Adjuvant Effect of Molecular Iodine in Conventional Chemotherapy for Breast Cancer. Randomized Pilot Study
Abstract
1. Introduction
2. Materials and Methods
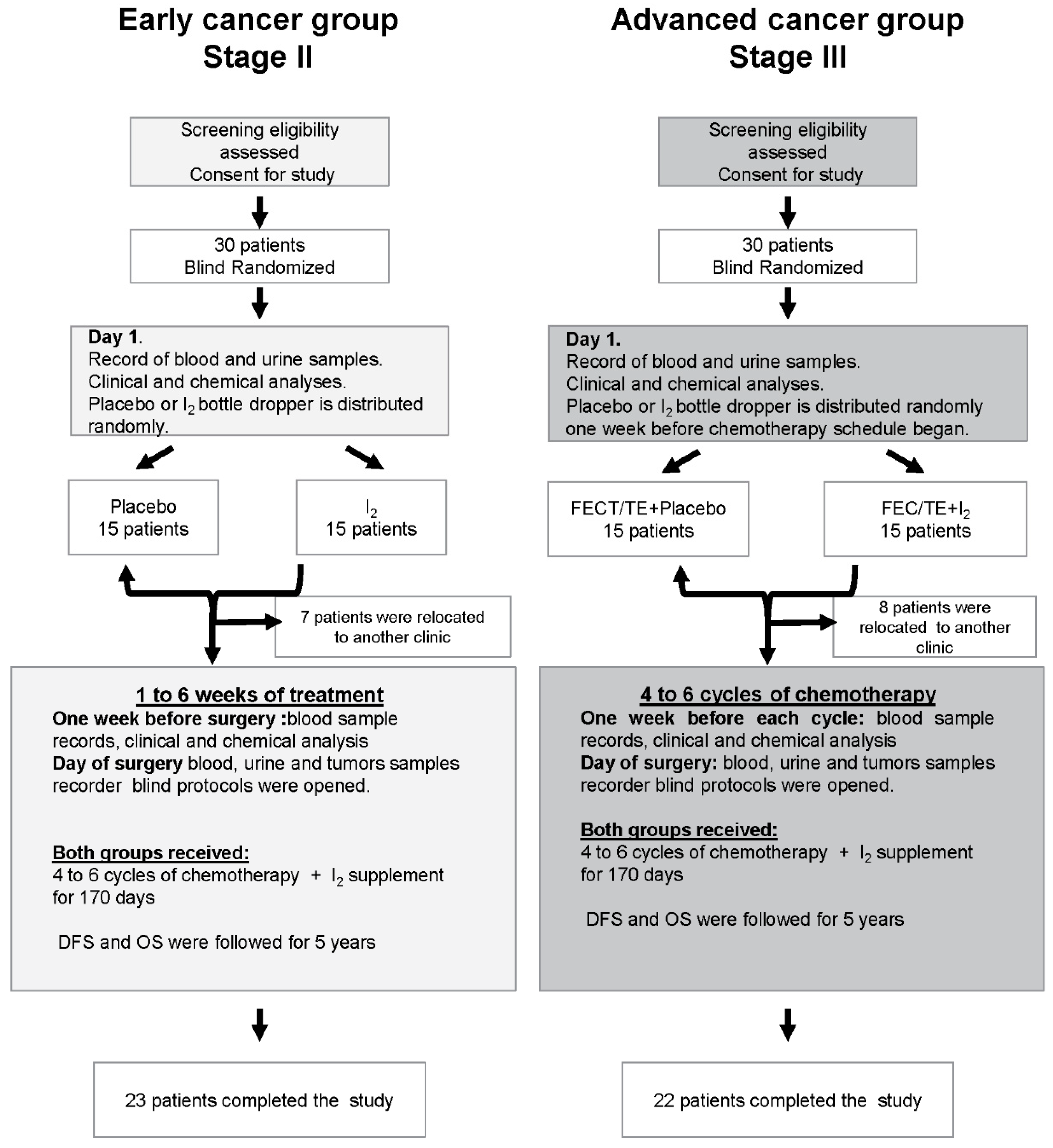
2.1. Study Design
2.2. Adverse Reactions
2.3. Evaluation of Treatment Efficacy
2.4. Follow-Up
2.5. Iodine and Thyroid Hormones
2.6. Molecular Markers Expression
2.7. Immunohistochemistry
2.8. Western Blotting
2.9. RNA-Seq and Transcriptomic Analysis
2.10. Statistics Analysis
2.11. Study Approval
3. Results
3.1. Patients
3.2. Toxicity and Safety
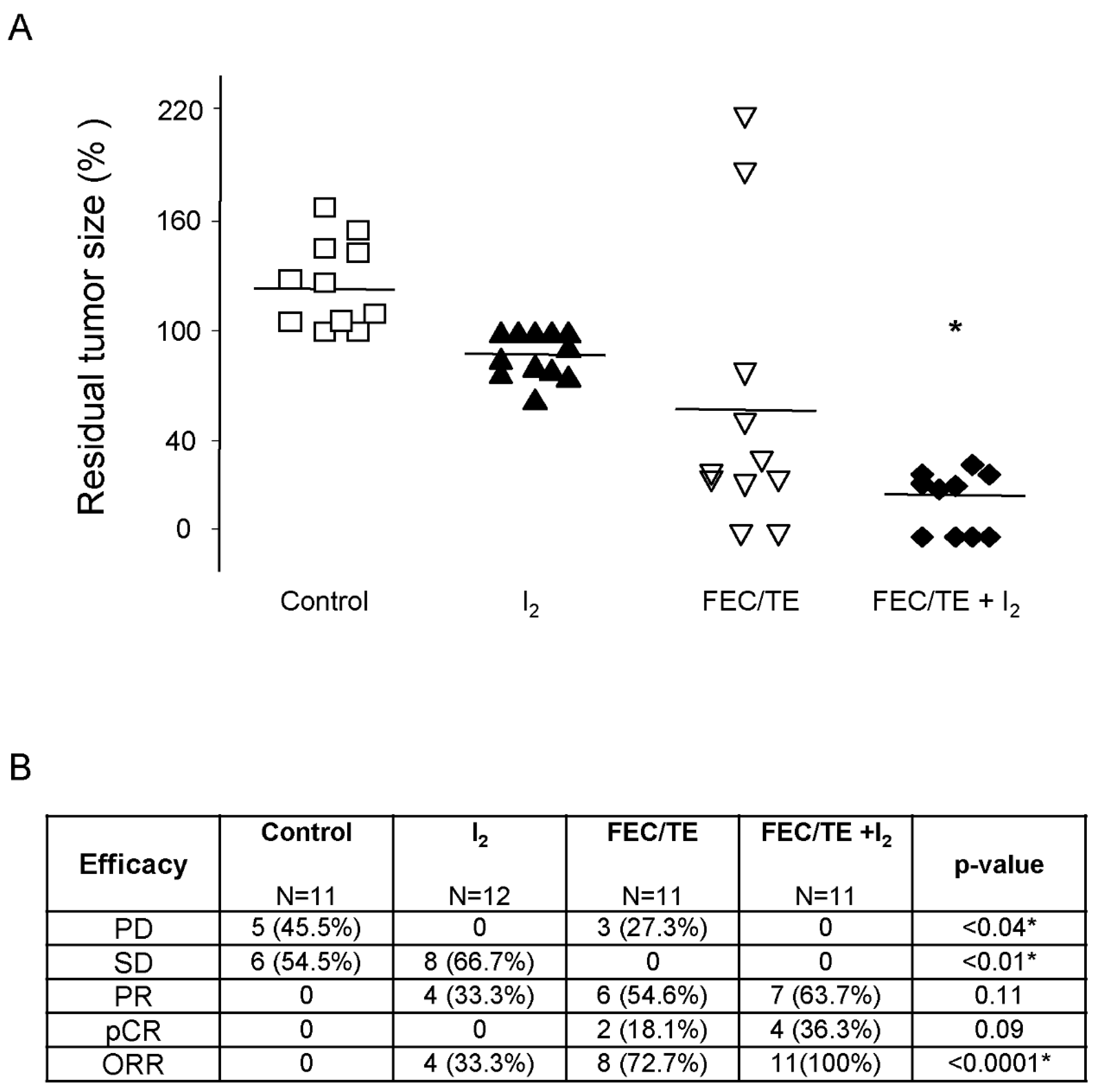
3.3. Tumoral Response
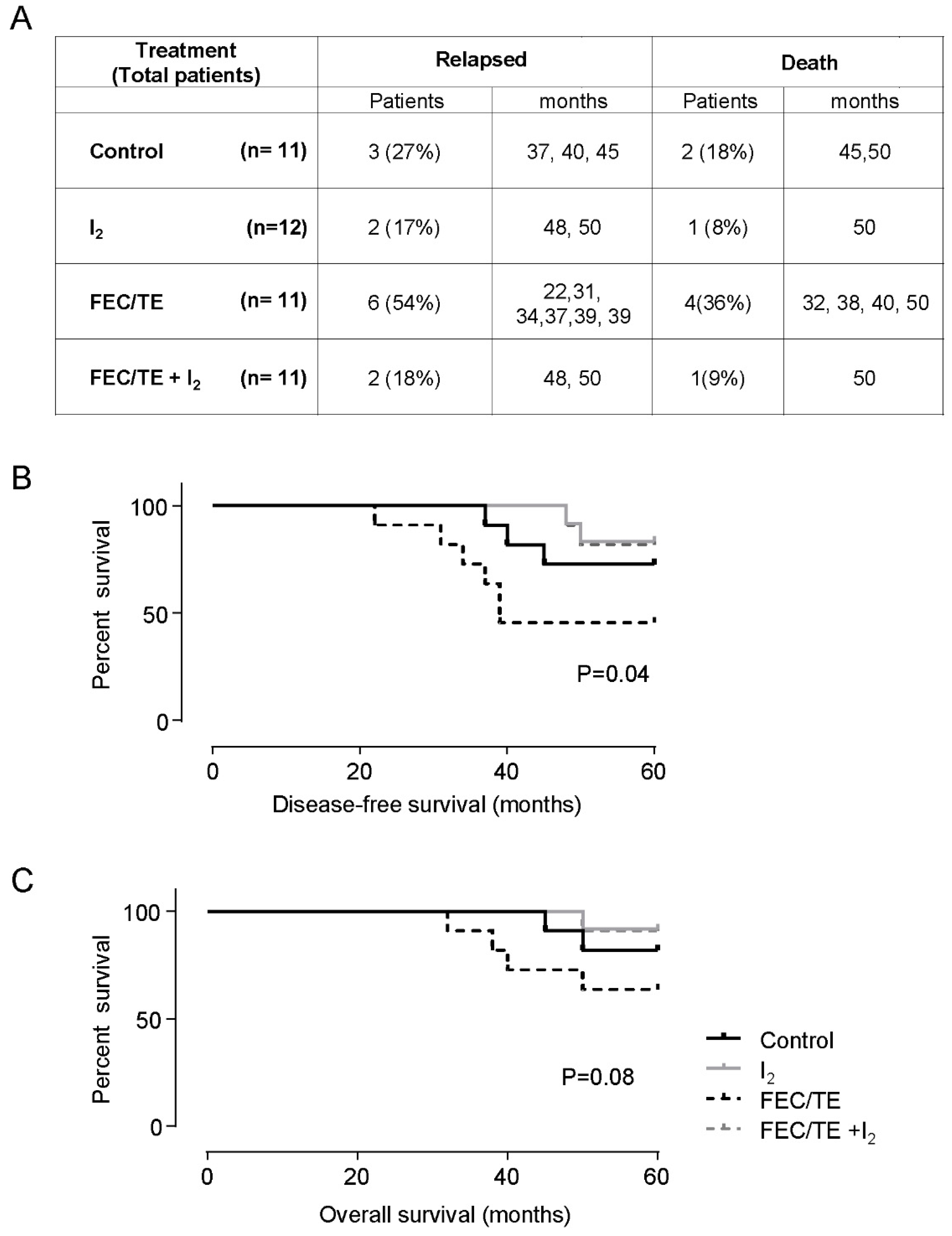
3.4. Follow Up
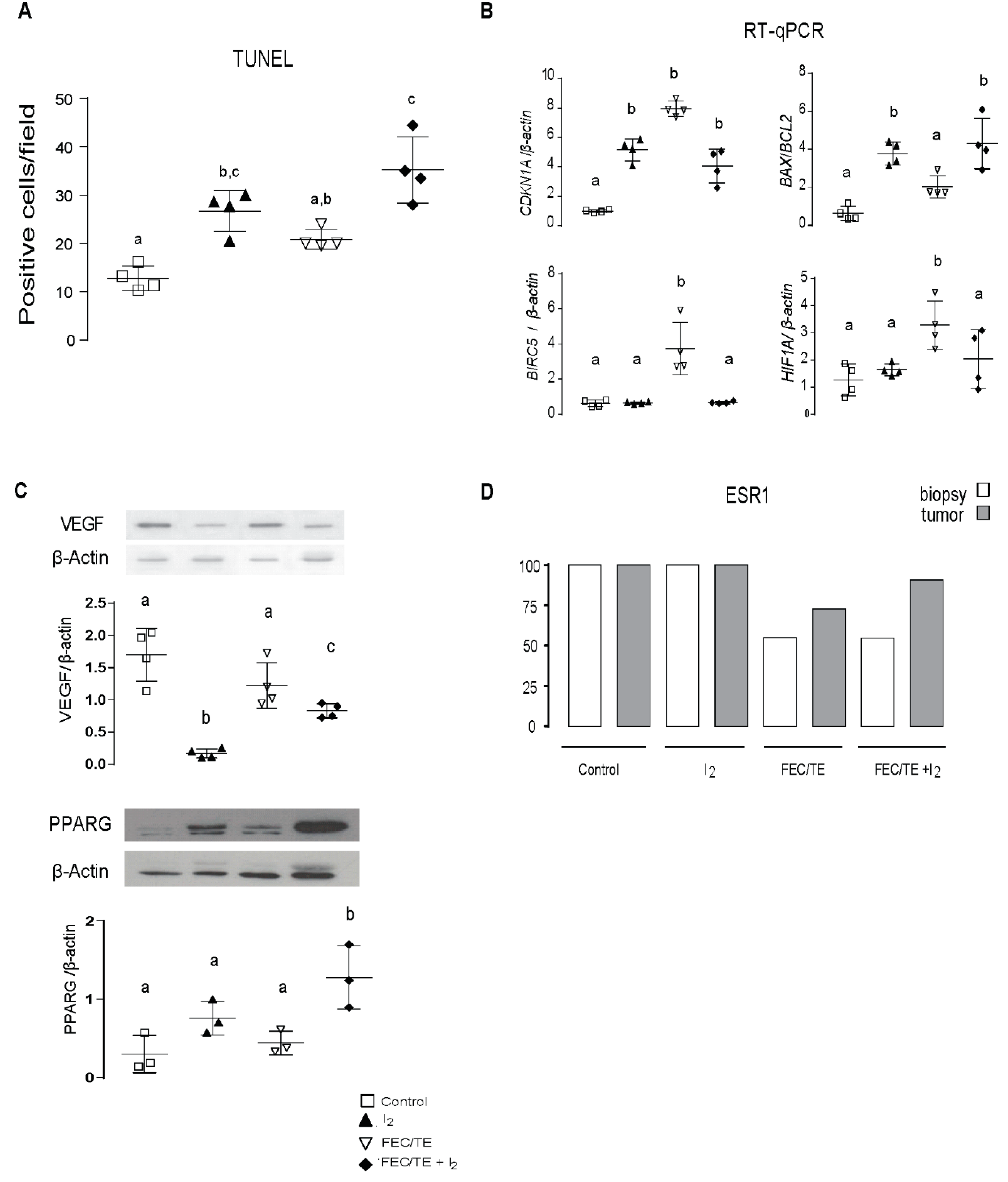
3.5. Molecular Tumoral Analysis
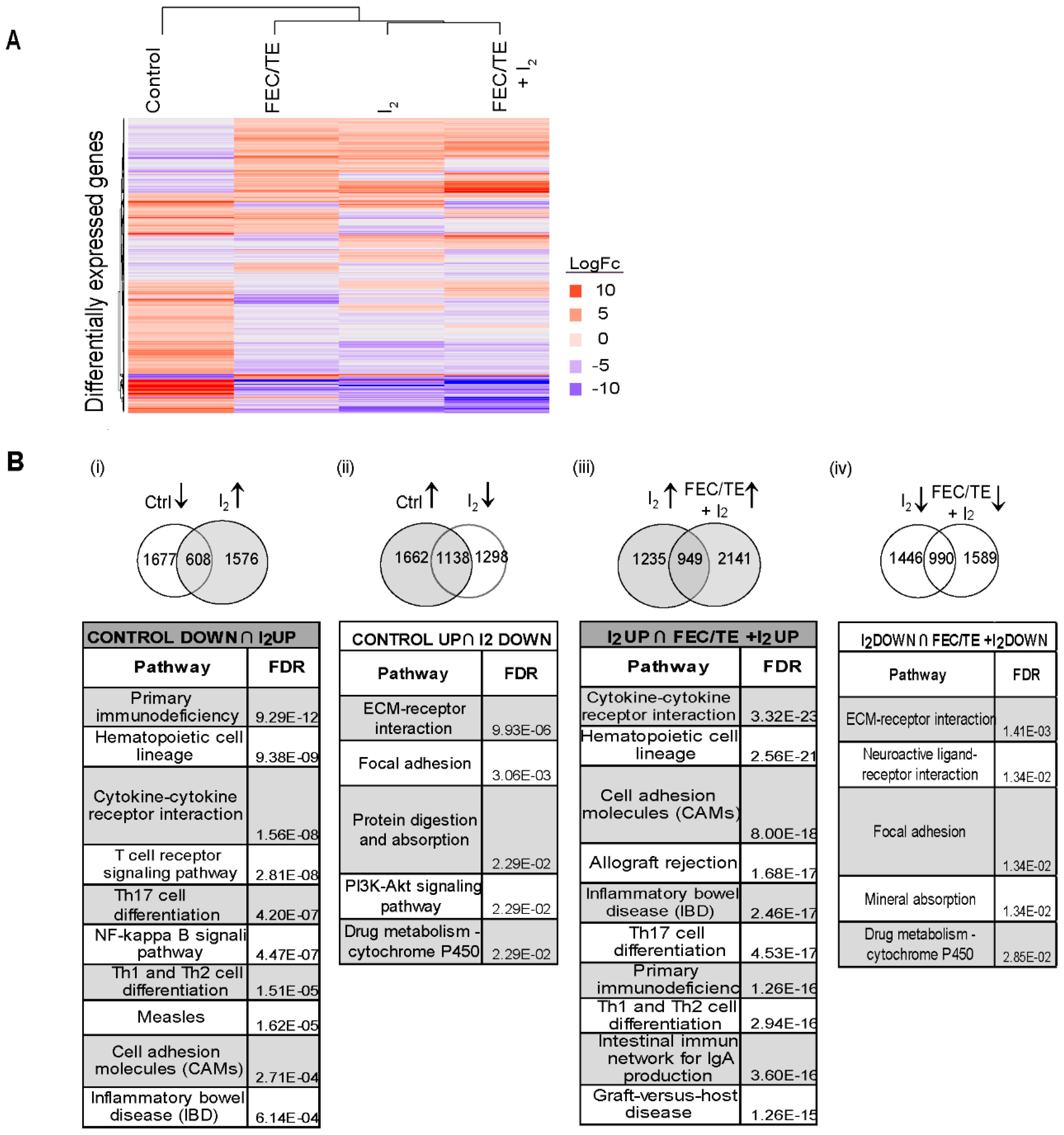
3.6. Effect of I2 and/or FEC/TE + I2 Treatment on General Tumor Gene Expression
3.7. Analysis of EMT-Related Gene and Protein Expression after I2 Treatment
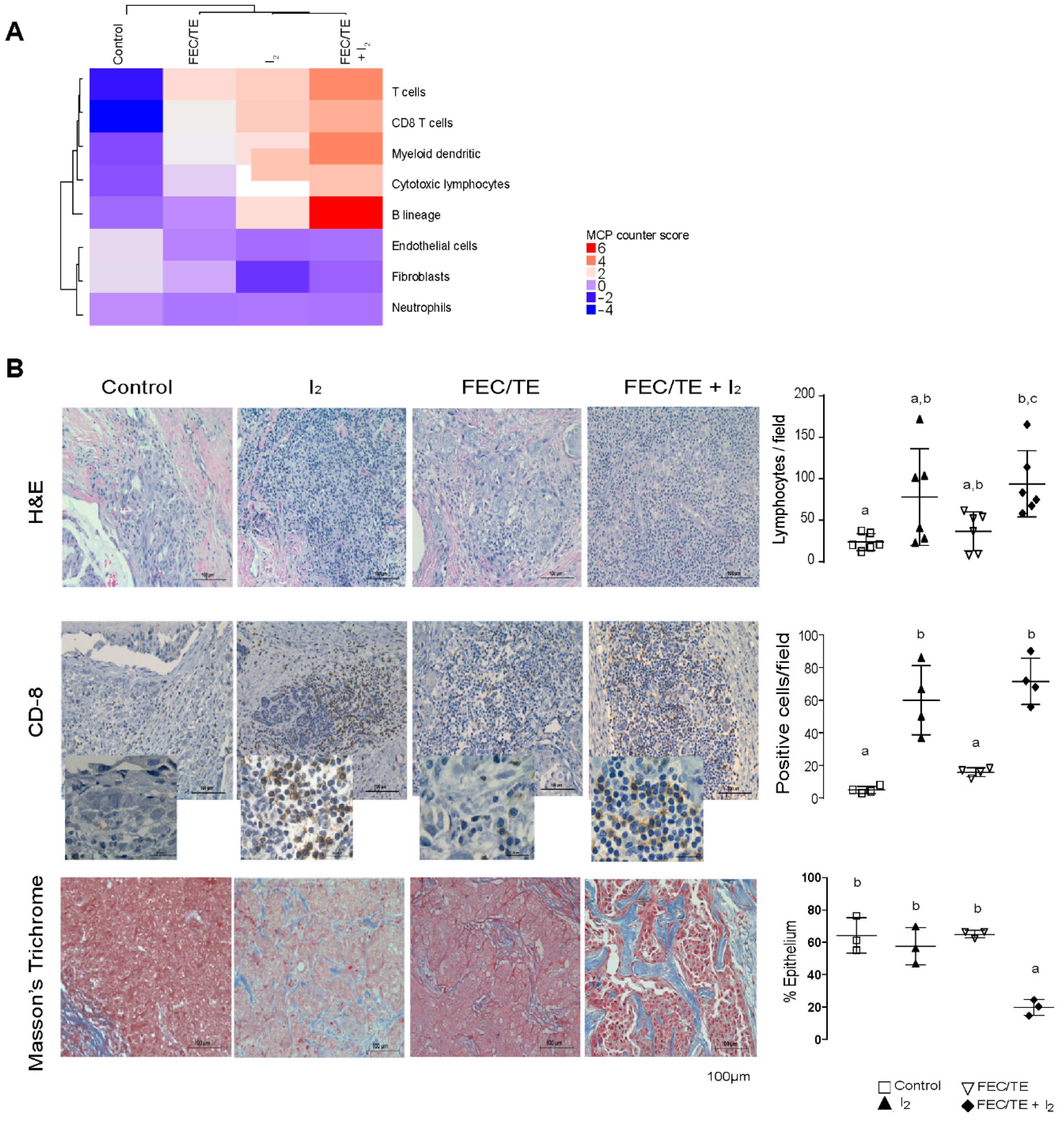
3.8. Immune Response Activation after I2 Treatment
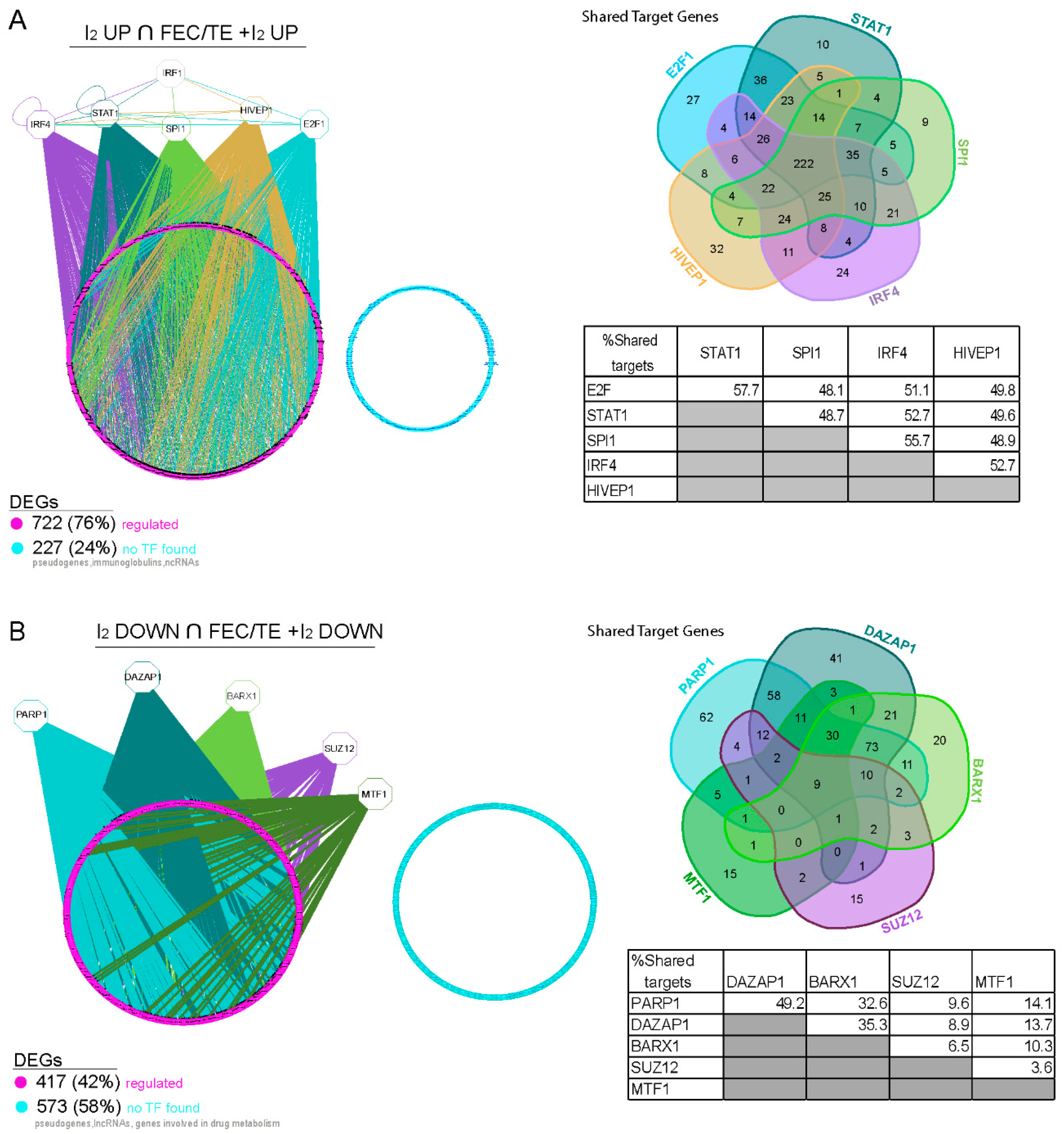
3.9. Transcriptional Regulation of Common Genes in the I2- and FEC/TE+I2-Treated Tumors
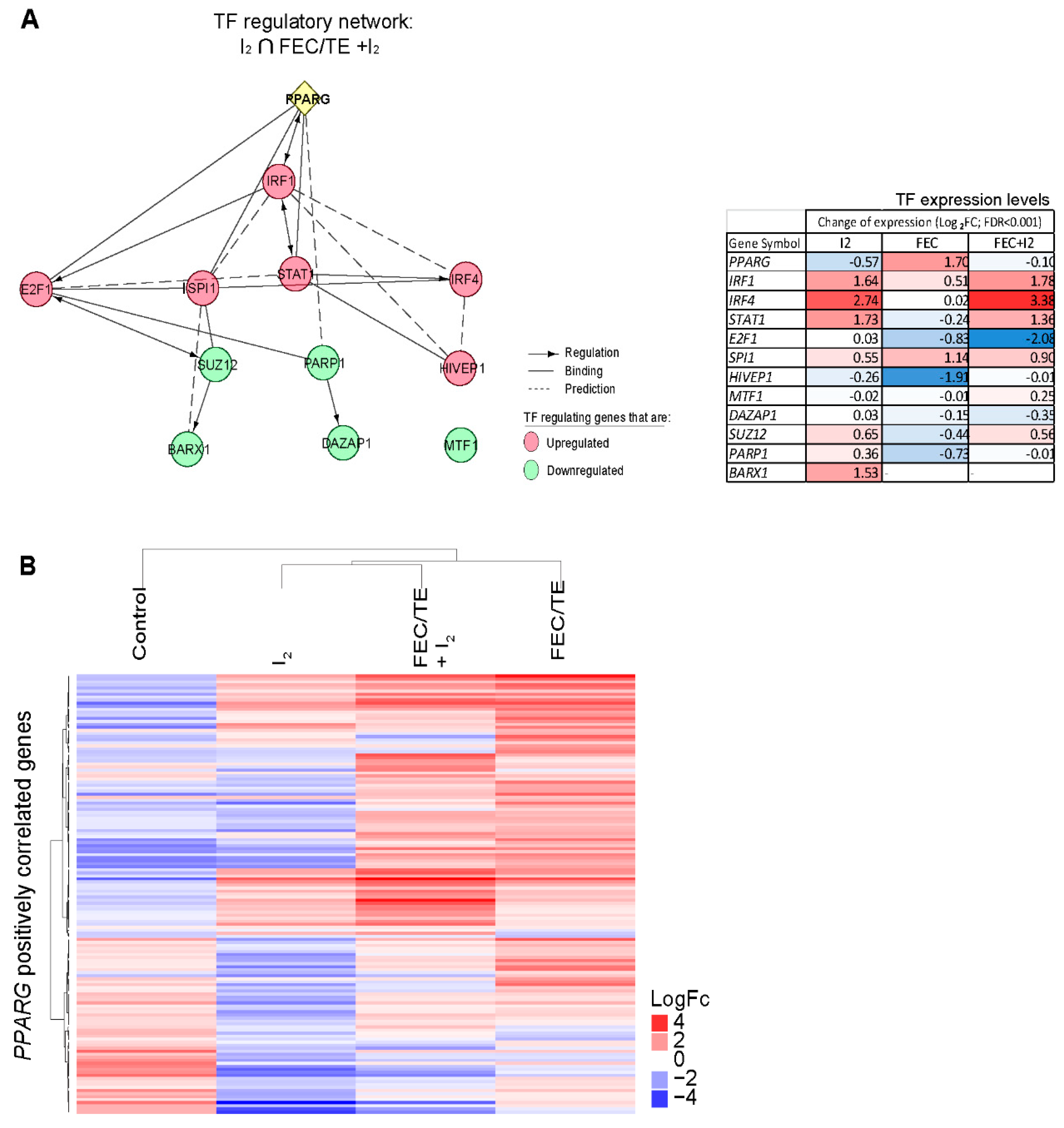
3.10. Role of PPARG in the Observed I2-Mediated Changes
4. Discussion
5. Conclusions
6. Patents
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- National Cancer Institute at the National Institutes of Health. Breast Cancer. Available online: http://www.cancer.gov/cancertopics/types/breast (accessed on 18 June 2019).
- Lu, W.; Kang, Y. Cell lineage determinants as regulators of breast cancer metastasis. Cancer Metastasis Rev. 2016, 35, 631–644. [Google Scholar] [CrossRef] [PubMed]
- Janni, W.; Harbeck, N.; Rack, B.; Augustin, D.; Jueckstock, J.; Wischnik, A.; Annecke, K.; Scholz, C.; Huober, J.; Zwingers, T.; et al. Randomised phase III trial of FEC120 vs EC-docetaxel in patients with high-risk node-positive primary breast cancer: Final survival analysis of the ADEBAR study. Br. J. Cancer 2016, 114, 863–871. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sawyer, D.B.; Peng, X.; Chen, B.; Pentassuglia, L.; Lim, C.C. Mechanisms of anthracycline cardiac injury: Can we identify strategies for cardioprotection? Prog. Cardiovasc. Dis. 2010, 53, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Ghent, W.R.; Eskin, B.A.; Low, D.A.; Hill, L.P. Iodine replacement in fibrocystic disease of the breast. Can. J. Surg. 1993, 36, 453–460. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kessler, J.H. The effect of supraphysiologic levels of iodine on patients with cyclic mastalgia. Breast J. 2004, 10, 328–336. [Google Scholar] [CrossRef] [PubMed]
- García-Solís, P.; Alfaro, Y.; Anguiano, B.; Delgado, G.; Guzman, R.C.; Nandi, S.; Díaz-Muñoz, M.; Vázquez-Martínez, O.; Aceves, C. Inhibition of N-methyl-N-nitrosourea-induced mammary carcinogenesis by molecular iodine (I2) but not by iodide (I−) treatment: Evidence that I2 prevents cancer promotion. Mol. Cell Endocrinol. 2005, 236, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Aceves, C.; Anguiano, B.; Delgado, G. The extrathyronine actions of iodine as antioxidant, apoptotic, and differentiation factor in various tissues. Thyroid 2013, 23, 938–946. [Google Scholar] [CrossRef]
- Rosner, H.; Torremante, P.; Moller, W.; Gartner, R. Antiproliferative/cytotoxic activity of molecular iodine and iodolactones in various human carcinoma cell lines. No interfering with EGF-signaling, but evidence for apoptosis. Exp. Clin. Endocrinol. Diabetes 2010, 118, 410–419. [Google Scholar] [CrossRef]
- Alfaro, Y.; Delgado, G.; Cárabez, A.; Anguiano, B.; Aceves, C. Iodine and doxorubicin, a good combination for mammary cancer treatment: Antineoplastic adjuvancy, chemoresistance inhibition, and cardioprotection. Mol. Cancer 2013, 12, 45–55. [Google Scholar] [CrossRef]
- Zambrano-Estrada, X.; Landaverde-Quiroz, B.; Dueñas-Bocanegra, A.A.; De Paz-Campos, M.A.; Hernández-Alberto, G.; Solorio-Perusquia, B.; Trejo-Mandujano, M.; Pérez-Guerrero, L.; Delgado-González, E.; Anguiano, B.; et al. Molecular iodine/doxorubicin neoadjuvant treatment impair invasive capacity and attenuate side effect in canine mammary cancer. BMC Vet. Res. 2018, 14, 87–100. [Google Scholar] [CrossRef]
- Bontempo, A.; Ugalde-Villanueva, B.; Delgado-González, E.; Rodríguez, A.L.; Aceves, C. Molecular iodine impairs chemoresistance mechanisms, enhances doxorubicin retention and induces downregulation of CD44+/CD24+ and E-Cadherin+/vimentin+ subpopulations in MCF-7 cells resistant to low doses of doxorubicin. Oncol. Rep. 2017, 38, 867–876. [Google Scholar] [CrossRef] [PubMed]
- Common Terminology Criteria for Adverse Events (CTCAE). Updated June 14, 2010 (v4.03). Available online: https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htmQuickReference_5x7.pdf (accessed on 18 June 2019).
- Therasse, P.; Arbuck, S.G.; Eisenhauer, E.A.; Wanders, J.; Kaplan, R.S.; Rubinstein, L.; Verweij, J.; Van Glabbeke, M.; Van Oosterom, A.T.; Christian, M.C.; et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J. Natl. Cancer Inst. 2000, 92, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Delgado, G.; Muñoz-Torres, C.; Orozco-Esquivel, T.; Anguiano, B.; Aceves, C. Total iodine quantification in fluids and tissues from iodine-or iodide-supplemented rats by ion chromatography following microwave-assisted digestion. Thyroid 2015, 25, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Nava-Villalba, M.; Nuñez-Anita, R.E.; Bontempo, A.; Aceves, C. Activation of peroxisome proliferator-activated receptor gamma is crucial for antitumoral effects of 6-iodolactone. Mol. Cancer 2015, 14, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Immunehistochemistry and Transcriptomic Analysis of Iodine and Breast Cancer. Available online: https://dx.doi.org/10.17504/protocols.io.t7gerjw (accessed on 18 June 2019).
- Chen, J.; Bardes, E.E.; Aronow, B.J.; Jegga, A.G. ToppGene Suite for gene list enrichment analysis and candidate gene prioritization. Nucleic Acids Res. 2009, 37, W305–W311. [Google Scholar] [CrossRef] [PubMed]
- Ksiązkiewicz, M.; Markiewicz, A.; Zaczek, A.J. Epithelial-mesenchymal transition: A hallmark in metastasis formation linking circulating tumor cells and cancer stem cells. Pathobiology 2012, 79, 195–208. [Google Scholar] [CrossRef] [PubMed]
- Becht, E.; Giraldo, N.A.; Lacroix, L.; Buttard, B.; Elarouci, N.; Petitprez, F.; Selves, J.; Laurent-Puig, P.; Sautès-Fridman, C.; Fridman, W.H.; et al. Estimating the population abundance of tissue-infiltrating immune and stromal cell populations using gene expression. Genome Biol. 2016, 17, 218–238. [Google Scholar] [CrossRef]
- Janky, R.; Verfaillie, A.; Imrichová, H.; Van de Sande, B.; Standaert, L.; Christiaens, V.; Hulselmans, G.; Herten, K.; Naval Sanchez, M.; Potier, D.; et al. iRegulon: From a gene list to a gene regulatory network using large motif and track collections. PLoS Comput Biol. 2014, 10, e1003731. [Google Scholar] [CrossRef]
- Wu, G.; Feng, X.; Stein, L. A human functional protein interaction network and its application to cancer data analysis. Genome Biol. 2010, 11, R53. [Google Scholar] [CrossRef]
- Chen, H.; Sharp, B.M. Content-rich biological network constructed by mining PubMed abstracts. BMC Bioinform. 2004, 5, 147. [Google Scholar] [CrossRef]
- Pereira, B.; Chin, S.F.; Rueda, O.M.; Vollan, H.K.; Provenzano, E.; Bardwell, H.A.; Pugh, M.; Jones, L.; Russell, R.; Sammut, S.J.; et al. The somatic mutation profiles of 2,433 breast cancers refines their genomic and transcriptomic landscapes. Nat. Commun. 2016, 7, 11479. [Google Scholar] [CrossRef] [PubMed]
- Meischl, C.; Buermans, H.P.; Hazes, T.; Zuidwijk, M.J.; Musters, R.J.; Boer, C.; van Lingen, A.; Simonides, W.S.; Blankenstein, M.A.; Dupuy, C.; et al. H9c2 cardiomyoblasts produce thyroid hormone. Am. J. Physiol. Cell Physiol. 2008, 294, 1227–1233. [Google Scholar] [CrossRef] [PubMed]
- Quintero-García, M.; Delgado-González, E.; Sánchez-Tusie, A.; Vázquez, M.; Aceves, C.; Anguiano, B. Iodine prevents the increase of testosterone-induced oxidative stress in a model of rat prostatic hyperplasia. Free Radic. Biol. Med. 2018, 115, 298–308. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Wei, W.; Jiang, Y.; Li, Q.; Huang, Q.; Yang, H.; Liu, J. Comparison of the efficacy and survival analysis of neoadjuvant chemotherapy for Her-2-positive breast cancer. Drug Des. Devel. Ther. 2018, 12, 3085–3093. [Google Scholar] [CrossRef] [PubMed]
- Gartner, R.; Rank, P.; Ander, B. The role of iodine and delta-iodolactone in growth and apoptosis of malignant thyroid epithelial cells and breast cancer cells. Horm. Athens 2010, 9, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Namvaran, F.; Azarpira, N.; Rahimi-Moghaddam, P.; Dabbaghmanesh, M.H. Polymorphism of peroxisome proliferator-activated receptor γ (PPARγ) Pro12Ala in the Iranian population: Relation with insulin resistance and response to treatment with pioglitazone in type 2 diabetes. Eur. J. Pharmacol. 2011, 671, 1–6. [Google Scholar] [CrossRef]
- Reka, A.K.; Goswami, M.T.; Krishnapuram, R.; Standiford, T.J.; Keshamouni, V.G. Molecular cross-regulation between PPAR-γ and other signaling pathways: Implications for lung cancer therapy. Lung. Cancer 2011, 72, 154–159. [Google Scholar] [CrossRef]
- Burstein, H.J.; Demetri, G.D.; Mueller, E.; Sarraf, P.; Spiegelman, B.M.; Winer, E.P. Use of the peroxisome proliferator-activated receptor (PPAR) gamma ligand troglitazone as treatment for refractory breast cancer: A phase II study. Breast Cancer Res. Treat 2003, 79, 391–397. [Google Scholar] [CrossRef]
- Girnun, G.D.; Chen, L.; Silvaggi, J.; Drapkin, R.; Chirieac, L.R.; Padera, R.F.; Upadhyay, R.; Vafai, S.B.; Weissleder, R.; Mahmood, U.; et al. Regression of drug-resistant lung cancer by the combination of rosiglitazone and carboplatin. Clin. Cancer Res. 2008, 14, 6478–6486. [Google Scholar] [CrossRef]
- Bräutigam, K.; Biernath-Wüpping, J.M.; Bauerschlag, D.O.; von Kaisenberg, C.S.; Jonat, W.; Maass, N.; Arnold, N.; Meinhold-Heerlein, I. Combined treatment with TRAIL and PPARγ ligands overcomes chemoresistance of ovarian cancer cell lines. J. Cancer. Res. Clin. Oncol. 2011, 137, 875–886. [Google Scholar] [CrossRef]
- Tripathi, R.; Singh, P.; Singh, A.; Chagtoo, M.; Khan, S.; Tiwari, S.; Agarwal, G.; Meeran, S.M.; Godbole, M.M. Zoledronate and molecular iodine cause synergistic cell death in triple negative breast cancer through endoplasmic reticulum stress. Nutr. Cancer 2016, 68, 679–688. [Google Scholar] [CrossRef] [PubMed]
- Kim, P.K.; Armstrong, M.; Liu, Y.; Yan, P.; Bucher, B.; Zuckerbraun, B.S.; Gambotto, A.; Billiar, T.R.; Yim, J.H. IRF-1 expression induces apoptosis and inhibits tumor growth in mouse mammary cancer cells in vitro and in vivo. Oncogene 2004, 23, 1125–1135. [Google Scholar] [CrossRef] [PubMed]
- Buccione, C.; Fragale, A.; Polverino, F.; Ziccheddu, G.; Aricò, E.; Belardelli, F.; Proietti, E.; Battistini, A.; Moschella, F. Role of interferon regulatory factor 1 in governing Treg depletion, Th1 polarization, inflammasome activation and antitumor efficacy of cyclophosphamide. Int. J. Cancer. 2018, 142, 976–987. [Google Scholar] [CrossRef] [PubMed]
- Mendieta, I.; Nuñez-Anita, R.E.; Nava-Villalba, M.; Zambrano-Estrada, X.; Delgado-González, E.; Anguiano, B.; Aceves, C. Molecular iodine exerts antineoplastic effects by diminishing proliferation and invasive potential and activating the immune response in mammary cancer xenografts. BMC Cancer 2019, 19, 261–268. [Google Scholar] [CrossRef] [PubMed]
- Venturi, S. Evolutionary significance of iodine. Curr. Chem. Biol. 2011, 5, 155–162. [Google Scholar]
- Bilal, M.Y.; Dambaeva, S.; Kwak-Kim, J.; Gilman-Sachs, A.; Beaman, K.D. A role for iodide and thyroglobulin in modulating the function of human immune cells. Front. Immunol. 2017, 8, 1573–1587. [Google Scholar] [CrossRef]
- Costa, R.O.; Macedo, P.M.; Carvalhal, A.; Bernardes-Engemann, A.R. Use of potassium iodide in dermatology: Updates on an old drug. Bras. Derm. 2013, 88, 396–402. [Google Scholar] [CrossRef]
- Miyachi, Y.; Niwa, Y. Effects of potassium iodide, colchicine and dapsone on the generation of polymorphonuclear leukocyte-derived oxygen intermediates. Br. J. Derm. 1982, 107, 209–214. [Google Scholar] [CrossRef]
- Honma, K.; Saga, K.; Onodera, H.; Takahashi, M. Potassium iodide inhibits neutrophil chemotaxis. Acta Derm. Venereol. 1990, 70, 247–249. [Google Scholar]
- Moore, K.; Thomas, A.; Harding, K.G. Iodine released from the wound dressing iodosorb modulates the secretion of cytokines by human macrophages responding to bacterial lipopolysaccharide. Int. J. Biochem. Cell Biol. 1997, 29, 163–171. [Google Scholar] [CrossRef]
- Zhao, D.; Lim, C.P.; Miyanaga, K.; Tanji, Y. Iodine from bacterial iodide oxidization by Roseovarius spp. inhibits the growth of other bacteria. Appl. Microbiol. Biotechnol. 2013, 97, 2173–2182. [Google Scholar] [CrossRef] [PubMed]
| Adverse Events | Control/Placebo + FEC/TE + I2 N = 11 | I2 + FEC/TE + I2N = 12 | FEC/TE + Placebo+FEC/TE + I2 N = 11 | FEC/TE + I2 + FEC/TE + I2 N = 11 | P-Value |
|---|---|---|---|---|---|
| 1 grade neutropenia | 7 (63.6%) | 5 (41.6%) | 5 (45.5%) | 5 (45.5%) | 0.72 |
| 2–3 grade neutropenia | 0 | 0 | 4 (36.4%) | 0 | <0.003 * |
| 1 grade nausea, vomiting and diarrhea | 2 (18.1%) | 1 (8.3%) | 9 (81.2%) | 3 (27.3%) | <0.002 * |
| 2–3 grade nausea, vomiting and diarrhea | 0 | 0 | 2 (18.1%) | 0 | 0.090 |
| Myalgia | 8 (72.7%) | 9 (75%) | 9 (81.8%) | 9 (75%) | 0.99 |
| Hand-foot syndrome | 0 | 0 | 3 (27.3%) | 0 | <0.01 * |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moreno-Vega, A.; Vega-Riveroll, L.; Ayala, T.; Peralta, G.; Torres-Martel, J.M.; Rojas, J.; Mondragón, P.; Domínguez, A.; De Obaldía, R.; Avecilla-Guerrero, C.; et al. Adjuvant Effect of Molecular Iodine in Conventional Chemotherapy for Breast Cancer. Randomized Pilot Study. Nutrients 2019, 11, 1623. https://doi.org/10.3390/nu11071623
Moreno-Vega A, Vega-Riveroll L, Ayala T, Peralta G, Torres-Martel JM, Rojas J, Mondragón P, Domínguez A, De Obaldía R, Avecilla-Guerrero C, et al. Adjuvant Effect of Molecular Iodine in Conventional Chemotherapy for Breast Cancer. Randomized Pilot Study. Nutrients. 2019; 11(7):1623. https://doi.org/10.3390/nu11071623
Chicago/Turabian StyleMoreno-Vega, Aura, Laura Vega-Riveroll, Tonatiuh Ayala, Guillermo Peralta, José Miguel Torres-Martel, Joel Rojas, Perla Mondragón, Adriana Domínguez, Rodrigo De Obaldía, Carlos Avecilla-Guerrero, and et al. 2019. "Adjuvant Effect of Molecular Iodine in Conventional Chemotherapy for Breast Cancer. Randomized Pilot Study" Nutrients 11, no. 7: 1623. https://doi.org/10.3390/nu11071623
APA StyleMoreno-Vega, A., Vega-Riveroll, L., Ayala, T., Peralta, G., Torres-Martel, J. M., Rojas, J., Mondragón, P., Domínguez, A., De Obaldía, R., Avecilla-Guerrero, C., Anguiano, B., Delgado-González, E., Zambrano-Estrada, X., Cuenca-Micó, O., De La Puente Flores, O., Varela-Echavarría, A., & Aceves, C. (2019). Adjuvant Effect of Molecular Iodine in Conventional Chemotherapy for Breast Cancer. Randomized Pilot Study. Nutrients, 11(7), 1623. https://doi.org/10.3390/nu11071623





