Dietary Patterns and Renal Health Outcomes in the General Population: A Review Focusing on Prospective Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
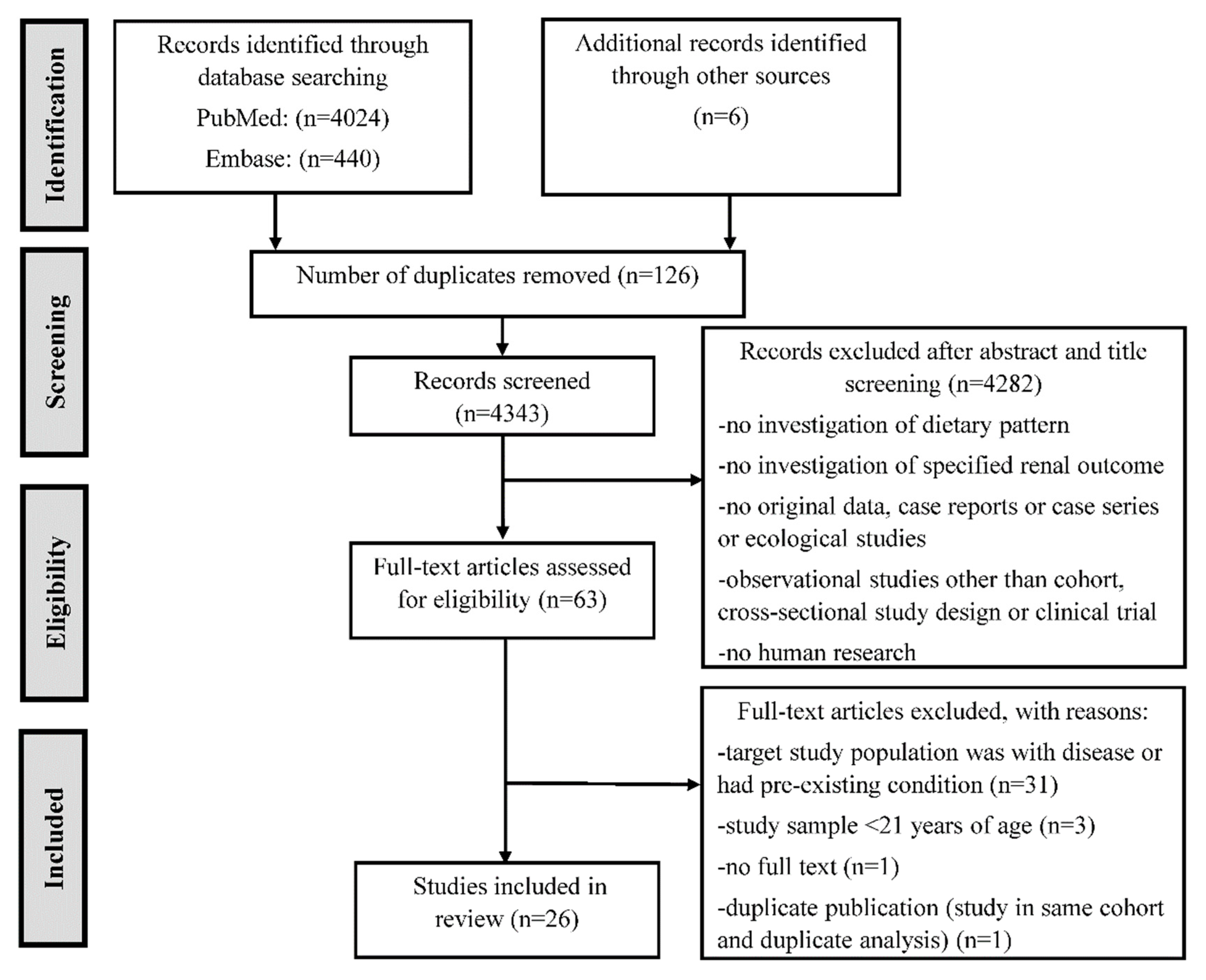
2.2. Literature Search, Screening and Data Extraction
3. Results
3.1. Dietary Approaches to Stop Hypertension (DASH) Diet
3.2. Mediterranean Diet
3.3. Vegetarian Diet
3.4. Other Diet Scores
3.5. A Posteriori Diet Patterns
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chapter 1: Definition and classification of CKD. Kidney Int. Suppl. 2013, 3, 19–62. [CrossRef] [PubMed]
- Hill, N.R.; Fatoba, S.T.; Oke, J.L.; Hirst, J.A.; O’Callaghan, C.A.; Lasserson, D.S.; Hobbs, F.D. Global Prevalence of Chronic Kidney Disease—A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0158765. [Google Scholar] [CrossRef] [PubMed]
- Anavekar, N.S.; McMurray, J.J.; Velazquez, E.J.; Solomon, S.D.; Kober, L.; Rouleau, J.L.; White, H.D.; Nordlander, R.; Maggioni, A.; Dickstein, K.; et al. Relation between renal dysfunction and cardiovascular outcomes after myocardial infarction. N. Engl. J. Med. 2004, 351, 1285–1295. [Google Scholar] [CrossRef] [PubMed]
- Foley, R.N.; Parfrey, P.S.; Sarnak, M.J. Epidemiology of cardiovascular disease in chronic renal disease. J. Am. Soc. Nephrol. 1998, 9, S16–S23. [Google Scholar] [CrossRef] [PubMed]
- Go, A.S.; Chertow, G.M.; Fan, D.; McCulloch, C.E.; Hsu, C.Y. Chronic kidney disease and the risks of death, cardiovascular events, and hospitalization. N. Engl. J. Med. 2004, 351, 1296–1305. [Google Scholar] [CrossRef] [PubMed]
- Manjunath, G.; Tighiouart, H.; Ibrahim, H.; MacLeod, B.; Salem, D.N.; Griffith, J.L.; Coresh, J.; Levey, A.S.; Sarnak, M.J. Level of kidney function as a risk factor for atherosclerotic cardiovascular outcomes in the community. J. Am. Coll. Cardiol. 2003, 41, 47–55. [Google Scholar] [CrossRef]
- United States Renal Data System. The 2018 Annual Data Report, Epidemiology of Kidney Disease in the United States; USRDS Coordinating Center United States Renal Data System 2018; United States Renal Data System: Minneapolis, MN, USA, 2018. [Google Scholar]
- Valerie, A.; Luyckx, M.T.; Stanifer, J.W. The global burden of kidney disease and the sustainable development goals. Bull. World Health Organ. 2018, 96, 369–440. [Google Scholar] [CrossRef]
- Couser, W.G.; Remuzzi, G.; Mendis, S.; Tonelli, M. The contribution of chronic kidney disease to the global burden of major noncommunicable diseases. Kidney Int. 2011, 80, 1258–1270. [Google Scholar] [CrossRef]
- Golestaneh, L.; Alvarez, P.J.; Reaven, N.L.; Funk, S.E.; McGaughey, K.J.; Romero, A.; Brenner, M.S.; Onuigbo, M. All-cause costs increase exponentially with increased chronic kidney disease stage. Am. J. Manag. Care 2017, 23, S163–S172. [Google Scholar]
- Hu, F.B. Dietary pattern analysis: A new direction in nutritional epidemiology. Curr. Opin. Lipidol. 2002, 13, 3–9. [Google Scholar] [CrossRef]
- Cespedes, E.M.; Hu, F.B. Dietary patterns: From nutritional epidemiologic analysis to national guidelines. Am. J. Clin. Nutr. 2015, 101, 899–900. [Google Scholar] [CrossRef]
- Whitham, D. Nutrition for the prevention and treatment of chronic kidney disease in diabetes. Can. J. Diabetes 2014, 38, 344–348. [Google Scholar] [CrossRef] [PubMed]
- Ko, G.J.; Kalantar-Zadeh, K.; Goldstein-Fuchs, J.; Rhee, C.M. Dietary Approaches in the Management of Diabetic Patients with Kidney Disease. Nutrients 2017, 9, 824. [Google Scholar] [CrossRef]
- Banerjee, T.; Liu, Y.; Crews, D.C. Dietary Patterns and CKD Progression. Blood Purif. 2016, 41, 117–122. [Google Scholar] [CrossRef]
- Kelly, J.T.; Palmer, S.C.; Wai, S.N.; Ruospo, M.; Carrero, J.J.; Campbell, K.L.; Strippoli, G.F. Healthy Dietary Patterns and Risk of Mortality and ESRD in CKD: A Meta-Analysis of Cohort Studies. Clin. J. Am. Soc. Nephrol. 2017, 12, 272–279. [Google Scholar] [CrossRef]
- Kalantar-Zadeh, K.; Fouque, D. Nutritional Management of Chronic Kidney Disease. N. Engl. J. Med. 2017, 377, 1765–1776. [Google Scholar] [CrossRef]
- Devries, M.C.; Sithamparapillai, A.; Brimble, K.S.; Banfield, L.; Morton, R.W.; Phillips, S.M. Changes in Kidney Function Do Not Differ between Healthy Adults Consuming Higher- Compared with Lower- or Normal-Protein Diets: A Systematic Review and Meta-Analysis. J. Nutr. 2018, 148, 1760–1775. [Google Scholar] [CrossRef]
- Kamper, A.L.; Strandgaard, S. Long-Term Effects of High-Protein Diets on Renal Function. Ann. Rev. Nutr. 2017, 37, 347–369. [Google Scholar] [CrossRef]
- Marckmann, P.; Osther, P.; Pedersen, A.N.; Jespersen, B. High-protein diets and renal health. J. Ren. Nutr. 2015, 25, 1–5. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G. Comparison of high vs. normal/low protein diets on renal function in subjects without chronic kidney disease: A systematic review and meta-analysis. PLoS ONE 2014, 9, e97656. [Google Scholar] [CrossRef]
- Toto, R.D. Microalbuminuria: Definition, detection, and clinical significance. J. Clin. Hypertens. 2004, 6, 2–7. [Google Scholar] [CrossRef]
- Bjornstad, P.; Cherney, D.Z.; Snell-Bergeon, J.K.; Pyle, L.; Rewers, M.; Johnson, R.J.; Maahs, D.M. Rapid GFR decline is associated with renal hyperfiltration and impaired GFR in adults with Type 1 diabetes. Nephrol. Dial. Transpl. 2015, 30, 1706–1711. [Google Scholar] [CrossRef]
- Traynor, J.; Mactier, R.; Geddes, C.C.; Fox, J.G. How to measure renal function in clinical practice. BMJ 2006, 333, 733–737. [Google Scholar] [CrossRef]
- Appel, L.J.; Brands, M.W.; Daniels, S.R.; Karanja, N.; Elmer, P.J.; Sacks, F.M.; American Heart, A. Dietary approaches to prevent and treat hypertension: A scientific statement from the American Heart Association. Hypertension 2006, 47, 296–308. [Google Scholar] [CrossRef]
- Fung, T.T.; Chiuve, S.E.; McCullough, M.L.; Rexrode, K.M.; Logroscino, G.; Hu, F.B. Adherence to a DASH-style diet and risk of coronary heart disease and stroke in women. Arch. Intern. Med. 2008, 168, 713–720. [Google Scholar] [CrossRef]
- Mellen, P.B.; Gao, S.K.; Vitolins, M.Z.; Goff, D.C., Jr. Deteriorating dietary habits among adults with hypertension: DASH dietary accordance, NHANES 1988-1994 and 1999-2004. Arch. Intern. Med. 2008, 168, 308–314. [Google Scholar] [CrossRef]
- Asghari, G.; Yuzbashian, E.; Mirmiran, P.; Azizi, F. The association between Dietary Approaches to Stop Hypertension and incidence of chronic kidney disease in adults: The Tehran Lipid and Glucose Study. Nephrol. Dial. Transpl. 2017, 32, ii224–ii230. [Google Scholar] [CrossRef]
- Rebholz, C.M.; Crews, D.C.; Grams, M.E.; Steffen, L.M.; Levey, A.S.; Miller, E.R.; Appel, L.J.; Coresh, J. DASH (Dietary Approaches to Stop Hypertension) Diet and Risk of Subsequent Kidney Disease. Am. J. Kidney Dis. 2016, 68, 853–861. [Google Scholar] [CrossRef]
- Liu, Y.; Kuczmarski, M.F.; Miller, E.R.; Nava, M.B.; Zonderman, A.B.; Evans, M.K.; Powe, N.R.; Crews, D.C. Dietary Habits and Risk of Kidney Function Decline in an Urban Population. J. Ren. Nutr. 2017, 27, 16–25. [Google Scholar] [CrossRef]
- Crews, D.C.; Kuczmarski, M.F.; Miller, E.R.; Zonderman, A.B.; Evans, M.K.; Powe, N.R. Dietary habits, poverty, and chronic kidney disease in an urban population. J. Ren. Nutr. 2015, 25, 103–110. [Google Scholar] [CrossRef]
- Lee, H.S.; Lee, K.B.; Hyun, Y.Y.; Chang, Y.; Ryu, S.; Choi, Y. DASH dietary pattern and chronic kidney disease in elderly Korean adults. Eur. J. Clin. Nutr. 2017, 71, 755–761. [Google Scholar] [CrossRef]
- Taylor, E.N.; Fung, T.T.; Curhan, G.C. DASH-style diet associates with reduced risk for kidney stones. J. Am. Soc. Nephrol. 2009, 20, 2253–2259. [Google Scholar] [CrossRef]
- Ferraro, P.M.; Taylor, E.N.; Gambaro, G.; Curhan, G.C. Dietary and Lifestyle Risk Factors Associated with Incident Kidney Stones in Men and Women. J. Urol. 2017, 198, 858–863. [Google Scholar] [CrossRef]
- Chang, A.; Van Horn, L.; Jacobs, D.R., Jr.; Liu, K.; Muntner, P.; Newsome, B.; Shoham, D.A.; Durazo-Arvizu, R.; Bibbins-Domingo, K.; Reis, J.; et al. Lifestyle-related factors, obesity, and incident microalbuminuria: The CARDIA (Coronary Artery Risk Development in Young Adults) study. Am. J. Kidney Dis. 2013, 62, 267–275. [Google Scholar] [CrossRef]
- Lin, J.; Fung, T.T.; Hu, F.B.; Curhan, G.C. Association of dietary patterns with albuminuria and kidney function decline in older white women: A subgroup analysis from the Nurses’ Health Study. Am. J. Kidney Dis. 2011, 57, 245–254. [Google Scholar] [CrossRef]
- Smyth, A.; Griffin, M.; Yusuf, S.; Mann, J.F.; Reddan, D.; Canavan, M.; Newell, J.; O’Donnell, M. Diet and Major Renal Outcomes: A Prospective Cohort Study. The NIH-AARP Diet and Health Study. J. Ren. Nutr. 2016, 26, 288–298. [Google Scholar] [CrossRef]
- Willett, W.C.; Sacks, F.; Trichopoulou, A.; Drescher, G.; Ferro-Luzzi, A.; Helsing, E.; Trichopoulos, D. Mediterranean diet pyramid: A cultural model for healthy eating. Am. J. Clin. Nutr. 1995, 61, 1402S–1406S. [Google Scholar] [CrossRef]
- Bach-Faig, A.; Berry, E.M.; Lairon, D.; Reguant, J.; Trichopoulou, A.; Dernini, S.; Medina, F.X.; Battino, M.; Belahsen, R.; Miranda, G.; et al. Mediterranean diet pyramid today. Science and cultural updates. Public Health Nutr. 2011, 14, 2274–2284. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef]
- Panagiotakos, D.B.; Pitsavos, C.; Arvaniti, F.; Stefanadis, C. Adherence to the Mediterranean food pattern predicts the prevalence of hypertension, hypercholesterolemia, diabetes and obesity, among healthy adults; the accuracy of the MedDietScore. Prev. Med. 2007, 44, 335–340. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Orfanos, P.; Norat, T.; Bueno-de-Mesquita, B.; Ocke, M.C.; Peeters, P.H.; van der Schouw, Y.T.; Boeing, H.; Hoffmann, K.; Boffetta, P.; et al. Modified Mediterranean diet and survival: EPIC-elderly prospective cohort study. BMJ 2005, 330, 991. [Google Scholar] [CrossRef]
- Asghari, G.; Farhadnejad, H.; Mirmiran, P.; Dizavi, A.; Yuzbashian, E.; Azizi, F. Adherence to the Mediterranean diet is associated with reduced risk of incident chronic kidney diseases among Tehranian adults. Hypertens. Res. 2017, 40, 96–102. [Google Scholar] [CrossRef]
- Khatri, M.; Moon, Y.P.; Scarmeas, N.; Gu, Y.; Gardener, H.; Cheung, K.; Wright, C.B.; Sacco, R.L.; Nickolas, T.L.; Elkind, M.S. The association between a Mediterranean-style diet and kidney function in the Northern Manhattan Study cohort. Clin. J. Am. Soc. Nephrol. 2014, 9, 1868–1875. [Google Scholar] [CrossRef]
- Leone, A.; Fernandez-Montero, A.; de la Fuente-Arrillaga, C.; Martinez-Gonzalez, M.A.; Bertoli, S.; Battezzati, A.; Bes-Rastrollo, M. Adherence to the Mediterranean Dietary Pattern and Incidence of Nephrolithiasis in the Seguimiento Universidad de Navarra Follow-up (SUN) Cohort. Am. J. Kidney Dis. 2017, 70, 778–786. [Google Scholar] [CrossRef]
- Chrysohoou, C.; Panagiotakos, D.B.; Pitsavos, C.; Skoumas, J.; Toutouza, M.; Papaioannou, I.; Stefanadis, C. Renal function, cardiovascular disease risk factors’ prevalence and 5-year disease incidence; the role of diet, exercise, lipids and inflammation markers: The ATTICA study. QJM Mon. J. Assoc. Physicians 2010, 103, 413–422. [Google Scholar] [CrossRef][Green Version]
- Asghari, G.; Momenan, M.; Yuzbashian, E.; Mirmiran, P.; Azizi, F. Dietary pattern and incidence of chronic kidney disease among adults: A population-based study. Nutr. Metab. 2018, 15, 88. [Google Scholar] [CrossRef]
- Liu, H.W.; Tsai, W.H.; Liu, J.S.; Kuo, K.L. Association of Vegetarian Diet with Chronic Kidney Disease. Nutrients 2019, 11, 279. [Google Scholar] [CrossRef]
- Turney, B.W.; Appleby, P.N.; Reynard, J.M.; Noble, J.G.; Key, T.J.; Allen, N.E. Diet and risk of kidney stones in the Oxford cohort of the European Prospective Investigation into Cancer and Nutrition (EPIC). Eur. J. Epidemiol. 2014, 29, 363–369. [Google Scholar] [CrossRef]
- Lin, C.K.; Lin, D.J.; Yen, C.H.; Chen, S.C.; Chen, C.C.; Wang, T.Y.; Chou, M.C.; Chang, H.R.; Lee, M.C. Comparison of renal function and other health outcomes in vegetarians versus omnivores in Taiwan. J. Health Popul. Nutr. 2010, 28, 470–475. [Google Scholar] [CrossRef]
- Rebholz, C.; Crews, D.; Grams, M.; Levey, A.; Miller, I.E.; Appel, L.; Coresh, J. Adherence to the dash diet and risk of incident chronic kidney disease: The atherosclerosis risk in communities (ARIC) study. FASEB J. 2015, 29, 119.1. [Google Scholar]
- Foster, M.C.; Hwang, S.J.; Massaro, J.M.; Jacques, P.F.; Fox, C.S.; Chu, A.Y. Lifestyle factors and indices of kidney function in the Framingham Heart Study. Am. J. Nephrol. 2015, 41, 267–274. [Google Scholar] [CrossRef]
- Gopinath, B.; Harris, D.C.; Flood, V.M.; Burlutsky, G.; Mitchell, P. A better diet quality is associated with a reduced likelihood of CKD in older adults. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 937–943. [Google Scholar] [CrossRef]
- Xu, H.; Sjogren, P.; Arnlov, J.; Banerjee, T.; Cederholm, T.; Riserus, U.; Lindholm, B.; Lind, L.; Carrero, J.J. A proinflammatory diet is associated with systemic inflammation and reduced kidney function in elderly adults. J. Nutr. 2015, 145, 729–735. [Google Scholar] [CrossRef]
- Rebholz, C.M.; Anderson, C.A.; Grams, M.E.; Bazzano, L.A.; Crews, D.C.; Chang, A.R.; Coresh, J.; Appel, L.J. Relationship of the AHA impact goals (life’s simple 7) with risk of chronic kidney disease: Results from the aric cohort study. Circulation 2015, 132, A10922. [Google Scholar]
- Mazidi, M.; Gao, H.-K.; Kengne, A.P. Food Patterns are Associated with Likelihood of CKD in US Adults. Sci. Rep. 2018, 8, 10696. [Google Scholar] [CrossRef]
- Shi, Z.; Taylor, A.W.; Riley, M.; Byles, J.; Liu, J.; Noakes, M. Association between dietary patterns, cadmium intake and chronic kidney disease among adults. Clin. Nutr. 2018, 37, 276–284. [Google Scholar] [CrossRef]
- Paterson, E.N.; Neville, C.E.; Silvestri, G.; Montgomery, S.; Moore, E.; Silvestri, V.; Cardwell, C.R.; MacGillivray, T.J.; Maxwell, A.P.; Woodside, J.V.; et al. Dietary patterns and chronic kidney disease: A cross-sectional association in the Irish Nun Eye Study. Sci. Rep. 2018, 8, 6654. [Google Scholar] [CrossRef]
- Nettleton, J.A.; Steffen, L.M.; Palmas, W.; Burke, G.L.; Jacobs, D.R., Jr. Associations between microalbuminuria and animal foods, plant foods, and dietary patterns in the Multiethnic Study of Atherosclerosis. Am. J. Clin. Nutr. 2008, 87, 1825–1836. [Google Scholar] [CrossRef]
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R.; Simons-Morton, D.G.; et al. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group. N. Engl. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef]
- Lopes, H.F.; Martin, K.L.; Nashar, K.; Morrow, J.D.; Goodfriend, T.L.; Egan, B.M. DASH diet lowers blood pressure and lipid-induced oxidative stress in obesity. Hypertension 2003, 41, 422–430. [Google Scholar] [CrossRef]
- Rifai, L.; Pisano, C.; Hayden, J.; Sulo, S.; Silver, M.A. Impact of the DASH diet on endothelial function, exercise capacity, and quality of life in patients with heart failure. Bayl. Univ. Med. Cent. Proc. 2015, 28, 151–156. [Google Scholar] [CrossRef]
- Rasmussen, B.M.; Vessby, B.; Uusitupa, M.; Berglund, L.; Pedersen, E.; Riccardi, G.; Rivellese, A.A.; Tapsell, L.; Hermansen, K.; Group, K.S. Effects of dietary saturated, monounsaturated, and n-3 fatty acids on blood pressure in healthy subjects. Am. J. Clin. Nutr. 2006, 83, 221–226. [Google Scholar] [CrossRef]
- Hinderliter, A.L.; Babyak, M.A.; Sherwood, A.; Blumenthal, J.A. The DASH diet and insulin sensitivity. Curr. Hypertens. Rep. 2011, 13, 67–73. [Google Scholar] [CrossRef]
- Widmer, R.J.; Flammer, A.J.; Lerman, L.O.; Lerman, A. The Mediterranean diet, its components, and cardiovascular disease. Am. J. Med. 2015, 128, 229–238. [Google Scholar] [CrossRef]
- Chrysohoou, C.; Pitsavos, C.; Panagiotakos, D.; Skoumas, J.; Lazaros, G.; Oikonomou, E.; Galiatsatos, N.; Striggou, M.; Xynogala, M.; Stefanadis, C. Long-term fish intake preserves kidney function in elderly individuals: The Ikaria study. J. Ren. Nutr. 2013, 23, e75–e82. [Google Scholar] [CrossRef]
- Tonelli, M.; Sacks, F.; Pfeffer, M.; Jhangri, G.S.; Curhan, G. Biomarkers of inflammation and progression of chronic kidney disease. Kidney Int. 2005, 68, 237–245. [Google Scholar] [CrossRef]
- Syauqy, A.; Hsu, C.Y.; Rau, H.H.; Chao, J.C. Association of Dietary Patterns with Components of Metabolic Syndrome and Inflammation among Middle-Aged and Older Adults with Metabolic Syndrome in Taiwan. Nutrients 2018, 10, 143. [Google Scholar] [CrossRef]
- Nettleton, J.A.; Steffen, L.M.; Mayer-Davis, E.J.; Jenny, N.S.; Jiang, R.; Herrington, D.M.; Jacobs, D.R., Jr. Dietary patterns are associated with biochemical markers of inflammation and endothelial activation in the Multi-Ethnic Study of Atherosclerosis (MESA). Am. J. Clin. Nutr. 2006, 83, 1369–1379. [Google Scholar] [CrossRef]
- Lopez-Garcia, E.; Schulze, M.B.; Fung, T.T.; Meigs, J.B.; Rifai, N.; Manson, J.E.; Hu, F.B. Major dietary patterns are related to plasma concentrations of markers of inflammation and endothelial dysfunction. Am. J. Clin. Nutr. 2004, 80, 1029–1035. [Google Scholar] [CrossRef]
- Slavin, J.L.; Lloyd, B. Health benefits of fruits and vegetables. Adv. Nutr. 2012, 3, 506–516. [Google Scholar] [CrossRef]
- Estruch, R.; Salas-Salvado, J. Towards an even healthier Mediterranean diet. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 1163–1166. [Google Scholar] [CrossRef]
- Williams, P.G.; Grafenauer, S.J.; O’Shea, J.E. Cereal grains, legumes, and weight management: A comprehensive review of the scientific evidence. Nutr. Rev. 2008, 66, 171–182. [Google Scholar] [CrossRef]
- Gannon, R.H.; Millward, D.J.; Brown, J.E.; Macdonald, H.M.; Lovell, D.P.; Frassetto, L.A.; Remer, T.; Lanham-New, S.A. Estimates of daily net endogenous acid production in the elderly UK population: Analysis of the National Diet and Nutrition Survey (NDNS) of British adults aged 65 years and over. Br. J. Nutr. 2008, 100, 615–623. [Google Scholar] [CrossRef]
- Engberink, M.F.; Bakker, S.J.; Brink, E.J.; van Baak, M.A.; van Rooij, F.J.; Hofman, A.; Witteman, J.C.; Geleijnse, J.M. Dietary acid load and risk of hypertension: The Rotterdam Study. Am. J. Clin. Nutr. 2012, 95, 1438–1444. [Google Scholar] [CrossRef]
- Remer, T.; Dimitriou, T.; Manz, F. Dietary potential renal acid load and renal net acid excretion in healthy, free-living children and adolescents. Am. J. Clin. Nutr. 2003, 77, 1255–1260. [Google Scholar] [CrossRef]
- Murakami, K.; Sasaki, S.; Takahashi, Y.; Uenishi, K. Association between dietary acid-base load and cardiometabolic risk factors in young Japanese women. Br. J. Nutr. 2008, 100, 642–651. [Google Scholar] [CrossRef]
- Scialla, J.J.; Anderson, C.A. Dietary acid load: A novel nutritional target in chronic kidney disease? Adv. Chronic Kidney Dis. 2013, 20, 141–149. [Google Scholar] [CrossRef]
- Wesson, D.E.; Simoni, J.; Broglio, K.; Sheather, S. Acid retention accompanies reduced GFR in humans and increases plasma levels of endothelin and aldosterone. Am. J. Physiol. Ren. Physiol. 2011, 300, F830–F837. [Google Scholar] [CrossRef]
- Wesson, D.E.; Simoni, J. Increased tissue acid mediates a progressive decline in the glomerular filtration rate of animals with reduced nephron mass. Kidney Int. 2009, 75, 929–935. [Google Scholar] [CrossRef]
- Kontessis, P.A.; Bossinakou, I.; Sarika, L.; Iliopoulou, E.; Papantoniou, A.; Trevisan, R.; Roussi, D.; Stipsanelli, K.; Grigorakis, S.; Souvatzoglou, A. Renal, metabolic, and hormonal responses to proteins of different origin in normotensive, nonproteinuric type I diabetic patients. Diabetes Care 1995, 18, 1233. [Google Scholar] [CrossRef]
- Barbaresko, J.; Koch, M.; Schulze, M.B.; Nothlings, U. Dietary pattern analysis and biomarkers of low-grade inflammation: A systematic literature review. Nutr. Rev. 2013, 71, 511–527. [Google Scholar] [CrossRef]

| First Author (Publication Year), Country | Population, Sample Size (Sex) | Age, Years | Outcome Ascertainment | Diet Assessment Method (No. of Items) | Follow-Up, Years | Outcome (Definition) | Dietary Pattern Identified (Method Used) | Association Measures with Outcomes (OR, RR, HR, sHR, IRR, and 95% CI) or Major Findings | Covariates in Fully Adjusted Model |
|---|---|---|---|---|---|---|---|---|---|
| Lin et al. (2011) [25], USA | NHS I, 3121 (women) | Median age of study sample: 67 | -Urinary creatinine via modified Jaffe method using urine sample collected in 2000. -Urinary albumin via solid-phase fluorescence immunoassay using urine sample collected in 2000. -Plasma creatinine via modified kinetic Jaffe method from plasma samples collected in 1989 and 2000. -eGFR calculated via the MDRD equation. | FFQ (116). The dietary pattern was calculated from the cumulative average dietary pattern from FFQ on five visits from 1984–1998 for microalbuminuria and three visits from 1984–1990 for eGFR decline. | 11 | 1. eGFR decline (≥30% between 1989 and 2000). 2. Microalbuminuria (UACR ≥ 25 mcg/mg). | (1) DASH-style diet (diet score) (2) Western diet (PCA) (3) Prudent (PCA) | eGFR decline (OR) Prudent Q1: 1.00 (ref.) Q2: 1.43 (1.04, 1.98) Q3: 1.07 (0.76, 1.51) Q4: 0.81 (0.55, 1.19) Western Q1: 1.00 (ref.) Q2: 1.22 (0.87, 1.73) Q3: 1.57 (1.08, 2.28) Q4: 1.48 (0.95, 2.30) DASH-Style Q1: 1.00 (ref.) Q2: 0.86 (0.63, 1.17) Q3: 0.79 (0.57, 1.09) Q4: 0.55 (0.38, 0.80) Microalbuminuria (OR) Prudent Q1: 1.00 (ref.) Q2: 0.89 (0.57, 1.42) Q3: 1.05 (0.66, 1.67) Q4: 0.97 (0.58, 1.61) Western Q1: 1.00 (ref.) Q2: 1.11 (0.68, 1.81) Q3: 1.12 (0.66, 1.92) Q4: 2.17 (1.18, 3.96) DASH-Style Q1: 1.00 (ref.) Q2: 0.80 (0.52, 1.23) Q3: 0.77 (0.49, 1.21) Q4: 0.71 (0.44, 1.14) | Age, BMI, hypertension, physical activity, energy intake, cigarette smoking, diabetes, cardiovascular disease, and ACE-inhibitor/ARB medication use |
| Smyth et al. (2016) [26], USA | NIH-AARP, 544,635 (both) | Mean age of study sample: 62.2 (SD: 5.4) | -Vital status ascertained from Social Security Administration Death Master File and NDI. -Self-reported dialysis was noted on the study follow-up questionnaire. | FFQ (124). Dietary pattern calculated from FFQ administered once at baseline from 1995–1996. | 14.3 | 1. Composite death due to renal cause and initiation of dialysis (death where chronic renal disease was primary or contributing cause of death based on ICD coding system, censored 31st December 2011). 2. Self-reported dialysis questionnaire). 3. Death due to renal cause. | 1. AHEI, 2010 (diet score) 2. HEI, 2010 (diet score) 3.MDS (diet score) 4. RFS (diet score) 5. DASH (diet score) | Composite death due to renal cause and initiation of dialysis (sHR) 1. AHEI-2010, HEI-2010, MDS, DASH but not RFS scores were significantly associated Self-reported dialysis (sHR) 1. None of the diet scores were significantly associated Death due to a renal cause (sHR) 1. AHEI-2010, HEI-2010, MDS, DASH but not RFS scores were significantly associated | Age, gender, BMI, smoking, education, ethnicity, physical activity, diabetes, heart disease, and stroke |
| Asghari et al. (2016) [27], Iran | TLGS, 1212 (both) | Mean age of study sample: 43.5 (SD: 9.4) | -Serum creatinine measured via Jaffe kinetic reaction with blood sample collected during fifth study visit between 2012–2015. -eGFR calculated via the MDRD equation. | FFQ (168). Dietary pattern calculated using FFQ administered during third study visit from 2006–2008. | 6.1 | 1. Incident CKD (eGFR < 60 mL/min/1.73 m2). | MDS (diet score) | Incident CKD (OR) Q1: 1.0 (ref.) Q2: 0.95 (0.64, 1.40) Q3: 0.85 (0.55, 1.32) Q4: 0.53 (0.31, 0.91) P for trend: 0.030 | Age, BMI, gender, smoking status, physical activity, total calorie intake, diabetes, hypertension, baseline eGFR |
| Khatri et al. (2014) [28], USA | NOMAS, 900 (both) | Mean baseline age of study sample: 64 | -Serum creatinine measured via Jaffe reaction with blood sample collected between 2003–2008. -eGFR calculated via the MDRD equation. | FFQ (147). Dietary pattern calculated via FFQ administered at baseline in 1998. | 6.9 | 1. Incident eGFR < 60 mL/min/1.73 m2 (started with eGFR ≥ 60 mL/min/1.73 m2 at baseline and had an eGFR < 60 mL/min/1.73 m2 at follow-up exam). 2. Upper quartile of eGFR decline. 3. Annualized eGFR decline. | MDS (diet score) | Incident eGFR < 60 mL/min/1.73 m2 (OR) Q1: 1.0 (ref.) Q2: 1.61 (0.88, 2.97) Q3: 0.51 (0.26, 1.02) Q4: 0.76 (0.39, 1.46) Upper quartile of eGFR decline (OR) Q1: 1.0 (ref.) Q2: 1.01 (0.62, 1.66) Q3: 0.49 (0.29, 0.82) Q4: 0.67 (0.41, 1.10) Annualized change in eGFR (parameter estimate) Q1: 1.0 (ref.) Q2: −0.37 (−0.86, 0.12) Q3: 0.11 (−0.37, 0.60) Q4: 0.20 (−0.28, 0.67) | Age, gender, BMI race/ethnicity, education, insurance status, physical activity, diabetes, smoking status, hypertension, LDL, HDL, baseline eGFR, and ACE inhibitor/ARB usage |
| Leone et al. (2017) [29], Spain | SUN Project, 16,094 (both) | Median baseline age of study sample: 36 (IQR: 28–46) | -Nephrolithiasis self-reported via study follow-up questionnaire (enough participants with at least one follow-up questionnaire by 2013). | FFQ (136). Dietary pattern calculated using FFQ administered at baseline in 1999. | 9.6 | 1. Incident nephrolithiasis (was free of nephrolithiasis at baseline and reported nephrolithiasis diagnosis at study follow-up). | MDS (diet score) | Incident nephrolithiasis (kidney stones) (HR) T1: 1.0 (ref.) T2: 0.93 (0.79, 1.09) T3: 0.64 (0.48, 0.87) P for trend: 0.01 | Gender, BMI, hypertension, diabetes, marital status, education, number of working hours per week, smoking, physical activity, time spent watching television, total energy intake, total water intake, calcium supplementation, vitamin D supplementation, and following a nutritional therapy |
| Rebholz et al. (2016) [30], USA | ARIC, 14,882 (both) | Age range of cohort: 45–65 | -Serum creatinine measured via modified Jaffe Kinetic reaction using blood collected at any of the five follow-up exams.-eGFR calculated via the CKD-EPI equation. -Kidney disease related hospitalization or death based on (ICD)-9/10 codes identified via surveillance and linkage to the NDI. -End-stage renal disease (dialysis or transplantation) identified by linkage to the USRDS registry between baseline and follow-up exam. | FFQ (66). Dietary pattern calculated from cumulative average of diet from FFQ administered at baseline between 1987–1989 and third visit from 1993–1995. | 23 | 1. Kidney disease (meeting one of the following: 1. < 60 mL/min/1.73 m2 with 25% eGFR decline at any follow-up study visit relative to baseline. 2. Kidney disease related hospitalization or death. 3. End-stage renal disease (dialysis or transplantation) at follow-up study relative to baseline. | DASH (diet score) | Kidney disease (HR) T1: 1.16 (1.07, 1.27) T2: 1.09 (1.00, 1.18) T3: 1.00 (ref.) P for trend: <0.001 | Age, gender, race-center, education level, smoking status, physical activity, total caloric intake, baseline eGFR, overweight/obese status, diabetes, hypertension, systolic blood pressure, use of angiotensin-converting enzyme inhibitors or angiotensin receptor blockers |
| Chang et al. (2013) [31], USA | CARDIA, 2354, (both) | Age range of study sample: 28–40 | -Urine albumin measured via nephelometric procedure with specific anti-albumin antibody using urine samples collected from 2000–2001, 2005–2006 or 2010–2011. -Urine and serum creatinine measured via Jaffe method using urine samples collected from 2000–2001, 2005–2006 or 2010–2011. | Interview-administered diet history. Dietary pattern calculated via diet info. calculated during study baseline year from 1995–1996. | 15 | 1. Incident Microalbuminuria. | DASH (diet score) | Incident Microalbuminuria (OR) Q1 (lowest score): 2.0 (1.1,3.4) Q5: 0.0 (ref.) | Age, gender, baseline obesity, race, family history of kidney disease, education, total energy intake, and baseline (year-10) ACR |
| Taylor et al., (2009) [32], USA | NHS I, 94,108 (women) | Age range of cohort: 30–55 | -Kidney stone self-reported via study questionnaire and diagnosis was confirmed through review of medical records. | FFQ (>130). Dietary pattern calculated using FFQ administered at baseline in 1986. FFQ info updated every 4 years. | 18 | 1. Incident kidney stone accompanied by pain or hematuria. Follow-up years calculated from to baseline to the date of a kidney stone or death or to 31 May 2004. | DASH (diet score) | Incident kidney stone accompanied by pain or hematuria (RR) Q1: 1.00 (ref.) Q2: 0.89 (0.77, 1.02) Q3:0.76 (0.65, 0.87) Q4: 0.64 (0.54, 0.74) Q5: 0.58 (0.49, 0.68) P for trend: <0.001 | Age, BMI, total energy intake, use of thiazide diuretics, fluid intake, caffeine, alcohol use, history of hypertension, and history of diabetes |
| Taylor et al. (2009) [32], USA | NHS II, 101,837 (women) | Age range of cohort: 25–42 | -Kidney stone self-reported via study questionnaire and diagnosis was confirmed through review of medical records. | FFQ (>130) Dietary pattern calculated using FFQ administered at baseline in 1991. FFQ info updated every 4 years. | 14 | 1. Incident kidney stone accompanied by pain or hematuria. Follow-up years calculated from baseline to the date of a kidney stone or death or to 31 May 2005. | DASH (diet score) | Incident kidney stone accompanied by pain or hematuria (RR) Q1: 1.00 (ref.) Q2: 0.92 (0.81, 1.03) Q3: 0.77 (0.68, 0.88) Q4: 0.75 (0.66, 0.86) Q5: 0.60(0.52, 0.70) P for trend: <0.001 | Age, BMI, total energy intake, use of thiazide diuretics, fluid intake, caffeine, alcohol use, history of hypertension, and history of diabetes |
| Taylor et al. (2009) [32], USA | HPFS, 45,821 (men) | Age range of cohort: 40–75 | -Kidney stone self-reported via study questionnaire and diagnosis was confirmed through review of medical records. | FFQ (>130) Dietary pattern calculated using FFQ administered at baseline in 1986. FFQ info updated every 4 years. | 18 | 1. Incident kidney stone accompanied by pain or hematuria. Follow-up years calculated from baseline to the date of a kidney stone or death or to 31 January 2004. | DASH (diet score) | Incident kidney stone accompanied by pain or hematuria (RR) Q1:1.00 (ref.) Q2:0.89 (0.77, 1.01) Q3:0.77 (0.67, 0.89) Q4:0.64 (0.54, 0.74) Q5:0.55 (0.46, 0.65) P for trend: <0.001 | Age, BMI, total energy intake, use of thiazide diuretics, fluid intake, caffeine, alcohol use, history of hypertension, and history of diabetes |
| Ferraro et al. (2017) [33], USA | NHS I, 59,740, (both) | Mean age of study sample: (52.9, SD: 7.1) | -Kidney stone self-reported via supplementary questionnaire and diagnosis was confirmed through review of medical records. | FFQ (>130). Dietary pattern calculated using baseline FFQ in 1986. | 12.1 | 1. Incident kidney stone accompanied by pain or hematuria. Follow-up years of calculated from day of return of baseline questionnaire to incident kidney stone. | DASH-style diet (diet score) | Incident kidney stone accompanied by pain or hematuria (IRR) Q5:1.00 (ref.) Q4:0.98 (0.82, 1.17) Q3:1.22 (1.03, 1.44) Q2:1.32 (1.12, 1.56) Q1:1.47(1.25, 1.73) | Age, BMI, fluid, race, geographic area, use of thiazide diuretics, history of diabetes, and history of hypertension |
| Ferraro et al. (2017) [33], USA | NHS II, 90,449, (both) | Mean age of study sample: 36.6, (SD: 4.6) | -Kidney stone self-reported via supplementary questionnaire and diagnosis was confirmed through review of medical records. | FFQ (>130) Dietary pattern calculated using baseline FFQ in 1991. | 11.3 | 1. Incident kidney stone accompanied by pain or hematuria. Follow-up years of calculated from day of return of baseline questionnaire to incident kidney stone. | DASH-style diet (diet score) | Incident kidney stone accompanied by pain or hematuria (IRR) Q5:1.00 (ref.) Q4:1.18 (1.04, 1.34) Q3:1.09 (0.96, 1.24) Q2:1.33 (1.18, 1.51) Q1:1.37 (1.21, 1.55) | Age, BMI, fluid, race, geographic area, use of thiazide diuretics, history of diabetes, and history of hypertension |
| Ferraro et al. (2017) [33], USA | HPFS, 41,937 (men) | Mean age of study sample: 54.2 (SD: 9.7) | -Kidney stone self-reported via supplementary questionnaire and diagnosis was confirmed through review of medical records. | FFQ (>130) Dietary pattern calculated using baseline FFQ in 1986. | 11.5 | 1. Incident kidney stone accompanied by pain or hematuria. Follow-up years of calculated from day of return of baseline questionnaire to incident kidney stone. | DASH-style diet (diet score) | Incident kidney stone accompanied by pain or hematuria (IRR) Q5:1.00 (ref.) Q4:1.06 (0.90, 1.25) Q3:1.21 (1.04, 1.42) Q2:1.36 (1.17, 1.59) Q1:1.53 (1.31, 1.78) | Age, BMI, fluid, race, geographic area, use of thiazide diuretics, history of diabetes, and history of hypertension |
| Asghari et al. (2017) [34], Iran | TGLS, 1630 (both) | Mean age of study sample: 42.8 (SD: 11.2) | -Serum creatinine measured via Jaffe kinetic reaction method with blood sample collected during fifth study visit from 2012–2015. -eGFR calculated via the MDRD equation. | FFQ (168) Dietary pattern calculated using FFQ administered during third study visit from 2006–2008. | 6.1 | 1. Incident CKD (eGFR < 60 mL/min/1.73 m2). | DASH-style diet (diet score) | Incident CKD (OR) Q1:1.00 (ref.) Q5:0.41 (0.24, 0.70) P for trend: <0.001 | Age, gender, BMI smoking, total energy intake, eGFR, triglycerides, physical activity, hypertension, and diabetes |
| Liu et al. (2017) [35], USA | NIA-HANDLS, (1534) (both) | Mean age of study sample: 48 | -Blood creatinine measured via modified kinetic Jaffe method and isotope dilution mass spectrometry using blood samples collected from 2009–2013. | 24-hr food recall. Dietary pattern calculated via diet information collected at baseline from 2004–2008. | 5 | 1. Incident CKD (follow-up eGFR < 60 mL/min/1.73 m2). 2. eGFR decline (>25% from baseline). | DASH (diet score) | Incident CKD (RR) High DASH accordance:1.00 (ref.) Low DASH accordance:1.49 (0.84, 2.63) eGFR decline >25% (RR) High DASH accordance:1.00 (ref.) Low DASH accordance: 1.36 (0.78, 2.37) | Age, gender, and race |
| Rebholz et al. (2015) [36], USA | ARIC, 14,832 (both) | Mean age of study sample: 54 | -Blood creatinine measured via modified kinetic Jaffe method using blood samples on five study visits from 1990–1992, 1993–1995, 1996–1998, and 2011–2013. -eGFR calculated via the CKD-EPI equation. | FFQ (66) Dietary pattern calculated using baseline FFQ from 1987–1989. | 22 | 1. Incident CKD (meets one of the following criteria 1. development of eGFR < 60 mL/min/1.73 m2 accompanied by ≥25% eGFR decline from baseline. 2. ICD 9/10 code for hospitalization due to CKD identified by surveillance of hospitalization and annual follow-up phone calls with study participants. 3. ICD 9/10 code for death due to CKD identified by linkage to the NDI. 4. ESRD identified by linkage to the US renal data system registry). | American Heart Association Life’s Simple 7 Healthy Diet Score (diet Score) | Incident CKD (HR) Poor healthy diet score: 1.00 (ref.) Intermediate healthy diet score: 1.02 (0.93, 1.13) Ideal healthy diet score: 0.99 (0.83, 1.18) P for trend: 0.55 | Age, gender, race, and baseline eGFR |
| Foster et al. (2015) [37], USA | The Framingham Offspring Cohort, 1802, (both) | Mean age of study sample: 59 | -Serum creatinine measured via modified Jaffe method using blood samples collected at baseline (1998–2001) and follow-up (2005–2008) and study visits. -eGFR calculated via the CKD-EPI equation. | FFQ (131). Dietary pattern calculated using FFQ administered at baseline from 1998–2001. | 6.6 | 1. Incident eGFR < 60 mL/min/1.73 m2 (presence of eGFR <60 mL/min/1.73 m2 at follow up among participants with eGFR > 60 mL/min/1.73 m2 at baseline. 2. Rapid eGFR decline (annual decrease in eGFR ≥ 3 mL/min/1.73 m2). | DGAI (diet score) | Incident eGFR < 60 mL/min/1.73 m2 (OR) Q1 (lowest quality): 1.0 (ref.) Q2: 0.77 (0.47, 1.27) Q3: 0.52 (0.31, 0.89) Q4 (highest quality): 0.63 (0.38, 1.07) P for trend: 0.045 Rapid eGFR decline (OR) Q1 (lowest quality): 1.0 (ref.) Q2: 0.83 (0.56, 1.22) Q3: 0.73 (0.49, 1.10) Q4 (highest quality): 0.69 (0.45, 1.05) P for trend: 0.07 | Age, gender, baseline eGFR, BMI, hypertension, diabetes, and dipstick proteinuria |
| Gopinath et al. (2013) [38], Australia | Blue Mountain Eye Study, 1952, (both) | Study sample age: ≥50 | -Serum creatinine measured via isotope dilution mass spectrometry using blood samples collected at follow-up examination from 2002–2004 -eGFR calculated via the MDRD equation. | FFQ (145). Diet score calculated via FFQ administered at baseline from 1992–1994. | 10 | 1.Incident CKD (eGFR < 60 mL/min/1.73 m2). | TDS (diet Score) | Incident CKD (OR): Q1: 1.00 (ref.) Q2: 0.99 (0.60, 1.64) Q3: 0.78 (0.46, 1.31) Q4: 0.68 (0.40, 1.15) P for trend: 0.10 | Age, serum total cholesterol, hypertension, and history of diagnosed diabetes |
| Turney et al. (2014) [39], United Kingdom | EPIC, 51,336 (both) | Study sample age: ≥20 | -Incidence of kidney stones determined by reviewing hospital records of study participants with (ICD)-9/10 codes. | FFQ (130). Dietary pattern from FFQ administered at baseline from 1993–1999. | 716,105 person-years | 1. Incidence of kidney stones. Follow-up calculated from the date of recruitment to the study to the earliest of date of kidney stone diagnosis, death or emigration. | 1. Vegetarian diet | Incident kidney stones (HR) Meat eater (>100g/day): 1.00 (ref.) Vegetarian: 0.69 (0.48, 0.98) P for trend: 0.04 | Smoking, BMI alcohol consumption, self-reported prior diabetes, and energy intake |
| Asghari et al. (2018) [40], Iran | TLGS, 1630 (both) | Mean age of study sample: 42.8 (SD: 11.2) | -Serum creatinine measured via Jaffe Kinetic reaction method with blood sample collected during fifth study visit from 2012–2015. | FFQ (168). Dietary pattern calculated using FFQ administered during third visit from 2006–2008. | 6.1 | 1. Incident CKD (eGFR < 60 mL/min/1.73 m2). | 1. Lacto-vegatarian (PCA) 2. Traditional Iranian (PCA) 3. High fat, high sugar (PCA) | Incident CKD (OR) Lacto-vegetarian T1: 1.0 (ref.) T2: 0.85 (0.62,1.15) T3: 0.57 (0.41, 0.80) P for trend: 0.002 Traditional Iranian: T1: 1.0 (ref.) T2: 1.26 (0.93, 1.72) T3: 0.91 (0.64, 1.32) P for trend: 0.698 High fat, high sugar: T1: 1.0 (ref) T2: 1.21 (0.87, 1.70) T3: 1.46 (1.03, 2.09) P for trend: 0.036 | Age, gender, BI smoking total energy intake, physical activity, diabetes, and hypertension |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ajjarapu, A.S.; Hinkle, S.N.; Li, M.; Francis, E.C.; Zhang, C. Dietary Patterns and Renal Health Outcomes in the General Population: A Review Focusing on Prospective Studies. Nutrients 2019, 11, 1877. https://doi.org/10.3390/nu11081877
Ajjarapu AS, Hinkle SN, Li M, Francis EC, Zhang C. Dietary Patterns and Renal Health Outcomes in the General Population: A Review Focusing on Prospective Studies. Nutrients. 2019; 11(8):1877. https://doi.org/10.3390/nu11081877
Chicago/Turabian StyleAjjarapu, Aparna S., Stefanie N. Hinkle, Mengying Li, Ellen C. Francis, and Cuilin Zhang. 2019. "Dietary Patterns and Renal Health Outcomes in the General Population: A Review Focusing on Prospective Studies" Nutrients 11, no. 8: 1877. https://doi.org/10.3390/nu11081877
APA StyleAjjarapu, A. S., Hinkle, S. N., Li, M., Francis, E. C., & Zhang, C. (2019). Dietary Patterns and Renal Health Outcomes in the General Population: A Review Focusing on Prospective Studies. Nutrients, 11(8), 1877. https://doi.org/10.3390/nu11081877






