Infant Feeding, Vitamin D and IgE Sensitization to Food Allergens at 6 Years in a Longitudinal Icelandic Cohort
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants and Design
2.2. Dietary Assessments
2.3. Blood Sampling and Biochemical Analysis
2.4. Other Variables
2.5. Statistical Analyses
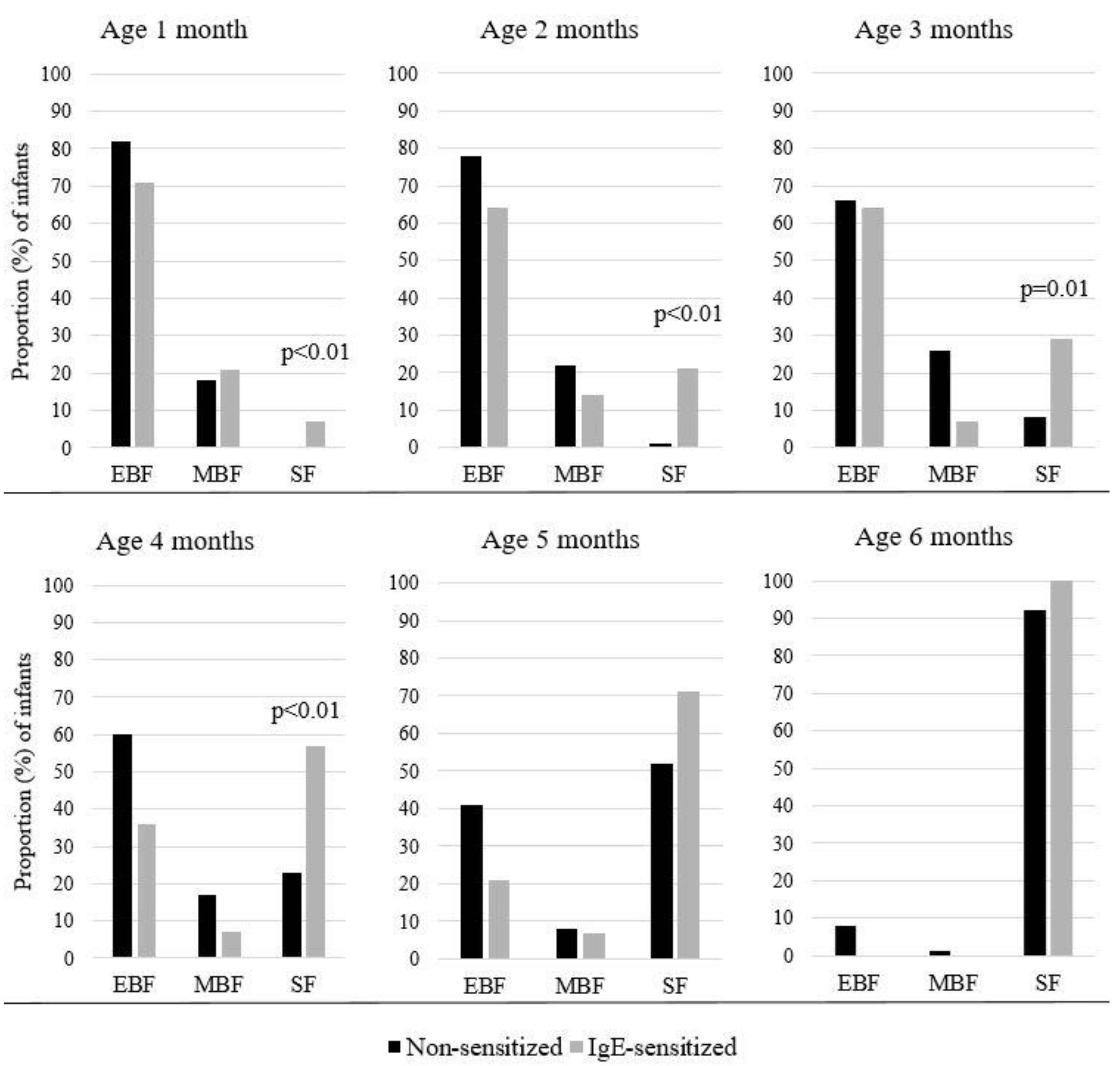
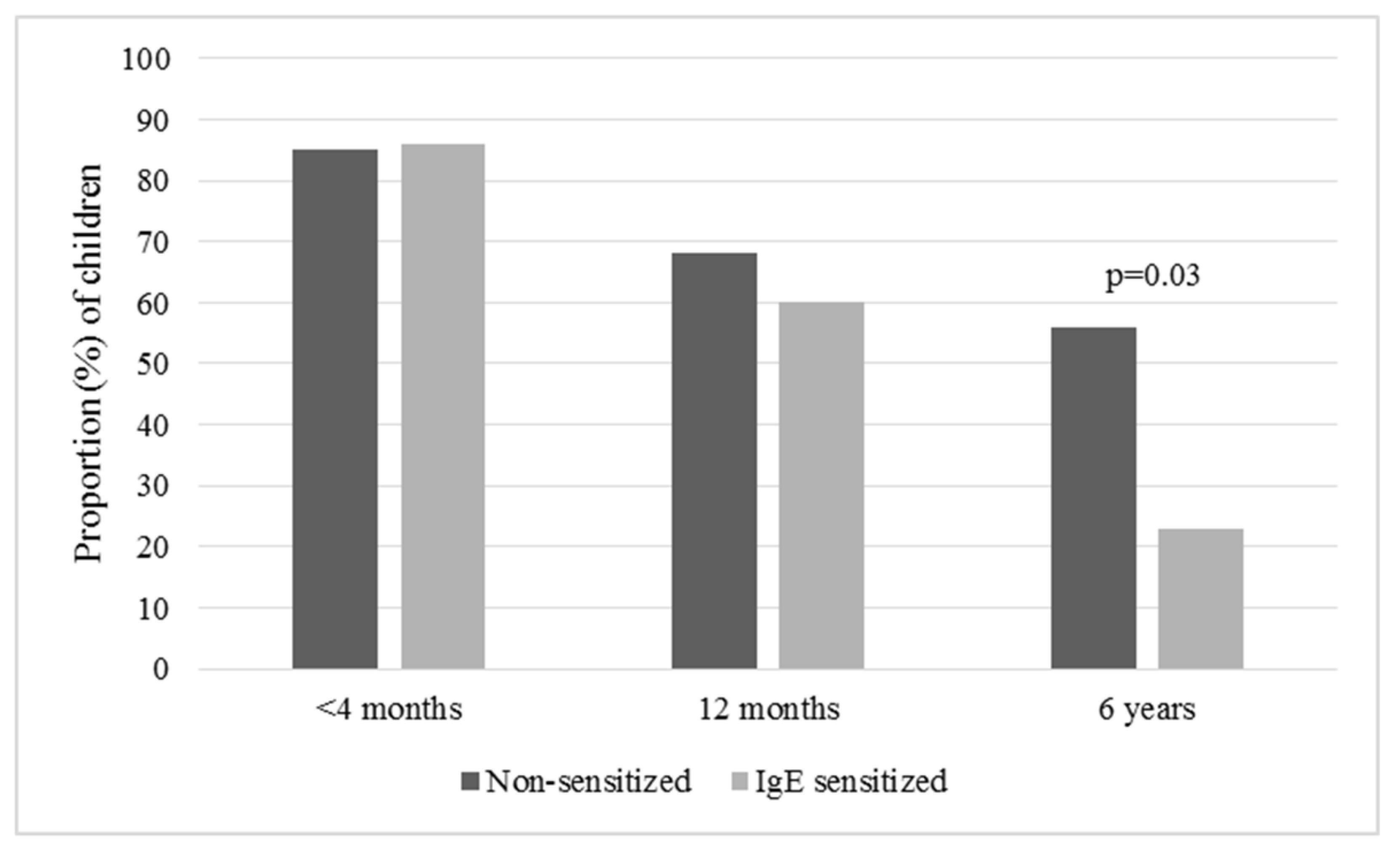
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ierodiakonou, D.; Garcia-Larsen, V.; Logan, A.; Groome, A.; Cunha, S.; Chivinge, J.; Robinson, Z.; Geoghegan, N.; Jarrold, K.; Reeves, T.; et al. Timing of allergenic food introduction to the infant diet and risk of allergic or autoimmune disease: A systematic review and meta-analysis. JAMA 2016, 316, 1181–1192. [Google Scholar] [CrossRef] [PubMed]
- Grimshaw, K.; Logan, K.; O’Donovan, S.; Kiely, M.; Patient, K.; van Bilsen, J.; Beyer, K.; Campbell, D.E.; Garcia-Larsen, V.; Grabenhenrich, L.; et al. Modifying the infant’s diet to prevent food allergy. Arch. Dis. Child. 2017, 102, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Nordic Council of Ministers. Nordic Nutrition Recommendations 2012: Integrating Nutrition and Physical Activity; Nordic Council of Ministers: Copenhagen, Denmark, 2014. [Google Scholar]
- Hornell, A.; Lagstrom, H.; Lande, B.; Thorsdottir, I. Breastfeeding, introduction of other foods and effects on health: A systematic literature review for the 5th Nordic nutrition recommendations. Food Nutr. Res. 2013, 57, 20823. [Google Scholar] [CrossRef] [PubMed]
- Sundhedsdatastyrelsen. Den Nationale Børnedatabase (BDB). Available online: https://sundhedsdatastyrelsen.dk/da/registre-og-services/om-de-nationale-sundhedsregistre/graviditet-foedsler-og-boern/boernedatabasen (accessed on 6 June 2019).
- Lande, B.; Helleve, A. Amming og Spedbarns Kosthold. Landsomfattende Undersøkelse; Rapport 2013; Helsedirektoratet: Oslo, Norway, 2014. [Google Scholar]
- Socialstyrelsen-The National Board of Health and Welfare. Statistics on breastfeeding 2016. In Official Statistics of Sweden. Health and Medical Care; Art. no. 2018-2019-2013; The National Board of Health and Welfare: Stockholm, Sweden, 2018; ISSN 1400-3511. [Google Scholar]
- Greer, F.R.; Sicherer, S.H.; Burks, A.W. Effects of early nutritional interventions on the development of atopic disease in infants and children: The role of maternal dietary restriction, breastfeeding, timing of introduction of complementary foods, and hydrolyzed formulas. Pediatrics 2008, 121, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Chan, E.S.; Cummings, C. Dietary exposures and allergy prevention in high-risk infants: A joint statement with the Canadian society of allergy and clinical immunology. Pediatr. Child Health 2013, 18, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Muraro, A.; Halken, S.; Arshad, S.H.; Beyer, K.; Dubois, A.E.; Du Toit, G.; Eigenmann, P.A.; Grimshaw, K.E.; Hoest, A.; Lack, G.; et al. EAACI food allergy and anaphylaxis guidelines. Primary prevention of food allergy. Allergy 2014, 69, 590–601. [Google Scholar] [CrossRef] [PubMed]
- Gungor, D.; Nadaud, P.; LaPergola, C.C.; Dreibelbis, C.; Wong, Y.P.; Terry, N.; Abrams, S.A.; Beker, L.; Jacobovits, T.; Jarvinen, K.M.; et al. Infant milk-feeding practices and food allergies, allergic rhinitis, atopic dermatitis, and asthma throughout the life span: A systematic review. Am. J. Clin. Nutr. 2019, 109, 772s–799s. [Google Scholar] [CrossRef] [PubMed]
- Obbagy, J.E.; English, L.K.; Wong, Y.P.; Butte, N.F.; Dewey, K.G.; Fleischer, D.M.; Fox, M.K.; Greer, F.R.; Krebs, N.F.; Scanlon, K.S.; et al. Complementary feeding and food allergy, atopic dermatitis/eczema, asthma, and allergic rhinitis: A systematic review. Am. J. Clin. Nutr. 2019, 109, 890s–934s. [Google Scholar] [CrossRef] [PubMed]
- Thorisdottir, B.; Gunnarsdottir, I.; Steingrimsdottir, L.; Palsson, G.I.; Birgisdottir, B.E.; Thorsdottir, I. Vitamin D intake and status in 6-year-old Icelandic children followed up from infancy. Nutrients 2016, 8, 75. [Google Scholar] [CrossRef] [PubMed]
- Thorisdottir, B.; Gunnarsdottir, I.; Steingrimsdottir, L.; Palsson, G.I.; Thorsdottir, I. Vitamin D intake and status in 12-month-old infants at 63–66° N. Nutrients 2014, 6, 1182–1193. [Google Scholar] [CrossRef] [PubMed]
- Sharief, S.; Jariwala, S.; Kumar, J.; Muntner, P.; Melamed, M.L. Vitamin D levels and food and environmental allergies in the United States: Results from the National health and nutrition examination survey 2005–2006. J. Allergy Clin. Immunol. 2011, 127, 1195–1202. [Google Scholar] [CrossRef] [PubMed]
- Baek, J.H.; Shin, Y.H.; Chung, I.H.; Kim, H.J.; Yoo, E.G.; Yoon, J.W.; Jee, H.M.; Chang, Y.E.; Han, M.Y. The link between serum vitamin D level, sensitization to food allergens, and the severity of atopic dermatitis in infancy. J. Pediatr. 2014, 165, 849–854. [Google Scholar] [CrossRef] [PubMed]
- Hypponen, E.; Sovio, U.; Wjst, M.; Patel, S.; Pekkanen, J.; Hartikainen, A.L.; Jarvelinb, M.R. Infant vitamin D supplementation and allergic conditions in adulthood: Northern Finland birth cohort 1966. Ann. N. Y. Acad. Sci. 2004, 1037, 84–95. [Google Scholar] [CrossRef] [PubMed]
- Kull, I.; Bergstrom, A.; Melen, E.; Lilja, G.; van Hage, M.; Pershagen, G.; Wickman, M. Early-life supplementation of vitamins A and D, in water-soluble form or in peanut oil, and allergic diseases during childhood. J. Allergy Clin. Immunol. 2006, 118, 1299–1304. [Google Scholar] [CrossRef] [PubMed]
- Hughes, A.M.; Lucas, R.M.; Ponsonby, A.L.; Chapman, C.; Coulthard, A.; Dear, K.; Dwyer, T.; Kilpatrick, T.J.; McMichael, A.J.; Pender, M.P.; et al. The role of latitude, ultraviolet radiation exposure and vitamin D in childhood asthma and hayfever: An Australian multicenter study. Pediatr. Allergy Immunol. Off. Publ. Eur. Soc. Pediatr. Allergy Immunol. 2011, 22, 327–333. [Google Scholar] [CrossRef]
- Hamilton, R.G.; Oppenheimer, J. Serological IgE analyses in the diagnostic algorithm for allergic disease. J. Allergy Clin. Immunol Pract. 2015, 3, 833–840. [Google Scholar] [CrossRef] [PubMed]
- Eigenmann, P.A. Do we still need oral food challenges for the diagnosis of food allergy? Pediatr. Allergy Immunol. Off. Publ. Eur. Soc. Pediatr. Allergy Immunol. 2018, 29, 239–242. [Google Scholar] [CrossRef]
- Thorisdottir, A.V.; Thorsdottir, I.; Palsson, G.I. Nutrition and iron status of 1-year olds following a revision in infant dietary recommendations. Anemia 2011, 2011, 986303. [Google Scholar] [CrossRef]
- Thorisdottir, B.; Gunnarsdottir, I.; Palsson, G.I.; Halldorsson, T.I.; Thorsdottir, I. Animal protein intake at 12 months is associated with growth factors at the age of six. Acta Paediatr. 2014, 103, 512–517. [Google Scholar] [CrossRef]
- Thorisdottir, A.V.; Gunnarsdottir, I.; Palsson, G.I.; Gretarsson, S.J.; Thorsdottir, I. Iron status and developmental scores in 6-year-olds highlights ongoing need to tackle iron deficiency in infants. Acta Paediatr. 2013, 102, 914–919. [Google Scholar] [CrossRef]
- Matis. The Icelandic Food Composition Database ISGEM. Available online: http://www.matis.is/neytendur/naeringargildi-matvaela/um-gagnagrunninn/ (accessed on 6 July 2018).
- Roche Diagnostics. Vitamin D Total. 25-Hydroxyvitamin D Manual; Roche Diagnostics: Mannheim, Germany, 2013. [Google Scholar]
- Matsui, T.; Tanaka, K.; Yamashita, H.; Saneyasu, K.I.; Tanaka, H.; Takasato, Y.; Sugiura, S.; Inagaki, N.; Ito, K. Food allergy is linked to season of birth, sun exposure, and vitamin D deficiency. Allergol. Int. Off. J. Jpn. Soc. Allergol. 2019, 68, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Elholm, G.; Linneberg, A.; Husemoen, L.L.; Omland, O.; Gronager, P.M.; Sigsgaard, T.; Schlunssen, V. The Danish urban-rural gradient of allergic sensitization and disease in adults. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2016, 46, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Strachan, D.P.; Ait-Khaled, N.; Foliaki, S.; Mallol, J.; Odhiambo, J.; Pearce, N.; Williams, H.C. Siblings, asthma, rhinoconjunctivitis and eczema: A worldwide perspective from the International study of asthma and allergies in childhood. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2015, 45, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Ciaccio, C.E.; Gentile, D. Effects of tobacco smoke exposure in childhood on atopic diseases. Curr. Allergy Asthma Rep. 2013, 13, 687–692. [Google Scholar] [CrossRef] [PubMed]
- Thacher, J.D.; Gruzieva, O.; Pershagen, G.; Neuman, A.; van Hage, M.; Wickman, M.; Kull, I.; Melen, E.; Bergstrom, A. Parental smoking and development of allergic sensitization from birth to adolescence. Allergy 2016, 71, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Thorisdottir, A.V.; Gunnarsdottir, I.; Thorsdottir, I. Revised infant dietary recommendations: The impact of maternal education and other parental factors on adherence rates in Iceland. Acta Paediatr. 2013, 102, 143–148. [Google Scholar] [CrossRef]
- World Health Organization. Growth Reference Data for 5–19 Years; WHO Reference: Geneva, Switzerland, 2007; Available online: www.who.int/growthref/en (accessed on 4 July 2019).
- Cole, T.J.; Bellizzi, M.C.; Flegal, K.M.; Dietz, W.H. Establishing a standard definition for child overweight and obesity worldwide: International survey. BMJ Clin. Res. 2000, 320, 1240–1243. [Google Scholar] [CrossRef]
- Clausen, M.; Jonasson, K.; Keil, T.; Beyer, K.; Sigurdardottir, S.T. Fish oil in infancy protects against food allergy in Iceland—Results from a birth cohort study. Allergy 2018, 73, 1305–1312. [Google Scholar] [CrossRef]
- Clausen, M.; Kristjansson, S.; Haraldsson, A.; Bjorksten, B. High prevalence of allergic diseases and sensitization in a low allergen country. Acta Paediatr. 2008, 97, 1216–1220. [Google Scholar] [CrossRef]
- Kristinsdottir, H.; Clausen, M.; Ragnarsdottir, H.S.; Halldorsdottir, I.H.; McBride, D.; Beyer, K.; Sigurdardottir, S.T. Prevalence of food allergy in Icelandic infants during first year of life. Laeknabladid 2011, 97, 11–18. [Google Scholar]
- Longo, G.; Berti, I.; Burks, A.W.; Krauss, B.; Barbi, E. IgE-mediated food allergy in children. Lancet 2013, 382, 1656–1664. [Google Scholar] [CrossRef]
- Laan, M.P.; Baert, M.R.; Bijl, A.M.; Vredendaal, A.E.; De Waard-van der Spek, F.B.; Oranje, A.P.; Savelkoul, H.F.; Neijens, H.J. Markers for early sensitization and inflammation in relation to clinical manifestations of atopic disease up to 2 years of age in 133 high-risk children. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2000, 30, 944–953. [Google Scholar] [CrossRef]
- Kulig, M.; Bergmann, R.; Klettke, U.; Wahn, V.; Tacke, U.; Wahn, U. Natural course of sensitization to food and inhalant allergens during the first 6 years of life. J. Allergy Clin. Immunol. 1999, 103, 1173–1179. [Google Scholar] [CrossRef]
- Fewtrell, M.; Bronsky, J.; Campoy, C.; Domellof, M.; Embleton, N.; Fidler Mis, N.; Hojsak, I.; Hulst, J.M.; Indrio, F.; Lapillonne, A.; et al. Complementary feeding: A position paper by the European society for paediatric gastroenterology, hepatology, and nutrition (ESPGHAN) committee on nutrition. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 119–132. [Google Scholar] [CrossRef] [PubMed]
- Naylor, A.J.; Morrow, A. (Eds.) Developmental Readiness of Normal Full Term Infants to Progress from Exclusive Breastfeeding to the Introduction of Complementary Foods: Reviews of the Relevant Literature Concerning Infant Immunologic, Gastrointestinal, Oral Motor and Maternal Reproductive and Lactational Development; Academy for Educational Development, The LINKAGES Project, Wellstart International: Washington, DC, USA, 2001. [Google Scholar]
- West, C.E.; Ryden, P.; Lundin, D.; Engstrand, L.; Tulic, M.K.; Prescott, S.L. Gut microbiome and innate immune response patterns in IgE-associated eczema. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2015, 45, 1419–1429. [Google Scholar] [CrossRef] [PubMed]
- Perkin, M.R.; Logan, K.; Tseng, A.; Raji, B.; Ayis, S.; Peacock, J.; Brough, H.; Marrs, T.; Radulovic, S.; Craven, J.; et al. Randomized trial of introduction of allergenic foods in breast-fed infants. N. Engl. J. Med. 2016, 374, 1733–1743. [Google Scholar] [CrossRef] [PubMed]
- Du Toit, G.; Roberts, G.; Sayre, P.H.; Bahnson, H.T.; Radulovic, S.; Santos, A.F.; Brough, H.A.; Phippard, D.; Basting, M.; Feeney, M.; et al. Randomized trial of peanut consumption in infants at risk for peanut allergy. N. Engl. J. Med. 2015, 372, 803–813. [Google Scholar] [CrossRef]
- Wei-Liang Tan, J.; Valerio, C.; Barnes, E.H.; Turner, P.J.; Van Asperen, P.A.; Kakakios, A.M.; Campbell, D.E. A randomized trial of egg introduction from 4 months of age in infants at risk for egg allergy. J. Allergy Clin. Immunol. 2017, 139, 1621–1628. [Google Scholar] [CrossRef]
- Nwaru, B.I.; Takkinen, H.M.; Niemela, O.; Kaila, M.; Erkkola, M.; Ahonen, S.; Tuomi, H.; Haapala, A.M.; Kenward, M.G.; Pekkanen, J.; et al. Introduction of complementary foods in infancy and atopic sensitization at the age of 5 years: Timing and food diversity in a Finnish birth cohort. Allergy 2013, 68, 507–516. [Google Scholar] [CrossRef]
- Grimshaw, K.E.; Maskell, J.; Oliver, E.M.; Morris, R.C.; Foote, K.D.; Mills, E.N.; Roberts, G.; Margetts, B.M. Introduction of complementary foods and the relationship to food allergy. Pediatrics 2013, 132, e1529–e1538. [Google Scholar] [CrossRef]
- Bellach, J.; Schwarz, V.; Ahrens, B.; Trendelenburg, V.; Aksunger, O.; Kalb, B.; Niggemann, B.; Keil, T.; Beyer, K. Randomized placebo-controlled trial of hen’s egg consumption for primary prevention in infants. J. Allergy Clin. Immunol. 2017, 139, 1591–1599. [Google Scholar] [CrossRef] [PubMed]
- Daniels, L.; Mallan, K.M.; Fildes, A.; Wilson, J. The timing of solid introduction in an ‘obesogenic’ environment: A narrative review of the evidence and methodological issues. Austr. N. Z. J. Public Health 2015, 39, 366–373. [Google Scholar] [CrossRef] [PubMed]
- Durmus, B.; Heppe, D.H.; Gishti, O.; Manniesing, R.; Abrahamse-Berkeveld, M.; van der Beek, E.M.; Hofman, A.; Duijts, L.; Gaillard, R.; Jaddoe, V.W. General and abdominal fat outcomes in school-age children associated with infant breastfeeding patterns. Am. J. Clin. Nutr. 2014, 99, 1351–1358. [Google Scholar] [CrossRef] [PubMed]
- Grote, V.; Schiess, S.A.; Closa-Monasterolo, R.; Escribano, J.; Giovannini, M.; Scaglioni, S.; Stolarczyk, A.; Gruszfeld, D.; Hoyos, J.; Poncelet, P.; et al. The introduction of solid food and growth in the first 2 y of life in formula-fed children: Analysis of data from a European cohort study. Am. J. Clin. Nutr. 2011, 94, 1785s–1793s. [Google Scholar] [PubMed]
- Huh, S.Y.; Rifas-Shiman, S.L.; Taveras, E.M.; Oken, E.; Gillman, M.W. Timing of solid food introduction and risk of obesity in preschool-aged children. Pediatrics 2011, 127, e544–e551. [Google Scholar] [CrossRef] [PubMed]
- Weng, S.F.; Redsell, S.A.; Swift, J.A.; Yang, M.; Glazebrook, C.P. Systematic review and meta-analyses of risk factors for childhood overweight identifiable during infancy. Arch. Dis. Child. 2012, 97, 1019–1026. [Google Scholar] [CrossRef] [PubMed]
- Anderson, E.L.; Fraser, A.; Martin, R.M.; Kramer, M.S.; Oken, E.; Patel, R.; Tilling, K. Associations of postnatal growth with asthma and atopy: The PROBIT Study. Pediatr. Allergy Immunol. Off. Publ. Eur. Soc. Pediatr. Allergy Immunol. 2013, 24, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Matos, S.M.; Jesus, S.R.; Saldiva, S.R.; Prado, M.S.; D’Innocenzo, S.; Assis, A.M.; Rodrigues, L.C.; Alcantara-Neves, N.M.; Cruz, A.A.; Simoes, S.D.M.; et al. Weight gain in the first two years of life, asthma and atopy: The SCAALA cohort study. Public Health Nutr. 2014, 17, 2537–2545. [Google Scholar] [CrossRef] [PubMed]
- Sonnenschein-van der Voort, A.M.; Jaddoe, V.W.; Raat, H.; Moll, H.A.; Hofman, A.; de Jongste, J.C.; Duijts, L. Fetal and infant growth and asthma symptoms in preschool children: The generation R study. Am. J. Respir. Critic. Care Med. 2012, 185, 731–737. [Google Scholar] [CrossRef]
- Tedner, S.G.; Ortqvist, A.K.; Almqvist, C. Fetal growth and risk of childhood asthma and allergic disease. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2012, 42, 1430–1447. [Google Scholar] [CrossRef]
- Ortqvist, A.K.; Lundholm, C.; Carlstrom, E.; Lichtenstein, P.; Cnattingius, S.; Almqvist, C. Familial factors do not confound the association between birth weight and childhood asthma. Pediatrics 2009, 124, e737–e743. [Google Scholar] [CrossRef] [PubMed]
- Lucas, J.S.; Inskip, H.M.; Godfrey, K.M.; Foreman, C.T.; Warner, J.O.; Gregson, R.K.; Clough, J.B. Small size at birth and greater postnatal weight gain: Relationships to diminished infant lung function. Am. J. Respir. Critic. Care Med. 2004, 170, 534–540. [Google Scholar] [CrossRef] [PubMed]
- Tauber, E.; Gartner, C.; Halmerbauer, G.; Rath, R.; Schelberger, B.; Schierl, M.; Frischer, T. Predictors of lung function in infants at high risk of atopy: Effect of allergen avoidance. Respir. Med. 2002, 96, 230–235. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hancox, R.J.; Poulton, R.; Greene, J.M.; McLachlan, C.R.; Pearce, M.S.; Sears, M.R. Associations between birth weight, early childhood weight gain and adult lung function. Thorax 2009, 64, 228–232. [Google Scholar] [CrossRef] [PubMed]
- Pike, K.C.; Crozier, S.R.; Lucas, J.S.; Inskip, H.M.; Robinson, S.; Roberts, G.; Godfrey, K.M. Patterns of fetal and infant growth are related to atopy and wheezing disorders at age 3 years. Thorax 2010, 65, 1099–1106. [Google Scholar] [CrossRef] [PubMed]
- Hersoug, L.G.; Linneberg, A. The link between the epidemics of obesity and allergic diseases: Does obesity induce decreased immune tolerance? Allergy 2007, 62, 1205–1213. [Google Scholar] [CrossRef] [PubMed]
- Boulet, L.P. Obesity and atopy. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2015, 45, 75–86. [Google Scholar] [CrossRef]
- Imai, C.M.; Gunnarsdottir, I.; Thorisdottir, B.; Halldorsson, T.I.; Thorsdottir, I. Associations between infant feeding practice prior to six months and body mass index at six years of age. Nutrients 2014, 6, 1608–1617. [Google Scholar] [CrossRef]
- Thorisdottir, B.; Gunnarsdottir, I.; Thorisdottir, A.V.; Palsson, G.I.; Halldorsson, T.I.; Thorsdottir, I. Nutrient intake in infancy and body mass index at six years in two population-based cohorts recruited before and after revision of infant dietary recommendations. Ann. Nutr. Metab. 2013, 63, 145–151. [Google Scholar] [CrossRef]
- Smith, H.A.; Becker, G.E. Early additional food and fluids for healthy breastfed full-term infants. Cochrane Database Syst. Rev. 2016, Cd006462. [Google Scholar] [CrossRef]
- Gunnarsson, O.; Indriethason, O.S.; Franzson, L.; Halldorsdottir, E.; Sigurethsson, G. Vitamin-D homeostasis amongst adult Icelandic population. Laeknabladid 2004, 90, 29–36. [Google Scholar] [PubMed]
- Steingrimsdottir, L.; Gunnarsson, O.; Indridason, O.S.; Franzson, L.; Sigurdsson, G. Relationship between serum parathyroid hormone levels, vitamin D sufficiency, and calcium intake. JAMA 2005, 294, 2336–2341. [Google Scholar] [CrossRef] [PubMed]
- Hypponen, E.; Berry, D.J.; Wjst, M.; Power, C. Serum 25-hydroxyvitamin D and IgE—A significant but nonlinear relationship. Allergy 2009, 64, 613–620. [Google Scholar] [CrossRef] [PubMed]
- Hollams, E.M.; Teo, S.M.; Kusel, M.; Holt, B.J.; Holt, K.E.; Inouye, M.; De Klerk, N.H.; Zhang, G.; Sly, P.D.; Hart, P.H.; et al. Vitamin D over the first decade and susceptibility to childhood allergy and asthma. J. Allergy Clin. Immunol. 2017, 139, 472–481. [Google Scholar] [CrossRef] [PubMed]
- Thorisdottir, B. Vitamin D in Northern Latitudes. Intake and Status in Icelandic Children. Ph.D. Thesis, Faculty of Food Science and Nutrition, University of Iceland, Reykjavík, Iceland, 2018. [Google Scholar]
- Suaini, N.H.; Zhang, Y.; Vuillermin, P.J.; Allen, K.J.; Harrison, L.C. Immune modulation by Vitamin D and its relevance to food allergy. Nutrients 2015, 7, 6088–6108. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. The relationship between the fatty acid composition of immune cells and their function. Prostaglandins Leukotrienes Essent. Fatty Acids 2008, 79, 101–108. [Google Scholar] [CrossRef] [PubMed]
| Non-Sensitized n = 130 | IgE Sensitized n = 14 | p | |
|---|---|---|---|
| Infant information | |||
| Boys, n (%) | 65 (50) | 9 (64) | 0.31 |
| Birth in winter/spring, n (%) | 70 (54) | 8 (57) | 0.81 |
| Urban living, n (%) | 91 (70) | 11 (79) | 0.50 |
| First born, n (%) | 42 (36) | 2 (17) | 0.18 |
| Exclusive breastfeeding, months | 4 (2, 5) | 4 (0, 4) | 0.18 |
| Any breastfeeding, months | 8 (6, 10) | 8 (7, 11) | 0.97 |
| Weight gain 0–2 months, kg | 1.8 ± 0.5 | 2.2 ± 0.4 | 0.04 |
| Weight gain 2–6 months, kg | 2.4 ± 0.7 | 2.3 ± 0.7 | 0.59 |
| Length gain 0–6 months 1, cm | 16.8 ± 2.0 | 17.8 ± 1.7 | 0.09 |
| Head circumference gain 0–2 months, cm | 4.2 ± 1.0 | 4.9 ± 1.2 | 0.02 |
| Head circumference gain 2–6 months, cm | 4.2 ± 0.7 | 3.9 ± 0.8 | 0.15 |
| Parent information 2 | |||
| Maternal smoking, n (%) | 4 (3) | 2 (15) | 0.05 |
| Paternal smoking, n (%) | 18 (16) | 3 (23) | 0.49 |
| Maternal age, years | 31.3 ± 4.8 | 33.2 ± 5.4 | 0.18 |
| Paternal age, years | 34.1 ± 5.7 | 36.5 ± 6.6 | 0.16 |
| Maternal BMI, kg/m2 | 23.5 (21.4, 26.5) | 26.6 (21.8, 33.2) | 0.21 |
| Paternal BMI, kg/m2 | 26.0 (24.3, 28.1) | 26.6 (23.4, 28.7) | 0.82 |
| Basic education mother 3, n (%) | 19 (16) | 4 (31) | 0.19 |
| Basic education father 3, n (%) | 26 (23) | 4 (31) | 0.52 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thorisdottir, B.; Gunnarsdottir, I.; Vidarsdottir, A.G.; Sigurdardottir, S.; Birgisdottir, B.E.; Thorsdottir, I. Infant Feeding, Vitamin D and IgE Sensitization to Food Allergens at 6 Years in a Longitudinal Icelandic Cohort. Nutrients 2019, 11, 1690. https://doi.org/10.3390/nu11071690
Thorisdottir B, Gunnarsdottir I, Vidarsdottir AG, Sigurdardottir S, Birgisdottir BE, Thorsdottir I. Infant Feeding, Vitamin D and IgE Sensitization to Food Allergens at 6 Years in a Longitudinal Icelandic Cohort. Nutrients. 2019; 11(7):1690. https://doi.org/10.3390/nu11071690
Chicago/Turabian StyleThorisdottir, Birna, Ingibjorg Gunnarsdottir, Anna Gudrun Vidarsdottir, Sigurveig Sigurdardottir, Bryndis Eva Birgisdottir, and Inga Thorsdottir. 2019. "Infant Feeding, Vitamin D and IgE Sensitization to Food Allergens at 6 Years in a Longitudinal Icelandic Cohort" Nutrients 11, no. 7: 1690. https://doi.org/10.3390/nu11071690
APA StyleThorisdottir, B., Gunnarsdottir, I., Vidarsdottir, A. G., Sigurdardottir, S., Birgisdottir, B. E., & Thorsdottir, I. (2019). Infant Feeding, Vitamin D and IgE Sensitization to Food Allergens at 6 Years in a Longitudinal Icelandic Cohort. Nutrients, 11(7), 1690. https://doi.org/10.3390/nu11071690





