Sedentary Behavior and Alcohol Consumption Increase Breast Cancer Risk Regardless of Menopausal Status: A Case-Control Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Design and Sample Size
2.2. Selection of Cases and Controls
2.3. Measurements
2.4. Quality Control
2.5. Statistical Analysis
3. Results
4. Discussion
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Instituto Nacional de Cancer José Alencar Gomes da Silva (INCA). Estimate 2018: Cancer Incidence in Brazil; INCA: Rio de Janeiro (RJ), Brazil, 2017.
- Allemani, C.; Matsuda, T.; Di Carlo, V.; Harewood, R.; Matz, M.; Nikšić, M.; Bonaventure, A.; Valkov, M.; Johnson, C.J.; Estève, J.; et al. Global surveillance of trends in cancer survival 2000-14 (CONCORD-3): Analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet 2018, 391, 1023–1075. [Google Scholar] [CrossRef]
- Simon, S.D.; Bines, J.; Werutsky, G.; Nunes, J.S.; Pacheco, F.C.; Segalla, J.G.; Gomes, A.J.S.; Adam, V.E.B.; Gimenes, D.L.; Crocamo, S.; et al. Characteristics and prognosis of stage I-III breast cancer subtypes in Brazil: The AMAZONA retrospective cohort study. Breast 2019, 44, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Kolak, A.; Kamińska, M.; Sygit, K.; Budny, A.; Surdyka, D.; Kukiełka-Budny, B.; Burdan, F. Primary and secondary prevention of breast cancer. Ann. Agric. Environ. Med. 2017, 24, 549–553. [Google Scholar] [CrossRef] [PubMed]
- McKenzie, F.; Ferrari, P.; Freisling, H.; Chajès, V.; Rinaldi, S.; de Batlle, J.; Dahm, C.C.; Overvad, K.; Baglietto, L.; Dartois, L.; et al. Healthy lifestyle and risk of breast cancer among postmenopausal women in the European Prospective Investigation into Cancer and Nutrition cohort study. Int. J. Cancer 2015, 136, 2640–2648. [Google Scholar] [CrossRef] [PubMed]
- Couch, F.J.; Shimelis, H.; Hu, C.; Hart, S.N.; Polley, E.C.; Na, J.; Hallberg, E.; Moore, R.; Thomas, A.; Lilyquist, J.; et al. Associations Between Cancer Predisposition Testing Panel Genes and Breast Cancer. JAMA Oncol. 2017, 3, 1190–1196. [Google Scholar] [CrossRef] [PubMed]
- Tamimi, R.M.; Spiegelman, D.; Smith-Warner, S.A.; Wang, M.; Pazaris, M.; Willett, W.C.; Eliassen, A.H.; Hunter, D.J. Population Attributable Risk of Modifiable and Nonmodifiable Breast Cancer Risk Factors in Postmenopausal Breast Cancer. Am. J. Epidemiol. 2016, 184, 884–893. [Google Scholar] [CrossRef] [PubMed]
- Godinho-Mota, J.C.M.; Martins, K.A.; Vaz-Gonçalves, L.; Mota, J.F.; Soares, L.R.; Freitas-Junior, R. Visceral adiposity increases the risk of breast cancer: A case-control study. Nutr. Hosp. 2018, 35, 576–581. [Google Scholar] [PubMed]
- Arthur, R.; Wassertheil-Smoller, S.; Manson, J.E.; Luo, J.; Snetselaar, L.; Hastert, T.; Caan, B.; Qi, L.; Rohan, T. The Combined Association of Modifiable Risk Factors with Breast Cancer Risk in the Women’s Health Initiative. Cancer Prev. Res. (Phila) 2018, 11, 317–326. [Google Scholar] [CrossRef]
- Godinho-Mota, J.C.M.; Gonçalves, L.V.; Soares, L.R.; Mota, J.F.; Martins, K.A.; Freitas-Junior, I.; Freitas-Junior, R. Abdominal Adiposity and Physical Inactivity Are Positively Associated with Breast Cancer: A Case-Control Study. BioMed Res. Int. 2018, 2018, 4783710. [Google Scholar] [CrossRef]
- Picon-Ruiz, M.; Morata-Tarifa, C.; Valle-Goffin, J.J.; Friedman, E.R.; Slingerland, J.M. Obesity and adverse breast cancer risk and outcome: Mechanistic insights and strategies for intervention. CA Cancer J. Clin. 2017, 67, 378–397. [Google Scholar] [CrossRef] [PubMed]
- Inumaru, L.E.; Irineu Gomes Duarte Quintanilha, M.; Aparecida da Silveira, É.; Veloso Naves, M.M. Risk and protective factors for breast cancer in Midwest of Brazil. J. Environ. Public Health 2012, 2012, 356851. [Google Scholar] [CrossRef] [PubMed]
- Winters, S.; Martin, C.; Murphy, D.; Shokar, N.K. Breast Cancer Epidemiology, Prevention, and Screening. Prog. Mol. Biol. Transl. Sci. 2017, 151, 1–32. [Google Scholar] [PubMed]
- Morales, L.; Alvarez-Garriga, C.; Matta, J.; Ortiz, C.; Vergne, Y.; Vargas, W.; Acosta, H.; Ramírez, J.; Perez-Mayoral, J.; Bayona, M. Factors associated with breast cancer in Puerto Rican women. J. Epidemiol. Glob. Health 2013, 3, 205–215. [Google Scholar] [CrossRef] [PubMed]
- Craig, C.L.; Marshall, A.L.; Sjöström, M.; Bauman, A.E.; Booth, M.L.; Ainsworth, B.E.; Pratt, M.; Ekelund, U.; Yngve, A.; Sallis, J.F.; et al. International physical activity questionnaire: 12-country reliability and validity. Med. Sci. Sports Exerc. 2003, 35, 1381–1395. [Google Scholar] [CrossRef] [PubMed]
- Gaudet, M.M.; Gapstur, S.M.; Sun, J.; Diver, W.R.; Hannan, L.M.; Thun, M.J. Active smoking and breast cancer risk: Original cohort data and meta-analysis. J. Natl. Cancer Inst. 2013, 105, 515–525. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Heng, Y.J.; Eliassen, A.H.; Tamimi, R.M.; Hazra, A.; Carey, V.J.; Ambrosone, C.B.; de Andrade, V.P.; Brufsky, A.; Couch, F.J.; et al. Alcohol consumption and breast tumor gene expression. Breast Cancer Res. 2017, 19, 108. [Google Scholar] [CrossRef]
- Habicht, J. Estandarización de Métodos Epidemiológicos Cuantitativos Sobre el Terreno. Bol. Oficina Sanit. Panam. 1974, 76, 375–384. [Google Scholar]
- Organization, W.H. Obesity: Preventing and Managing the Global Epidemic; World Health Organization: Geneva, Switzerland, 2000. [Google Scholar]
- Lohman, T.G.; Roche, A.F.; Martorell, R. Anthropometric Standardization Reference Manual; Human Kinetics Books: Champaign, IL, USA, 1988; 177p. [Google Scholar]
- Lean, M.E.; Han, T.S.; Morrison, C.E. Waist circumference as a measure for indicating need for weight management. BMJ 1995, 311, 158–161. [Google Scholar] [CrossRef]
- Kerr, J.; Anderson, C.; Lippman, S.M. Physical activity, sedentary behaviour, diet, and cancer: An update and emerging new evidence. Lancet Oncol. 2017, 18, e457–e471. [Google Scholar] [CrossRef]
- Liu, Y.; Colditz, G.A.; Rosner, B.; Berkey, C.S.; Collins, L.C.; Schnitt, S.J.; Connolly, J.L.; Chen, W.Y.; Willett, W.C.; Tamimi, R.M. Alcohol intake between menarche and first pregnancy: A prospective study of breast cancer risk. J. Natl. Cancer Inst. 2013, 105, 1571–1578. [Google Scholar] [CrossRef] [PubMed]
- Hamajima, N.; Hirose, K.; Tajima, K.; Rohan, T.; Calle, E.E.; Heath, C.W.; Coates, R.J.; Liff, J.M.; Talamini, R.; Chantarakul, N.; et al. Alcohol, tobacco and breast cancer—collaborative reanalysis of individual data from 53 epidemiological studies, including 58,515 women with breast cancer and 95,067 women without the disease. Br. J. Cancer 2002, 87, 1234–1245. [Google Scholar] [PubMed]
- Bagnardi, V.; Rota, M.; Botteri, E.; Tramacere, I.; Islami, F.; Fedirko, V.; Scotti, L.; Jenab, M.; Turati, F.; Pasquali, E.; et al. Light alcohol drinking and cancer: A meta-analysis. Ann Oncol. 2013, 24, 301–308. [Google Scholar] [CrossRef] [PubMed]
- Eliassen, A.H.; Hankinson, S.E.; Rosner, B.; Holmes, M.D.; Willett, W.C. Physical activity and risk of breast cancer among postmenopausal women. Arch. Intern. Med. 2010, 170, 1758–1764. [Google Scholar] [CrossRef] [PubMed]
- Kawai, M.; Kakugawa, Y.; Nishino, Y.; Hamanaka, Y.; Ohuchi, N.; Minami, Y. Anthropometric factors, physical activity, and breast cancer risk in relation to hormone receptor and menopausal status in Japanese women: A case-control study. Cancer Causes Control 2013, 24, 1033–1044. [Google Scholar] [CrossRef] [PubMed]
- Moore, S.C.; Lee, I.M.; Weiderpass, E.; Campbell, P.T.; Sampson, J.N.; Kitahara, C.M.; Keadle, S.K.; Arem, H.; Berrington de Gonzalez, A.; Hartge, P.; et al. Association of Leisure-Time Physical Activity with Risk of 26 Types of Cancer in 1.44 Million Adults. JAMA Intern. Med. 2016, 176, 816–825. [Google Scholar] [CrossRef] [PubMed]
- Ma, H.; Xu, X.; Clague, J.; Lu, Y.; Togawa, K.; Wang, S.S.; Clarke, C.A.; Lee, E.; Park, H.L.; Sullivan-Halley, J.; et al. Recreational physical activity and risk of triple negative breast cancer in the California Teachers Study. Breast Cancer Res. 2016, 18, 62. [Google Scholar] [CrossRef]
- Lope, V.; Martín, M.; Castelló, A.; Casla, S.; Ruiz, A.; Baena-Cañada, J.M.; Casas, A.M.; Calvo, L.; Bermejo, B.; Muñoz, M.; et al. Physical activity and breast cancer risk by pathological subtype. Gynecol. Oncol. 2017, 144, 577–585. [Google Scholar] [CrossRef]
- Ballard-Barbash, R.; Friedenreich, C.M.; Courneya, K.S.; Siddiqi, S.M.; McTiernan, A.; Alfano, C.M. Physical activity, biomarkers, and disease outcomes in cancer survivors: A systematic review. J. Natl. Cancer Inst. 2012, 104, 815–840. [Google Scholar] [CrossRef]
- De Boer, M.C.; Wörner, E.A.; Verlaan, D.; van Leeuwen, P.A.M. The Mechanisms and Effects of Physical Activity on Breast Cancer. Clin. Breast Cancer 2017, 17, 272–278. [Google Scholar] [CrossRef]
- Hibler, E. Epigenetics and Colorectal Neoplasia: The Evidence for Physical Activity and Sedentary Behavior. Curr. Colorectal. Cancer Rep. 2015, 11, 388–396. [Google Scholar] [CrossRef][Green Version]
- Hardefeldt, P.J.; Penninkilampi, R.; Edirimanne, S.; Eslick, G.D. Physical Activity and Weight Loss Reduce the Risk of Breast Cancer: A Meta-analysis of 139 Prospective and Retrospective Studies. Clin. Breast Cancer 2018, 18, e601–e612. [Google Scholar] [CrossRef] [PubMed]
- Paxton, R.J.; Garner, W.; Dean, L.T.; Logan, G.; Allen-Watts, K. Health Behaviors and Lifestyle Interventions in African American Breast Cancer Survivors: A Review. Front. Oncol. 2019, 9, 3. [Google Scholar] [CrossRef] [PubMed]
- Amadou, A.; Ferrari, P.; Muwonge, R.; Moskal, A.; Biessy, C.; Romieu, I.; Hainaut, P. Overweight, obesity and risk of premenopausal breast cancer according to ethnicity: A systematic review and dose-response meta-analysis. Obes. Rev. 2013, 14, 665–678. [Google Scholar] [CrossRef] [PubMed]
- Nagrani, R.; Mhatre, S.; Rajaraman, P.; Soerjomataram, I.; Boffetta, P.; Gupta, S.; Parmar, V.; Badwe, R.; Dikshit, R. Central obesity increases risk of breast cancer irrespective of menopausal and hormonal receptor status in women of South Asian Ethnicity. Eur. J. Cancer 2016, 66, 153–161. [Google Scholar] [CrossRef] [PubMed]
- White, A.J.; Nichols, H.B.; Bradshaw, P.T.; Sandler, D.P. Overall and central adiposity and breast cancer risk in the Sister Study. Cancer 2015, 121, 3700–3708. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.R.; Hwang, I.C.; Han, K.D.; Jung, J.; Seo, M.H. Waist circumference and risk of breast cancer in Korean women: A nationwide cohort study. Int. J. Cancer 2018, 142, 1554–1559. [Google Scholar] [CrossRef]
- Paxton, R.J.; King, D.W.; Garcia-Prieto, C.; Connors, S.K.; Hernandez, M.; Gor, B.J.; Jones, L.A. Associations between body size and serum estradiol and sex hormone-binding globulin levels in premenopausal African American women. J. Clin. Endocrinol. Metab. 2013, 98, E485–E490. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer. IARC monographs on the evaluation of carcinogenic risks to humans, volume 100 (E). A review of human carcinogens: Personal habits and indoor combustions. Lyon Fr. IARC 2012, 100, 43–213. [Google Scholar]
- Macacu, A.; Autier, P.; Boniol, M.; Boyle, P. Active and passive smoking and risk of breast cancer: A meta-analysis. Breast Cancer Res. Treat. 2015, 154, 213–224. [Google Scholar] [CrossRef]
- Gram, I.T.; Park, S.-Y.; Kolonel, L.N.; Maskarinec, G.; Wilkens, L.R.; Henderson, B.E.; Le Marchand, L. Smoking and risk of breast cancer in a racially/ethnically diverse population of mainly women who do not drink alcohol: The MEC Study. Am. J. Epidemiol. 2015, 182, 917–925. [Google Scholar] [CrossRef]
- World Health Organization (WHO). International Agency for Research on Cancer (IARC) Monographs on the Evaluation of Carcinogenic Risks to Humans. Tobacco Smoke and Involuntary Smoking. Available online: https://publications.iarc.fr/101 (accessed on 4 February 2019).
- Perera, F.P.; Estabrook, A.; Hewer, A.; Channing, K.; Rundle, A.; Mooney, L.A.; Whyatt, R.; Phillips, D.H. Carcinogen-DNA adducts in human breast tissue. Cancer Epidemiol. Biomark. Prev. 1995, 4, 233–238. [Google Scholar]
- Ko, K.P.; Kim, S.J.; Huzarski, T.; Gronwald, J.; Lubinski, J.; Lynch, H.T.; Armel, S.; Park, S.K.; Karlan, B.; Singer, C.F.; et al. The association between smoking and cancer incidence in BRCA1 and BRCA2 mutation carriers. Int. J. Cancer 2018, 142, 2263–2272. [Google Scholar] [CrossRef]
- Gao, C.M.; Ding, J.H.; Li, S.P.; Liu, Y.T.; Qian, Y.; Chang, J.; Tang, J.H.; Tajima, K. Active and passive smoking, and alcohol drinking and breast cancer risk in chinese women. Asian Pac. J. Cancer Prev. 2013, 14, 993–996. [Google Scholar] [CrossRef]
- Rosenthal, E.; Moyes, K.; Arnell, C.; Evans, B.; Wenstrup, R.J. Incidence of BRCA1 and BRCA2 non-founder mutations in patients of Ashkenazi Jewish ancestry. Breast Cancer Res. Treat. 2015, 149, 223–227. [Google Scholar] [CrossRef]
- Walsh, T.; Mandell, J.B.; Norquist, B.M.; Casadei, S.; Gulsuner, S.; Lee, M.K.; King, M.C. Genetic Predisposition to Breast Cancer Due to Mutations Other Than BRCA1 and BRCA2 Founder Alleles Among Ashkenazi Jewish Women. JAMA Oncol. 2017, 3, 1647–1653. [Google Scholar] [CrossRef]
- Koifman, S.; Koifman, R.J. Breast cancer mortality among Ashkenazi Jewish women in São Paulo and Porto Alegre, Brazil. Breast Cancer Res. 2001, 3, 270–275. [Google Scholar] [CrossRef]
- Shieh, Y.; Scott, C.G.; Jensen, M.R.; Norman, A.D.; Bertrand, K.A.; Pankratz, V.S.; Brandt, K.R.; Visscher, D.W.; Shepherd, J.A.; Tamimi, R.M.; et al. Body mass index, mammographic density, and breast cancer risk by estrogen receptor subtype. Breast Cancer Res. 2019, 21, 48. [Google Scholar] [CrossRef]
| Premenopausal 213 (39.37) | Postmenopausal 328 (60.63) | |||||
|---|---|---|---|---|---|---|
| Cases | Controls | p-Value | Cases | Controls | p-Value | |
| 80 (37.56) | 133 (62.44) | 117 (35.67) | 211 (64.33) | |||
| Age (years) | 41.84 ± 6.62 | 41.41 ± 7.01 | 0.488 | 59.21 ± 8.78 | 58.77 ± 8.22 | 0.722 |
| Marital status | 0.594 | 0.184 | ||||
| With partner | 54 (67.50) | 85 (63.91) | 61 (52.14) | 126 (59.72) | ||
| Without partner | 26 (32.50) | 48 (36.09) | 56 (47.86) | 85 (40.28) | ||
| Schooling | 0.328 * | <0.001 * | ||||
| Grade school | 5 (6.33) | 3 (2.34) | 14 (12.73) | 10 (5.03) | ||
| Incomplete high school | 9 (11.39) | 13 (10.16) | 44 (40.00) | 49 (24.62) | ||
| High school and undergrad course | 65 (82.28) | 112 (87.50) | 52 (47.27) | 140 (70.35) | ||
| Premenopausal 213 (39.37) | Postmenopausal 328 (60.63) | |||||
|---|---|---|---|---|---|---|
| Cases | Controls | p-Value | Cases | Controls | p-Value | |
| 80 (37.56) | 133 (62.44) | 117 (35.67) | 211 (64.33) | |||
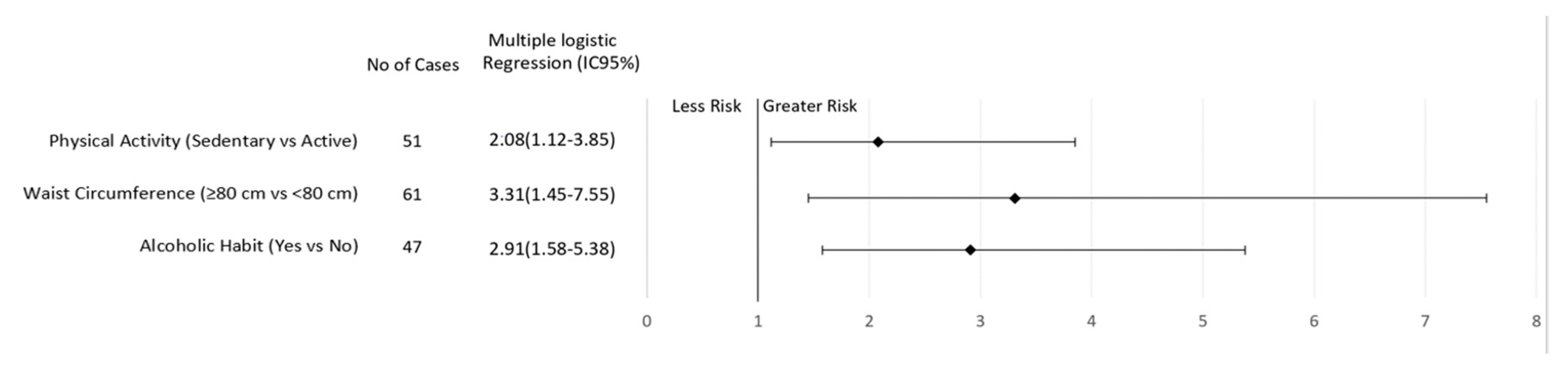
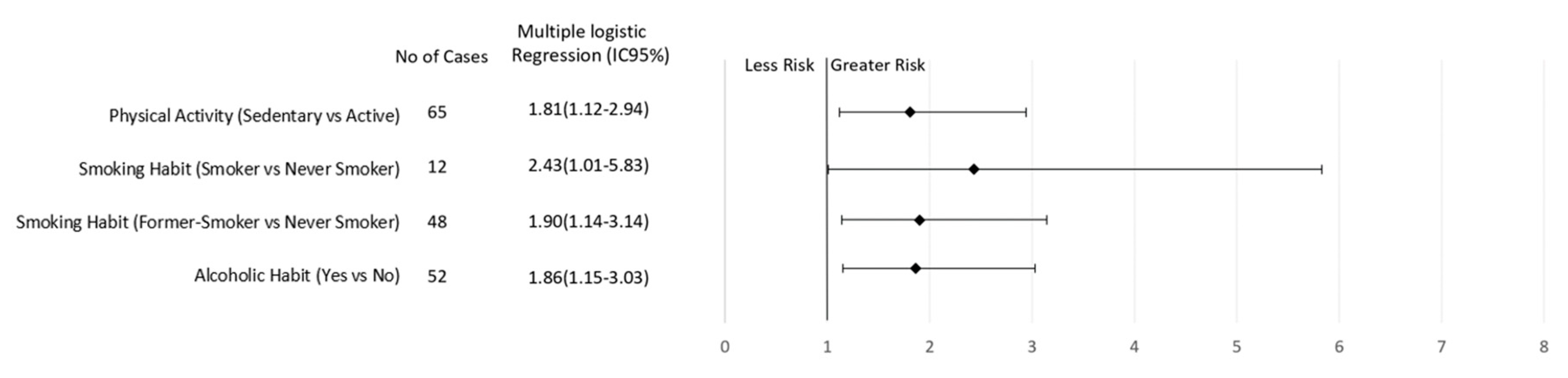
| Physical activity level * | 0.011 | 0.021 | ||||
| Active | 29 (36.25) | 73 (54.89) | 52 (44.44) | 123 (58.29) | ||
| Sedentary | 51 (63.75) | 60 (45.11) | 65 (55.56) | 88 (41.71) | ||
| Cigarette consumption (units/day) | 16.18 ± 9.99 | 8.72 ± 4.90 | <0.005 | 17.59 ± 12.48 | 9.22 ± 6.31 | <0.001 |
| Smoking habit * | 0.271 | 0.005 | ||||
| Never smoker | 58 (72.50) | 108 (81.20) | 57 (48.72) | 141 (66.82) | ||
| Smoker | 6 (7.50) | 5 (3.76) | 12 (10.26) | 12 (5.69) | ||
| Ex-smoker | 16 (20.00) | 20 (15.04) | 48 (41.03) | 58 (27.49) | ||
| Ethanol consumption (g/day) | 1.59 ± 1.04 | 0.82 ± 0.35 | <0.001 | 1.44 ± 0.76 | 0.90 ± 0.39 | <0.001 |
| Alcoholic habit * | 0.001 | 0.016 | ||||
| Not | 33 (41.25) | 86 (64.66) | 65 (55.56) | 146 (69.19) | ||
| Yes | 47 (58.75) | 47 (35.34) | 52 (44.44) | 65 (30.81) | ||
| Premenopausal 213 (39.37) | Postmenopausal 328 (60.63) | |||||
|---|---|---|---|---|---|---|
| Cases | Controls | p-Value | Cases | Controls | p-Value | |
| 80 (37.56) | 133 (62.44) | 117 (35.67) | 211 (64.33) | |||
| Height (meters) | 1.58 ± 0.06 | 1.60 ± 0.06 | 0.19 | 1.55 ± 0.06 | 1.57 ± 0.06 | 0.07 |
| Body weight (kg) | 66.01 ± 12.85 | 69.66 ± 13.84 | 0.04 | 68.37 ± 13.61 | 68.51 ± 13.14 | 0.78 |
| BMI | 26.26 ± 4.73 | 27.21 ± 5.34 | 0.19 | 28.28 ± 5.62 | 28.02 ± 5.09 | 0.96 |
| Normal weight * | 0 | 1 (0.75) | 0.06 | 2 (1.71) | 3 (1.42) | 0.74 |
| Overweight | 40 (50.00) | 85 (63.91) | 79 (67.52) | 151 (71.56) | ||
| Obesity | 40 (50.00) | 47 (35.34) | 36 (30.77) | 57 (27.01) | ||
| Waist circumference | 87.61 ± 11.79 | 87.75 ± 12.79 | 0.97 | 93.33 ± 12.08 | 91.73 ± 12.66 | 0.39 |
| Low risk (<80 cm) * | 16 (20.78) | 41 (32.54) | 0.07 | 12 (10.91) | 31 (15.05) | 0.31 |
| Increased risk (≥80 cm) | 61 (79.22) | 85 (67.46) | 98 (89.09) | 175 (84.95) | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Godinho-Mota, J.C.M.; Gonçalves, L.V.; Mota, J.F.; Soares, L.R.; Schincaglia, R.M.; Martins, K.A.; Freitas-Junior, R. Sedentary Behavior and Alcohol Consumption Increase Breast Cancer Risk Regardless of Menopausal Status: A Case-Control Study. Nutrients 2019, 11, 1871. https://doi.org/10.3390/nu11081871
Godinho-Mota JCM, Gonçalves LV, Mota JF, Soares LR, Schincaglia RM, Martins KA, Freitas-Junior R. Sedentary Behavior and Alcohol Consumption Increase Breast Cancer Risk Regardless of Menopausal Status: A Case-Control Study. Nutrients. 2019; 11(8):1871. https://doi.org/10.3390/nu11081871
Chicago/Turabian StyleGodinho-Mota, Jordana Carolina Marques, Larissa Vaz Gonçalves, Joao Felipe Mota, Leonardo Ribeiro Soares, Raquel Machado Schincaglia, Karine Anusca Martins, and Ruffo Freitas-Junior. 2019. "Sedentary Behavior and Alcohol Consumption Increase Breast Cancer Risk Regardless of Menopausal Status: A Case-Control Study" Nutrients 11, no. 8: 1871. https://doi.org/10.3390/nu11081871
APA StyleGodinho-Mota, J. C. M., Gonçalves, L. V., Mota, J. F., Soares, L. R., Schincaglia, R. M., Martins, K. A., & Freitas-Junior, R. (2019). Sedentary Behavior and Alcohol Consumption Increase Breast Cancer Risk Regardless of Menopausal Status: A Case-Control Study. Nutrients, 11(8), 1871. https://doi.org/10.3390/nu11081871






