Western Diet-Induced Metabolic Alterations Affect Circulating Markers of Liver Function before the Development of Steatosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Animal Model and Treatment
2.2. Reagents
2.3. Assessment of Liver Function and Immunohistochemical Study
2.4. Quantification of Gene Expression by qRT-PCR
2.5. Western Blot Analyses
2.6. Immunofluorescence Coupled with Confocal Microscopy
2.7. Bile Acid Extraction and LC-MS/MS Analysis
2.8. Statistical Analyses
3. Results
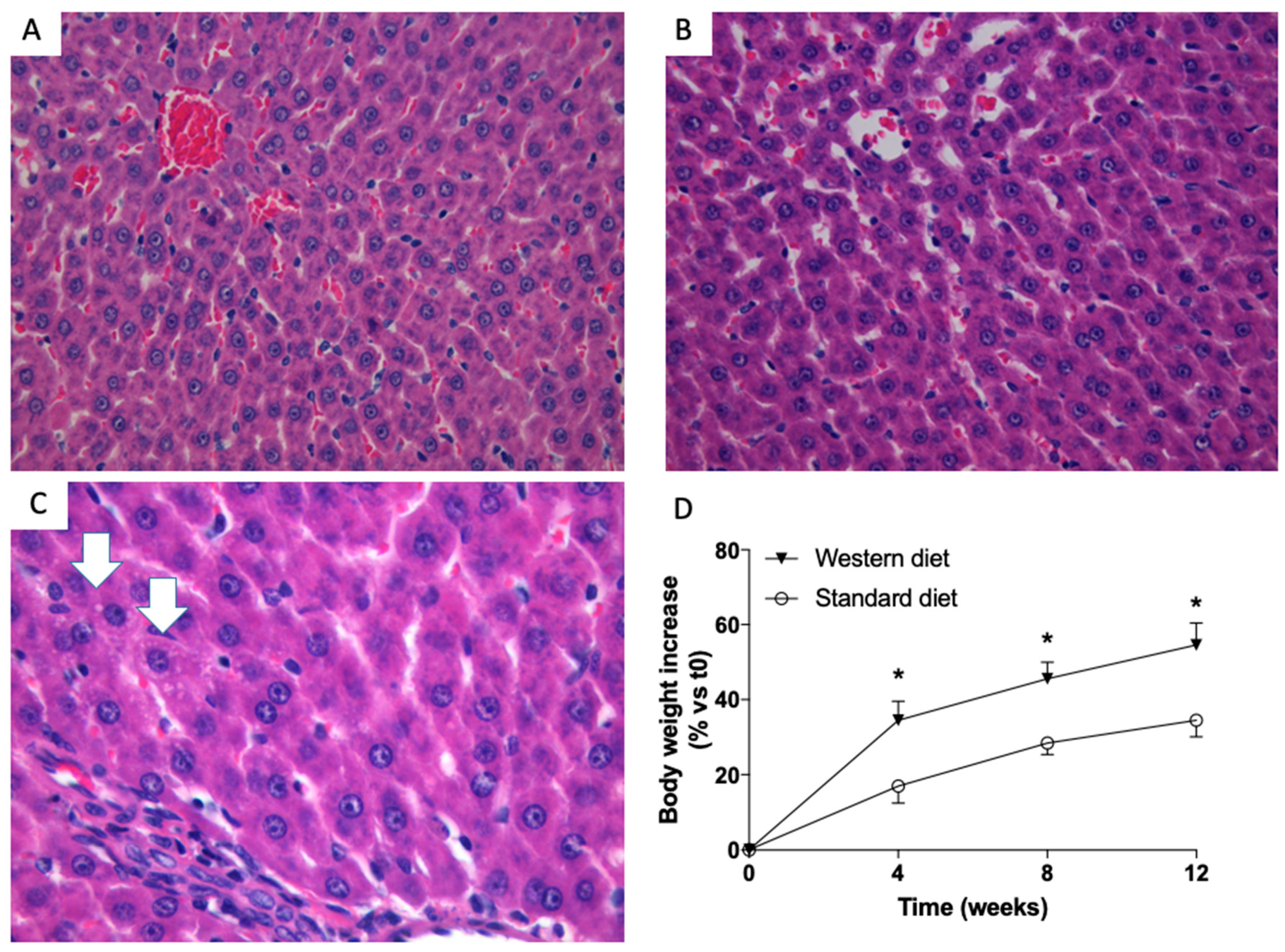
3.1. Effects of Western-Type Diet on Liver Histology, Body Weight and Plasma Biochemistry
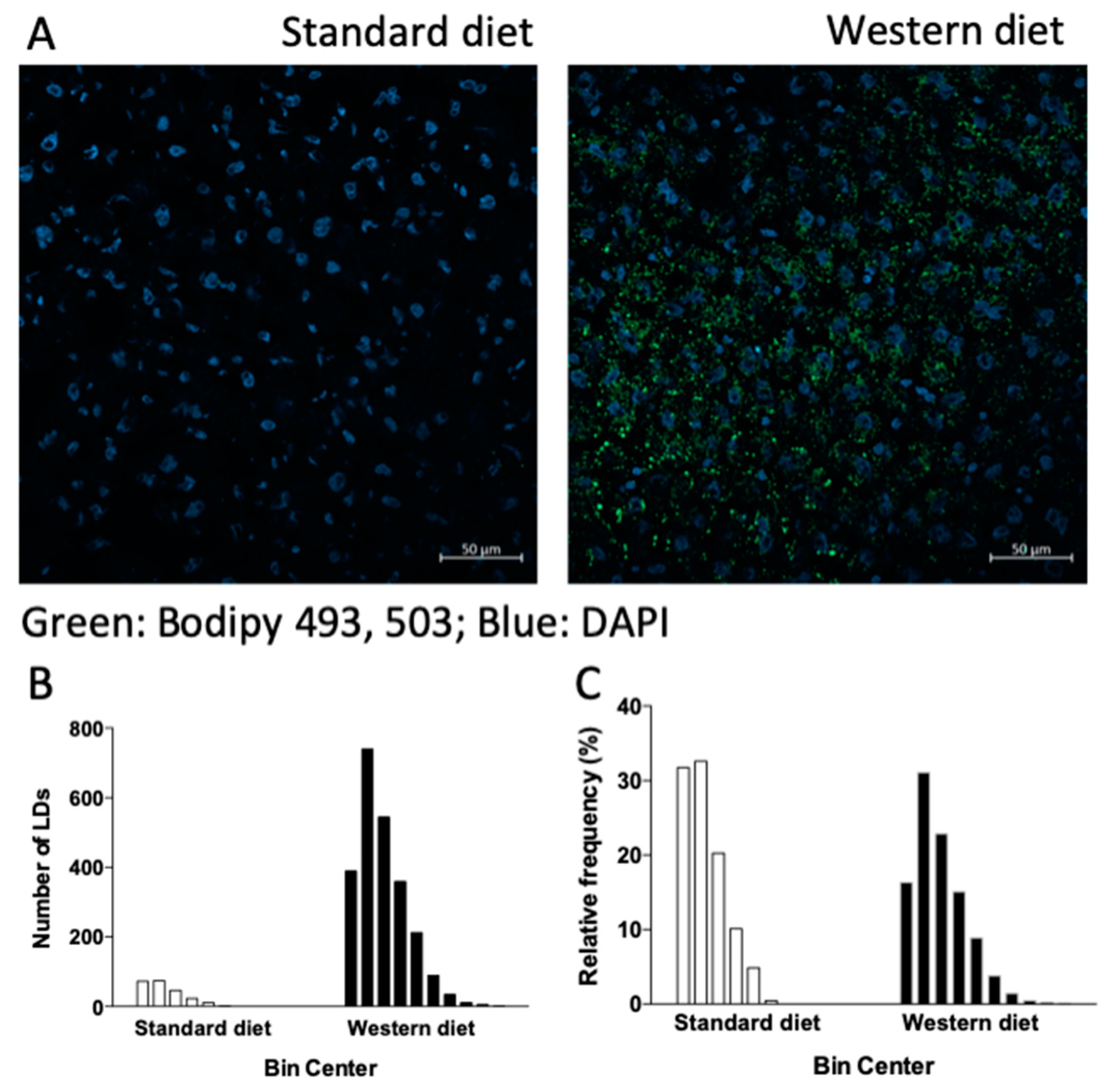
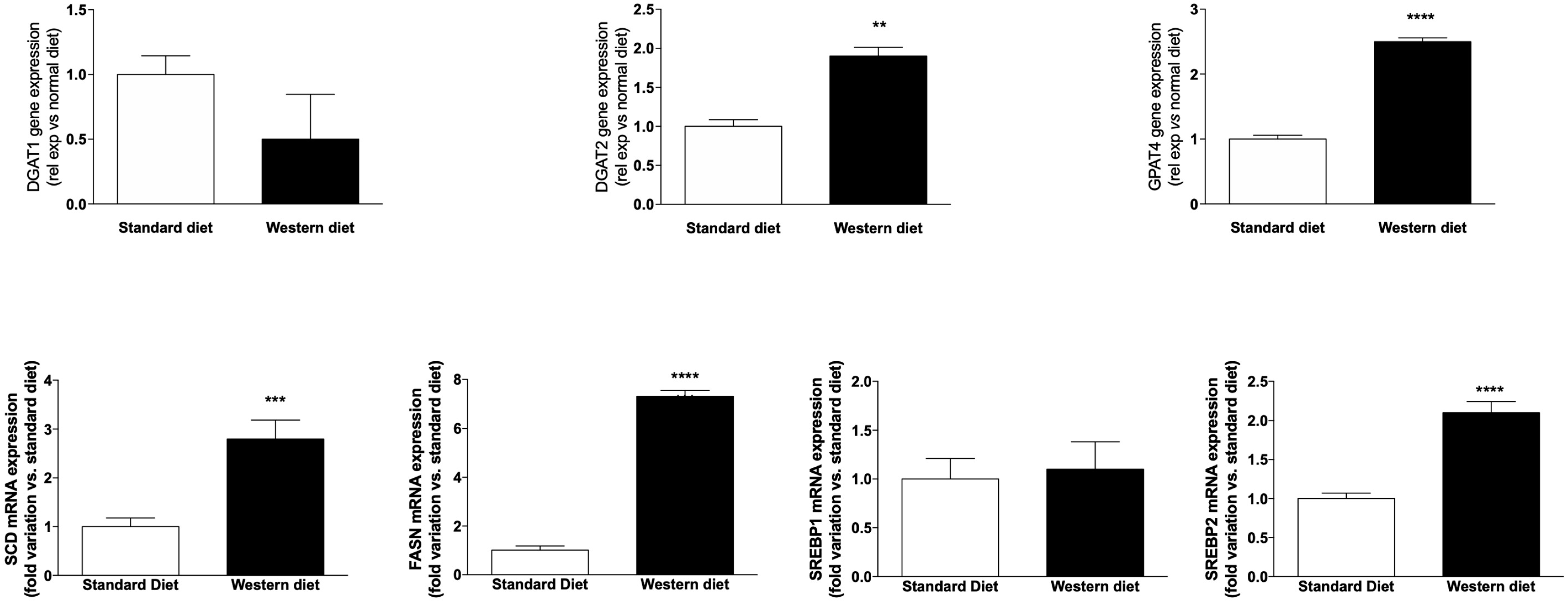
3.2. Effect of a Western-Type Diet on Hepatic Lipid Droplets and Hepatic Liver Metabolism
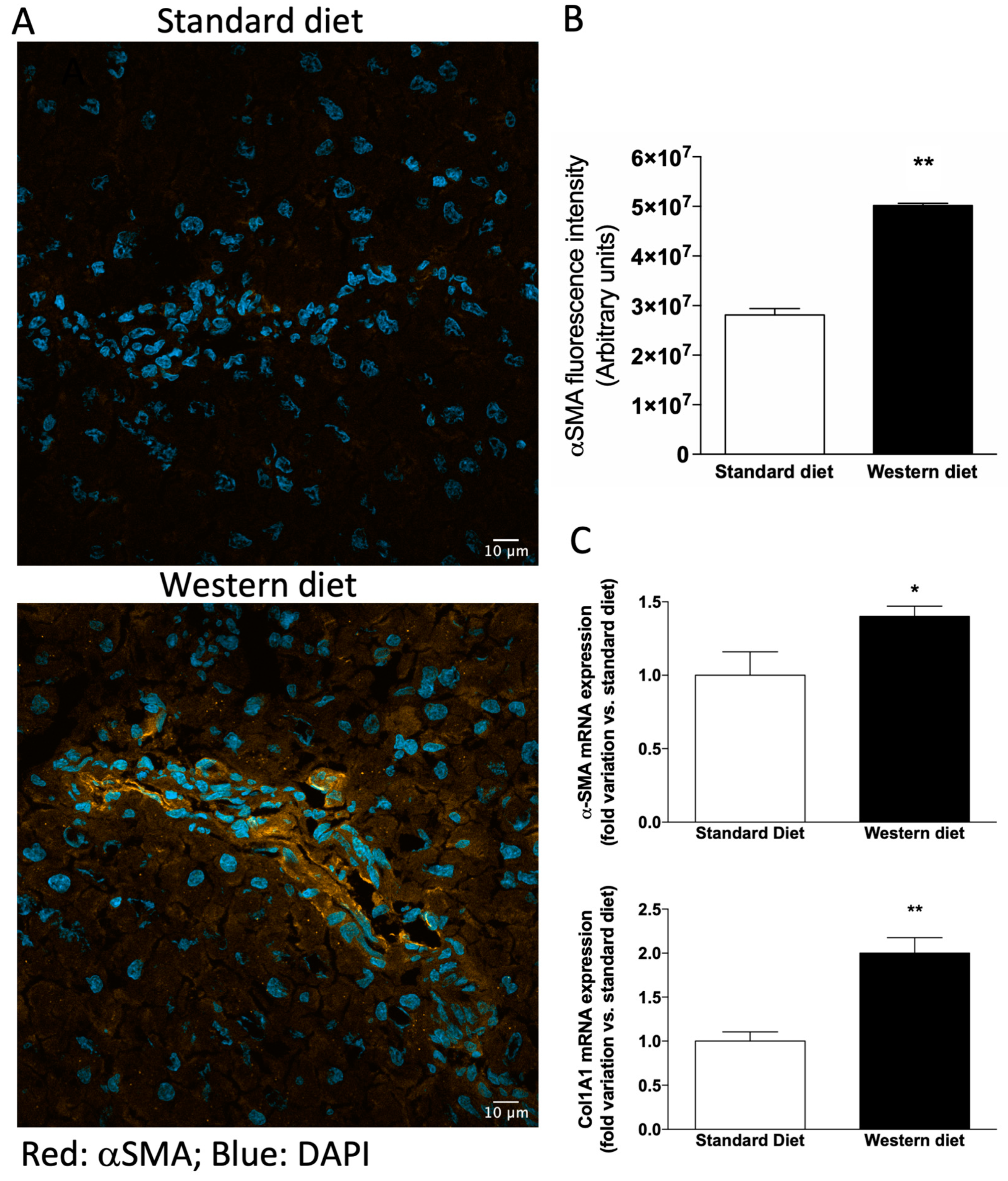
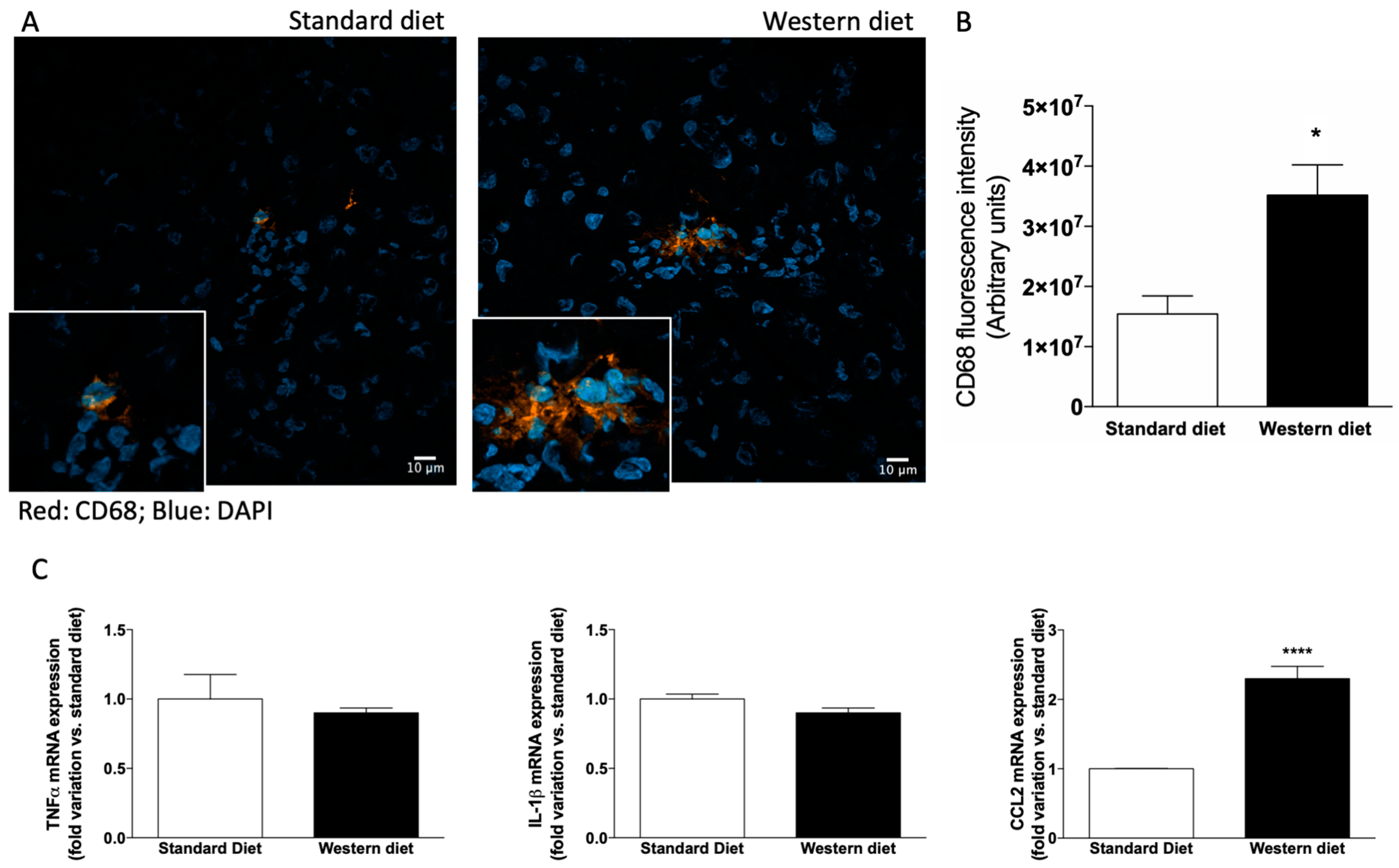
3.3. Effect of Western-Type Diet on the Activation of Hepatic Stellate Cells and Macrophages
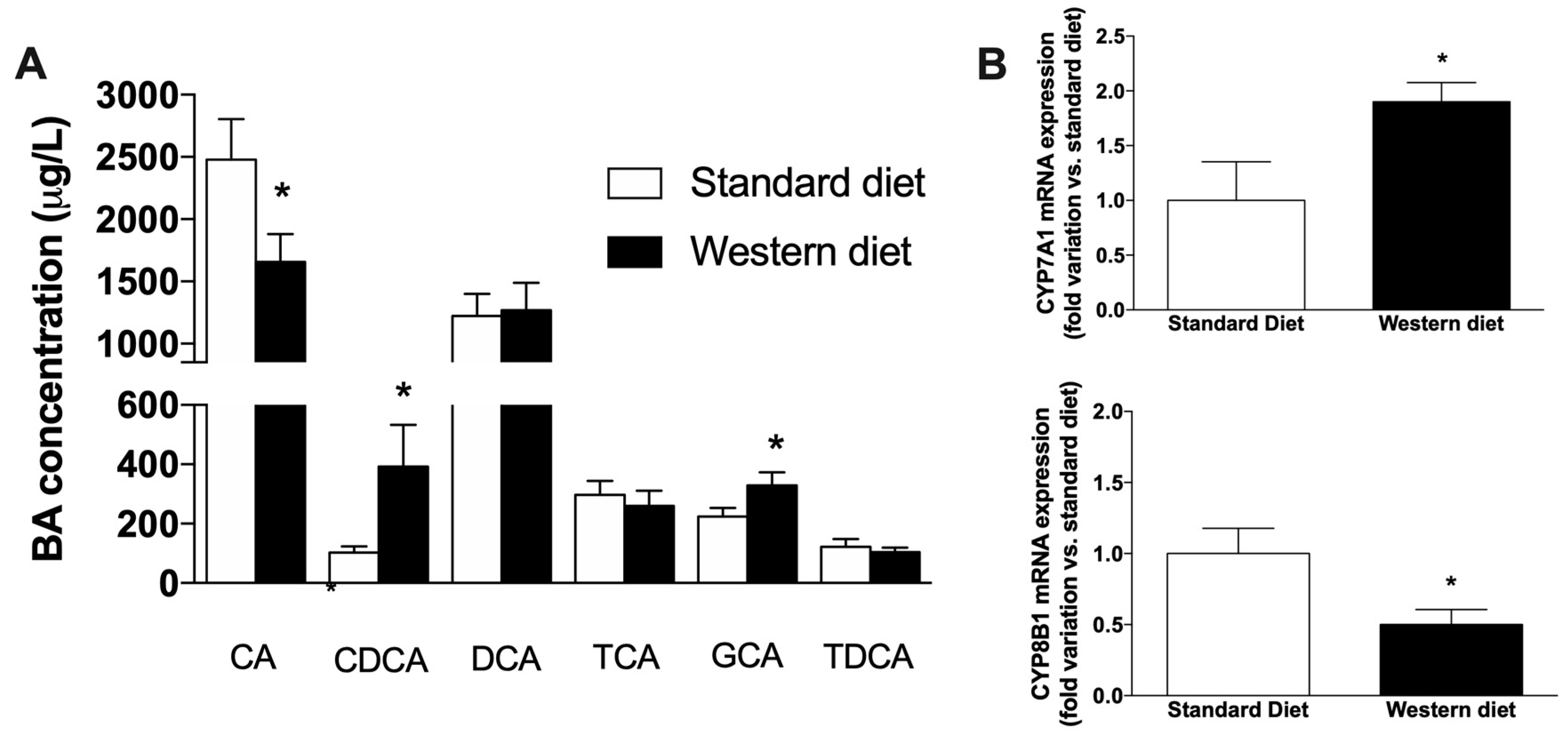
3.4. Effect of a Western-Type Diet on the Circulating Pool of Bile Acids
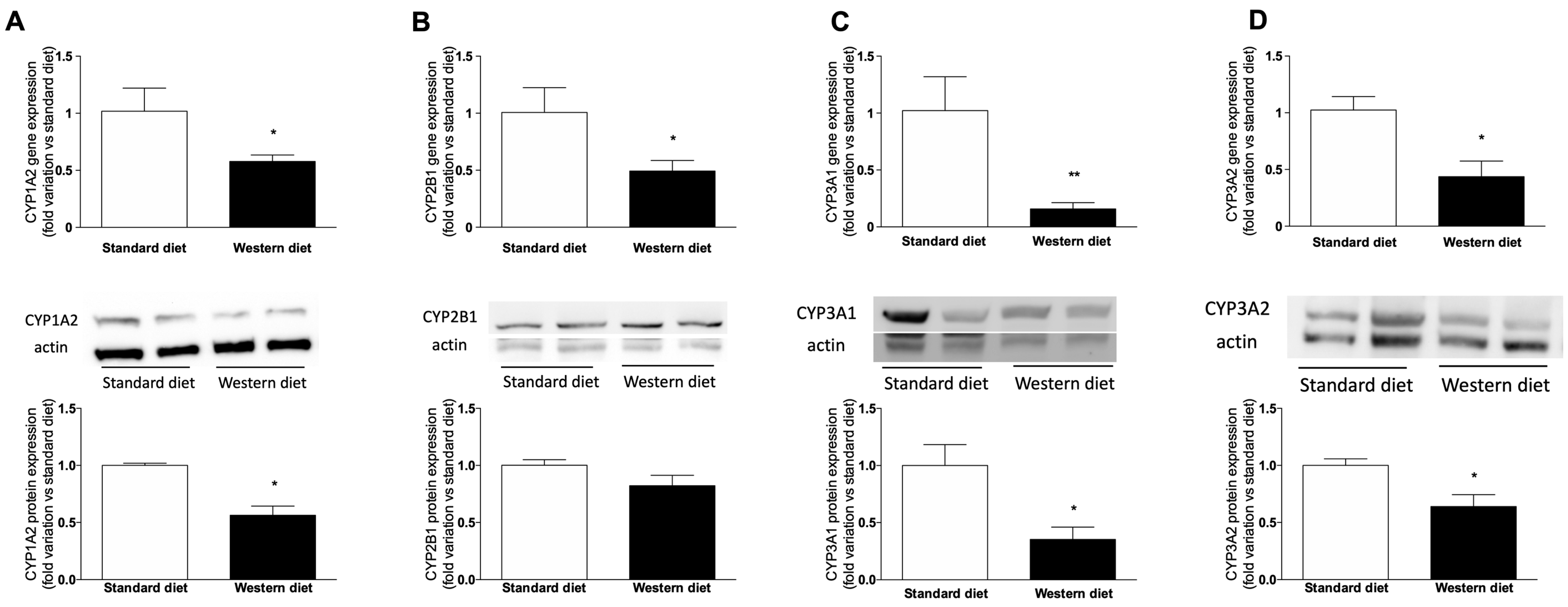
3.5. Effect of a Western-Type Diet on Hepatic Drug Metabolism
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nolan, C.J.; Damm, P.; Prentki, M. Type 2 diabetes across generations: From pathophysiology to prevention and management. Lancet Lond. Engl. 2011, 378, 169–181. [Google Scholar] [CrossRef]
- Dabelea, D.; Pettitt, D.J. Intrauterine diabetic environment confers risks for type 2 diabetes mellitus and obesity in the offspring, in addition to genetic susceptibility. J. Pediatr. Endocrinol. Metab. JPEM 2001, 14, 1085–1091. [Google Scholar] [CrossRef]
- Schwenk, R.W.; Vogel, H.; Schürmann, A. Genetic and epigenetic control of metabolic health. Mol. Metab. 2013, 2, 337–347. [Google Scholar] [CrossRef]
- Lozano, I.; Van der Werf, R.; Bietiger, W.; Seyfritz, E.; Peronet, C.; Pinget, M.; Jeandidier, N.; Maillard, E.; Marchioni, E.; Sigrist, S.; et al. High-fructose and high-fat diet-induced disorders in rats: Impact on diabetes risk, hepatic and vascular complications. Nutr. Metab. 2016, 13, 15. [Google Scholar] [CrossRef]
- Elliott, S.S.; Keim, N.L.; Stern, J.S.; Teff, K.; Havel, P.J. Fructose, weight gain, and the insulin resistance syndrome. Am. J. Clin. Nutr. 2002, 76, 911–922. [Google Scholar] [CrossRef]
- Ruxton, C.H.S.; Gardner, E.J.; McNulty, H.M. Is sugar consumption detrimental to health? A review of the evidence 1995–2006. Crit. Rev. Food Sci. Nutr. 2010, 50, 1–19. [Google Scholar] [CrossRef]
- Welsh, J.A.; Sharma, A.; Abramson, J.L.; Vaccarino, V.; Gillespie, C.; Vos, M.B. Caloric sweetener consumption and dyslipidemia among US adults. JAMA 2010, 303, 1490–1497. [Google Scholar] [CrossRef]
- Ludwig, D.S. Dietary glycemic index and obesity. J. Nutr. 2000, 130, 280S–283S. [Google Scholar] [CrossRef]
- Dekker, M.J.; Su, Q.; Baker, C.; Rutledge, A.C.; Adeli, K. Fructose: A highly lipogenic nutrient implicated in insulin resistance, hepatic steatosis, and the metabolic syndrome. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E685–E694. [Google Scholar] [CrossRef]
- Vasdev, S.; Longerich, L.; Gill, V. Prevention of fructose-induced hypertension by dietary vitamins. Clin. Biochem. 2004, 37, 1–9. [Google Scholar] [CrossRef]
- Fon Tacer, K.; Rozman, D. Nonalcoholic Fatty liver disease: Focus on lipoprotein and lipid deregulation. J. Lipids 2011, 2011, 783976. [Google Scholar] [CrossRef]
- Koo, S.-H. Nonalcoholic fatty liver disease: Molecular mechanisms for the hepatic steatosis. Clin. Mol. Hepatol. 2013, 19, 210–215. [Google Scholar] [CrossRef]
- Marchesini, G.; Brizi, M.; Bianchi, G.; Tomassetti, S.; Bugianesi, E.; Lenzi, M.; McCullough, A.J.; Natale, S.; Forlani, G.; Melchionda, N. Nonalcoholic fatty liver disease: A feature of the metabolic syndrome. Diabetes 2001, 50, 1844–1850. [Google Scholar] [CrossRef]
- Musso, G.; Cassader, M.; Gambino, R. Non-alcoholic steatohepatitis: Emerging molecular targets and therapeutic strategies. Nat. Rev. Drug Discov. 2016, 15, 249–274. [Google Scholar] [CrossRef]
- Takaki, A.; Kawai, D.; Yamamoto, K. Molecular mechanisms and new treatment strategies for non-alcoholic steatohepatitis (NASH). Int. J. Mol. Sci. 2014, 15, 7352–7379. [Google Scholar] [CrossRef]
- Byrne, C.D.; Targher, G. NAFLD: A multisystem disease. J. Hepatol. 2015, 62, S47–S64. [Google Scholar] [CrossRef]
- De Martin, S.; Gabbia, D.; Carrara, M.; Ferri, N. The brown algae fucus vesiculosus and ascophyllum nodosum reduce metabolic syndrome risk factors: A clinical study. Nat. Prod. Commun. 2018, 13, 1691–1694. [Google Scholar] [CrossRef]
- Lambert, J.E.; Ramos-Roman, M.A.; Browning, J.D.; Parks, E.J. Increased de novo lipogenesis is a distinct characteristic of individuals with nonalcoholic fatty liver disease. Gastroenterology 2014, 146, 726–735. [Google Scholar] [CrossRef]
- Arab, J.P.; Karpen, S.J.; Dawson, P.A.; Arrese, M.; Trauner, M. Bile acids and nonalcoholic fatty liver disease: Molecular insights and therapeutic perspectives. Hepatology 2017, 65, 350–362. [Google Scholar] [CrossRef]
- Gabbia, D.; Pozzo, L.; Zigiotto, G.; Roverso, M.; Sacchi, D.; Dalla Pozza, A.; Carrara, M.; Bogialli, S.; Floreani, A.; Guido, M.; et al. Dexamethasone counteracts hepatic inflammation and oxidative stress in cholestatic rats via CAR activation. PLoS ONE 2018, 13, e0204336. [Google Scholar] [CrossRef]
- Aleixandre de, A.A.; Miguel Castro, M. Experimental rat models to study the metabolic syndrome. Br. J. Nutr. 2009, 102, 1246–1253. [Google Scholar] [CrossRef]
- Gabbia, D.; Dall’Acqua, S.; Di Gangi, I.M.; Bogialli, S.; Caputi, V.; Albertoni, L.; Marsilio, I.; Paccagnella, N.; Carrara, M.; Giron, M.C.; et al. The Phytocomplex from Fucus vesiculosus and Ascophyllum nodosum Controls Postprandial Plasma Glucose Levels: An In Vitro and In Vivo Study in a Mouse Model of NASH. Mar. Drugs 2017, 15, 41. [Google Scholar] [CrossRef]
- Guido, M.; Sarcognato, S.; Russo, F.P.; Cardin, R.; Piciocchi, M.; Colloredo, G.; Farinati, F. Focus on histological abnormalities of intrahepatic vasculature in chronic viral hepatitis. Liver Int. Off. J. Int. Assoc. Study Liver 2018, 38, 1770–1776. [Google Scholar] [CrossRef]
- De Martin, S.; Gabbia, D.; Albertin, G.; Sfriso, M.M.; Mescoli, C.; Albertoni, L.; Paliuri, G.; Bova, S.; Palatini, P. Differential effect of liver cirrhosis on the pregnane X receptor-mediated induction of CYP3A1 and 3A2 in the rat. Drug Metab. Dispos. Biol. Fate Chem. 2014, 42, 1617–1626. [Google Scholar] [CrossRef]
- Castellani, G.; Paliuri, G.; Orso, G.; Paccagnella, N.; D’Amore, C.; Facci, L.; Cima, F.; Caicci, F.; Palatini, P.; Bova, S.; et al. An intracellular adrenomedullin system reduces IL-6 release via a NF-kB-mediated, cAMP-independent transcriptional mechanism in rat thymic epithelial cells. Cytokine 2016, 88, 136–143. [Google Scholar] [CrossRef]
- Gabbia, D.; Pozza, A.D.; Albertoni, L.; Lazzari, R.; Zigiotto, G.; Carrara, M.; Baldo, V.; Baldovin, T.; Floreani, A.; Martin, S.D. Pregnane X receptor and constitutive androstane receptor modulate differently CYP3A-mediated metabolism in early- and late-stage cholestasis. World J. Gastroenterol. 2017, 23, 7519–7530. [Google Scholar] [CrossRef]
- Floreani, M.; Gabbia, D.; Barbierato, M.; DE Martin, S.; Palatini, P. Differential inducing effect of benzo[a]pyrene on gene expression and enzyme activity of cytochromes P450 1A1 and 1A2 in Sprague-Dawley and Wistar rats. Drug Metab. Pharmacokinet. 2012, 27, 640–652. [Google Scholar] [CrossRef]
- De Martin, S.; Paliuri, G.; Belloni, A.; Orso, G.; Zanarella, E.; Stellin, G.; Milanesi, O.; Basso, G.; Ruga, E.M.; Frasson, C.; et al. Expression and distribution of the adrenomedullin system in newborn human thymus. PLoS ONE 2014, 9, e97592. [Google Scholar] [CrossRef]
- Antonioli, L.; Pellegrini, C.; Fornai, M.; Tirotta, E.; Gentile, D.; Benvenuti, L.; Giron, M.C.; Caputi, V.; Marsilio, I.; Orso, G.; et al. Colonic motor dysfunctions in a mouse model of high-fat diet-induced obesity: An involvement of A2B adenosine receptors. Purinergic Signal. 2017, 13, 497–510. [Google Scholar] [CrossRef]
- Di Francesco, L.; Dovizio, M.; Trenti, A.; Marcantoni, E.; Moore, A.; O’Gaora, P.; McCarthy, C.; Tacconelli, S.; Bruno, A.; Alberti, S.; et al. Dysregulated post-transcriptional control of COX-2 gene expression in gestational diabetic endothelial cells. Br. J. Pharmacol. 2015, 172, 4575–4587. [Google Scholar] [CrossRef]
- Gluchowski, N.L.; Becuwe, M.; Walther, T.C.; Farese, R.V. Lipid droplets and liver disease: From basic biology to clinical implications. Nat. Rev. Gastroenterol. Hepatol. 2017, 14, 343–355. [Google Scholar] [CrossRef]
- Marcher, A.-B.; Bendixen, S.M.; Terkelsen, M.K.; Hohmann, S.S.; Hansen, M.H.; Larsen, B.D.; Mandrup, S.; Dimke, H.; Detlefsen, S.; Ravnskjaer, K. Transcriptional regulation of Hepatic Stellate Cell activation in NASH. Sci. Rep. 2019, 9, 2324. [Google Scholar] [CrossRef]
- Ehling, J.; Tacke, F. Role of chemokine pathways in hepatobiliary cancer. Cancer Lett. 2016, 379, 173–183. [Google Scholar] [CrossRef]
- Ju, C.; Tacke, F. Hepatic macrophages in homeostasis and liver diseases: From pathogenesis to novel therapeutic strategies. Cell. Mol. Immunol. 2016, 13, 316–327. [Google Scholar] [CrossRef]
- Palatini, P.; Orlando, R.; De Martin, S. The effect of liver disease on inhibitory and plasma protein-binding displacement interactions: An update. Expert Opin. Drug Metab. Toxicol. 2010, 6, 1215–1230. [Google Scholar] [CrossRef]
- Yuan, Y.; Naito, H.; Jia, X.; Kitamori, K.; Nakajima, T. Combination of Hypertension Along with a High Fat and Cholesterol Diet Induces Severe Hepatic Inflammation in Rats via a Signaling Network Comprising NF-κB, MAPK, and Nrf2 Pathways. Nutrients 2017, 9, 1018. [Google Scholar] [CrossRef]
- Ramos-Romero, S.; Hereu, M.; Atienza, L.; Casas, J.; Taltavull, N.; Romeu, M.; Amézqueta, S.; Dasilva, G.; Medina, I.; Torres, J.L. Functional Effects of the Buckwheat Iminosugar d-Fagomine on Rats with Diet-Induced Prediabetes. Mol. Nutr. Food Res. 2018, 62, e1800373. [Google Scholar] [CrossRef]
- Biernacki, M.; Ambrożewicz, E.; Gęgotek, A.; Toczek, M.; Skrzydlewska, E. Long-term administration of fatty acid amide hydrolase inhibitor (URB597) to rats with spontaneous hypertension disturbs liver redox balance and phospholipid metabolism. Adv. Med. Sci. 2019, 64, 15–23. [Google Scholar] [CrossRef]
- Hintze, K.J.; Benninghoff, A.D.; Cho, C.E.; Ward, R.E. Modeling the Western Diet for Preclinical Investigations. Adv. Nutr. Bethesda MD 2018, 9, 263–271. [Google Scholar] [CrossRef]
- Lau, J.K.C.; Zhang, X.; Yu, J. Animal models of non-alcoholic fatty liver disease: Current perspectives and recent advances. J. Pathol. 2017, 241, 36–44. [Google Scholar] [CrossRef]
- Chang, Y.; Ryu, S.; Zhang, Y.; Son, H.J.; Kim, J.-Y.; Cho, J.; Guallar, E. A cohort study of serum bilirubin levels and incident non-alcoholic fatty liver disease in middle aged Korean workers. PLoS ONE 2012, 7, e37241. [Google Scholar] [CrossRef]
- Kwak, M.-S.; Kim, D.; Chung, G.E.; Kang, S.J.; Park, M.J.; Kim, Y.J.; Yoon, J.-H.; Lee, H.-S. Serum bilirubin levels are inversely associated with nonalcoholic fatty liver disease. Clin. Mol. Hepatol. 2012, 18, 383–390. [Google Scholar] [CrossRef]
- Luo, L.; An, P.; Jia, X.; Yue, X.; Zheng, S.; Liu, S.; Chen, Y.; An, W.; Winkler, C.A.; Duan, Z. Genetically Regulated Bilirubin and Risk of Non-alcoholic Fatty Liver Disease: A Mendelian Randomization Study. Front. Genet. 2018, 9, 662. [Google Scholar] [CrossRef]
- Zhou, H.; Hylemon, P.B. Bile acids are nutrient signaling hormones. Steroids 2014, 86, 62–68. [Google Scholar] [CrossRef]
- Jurate, K.; Rimantas, Z.; Jolanta, S.; Vladas, G.; Limas, K. Sensitivity and Specificity of Biochemical Tests for Diagnosis of Intrahepatic Cholestasis of Pregnancy. Ann. Hepatol. 2017, 16, 569–573. [Google Scholar] [CrossRef]
- Jericho, H.S.; Kaurs, E.; Boverhof, R.; Knisely, A.; Shneider, B.L.; Verkade, H.J.; Whitington, P.F. Bile acid pool dynamics in progressive familial intrahepatic cholestasis with partial external bile diversion. J. Pediatr. Gastroenterol. Nutr. 2015, 60, 368–374. [Google Scholar] [CrossRef]
- Lu, Y.; Du, Y.; Qin, L.; Wu, D.; Wang, W.; Ling, L.; Ma, F.; Ling, H.; Yang, L.; Wang, C.; et al. Gypenosides Altered Hepatic Bile Acids Homeostasis in Mice Treated with High Fat Diet. Evid. Based Complement. Altern. Med. ECAM 2018, 2018, 8098059. [Google Scholar] [CrossRef]
- Mouzaki, M.; Wang, A.Y.; Bandsma, R.; Comelli, E.M.; Arendt, B.M.; Zhang, L.; Fung, S.; Fischer, S.E.; McGilvray, I.G.; Allard, J.P. Bile Acids and Dysbiosis in Non-Alcoholic Fatty Liver Disease. PLoS ONE 2016, 11, e0151829. [Google Scholar] [CrossRef]
- Fan, L.; Joseph, J.F.; Durairaj, P.; Parr, M.K.; Bureik, M. Conversion of chenodeoxycholic acid to cholic acid by human CYP8B1. Biol. Chem. 2019, 400, 625–628. [Google Scholar] [CrossRef]
- Suppli, M.P.; Rigbolt, K.T.G.; Veidal, S.S.; Heebøll, S.; Eriksen, P.L.; Demant, M.; Bagger, J.I.; Nielsen, J.C.; Oró, D.; Thrane, S.W.; et al. Hepatic transcriptome signatures in patients with varying degrees of nonalcoholic fatty liver disease compared with healthy normal-weight individuals. Am. J. Physiol. Gastrointest. Liver Physiol. 2019, 316, G462–G472. [Google Scholar] [CrossRef]
- Fisher, C.D.; Lickteig, A.J.; Augustine, L.M.; Oude Elferink, R.P.J.; Besselsen, D.G.; Erickson, R.P.; Cherrington, N.J. Experimental non-alcoholic fatty liver disease results in decreased hepatic uptake transporter expression and function in rats. Eur. J. Pharmacol. 2009, 613, 119–127. [Google Scholar] [CrossRef]
- Fisher, C.D.; Lickteig, A.J.; Augustine, L.M.; Ranger-Moore, J.; Jackson, J.P.; Ferguson, S.S.; Cherrington, N.J. Hepatic cytochrome P450 enzyme alterations in humans with progressive stages of nonalcoholic fatty liver disease. Drug Metab. Dispos. Biol. Fate Chem. 2009, 37, 2087–2094. [Google Scholar] [CrossRef]
- Donato, M.T.; Jiménez, N.; Serralta, A.; Mir, J.; Castell, J.V.; Gómez-Lechón, M.J. Effects of steatosis on drug-metabolizing capability of primary human hepatocytes. Toxicol. In Vitro Int. J. Publ. Assoc. BIBRA 2007, 21, 271–276. [Google Scholar] [CrossRef]
- Gómez-Lechón, M.J.; Donato, M.T.; Castell, J.V.; Jover, R. Human hepatocytes in primary culture: The choice to investigate drug metabolism in man. Curr. Drug Metab. 2004, 5, 443–462. [Google Scholar] [CrossRef]
- Donato, M.T.; Lahoz, A.; Jiménez, N.; Pérez, G.; Serralta, A.; Mir, J.; Castell, J.V.; Gómez-Lechón, M.J. Potential impact of steatosis on cytochrome P450 enzymes of human hepatocytes isolated from fatty liver grafts. Drug Metab. Dispos. Biol. Fate Chem. 2006, 34, 1556–1562. [Google Scholar] [CrossRef]
- Dietrich, C.G.; Rau, M.; Jahn, D.; Geier, A. Changes in drug transport and metabolism and their clinical implications in non-alcoholic fatty liver disease. Expert Opin. Drug Metab. Toxicol. 2017, 13, 625–640. [Google Scholar] [CrossRef]
- Renaud, H.J.; Cui, J.Y.; Khan, M.; Klaassen, C.D. Tissue distribution and gender-divergent expression of 78 cytochrome P450 mRNAs in mice. Toxicol. Sci. Off. J. Soc. Toxicol. 2011, 124, 261–277. [Google Scholar] [CrossRef]
- Kubota, A.; Kawai, Y.K.; Yamashita, N.; Lee, J.S.; Kondoh, D.; Zhang, S.; Nishi, Y.; Suzuki, K.; Kitazawa, T.; Teraoka, H. Transcriptional profiling of cytochrome P450 genes in the liver of adult zebrafish, Danio rerio. J. Toxicol. Sci. 2019, 44, 347–356. [Google Scholar] [CrossRef]
- Ribeiro, A.; Igual-Perez, M.-J.; Santos Silva, E.; Sokal, E.M. Childhood Fructoholism and Fructoholic Liver Disease. Hepatol. Commun. 2019, 3, 44–51. [Google Scholar] [CrossRef]
- Geidl-Flueck, B.; Gerber, P.A. Insights into the Hexose Liver Metabolism-Glucose versus Fructose. Nutrients 2017, 9, 1026. [Google Scholar] [CrossRef]
- Tappy, L. Fructose-containing caloric sweeteners as a cause of obesity and metabolic disorders. J. Exp. Biol. 2018, 221 (Suppl. 1). [Google Scholar] [CrossRef]
- Zammit, V.A. Hepatic triacylglycerol synthesis and secretion: DGAT2 as the link between glycaemia and triglyceridaemia. Biochem. J. 2013, 451, 1–12. [Google Scholar] [CrossRef]
- Yu, J.; Loh, K.; Song, Z.-Y.; Yang, H.-Q.; Zhang, Y.; Lin, S. Update on glycerol-3-phosphate acyltransferases: The roles in the development of insulin resistance. Nutr. Diabetes 2018, 8, 34. [Google Scholar] [CrossRef]
- Madison, B.B. Srebp2: A master regulator of sterol and fatty acid synthesis. J. Lipid Res. 2016, 57, 333–335. [Google Scholar] [CrossRef]
- Marra, F.; Tacke, F. Roles for chemokines in liver disease. Gastroenterology 2014, 147, 577–594. [Google Scholar] [CrossRef]
| Standard Diet | Western Diet | |
|---|---|---|
| Albumin (g/L) | 41.05 ± 2.44 | 43.53 ± 2.26 |
| ALKP (U/L) | 167.5 ± 37.3 | 144.0 ± 11.1 |
| ALT (U/L) | 58.33 ± 11.25 | 63.67 ± 8.39 |
| AST (U/L) | 163.6 ± 36.4 | 265.0 ± 61.9 * |
| Total bilirubin (mmol/L) | 0.92 ± 0.32 | 1.88 ± 0.32 ** |
| Direct bilirubin (mmol/L) | 0.67 ± 0.31 | 1.37 ± 0.12 ** |
| Triglycerides (mmol/L) | 0.57 ± 0.19 | 0.97 ± 0.11 * |
| Total Cholesterol (mmol/L) | 2.18 ± 0.19 | 2.38 ± 0.06 |
| HDL Cholesterol (mmol/L) | 1.59 ± 0.20 | 1.73 ± 0.09 |
| LDL Cholesterol (mmol/L) | 0.35 ± 0.03 | 0.50 ± 0.05 *** |
| LDL:HDL ratio | 0.22 ± 0.02 | 0.29 ± 0.03 ** |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gabbia, D.; Roverso, M.; Guido, M.; Sacchi, D.; Scaffidi, M.; Carrara, M.; Orso, G.; Russo, F.P.; Floreani, A.; Bogialli, S.; et al. Western Diet-Induced Metabolic Alterations Affect Circulating Markers of Liver Function before the Development of Steatosis. Nutrients 2019, 11, 1602. https://doi.org/10.3390/nu11071602
Gabbia D, Roverso M, Guido M, Sacchi D, Scaffidi M, Carrara M, Orso G, Russo FP, Floreani A, Bogialli S, et al. Western Diet-Induced Metabolic Alterations Affect Circulating Markers of Liver Function before the Development of Steatosis. Nutrients. 2019; 11(7):1602. https://doi.org/10.3390/nu11071602
Chicago/Turabian StyleGabbia, Daniela, Marco Roverso, Maria Guido, Diana Sacchi, Michela Scaffidi, Maria Carrara, Genny Orso, Francesco Paolo Russo, Annarosa Floreani, Sara Bogialli, and et al. 2019. "Western Diet-Induced Metabolic Alterations Affect Circulating Markers of Liver Function before the Development of Steatosis" Nutrients 11, no. 7: 1602. https://doi.org/10.3390/nu11071602
APA StyleGabbia, D., Roverso, M., Guido, M., Sacchi, D., Scaffidi, M., Carrara, M., Orso, G., Russo, F. P., Floreani, A., Bogialli, S., & De Martin, S. (2019). Western Diet-Induced Metabolic Alterations Affect Circulating Markers of Liver Function before the Development of Steatosis. Nutrients, 11(7), 1602. https://doi.org/10.3390/nu11071602








