Abscisic Acid Standardized Fig (Ficus carica) Extracts Ameliorate Postprandial Glycemic and Insulinemic Responses in Healthy Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Study Treatments
2.3. Study Procedures
2.4. Data Analysis and Statistical Analysis
3. Results
3.1. Participant Characteristics
3.2. Tolerability of Test Beverages
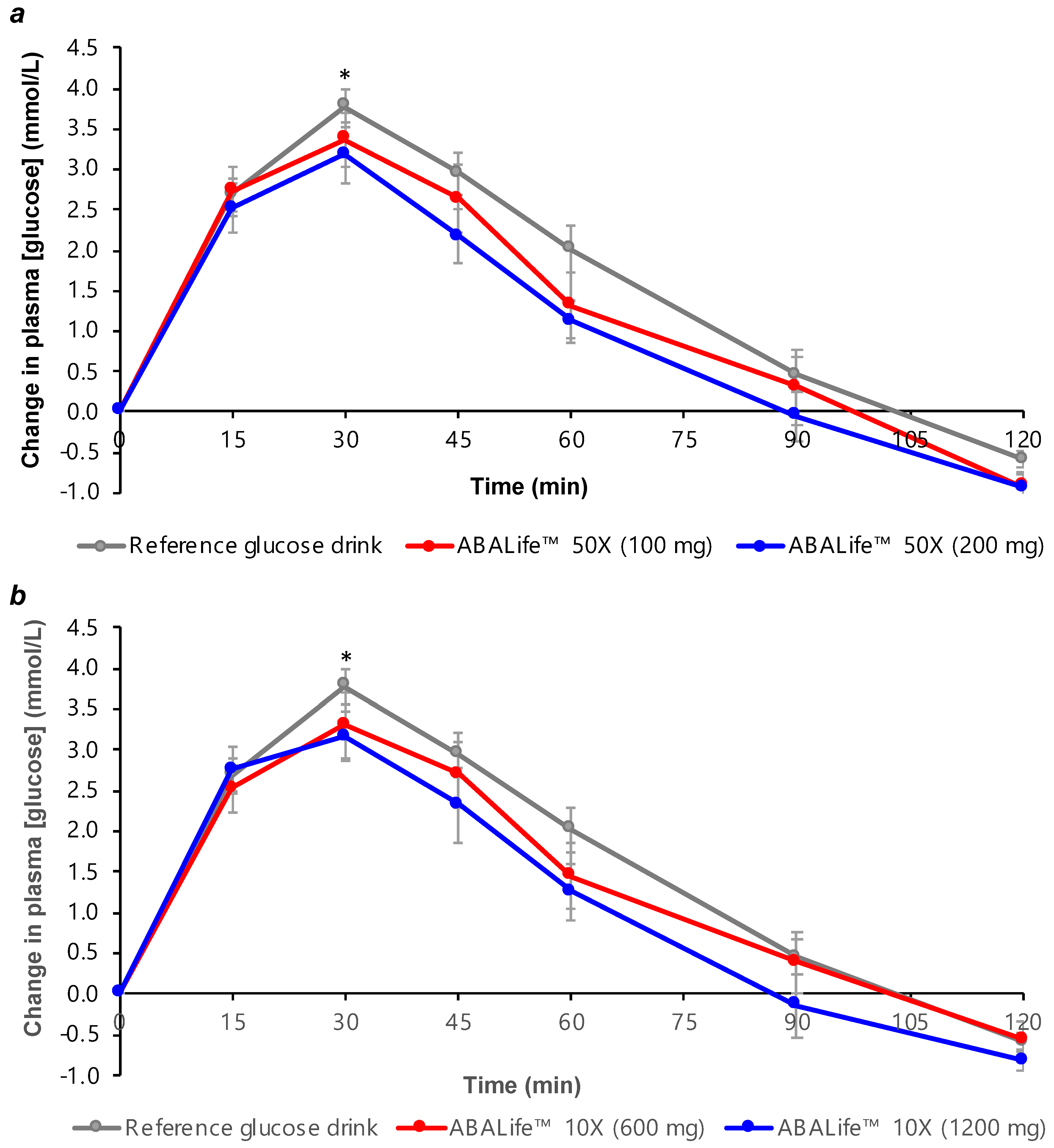
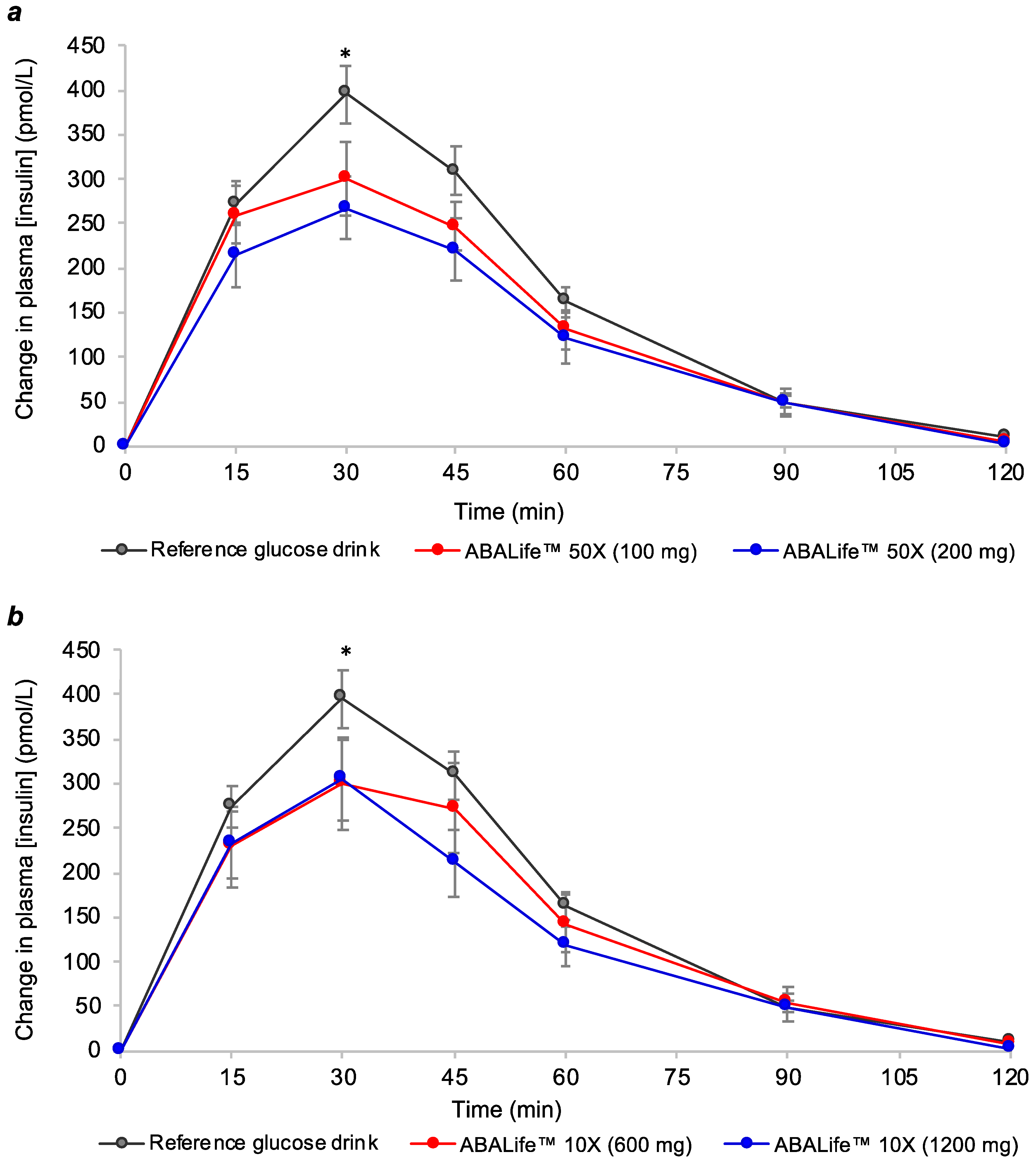
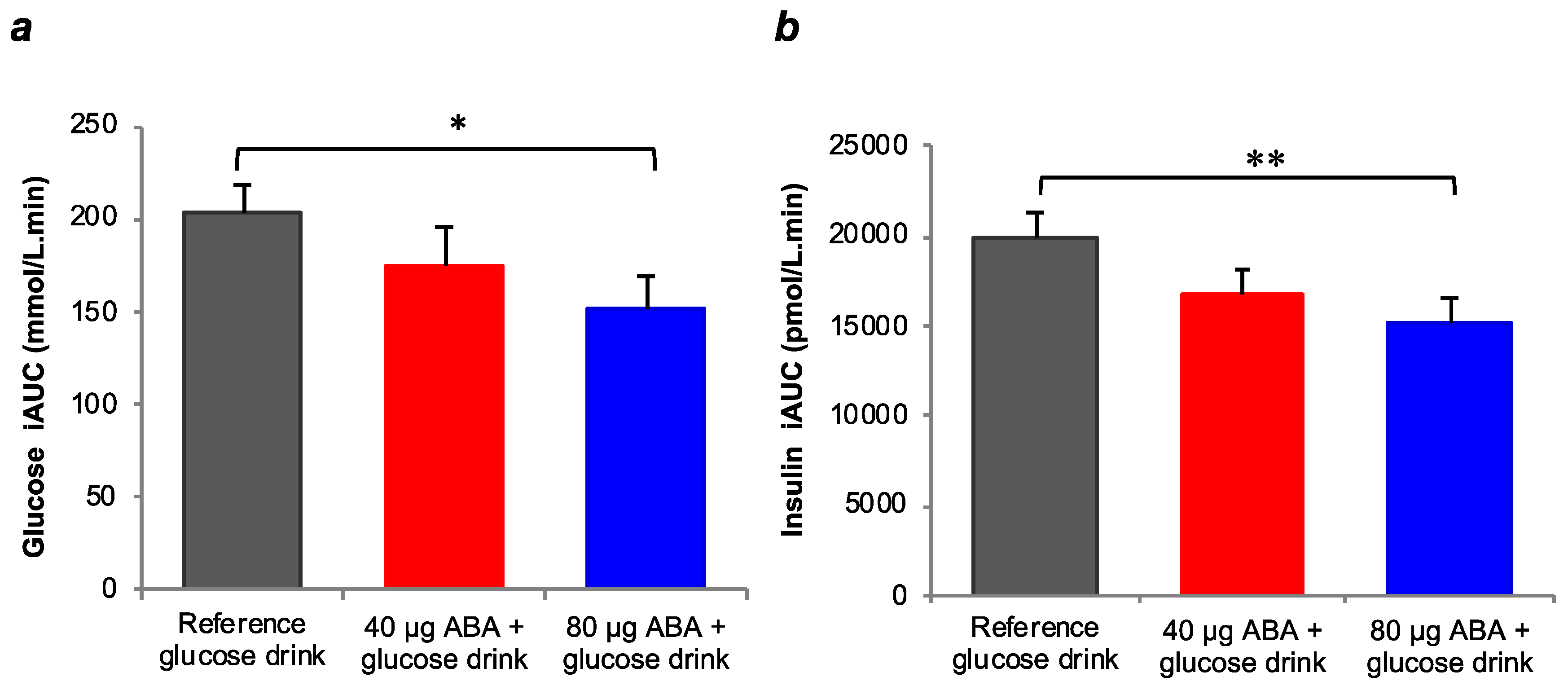
3.3. Two-Hour Plasma Glucose and Insulin Responses for Reference Beverage and Test Beverages
3.4. Glycemic Index and Insulinemic Index of Test Beverages
4. Discussion
5. Conclusions
6. Patents
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Edwards, C.M.; Cusi, K. Prediabetes: A worldwide epidemic. Endocrinol. Metab. Clin. N. Am. 2016, 45, 751–764. [Google Scholar] [CrossRef] [PubMed]
- International Diabetes Federation. IDF Diabetes Atlas, 8th ed.; International Diabetes Federation: Brussels, Belgium, 2017; Available online: http://www.diabetesatlas.org (accessed on 29 October 2018).
- Centers for Disease Control and Prevention. National Diabetes Statistics Report 2017; Centers for Disease Control and Prevention, U.S. Department of Health and Human Services: Atlanta, GA, USA, 2017. Available online: https://www.cdc.gov/diabetes/data/statistics/statistics-report.html (accessed on 29 October 2018).
- Handelsman, Y.; Bloomgarden, Z.T.; Grunberger, G.; Umpierrez, G.; Zimmerman, R.S.; Bailey, T.S.; Blonde, L.; Bray, G.A.; Cohen, A.J.; Dagogo-Jack, S.; et al. American association of clinical endocrinologists and American college of endocrinology—Clinical practice guidelines for developing a diabetes mellitus comprehensive care plan—2015. Endocr. Pract. 2015, 21, 1–87. [Google Scholar] [CrossRef] [PubMed]
- Bansal, N. Prediabetes diagnosis and treatment: A review. World J. Diabetes 2015, 6, 296–303. [Google Scholar] [CrossRef] [PubMed]
- Karimi, A.; Majlesi, M.; Rafieian-Kopaei, M. Herbal versus synthetic drugs; beliefs and facts. J. Nephropharmacol. 2015, 4, 27–30. [Google Scholar] [PubMed]
- Schulz, V. Safety of St. John’s Wort extract compared to synthetic antidepressants. Phytomedicine 2006, 13, 199–204. [Google Scholar] [CrossRef] [PubMed]
- Bruzzone, S.; Bodrato, N.; Usai, C.; Guida, L.; Moreschi, I.; Nano, R.; Antonioli, B.; Fruscione, F.; Magnone, M.; Scarfi, S.; et al. Abscisic Acid Is an Endogenous Stimulator of Insulin Release from Human Pancreatic Islets with Cyclic ADP Ribose as Second Messenger. J. Boil. Chem. 2008, 283, 32188–32197. [Google Scholar] [CrossRef]
- Guri, A.J.; Hontecillas, R.; Si, H.; Liu, D.; Bassaganya-Riera, J. Dietary abscisic acid ameliorates glucose tolerance and obesity-related inflammation in db/db mice fed high-fat diets. Clin. Nutr. 2007, 26, 107–116. [Google Scholar] [CrossRef]
- Qi, C.C.; Zhang, Z.; Fang, H.; Liu, J.; Zhou, N.; Ge, J.F.; Chen, F.H.; Xiang, C.B.; Zhou, J.N. Antidepressant effects of abscisic acid mediated by the downregulation of corticotrophin-releasing hormone gene expression in rats. Int. J. Neuropsychopharmacol. 2014, 18. [Google Scholar] [CrossRef]
- Lu, P.; Bevan, D.R.; Lewis, S.N.; Hontecillas, R.; Bassaganya-Riera, J. Molecular modeling of lanthionine synthetase component C-like protein 2: A potential target for the discovery of novel type 2 diabetes prophylactics and therapeutics. J. Mol. Model. 2011, 17, 543–553. [Google Scholar] [CrossRef]
- Sturla, L.; Fresia, C.; Guida, L.; Bruzzone, S.; Scarfi, S.; Usai, C.; Fruscione, F.; Magnone, M.; Millo, E.; Basile, G.; et al. LANCL2 Is Necessary for Abscisic Acid Binding and Signaling in Human Granulocytes and in Rat Insulinoma Cells. J. Boil. Chem. 2009, 284, 28045–28057. [Google Scholar] [CrossRef]
- Zeng, M.; Van Der Donk, W.A.; Chen, J. Lanthionine synthetase C-like protein 2 (LanCL2) is a novel regulator of Akt. Mol. Boil. Cell 2014, 25, 3954–3961. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Dehvari, N.; Dallner, O.S.; Olsen, J.M.; Csikasz, R.I.; Summers, R.J.; Hutchinson, D.S.; Bengtsson, T.; Öberg, A.I.; Sandström, A.L. Improving Type 2 Diabetes Through a Distinct Adrenergic Signaling Pathway Involving mTORC2 That Mediates Glucose Uptake in Skeletal Muscle. Diabetes 2014, 63, 4115–4129. [Google Scholar] [CrossRef] [PubMed]
- Bruzzone, S.; Ameri, P.; Briatore, L.; Mannino, E.; Basile, G.; Andraghetti, G.; Grozio, A.; Magnone, M.; Guida, L.; Scarfi, S.; et al. The plant hormone abscisic acid increases in human plasma after hyperglycemia and stimulates glucose consumption by adipocytes and myoblasts. FASEB J. 2012, 26, 1251–1260. [Google Scholar] [CrossRef] [PubMed]
- Ameri, P.; Bruzzone, S.; Mannino, E.; Sociali, G.; Andraghetti, G.; Salis, A.; Ponta, M.L.; Briatore, L.; Adami, G.F.; Ferraiolo, A.; et al. Impaired Increase of Plasma Abscisic Acid in Response to Oral Glucose Load in Type 2 Diabetes and in Gestational Diabetes. PLoS ONE 2015, 10, e0115992. [Google Scholar] [CrossRef] [PubMed]
- Vilsbøll, T.; Krarup, T.; Sonne, J.; Madsbad, S.; Vølund, A.; Juul, A.G.; Holst, J.J. Incretin Secretion in Relation to Meal Size and Body Weight in Healthy Subjects and People with Type 1 and Type 2 Diabetes Mellitus. J. Clin. Endocrinol. Metab. 2003, 88, 2706–2713. [Google Scholar] [CrossRef] [PubMed]
- Zocchi, E.; Hontecillas, R.; Leber, A.; Einerhand, A.; Carbo, A.; Bruzzone, S.; Tubau-Juni, N.; Philipson, N.; Zoccoli-Rodriguez, V.; Sturla, L.; et al. Abscisic Acid: A Novel Nutraceutical for Glycemic Control. Front. Nutr. 2017, 4, 24. [Google Scholar] [CrossRef] [PubMed]
- Magnone, M.; Ameri, P.; Salis, A.; Andraghetti, G.; Emionite, L.; Murialdo, G.; De Flora, A.; Zocchi, E. Microgram amounts of abscisic acid in fruit extracts improve glucose tolerance and reduce insulinemia in rats and in humans. FASEB J. 2015, 29, 4783–4793. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, D.J.; Wolever, T.M.; Taylor, R.H.; Barker, H.; Fielden, H.; Baldwin, J.M.; Bowling, A.C.; Newman, H.C.; Jenkins, A.L.; Goff, D.V. Glycemic index of foods: A physiological basis for carbohydrate exchange. Am. J. Clin. Nutr. 1981, 34, 362–366. [Google Scholar] [CrossRef] [PubMed]
- Wolever, T.M.S.; Brand-Miller, J.C.; Abernethy, J.; Astrup, A.; Atkinson, F.; Axelsen, M.; Björck, I.; Brighenti, F.; Brown, R.; Brynes, A.; et al. Measuring the glycemic index of foods: Interlaboratory study. Am. J. Clin. Nutr. 2008, 87, 247S–257S. [Google Scholar] [CrossRef] [PubMed]
- International Organization for Standardization. ISO 26642:2010: Food Products—Determination of the Glycaemic Index (GI) and Recommendation for Food Classification; ISO: Geneva, Switzerland, 2010. [Google Scholar]
- Liu, S.; Salas-Salvadó, J.; La Vecchia, C.; Poli, A.; Ha, M.-A.; Baer-Sinnott, S.; Brand-Miller, J.C.; Livesey, G.; Taylor, R.; Livesey, H.F.; et al. Dietary Glycemic Index and Load and the Risk of Type 2 Diabetes: A Systematic Review and Updated Meta-Analyses of Prospective Cohort Studies. Nutrients 2019, 11, 1280. [Google Scholar]
- Augustin, L.; Kendall, C.W.; Jenkins, D.J.; Willett, W.C.; Astrup, A.; Barclay, A.W.; Björck, I.; Brand-Miller, J.C.; Brighenti, F.; Buyken, A.E.; et al. Glycemic index, glycemic load and glycemic response: An International Scientific Consensus Summit from the International Carbohydrate Quality Consortium (ICQC). Nutr. Metab. Cardiovasc. Dis. 2015, 25, 795–815. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Li, Y.; Mu, W.; Li, Z.; Sun, J.; Wang, B.; Zhong, Z.; Luo, X.; Xie, C.; Huang, Y. Mulberry leaf extract reduces the glycemic indexes of four common dietary carbohydrates. Medicine 2018, 97, e11996. [Google Scholar] [CrossRef] [PubMed]
- Gibb, R.D.; McRorie, J.W.; Russell, D.A.; Hasselblad, V.; D’Alessio, D. Psyllium fiber improves glycemic control proportional to loss of glycemic control: A meta-analysis of data in euglycemic subjects, patients at risk of type 2 diabetes mellitus, and patients being treated for type 2 diabetes mellitus. Am. J. Clin. Nutr. 2015, 102, 1604–1614. [Google Scholar] [CrossRef] [PubMed]
- Pastors, J.G.; Blaisdell, P.W.; Balm, T.K.; Asplin, C.M.; Pohl, S.L. Psyllium fiber reduces rise in postprandial glucose and insulin concentrations in patients with non-insulin-dependent diabetes. Am. J. Clin. Nutr. 1991, 53, 1431–1435. [Google Scholar] [CrossRef] [PubMed]
- Groop, P.H.; Aro, A.; Stenman, S.; Groop, L. Long-term effects of guar gum in subjects with non-insulin-dependent diabetes mellitus. Am. J. Clin. Nutr. 1993, 58, 513–518. [Google Scholar] [CrossRef]
- Breneman, C.B.; Tucker, L. Dietary fibre consumption and insulin resistance—The role of body fat and physical activity. Br. J. Nutr. 2013, 110, 375–383. [Google Scholar] [CrossRef] [PubMed]
- Lloyd-Jones, D.M.; Hong, Y.; Labarthe, D.; Mozaffarian, D.; Appel, L.J.; Van Horn, L.; Greenlund, K.; Daniels, S.; Nichol, G.; Tomaselli, G.F.; et al. Defining and Setting National Goals for Cardiovascular Health Promotion and Disease Reduction. Circulation 2010, 121, 586–613. [Google Scholar] [CrossRef] [PubMed]
- Rehm, C.D.; Penalvo, J.L.; Afshin, A.; Mozaffarian, D. Dietary intake among US adults. 1999–2012. JAMA 2016, 315, 2542–2553. [Google Scholar] [CrossRef]
- Brand-Miller, J.; Hayne, S.; Petocz, P.; Colagiuri, S. Low-Glycemic Index Diets in the Management of Diabetes: A meta-analysis of randomized controlled trials. Diabetes Care 2003, 26, 2261–2267. [Google Scholar] [CrossRef]
- Elliott, E.J.; Thomas, D. Low glycaemic index, or low glycaemic load, diets for diabetes mellitus. Cochrane Database Syst. Rev. 2009. [Google Scholar] [CrossRef]
- Eleazu, C.O. The concept of low glycemic index and glycemic load foods as panacea for type 2 diabetes mellitus; prospects, challenges and solutions. Afr. Heal. Sci. 2016, 16, 468–479. [Google Scholar] [CrossRef] [PubMed]
| Test Food | GI Value | Mean GI Difference 1 | II Value | Mean II Difference 1 |
|---|---|---|---|---|
| Reference food (glucose drink) | 100 | ⁻ | 100 | ⁻ |
| ABALifeTM 50× (100 mg) + glucose drink | 87 ± 6 | −13 | 86 ± 5 # | −14 |
| ABALifeTM 50× (200 mg) + glucose drink | 75 ± 6 * | −25 | 76 ± 6 * | −24 |
| ABALifeTM 10× (600 mg) + glucose drink | 86 ± 5 | −14 | 84 ± 6 ** | −16 |
| ABALifeTM 10× (1200 mg) + glucose drink | 77 ± 5 ‡ | −24 | 76 ± 5 * | −24 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atkinson, F.S.; Villar, A.; Mulà, A.; Zangara, A.; Risco, E.; Smidt, C.R.; Hontecillas, R.; Leber, A.; Bassaganya-Riera, J. Abscisic Acid Standardized Fig (Ficus carica) Extracts Ameliorate Postprandial Glycemic and Insulinemic Responses in Healthy Adults. Nutrients 2019, 11, 1757. https://doi.org/10.3390/nu11081757
Atkinson FS, Villar A, Mulà A, Zangara A, Risco E, Smidt CR, Hontecillas R, Leber A, Bassaganya-Riera J. Abscisic Acid Standardized Fig (Ficus carica) Extracts Ameliorate Postprandial Glycemic and Insulinemic Responses in Healthy Adults. Nutrients. 2019; 11(8):1757. https://doi.org/10.3390/nu11081757
Chicago/Turabian StyleAtkinson, Fiona S., Agusti Villar, Anna Mulà, Andrea Zangara, Ester Risco, Carsten R. Smidt, Raquel Hontecillas, Andrew Leber, and Josep Bassaganya-Riera. 2019. "Abscisic Acid Standardized Fig (Ficus carica) Extracts Ameliorate Postprandial Glycemic and Insulinemic Responses in Healthy Adults" Nutrients 11, no. 8: 1757. https://doi.org/10.3390/nu11081757
APA StyleAtkinson, F. S., Villar, A., Mulà, A., Zangara, A., Risco, E., Smidt, C. R., Hontecillas, R., Leber, A., & Bassaganya-Riera, J. (2019). Abscisic Acid Standardized Fig (Ficus carica) Extracts Ameliorate Postprandial Glycemic and Insulinemic Responses in Healthy Adults. Nutrients, 11(8), 1757. https://doi.org/10.3390/nu11081757






