Vitamin D Deficiency and Effects of Vitamin D Supplementation on Disease Severity in Patients with Atopic Dermatitis: A Systematic Review and Meta-Analysis in Adults and Children
Abstract
1. Introduction
2. Methods
2.1. Search Procedures
2.2. Eligibility Criteria for Inclusion and Data Extraction
2.3. Primary Outcomes
- From observational studies, serum 25(OH)D levels in AD patients versus HC.
- From interventional trials, changes in SCORAD or EASI score in the VitD supplemented AD group compared to the placebo supplemented AD group.
2.4. Secondary Outcomes
- The relationship between serum 25(OH)D levels in AD with SCORAD or EASI score.
- Effects of VitD supplementation on secondary infections of skin in AD.
- The relationship between serum 25(OH)D levels, serum IgE levels and total eosinophil count (TEC) in the AD population and HC, including changes in these post-supplementation.
- In interventional trials only: Effects of the co-usage of topical steroids.
- The relationship, in AD patients, between serum 25(OH)D levels and cathelicidin LL-37 or Cathelicidin Antimicrobial Peptide (CAMP) and changes post-supplementation.
- The relationship, in AD patients, between serum 25(OH)D levels and serum cytokines, with changes post-supplementation.
- The relationship between serum 25(OH)D levels and atopic sensitisation.
2.5. Statistical Analysis
3. Results
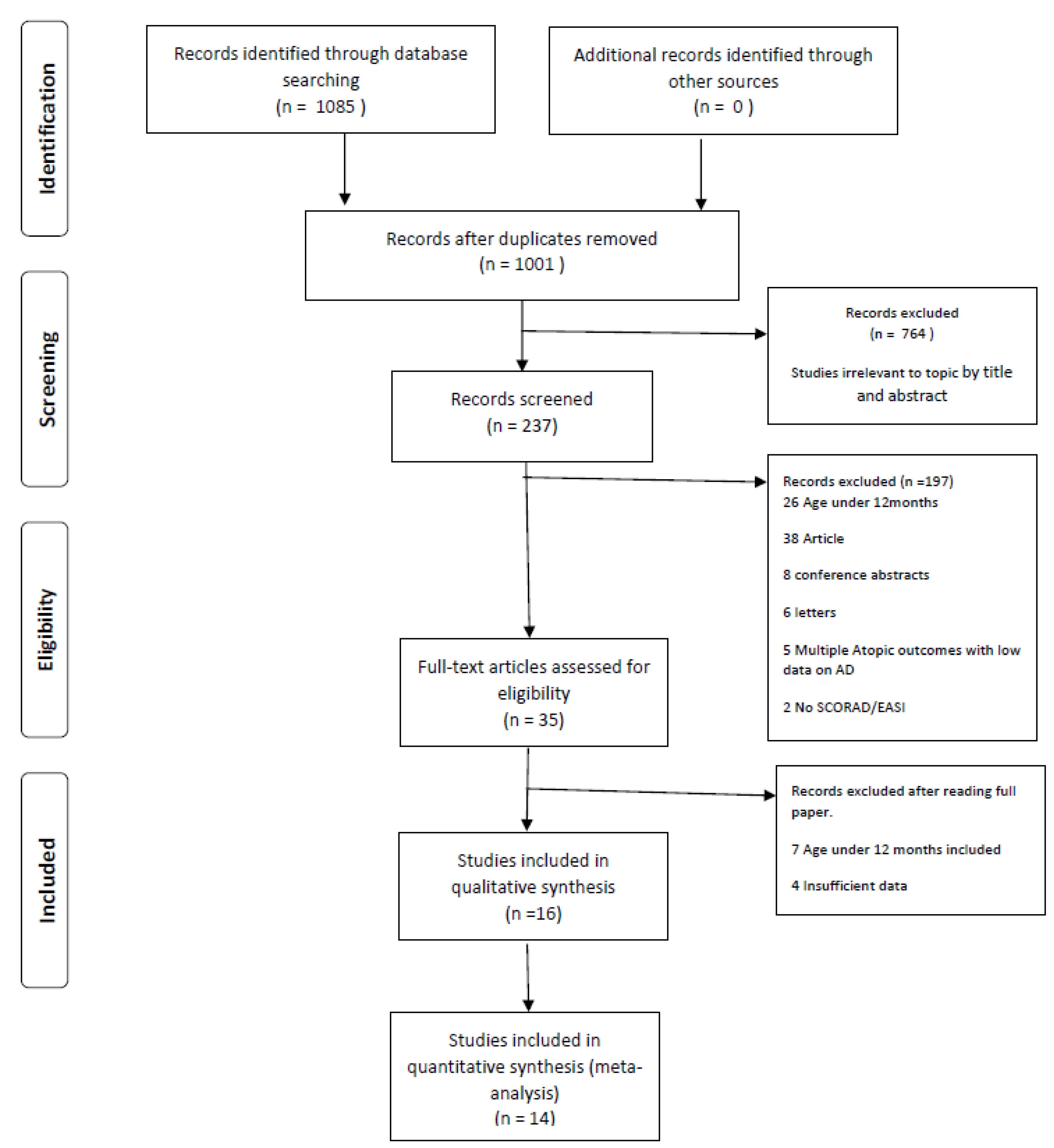
3.1. Systematic Review
3.2. Meta-Analysis
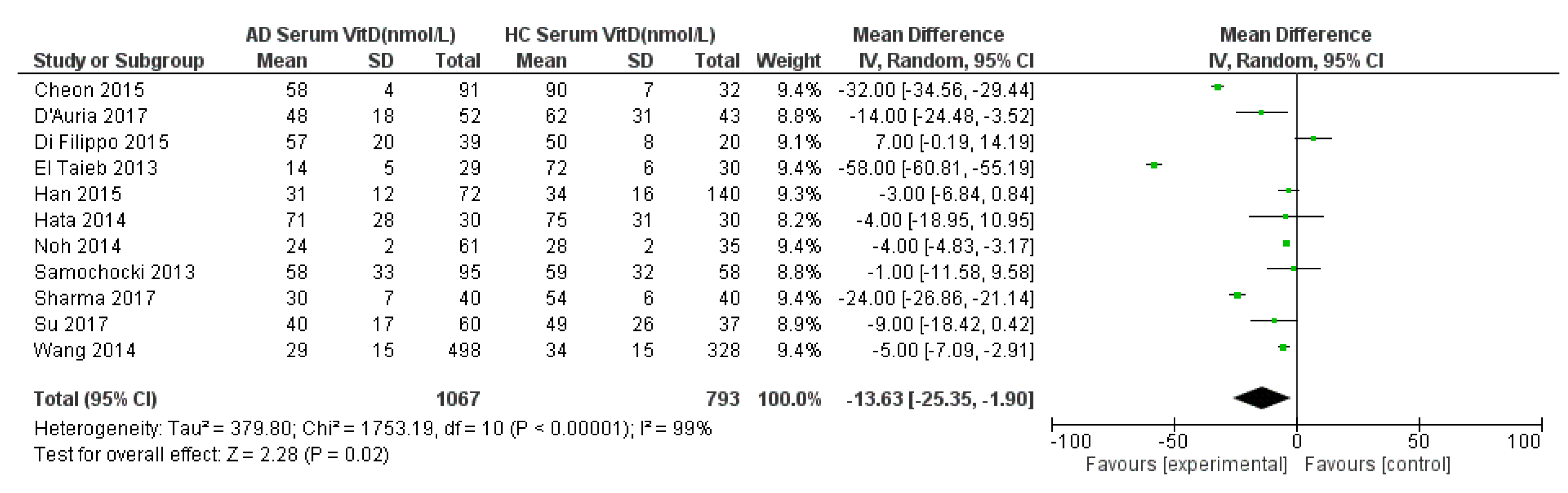
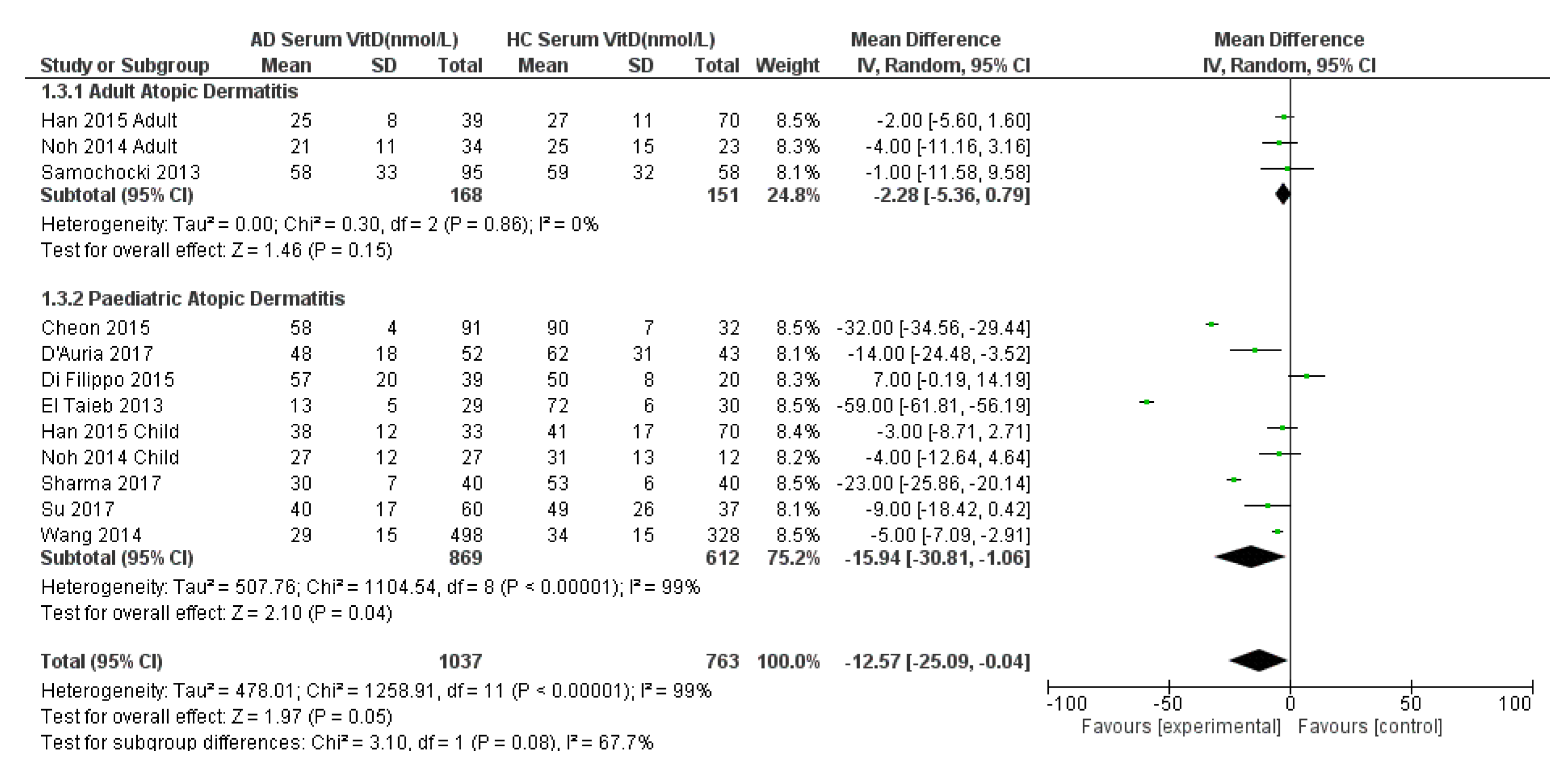
3.2.1. Serum VitD in AD Compared to HC, with Sub-Analysis of the Paediatric Population
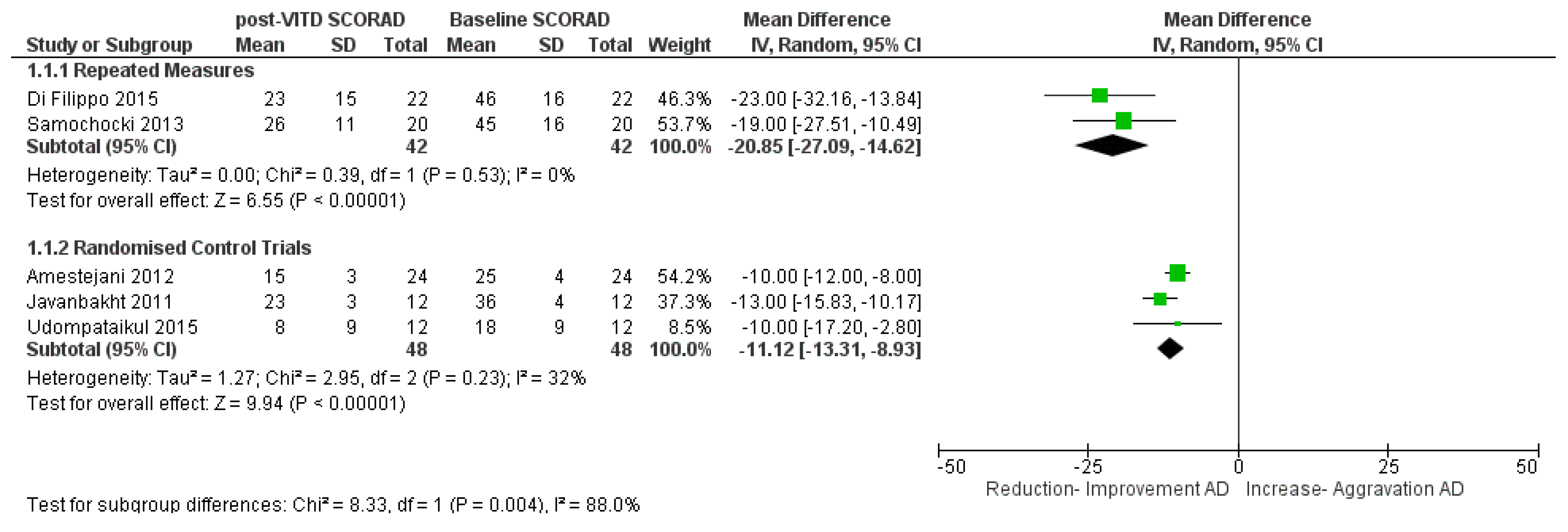
3.2.2. VitD Interventional Trials and Change in Clinical AD Severity (SCORAD)
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Coutanceau, C.; Stalder, J.F. Analysis of correlations between patient-oriented SCORAD (PO-SCORAD) and other assessment scores of atopic dermatitis severity and quality of life. Dermatology 2014, 229, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Boccardi, D.; D’Auria, E.; Turati, F.; DI, M.V.; Sortino, S.; Riva, E.; Cerri, A. Disease severity and quality of life in children with atopic dermatitis: PO-SCORAD in clinical practice. Minerva Pediatr. 2017, 69, 373–380. [Google Scholar] [PubMed]
- Boguniewicz, M.; Fonacier, L.; Guttman-Yassky, E.; Ong, P.Y.; Silverberg, J.; Farrar, J.R. Atopic dermatitis yardstick: Practical recommendations for an evolving therapeutic landscape. Ann. Allergy Asthma Immunol. 2018, 120, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Chaplin, S. Guide to treatments used for atopic dermatitis in adults. Prescriber 2016, 27, 30–39. [Google Scholar] [CrossRef]
- Weiland, S.K.; Husing, A.; Strachan, D.P.; Rzehak, P.; Pearce, N. Climate and the prevalence of symptoms of asthma, allergic rhinitis, and atopic eczema in children. Occup. Environ. Med. 2004, 61, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Drucker, A.M.; Wang, A.R.; Li, W.-Q.; Sevetson, E.; Block, J.K.; Qureshi, A.A. The Burden of Atopic Dermatitis: Summary of a Report for the National Eczema Association. J. Investig. Dermatol. 2017, 137, 26–30. [Google Scholar] [CrossRef] [PubMed]
- Shrestha, S.; Miao, R.; Wang, L.; Chao, J.; Yuce, H.; Wei, W. Burden of Atopic Dermatitis in the United States: Analysis of Healthcare Claims Data in the Commercial, Medicare, and Medi-Cal Databases. Adv. Ther. 2017, 34, 1989–2006. [Google Scholar] [CrossRef] [PubMed]
- Tokura, Y. Extrinsic and intrinsic types of atopic dermatitis. J. Dermatol. Sci. 2010, 58, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Su, C.; Yang, T.; Wu, Z.; Zhong, J.; Huang, Y.; Huang, T.; Zheng, E. Differentiation of T-helper cells in distinct phases of atopic dermatitis involves Th1/Th2 and Th17/Treg. Eur. J. Inflamm. 2017, 15, 46–52. [Google Scholar] [CrossRef]
- D’Auria, E.; Banderali, G.; Barberi, S.; Gualandri, L.; Pietra, B.; Riva, E.; Cerri, A. Atopic dermatitis: Recent insight on pathogenesis and novel therapeutic target. Asian Pac. J. Allergy Immunol. 2016, 34, 98–108. [Google Scholar]
- Von Kobyletzki, L.; Svensson, A.; Apfelbacher, C.; Schmitt, J. Factors that predict remission of infant atopic dermatitis: A systematic review. Acta Derm. Venereol. 2015, 95, 389–394. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.J.; Kim, S.N.; Lee, Y.W.; Choe, Y.B.; Ahn, K.J. Vitamin D status and efficacy of vitamin D supplementation in atopic dermatitis: A systematic review and meta-analysis. Nutrients 2016, 8, 789. [Google Scholar] [CrossRef] [PubMed]
- Kunz, B.; Oranje, A.P.; Labrèze, L.; Stalder, J.F.; Ring, J.; Taïeb, A. Clinical Validation and Guidelines for the SCORAD Index: Consensus Report of the European Task Force on Atopic Dermatitis. Dermatology 1997, 195, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Wollenberg, A.; Oranje, A.; Deleuran, M.; Simon, D.; Szalai, Z.; Kunz, B.; Svensson, A.; Barbarot, S.; Kobyletzki, L.; Taieb, A.; et al. ETFAD/EADV Eczema task force 2015 position paper on diagnosis and treatment of atopic dermatitis in adult and paediatric patients. J. Eur. Acad. Dermatol. Venereol. 2016, 30, 729–747. [Google Scholar] [CrossRef] [PubMed]
- Sidbury, R.; Sullivan, A.; Thadhani, R.; Camargo, C. Randomized controlled trial of vitamin D supplementation for winter-related atopic dermatitis in Boston: A pilot study. Br. J. Dermatol. 2008, 159, 245–247. [Google Scholar] [CrossRef] [PubMed]
- Camargo, C.A.; Ganmaa, D.; Sidbury, R.; Erdenedelger, K.; Radnaakhand, N.; Khandsuren, B. Randomized trial of vitamin D supplementation for winter-related atopic dermatitis in children. J. Allergy Clin. Immunol. 2014, 134, 831–835. [Google Scholar] [CrossRef] [PubMed]
- Liang, Y.; Chang, C.; Lu, Q. The Genetics and Epigenetics of Atopic Dermatitis-Filaggrin and Other Polymorphisms. Clin. Rev. Allergy Immunol. 2016, 51, 315–328. [Google Scholar] [CrossRef] [PubMed]
- Bieber, T. Atopic dermatitis. N. Engl. J. Med. 2008, 358, 1483–1494. [Google Scholar] [CrossRef]
- Oda, Y.; Uchida, Y.; Moradian, S.; Crumrine, D.; Elias, P.M.; Bikle, D.D. Vitamin D receptor and coactivators SRC2 and 3 regulate epidermis-specific sphingolipid production and permeability barrier formation. J. Investig. Dermatol. 2009, 129, 1367–1378. [Google Scholar] [CrossRef]
- Umar, M.; Sastry, K.S.; Al Ali, F.; Al-Khulaifi, M.; Wang, E.; Chouchane, A.I. Vitamin D and the Pathophysiology of Inflammatory Skin Diseases. Skin Pharmacol. Physiol. 2018, 31, 74–86. [Google Scholar] [CrossRef]
- Amon, U.; Baier, L.; Yaguboglu, R.; Ennis, M.; Holick, M.F.; Amon, J. Serum 25-hydroxyvitamin D levels in patients with skin diseases including psoriasis, infections, and atopic dermatitis. Dermatoendocrinology 2018, 10, e1442159. [Google Scholar] [CrossRef] [PubMed]
- SACN. Vitamin D and Health. Available online: https://www.gov.uk/government/groups/scientific-advisory-committee-on-nutrition (accessed on 21 October 2018).
- Kim, G.; Bae, J.H. Vitamin D and atopic dermatitis: A systematic review and meta-analysis. Nutrition 2016, 32, 913–920. [Google Scholar] [CrossRef] [PubMed]
- Huang, C.M.; Lara-Corrales, I.; Pope, E. Effects of Vitamin D levels and supplementation on atopic dermatitis: A systematic review. Pediatr. Dermatol. 2018, 35, 754–760. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Kaur, T.; Malhotra, S.K.; Rai, J.; Chaudhari, S. Correlation of Vitamin D3 Levels and SCORAD Index in Atopic Dermatits: A Case Control Study. J. Clin. Diagn. Res. 2017, 11, WC01–WC03. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.S.; Hon, K.L.; Kong, A.P.; Pong, H.N.; Wong, G.W.; Leung, T.F. Vitamin D deficiency is associated with diagnosis and severity of childhood atopic dermatitis. Pediatr. Allergy Immunol. 2014, 25, 30–35. [Google Scholar] [CrossRef]
- Cheon, B.R.; Shin, J.E.; Kim, Y.J.; Shim, J.W.; Kim, D.S.; Jung, H.L.; Park, M.S.; Shim, J.Y. Relationship between serum 25-hydroxyvitamin D and interleukin-31 levels, and the severity of atopic dermatitis in children. Korean J. Pediatr. 2015, 58, 96–101. [Google Scholar] [CrossRef] [PubMed]
- D’Auria, E.; Barberi, S.; Cerri, A.; Boccardi, D.; Turati, F.; Sortino, S.; Banderali, G.; Ciprandi, G. Vitamin D status and body mass index in children with atopic dermatitis: A pilot study in Italian children. Immunol. Lett. 2017, 181, 31–35. [Google Scholar] [CrossRef]
- Di Filippo, P.; Scaparrotta, A.; Rapino, D.; Cingolani, A.; Attanasi, M.; Petrosino, M.I.; Chuang, K.; Di Pillo, S.; Chiarelli, F. Vitamin D supplementation modulates the immune system and improves atopic dermatitis in children. Int. Arch. Allergy Immunol. 2015, 166, 91–96. [Google Scholar] [CrossRef]
- El Taieb, M.A.; Fayed, H.M.; Aly, S.S.; Ibrahim, A.K. Assessment of serum 25-hydroxyvitamin d levels in children with atopic dermatitis: Correlation with SCORAD index. Dermatitis 2013, 24, 296–301. [Google Scholar] [CrossRef]
- Han, T.Y.; Kong, T.S.; Kim, M.H.; Chae, J.D.; Lee, J.H.; Son, S.J. Vitamin D Status and Its Association with the SCORAD Score and Serum LL-37 Level in Korean Adults and Children with Atopic Dermatitis. Ann. Dermatol. 2015, 27, 10–14. [Google Scholar] [CrossRef]
- Noh, S.; Park, C.O.; Bae, J.M.; Lee, J.; Shin, J.U.; Hong, C.S.; Lee, K.H. Lower vitamin D status is closely correlated with eczema of the head and neck. J. Allergy Clin. Immunol. 2014, 133, 1767–1770. [Google Scholar] [CrossRef] [PubMed]
- Samochocki, Z.; Bogaczewicz, J.; Jeziorkowska, R.; Sysa-Jedrzejowska, A.; Glinska, O.; Karczmarewicz, E.; McCauliffe, D.P.; Wozniacka, A. Vitamin D effects in atopic dermatitis. J. Am. Acad. Dermatol. 2013, 69, 238–244. [Google Scholar] [CrossRef] [PubMed]
- Su, O.; Bahali, A.G.; Demir, A.D.; Ozkaya, D.B.; Uzuner, S.; Dizman, D.; Onsun, N. The relationship between severity of disease and vitamin D levels in children with atopic dermatitis. Adv. Dermatol. Allergol. 2017, 34, 224–227. [Google Scholar] [CrossRef] [PubMed]
- Hata, T.; Audish, D.; Kotol, P.; Coda, A.; Kabigting, F.; Miller, J.; Alexandrescu, D.; Boguniewicz, M.; Taylor, P.; Aertker, L.; et al. A randomized controlled double-blind investigation of the effects of vitamin D dietary supplementation in subjects with atopic dermatitis. J. Eur. Acad. Dermatol. Venereol. 2014, 28, 781–789. [Google Scholar] [CrossRef] [PubMed]
- Udompataikul, M.; Huajai, S.; Chalermchai, T.; Taweechotipatr, M.; Kamanamool, N. The effects of oral vitamin D supplement on atopic dermatitis: A clinical trial with staphylococcus aureus colonization determination. J. Med. Assoc. Thail. 2015, 98, S23–S30. [Google Scholar]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M.; Endocrine, S. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Albenali, L.H.; Danby, S.; Moustafa, M.; Brown, K.; Chittock, J.; Shackley, F.; Cork, M.J. Vitamin D and antimicrobial peptide levels in patients with atopic dermatitis and atopic dermatitis complicated by eczema herpeticum: A pilot study. J. Allergy Clin. Immunol. 2016, 138, 1715–1719. [Google Scholar] [CrossRef]
- Amestejani, M.; Salehi, B.; Vasigh, M.; Sobhkhiz, A.; Karami, M.; Alinia, H.; Kamrava, S.; Shamspour, N.; Ghalehbaghi, B.; Behzadi, A. Vitamin D supplementation in the treatment of atopic dermatitis: A clinical trial study. J. Drugs Dermatol. 2012, 11, 327–330. [Google Scholar]
- Javanbakht, M.; Keshavarz, S.; Djalali, M.; Siassi, F.; Eshraghian, M.; Firooz, A.; Seirafi, H.; Ehsani, A.; Chamari, M.; Mirshafiey, A. Randomized controlled trial using vitamins E and D supplementation in atopic dermatitis. J. Dermatol. Treat. 2011, 22, 144–150. [Google Scholar] [CrossRef]
- Tsotra, K.; Garoufi, A.; Kossiva, L.; Gourgiotis, D.; Tsoukatou, T.; Katsantoni, E.; Stavropoulos, P. The impact of vitamin D supplementation on serum cathelicidin levels and the clinical course of atopic dermatitis in children. Minerva Pediatr. 2017. [Google Scholar] [CrossRef]
- Schram, M.E.; Spuls, P.I.; Leeflang, M.M.; Lindeboom, R.; Bos, J.D.; Schmitt, J. EASI, (objective) SCORAD and POEM for atopic eczema: Responsiveness and minimal clinically important difference. Allergy 2012, 67, 99–106. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Scientific Opinion on the Tolerable Upper Intake of Vitamin D. EFSA J. 2012, 10, 2813. [Google Scholar]
- Vaughn, A.R.; Foolad, N.; Maarouf, M.; Tran, K.A.; Shi, V.Y. Micronutrients in Atopic Dermatitis: A Systematic Review. J. Altern. Complement. Med. 2019, 25, 567–577. [Google Scholar] [CrossRef] [PubMed]
- Gois, P.H.F.; Ferreira, D.; Olenski, S.; Seguro, A.C. Vitamin D and Infectious Diseases: Simple Bystander or Contributing Factor? Nutrients 2017, 9, 651. [Google Scholar] [CrossRef] [PubMed]
- Sharief, S.; Jariwala, S.; Kumar, J.; Muntner, P.; Melamed, M.L. Vitamin D levels and food and environmental allergies in the United States: Results from the National Health and Nutrition Examination Survey 2005–2006. J. Allergy Clin. Immunol. 2011, 127, 1195–1202. [Google Scholar] [CrossRef] [PubMed]
- Yip, K.H.; Kolesnikoff, N.; Yu, C.; Hauschild, N.; Taing, H.; Biggs, L.; Goltzman, D.; Gregory, P.A.; Anderson, P.H.; Samuel, M.S.; et al. Mechanisms of vitamin D(3) metabolite repression of IgE-dependent mast cell activation. J. Allergy Clin. Immunol. 2014, 133, 1356–1364. [Google Scholar] [CrossRef] [PubMed]
- Leaf, D.E.; Croy, H.E.; Abrahams, S.J.; Raed, A.; Waikar, S.S. Cathelicidin antimicrobial protein, vitamin D, and risk of death in critically ill patients. Crit. Care 2015, 19, 80. [Google Scholar] [CrossRef] [PubMed]
- Xie, Z.; Komuves, L.; Yu, Q.C.; Elalieh, H.; Ng, D.C.; Leary, C.; Chang, S.; Crumrine, D.; Yoshizawa, T.; Kato, S.; et al. Lack of the vitamin D receptor is associated with reduced epidermal differentiation and hair follicle growth. J. Investig. Dermatol. 2002, 118, 11–16. [Google Scholar] [CrossRef] [PubMed]
- Mallbris, L.; Carlen, L.; Wei, T.; Heilborn, J.; Nilsson, M.F.; Granath, F.; Stahle, M. Injury downregulates the expression of the human cathelicidin protein hCAP18/LL-37 in atopic dermatitis. Exp. Dermatol. 2010, 19, 442–449. [Google Scholar] [CrossRef]
- Matheson, E.M.; Mainous, A.G., 3rd; Hueston, W.J.; Diaz, V.A.; Everett, C.J. Vitamin D and methicillin-resistant Staphylococcus aureus nasal carriage. Scand. J. Infect. Dis. 2010, 42, 455–460. [Google Scholar] [CrossRef]
- Gilaberte, Y.; Sanmartin, R.; Aspiroz, C.; Hernandez-Martin, A.; Benito, D.; Sanz-Puertolas, P.; Alonso, M.; Torrelo, A.; Torres, C. Correlation Between Serum 25-Hydroxyvitamin D and Virulence Genes of Staphylococcus aureus Isolates Colonizing Children with Atopic Dermatitis. Pediatr. Dermatol. 2015, 32, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.W.; Hogan, P.G.; Hunstad, D.A.; Fritz, S.A. Vitamin D sufficiency and Staphylococcus aureus infection in children. Pediatr. Infect. Dis. J. 2015, 34, 544–545. [Google Scholar] [CrossRef] [PubMed]
- Narla, S.; Silverberg, J.I. Association between atopic dermatitis and serious cutaneous, multiorgan and systemic infections in US adults. Ann. Allergy Asthma Immunol. 2018, 120, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Tripkovic, L.; Wilson, L.R.; Hart, K.; Johnsen, S.; De Lusignan, S.; Smith, C.P.; Bucca, G.; Penson, S.; Chope, G.; Elliott, R.; et al. Daily supplementation with 15 μg vitamin D2 compared with vitamin D3 to increase wintertime 25-hydroxyvitamin D status in healthy South Asian and white European women: A 12-wk randomized, placebo-controlled food-fortification trial. Am. J. Clin. Nutr. 2017, 106, 481–490. [Google Scholar] [CrossRef] [PubMed]
| Case Control Study | Participants | Population-Total N, ‘n-AD’, n-HC | Primary Study Outcome | Review Outcome Serum 25(OH)D Levels Observed | p Value | Secondary Study Outcome |
|---|---|---|---|---|---|---|
| Cheon 2015 (South Korea) [27] | Paediatric OPD, median age 6 years | N: 123, n-AD: 91, n-HC: 32 | Serum 25(OH)D levels significantly lower in AD compared to HC. Lower levels in Moderate and Severe AD compared to Mild AD. | AD = 23 ± 2 ng/mL HC = 36 ± 3 ng/mL | <0.05 | |
| D’Auria 2017 (Italy) [28] | Paediatric OPD. Age 1–14 years, 43% Caucasians, skin phototype II or III according to Fitzpatrick skin type | N: 95, analysis-n-AD: 52, n-HC 43 | Serum 25(OH)D levels statistically significant higher in HC than AD even after adjustment for age, sex and season (p = 0.04) | AD = 19 ng/mL, HC = 25 ng/mL | 0.04 | No association was found between serum 25(OH)D levels and AD severity. |
| El Taieb 2013 (Egypt) [30] | Patients from the OPD Clinic. Age 2–12 years | N: 59, n-AD: 29, n-HC: 30 | Mean Value of Serum Vitamin D in AD is much lower than HC. | AD = 5 ± 2 ng/mL, HC = 29 ± 2 ng/mL | <0.001 | Mean Serum 25(OH)D levels significantly higher in Mild AD (15 ± 4 ng/mL) vs. Moderate AD (6 ± 3 ng/mL) or Severe AD (0.3 ± 0.1 ng/mL). Individual SCORAD values showed significant inverse correlation with serum 25(OH) D Levels, r = −0.88, p = 0.001 |
| Han 2015 [31] | Patients: adult >18 years, child <18. Age: Adults: 26.8 ± 8.25 (18–51), Child 9.5 ± 4.27 (1–16) years | N: 212, Adults n-AD: 39, n-HC: 70, Children n-AD: 33, n-HC: 70 | Serum 25(OH)D level significantly lower in AD children, not statistically different in AD adults. Overall not statistically different between 72 AD patients (12.43 ± 4.66 ng/mL) vs. 140 control (13.49 ± 6.23 ng/mL) (p = 0.05) All adults + 76% children with AD showed deficient levels of Serum 25(OH)D levels. | Child-AD = 15 ± 5 ng/mL, Child-HC = 16 ± 7 ng/mL. Adults-AD = 10 ± 4 ng/mL. Adult-HC = 11 ± 4 ng/mL | Child 0.04 | Difference in serum 25(OH)D levels of different AD severity not statistically different (p > 0.05). Significant inverse correlation between BMI and VitD level in AD (r = −0.32, p = 0.007) and HC (r = −0.335, p = 0.009). Significant inverse correlation between SCORAD and serum LL-37 (r = −0.3, p = 0.01) for total population and only significant in adults after subdividing (r = −0.36, p = 0.025) |
| Noh 2014 (South Korea) [32] | Patients AD-82, Asthma-38 HC-49 | N: 169, n-AD: 82, n-HC: 49. Analysis done with n-AD: 61, n-HC: 34 | AD patients had significantly lower Vitamin D levels compared to Asthmatic pts and healthy Controls (p = 0.01 and p < 0.001). Statistically significant negative correlation between Serum 25(OH)D levels and eczema involvement of the total area (r = −0.376, p = 0.001) | HC = 11 ± 1 ng/mL AD = 10 ± 1 ng/mL | 0.001 | Significant inverse correlation was observed for serum 25(OH)D levels and total body affected by eczema (r = −0.38, p = 0.001) Correlation found between serum 25(OH)D levels and different dermal area manifestations, age, eosinophil count, serum IgE levels. |
| Sharma 2017 (India) [25] | Patients from Dermatological OPD, ages 2–18 years | N: 80, n-AD: 40, n-HC: 40 | Lower Serum 25(OH)D levels in AD was statistically highly significant compared to HC | AD = 30 nmol/L, HC = 54 nmol/L | <0.001 | Significantly inverse correlation between Serum25(OH)D levels and SCORAD index. Mean serum 25(OH)D levels were significantly lower in AD vs. HC with superficial bacterial, fungal, viral infection. Levels were deficient in patients with AD (<20 nmol/L) and insufficient in (20–29 nmol/L) in HC with cutaneous infection. |
| Su 2017 (Turkey) [34] | Dermatology OPD, 2–16 years (mean 8·37); | N: 97, n-AD:60, n-HC: 37 | No statistical significant difference in Mean Serum 25(OH)D levels of AD and HC. | AD = 16 ± 7 ng/mL HC = 20 ± 10 ng/mL | 0.07 | Significant inverse relationship between SCORAD scores and Serum 25(OH)D levels. Serum 25(OH)D levels statistically significantly lower in moderate and severe AD compared with mild AD (p = 0.001, p = 0.004) Vit D showed negative correlation with Serum IgE levels (p = 0.007) |
| Wang 2014 (Hong Kong) [26] | Paediatric OPD (n = 405) Secondary school epidemiological study for obesity and diabetics (n = 95) | N: 826, n-AD: 498, n-HC:328 | serum 25(OH)D levels higher in HC than AD. Inverse association seen between Serum VitD levels and SCORAD, Ness Scores. | AD = 29 ± 15 HC = 34 ± 15 nM | <0.001 | Higher percentage of AD had Serum 25(OH)D <25 nM vs. HC (47.8% vs. 26.6%). Serum 25(OH)D levels showed inverse associations with short-term, long-term AD severity, serum IgE and eosinophil levels. |
| Study | Case Study Type | n-Cases | n-HC | Age: Adult ‘A’ Child ‘C’ | Serum 25(OH)VitD Levels AD Baseline (ng/mL) | Serum 25(OH)VitD Levels AD Baseline (nmol/L) | Serum 25(OH)VitD Levels Healthy Controls (ng/mL) | Serum 25(OH)VitD Levels Healthy Controls (nmol/L) | p Value | Geographical Location by Latitude | VitD Deficient(D) Sufficient(S) Insufficient(I) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Cheon 2015 [27] | Case-Control | 91 | 32 | C | 23 ± 2 | 58 ± 4 | 36 ± 3 | 90 ± 7 | <0.05 | Korea 37.7 | AD-I Controls-S |
| D’Auria 2017 [28] | Case-Control | 52 | 43 | C | 19 ± 7 | 48 ± 18 | 25 ± 13 | 62 ± 31 | 0.04 | Milan, Italy 45.5 | AD-D Controls-I |
| Di Filippo 2015 [29] | Interventional | 39 | 20 | C | 23 ± 8 | 57 ± 20 | 20 ± 3 | 50 ± 8 | Not Specified | Cheiti, Italy 42.4 | AD-I Controls-I |
| El Taieb 2013 [30] | Case-Control | 29 | 30 | C | 5 ± 2 | 14 ± 5 | 29 ± 2 | 72 ± 6 | <0.001 | Egypt 26.8 | AD-D Controls-I |
| Han 2015 [31] | Case-Control | 72 | 140 | A,C | 12 ± 5 | 31 ± 12 | 14 ± 6 | 34 ± 16 | p > 0.05 | Korea 37.7 | AD-D Controls-D |
| Han 2015 Adult [31] | Case-Control | 39 | 70 | A | 10 ± 3 | 26 ± 8 | 11 ± 4 | 27 ± 11 | p > 0.05 | Korea 37.7 | AD-D Controls-D |
| Han 2015 Child [31] | Case-Control | 33 | 70 | C | 15 ± 5 | 38 ± 11·58 | 16 ± 7 | 41 ± 17 | 0.04 | Korea 37.7 | AD-D Controls- D |
| Hata 2014 [35] | Interventional | 30 | 30 | A,C | 28 ± 11 | 71 ± 28 | 30 ± 12 | 75 ± 31 | Not specified | Multicentric 32.7–45.5 | AD-I Controls- S |
| Noh 2014 [32] | Case-Control | 82 | 49 | A,C | 10 ± 0.6 | 24 ± 2 | 11 ± 1 | 28 ± 2 | 0.001 | Korea 37. | AD-D Controls-D |
| Noh 2014—Child [32] | Case-Control | 27 | 12 | C | 11 ± 5 | 27 ± 12 | 13 ± 5 | 31 ± 14 | Retrieved from supplied data | Korea 37.7 | AD-D Controls-D |
| Noh 2014 Adult [32] | Case-Control | 34 | 23 | A | 9 ± 4 | 21 ± 11 | 10 ± 6 | 25 ± 15 | Retrieved from supplied data | Korea 37.7 | AD-D Controls-D |
| Samochocki 2013 [33] | Interventional | 95 | 58 | A | 23 ± 13 | 58 ± 33 | 24 ± 13 | 59 ± 32 | >0.05 | Poland 51.9 | AD-D Controls-D |
| Sharma 2017 [25] | Case-Control | 40 | 40 | C | 12 ± 3 | 30 ± 7 | 21 ± 3 | 54 ± 6 | <0.001 | Punjab, India 31.2 | AD-D Controls-I |
| Su 2017 [34] | Case-Control | 60 | 37 | C | 16 ± 7 | 40 ± 7 | 20 ± 10 | 49 ± 26 | 0.07 | Turkey 39 | AD-D Controls-D |
| Wang 2014 [26] | Case-Control | 498 | 328 | C | 11 ± 6 | 29 ± 15 | 14 ± 6 | 34 ± 15 | <0.001 | Hong Kong 22.4 | AD-D Controls-D |
| Study | Interventional Design Type | Participants | Population-n | Primary Study Outcome | p Value | Secondary Outcome | Serum 25(OH)D Levels Baseline (nmol/L) | VitD Deficient(D) Sufficient(S) Insufficient(I) |
|---|---|---|---|---|---|---|---|---|
| Albenali 2016 (U.K.) [38] | Clinical service evaluation of AD and Eczema Herpeticum (ADEH). VitD supplementation based on deficiency and additionally 6000 IU/daily for age 1–12 years, 10,000 IU/daily for age 12–18 years. Baseline population: 57% with VitD def, 26% suboptimal levels and 83% insufficient VitD level | Age 1–18 years | n = 47, n-AD = 12, n-ADEH = 35 | 25(OH)D level and SCORAD showed significant inverse relationship (p = 0.01) r = −0.36. at baseline. After 2 months VitD supplementation, 42% reduction in SCORAD (p = 0.001) | 0.001 | Serum of AD was significantly correlated with LL-37 levels (r = −0.32 p = 0.01) suggesting causal relationship. LL 37 levels (known to help with re-epithelization) significantly different; reduced in severe AD, post-supplementation. | - | - |
| Amestejani 2012 (Iran) [39] | Randomized double-blind placebo controlled trial, 1600 IU/daily cholecalciferol given for 60 days | Age 14 and older. | Population n = 60 n-VitD = 24 n-placebo = 30 | Significant improvement of SCORAD and TIS (Three Item Severity Score) value under in VitD group of mild, moderate and severe AD (p < 0.005) not seen in placebo group (p > 0.05). | <0.005 | 23 ± 3 | D | |
| Di Filippo 2015 (Italy) [29] | Interventional study—prospective longitudinal 1000 IU/daily 3 months. | Age 4 ± 3.15 year with AD and pre-pubertal Tanner stage 1. | n-AD = 39, final compliers AD = 22, n-HC = 20 | Standard mean value of serum Vit D levels was insufficient and comparable with healthy controls. AD = 23 ± 8 HC = 20 ± 3. Improvement from baseline levels of serum 25(OH)D correlated with reduction in SCORAD index. Significant negative correlation between VitD change and SCORAD change (r = −0.49 p = 0.02) | 0.01 | In AD, high rate of VitD insufficiency (74%) and deficiency (7%). Improvement in VitD levels after 3 months correlated with reduction in SCORAD and reduced inflammatory cytokines. | 57 ± 20 | I |
| Hata 2014 (U.S.A-Multicenter) [35] | Randomized double-blinded placebo controlled trial VitD3: 4000 IU/daily for 21 days | AD interventional = 30 (mean age of 31.2 year); placebo = 30 (mean age 31.9 year) | AD subject with Fitzpatrick Type V/VI skin had significantly lower serum 25(OH)D level 19 ng/mL comparing to type III/IV with a mean 25 OHD of 29 ng/mL (p = 0.04). Increase in serum 25(OH)D from mean of 30 ng/mL to 40 ng/mL. p < 0.01 | 0.05 | AD subject serum 25(OH)D inversely correlated with BMI (r = −0.31. p = 0.04). | 29 (11) | D | |
| Javanbakht 2011 (Iran) [40] | Randomized double-blind placebo controlled trial. 1600 IU/daily for 60 days | Age 13–45, SCORAD 10–70, | n-AD with VitD = 12, n-placebo = 11 | SCORAD: significant reduction of 34.8% in VitD group, placebo group: 28.9%. Change in objective SCORAD: VitD group: 38%, placebo group: 31%. Change in intensity 37% VitD group, 25% in placebo group. | 0.004 | No associate relationship with SCORAD and serum 25(OH)VitD3 level. Strong reduction in usage of topical steroid. | - | - |
| Samochocki 2013 (Poland) [33] | Interventional study from a cross-sectional study not randomised or controlled. Blinding done for supplementation and SCORAD evaluation. 2000 IU/daily 3 months, Jan to March | Age 18–50 years | n-AD:95, n-HC:58 observational, For interventional study n = 20 | Supplementation improved Vit D levels from deficient to insufficient levels, statistically significant decrease in AD severity (p < 0.05). All SCORAD parameters except lichenification significantly decreased. Correlation of improvement in SCORAD and VitD (p = 0.001) | <0.05 | After 3 months supplementation mean total IgE level significantly lower than before (995 ± 1681 vs. 1148 ± 19 IU/mL) p < 0.001. | 19 ± 8 | D |
| Tsotra 2017 (Greece) [41] | Interventional study 2 months, Vitamin D supplementation 1200 IU in mild AD (SCORAD < 40), 2400 IU in severe AD (SCORAD > 40) | Children with AD, Severe and mild (no other information) | n-AD: 50, | Baseline SCORAD differed significantly between mild AD, severe AD group, p < 0.001. Post 20 days supplementation, the difference disappeared (p = 0.65) and statistically insignificant at 45 days (p = 0.61) and 60 days (p = 0.48). Significant “downregulation” of symptom severity in severe AD group on 2400 IU/daily oral VitD supplementation. | 0.001 | Levels of serum cathelicidin child with AD significantly higher than those with children in control group. AD 61 (261–129) p = 0.02, HC 50 (0.2–93.6). No correlation seen with severity of SCORAD and baseline serum cathelicidin and cathelicidin in tissue preparation for atopic skin. | - | - |
| Udompataikul 2015 (Thailand) [36] | Randomized double-blind placebo controlled trial VitD 2000 IU/daily. | 1–18 years old. Mean age 8–28 years | n-Vit D: 12, n-placebo: 12 | Changes of VitD group vs. placebo group at week 4: Statistically significant reduction in SCORAD (p = 0.02), lower S. aureus colony count (p = 0.03). Statistically significant reduction in Erythema index (p = 0.01). Oedema, excoriation, pruritus better. lichenification, dryness and sleeplessness not better. Skin conductance not statistically significantly difference between group at each visit (p = 0.01, 0.65 and 0.55 respectively) Serum VitD levels inversely correlate with SCORAD and S. aureus colonisation with statistically significant difference (r = −1.0, p = < 0.01). Obvious increment of 25(OH)D levels in supplemented group compared to placebo group significant difference 8 mg/mL vs. 0.8 mg/mL (p = 0.01) | 0.02 | Significant reduction documented in S. aureus colonisation in Vitamin D group at week 4 vs. placebo group, p = 0.03 | 43 | D |
| Study | Age: Adult ‘A’ Child ‘C’ | n-Cases | n-Controls | Serum 25(OH)D Levels Baseline(n/) | Serum 25(OH)D Levels after VitD Intervention (n/) | SCORAD before Intervention | SCORAD after Intervention | Percentage Improvement of SCORAD | p Value | Trial Period in Months | VitD Dosage IU. | Percentage Weight of Study as per Forest Plot | Calculation of Weighted Mean Dose per Study (I.U) | VitD Deficient(D) Sufficient(S) Insufficient(I) |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amestejani 2012 [39]. | A, C | 24 | 24 | 23 ± 3 | 55.4 ± 10.8 | 25 ± 4 | 15 ± 3 | 38% | <0.05 | 2 | 1600 | 31 | 496 | D |
| Di Filippo 2015 [29] | C | 22 | 22 | 57 ± 20 | 74 ± 26.8 | 46 ± 16 | 23 ± 15 | 51% | <0.001 | 3 | 1000 | 12.6 | 252 | I |
| Javanbakht 2011 [40] | A,C | 12 | 12 | - | - | 36 ± 4 | 23 ± 3 | 35% | 0.004 | 2 | 1600 | 11.5 | 115 | - |
| Samochocki 2013 [33] | A | 20 | 20 | 19 ± 8 | 33 ± 14 | 45 ± 16 | 26 ± 11 | 43% | 0.001 | 3 | 2000 | 29.3 | 469 | D |
| Udompataikul 2015 [36] | C | 12 | 12 | 43 | 63 | 18·± 9 | 8·± 9 | 56% | 0.02 | 1 | 2000 | 15.6 | 312 | D |
| Weighted Mean Dose * | 100% | 1600 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hattangdi-Haridas, S.R.; Lanham-New, S.A.; Wong, W.H.S.; Ho, M.H.K.; Darling, A.L. Vitamin D Deficiency and Effects of Vitamin D Supplementation on Disease Severity in Patients with Atopic Dermatitis: A Systematic Review and Meta-Analysis in Adults and Children. Nutrients 2019, 11, 1854. https://doi.org/10.3390/nu11081854
Hattangdi-Haridas SR, Lanham-New SA, Wong WHS, Ho MHK, Darling AL. Vitamin D Deficiency and Effects of Vitamin D Supplementation on Disease Severity in Patients with Atopic Dermatitis: A Systematic Review and Meta-Analysis in Adults and Children. Nutrients. 2019; 11(8):1854. https://doi.org/10.3390/nu11081854
Chicago/Turabian StyleHattangdi-Haridas, Sonal R, Susan A Lanham-New, Wilfred Hing Sang Wong, Marco Hok Kung Ho, and Andrea L Darling. 2019. "Vitamin D Deficiency and Effects of Vitamin D Supplementation on Disease Severity in Patients with Atopic Dermatitis: A Systematic Review and Meta-Analysis in Adults and Children" Nutrients 11, no. 8: 1854. https://doi.org/10.3390/nu11081854
APA StyleHattangdi-Haridas, S. R., Lanham-New, S. A., Wong, W. H. S., Ho, M. H. K., & Darling, A. L. (2019). Vitamin D Deficiency and Effects of Vitamin D Supplementation on Disease Severity in Patients with Atopic Dermatitis: A Systematic Review and Meta-Analysis in Adults and Children. Nutrients, 11(8), 1854. https://doi.org/10.3390/nu11081854




