The Role of the Anabolic Properties of Plant- versus Animal-Based Protein Sources in Supporting Muscle Mass Maintenance: A Critical Review
Abstract
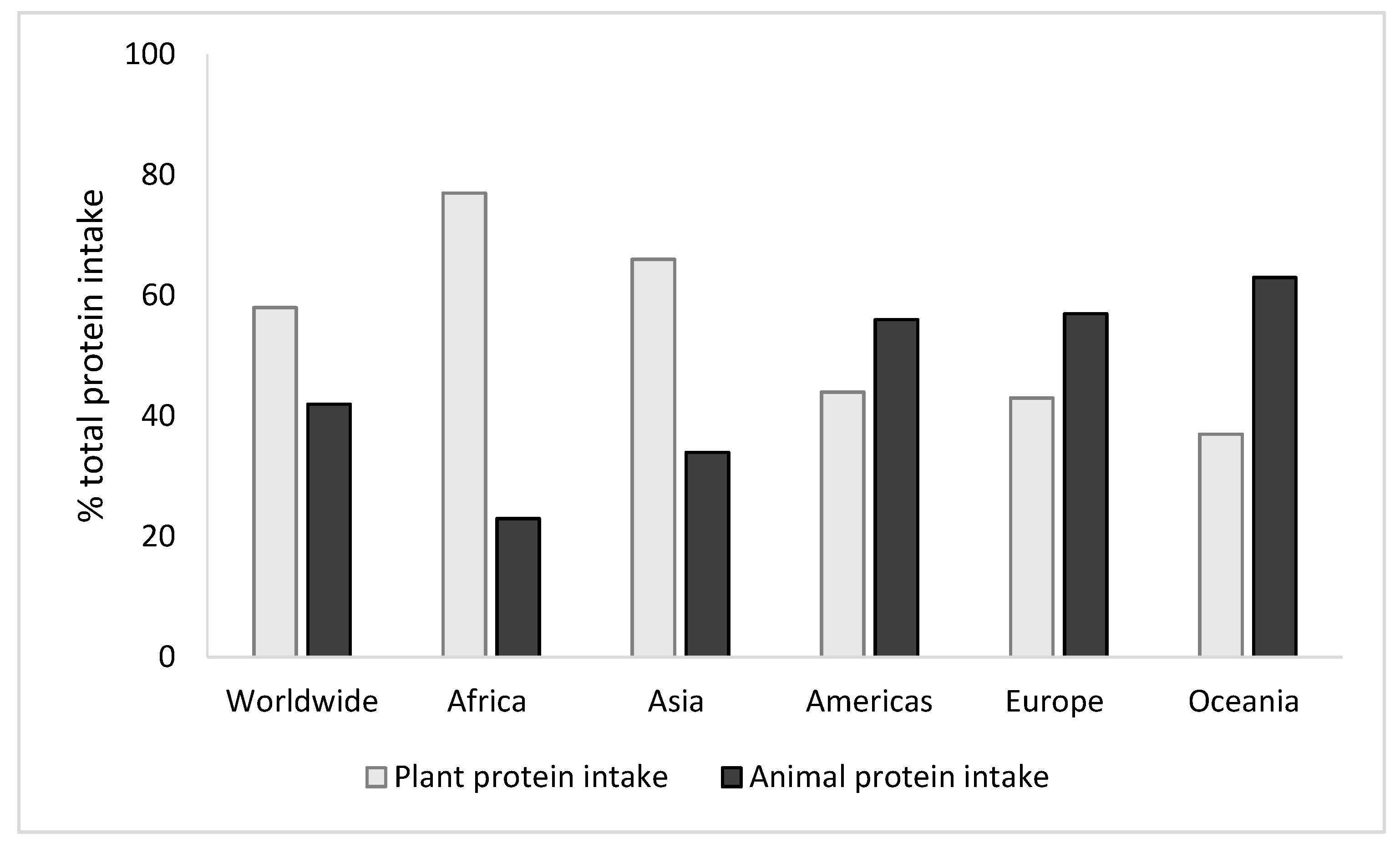
1. Introduction
2. Protein Quality of Plant- Versus Animal-Based Proteins
3. Anabolic Properties of Plant-Based Proteins: Consequences on Muscle Protein Metabolism
3.1. Acute Clinical Studies on Plant- versus Animal-Based Proteins Enrolling Young and Older Subjects
3.2. Chronic Animal Studies on Plant- versus Animal-Based Proteins Enrolling Young Individuals
3.3. Chronic Clinical Studies on Plant- versus Animal-Based Proteins Enrolling Young Subjects
3.4. Chronic Clinical Studies on Plant- versus Animal-Based Diets Enrolling Young and Older Subjects
4. Main Strategies to Improve the Anabolic Properties of Plant-Based Protein Sources
4.1. Increased Protein Intake
4.2. Supplementation with Limiting Amino Acids or Branched-Chain Amino Acids
4.3. Protein Blending
4.3.1. Blending Different Plant-Based Protein Sources
4.3.2. Blending Plant- and Animal-Based Protein Sources
4.4. Using Plant Selections with More Balanced Amino Acid Composition
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Koopman, R.; van Loon, L.J.C. Aging, exercise, and muscle protein metabolism. J. Appl. Physiol. 2009, 106, 2040–2048. [Google Scholar] [CrossRef] [PubMed]
- Koopman, R. Dietary protein and exercise training in ageing. Proc. Nutr. Soc. 2011, 70, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Boirie, Y.; Gachon, P.; Corny, S.; Fauquant, J.; Maubois, J.L.; Beaufrère, B. Acute postprandial changes in leucine metabolism as assessed with an intrinsically labeled milk protein. Am. J. Physiol. 1996, 271, E1083–E1091. [Google Scholar] [CrossRef] [PubMed]
- Volpi, E.; Lucidi, P.; Cruciani, G.; Monacchia, F.; Reboldi, G.; Brunetti, P.; Bolli, G.B.; De Feo, P. Contribution of amino acids and insulin to protein anabolism during meal absorption. Diabetes 1996, 45, 1245–1252. [Google Scholar] [CrossRef] [PubMed]
- Pacy, P.J.; Price, G.M.; Halliday, D.; Quevedo, M.R.; Millward, D.J. Nitrogen homeostasis in man: The diurnal responses of protein synthesis and degradation and amino acid oxidation to diets with increasing protein intakes. Clin. Sci. 1994, 86, 103–116. [Google Scholar] [CrossRef]
- Dardevet, D.; Rémond, D.; Peyron, M.-A.; Papet, I.; Savary-Auzeloux, I.; Mosoni, L. Muscle wasting and resistance of muscle anabolism: The “anabolic threshold concept” for adapted nutritional strategies during sarcopenia. Sci. World J. 2012, 2012, 269–531. [Google Scholar] [CrossRef]
- Walrand, S.; Gryson, C.; Salles, J.; Giraudet, C.; Migné, C.; Bonhomme, C.; Le Ruyet, P.; Boirie, Y. Fast-digestive protein supplement for ten days overcomes muscle anabolic resistance in healthy elderly men. Clin. Nutr. 2016, 35, 660–668. [Google Scholar] [CrossRef]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.M.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Avan Aihie, S.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2018, 0, 1–16. [Google Scholar]
- Volpi, E.; Mittendorfer, B.; Rasmussen, B.B.; Wolfe, R.R. The response of muscle protein anabolism to combined hyperaminoacidemia and glucose-induced hyperinsulinemia is impaired in the elderly. J. Clin. Endocrinol. Metab. 2000, 85, 4481–4490. [Google Scholar] [CrossRef]
- Guillet, C.; Prod’homme, M.; Balage, M.; Gachon, P.; Giraudet, C.; Morin, L.; Grizard, J.; Boirie, Y. Impaired anabolic response of muscle protein synthesis is associated with S6K1 dysregulation in elderly humans. FASEB J. 2004, 18, 1586–1587. [Google Scholar] [CrossRef]
- Guillet, C.; Zangarelli, A.; Gachon, P.; Morio, B.; Giraudet, C.; Rousset, P.; Boirie, Y. Whole body protein breakdown is less inhibited by insulin, but still responsive to amino acid, in nondiabetic elderly subjects. J. Clin. Endocrinol. Metab. 2004, 89, 6017–6024. [Google Scholar] [CrossRef] [PubMed]
- Dardevet, D.; Sornet, C.; Balage, M.; Grizard, J. Stimulation of in vitro rat muscle protein synthesis by leucine decreases with age. J. Nutr. 2000, 130, 2630–2635. [Google Scholar] [CrossRef] [PubMed]
- Dardevet, D.; Sornet, C.; Bayle, G.; Prugnaud, J.; Pouyet, C.; Grizard, J. Postprandial stimulation of muscle protein synthesis in old rats can be restored by a leucine-supplemented meal. J. Nutr. 2002, 132, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Cuthbertson, D.; Smith, K.; Babraj, J.; Leese, G.; Waddell, T.; Atherton, P.; Wackerhage, H.; Taylor, P.M.; Rennie, M.J. Anabolic signaling deficits underlie amino acid resistance of wasting, aging muscle. FASEB J. 2005, 19, 422–424. [Google Scholar] [CrossRef] [PubMed]
- Katsanos, C.S.; Kobayashi, H.; Sheffield-Moore, M.; Aarsland, A.; Wolfe, R.R. Aging is associated with diminished accretion of muscle proteins after the ingestion of a small bolus of essential amino acids. Am. J. Clin. Nutr. 2005, 82, 1065–1073. [Google Scholar] [CrossRef] [PubMed]
- Rieu, I.; Balage, M.; Sornet, C.; Debras, E.; Ripes, S.; Rochon-Bonhomme, C.; Pouyet, C.; Grizard, J.; Dardevet, D. Increased availability of leucine with leucine-rich whey proteins improves postprandial muscle protein synthesis in aging rats. Nutrition 2007, 23, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Gryson, C.; Walrand, S.; Giraudet, C.; Rousset, P.; Migné, C.; Bonhomme, C.; Le Ruyet, P.; Boirie, Y. “Fast proteins” with a unique essential amino acid content as an optimal nutrition in the elderly: Growing evidence. Clin. Nutr. 2014, 33, 642–648. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.R.; Churchward-Venne, T.A.; Witard, O.; Breen, L.; Burd, N.A.; Tipton, K.D.; Phillips, S.M. Protein ingestion to stimulate myofibrillar protein synthesis requires greater relative protein intakes in healthy older versus younger men. J. Gerontol. A Biol. Sci. Med. Sci. 2015, 70, 57–62. [Google Scholar] [CrossRef]
- Martínez, J.A.; Larralde, J. Influence of diets containing different levels of Vicia faba L. as source of protein on body Protein composition and nitrogen balance of growing rats. Ann. Nutr. Metab. 1984, 28, 174–180. [Google Scholar]
- Martínez, J.A.; Larralde, J. Muscle protein turnover in rats fed on diets containing different levels of Vicia faba L. and casein as source of protein. Rev. Esp. Fisiol. 1984, 40, 109–115. [Google Scholar]
- Martínez, J.A.; Larralde, J. Developmental changes on protein turnover in growing rats fed on diets containing field beans (Vicia faba L.) as source of protein. Arch. Latinoam. Nutr. 1984, 34, 466–476. [Google Scholar]
- Martínez, J.A.; Barcina, Y.; Larralde, J. Induced biochemical and physiological changes in young and adult growing rats fed on a vegetable or animal protein diet. Growth 1986, 50, 178–184. [Google Scholar]
- Combe, E.; Pirman, T.; Stekar, J.; Houlier, M.-L.; Mirand, P.P. Differential effect of lentil feeding on proteosynthesis rates in the large intestine, liver and muscle of rats. J. Nutr. Biochem. 2004, 15, 12–17. [Google Scholar] [CrossRef]
- Pirman, T.; Combe, E.; Ribeyre, M.C.; Prugnaud, J.; Stekar, J.; Patureau Mirand, P. Differential effects of cooked beans and cooked lentils on protein metabolism in intestine and muscle in growing rats. Ann. Nutr. Metab. 2006, 50, 197–205. [Google Scholar] [CrossRef]
- Pirman, T.; Stekar, J.M.A.; Combe, E.; Orešnik, A. Nutritional value of beans and lentils in rats. Krmiva 2001, 43, 133–143. [Google Scholar]
- Bos, C.; Metges, C.C.; Gaudichon, C.; Petzke, K.J.; Pueyo, M.E.; Morens, C.; Everwand, J.; Benamouzig, R.; Tomé, D. Postprandial kinetics of dietary amino acids are the main determinant of their metabolism after soy or milk protein ingestion in humans. J. Nutr. 2003, 133, 1308–1315. [Google Scholar] [CrossRef]
- Alonso, R.; Grant, G.; Frühbeck, G.; Marzo, F. Muscle and liver protein metabolism in rats fed raw or heat-treated pea seeds. J. Nutr. Biochem. 2002, 13, 611–618. [Google Scholar] [CrossRef]
- Hartman, J.W.; Tang, J.E.; Wilkinson, S.B.; Tarnopolsky, M.A.; Lawrence, R.L.; Fullerton, A.V.; Phillips, S.M. Consumption of fat-free fluid milk after resistance exercise promotes greater lean mass accretion than does consumption of soy or carbohydrate in young, novice, male weightlifters. Am. J. Clin. Nutr. 2007, 86, 373–381. [Google Scholar] [CrossRef]
- Norton, L.E.; Wilson, G.J.; Layman, D.K.; Moulton, C.J.; Garlick, P.J. Leucine content of dietary proteins is a determinant of postprandial skeletal muscle protein synthesis in adult rats. Nutr. Metab. 2012, 9, 67. [Google Scholar] [CrossRef]
- Kanda, A.; Nakayama, K.; Sanbongi, C.; Nagata, M.; Ikegami, S.; Itoh, H. Effects of whey, caseinate, or milk protein ingestion on muscle protein synthesis after exercise. Nutrients 2016, 8, 339. [Google Scholar] [CrossRef]
- Wróblewska, B.; Juśkiewicz, J.; Kroplewski, B.; Jurgoński, A.; Wasilewska, E.; Złotkowska, D.; Markiewicz, L. The effects of whey and soy proteins on growth performance, gastrointestinal digestion, and selected physiological responses in rats. Food Funct. 2018, 9, 1500–1509. [Google Scholar] [CrossRef]
- Martinez, J.; Marcos, R.; Macarulla, M.; Larralde, J. Growth, hormonal status and protein-turnover in rats fed on a diet containing peas (Pisum-Sativum L.) as the source of protein. Plant Foods Hum. Nutr. 1995, 47, 211–220. [Google Scholar] [CrossRef]
- Luiking, Y.C.; Deutz, N.E.P.; Jäkel, M.; Soeters, P.B. Casein and soy protein meals differentially affect whole-body and splanchnic protein metabolism in healthy humans. J. Nutr. 2005, 135, 1080–1087. [Google Scholar] [CrossRef]
- Laleg, K.; Salles, J.; Berry, A.; Giraudet, C.; Patrac, V.; Guillet, C.; Denis, P.; Tessier, F.J.; Guilbaud, A.; Howsam, M.; et al. Nutritional evaluation of mixed wheat-faba bean pasta in growing rats: Impact of protein source and drying temperature on protein digestibility and retention. Br. J. Nutr. 2019, 121, 496–507. [Google Scholar] [CrossRef]
- Giménez, M.A.; Drago, S.R.; Bassett, M.N.; Lobo, M.O.; Sammán, N.C. Nutritional improvement of corn pasta-like product with broad bean (Vicia faba) and quinoa (Chenopodium quinoa). Food Chem. 2016, 199, 150–156. [Google Scholar] [CrossRef]
- Phillips, S.M. Nutrient-rich meat proteins in offsetting age-related muscle loss. Meat Sci. 2012, 92, 174–178. [Google Scholar] [CrossRef]
- Volek, J.S.; Volk, B.M.; Gómez, A.L.; Kunces, L.J.; Kupchak, B.R.; Freidenreich, D.J.; Aristizabal, J.C.; Saenz, C.; Dunn-Lewis, C.; Ballard, K.D.; et al. Whey protein supplementation during resistance training augments lean body mass. J. Am. Coll. Nutr. 2013, 32, 122–135. [Google Scholar] [CrossRef]
- Mobley, C.B.; Haun, C.T.; Roberson, P.A.; Mumford, P.W.; Romero, M.A.; Kephart, W.C.; Anderson, R.G.; Vann, C.G.; Osburn, S.C.; Pledge, C.D.; et al. Effects of whey, soy or leucine supplementation with 12 weeks of resistance training on strength, body composition, and skeletal muscle and adipose tissue histological attributes in college-aged males. Nutrients 2017, 9, 972. [Google Scholar] [CrossRef]
- Banaszek, A.; Townsend, J.R.; Bender, D.; Vantrease, W.C.; Marshall, A.C.; Johnson, K.D. The effects of whey vs. pea protein on physical adaptations following 8-weeks of high-intensity functional training (HIFT): A pilot study. Sports 2019, 7, 12. [Google Scholar] [CrossRef]
- Wilkinson, S.B.; Tarnopolsky, M.A.; Macdonald, M.J.; Macdonald, J.R.; Armstrong, D.; Phillips, S.M. Consumption of fluid skim milk promotes greater muscle protein accretion after resistance exercise than does consumption of an isonitrogenous and isoenergetic soy-protein beverage. Am. J. Clin. Nutr. 2007, 85, 1031–1040. [Google Scholar] [CrossRef]
- Pannemans, D.L.; Wagenmakers, A.J.; Westerterp, K.R.; Schaafsma, G.; Halliday, D. Effect of protein source and quantity on protein metabolism in elderly women. Am. J. Clin. Nutr. 1998, 68, 1228–1235. [Google Scholar] [CrossRef]
- Yang, Y.; Churchward-Venne, T.A.; Burd, N.A.; Breen, L.; Tarnopolsky, M.A.; Phillips, S.M. Myofibrillar protein synthesis following ingestion of soy protein isolate at rest and after resistance exercise in elderly men. Nutr. Metab. 2012, 9, 57. [Google Scholar] [CrossRef]
- Mitchell, C.J.; Della Gatta, P.A.; Petersen, A.C.; Cameron-Smith, D.; Markworth, J.F. Soy protein ingestion results in less prolonged p70S6 kinase phosphorylation compared to whey protein after resistance exercise in older men. J. Int. Soc. Sports Nutr. 2015, 12, 6. [Google Scholar] [CrossRef]
- Gorissen, S.H.; Horstman, A.M.; Franssen, R.; Crombag, J.J.; Langer, H.; Bierau, J.; Respondek, F.; van Loon, L.J. Ingestion of wheat protein increases in vivo muscle protein synthesis rates in healthy older men in a randomized trial. J. Nutr. 2016, 146, 1651–1659. [Google Scholar] [CrossRef]
- FAOSTAT Food Balance Sheets; FAO UN Statistics Division; FAOSTAT: Rome, Italy, 2009.
- Lynch, H.; Johnston, C.; Wharton, C. Plant-based diets: Considerations for environmental impact, protein quality, and exercise performance. Nutrients 2018, 10, 1841. [Google Scholar] [CrossRef]
- Moore, D.R.; Soeters, P.B. The Biological Value of Protein. Nestle Nutr. Inst. Workshop Ser. 2015, 82, 39–51. [Google Scholar]
- Mariotti, F.; Mahé, S.; Benamouzig, R.; Luengo, C.; Daré, S.; Gaudichon, C.; Tomé, D. Nutritional value of [15N]-soy protein isolate assessed from ileal digestibility and postprandial protein utilization in humans. J. Nutr. 1999, 129, 1992–1997. [Google Scholar] [CrossRef]
- FAO/WHO. Protein Quality Evaluation: Report of the Joint FAO/WHO Expert Consultation 1989; FAO Food and Nutrition Paper 51; FAO: Rome, Italy, 1991. [Google Scholar]
- FAO. Dietary Protein Evaluation in Human Nutrition: Report of an FAO Expert Consultation 2011; FAO Food and Nutrition Paper 92; FAO: Rome, Italy, 2013. [Google Scholar]
- Boye, J.; Zare, F.; Pletch, A. Pulse proteins: Processing, characterization, functional properties and applications in food and feed. Food Res. Int. 2010, 43, 414–431. [Google Scholar] [CrossRef]
- Hoffman, J.R.; Falvo, M.J. Protein-which is best? J. Sports Sci. Med. 2004, 3, 118–130. [Google Scholar]
- Van Vliet, S.; Burd, N.A.; van Loon, L.J.C. The skeletal muscle anabolic response to plant- versus animal-based protein consumption. J. Nutr. 2015, 145, 1981–1991. [Google Scholar] [CrossRef]
- Sarwar, G.; Peace, R.W.; Botting, H.G.; Brulé, D. Digestibility of protein and amino acids in selected foods as determined by a rat balance method. Plant Foods Hum. Nutr. 1989, 39, 23–32. [Google Scholar] [CrossRef]
- Marinangeli, C.P.F.; House, J.D. Potential impact of the digestible indispensable amino acid score as a measure of protein quality on dietary regulations and health. Nutr. Rev. 2017, 75, 658–667. [Google Scholar] [CrossRef]
- Mathai, J.K.; Liu, Y.; Stein, H.H. Values for digestible indispensable amino acid scores (DIAAS) for some dairy and plant proteins may better describe protein quality than values calculated using the concept for protein digestibility-corrected amino acid scores (PDCAAS). Br. J. Nutr. 2017, 117, 490–499. [Google Scholar] [CrossRef]
- ANSES. Apport en Protéines: Consommation, Qualité, Besoins et Recommandations; Agence Nationale de Sécurité Sanitaire des Aliments, de L’environnement et du Travail: Paris, France, 2007. [Google Scholar]
- Rutherfurd, S.M.; Fanning, A.C.; Miller, B.J.; Moughan, P.J. Protein digestibility-corrected amino acid scores and digestible indispensable amino acid scores differentially describe protein quality in growing male rats. J. Nutr. 2015, 145, 372–379. [Google Scholar] [CrossRef]
- Sarwar, G. The protein digestibility-corrected amino acid score method overestimates quality of proteins containing antinutritional factors and of poorly digestible proteins supplemented with limiting amino acids in rats. J. Nutr. 1997, 127, 758–764. [Google Scholar] [CrossRef]
- Laleg, K.; Cassan, D.; Barron, C.; Prabhasankar, P.; Micard, V. Structural, culinary, nutritional and anti-nutritional properties of high protein, gluten free, 100% legume pasta. PLoS ONE 2016, 11, e0160721. [Google Scholar] [CrossRef]
- Gorissen, S.H.M.; Witard, O.C. Characterising the muscle anabolic potential of dairy, meat and plant-based protein sources in older adults. Proc. Nutr. Soc. 2018, 77, 20–31. [Google Scholar] [CrossRef]
- WHO/FAO/UNU. Protein and Amino Acid Requirements in Human Nutrition. Report of the Joint FAO/WHO/UNU Expert Consultation; World Health Organization Technical Report Series 935; WHO: Geneva, Switzerland, 2007. [Google Scholar]
- Lorieau, L.; Halabi, A.; Ligneul, A.; Hazart, E.; Dupont, D.; Floury, J. Impact of the dairy product structure and protein nature on the proteolysis and amino acid bioaccessiblity during in vitro digestion. Food Hydrocoll. 2018, 82, 399–411. [Google Scholar] [CrossRef]
- Carbonaro, M.; Maselli, P.; Nucara, A. Relationship between digestibility and secondary structure of raw and thermally treated legume proteins: A Fourier transform infrared (FT-IR) spectroscopic study. Amino Acids 2012, 43, 911–921. [Google Scholar] [CrossRef]
- Nguyen, T.T.P.; Bhandari, B.; Cichero, J.; Prakash, S. Gastrointestinal digestion of dairy and soy proteins in infant formulas: An in vitro study. Food Res. Int. 2015, 76, 348–358. [Google Scholar] [CrossRef]
- Duodu, K.G.; Taylor, J.R.N.; Belton, P.S.; Hamaker, B.R. Factors affecting sorghum protein digestibility. J. Cereal Sci. 2003, 38, 117–131. [Google Scholar] [CrossRef]
- Gilani, G.S.; Cockell, K.A.; Sepehr, E. Effects of antinutritional factors on protein digestibility and amino acid availability in foods. J. AOAC Int. 2005, 88, 967–987. [Google Scholar]
- Multari, S.; Stewart, D.; Russell, W. Potential of fava bean as future protein supply to partially replace meat intake in the human diet. Compr. Rev. Food Sci. Saf. 2015, 14, 511–522. [Google Scholar] [CrossRef]
- Cheryan, M. Phytic acid interactions in food systems. Crit. Rev. Food Sci. Nutr. 1980, 13, 297–335. [Google Scholar] [CrossRef]
- Chouchene, A.; Micard, V.; Lullien-Pellerin, V. Evidence of a synergistic effect between pea seed and wheat grain endogenous phytase activities. J. Agric. Food Chem. 2018, 66, 12034–12041. [Google Scholar] [CrossRef]
- Boirie, Y.; Dangin, M.; Gachon, P.; Vasson, M.P.; Maubois, J.L.; Beaufrère, B. Slow and fast dietary proteins differently modulate postprandial protein accretion. Proc. Natl. Acad. Sci. USA 1997, 94, 14930–14935. [Google Scholar] [CrossRef]
- Dangin, M.; Boirie, Y.; Garcia-Rodenas, C.; Gachon, P.; Fauquant, J.; Callier, P.; Ballèvre, O.; Beaufrère, B. The digestion rate of protein is an independent regulating factor of postprandial protein retention. Am. J. Physiol. Endocrinol. Metab. 2001, 280, E340–E348. [Google Scholar] [CrossRef]
- Dangin, M.; Boirie, Y.; Guillet, C.; Beaufrère, B. Influence of the protein digestion rate on protein turnover in young and elderly subjects. J. Nutr. 2002, 132, 3228S–3233S. [Google Scholar] [CrossRef]
- Dangin, M.; Guillet, C.; Garcia-Rodenas, C.; Gachon, P.; Bouteloup-Demange, C.; Reiffers-Magnani, K.; Fauquant, J.; Ballèvre, O.; Beaufrère, B. The rate of protein digestion affects protein gain differently during aging in humans. J. Physiol. 2003, 549, 635–644. [Google Scholar] [CrossRef]
- Tang, J.E.; Moore, D.R.; Kujbida, G.W.; Tarnopolsky, M.A.; Phillips, S.M. Ingestion of whey hydrolysate, casein, or soy protein isolate: Effects on mixed muscle protein synthesis at rest and following resistance exercise in young men. J. Appl. Physiol. 2009, 107, 987–992. [Google Scholar] [CrossRef]
- Anthony, J.C.; Anthony, T.G.; Kimball, S.R.; Jefferson, L.S. Signaling pathways involved in translational control of protein synthesis in skeletal muscle by leucine. J. Nutr. 2001, 131, 856S–860S. [Google Scholar] [CrossRef]
- Suryawan, A.; Orellana, R.A.; Fiorotto, M.L.; Davis, T.A. Leucine acts as a nutrient signal to stimulate protein synthesis in neonatal pigs. J. Anim. Sci. 2011, 89, 2004–2016. [Google Scholar] [CrossRef]
- Gorissen, S.H.M.; Crombag, J.J.R.; Senden, J.M.G.; Waterval, W.A.H.; Bierau, J.; Verdijk, L.B.; van Loon, L.J.C. Protein content and amino acid composition of commercially available plant-based protein isolates. Amino Acids 2018, 50, 1685–1695. [Google Scholar] [CrossRef]
- Tujioka, K.; Ohsumi, M.; Hayase, K.; Yokogoshi, H. Effect of the quality of dietary amino acids composition on the urea synthesis in rats. J. Nutr. Sci. Vitaminol. 2011, 57, 48–55. [Google Scholar] [CrossRef]
- Gaudichon, C.; Mahé, S.; Benamouzig, R.; Luengo, C.; Fouillet, H.; Daré, S.; Van Oycke, M.; Ferrière, F.; Rautureau, J.; Tomé, D. Net postprandial utilization of [15N]-labeled milk protein nitrogen is influenced by diet composition in humans. J. Nutr. 1999, 129, 890–895. [Google Scholar] [CrossRef]
- Bos, C.; Juillet, B.; Fouillet, H.; Turlan, L.; Daré, S.; Luengo, C.; N’tounda, R.; Benamouzig, R.; Gausserès, N.; Tomé, D.; et al. Postprandial metabolic utilization of wheat protein in humans. Am. J. Clin. Nutr. 2005, 81, 87–94. [Google Scholar] [CrossRef]
- FAO. Research Approaches and Methods for Evaluating the Protein Quality of Human Foods: Report of a FAO Expert Working Group 2 – 5 March 2014 Bangalore, India; FAO: Rome, Italy, 2014. [Google Scholar]
- Tomé, D.; Bos, C. Dietary protein and nitrogen utilization. J. Nutr. 2000, 130, 1868S–1873S. [Google Scholar] [CrossRef]
- Löhrke, B.; Saggau, E.; Schadereit, R.; Beyer, M.; Bellmann, O.; Kuhla, S.; Hagemeister, H. Activation of skeletal muscle protein breakdown following consumption of soyabean protein in pigs. Br. J. Nutr. 2001, 85, 447–457. [Google Scholar] [CrossRef]
- Millward, D.J.; Fereday, A.; Gibson, N.R.; Cox, M.C.; Pacy, P.J. Efficiency of utilization of wheat and milk protein in healthy adults and apparent lysine requirements determined by a single-meal [1-13C]leucine balance protocol. Am. J. Clin. Nutr. 2002, 76, 1326–1334. [Google Scholar] [CrossRef]
- Fouillet, H.; Bos, C.; Gaudichon, C.; Tomé, D. Approaches to quantifying protein metabolism in response to nutrient ingestion. J. Nutr. 2002, 132, 3208S–3218S. [Google Scholar] [CrossRef]
- Aubertin-Leheudre, M.; Adlercreutz, H. Relationship between animal protein intake and muscle mass index in healthy women. Br. J. Nutr. 2009, 102, 1803–1810. [Google Scholar] [CrossRef]
- Sahni, S.; Mangano, K.M.; Hannan, M.T.; Kiel, D.P.; McLean, R.R. Higher protein intake is associated with higher lean mass and quadriceps muscle strength in adult men and women. J. Nutr. 2015, 145, 1569–1575. [Google Scholar] [CrossRef] [PubMed]
- Mangano, K.M.; Sahni, S.; Kiel, D.P.; Tucker, K.L.; Dufour, A.B.; Hannan, M.T. Dietary protein is associated with musculoskeletal health independently of dietary pattern: The Framingham Third Generation study. Am. J. Clin. Nutr. 2017, 105, 714–722. [Google Scholar] [CrossRef] [PubMed]
- Campbell, W.W.; Barton, M.L.; Cyr-Campbell, D.; Davey, S.L.; Beard, J.L.; Parise, G.; Evans, W.J. Effects of an omnivorous diet compared with a lactoovovegetarian diet on resistance-training-induced changes in body composition and skeletal muscle in older men. Am. J. Clin. Nutr. 1999, 70, 1032–1039. [Google Scholar] [CrossRef] [PubMed]
- Haub, M.D.; Wells, A.M.; Tarnopolsky, M.A.; Campbell, W.W. Effect of protein source on resistive-training-induced changes in body composition and muscle size in older men. Am. J. Clin. Nutr. 2002, 76, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Lord, C.; Chaput, J.P.; Aubertin-Leheudre, M.; Labonté, M.; Dionne, I.J. Dietary animal protein intake: Association with muscle mass index in older women. J. Nutr. Health Aging 2007, 11, 383–387. [Google Scholar] [PubMed]
- Isanejad, M.; Mursu, J.; Sirola, J.; Kröger, H.; Rikkonen, T.; Tuppurainen, M.; Erkkilä, A.T. Association of protein intake with the change of lean mass among elderly women: The Osteoporosis Risk Factor and Prevention—Fracture Prevention Study (OSTPRE-FPS). J. Nutr. Sci. 2015, 4, e41. [Google Scholar] [CrossRef] [PubMed]
- Chan, R.; Leung, J.; Woo, J.; Kwok, T. Associations of dietary protein intake on subsequent decline in muscle mass and physical functions over four years in ambulant older Chinese people. J. Nutr. Health Aging 2014, 18, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.-Y.; Yang, K.-C.; Chang, H.-H.; Lee, L.-T.; Lu, C.-W.; Huang, K.-C. The association between total protein and vegetable protein intake and low muscle mass among the community-dwelling elderly population in Northern Taiwan. Nutrients 2016, 8, 373. [Google Scholar] [CrossRef]
- Miki, A.; Hashimoto, Y.; Matsumoto, S.; Ushigome, E.; Fukuda, T.; Sennmaru, T.; Tanaka, M.; Yamazaki, M.; Fukui, M. Protein intake, especially vegetable protein intake, is associated with higher skeletal muscle mass in elderly patients with type 2 diabetes. J. Diabetes Res. 2017, 2017, 7985728. [Google Scholar] [CrossRef]
- Verreijen, A.M.; Engberink, M.F.; Houston, D.K.; Brouwer, I.A.; Cawthon, P.M.; Newman, A.B.; Tylavsky, F.A.; Harris, T.B.; Weijs, P.J.M.; Visser, M. Dietary protein intake is not associated with 5-y change in mid-thigh muscle cross-sectional area by computed tomography in older adults: The Health, Aging, and Body Composition (Health ABC) Study. Am. J. Clin. Nutr. 2019, 109, 535–543. [Google Scholar] [CrossRef] [PubMed]
- Babault, N.; Païzis, C.; Deley, G.; Guérin-Deremaux, L.; Saniez, M.-H.; Lefranc-Millot, C.; Allaert, F.A. Pea proteins oral supplementation promotes muscle thickness gains during resistance training: A double-blind, randomized, Placebo-controlled clinical trial vs. Whey protein. J. Int. Soc. Sports Nutr. 2015, 12, 3. [Google Scholar] [CrossRef] [PubMed]
- Brown, E.C.; DiSilvestro, R.A.; Babaknia, A.; Devor, S.T. Soy versus whey protein bars: Effects on exercise training impact on lean body mass and antioxidant status. Nutr. J. 2004, 3, 22. [Google Scholar] [CrossRef] [PubMed]
- Joy, J.M.; Lowery, R.P.; Wilson, J.M.; Purpura, M.; De Souza, E.O.; Wilson, S.M.; Kalman, D.S.; Dudeck, J.E.; Jäger, R. The effects of 8 weeks of whey or rice protein supplementation on body composition and exercise performance. Nutr. J. 2013, 12, 86. [Google Scholar] [CrossRef] [PubMed]
- Bauer, J.; Biolo, G.; Cederholm, T.; Cesari, M.; Cruz-Jentoft, A.J.; Morley, J.E.; Phillips, S.; Sieber, C.; Stehle, P.; Teta, D.; et al. Evidence-based recommendations for optimal dietary protein intake in older people: A position paper from the PROT-AGE Study Group. J. Am. Med. Dir. Assoc. 2013, 14, 542–559. [Google Scholar] [CrossRef] [PubMed]
- Traylor, D.A.; Gorissen, S.H.M.; Phillips, S.M. Perspective: Protein requirements and optimal intakes in aging: Are we ready to recommend more than the recommended daily allowance? Adv. Nutr. 2018, 9, 171–182. [Google Scholar] [CrossRef] [PubMed]
- Gilani, G.S.; Sepehr, E. Protein digestibility and quality in products containing antinutritional factors are adversely affected by old age in rats. J. Nutr. 2003, 133, 220–225. [Google Scholar] [CrossRef]
- Norton, L.E.; Layman, D.K.; Bunpo, P.; Anthony, T.G.; Brana, D.V.; Garlick, P.J. The leucine content of a complete meal directs peak activation but not duration of skeletal muscle protein synthesis and mammalian target of rapamycin signaling in rats. J. Nutr. 2009, 139, 1103–1109. [Google Scholar] [CrossRef] [PubMed]
- Engelen, M.P.K.J.; Rutten, E.P.A.; De Castro, C.L.N.; Wouters, E.F.M.; Schols, A.M.W.J.; Deutz, N.E.P. Supplementation of soy protein with branched-chain amino acids alters protein metabolism in healthy elderly and even more in patients with chronic obstructive pulmonary disease. Am. J. Clin. Nutr. 2007, 85, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Zhai, F.; Zhang, D.; An, Y.; Liu, Y.; He, Y.; Ge, K.; Scrimshaw, N.S. Lysine-fortified wheat flour improves the nutritional and immunological status of wheat-eating families in northern China. Food Nutr. Bull. 2004, 25, 123–129. [Google Scholar] [CrossRef]
- Hussain, T.; Abbas, S.; Khan, M.A.; Scrimshaw, N.S. Lysine fortification of wheat flour improves selected indices of the nutritional status of predominantly cereal-eating families in Pakistan. Food Nutr. Bull. 2004, 25, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Duranti, M. Grain legume proteins and nutraceutical properties. Fitoterapia 2006, 77, 67–82. [Google Scholar] [CrossRef] [PubMed]
- Casagrandi, D.A.; Canniatti-Brazaca, S.G.; Salgado, J.M.; Pizzinato, A.; Novaes, N.J. Technological, nutritional and sensorial analysis of macaroni elaborated with wheat and pigeon pea flours. Rev. Nutr. 1999, 12, 137–143. [Google Scholar] [CrossRef]
- Torres, A.; Frias, J.; Granito, M.; Guerra, M.; Vidal-Valverde, C. Chemical, biological and sensory evaluation of pasta products supplemented with α-galactoside-free lupin flours. J. Sci. Food Agric. 2007, 87, 74–81. [Google Scholar] [CrossRef]
- Torres, A.; Frias, J.; Granito, M.; Vidal-Valverde, C. Germinated Cajanus cajan seeds as ingredients in pasta products: Chemical, biological and sensory evaluation. Food Chem. 2007, 101, 202–211. [Google Scholar] [CrossRef]
- Torres, A.; Frias, J.; Granito, M.; Vidal-Valverde, C. Fermented pigeon pea (Cajanus cajan) ingredients in pasta products. J. Agric. Food Chem. 2006, 54, 6685–6691. [Google Scholar] [CrossRef] [PubMed]
- Márquez-Mota, C.C.; Rodriguez-Gaytan, C.; Adjibade, P.; Mazroui, R.; Gálvez, A.; Granados, O.; Tovar, A.R.; Torres, N. The mTORC1-signaling pathway and hepatic polyribosome profile are enhanced after the recovery of a protein restricted diet by a combination of soy or black bean with corn protein. Nutrients 2016, 8, 573. [Google Scholar] [CrossRef] [PubMed]
- Acevedo-Pacheco, L.; Serna-Saldívar, S.O. In vivo protein quality of selected cereal-based staple foods enriched with soybean proteins. Food Nutr. Res. 2016, 60, 31382. [Google Scholar] [CrossRef]
- FAO/WHO/UNU. Energy and Protein Requirements: Report of a joint FAO/WHO/UNU Expert Consultation; WHO Tech Rep Ser No.724; WHO: Geneva, Switzerland, 1985. [Google Scholar]
- Reidy, P.T.; Walker, D.K.; Dickinson, J.M.; Gundermann, D.M.; Drummond, M.J.; Timmerman, K.L.; Cope, M.B.; Mukherjea, R.; Jennings, K.; Volpi, E.; et al. Soy-dairy protein blend and whey protein ingestion after resistance exercise increases amino acid transport and transporter expression in human skeletal muscle. J. Appl. Physiol. 2014, 116, 1353–1364. [Google Scholar] [CrossRef]
- Reidy, P.T.; Borack, M.S.; Markofski, M.M.; Dickinson, J.M.; Deer, R.R.; Husaini, S.H.; Walker, D.K.; Igbinigie, S.; Robertson, S.M.; Cope, M.B.; et al. Protein supplementation has minimal effects on muscle adaptations during resistance exercise training in young men: A double-blind randomized clinical trial. J. Nutr. 2016, 146, 1660–1669. [Google Scholar] [CrossRef]
- Reidy, P.T.; Walker, D.K.; Dickinson, J.M.; Gundermann, D.M.; Drummond, M.J.; Timmerman, K.L.; Fry, C.S.; Borack, M.S.; Cope, M.B.; Mukherjea, R.; et al. Protein blend ingestion following resistance exercise promotes human muscle protein synthesis. J. Nutr. 2013, 143, 410–416. [Google Scholar] [CrossRef] [PubMed]
- Borack, M.S.; Reidy, P.T.; Husaini, S.H.; Markofski, M.M.; Deer, R.R.; Richison, A.B.; Lambert, B.S.; Cope, M.B.; Mukherjea, R.; Jennings, K.; et al. Soy-dairy protein blend or whey protein isolate ingestion induces similar postexercise muscle mechanistic target of rapamycin complex 1 signaling and protein synthesis responses in older men. J. Nutr. 2016, 146, 2468–2475. [Google Scholar] [CrossRef] [PubMed]
- Berrazaga, I.; Mession, J.L.; Laleg, K.; Salles, J.; Guillet, C.; Patrac, V.; Giraudet, C.; Le Bacquer, O.; Boirie, Y.; Micard, V.; et al. Formulation, process conditions, and biological evaluation of dairy mixed gels containing fava bean and milk proteins: Effect on protein retention in growing young rats. J. Dairy Sci. 2018, 102, 1066–1082. [Google Scholar] [CrossRef] [PubMed]
- El-Moghazy, G.; Dina, M.; Abd El Ghafar, N. Effect of fermentation of faba bean (Vicia faba) on its nutritive and sensory properties. J. Food Dairy Sci. 2011, 2, 237–250. [Google Scholar]
- Prasanna, B.M.; Vasal, S.K.; Kassahun, B.; Singh, N.N. Quality protein maize. Curr. Sci. 2001, 81, 1308–1319. [Google Scholar]
- Vasal, S. The quality protein maize story. Food Nutr. Bull. 2000, 21, 445–450. [Google Scholar] [CrossRef]
- Fufa, H.; Akalu, G.; Wondimu, A.; Taffesse, S.; Gebre, T.; Schlosser, K.; Noetzold, H.; Henle, T. Assessment of protein nutritional quality and effects of traditional processes: A comparison between Ethiopian quality protein maize and five Ethiopian adapted normal maize cultivars. Nahrung 2003, 47, 269–273. [Google Scholar] [CrossRef]
- Akalu, G.; Taffesse, S.; Gunaratna, N.S.; De Groote, H. The effectiveness of quality protein maize in improving the nutritional status of young children in the Ethiopian highlands. Food Nutr. Bull. 2010, 31, 418–430. [Google Scholar] [CrossRef]
- Gunaratna, N.S.; De Groote, H.; Nestel, P.; Pixley, K.V.; McCabe, G.P. A meta-analysis of community-based studies on quality protein maize. Food Policy 2010, 35, 202–210. [Google Scholar] [CrossRef]
- Asif, M.; Rooney, L.W.; Ali, R.; Riaz, M.N. Application and opportunities of pulses in food system: A review. Crit. Rev. Food Sci. Nutr. 2013, 53, 1168–1179. [Google Scholar] [CrossRef]
- Petitot, M.; Boyer, L.; Minier, C.; Micard, V. Fortification of pasta with split pea and faba bean flours: Pasta processing and quality evaluation. Food Res. Int. 2010, 43, 634–641. [Google Scholar] [CrossRef]
| Protein Type | Protein Digestibility (%) | Biological Value (%) | Net Protein Utilization (%) | PDCAAS | DIAAS |
|---|---|---|---|---|---|
| Animal source | |||||
| Red meat 1 | 80 | 73 | 92 | ||
| Casein 1,3,6 | 99 | 77 | 76–82 | 100 | |
| Whey 1 | 104 | 92 | 100 | ||
| Milk 1,4,6 | 96 | 91 | 82 | 100 | 114 |
| Egg 1,4,6 | 98 | 100 | 94 | 100 | 113 |
| Plant source | |||||
| Black bean 1,6,8 | 70 | 75 | |||
| Cooked black bean 7,8 | 83 | 65 | 59 | ||
| Soy flour 5,8 | 80 | 93 | 89(SAA) | ||
| Soy protein isolate1,6 | 98 | 74 | 61 | 100 | |
| Green lentil 3,4 | 84 | 63 | 65 | ||
| Yellow split pea 4,6 | 88 | 64 | 73 | ||
| Cooked pea 7 | 89 | 60 | 58 | ||
| Pea protein concentrate 7 | 99 | 89 | 82 | ||
| Chickpea 3,4 | 89 | 74 | 83 | ||
| Peanuts 1 | 52 | ||||
| Roasted peanuts 7 | 98 | 51 | 43 | ||
| Peanut butter 3,4 | 98 | 45 | 46 | ||
| Whole grains 2 | 45 | ||||
| Wheat 3,5,6 | 91 | 56–68 | 53–65 | 51 | 45(Lys) |
| Wheat gluten 1 | 64 | 67 | 25 | ||
| White bread 4,6 | 93 | 28 | 29 | ||
| White rice 4,6 | 93 | 56 | 57 | ||
| Cooked rice 7 | 87 | 62 | 60 | ||
| Plant-Based Proteins | Animal-Based Proteins | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Wheat | Maize | Soybean | Pea | Faba Bean | Lentil | Whey | Casein | Milk | Beef | |
| Essential amino acid scores (%) 1 | ||||||||||
| Histidine | 140 | 187 | 173 | 167 | 231 | 176 | 127 | 180 | 180 | 240 |
| Isoleucine | 137 | 127 | 157 | 153 | 112 | 154 | 213 | 167 | 170 | 167 |
| Leucine | 115 | 219 | 136 | 125 | 121 | 132 | 168 | 151 | 161 | 144 |
| Lysine | 31 | 62 | 147 | 182 | 158 | 160 | 204 | 169 | 153 | 207 |
| Methionine + Cysteine | 120 | 127 | 91 | 73 | 79 | 91 | 130 | 125 | 134 | 157 |
| Phenylalanine + Tyrosine | 290 | 300 | 277 | 267 | 247 | 263 | 227 | 343 | 313 | 280 |
| Threonine | 109 | 161 | 174 | 191 | 156 | 165 | 291 | 187 | 174 | 209 |
| Valine | 108 | 128 | 126 | 131 | 95 | 135 | 162 | 162 | 159 | 133 |
| Study Type | Study (Reference) | Design | Method | Outcomes |
|---|---|---|---|---|
| Acute | Kanda et al. [30] Animal study | Young male Sprague-Dawley rats: n = 237 Oral administration of 3.1 g protein/kg BW Milk protein CC Whey protein CC Caseinate Soy protein CC | Isotope tracer | Soy proteins had an inferior effect on muscle protein synthesis after exercise compared with dairy proteins |
| Norton et al. [29] Animal study | Young rats: n = 52 (Exp 1) n = 18 (Exp 2) Exp 2: 4 g meals (16% protein) Whey protein isolate Wheat gluten Wheat gluten+ Leu | Isotope tracer | Exp 2: Fortifying wheat with leucine to match the leucine content of whey diet induced similar anabolic responses, i.e., similar muscle protein synthesis rates | |
| Tang et al. [75] Clinical study | 18 M Young subjects: 19–27 years Whey hydrolysate: 21.4 g Casein micelle: 21.9 g Soy protein isolate: 22.2 g | Isotope tracer | Muscle protein synthesis rates were in this order at rest condition: whey ≈ soy > casein after resistance exercise: whey > soy > casein | |
| Yang et al. [42] Clinical study | 30 M Older subjects: 66–76 years 20 or 40 g Whey protein isolate Soy protein isolate | Isotope tracer | Soy protein isolate had less ability to stimulate muscle protein synthesis, compared to whey protein isolate under both rested and post-exercise conditions | |
| Gorissen et al. [44] Clinical study | 60 M Older subjects: 70–72 years 35 or 60 g Whey protein isolate Micellar casein Wheat protein hydrolysate | Isotope tracer | Muscle protein synthesis rates were lower after ingesting 35 g wheat protein than after the same amount of casein. Ingesting a larger quantity of wheat protein (i.e., 60 g) substantially improved muscle protein synthesis rates in elderly men | |
| Laleg et al. [34] Animal study | Young male Wistar Rats n = 50 Ad libitum consumption of isoproteic and isocaloric diets for 3 weeks Faba bean-enriched pasta Wheat gluten pasta Casein | Echo-MRI | Rats fed legume-enriched pasta or wheat gluten pasta had a lower LM than rats fed casein | |
| Chronic | Volek et al. [37] Clinical study | 63 M + F Young subjects: 18–35 years Daily consumption of supplements containing whey or soy proteins + Whole-body periodized resistance training program for 36 weeks | DXA | Daily supplementation with whey was more effective than isoproteic and isocaloric supplement containing soy protein in enhancing LM during resistance training |
| Mobley et al. [38] Clinical study | 75 F Young subjects: 20–22 years Daily consumption (twice) of supplements containing whey or soy proteins + Whole-body resistance training (3 d/week) for 12 weeks | DXA | Whey and soy supplement groups showed similar increases in total body skeletal muscle mass and type I and II fiber cross-sectional area during resistance training | |
| Banaszek et al. [39] Clinical study | 15 M + F Young and adult subjects: 26–51 years Consumption of supplements containing whey or pea proteins on training day + High-intensity functional training (4 sessions/week) for 8 weeks | BIA | Ingestion of whey and pea protein produced similar outcomes in measurements of body composition, especially LM and muscle thickness | |
| Chan et al. [94] Clinical study | 1411 M + 1315 F Older subjects: 65 years and older L (4 years), FFQ | DXA | Higher plant (but not total and animal) protein intakes were associated with reduced muscle loss | |
| Chronic | Isanejad et al. [93] Clinical study | 554 F Older subjects: 65–72 years L (3 years); 3 d food record | DXA | Higher total and animal protein intakes were associated with increased LM and ALM Higher plant protein intake was associated with less reduction in ALM |
| Sahni et al. [88] Clinical study | 1139 M + 1497 F Young and older subjects: 29–86 years CS; FFQ | DXA | Higher total and animal (but not plant) protein intakes were associated with higher LM in the leg | |
| Miki et al. [96] Clinical study | 168 M + F with type 2 diabetes Older subjects: ≥ 65 years CS; BDHQ | BIA | Total and plant protein intakes were positively associated with skeletal muscle mass | |
| Huang et al. [95] Clinical study | 327 M + F Older subjects: 66–76 years CS; FFQ | BIA | Low total and plant protein intakes were associated with a higher risk for low muscle mass | |
| Verreijen et al. [97] Clinical study | 3075 M + F Older subjects: 70–79 years L (5 years); FFQ | DXA | Higher total, animal and plant protein intakes were not associated with changes in mid-thigh-muscle CSA | |
| Mangano et al. [89] Clinical study | 2986 M + F Young and older subjects: 19–72 years L; FFQ Food clusters 1. Fast food 2. Red meat 3. Fish 4. Chicken 5. Low-fat milk 6. Legumes | DXA | Individuals in the legume protein food cluster had significantly lower ALM compared with subjects in all other protein food clusters. No associations between protein clusters and any musculoskeletal outcomes in adjusted models. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Berrazaga, I.; Micard, V.; Gueugneau, M.; Walrand, S. The Role of the Anabolic Properties of Plant- versus Animal-Based Protein Sources in Supporting Muscle Mass Maintenance: A Critical Review. Nutrients 2019, 11, 1825. https://doi.org/10.3390/nu11081825
Berrazaga I, Micard V, Gueugneau M, Walrand S. The Role of the Anabolic Properties of Plant- versus Animal-Based Protein Sources in Supporting Muscle Mass Maintenance: A Critical Review. Nutrients. 2019; 11(8):1825. https://doi.org/10.3390/nu11081825
Chicago/Turabian StyleBerrazaga, Insaf, Valérie Micard, Marine Gueugneau, and Stéphane Walrand. 2019. "The Role of the Anabolic Properties of Plant- versus Animal-Based Protein Sources in Supporting Muscle Mass Maintenance: A Critical Review" Nutrients 11, no. 8: 1825. https://doi.org/10.3390/nu11081825
APA StyleBerrazaga, I., Micard, V., Gueugneau, M., & Walrand, S. (2019). The Role of the Anabolic Properties of Plant- versus Animal-Based Protein Sources in Supporting Muscle Mass Maintenance: A Critical Review. Nutrients, 11(8), 1825. https://doi.org/10.3390/nu11081825





