [CrossRef]
- Machowetz, A.; Poulsen, H.E.; Gruendel, S.; Weimann, A.; Fitó, M.; Marrugat, J.; de la Torre, R.; Salonen, J.T.; Nyyssönen, K.; Mursu, J.; et al. Effect of Olive Oils on Biomarkers of Oxidative DNA Stress in Northern and Southern Europeans. FASEB J. 2007. [Google Scholar] [CrossRef]
- Fitó, M.; Cladellas, M.; de la Torre, R.; Martí, J.; Muñoz, D.; Schröder, H.; Alcántara, M.; Pujadas-Bastardes, M.; Marrugat, J.; Ló-Sabater, M.C.; et al. Anti-Inflammatory Effect of Virgin Olive Oil in Stable Coronary Disease Patients: A Randomized, Crossover, Controlled Trial. Eur. J. Clin. Nutr. 2008. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.; Turati, F.; Lagiou, P.; Trichopoulos, D.; Augustin, L.S.; La Vecchia, C.; Trichopoulou, A. Mediterranean Diet and Glycaemic Load in Relation to Incidence of Type 2 Diabetes: Results from the Greek Cohort of the Population-Based European Prospective Investigation into Cancer and Nutrition (EPIC). Diabetologia 2013, 56, 2405–2413. [Google Scholar] [CrossRef] [PubMed]
- Praud, D.; Bertuccio, P.; Bosetti, C.; Turati, F.; Ferraroni, M.; La Vecchia, C. Adherence to the Mediterranean Diet and Gastric Cancer Risk in Italy. Int. J. Cancer 2014, 134, 2935–2941. [Google Scholar] [CrossRef] [PubMed]
- Pitsavos, C.; Panagiotakos, D.B.; Tzima, N.; Chrysohoou, C.; Economou, M.; Zampelas, A.; Stefanadis, C. Adherence to the Mediterranean Diet Is Associated with Total Antioxidant Capacity in Healthy Adults: The ATTICA Study. Am. J. Clin. Nutr. 2005, 82, 694–699. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Jiménez, J.; Elena Díaz-Rubio, M.; Saura-Calixto, F. Contribution of Macromolecular Antioxidants to Dietary Antioxidant Capacity: A Study in the Spanish Mediterranean Diet. Plant Foods Hum. Nutr. 2015. [Google Scholar] [CrossRef] [PubMed]
- Castelló, A.; Boldo, E.; Pérez-Gómez, B.; Lope, V.; Altzibar, J.M.; Martín, V.; Castaño-Vinyals, G.; Guevara, M.; Dierssen-Sotos, T.; Tardón, A.; et al. Adherence to the Western, Prudent and Mediterranean Dietary Patterns and Breast Cancer Risk: MCC-Spain Study. Maturitas 2017, 103, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; An, S.; Hou, L.; Chen, P.; Lei, C.; Tan, W. Red and Processed Meat Intake and Risk of Bladder Cancer: A Meta-Analysis. Int. J. Clin. Exp. Med. 2014, 7, 2100. [Google Scholar] [PubMed]
- Giacosa, A.; Barale, R.; Bavaresco, L.; Gatenby, P.; Gerbi, V.; Janssens, J.; Johnston, B.; Kas, K.; La Vecchia, C.; Mainguet, P.; et al. Cancer Prevention in Europe. Eur. J. Cancer Prev. 2013, 22, 90–95. [Google Scholar] [CrossRef]
- Bosetti, C.; Turati, F.; Pont, A.D.; Ferraroni, M.; Polesel, J.; Negri, E.; Serraino, D.; Talamini, R.; La Vecchia, C.; Zeegers, M.P. The Role of Mediterranean Diet on the Risk of Pancreatic Cancer. Br. J. Cancer 2013, 109, 1360–1366. [Google Scholar] [CrossRef]
- Filomeno, M.; Bosetti, C.; Garavello, W.; Levi, F.; Galeone, C.; Negri, E.; La Vecchia, C. The Role of a Mediterranean Diet on the Risk of Oral and Pharyngeal Cancer. Br. J. Cancer 2014, 111, 981–986. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Lagiou, P.; Kuper, H.; Trichopoulos, D. Cancer and Mediterranean Dietary Traditions. Cancer Epidemiol. Biomark. Prev. 2000, 9, 869–873. [Google Scholar]
- Pelucchi, C.; Bosetti, C.; Rossi, M.; Negri, E.; La Vecchia, C. Selected Aspects of Mediterranean Diet and Cancer Risk. Nutr. Cancer 2009. [Google Scholar] [CrossRef]
- Verberne, L.; Bach-Faig, A.; Buckland, G.; Serra-Majem, L. Association between the Mediterranean Diet and Cancer Risk: A Review of Observational Studies. Nutr. Cancer 2010. [Google Scholar] [CrossRef] [PubMed]
- Schwingshackl, L.; Hoffmann, G. Does a Mediterranean-Type Diet Reduce Cancer Risk? Curr. Nutr. Rep. 2016, 5, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Fasanelli, F.; Zugna, D.; Giraudo, M.T.; Krogh, V.; Grioni, S.; Panico, S.; Mattiello, A.; Masala, G.; Caini, S.; Tumino, R.; et al. Abdominal Adiposity Is Not a Mediator of the Protective Effect of Mediterranean Diet on Colorectal Cancer. Int. J. Cancer 2017, 140, 2265–2271. [Google Scholar] [CrossRef]
- Bravi, F.; Spei, M.E.; Polesel, J.; Di Maso, M.; Montella, M.; Ferraroni, M.; Serraino, D.; Libra, M.; Negri, E.; La Vecchia, C.; et al. Mediterranean Diet and Bladder Cancer Risk in Italy. Nutrients 2018, 10, 1061. [Google Scholar] [CrossRef] [PubMed]
- Kontou, N.; Psaltopoulou, T.; Soupos, N.; Polychronopoulos, E.; Linos, A.; Xinopoulos, D.; Panagiotakos, D.B. The Role of Number of Meals, Coffee Intake, Salt and Type of Cookware on Colorectal Cancer Development in the Context of the Mediterranean Diet. Public Health Nutr. 2013, 16, 928–935. [Google Scholar] [CrossRef]
- Kontou, N.; Psaltopoulou, T.; Soupos, N.; Polychronopoulos, E.; Xinopoulos, D.; Linos, A.; Panagiotakos, D.B. The Mediating Effect of Mediterranean Diet on the Relation between Smoking and Colorectal Cancer: A Case-Control Study. Eur. J. Public Health 2013, 23, 742–746. [Google Scholar] [CrossRef][Green Version]
- Russo, G.I.; Solinas, T.; Urzì, D.; Privitera, S.; Campisi, D.; Cocci, A.; Carini, M.; Madonia, M.; Cimino, S.; Morgia, G. Adherence to Mediterranean Diet and Prostate Cancer Risk in Sicily: Population-Based Case–Control Study. Int. J. Impot. Res. 2018. [Google Scholar] [CrossRef]
- Lauby-Secretan, B.; Scoccianti, C.; Loomis, D.; Grosse, Y.; Bianchini, F.; Straif, K. Body Fatness and Cancer—Viewpoint of the IARC Working Group. N. Engl. J. Med. 2016. [Google Scholar] [CrossRef]
- Mayne, S.T.; Playdon, M.C.; Rock, C.L. Diet, Nutrition, and Cancer: Past, Present and Future. Nat. Rev. Clin. Oncol. 2016, 13, 504–515. [Google Scholar] [CrossRef] [PubMed]
- Jacobs, D.R.; Gross, M.D.; Tapsell, L.C. Food Synergy: An Operational Concept for Understanding Nutrition. Am. J. Clin. Nutr. 2009. [Google Scholar] [CrossRef] [PubMed]
- Grosso, G.; Buscemi, S.; Galvano, F.; Mistretta, A.; Marventano, S.; La Vela, V.; Drago, F.; Gangi, S.; Basile, F.; Biondi, A. Mediterranean Diet and Cancer: Epidemiological Evidence and Mechanism of Selected Aspects. BMC Surg. 2013, 13, S14. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Li, S.; Meng, X.; Gan, R.Y.; Zhang, J.J.; Li, H. Bin. Dietary Natural Products for Prevention and Treatment of Breast Cancer. Nutrients 2017, 9, 728. [Google Scholar] [CrossRef] [PubMed]
- Amor, S.; Châlons, P.; Aires, V.; Delmas, D. Polyphenol Extracts from Red Wine and Grapevine: Potential Effects on Cancers. Diseases 2018, 6, 106. [Google Scholar] [CrossRef]
- Tosti, V.; Bertozzi, B.; Fontana, L. Health Benefits of the Mediterranean Diet: Metabolic and Molecular Mechanisms. J. Gerontol. Series A Biol. Sci. Med Sci. 2018. [Google Scholar] [CrossRef]
- Akram, M.; Iqbal, M.; Daniyal, M.; Khan, A.U. Awareness and Current Knowledge of Breast Cancer. Biol. Res. 2017, 50, 33. [Google Scholar] [CrossRef]
- van den Brandt, P.A.; Schulpen, M. Mediterranean Diet Adherence and Risk of Postmenopausal Breast Cancer: Results of a Cohort Study and Meta-Analysis. Int. J. Cancer 2017, 140, 2220–2231. [Google Scholar] [CrossRef]
- Tfayli, A.; Temraz, S.; Abou Mrad, R.; Shamseddine, A. Breast Cancer in Low- and Middle-Income Countries: An Emerging and Challenging Epidemic. J. Oncol. 2010. [Google Scholar] [CrossRef]
- Turati, F.; Carioli, G.; Bravi, F.; Ferraroni, M.; Serraino, D.; Montella, M.; Giacosa, A.; Toffolutti, F.; Negri, E.; Levi, F.; et al. Mediterranean Diet and Breast Cancer Risk. Nutrients 2018, 10, 326. [Google Scholar] [CrossRef]
- Buckland, G.; Travier, N.; Cottet, V.; González, C.A.; Luján-Barroso, L.; Agudo, A.; Trichopoulou, A.; Lagiou, P.; Trichopoulos, D.; Peeters, P.H.; et al. Adherence to the Mediterranean Diet and Risk of Breast Cancer in the European Prospective Investigation into Cancer and Nutrition Cohort Study. Int. J. Cancer 2013, 132, 2918–2927. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Nguyen, N.; Colditz, G.A. Links between Alcohol Consumption and Breast Cancer: A Look at the Evidence. Women’s Health 2015, 11, 65–77. [Google Scholar] [CrossRef] [PubMed]
- Shield, K.D.; Soerjomataram, I.; Rehm, J. Alcohol Use and Breast Cancer: A Critical Review. Alcohol. Clin. Exp. Res. 2016, 40, 1166–1181. [Google Scholar] [CrossRef] [PubMed]
- Krusinska, B.; Hawrysz, I.; Wadolowska, L.; Slowinska, M.A.; Biernacki, M.; Czerwinska, A.; Golota, J.J. Associations of Mediterranean Diet and a Posteriori Derived Dietary Patterns with Breast and Lung Cancer Risk: A Case-Control Study. Nutrients 2018, 10, 470. [Google Scholar] [CrossRef] [PubMed]
- Toklu, H.; Nogay, N.H. Effects of Dietary Habits and Sedentary Lifestyle on Breast Cancer among Women Attending the Oncology Day Treatment Center at a State University in Turkey. Niger. J. Clin. Pract. 2018, 21, 1576–1584. [Google Scholar] [CrossRef]
- Reedy, J.; Mitrou, P.N.; Krebs-Smith, S.M.; Wirfält, E.; Flood, A.; Kipnis, V.; Leitzmann, M.; Mouw, T.; Hollenbeck, A.; Schatzkin, A.; et al. Index-Based Dietary Patterns and Risk of Colorectal Cancer. Am. J. Epidemiol. 2008, 168, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Park, S.Y.; Boushey, C.J.; Wilkens, L.R.; Haiman, C.A.; Le Marchand, L. High-Quality Diets Associate With Reduced Risk of Colorectal Cancer: Analyses of Diet Quality Indexes in the Multiethnic Cohort. Gastroenterology 2017, 153, 386–394. [Google Scholar] [CrossRef]
- Barrubés, L.; Babio, N.; Mena-Sánchez, G.; Toledo, E.; Ramírez-Sabio, J.B.; Estruch, R.; Ros, E.; Fitó, M.; Arós, F.; Fiol, M.; et al. Dairy Product Consumption and Risk of Colorectal Cancer in an Older Mediterranean Population at High Cardiovascular Risk. Int. J. Cancer 2018, 143, 1356–1366. [Google Scholar] [CrossRef]
- Aune, D.; Chan, D.S.M.; Vieira, A.R.; Navarro Rosenblatt, D.A.; Vieira, R.; Greenwood, D.C.; Kampman, E.; Norat, T. Red and Processed Meat Intake and Risk of Colorectal Adenomas: A Systematic Review and Meta-Analysis of Epidemiological Studies. Cancer Causes Control. 2013. [Google Scholar] [CrossRef]
- Cai, S.; Li, Y.; Ding, Y.; Chen, K.; Jin, M. Alcohol Drinking and the Risk of Colorectal Cancer Death: A Meta-Analysis. Eur. J. Cancer Prev. 2014, 23, 532–539. [Google Scholar] [CrossRef]
- Johnson, I.T. The Cancer Risk Related to Meat and Meat Products. Br. Med. Bull. 2017, 121, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Castelló, A.; Amiano, P.; Fernández de Larrea, N.; Martín, V.; Alonso, M.H.; Castaño-Vinyals, G.; Pérez-Gómez, B.; Olmedo-Requena, R.; Guevara, M.; Fernandez-Tardon, G.; et al. Low Adherence to the Western and High Adherence to the Mediterranean Dietary Patterns Could Prevent Colorectal Cancer. Eur. J. Nutr. 2018, 58, 1–11. [Google Scholar] [CrossRef]
- Rosato, V.; Guercio, V.; Bosetti, C.; Negri, E.; Serraino, D.; Giacosa, A.; Montella, M.; La Vecchia, C.; Tavani, A. Mediterranean Diet and Colorectal Cancer Risk: A Pooled Analysis of Three Italian Case-Control Studies. Br. J. Cancer 2016, 115, 862–865. [Google Scholar] [CrossRef] [PubMed]
- Ratjen, I.; Schafmayer, C.; di Giuseppe, R.; Waniek, S.; Plachta-Danielzik, S.; Koch, M.; Nöthlings, U.; Hampe, J.; Schlesinger, S.; Lieb, W. Postdiagnostic Mediterranean and Healthy Nordic Dietary Patterns Are Inversely Associated with All-Cause Mortality in Long-Term Colorectal Cancer Survivors. J. Nutr. 2017, 147, 636–644. [Google Scholar] [CrossRef]
- Agnoli, C.; Grioni, S.; Sieri, S.; Palli, D.; Masala, G.; Sacerdote, C.; Vineis, P.; Tumino, R.; Giurdanella, M.C.; Pala, V.; et al. Italian Mediterranean Index and Risk of Colorectal Cancer in the Italian Section of the EPIC Cohort. Int. J. Cancer 2013, 132, 1404–1411. [Google Scholar] [CrossRef] [PubMed]
- Wilson, K.M.; Giovannucci, E.L.; Mucci, L.A. Lifestyle and Dietary Factors in the Prevention of Lethal Prostate Cancer. Asian J. Androl. 2012. [Google Scholar] [CrossRef] [PubMed]
- Erdrich, S.; Bishop, K.S.; Karunasinghe, N.; Han, D.Y.; Ferguson, L.R. A Pilot Study to Investigate If New Zealand Men with Prostate Cancer Benefit from a Mediterranean-Style Diet. PeerJ 2015, 3, e1080. [Google Scholar] [CrossRef] [PubMed]
- Schneider, L.; Su, L.J.; Arab, L.; Bensen, J.T.; Farnan, L.; Fontham, E.T.H.; Song, L.; Hussey, J.; Merchant, A.T.; Mohler, J.L.; et al. Dietary Patterns Based on the Mediterranean Diet and DASH Diet Are Inversely Associated with High Aggressive Prostate Cancer in PCaP. Ann. Epidemiol. 2019, 29, 16–22.e1. [Google Scholar] [CrossRef]
- Kenfield, S.A.; DuPre, N.; Richman, E.L.; Stampfer, M.J.; Chan, J.M.; Giovannucci, E.L. Mediterranean Diet and Prostate Cancer Risk and Mortality in the Health Professionals Follow-up Study. Eur. Urol. 2015, 65, 887–894. [Google Scholar] [CrossRef] [PubMed]
- Richard, A.; Faeh, D.; Rohrmann, S.; Braun, J.; Tarnutzer, S.; Bopp, M. Italianity Is Associated with Lower Risk of Prostate Cancer Mortality in Switzerland. Cancer Causes Control 2014, 25, 1523–1529. [Google Scholar] [CrossRef]
- Plummer, M.; Franceschi, S.; Vignat, J.; Forman, D.; de Martel, C. Global Burden of Gastric Cancer Attributable to Helicobacter Pylori. Int. J. Cancer 2015. [Google Scholar] [CrossRef] [PubMed]
- Schulpen, M.; Peeters, P.H.; van den Brandt, P.A. Mediterranean Diet Adherence and Risk of Pancreatic Cancer: A Pooled Analysis of Two Dutch Cohorts. Int. J. Cancer 2019, 144, 1550–1560. [Google Scholar] [CrossRef] [PubMed]
- de Martel, C.; Forman, D.; Plummer, M. Gastric Cancer. Epidemiology and Risk Factors. Gastroenterol. Clin. N. Am. 2013. [Google Scholar] [CrossRef] [PubMed]
- Karimi, P.; Islami, F.; Anandasabapathy, S.; Freedman, N.D.; Kamangar, F. Gastric Cancer: Descriptive Epidemiology, Risk Factors, Screening, and Prevention. Cancer Epidemiol. Biomark. Prev. 2014. [Google Scholar] [CrossRef]
- McKeown, P.P.; Logan, K.; McKinley, M.C.; Young, I.S.; Woodside, J.V. Session 4: CVD, Diabetes and Cancer Evidence for the Use of the Mediterranean Diet in Patients with CHD. Proc. Nutr. Soc. 2010, 69, 45–60. [Google Scholar] [CrossRef]
- Buckland, G.; Travier, N.; Huerta, J.M.; Bueno-De-Mesquita, H.B.; Siersema, P.D.; Skeie, G.; Weiderpass, E.; Engeset, D.; Ericson, U.; Ohlsson, B.; et al. Healthy Lifestyle Index and Risk of Gastric Adenocarcinoma in the EPIC Cohort Study. Int. J. Cancer 2015, 137, 598–606. [Google Scholar] [CrossRef] [PubMed]
- Piyathilake, C. Dietary Factors Associated with Bladder Cancer. Investig. Clin. Urol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Pelucchi, C.; Bosetti, C.; Negri, E.; Malvezzi, M.; La Vecchia, C. Mechanisms of Disease: The Epidemiology of Bladder Cancer. Nat. Clin. Pract. Urol. 2006. [Google Scholar] [CrossRef] [PubMed]
- Brinkman, M.T.; Buntinx, F.; Kellen, E.; Van Dongen, M.C.J.M.; Dagnelie, P.C.; Muls, E.; Zeegers, M.P. Consumption of Animal Products, Olive Oil and Dietary Fat and Results from the Belgian Case-Control Study on Bladder Cancer Risk. Eur. J. Cancer 2011, 47, 436–442. [Google Scholar] [CrossRef]
- Witlox, W.J.A.; van Osch, F.H.M.; Brinkman, M.; Jochems, S.; Goossens, M.E.; Weiderpass, E.; White, E.; van den Brandt, P.A.; Giles, G.G.; Milne, R.L.; et al. An Inverse Association between the Mediterranean Diet and Bladder Cancer Risk: A Pooled Analysis of 13 Cohort Studies. Eur. J. Nutr. 2019, 1–10. [Google Scholar] [CrossRef]
- Serrano, B.; Brotons, M.; Bosch, F.X.; Bruni, L. Epidemiology and Burden of HPV-Related Disease. Best Pract. Res. Clin. Obstet. Gynaecol. 2018. [Google Scholar] [CrossRef] [PubMed]
- García-Closas, R.; Castellsagué, X.; Bosch, X.; González, C.A. The Role of Diet and Nutrition in Cervical Carcinogenesis: A Review of Recent Evidence. Int. J. Cancer 2005. [Google Scholar] [CrossRef] [PubMed]
- Field, C.J.; Johnson, I.R.; Schley, P.D. Nutrients and Their Role in Host Resistance to Infection. J. Leukoc. Biol. 2002, 71, 16–32. [Google Scholar] [PubMed]
- Li, Y.; Xu, C. Human Papillomavirus-Related Cancers BT—Infectious Agents Associated Cancers: Epidemiology and Molecular Biology; Cai, Q., Yuan, Z., Lan, K., Eds.; Springer: Singapore, 2017; pp. 23–34. [Google Scholar]
- Ferlay, J.; Steliarova-Foucher, E.; Lortet-Tieulent, J.; Rosso, S.; Coebergh, J.W.W.; Comber, H.; Forman, D.; Bray, F. Reprint of: Cancer Incidence and Mortality Patterns in Europe: Estimates for 40 Countries in 2012. Eur. J. Cancer 2015. [Google Scholar] [CrossRef]
- Liang, J.; Shang, Y. Estrogen and Cancer. Annu. Rev. Physiol. 2012. [Google Scholar] [CrossRef]
- Kaaks, R.; Lukanova, A.; Kurzer, M.S. Obesity, Endogenous Hormones, and Endometrial Cancer Risk: A Synthetic Review. Cancer Epidemiol. Biomark. Prev. 2002, 11, 1531–1543. [Google Scholar]
- Onstad, M.A.; Schmandt, R.E.; Lu, K.H. Addressing the Role of Obesity in Endometrial Cancer Risk, Prevention, and Treatment. J. Clin. Oncol. 2016, 34, 4225–4230. [Google Scholar] [CrossRef] [PubMed]
- Gaskins, A.J.; Mumford, S.L.; Zhang, C.; Wactawski-Wende, J.; Hovey, K.M.; Whitcomb, B.W.; Howards, P.P.; Perkins, N.J.; Yeung, E.; Schisterman, E.F. Effect of Daily Fiber Intake on Reproductive Function: The BioCycle Study. Am. J. Clin. Nutr. 2009. [Google Scholar] [CrossRef]
- Filomeno, M.; Bosetti, C.; Bidoli, E.; Levi, F.; Serraino, D.; Montella, M.; La Vecchia, C.; Tavani, A. Mediterranean Diet and Risk of Endometrial Cancer: A Pooled Analysis of Three Italian Case-Control Studies. Br. J. Cancer 2015, 112, 1816–1821. [Google Scholar] [CrossRef]
- Jemal, A.; Bray, F.; Ferlay, J. Global Cancer Statistics: 2011. CA Cancer J. Clin. 2008. [Google Scholar] [CrossRef]
- Li, W.Q.; Park, Y.; Wu, J.W.; Goldstein, A.M.; Taylor, P.R.; Hollenbeck, A.R.; Freedman, N.D.; Abnet, C.C. Index-Based Dietary Patterns and Risk of Head and Neck Cancer in a Large Prospective Study. Am. J. Clin. Nutr. 2014, 99, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Garavello, W.; Lucenteforte, E.; Bosetti, C.; Talamini, R.; Levi, F.; Tavani, A.; Franceschi, S.; Negri, E.; Vecchia, C.L. Diet Diversity and the Risk of Laryngeal Cancer: A Case-Control Study from Italy and Switzerland. Oral Oncol. 2009. [Google Scholar] [CrossRef] [PubMed]
- Chang, E.T.; Adami, H.O. The Enigmatic Epidemiology of Nasopharyngeal Carcinoma. Cancer Epidemiol. Biomark. Prev. 2006. [Google Scholar] [CrossRef] [PubMed]
- Misra, S.; Chaturvedi, A.; Misra, N.C.; Sharma, I.D. Carcinoma of the Gallbladder. Lancet 2003, 4, 167–176. [Google Scholar] [CrossRef]
- Larsson, S.C.; Håkansson, N.; Wolk, A. Healthy Dietary Patterns and Incidence of Biliary Tract and Gallbladder Cancer in a Prospective Study of Women and Men. Eur. J. Cancer 2017, 70, 42–47. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.D.; Bray, F. Cancer Incidence and Mortality Worldwide: IARC Cancer Base. Lyon Int. Agency Res. Cancer 2015. [Google Scholar] [CrossRef]
- McGuigan, A.; Kelly, P.; Turkington, R.C.; Jones, C.; Coleman, H.G.; McCain, R.S. Pancreatic Cancer: A Review of Clinical Diagnosis, Epidemiology, Treatment and Outcomes. World J. Gastroenterol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Rosato, V.; Polesel, J.; Bosetti, C.; Serraino, D.; Negri, E.; La Vecchia, C. Population Attributable Risk for Pancreatic Cancer in Northern Italy. Pancreas 2015, 44, 216–220. [Google Scholar] [CrossRef] [PubMed]
- Maisonneuve, P.; Shivappa, N.; Hébert, J.R.; Bellomi, M.; Rampinelli, C.; Bertolotti, R.; Spaggiari, L.; Palli, D.; Veronesi, G.; Gnagnarella, P. Dietary Inflammatory Index and Risk of Lung Cancer and Other Respiratory Conditions among Heavy Smokers in the COSMOS Screening Study. Eur. J. Nutr. 2016, 55, 1069–1079. [Google Scholar] [CrossRef] [PubMed]
- Mitrou, P.N.; Kipnis, V.; Thiébaut, A.C.M.; Reedy, J.; Subar, A.F.; Wirfält, E.; Flood, A.; Mouw, T.; Hollenbeck, A.R.; Leitzmann, M.F.; et al. Mediterranean Dietary Pattern and Prediction of All-Cause Mortality in a US Population: Results from the NIH-AARP Diet and Health Study. Arch. Intern. Med. 2007, 167, 2461–2468. [Google Scholar] [CrossRef]
- Anic, G.M.; Park, Y.; Subar, A.F.; Schap, T.E.; Reedy, J. Index-Based Dietary Patterns and Risk of Lung Cancer in the NIH-AARP Diet and Health Study. Eur. J. Clin. Nutr. 2016, 70, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Gnagnarella, P.; Maisonneuve, P.; Bellomi, M.; Rampinelli, C.; Bertolotti, R.; Spaggiari, L.; Palli, D.; Veronesi, G. Red Meat, Mediterranean Diet and Lung Cancer Risk among Heavy Smokers in the Cosmos Screening Study. Ann. Oncol. 2013, 24, 2606–2611. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer. Agents Classified by the IARC Monographs; International Agency for Research on Cancer: Lyon, France, 2019; pp. 1–124. Available online: https://monographs.iarc.fr/agents-classified-by-the-iarc/ (accessed on 30 June 2019).
- Jeong, S.H.; Kang, D.; Lim, M.W.; Kang, C.S.; Sung, H.J. Risk Assessment of Growth Hormones and Antimicrobial Residues in Meat. Toxicol. Res. 2013. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Chang, Y.; Zheng, Q.; Zhang, R.; Hu, C.; Jia, W. Altered Intestinal Microbiota Associated with Colorectal Cancer. Front. Med. 2019. [Google Scholar] [CrossRef] [PubMed]

| Study | Study Characteristics | MD Adherence | Objective | Statistical Method | Results |
|---|---|---|---|---|---|
| Buckland [97] (2013) | Prospective Study Case-control Sample: 335,062 women Period: 1992 to 2000 Place: Europe | arMED | Incidence of cancer | Cox proportional hazard regression model | HRarMEDhigh vs. arMEDlow = 0.94 (0.88–1.00) HRarMEDhigh vs. arMEDlow = 0.93 (0.87–0.99) HRarMEDhigh vs. arMEDlow = 0.80 (0.65–0.99) |
| Van den Brandt [94] (2017) | Prospective Study Case-control Sample: 62,573 women aged 55–69 years Period: 1986–2007 Place: Netherlands | Mediterranean Diet Score | Incidence of cancer | Cox proportional hazard regression model | HRMD high vs. MD low = 0.60, 95% CI: 0.39–0.93 |
| Turati [96] (2018) | Prospective Study Case-control Sample: 6426 women Period: 1991–2008 Place: Italy and Switz | Mediterranean Diet Score | Incidence of cancer | Logistic regression | ORMDS=4-5 vs. MDS=0-3 = 0.86 (0.76–0.98) ORMDS=6-9 vs. MDS=0-3 = 0.82 (0.71–0.95) ORMDS=4-5 vs. MDS=0-3 = 0.81, (0.71–0.91) |
| Study | Study Characteristics | MD Adherence | Objective | Statistical Method | Results |
|---|---|---|---|---|---|
| Castello [108] (2018) | Multicase-control study Sample: 5138 Period: 2008–2013 Place: 11 Spanish provinces | A posteriori score | Incidence of cancer | Logistic regression | Men: ORQ4 vs. Q1 = 0.71 (0.55–0.92) Women: ORQ4 vs. Q1 = 0.65, (0.40–0.77) Proximal colon: ORQ4 vs. Q1 = 0.70 (0.51–0.97) Distal colon: ORQ4 vs. Q1 = 0.65 (0.48–0.89) Rectum: ORQ4 vs. Q1 = 0.60, (0.45–0.81) |
| Fliss-Isakov [13] (2018) | Case-control study Sample: 783 patients Period: 2010–2015 Place: Israel | A posteriori score | Incidence of cancer | Multivariate logistic regression | ORMDS = 3-4 = 0.34 (0.17–0.65), ORMDS = 5-7 = 0.22 (0.11–0.43); ORMDS = 8-10 = 0.18 (0.07–0.47) |
| Rosato [109] (2016) | Case-control study Sample: 10,549 patients Period: 1985–1991 Place: Milan (Italy) | Mediterranean Diet Score | Incidence of cancer | Unconditional logistic regression | OR = 0.89, 95% CI: 0.86–0.91 (for each 1-point increase of MD) |
| Ratjen [110] (2017) | Prospective cohort study Sample: 1404 CRC patients Period: 2004–2007 Place: Northern Germany | A posteriori score | Mortality rate in CRC patients | Cox proportional hazard regression model | HRhighest quartile vs. lowest quartile = 0.48 (0.32–0.74) HRhighest quartile vs. lowest quartile = 0.88 (0.81–0.96) (for each 1-point increase of MD) |
| Study | Study Characteristics | MD Adherence Measurement | Objective | Statistical Method | Results |
|---|---|---|---|---|---|
| Schneider [114] (2019) | Prospective study Sample: 2258 patients Period:2004–2009 Place: North Caroline, Louisiana (USA) | Mediterranean Diet Score | Incidence of cancer | Multivariate logistic regression | ORhigh score vs. low score = 0.66 (0.46–0.95) |
| Kenfield [115] (2015) | Prospective study Sample: 47,867 men Period: 1986–2010 Place: USA | Mediterranean Diet Score | Mortality rate in patients without metastasis | Cox proportional hazard regression model | HR = 0.78 (0.67–0.90) |
| Russo [85] (2018) | Case-control Sample: 356 patients Period: 2015–2016 Place: Catania (Italy) | MEDILITE score | Incidence of cancer | Multivariate logistic regression | OR = 0.86 (0.77–0.96) (for each 1-point increase of MD score) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mentella, M.C.; Scaldaferri, F.; Ricci, C.; Gasbarrini, A.; Miggiano, G.A.D. Cancer and Mediterranean Diet: A Review. Nutrients 2019, 11, 2059. https://doi.org/10.3390/nu11092059
Mentella MC, Scaldaferri F, Ricci C, Gasbarrini A, Miggiano GAD. Cancer and Mediterranean Diet: A Review. Nutrients. 2019; 11(9):2059. https://doi.org/10.3390/nu11092059
Chicago/Turabian StyleMentella, Maria Chiara, Franco Scaldaferri, Caterina Ricci, Antonio Gasbarrini, and Giacinto Abele Donato Miggiano. 2019. "Cancer and Mediterranean Diet: A Review" Nutrients 11, no. 9: 2059. https://doi.org/10.3390/nu11092059
APA StyleMentella, M. C., Scaldaferri, F., Ricci, C., Gasbarrini, A., & Miggiano, G. A. D. (2019). Cancer and Mediterranean Diet: A Review. Nutrients, 11(9), 2059. https://doi.org/10.3390/nu11092059





