The Nutritional Components of Beer and Its Relationship with Neurodegeneration and Alzheimer’s Disease
Abstract
1. Introduction
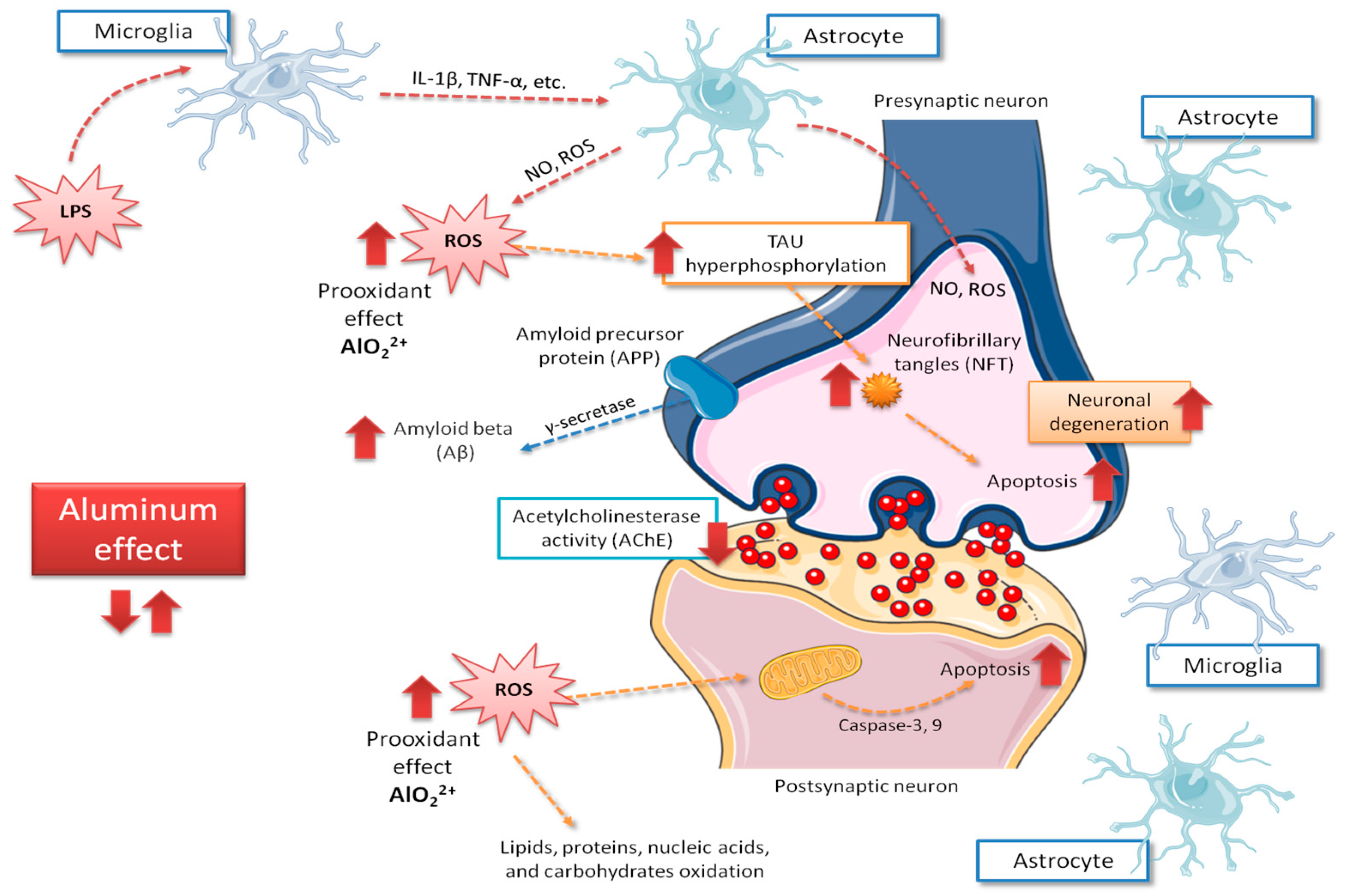
2. Aluminum as an Alzheimer’s Disease Risk Factor
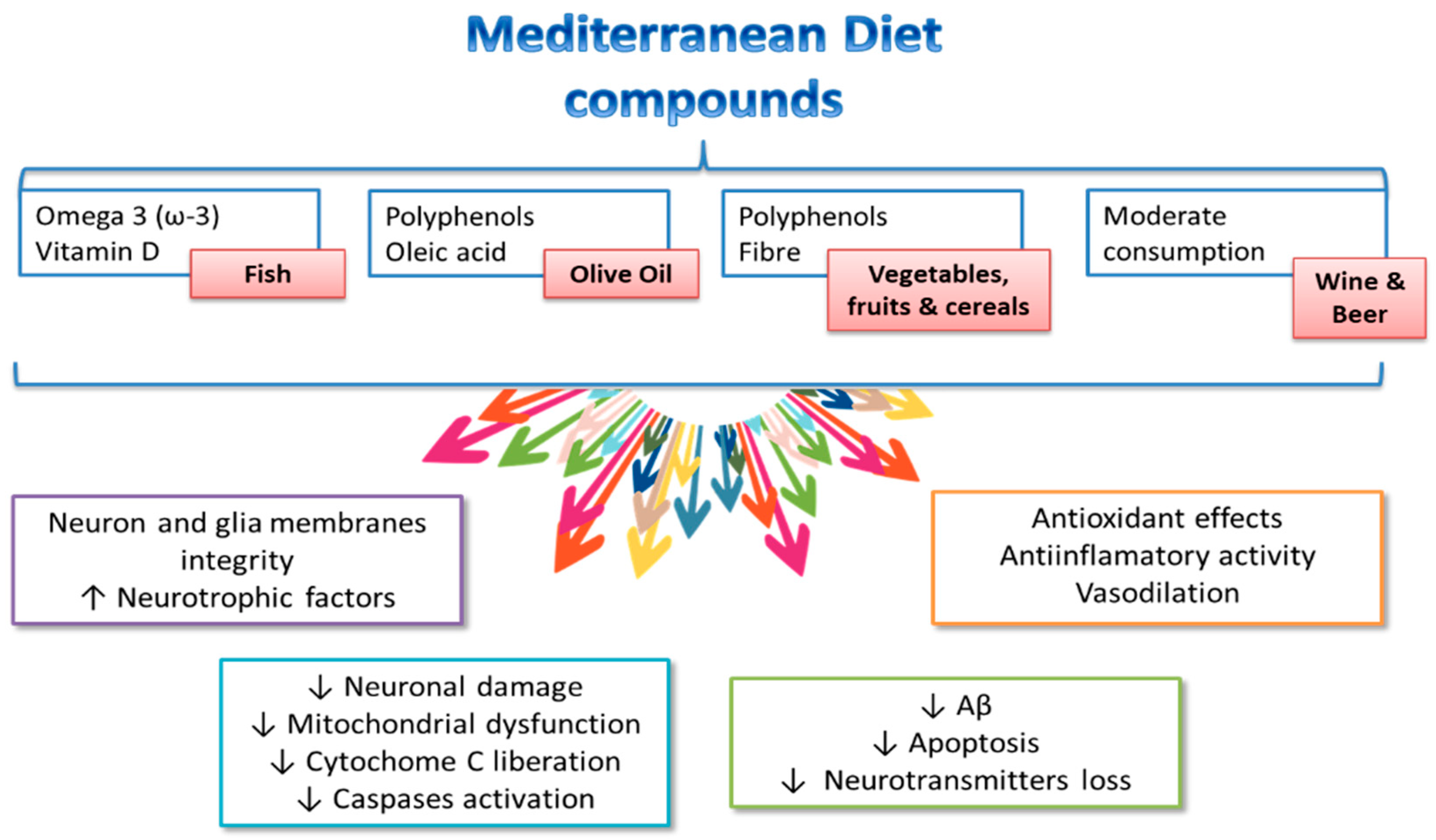
3. Nutritional Factors and Neurological Health—Beer as a Component of the Mediterranean Diet
4. Composition of Beer—Beneficial Aspects on Alzheimer’s Disease
4.1. Hops (Humulus lupulus L.)
4.2. Silicon
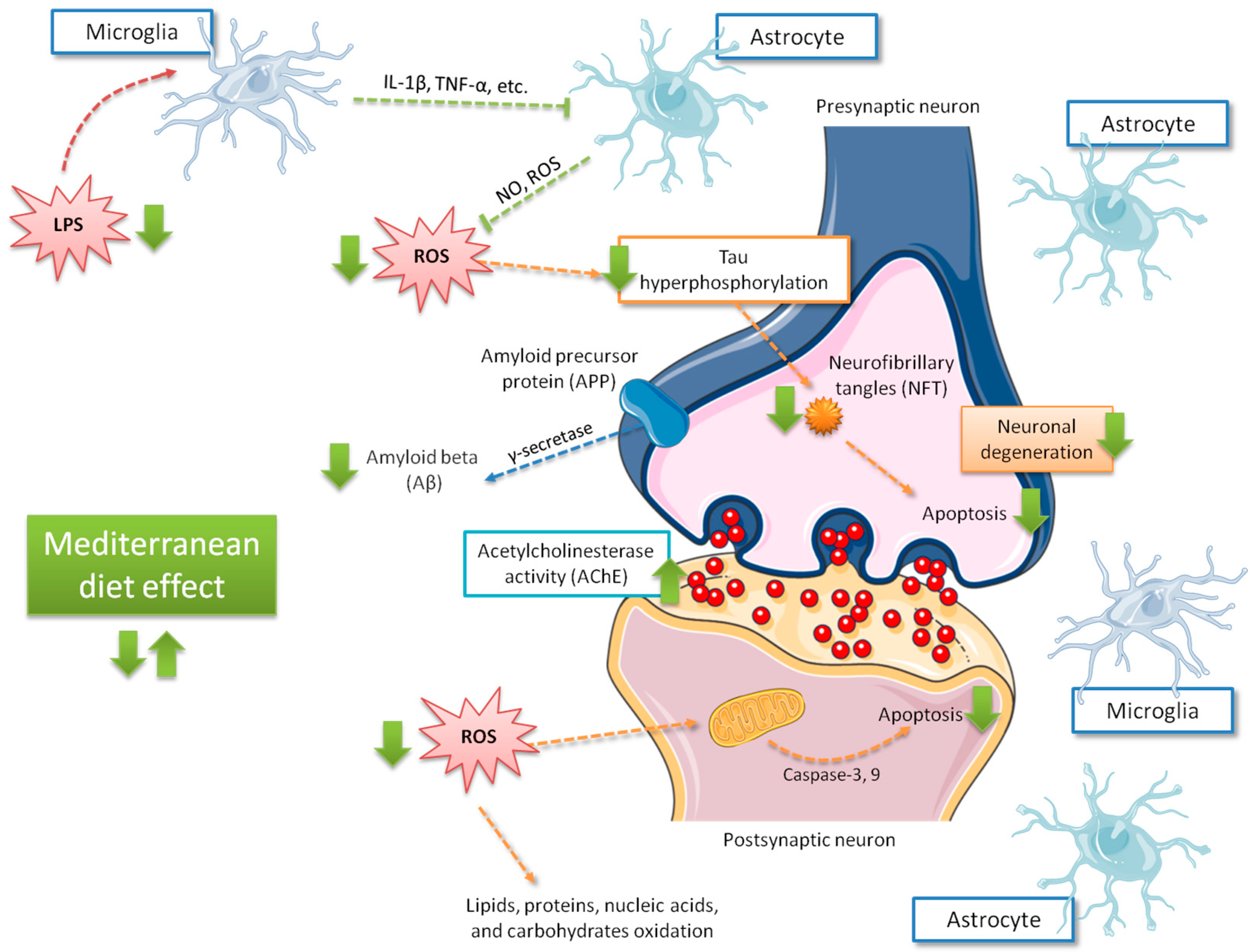
5. Effects of Beer on Aluminum Bioavailability
6. Effect of Beer on Brain Antioxidant and Inflammatory Status
7. Effect of Beer on Metal Homeostasis in the Brain
8. Effect of Non-Alcoholic Beer, Silicon, and Hops on Brain Damage and Behavioral Changes Induced by Aluminum
9. Conclusions
- In vitro and in vivo models are plausible tools to study brain mechanisms related to changes in behavior, Alzheimer’s disease, and dementia.
- Aluminum induces several mechanisms engaged to brain damage and behavioral disturbances, through mechanisms that mainly involve apoptosis, tau phosphorylation, Aβ accumulation, ROS formation, necrosis of neuronal cells, regulation of metal imbalance, and changes in the antioxidant defense system.
- The conjoint addition of aluminum and beer, or its ingredients and compounds, proved that they can partially block the negative effects of neurodegeneration or neurotoxics, such as aluminum, in several cell, rodent, and human models.
- Due to its alcohol content, regular beer consumption can be non-adequate for some risk-group populations (pregnancy, children, people affected by liver diseases), and the consumption of non-alcoholic beer is highly recommended instead of regular beers.
- Given the results observed by our groups and others, dementia or the pathognomic factors can be blocked by promoting increased levels of silicon consumption in aluminum-intoxicated patients.
- As silicon is “attracted by” the presence of aluminum at the intestine and brain, among other body places, strategies should be formulated to adjust silicon consumption to aluminum exposure and to increase the brain uptake of silicon.
10. Future Remarks
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Prince, M.; Ali, G.C.; Guerchet, M.; Prina, A.M.; Albanese, E.; Wu, Y.T. Recent global trends in the prevalence and incidence of dementia, and survival with dementia. Alzheimers Res. Ther. 2016, 30, 23. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Risk Reduction of Cognitive Decline and Dementia: WHO Guidelines; WHO: Geneva, Switzerland, 2019. [Google Scholar]
- Blennow, K.; de Leon, M.J.; Zetterberg, H. Alzheimer’s disease. Lancet 2006, 368, 387–403. [Google Scholar] [CrossRef]
- Cuervo, A.M.; Wong, E. Chaperone-mediated autophagy: Roles in disease and aging. Cell Res. 2014, 24, 92–104. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Villegas, A.; Galbete, C.; Martínez-González, M.A.; Martinez, J.A.; Razquín, C.; Salas-Salvadó, J.; Estruch, R.; Buil-Cosiales, P.; Martí, A. The effect of the Mediterranean diet on plasma brain-derived neurotrophic factor (BDNF) levels: The PREDIMED-NAVARRA randomized trial. Nutr. Neurosci. 2014, 14, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Gosche, K.M.; Mortimer, J.A.; Smith, C.D.; Markesbery, W.R.; Snowdon, D.A. Hippocampal volume as an index of Alzheimer neuropathology: Findings from the nun study. Neurology 2002, 58, 1476–1482. [Google Scholar] [CrossRef] [PubMed]
- Barnes, D.E. The Mediterranean diet: Good for health good for the brain? Ann. Neurol. 2011, 69, 226–228. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.L.; Atti, A.R.; Gatz, M.; Pedersen, N.L.; Johansson, B.; Fratiglioni, L. Midlife overweight and obesity increase late-life dementia risk: A population-based twin study. Neurology 2011, 76, 1568–1574. [Google Scholar] [CrossRef] [PubMed]
- Medina-Santillán, R.; Martínez-Sámano, J. Implicaciones de la obesidad: Neuroinflamación y neurodegeneración [Obesity implications: Neuroinflammation and Neurodegenaration]. In IV y V Cursos Avanzados Sobre Obesidad y Síndrome Metabólico; Sánchez-Muniz, F.J., Bastida, S., Gesteiro, E., Garcimartín, A., Eds.; Real Academia Nacional de Farmacia e Instituto de España: Madrid, Spain, 2018; pp. 101–116. [Google Scholar]
- Nuzzo, D.; Picone, P.; Baldassano, S.; Caruana, L.; Messina, E.; Marino Gammazza, A.; Di Carlo, M. Insulin resistance as common molecular denominator linking obesity to Alzheimer’s disease. Curr. Alzheimers Res. 2015, 12, 723–735. [Google Scholar] [CrossRef]
- Nuzzo, D.; Amato, A.; Picone, P.; Terzo, S.; Galizzi, G.; Bonina, F.; Di Carlo, M. A natural dietary supplement with a combination of nutrients prevents neurodegeneration induced by a high fat diet in mice. Nutrients 2018, 10, 1130. [Google Scholar] [CrossRef]
- Sánchez-Muniz, F.J.; Simón, C. Clock genes, chronodysruption, nutrition and obesity. Curr. Res. Diabetes Obes. J. 2017, 3, 555607. [Google Scholar]
- García-Huerta, P.; Troncoso-Escudero, P.; Jerez, C.; Hetz, C.; Vidal, R.L. The intersection between growth factors, autophagy and ER stress: A new target to treat neurodegenerative diseases? Brain Res. 2016, 1649 Pt B, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Valero, T. Mitochondrial Biogenesis: Pharmacological Approaches. Curr. Pharm. Des. 2014, 20, 5507–5509. [Google Scholar] [CrossRef] [PubMed]
- Miklossy, J. Emerging roles of pathogens in Alzheimer disease. Exp. Rev. Mol. Med. 2011, 13, e30. [Google Scholar] [CrossRef] [PubMed]
- Goyer, R.A. Toxic and essential metal interactions. Ann. Rev. Nutr. 1997, 17, 37–50. [Google Scholar] [CrossRef] [PubMed]
- Colomina, M.T.; Peris-Sampedro, F. Aluminium and Alzheimer disease. Adv. Neurobiol. 2017, 18, 183–197. [Google Scholar] [PubMed]
- Noremberg, S.; Bohrer, D.; Schetinger, M.R.; Bairros, A.V.; Gutierres, J.; Gonçalves, J.F.; Veiga, M.; Santos, F.W. Silicon reverses lipid peroxidation but not acetylcholinesterase activity induced by long-term exposure to low aluminum levels in rat brain regions. Biol. Trace Elem. Res. 2016, 169, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Singh, A. A review on Alzheimer’s disease pathophysiology and its management: An update. Pharmacol. Rep. 2015, 67, 195–203. [Google Scholar] [CrossRef]
- El-Swefy, S.H.; Atteia, H.H. Molecular aspects of the Mediterranean diet: Adiponectin, brain, amyloid precursor protein, apolipoprotein E and Caspase-3 mRNA. In The Mediterranean Diet: An Evidence-Based Approach; Preedy, V., Watson, R., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 441–453. [Google Scholar]
- Hersi, M.; Irvine, B.; Gupta, P.; Gomes, J.; Birkett, N.; Krewski, D. Risk factors associated with the onset and progression of Alzheimer’s disease: A systematic review of the evidence. Neurotoxicology 2017, 61, 143–187. [Google Scholar] [CrossRef]
- Campbell, A. The role of aluminum and cooper on neuroinflammation and Alzheimer’s disease. J Alzheimers Dis. 2006, 10, 165–172. [Google Scholar] [CrossRef]
- Perry, C.C.; Keeling-Tucker, T. Aspects of the bioinorganic chemistry of silicon in conjunction with the biometals calcium, iron and aluminum. J. Inorg. Biochem. 1998, 69, 181–191. [Google Scholar] [CrossRef]
- Aikoh, H.; Nakamura, K.; Yamato, M.; Shibahara, T. Studies on the amount of aluminum and calcium administration with and without amino acid. Physiol. Chem. Phys. Med. NMR 2005, 37, 65–69. [Google Scholar]
- Baydar, T.; Papp, A.; Aydin, A.; Nagymajtenyi, L.; Schulz, H.; Isimer, A.; Sahin, G. Accumulation of aluminum in rat brain: Does it lead to behavioral and electrophysiological changes? Biol. Trace Elem. Res. 2003, 92, 231–244. [Google Scholar] [CrossRef]
- Rondeau, V.; Iron, A.; Letenneur, L.; Commenges, D.; Duchene, F.; Arveiler, B.; Dartigues, J.F. Analysis of the effect of aluminum in drinking water and transferrin C2 allele on Alzheimer’s disease. Eur. J. Neurol. 2006, 13, 1022–1025. [Google Scholar] [CrossRef] [PubMed]
- Inan-Eroglu, E.; Ayaz, A. Is aluminum exposure a risk factor for neurological disorders? J. Res. Med. Sci. 2018, 23, 51. [Google Scholar] [PubMed]
- Yuan, C.Y.; Lee, Y.J.; Hsu, G.S. Aluminium overload increases oxidative stress in four functional brain areas of neonatal rats. J. Biomed. Sci. 2012, 19, 51. [Google Scholar] [CrossRef] [PubMed]
- Nübling, G.; Bader, B.; Levin, J.; Hildebrandt, J.; Kretzschmar, H.; Giese, A. Synergistic influence of phosphorylation and metal ions on tau oligomer formation and coaggregation with α-synuclein at the single molecule level. Mol. Neurodegener. 2012, 23, 35. [Google Scholar] [CrossRef]
- Oshiro, S.; Kawahara, M.; Kuroda, Y.; Zhang, C.; Cai, Y.; Kitajima, S.; Shirao, M. Glial cells contribute more to iron and aluminum accumulation but are more resistant to oxidative stress than neuronal cells. Biochim. Biophys. Acta 2000, 1502, 405–414. [Google Scholar] [CrossRef]
- Exley, C. A molecular mechanism of aluminum-induced Alzheimer’s disease? J. Inorg. Biochem. 1999, 76, 133–140. [Google Scholar] [CrossRef]
- Kawahara, M. Effect of aluminum on the nervous system and its possible link with neurodegenerative diseases. J. Alzheimers Dis. 2005, 8, 171–182. [Google Scholar] [CrossRef]
- Perl, D.P.; Moalem, S. Aluminium and Alzheimer’s disease, a personal perspective after 25 years. J. Alzheimers Dis. 2006, 9, 291–300. [Google Scholar] [CrossRef]
- McLachlan, D.R.; Bergeron, C.; Alexandrov, P.N.; Walsh, W.J.; Pogue, A.I.; Percy, M.E.; Kruck, T.P.A.; Fang, Z.; Sharfman, N.M.; Jaber, V.; et al. Aluminium in Neurological and Neurodegenerative Disease. Mol. Neurobiol. 2019, 56, 1531–1538. [Google Scholar] [CrossRef] [PubMed]
- Kawahara, M.; Kato-Negishi, M. Link between aluminum and the pathogenesis of Alzheimer’s disease: The integration of the aluminum and amyloid cascade hypotheses. Int. J. Alzheimers Dis. 2011, 2011, 276393. [Google Scholar] [CrossRef] [PubMed]
- Walton, J.R. Aluminium in hippocampal neurons from humans with Alzheimer’s disease. Neurotoxicology 2006, 27, 385–394. [Google Scholar] [CrossRef] [PubMed]
- Bolognin, S.; Messori, L.; Zatta, P. Metal ion physiopathology in neurodegenerative disorders. Neuromol. Med. 2009, 11, 223–238. [Google Scholar] [CrossRef] [PubMed]
- Exley, C. The aluminum-amyloid cascade hypothesis and Alzheimer’s disease. Subcell Biochem. 2005, 38, 225–234. [Google Scholar] [PubMed]
- Exley, C. Aluminium and iron, but neither copper nor zinc, are key to the precipitation of beta-sheets of Abets in senile plaque cores in Alzheimer’s disease. J. Alzheimers Dis. 2006, 10, 173–177. [Google Scholar] [CrossRef]
- Pratico, D.; Uryu, K.; Sung, S.; Tnag, S.; Trojanowski, J.Q.; Lee, V.M. Aluminium odulates brain amyloidosis though oxidative stress in APP transgenic mice. FASEB J. 2002, 16, 1138–1140. [Google Scholar] [CrossRef]
- Bondy, S.C.; Kirstein, S. The promotion of iron-induced generation of reactive oxygen species in nerve tissue by aluminum. Mol. Chem. Neuropathol. 1996, 27, 185–194. [Google Scholar] [CrossRef]
- Becaria, A.; Campbell, A.; Bondy, S.C. Aluminium and copper interact in the promotion of oxidative but not inflammatory events: Implications for Alzheimer’s disease. J. Alzheimers Dis. 2003, 5, 31–38. [Google Scholar] [CrossRef]
- González-Muñoz, M.J.; Garcimartín, A.; Meseguer, I.; Mateos-Vega, C.J.; Orellana, J.M.; Peña-Fernández, A.; Benedí, J.; Sánchez-Muniz, F.J. Silicic acid and beer consumption reverses the metal imbalance and the prooxidant status induced by aluminum nitrate in mouse brain. J. Alzheimers Dis. 2017, 56, 917–927. [Google Scholar] [CrossRef]
- Fattoretti, P.; Bertoni-Freddari, C.; Balietti, M.; Giorgetti, B.; Solazzi, M.; Zatta, P. Chronic aluminum administration to old rats results in increased levels of brain metal ions and enlarged hippocampal mossy fibers. Ann. N. Y. Acad. Sci. 2004, 1019, 44–47. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Iglesias, S.; Méndez-Alvarez, E.; Iglesias-González, J.; Muñoz-Patiño, A.; Sánchez-Sellero, I.; Labandeira-García, J.L.; Soto-Otero, R. Brain oxidative stress and selective behaviour of aluminum in specific areas of rat brain: Potential effects in a 6-OHDA-induced model of Parkinson’s disease. J. Neurochem. 2009, 109, 879–888. [Google Scholar] [CrossRef] [PubMed]
- Sharma, P.; Ahmad-Shah, Z.; Kumar, A.; Islam, F.; Mishra, K.P. Role of combined administration of Tiron and glutathione against aluminum-induced oxidative stress in rat brain. J. Trace Elem. Med. Biol. 2007, 21, 63–70. [Google Scholar] [CrossRef] [PubMed]
- Moumen, R.; Ait-Oukhata, N.; Bureau, F.; Fleury, C.; Bougle, D.; Arhan, P. Aluminium increases xanthine oxidase activity and disturbs antioxidant status in the rat. J. Trace Elem. Med. Biol. 2001, 15, 89–93. [Google Scholar] [CrossRef]
- González-Muñoz, M.J.; Meseguer, I.; Sánchez-Reus, M.I.; Schultz, A.; Olivero, R.; Benedí, J.; Sánchez-Muniz, F.J. Beer consumption reduces cerebral oxidation caused by aluminum toxicity by normalizing gene expression of tumor necrotic factor alpha and several antioxidant enzymes. Food Chem. Toxicol. 2008, 46, 1111–1118. [Google Scholar] [CrossRef] [PubMed]
- Tarkowski, E.; Andreasen, N.; Tarkowski, A.; Blennow, K. Intrathecal inflammation precedes development of Alzheimer’s disease. J. Neurol. Neurosurg. Psychiatry 2003, 74, 1200–1205. [Google Scholar] [CrossRef] [PubMed]
- Becaria, A.; Lahiri, D.K.; Bondy, S.C.; Chen, D.; Hamadeh, A.; Li, H. Aluminium and cooper in drinking water enhance inflammatory or oxidative events specifically in the brain. J. Neuroinmunol. 2006, 176, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Lukiw, W.J.; Percy, M.E.; Kruck, T.P. Nanomolar aluminum induces pro-inflammatory and pro-apoptotic gene expression in human brain cells in primary culture. J. Inorg. Biochem. 2005, 99, 1895–1898. [Google Scholar] [CrossRef]
- Abdel-Aal, R.A.; Assi, A.A.; Kostandy, B.B. Rivastigmine reverses aluminum-induced behavioral changes in rats. Eur. J. Pharmacol. 2011, 659, 169–176. [Google Scholar] [CrossRef]
- Martinez, C.S.; Escobar, A.G.; Uranga-Ocio, J.A.; Peçanha, F.M.; Vassallo, D.V.; Exley, C.; Miguel, M.; Wiggers, G.A. Aluminium exposure for 60 days at human dietary levels impairs spermatogenesis and sperm quality in rats. Reprod. Toxicol. 2017, 73, 128–141. [Google Scholar] [CrossRef]
- Kaizer, R.R.; Correa, M.C.; Spanevello, R.M.; Morsch, V.M.; Mazzanti, C.M.; Gonçalves, J.F.; Schetinger, M.R. Acetylcholinesterase activation and enhanced lipid peroxidation after long-term exposure to low levels of aluminum on different mouse brain regions. J. Inorg. Biochem. 2005, 9, 1865–1870. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S. Aluminium-induced biphasic effect. Med. Hypotheses 1999, 52, 557–559. [Google Scholar] [CrossRef]
- Solfrizzi, V.; Panza, F.; Torres, F.; Mastroianni, F.; Del Parigi, A.; Venezia, A.; Capurso, A. High monounsaturated fatty acids intake protects against age-related cognitive decline. Neurology 1999, 52, 1563–1569. [Google Scholar] [CrossRef] [PubMed]
- Berr, C.; Portet, F.; Carriere, I.; Akbaraly, T.N.; Feart, C.; Gourlet, V.; Combe, N.; Barberger-Gateau, P.; Ritchie, K. Olive oil and cognition: Results from the three-city study. Dement. Geriatr. Cogn. Disord. 2009, 28, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Yannakoulia, M.; Kontogianni, M.; Scarmeas, N. Cognitive health and Mediterranean diet: Just diet or lifestyle pattern? Ageing Res. Rev. 2015, 20, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Jacka, F.N.; Cherbuin, N.; Anstey, K.J.; Sachdev, P.; Butterworth, P. Western diet is associated with a smaller hippocampus: A longitudinal investigation. BMC Med. 2015, 8, 215. [Google Scholar] [CrossRef] [PubMed]
- Gu, Y.; Brickman, A.M.; Stern, Y.; Habeck, C.G.; Razlighi, Q.R.; Luchsinger, J.A.; Manly, J.J.; Schupf, N.; Mayeux, R.; Scarmeas, N. Mediterranean diet and brain structure in a multiethnic elderly cohort. Neurology 2015, 85, 1744–1751. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.C.; Davies, M.; Murray, J.; Williams, S.; Tsui, W.H.; Li, Y.; Andrews, R.D.; Lukic, A.; McHugh, P.; Vallabhajosula, S.; et al. Physical activity, Mediterranean diet and biomarkers-assessed risk of Alzheimer is a multi-modality brain imaging study. Adv. J. Mol. Imaging 2014, 4, 43–57. [Google Scholar] [CrossRef] [PubMed]
- Turner, P.L.; Principal, M.A. Circadian photoreception: Aging and the eye’s important role in systemic health. Br. J. Ophthalmol. 2008, 92, 1439–1444. [Google Scholar] [CrossRef] [PubMed]
- Gesteiro, E.; Sánchez-Muniz, F.J.; Bastida, S. Hypercortisolemia and hyperinsulinemia interaction and their impact upon insulin resistance/sensitivity markers at birth. In Blood Banking for Clinical Application and Regenerative Medicine; Colette-Mauricio, A., Ed.; InTech: Rijeka, Croatia, 2017; pp. 491–503. [Google Scholar]
- Del Chierico, F.; Vernocchi, P.; Dallapiccola, B.; Putignani, L. Mediterranean diet and health: Food effects on gut microbiota and disease control. Int. J. Mol. Sci. 2014, 15, 11678–11699. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Martínez-González, M.A.; Tong, T.Y.; Forouhi, N.G.; Khandelwal, S.; Prabhakaran, D.; Mozaffarian, D.; de Lorgeril, M. Definitions and potential health benefits of the Mediterranean diet: Views from experts around the world. BMC Med. 2014, 12, 112. [Google Scholar] [CrossRef] [PubMed]
- Castro-Quezada, I.; Román-Viñas, B.; Serra-Majem, L. Nutritional adequacy of the Mediterranean diet. In The Mediterranean Diet: An Evidence-Based Approach; Preedy, V.R., Ross-Watson, R., Eds.; Academic Press: Amsterdam, The Netherlands, 2015; pp. 13–21. [Google Scholar]
- Trichopoulou, A. Traditional Mediterranean diet and longevity in the elderly: A review. Public Health Nutr. 2004, 7, 943–947. [Google Scholar] [CrossRef] [PubMed]
- Vasto, S.; Buscemi, S.; Barera, A.; Di Carlo, M.; Accardi, G.; Caruso, C. Mediterranean diet and healthy ageing: A Sicilian perspective. Gerontology 2014, 60, 508–518. [Google Scholar] [CrossRef] [PubMed]
- Vasto, S.; Barera, A.; Rizzo, C.; Di Carlo, M.; Caruso, C.; Panotopoulos, G. Mediterranean diet and longevity: An example of nutraceuticals? Curr. Vasc. Pharmacol. 2014, 12, 735–738. [Google Scholar] [CrossRef] [PubMed]
- Keys, A.; Menotti, A.; Karvonen, M.J.; Aravanis, C.; Blackburn, H.; Buzina, R. The diet and 15-years dea Beer Drinking Associates with Lower Burden of Amyloid Beta Aggregation in the Brain: Helsinki Sudden Death Series. Alcohol. Clin. Exp. Res. 1986, 40, 1473–1478. [Google Scholar]
- Ruiz-Liso, J.M.; Ruiz-García, J. Temple of health based on the Mediterranean diet. J. Neg. Non Pos. Res. 2019, 4, 492–506. [Google Scholar]
- González-Gross, M.; Gómez-Llorente, J.J.; Valtueña, J.; Ortiz, J.C.; Meléndez, A. La “pirámide de la guía de estilo de vida saludable” para niños y adolescentes. Nutr. Hosp. 2008, 23, 159–168. [Google Scholar]
- Aranceta-Bartrina, J.; Arija-Val, V.; Maíz-Aldalur, E.; Martínez de la Victoria-Muñoz, E.; Ortega-Anta, R.M.; Pérez-Rodrigo, C.; Quiles-Izquierdo, J.; Rodríguez-Martín, A.; Román-Viñas, B.; Salvador-Castell, G.; et al. SENC-Dietary Guidelines. Nutr. Hosp. 2016, 33, 1–48. [Google Scholar]
- Sánchez-Muniz, F.J. Aceite de oliva, clave de vida en la Cuenca Mediterránea. Anales de la Real Academia Nacional de Farmacia 2007, 73, 653–692. [Google Scholar]
- Bastida, S.; Sánchez-Muniz, F.J. Frying a cultural way of cooking in the Mediterranean diet. In The Mediterranean Diet: An Evidence-Based Approach; Preedy, V.R., Watson, R.R., Eds.; Elsevier: Amsterdam, The Netherlands, 2015; pp. 217–234. [Google Scholar]
- Arranz, A.; Chiva-Blanch, G.; Valderas Martínez, P.; Casas, R.; Estruch, R. Beer: Beneficial aspects and contribution to the Mediterranean diet. In The Mediterranean Diet: An Evidence-Based Approach; Preedy, V.R., Ross-Watson, R., Eds.; Academic Press: Amsterdam, The Netherlands, 2015; pp. 153–164. [Google Scholar]
- Noah, A.; Truswell, A.S. There are many Mediterranean diets. Asia Pacific J. Clin. Nutr. 2001, 10, 2–9. [Google Scholar] [CrossRef]
- Davis, C.; Bryan, J.; Hodgson, J.; Murphy, K. Definition of the Mediterranean Diet: A Literature Review. Nutrients 2015, 7, 9139–9153. [Google Scholar] [CrossRef] [PubMed]
- Kirin Holding Company. Global Beer Consumption by Country in 2016. 2017. Available online: www.kirinholdings.co.jp/english/news/2017/1221_01.html (accessed on 1 May 2019).
- Otaegui-Arrazola, A.; Amiano, P.; Elbusto, A.; Urdaneta, E.; Martínez-Lage, P. Diet, cognition, and Alzheimer’s disease: Food for thought. Eur. J. Nutr. 2014, 53, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Lourida, I.; Soni, M.; Thompson-Coon, J.; Purandare, N.; Lang, I.A.; Ukoumunne, O.C.; Llewellyn, D.J. Mediterranean diet, cognitive function, and dementia: A systematic review. Epidemiology 2013, 24, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Psaltopoulou, T.; Sergentanis, T.N.; Panagiotakos, D.B.; Sergentanis, I.N.; Kosti, R.; Scarmeas, N. Mediterranean diet, stroke, cognitive impairment, and depression: A meta-analysis. Ann. Neurol. 2013, 74, 580–591. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.; Parsaik, A.K.; Mielke, M.M. Association of Mediterranean diet with mild cognitive impairment and Alzheimer’s disease: A systematic review and meta-analysis. J. Alzheimers Dis. 2014, 39, 271–282. [Google Scholar] [CrossRef] [PubMed]
- Macpherson, H.; Lee, J.; Villalon, L.; Pase, M.; Pipingas, A.; Scholey, A. The influence of Mediterranean diet on cognitive health. In The Mediterranean Diet. An Evidence-Based Approach; Preedy, V., Watson, R.R., Eds.; Academic Press: Amsterdam, The Netherlands, 2015; pp. 81–88. [Google Scholar]
- Samieri, C.; Okereke, O.I.; Devore, E.; Grodstein, F. Long-term adherence to the Mediterranean diet is associated with overall cognitive status, but not cognitive decline, in women. J. Nutr. 2013, 143, 493–499. [Google Scholar] [CrossRef] [PubMed]
- Titova, O.E.; Ax, E.; Brooks, S.J.; Sjögren, P.; Cederholm, T.; Kilander, L.; Kullberg, J.; Larsson, E.M.; Johansson, L.; Ahlström, H.; et al. Mediterranean diet habits in older individuals: Associations with cognitive functioning and brain volumes. Exp. Gerontol. 2013, 48, 1443–1448. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Lapiscina, E.H.; Clavero, P.; Toledo, E.; Estruch, R.; Salas-Salvadó, J.; San Julián, B.; Sanchez-Tainta, A.; Ros, E.; Valls-Pedret, C.; Martinez-Gonzalez, M.Á. Mediterranean diet improves cognition: The PREDIMED-NAVARRA randomised trial. J. Neurol. Neurosurg. Psychiatry 2013, 84, 1318–1325. [Google Scholar] [CrossRef] [PubMed]
- Arranz, S.; Chiva-Blanch, G.; Valderas Martínez, P.; Medina-Remón, A.; Lamuela-Raventós, R.M.; Estruch, R. Wine, beer, alcohol and polyphenols on cardiovascular disease and cancer. Nutrients 2012, 4, 759–781. [Google Scholar] [CrossRef] [PubMed]
- Chiva-Blanch, G.; Magraner, E.; Condines, X.; Valderas-Martínez, P.; Roth, I.; Arranz, S.; Casas, R.; Navarro, M.; Hervas, A.; Sisó, A.; et al. Effects of alcohol and polyphenols from beer on atherosclerotic biomarkers in high cardiovascular risk men: A randomized feeding trial. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Anastasiou, C.A.; Yannakoulia, M.; Kosmidis, M.H.; Dardiotis, E.; Hadjigeorgiou, G.M.; Sakka, P.; Arampatzi, X.; Bougea, A.; Labropoulos, I.; Scarmeas, N. Mediterranean diet and cognitive health: Initial results from the Hellenic Longitudinal Investigation of Ageing and Diet. PLoS ONE 2017, 12, e0182048. [Google Scholar] [CrossRef] [PubMed]
- Gorelick, P.B.; Scuteri, A.; Black, S.E. American Heart Association Stroke Council, Council on Epidemiology and Prevention, Councilon Cardiovascular Nursing, Council on Cardiovascular Radiology and Intervention, and Council on Cardiovascular Surgery and Anesthesia. Vascular contributions to cognitive impairment and dementia: A statement for health care professionals from the American heart association/american stroke association. Stroke 2011, 42, 2672–2713. [Google Scholar] [PubMed]
- Loy, C.T.; Twigg, S.M. Growth factors, AGEing, and the diabetes link in Alzheimer’s disease. J. Alzheimers Dis. 2009, 16, 823–831. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.J.; Blumenthal, J.A.; Hoffman, B.M.; Cooper, H.; Strauman, T.A.; Welsh-Bohmer, K.; Browndyke, J.N.; Sherwood, A. Aerobic exercise and neurocognitive performance: A meta-analytic review of randomized controlled trials. Psychosom. Med. 2010, 72, 239–252. [Google Scholar] [CrossRef] [PubMed]
- Valls-Pedret, C.; Sala-Vila, A.; Serra-Mir, M.; Corella, D.; de la Torre, R.; Martínez-González, M.Á.; Martínez-Lapiscina, E.H.; Fitó, M.; Pérez-Heras, A.; Salas-Salvadó, J.; et al. Mediterranean diet and age-related cognitive decline: A randomized clinical trial. JAMA Intern. Med. 2015, 175, 1094–1103. [Google Scholar] [CrossRef] [PubMed]
- Scarmeas, N.; Stern, Y.; Mayeux, R.; Manly, J.J.; Schupf, N.; Luchsinger, J.A. Mediterranean diet and mild cognitive impairment. Arch. Neurol. 2009, 66, 216–225. [Google Scholar] [CrossRef] [PubMed]
- Gardener, S.; Gu, Y.; Rainey-Smith, S.R.; Keogh, J.B.; Clifton, P.M.; Mathieson, S.L.; Taddei, K.; Mondal, A.; Ward, V.K.; Scarmeas, N.; et al. Adherence to a Mediterranean diet and Alzheimer’s disease risk in an Australian population. Transl. Psychiatry 2012, 2, e164. [Google Scholar] [CrossRef] [PubMed]
- Pitsavos, C.; Panagiotakos, D.B.; Tzima, N.; Chrysohoou, C.; Economou, M.; Zampelas, A.; Stefanadis, C. Adherence to the Mediterranean diet is associated with total antioxidant capacity in healthy adults: The ATTICA study. Am. J. Clin. Nutr. 2005, 82, 694–699. [Google Scholar] [CrossRef]
- Salas-Salvadó, J.; Garcia-Arellano, A.; Estruch, R.; Marquez-Sandoval, F.; Corella, D.; Fiol, M.; Gómez-Gracia, E.; Viñoles, E.; Arós, F.; Herrera, C.; et al. Components of the Mediterranean-type food pattern and serum inflammatory markers among patients at high risk for cardiovascular disease. Eur. J. Clin. Nutr. 2008, 62, 651–659. [Google Scholar] [CrossRef]
- Von Bernhardi, R.; Eugenín, J. Alzheimer’s disease: Redox dysregulation as a common denominator for diverse pathogenic mechanisms. Antioxid. Redox Signal. 2012, 16, 974–1031. [Google Scholar] [CrossRef]
- Sastre, M.; Klockgether, T.; Heneka, M.T. Contribution of inflammatory processes to Alzheimer’s disease: Molecular mechanisms. Int. J. Dev. Neurosci. 2006, 24, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Abuznait, A.H.; Qosa, H.; Busnena, B.A.; ElSayed, K.A.; Kaddoumi, A. Olive-oil-derived oleocanthal enhances β-amyloid clearance as a potential neuroprotective mechanism against Alzheimer’s disease: In vitro and in vivo studies. ACS Chem. Neurosci. 2013, 4, 973–982. [Google Scholar] [CrossRef] [PubMed]
- Bulló, M.; Lamuela-Raventós, R.; Salas-Salvadó, J. Mediterranean diet and oxidation: Nuts and olive oil as important sources of fat and antioxidants. Curr. Top. Med. Chem. 2011, 11, 1797–1810. [Google Scholar] [CrossRef] [PubMed]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly) phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antiox. Redox Signal. 2013, 18, 1818–1892. [Google Scholar] [CrossRef] [PubMed]
- Solfrizzi, V.; Panza, F.; Frisardi, V.; Seripa, D.; Logroscino, G.; Imbimbo, B.P.; Pilotto, A. Diet and Alzheimer’s disease risk factors or prevention: The current evidence. Expert Rev. Neurother. 2011, 11, 677–708. [Google Scholar] [CrossRef] [PubMed]
- Escamilla-Hurtado, M.L.; Escamilla-Hurtado, M.G. Los alimentos fermentados que consumían nuestros bisabuelos prehispánicos. Ciencia 2007, 58, 75–84. [Google Scholar]
- Nelson, M. The Barbarian’s Beverage: A History of Beer in Ancient Europe; Routledge: London, UK, 2005. [Google Scholar]
- Beer Composition and Nutritional Elements. Data from the National Nutrient Database for Standard Reference (USDA). Available online: https://ndb.nal.usda.gov/ndb/search/list (accessed on 1 May 2019).
- Ares-Peón, I.A.; Garrote, G.; Domínguez, H.; Parajó, J.C. Phenolics production from alkaline hydrolysis of autohydrolysis liquors. CyTA J. Food 2016, 14, 255–265. [Google Scholar] [CrossRef]
- Porretta, S.; Donadini, G. A Preference Study for No Alcohol Beer in Italy Using Quantitative Concept Analysis. J. Inst. Brew. 2008, 114, 315–321. [Google Scholar] [CrossRef]
- INSERM Collective Expertise Centre. Alcohol: Social Damages, Abuse, and Dependence. Institut National de la Santé et de la Recherche Médicale, 2003. Available online: https://www.ncbi.nlm.nih.gov/books/NBK10785/ (accessed on 30 May 2019).
- Kok, E.H.; Karppinen, T.T.; Luoto, T.; Alafuzoff, I.; Karhunen, P.J. Beer Drinking Associates with Lower Burden of Amyloid Beta Aggregation in the Brain: Helsinki Sudden Death Series. Alcohol Clin. Exp Res. 2016, 40, 1473–1478. [Google Scholar] [CrossRef]
- Collins, M.A.; Neafsey, E.J.; Mukamal, K.J.; Gray, M.O.; Parks, D.A.; Das, D.K.; Korthuis, R.J. Alcohol in moderation, cardioprotection, and neuroprotection: Epidemiological considerations and mechanistic studies. Alcohol Clin. Exp. Res. 2009, 33, 206–219. [Google Scholar] [CrossRef]
- Downer, B.; Jiang, Y.; Zanjani, F.; Fardo, D. Effects of alcohol consumption on cognition and regional brain volumes among older adults. Am. J. Alzheimers Dis. Other Demen. 2015, 30, 364–374. [Google Scholar] [CrossRef] [PubMed]
- Heymann, D.; Stern, Y.; Cosentino, S.; Tatarina-Nulman, O.; N Dorrejo, J.; Gu, Y. The association between alcohol use and the progression of Alzheimer’s disease. Curr. Alzheimers Res. 2016, 13, 1356–1362. [Google Scholar] [CrossRef]
- Mukamal, K.J.; Kuller, L.H.; Fitzpatrick, A.L.; Longstreth-Jr, W.T.; Mittleman, M.A.; Siscovick, D.S. Prospective study of alcohol consumption and risk of dementia in older adults. JAMA 2003, 289, 1405–1413. [Google Scholar] [CrossRef] [PubMed]
- Luchsinger, J.A.; Tang, M.X.; Siddiqui, M.; Shea, S.; Mayeux, R. Alcohol intake and risk of dementia. J. Am. Geriatrics Soc. 2004, 52, 540–546. [Google Scholar] [CrossRef] [PubMed]
- Winkler, C.; Wirleitner, B.; Schroecksnadel, K.; Schennach, H.; Fuchs, D. Beer down-regulates activated peripheral blood mononuclear cells in vitro. Int. Immunopharmacol. 2006, 6, 390–395. [Google Scholar] [CrossRef] [PubMed]
- Polak, J.; Bartoszek, M.; Stanimirova, I. A study of the antioxidant properties of beers using electron paramagnetic resonance. Food Chem. 2013, 14, 3042–3049. [Google Scholar] [CrossRef] [PubMed]
- Sripanyakorn, S.; Jugdaohsingh, R.; Elliott, H.; Walker, C.; Mehta, P.; Shoukru, S. The silicon content of beer and its bioavailability in healthy volunteers. Brit. J. Nutr. 2004, 91, 403–409. [Google Scholar] [CrossRef]
- Peña, A.; Meseguer, I.; González-Muñoz, M.J. Influencia del consumo moderado de cerveza sobre la toxicocinética del aluminio: Estudio agudo. Nutr. Hosp. 2007, 22, 371–376. [Google Scholar]
- Granero, S.; Vicente, M.; Aguilar, V.; Martínez-Para, M.C.; Domingo, J.L. Effects of beer as a source of dietary silicon on aluminum absorption and retention in mice. Trace Elem. Electrol. 2004, 21, 28–32. [Google Scholar] [CrossRef]
- González-Muñoz, M.J.; Peña, A.; Meseguer, M.I. Role of beer as a possible protective factor in preventing Alzheimer’s disease. Food Chem. Toxicol. 2008, 46, 49–56. [Google Scholar] [CrossRef]
- Merino, P.; Santos-López, J.A.; Mateos, C.; Meseguer, I.; Garcimartín, A.; Bastida, S.; Sánchez-Muniz, F.J.; Benedí, J.; González-Muñoz, M.J. Can nonalcoholic beer, silicon and hops reduce the brain damage and behavioral changes induced by aluminum nitrate in young male Wistar rats? Food Chem. Toxicol. 2018, 118, 784–794. [Google Scholar] [CrossRef] [PubMed]
- Sasaoka, N.; Sakamoto, M.; Kanemori, S.; Kan, M.; Tsukano, C.; Takemoto, Y.; Kakizuka, A. Long-term oral administration of hop flower extracts mitigates Alzheimer phenotypes in mice. PLoS ONE 2014, 9, e87185. [Google Scholar] [CrossRef] [PubMed]
- Callemien, D.; Jerkovic, V.; Rozenberg, R.; Collin, S. Hop as an interesting source of resveratrol for brewers: Optimization of the extraction and quantitative study by liquid chromatography/atmospheric pressure chemical ionization tandem mass spectrometry. J. Agric. Food Chem. 2005, 53, 424–429. [Google Scholar] [CrossRef] [PubMed]
- Gillette-Guyonnet, S.; Andrieu, S.; Nourhashemi, F.; de la Gueronniere, V.; Grandjean, H.; Vellas, B. Cognitive impairment and composition of drinking water in women: Finding of the EPIDOS study. Am. J. Clin. Nutr. 2005, 81, 897–902. [Google Scholar] [CrossRef] [PubMed]
- Gillette-Guyonnet, S.; Andrieu, S.; Vellas, B. The potential influence of silica presents in drinking water on Alzheimer’s disease and associated disorders. J. Nutr. Health Aging 2007, 11, 119–124. [Google Scholar] [PubMed]
- Garcimartín, A.; Merino, J.J.; Santos-López, J.A.; López-Oliva, M.E.; González, M.P.; Sánchez-Muniz, F.J.; Benedí, J. Silicon as neuroprotector or neurotoxic in the human neuroblastoma SH-SY5Y cell line. Chemosphere 2015, 135, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Maldonado, M.D.; Moreno, H.; Calvo, J.R. Melatonin present in beer contributes to increase the levels of melatonin and antioxidant capacity of the human serum. Clin. Nutr. 2009, 28, 188–191. [Google Scholar] [CrossRef] [PubMed]
- Rosales-Corral, S.A.; Acuña-Castroviejo, D.; Coto-Montes, A.; Boga, J.A.; Manchester, L.C.; Fuentes-Broto, L.; Reiter, R.J. Alzheimer’s disease: Pathological mechanisms and the beneficial role of melatonin. J. Pineal Res. 2012, 52, 167–202. [Google Scholar] [CrossRef] [PubMed]
- Matsubara, E.; Bryant-Thomas, T.; Pacheco Quinto, J.; Henry, T.L.; Poeggeler, B.; Herbert, D.; Shoji, M. Melatonin increases survival and inhibits oxidative and amyloid pathology in a transgenic model of Alzheimer’s disease. J. Neurochem. 2003, 85, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- Masilamoni, J.G.; Jesudason, E.P.; Dhandayuthapani, S.; Ashok, B.S.; Vignesh, S.; Jebaraj, W.C.; Paul, S.F.; Jayakumar, R. The neuroprotective role of melatonin against amyloid beta peptide injected mice. Free Radic. Res. 2008, 42, 661–673. [Google Scholar] [CrossRef]
- Hornedo-Ortega, R.; Cerezo, A.B.; Troncoso, A.M.; Garcia-Parrilla, M.C.; Mas, A. Melatonin and other tryptophan metabolites produced by yeasts: Implications in cardiovascular and neurodegenerative diseases. Front. Microbiol. 2016, 6, 1565. [Google Scholar] [CrossRef]
- Szwajgier, D. Anticholinesterase activity of phenolic acids and their derivatives. Zeitschrift für Naturforschung C 2013, 68, 125–132. [Google Scholar] [CrossRef]
- Szwajgier, D.; Borowiec, K. Phenolic acids from malt are efficient acetylcholinesterase and butyrylcholinesterase inhibitors. J. Inst. Brew. 2012, 118, 40–48. [Google Scholar] [CrossRef]
- Hamaguchi, T.; Ono, K.; Murase, A.; Yamada, M. Phenolic compounds prevent Alzheimer’s pathology through different effects on the amyloid-β aggregation pathway. Am. J. Patol. 2009, 175, 2557–2565. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Wang, J.; Chen, X.; Liu, P.; Wang, S.; Song, F.; Zhang, Z.; Zhu, F.; Huang, X.; Liu, J.; et al. The prenylflavonoid xanthohumol reduces Alzheimer-like changes and modulates multiple pathogenic molecular pathways in the neuro2a/APPswe cell model of AD. Front. Pharmacol. 2018, 9, 199. [Google Scholar] [CrossRef] [PubMed]
- Ano, Y.; Dohata, A.; Taniguchi, Y.; Hoshi, A.; Uchida, K.; Takashima, A.; Nakayama, H. Iso-α-acids, bitter components of beer, prevent inflammation and cognitive decline induced in a mouse model of Alzheimer’s disease. J. Biol. Chem. 2017, 292, 3720–3728. [Google Scholar] [CrossRef] [PubMed]
- Ano, Y.; Hoshi, A.; Ayabe, T.; Takashima, A.; Nakayama, H. Iso-α-acids, bitter components in beer, improve hippocampus-dependent memory via dopamine release. Alzheimers Demen. J Alzheimer Assoc. 2018, 14, 304. [Google Scholar] [CrossRef]
- Ano, Y.; Yoshikawa, M.; Takaichi, Y.; Michikawa, M.; Uchida, K.; Nakayama, H.; Takashima, A. Iso-α-Acids, bitter components in beer, suppress inflammatory responses and attenuate neural hyperactivation in the hippocampus. Front. Pharmacol. 2019, 10, 81. [Google Scholar] [CrossRef]
- Fukuda, T.; Uchida, K.; Nakayama, H.; Ano, Y. Short-term administration of iso-α-acids increases transthyretin transcription in the hippocampus. Biochem. Biophys. Res. Commun. 2018, 507, 471–475. [Google Scholar] [CrossRef] [PubMed]
- Ayabe, T.; Ohya, R.; Ano, Y. Hop-Derived Iso-α-Acids in Beer Improve Visual Discrimination and Reversal Learning in Mice as Assessed by a Touch Panel Operant System. Front. Behav. Neurosci. 2019, 13, 67. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.W.; Barofsky, E.; Kennedy, J.A.; Deinzer, M.L. Hop (Humulus lupulus L.) proanthocyanidins characterized by mass spectrometry, acid catalysis, and gel permeation chromatography. J. Agric. Food Chem. 2003, 51, 4101–4110. [Google Scholar] [CrossRef] [PubMed]
- De Keukeleire, D.; De Cooman, L.; Rong, H.; Heyerick, A.; Kalita, J.; Milligan, S.R. Functional properties of hop polyphenols. Basic Life Sci. 1999, 66, 739–760. [Google Scholar] [PubMed]
- Nikolic, D.; Li, Y.; Chadwick, L.R.; Grubjesic, S.; Schwab, P.; Metz, P.; van Breemen, R.B. Metabolism of 8-prenylnaringenin, a potent phytoestrogen from hops (Humulus lupulus), by human liver microsomes. Drug Metab. Dispos. 2004, 32, 272–279. [Google Scholar] [CrossRef] [PubMed]
- Dong, L.; Hou, Y.; Li, F.; Piao, Y.; Zhang, X.; Zhang, X.; Li, C.; Zhao, C. Characterization of volatile aroma compounds in different brewing barley cultivars. J. Sci. Food Agric. 2015, 95, 915–921. [Google Scholar] [CrossRef] [PubMed]
- Van Cleemput, M.; Heyerick, A.; Libert, C.; Swerts, K.; Philippé, J.; De Keukeleire, D.; Haegeman, G.; De Bosscher, K. Hop bitter acids efficiently block inflammation independent of GRalpha, PPARalpha, or PPARgamma. Mol. Nutr. Food Res. 2009, 53, 1143–1155. [Google Scholar] [CrossRef]
- Rancán, L.; Paredes, S.D.; García, I.; Muñoz, P.; García, C.; López de Hontanar, G.; de la Fuente, M.; Vara, E.; Tresguerres, J.A.F. Protective effect of xanthohumol against age-related brain damage. J. Nutr. Biochem. 2017, 49, 133–140. [Google Scholar] [CrossRef]
- Chumlea, W.C. Editorial: Silica, a mineral of unknown but emerging health importance. J. Nutr. Health Aging 2007, 11, 93. [Google Scholar]
- Santos-López, J.A.; Garcimartín, A.; Merino, P.; López-Oliva, M.E.; Bastida, S.; Benedí, J.; Sánchez-Muniz, F.J. Effects of silicon vs. hydroxytyrosol-enriched restructured pork on liver oxidation status of aged rats fed high-Saturated/high-Cholesterol Diet. PLoS ONE 2016, 11, e0147469. [Google Scholar] [CrossRef]
- Wu, Z.H.; Du, Y.M.; Xue, H.; Wu, Y.; Zhou, B. Aluminium induces neurodegeneration and its toxicity arises from increased iron accumulation and reactive oxygen species (ROS) production. Neurobiol. Aging 2012, 33, e1–e199. [Google Scholar] [CrossRef]
- Parry, R.; Plowman, D.; Trevor Delves, H.; Roberts, N.B.; Birchall, J.D.; Bellia, J.P. Silicon and aluminum interactions in haemodialysis patients. Nephrol. Dial. Transp. 1998, 13, 1759–1762. [Google Scholar] [CrossRef][Green Version]
- Reffitt, D.M.; Jugdaohsingh, R.; Thompson, R.P.; Powell, J.J.; Hampson, G.N. Silicic acid: Its gastrointestinal uptake and urinary excretion in men and effects on aluminum excretion. Inorganic Biochem. 1999, 76, 141–147. [Google Scholar] [CrossRef]
- Birchall, J.D.; Chappell, J.S. Aluminium, water chemistry, and Alzheimer’s disease. Lancet 1989, 29, 953. [Google Scholar] [CrossRef]
- Martin, K.R. Silicon: The health benefits of a metalloid. Met Ions Life Sci. 2013, 13, 451–473. [Google Scholar] [PubMed]
- Vargas, C.R.; Wajner, M.; Sirtori, L.R.; Goulart, L.; Chiochetta, M.; Coelho, D. Evidence that oxidative stress is increased in patients with X-linked adrenoleukodystrophy. Biochem. Biophys. Acta 2004, 1688, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Gómez, M.; Esperza, J.L.; Nogués, M.R.; Giralt, M.; Cabré, M.; Domingo, J.L. Pro-oxidant activity of aluminum in the rat hippocampus: Gene expression of antioxidant enzymes alters melatonin administration. Free Rad. Biol. Med. 2005, 38, 104–111. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, R.A.; Jain, J.C. Scavenger receptor class B type I expression and elemental analysis in cerebellum and parietal cortex regions of the Alzheimer’s disease brain. J. Neurol. Sci. 2002, 196, 45–52. [Google Scholar] [CrossRef]
- Valls-Belles, V.; Codoner-Franch, P.; Villarino Marin, A.; Martinez-Alvarez, J.R. Effect of hop supplementation in oxidative metabolism and inflammation parameters. In Proceedings of the Functional Foods in Europe: International Developments in Science and Health Claims: Summary Report of an International Symposium, Portomaso, Malta, 9–11 May 2007; p. 45. [Google Scholar]
- Thambyrajah, J.; Townend, J.N. Homocysteine and atherothrombosis mechanism for injury. Eur. Heart J. 2000, 21, 967–974. [Google Scholar] [CrossRef] [PubMed]
- Ravaglia, G.; Forti, P.; Maioli, F.; Muscari, A.; Scchetti, L. Homocysteine and cognitive function in healthy elderly community dwellers in Italy. Am. J. Clin. Nutr. 2003, 77, 668–673. [Google Scholar] [CrossRef] [PubMed]
- Whitmer, R.A.; Haan, M.N.; Miller, J.W.; Yaffe, K. Hormone replacement therapy and cognitive performance: The role of homocysteine. J. Gerontol. Biol. Sci. Med. Sci. 2003, 58, 324–330. [Google Scholar] [CrossRef]
- Reiter, R.J.; Tan, D.X.; Maldonado, M.D. Melatonin as an antioxidant: Physiology versus pharmacology. J. Pineal Res. 2005, 39, 215–216. [Google Scholar] [CrossRef]
- Bieschke, J.; Zhang, Q.; Powers, E.T.; Lerner, R.A.; Kelly, J.W. Oxidative metabolites accelerate Alzheimer’s amyloidogenesis by a two-step mechanism, eliminating the requirement for nucleation. Biochemistry 2005, 44, 4977–4983. [Google Scholar] [CrossRef] [PubMed]
- Walton, R.J. Chronic aluminum intake causes Alzheimer’s disease: Applied Sir Austin Bradford Hill’s causality criteria. J. Alzheimers Dis. 2014, 40, 765–838. [Google Scholar] [CrossRef] [PubMed]
- Gordillo Bastidas, D.; Gordillo Bastidas, E. (Eds.) Metabolismo del alcohol. In Nutrición Molecular; McGraw Hill Education: Mexico City, Mexico, 2015; pp. 179–188. [Google Scholar]
- Casarrubea, M.; Sorbera, F.; Crescimanno, G. Structure of rat behavior in hole-board: I) multivariate analysis of response to anxiety. Physiol. Behav. 2009, 96, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Espejo, E.F.; Mir, D. Structure of the rat’s behaviour in the hot plate test. Behav. Brain Res. 1993, 56, 171–176. [Google Scholar] [CrossRef]
- Platt, B.; Fiddler, G.; Riedel, G.; Henderson, Z. Toxicidad por aluminio en el cerebro de rata: Pruebas histoquímicas e inmunocitoquímicas. Brain Res. Bull. 2001, 55, 257–267. [Google Scholar] [CrossRef]
- Jacqmin-Gadda, H.; Commenges, D.; Letenneur, L.; Dartigues, J.F. Silica and aluminum in drinking water and cognitive impairment in the elderly. Epidemiology 1996, 7, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Ogasawara, Y.; Ohata, E.; Sakamoto, T.; Ishii, K.; Takahashi, H.; Tanabe, S. A model of aluminum exposure associated with lipid peroxidation in rat brain. Biol. Trace Elem. Res. 2003, 96, 91–201. [Google Scholar] [CrossRef]
- Lang, U.E.; Beglinger, C.; Schweinfurth, N.; Walter, M.; Borgwardt, S. Nutritional aspects of depression. Cell Physiol. Biochem. 2015, 37, 1029–1043. [Google Scholar] [CrossRef]
- Gordillo-Bastidas, E.; Panduro, A.; Gordillo-Bastidas, D.; Zepeda-Carrillo, E.A.; García-Bañuelos, J.J.; Muñoz-Valle, J.F.; Bastidas-Ramírez, B.E. Polymorphisms of alcohol metabolizing enzymes in indigenous Mexican population: Unusual high frequency of CYP2E1*c2 allele. Alcohol Clin. Exp. Res. 2010, 34, 142–149. [Google Scholar] [CrossRef]
- Fraga Elenes, R.A.; Hagelsieb Pizano, O. Enfermedad de Alzheimer: Impacto de los ácidos grasos. In Nutrición Molecular; Gordillo Bastidas, D., Gordillo Bastidas, E., Eds.; McGraw Hill Education: Mexico City, Mexico, 2015; pp. 293–306. [Google Scholar]
- Zou, F.; Chai, H.S.; Younkin, C.S.; Allen, M.; Crook, J.; Pankratz, V.S.; Carrasquillo, M.M.; Rowley, C.N.; Nair, A.A.; Middha, S.; et al. Brain expression genome-wide association study (eGWAS) identifies human disease-associated variants. PLoS Genet. 2012, 8, e1002707. [Google Scholar] [CrossRef]
- Li, Z.; Zhu, H.; Zhang, L.; Qin, C. The intestinal microbiome and Alzheimer’s disease: A review. Animal Model Exp. Med. 2018, 1, 180–188. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Li, G.; Huang, P.; Liu, Z.; Zhao, B. The Gut Microbiota and Alzheimer’s Disease. J. Alzheimers Dis. 2017, 58, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Eguilaz, M.; Ramon-Trapero, J.L.; Perez-Martinez, L.; Blanco, J.R. The microbiota-gut-brain axis and its great projections. Rev. Neurol. 2019, 68, 111–117. [Google Scholar] [PubMed]
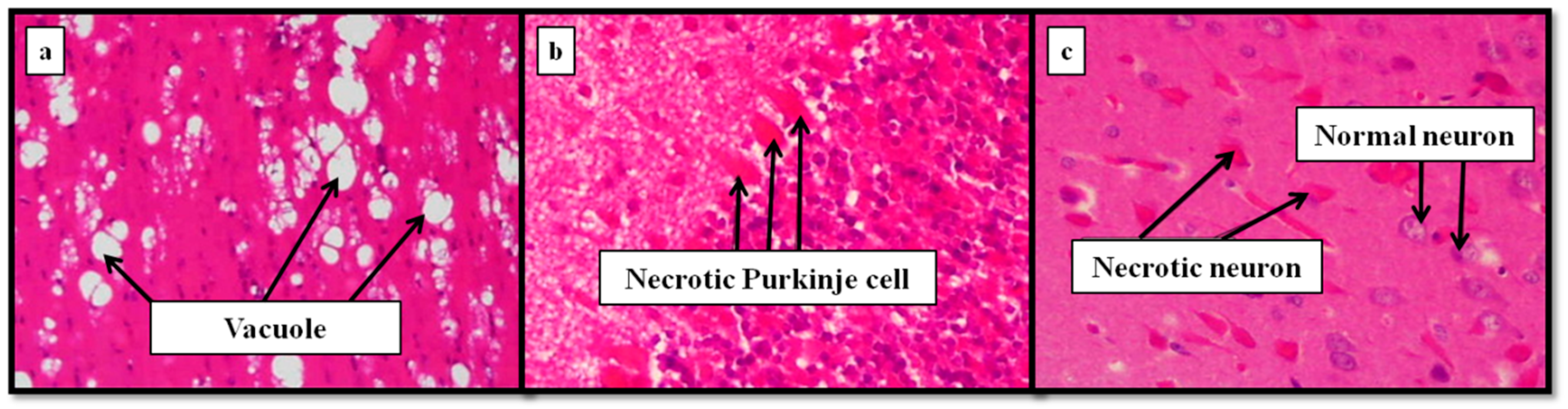
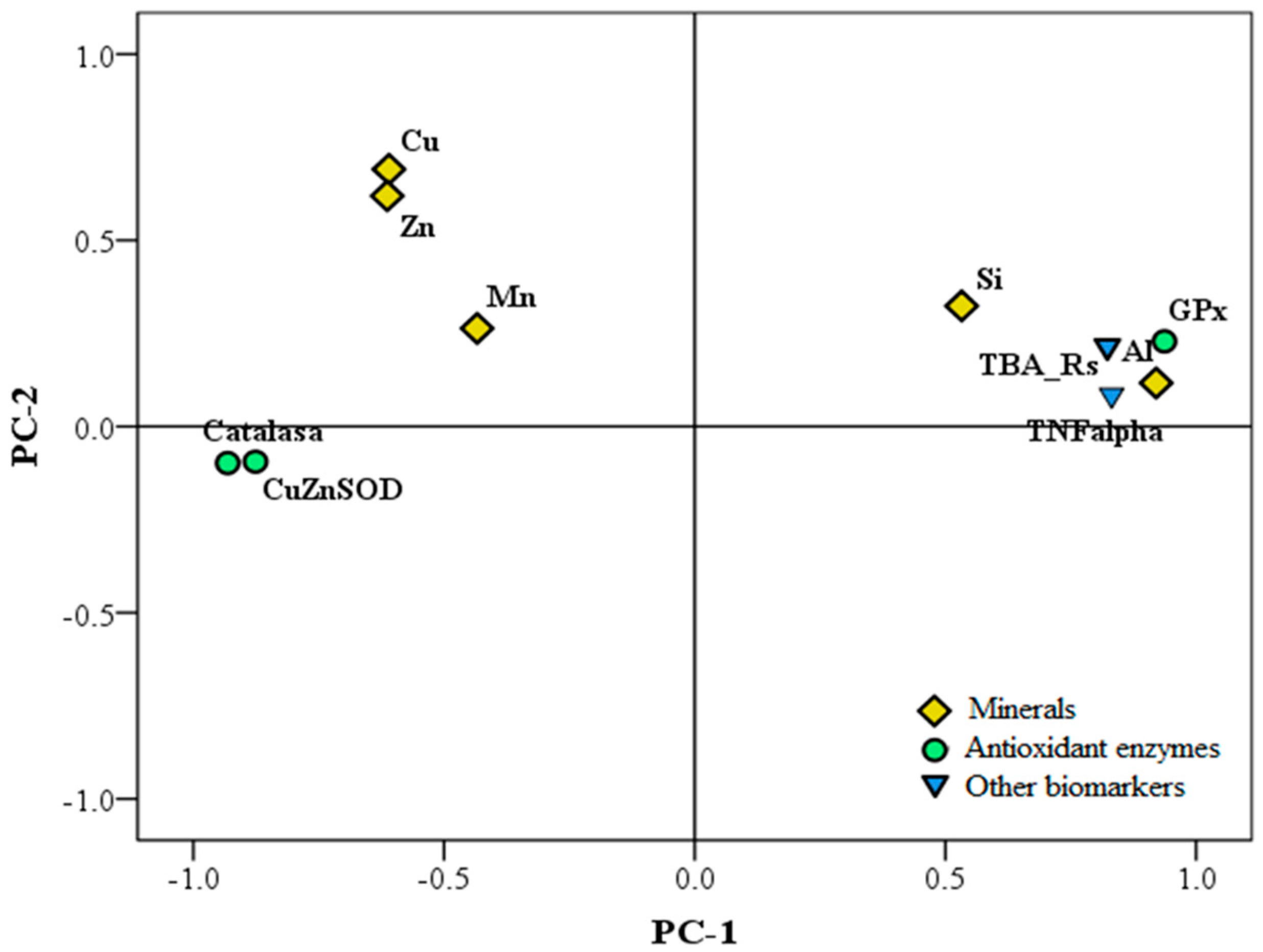
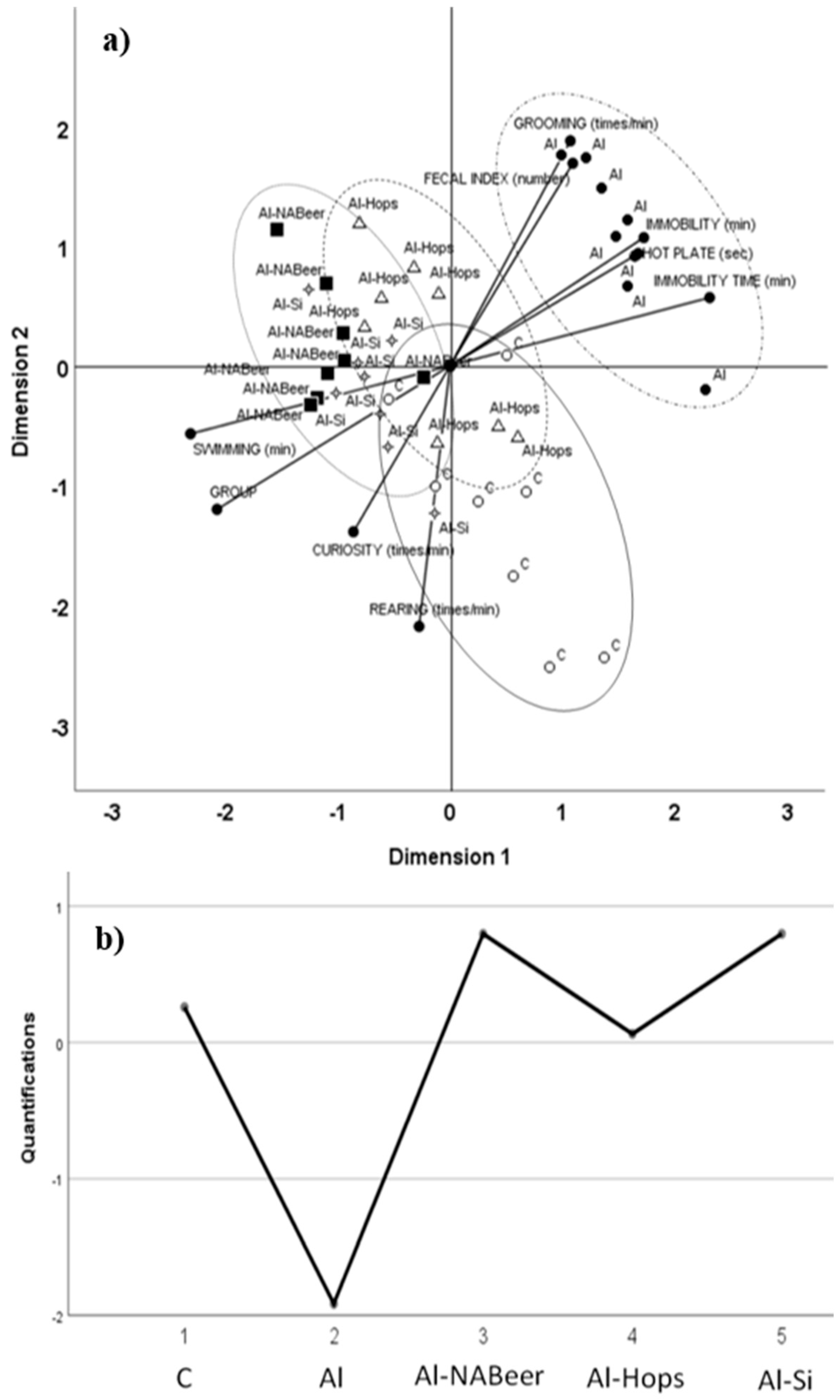
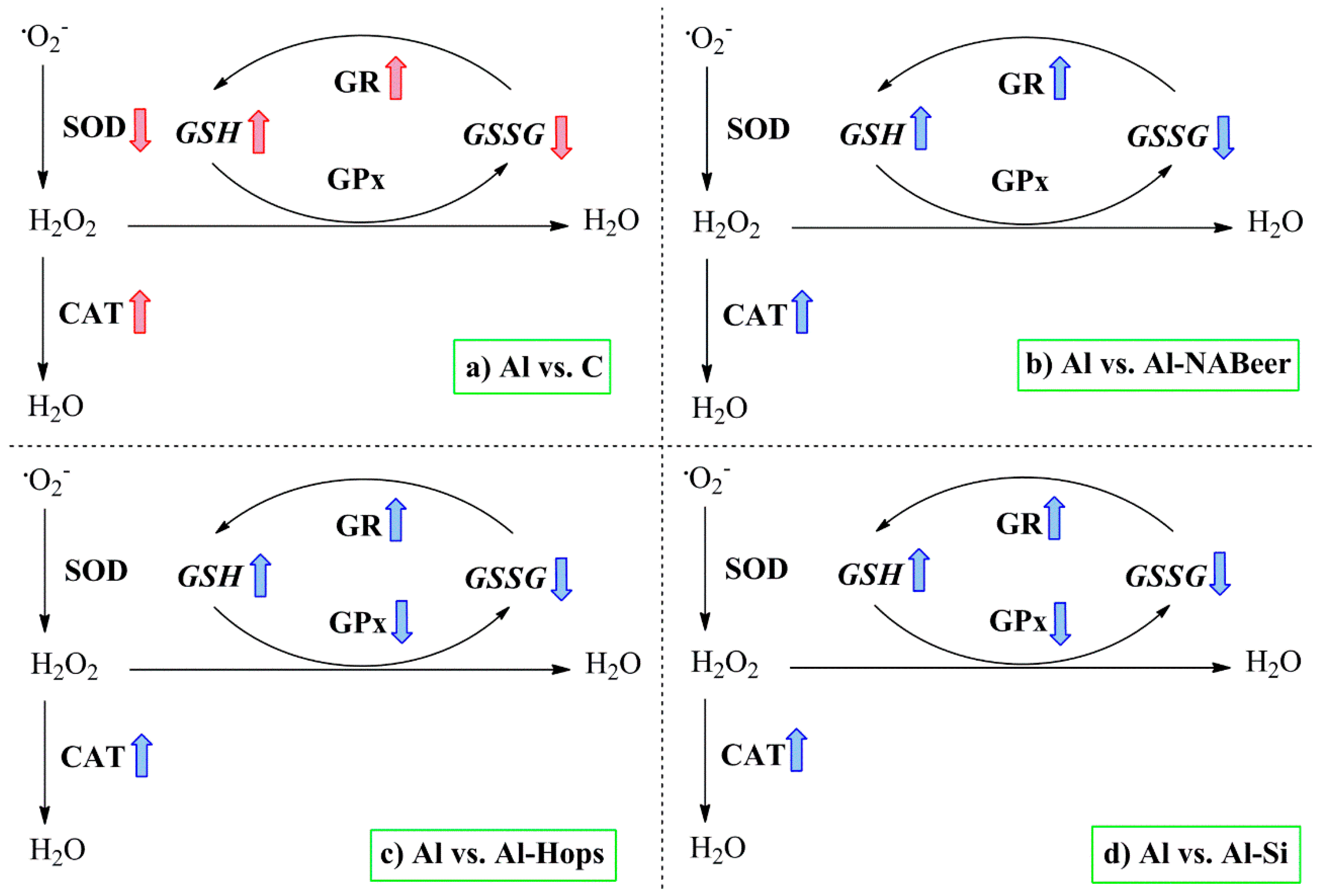
| Country | L Per Capita | Total National Consumption |
|---|---|---|
| China | 29 | 43266 |
| United States | 74.8 | 24245 |
| Brazil | 60.4 | 12654 |
| Germany | 104.2 | 8412 |
| Russia | 58.60 | 8405 |
| Mexico | 65.1 | 7988 |
| Japan | 41.4 | 5251 |
| Spain | 84.8 | 3909 |
| Poland | 100.8 | 3892 |
| Canada | 57.7 | 2093 |
| Argentina | 49 | 1980 |
| Czech Republic | 143.3 | 1959 |
| Netherlands | 69.8 | 1186 |
| Austria | 106 | 928 |
| Belgium | 67.4 | 769 |
| Nutrient | Units | Mean Content/100 g |
|---|---|---|
| Proximates | ||
| Energy | Kcal | 91.97 |
| Water | g | 43 |
| Protein | g | 0.46 |
| Total lipid (fat) | g | 0.0 |
| Carbohydrate, by difference | g | 3.55 |
| Fiber, total dietary | g | 0.0 |
| Minerals | ||
| Calcium (Ca) | mg | 4 |
| Cooper (Cu) | mg | 0.01 |
| Iron (Fe) | mg | 0.02 |
| Magnesium (Mg) | mg | 6 |
| Manganese (Mn) | mg | 0.02 |
| Silicon (Si) | mg | 1.92 |
| Selenium (Se) | µg | 0.6 |
| Fluoride (F) | µg | 44.2 |
| Phosphorus (P) | mg | 14 |
| Potassium (K) | mg | 27 |
| Sodium (Na) | mg | 4 |
| Zinc (Zn) | mg | 0.01 |
| Vitamins | ||
| Vitamin C, total ascorbic acid | mg | 0.0 |
| Thiamin B1 | mg | 0.005 |
| Riboflavin B2 | mg | 0.025 |
| Niacin B3 | mg | 0.513 |
| Pantothenic acid B5 | mg | 0.041 |
| Vitamin B-6 | mg | 0.046 |
| Folate, DFE | µg | 6 |
| Choline, total | mg | 10.1 |
| Cobalamine B12 | µg | 0.0 |
| Vitamin A, RAE | µg | 0.0 |
| Vitamin E (α-tocophenol) | mg | 0.0 |
| Vitamin D | IU | 0.0 |
| Vitamin K (phylloquinine) | µg | 0.0 |
| Lipids | ||
| Saturated fatty acids | g | 0 |
| Monounsaturated fatty acids | g | 0 |
| Polyunsaturated fatty acids | g | 0 |
| Cholesterol | mg | 0 |
| Aminoacids | ||
| Alanine | g | 0.012 |
| Aspartic acid | g | 0.016 |
| Glutamic acid | g | 0.047 |
| Glycine | g | 0.013 |
| Proline | g | 0.035 |
| Ethyl alcohol | g | 3.9 * |
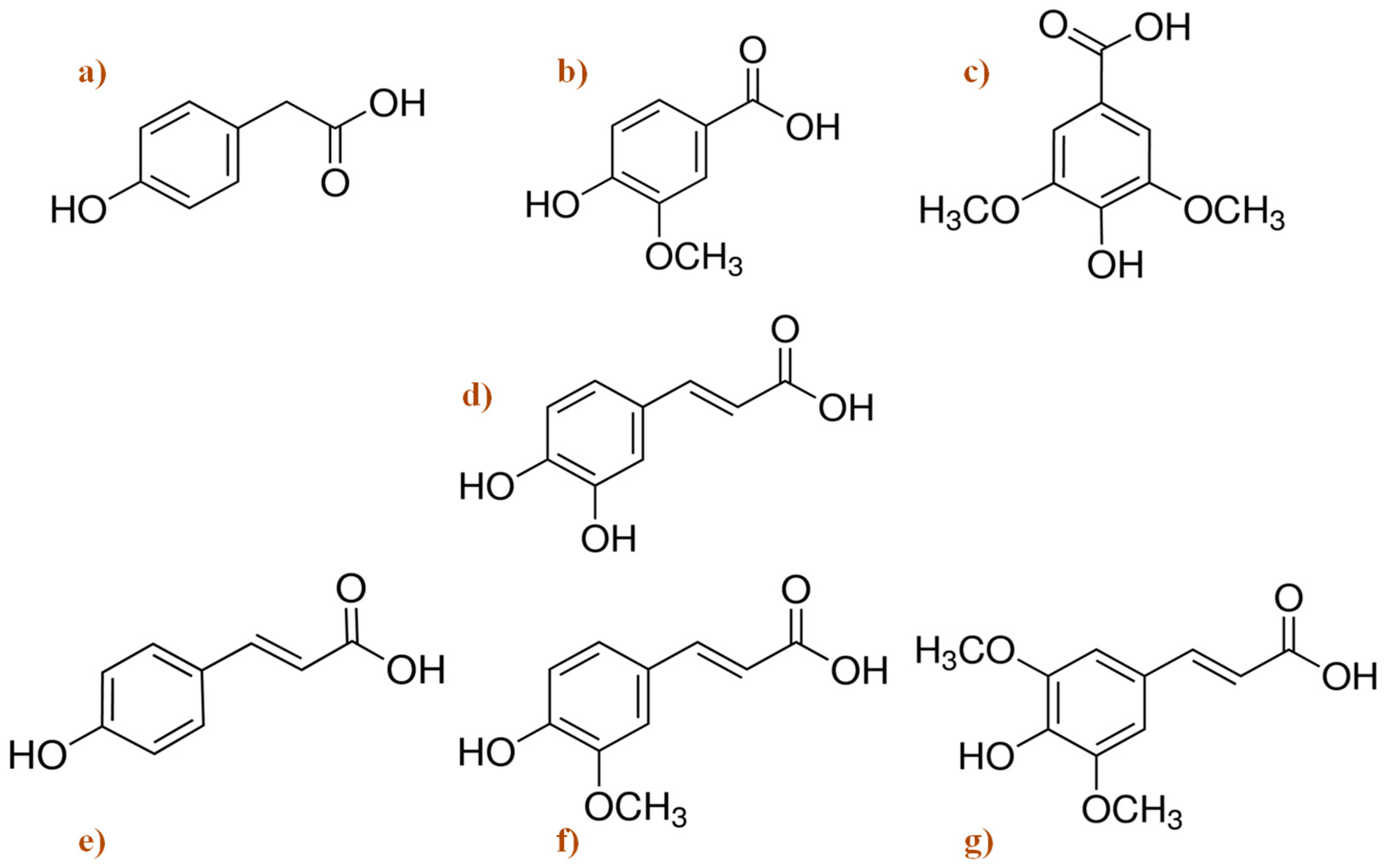
| Phenolic Compound (g/100 mL) | |||
|---|---|---|---|
| Alkylmethoxyphenols | 5-Caffeoylquinic acid | 0.08 | |
| 4-Vinylguaiachol | 0.15 | Caffeic acid | 0.03 |
| Alkylphenols | Ferulic acid | 0.26 | |
| 3-Methylcatechol | 1.00 × 10−4 | Sinapic acid | 0.02 |
| 4-Ethylcatechol | 6.00 × 10−4 | Chalcones | |
| 4-Vinyphenol | 4.53 × 10−3 | Xanthohumol | 1.41 × 10−3 |
| Hydroxybenzaldehydes | Flavanones | ||
| Vanillin | 0.02 | Isoxanthohumol | 0.04 |
| Hydroxybenzoketones | Naringin | 7.5 × 10−4 | |
| 2,3-Dihydroxy-1-guaiacylpropanone | 3.4 × 10−3 | 8-Prenylnaringenin | 1.04 × 10−3 |
| Hidroxycoumarins | 6-Prenylnaringenin | 2.59 × 10−3 | |
| Esculin | 0.02 | 6-Genarnylnaringenin | 4.29 × 10−4 |
| Umbelliferone | 1.67 × 10−3 | Hydroxyphenylacetic acids | |
| 4-Hidroxycoumarin | 0.11 | 4-Hydroxyphenilacetic acid | 0.03 |
| Hidroxyphenylacetic acids | Homovanillic acid | 0.05 | |
| 4-Hydroxyphenylacetic acid | 0.03 | Tyrosol | 0.32 |
| Homovanillic acid | 0.05 | (+)-Catechin | 0.11 |
| Benzoic acid derivatives | (-)-Epicatechin | 0.06 | |
| Gallic acid 3-O-gallate | 0.3 | Procyanidin dimer B3 | 0.16 |
| 2,6-Dihydroxybenzoic acid | 0.09 | Prodelphinidin trimer GCGCC | 0.04 |
| 2-Hydroxybenzoic acid | 0.20 | Prodelphinidin trimer GCCC | 1.00 × 10−2 |
| 3-Hydroxybenzoic acid | 0.03 | Prodelphinidin trimer CGCC | 0.02 |
| 3,5-Dihydroxybenzoic acid | 0.03 | Flavonols | |
| Syringic acid | 0.02 | Quercetin (3-O-arabinoside) | 5.83 × 10−4 |
| Protocatechuic acid | 0.05 | Quercetin | 6.67 × 10−3 |
| Vanillic acid | 0.07 | 3,7-Dimetilquercitin | 2.50 × 10−4 |
| Gallic acid | 0.07 | Myricetin | 6.67 × 10−4 |
| Gentisic acid | 0.03 | Quercetin (3-O-rutinoside) | 0.09 |
| Cinnamic acids | Isoflavonoids | ||
| p-Coumaric acid | 0.10 | Daidzein | 0.005 |
| m-Coumaric acid | 0.02 | Genistein | 0.01 |
| o- Coumaric acid | 0.15 | Biochanin A | 0.005 |
| 4-Caffeoylquinic acid | 0.01 | Flavones | |
| Other polyphenols | Apigenin | 4.17 × 10−3 | |
| Catechol | 1.10 × 10−3 | α-acids (humolones) | 0.17 |
| Pyrogallol | 4.70 × 10−3 | Iso-α-acids (iso-humolones) | 0.06 × 10−10 |
| Compound | Species and Trial Mode | Formulation and Doses | Results | Reference |
|---|---|---|---|---|
| Beer | Clinical trial in humans with 125 males of the Helsinki Sudden Death autopsy | Total life consumption | Neuroprotective effect by a possible depletion of Aβ aggregation in brain | Kok et al. [111] |
| Beer | Cohort study with 360 patients in early AD, biannually evaluation up to 19–28 years. | Heavy drinkers (≥8 alcoholic drinks/week); mild-moderate drinkers (1–7 alcoholic drinks/week); abstainers | Increasing standard drinks of hard liquor, but not beer or wine, was associated with a faster rate of cognitive decline, such as AD | Heymann et al [114] |
| Beer | Cardiovascular Health Study, cohort study with 5888 men and women aged ≥65 years and 5–7 years follow up. | 12oz of beer with later magnetic resonance imaging of the brain and cognitive capacity evaluation. | Limited beer consumption resulted in a decreased risk of dementia or AD | Mukamal et al. [115] |
| Beer | Cohort study with 980 community-dwelling individuals aged ≥65 years without dementia at baseline, annually evaluation. | Light drinkers (1 serving/month to 6 servings/week); moderate drinkers (1–3 servings/day); heavy drinkers (≥4 servings/day) | Light to moderate alcohol intake was associated with a lower risk of dementia and AD, whereas intake of beer and liquor was not associated with incident dementia. | Luchsinger et al. [116] |
| Beer | Review of the observational studies, trials, reviews, and meta-analyses in humans | Review from 45 reports since the early 1990’s | More than half of the papers indicate that low consumption of beer reduced the risk of dementia. While a minority suggests the risk of neurodegeneration due to its ethanol content | Collins et al. [112] |
| Beer | Immune response evaluation in human peripheral blood mononuclear cells with 48-h treatment | Different beer types with 2%–4% (v/v) ethanol | Beer reduces the production of neopterin and the tryptophan degradation. Its immunosuppressive capacity seems related to its anti-inflammatory mechanisms. | Winkler et al. [117] |
| Beer | In vitro experiments of pure samples | Electron paramagnetic resonance spectroscopy and antioxidant activity of different types of beer | Beers exhibit antioxidant properties | Polak et al. [118] |
| Alcohol | Review of the human, rodents and cell culture neuroprotection evaluations and epidemiological studies | Wine, beer, and liquor administration and consumption | Alcohol-dependent neuroprotected state appears linked to an activation of signal transduction processes of reactive oxygen species. The alcohol intake ameliorates inflammatory pathways and increases hippocampal acetylcholine release. Alcohol exposure is inversely associated with dementia through protective changes in cerebral vasculature | Collins et al. [112] |
| Beer/silicon | Human intake with 6-h bioavailability evaluation of silicon-enriched beer | 0.6 L beer containing 22.5 mg Si and 4.6% (v/v) ethanol | Silicon in beer in monomeric form, is readily bioavailable in healthy volunteers | Sripanyakorn et al. [119] |
| Beer/silicic acid | Acute three-day study with male NMRI mice | Equivalent to moderate-high consumption in humans (1 L/day; 55 g alcohol/day). | Beer, mainly associated with its silicon content, reduces dietary aluminum toxicokinetics and bioavailability through a reduction of aluminum uptake in the digestive tract and by increasing its fecal excretion | Peña et al. [120] |
| Beer/silicon | Male NMRI mice on 3-month trial with neuroprotective evaluation | 2.5 mL beer/per week (5.5% (v/v)), and 40 mg silicon/L/day | Silicon appears to be effective in preventing aluminum accumulation in mouse’s brain. Nonetheless, silicon could act either as neuroprotector or neurotoxic | Granero et al. [121] |
| Beer/silicic acid | Male NMRI mice on 3-month evaluation | 450 mg of aluminum nitrate,0.5 mL beer* (5.5% (v/v))/day, and 9 µg silicon/day *equivalent to moderate to high consumption in humans (1 L/day) | Beer consumption, and its content on bioavailable silicon, reduces the accumulation of aluminum in the body and brain tissue, the lipid peroxidation, and protected against the neurotoxic effects through the regulation of antioxidant enzymes | González-Muñoz et al. [48,122] |
| Beer/silicic acid | Male NMRI mice on 3-month trial | 450 mg of aluminum nitrate,0.5 mL beer* (5.5% (v/v))/day, and 9 µg silicon/day *equivalent to moderate to high consumption in humans (1 L/day) | Silicic acid and beer block the metal imbalance, inflammation, and antioxidant defense impairment induced by aluminum intoxication in the brain | González-Muñoz et al. [43] |
| Non-alcoholic beer/hops extract/organic silicon (Silicium organique G57™) | Male Wistar rats on 3-month trial with behavioral, organs, and in vitro studies of neurodegeneration | 450 µg aluminum nitrate/kg/day; 2 mg hops extract/day; 250 µg silicon/day; 5 mL NA-beer/day* *equivalent to moderate/high consumption in humans (1 L/day) | NA-beer, hops, and silicon ameliorated behavioral modifications, blocked the negative effect on the in vivo and in vitro antioxidant status, and reduced the inflammation markers in brain induced by aluminum intoxication | Merino et al. [123] |
| Hops extract | Homozygous transgenic mice (V717F) and heterozygous transgenic mice (V717F/P267S) on a 2-, 6-, 11-, and 18-month trial, and HEK293A cell culture | Hops extract added to drinking water at a dose of 2 g extract/L | Hops extract reduced Alzheimer’s phenotypes in mice and prevented the emotional disturbance at the 18 months AD-mice. The extract significantly reduced Aβ production in cultured cells and presented γ-secretase inhibitory activity | Sasaoka et al. [124] |
| Hops pellets | Hops pellets to obtain its chemical characterization | Chemical and quantitative determination of hops compounds | The presence of resveratrol in hops highlights the potential health-promoting effect of moderate beer consumption | Callemien et al. [125] |
| Silicon | Human trial in 7598 women ≥75 years for evaluation of cognitive function and neuroprotective effect | Silica content in drinking water | Silicon in drinking water might reduce the risk of AD | Gillette-Guyonnet et al. [126] |
| Silicon | Review from human trials that evaluate the neuroprotective effect of silica in drinking water | Reviews from tidies of silica in drinking water | Aluminum in water seems to have a deleterious effect when the silica concentrations were low, while the risk of AD was reduced in subjects who had higher daily silica intake | Gillette-Guyonnet et al. [127] |
| Silicon | Male Wistar rats on a 12-week trial | 0.5 mg aluminum/kg/day, and 2 mg silicon/kg/day | Silicon is considered an important protector against lipid peroxidation induced by aluminum intake | Noremberg et al. [18] |
| Silicon | SH-5HSY neuroblastoma cells line with a 24-h treatment | Ladder concentration of 50–250 ng silicon/mL | Silicon treatment reduced TBARS levels, it also may act as neuroprotector by inducing antiapoptotic effects at low doses and may act as neurotoxic by regulating necrosis and apoptosis mechanisms at high doses. | Garcimartín et al. [128] |
| Beer/melatonin | Human trial with healthy volunteers (4 men and 3 women) aged 20 to 30 years. | Different beer brands with diverse ethanol content | Melatonin contained in beer showed antioxidant, oncostatic, and immune enhancer activities | Maldonado et al. [129] |
| Melatonin | Review of the main effects on AD pathology | Main research publications on the melatonin pathological mechanisms related to AD through different approaches | Prevention of amyloid overproduction, reduction of tau phosphorylation, antioxidant ability, modulates inflammation, anticholinesterase agent, prevents mitochondrial damage and apoptosis. | Rosales-Corral et al. [130] |
| Melatonin | Transgenic mice Tg2576 on an 8-, 9.5-, 11-, and 15.5-month survival study trial to evaluate neuroprotective effect | 0.5 mg/mL melatonin administration to obtain Aβ measurements in brain, quantitative immunoblots of APP levels, and nitrotyrosine measurements | The melatonin administration proved a reduction of major AD markers and brain Aβ levels | Matsubara et al. [131] |
| Melatonin | Male swiss albino mice in a 5-day neuroprotective effect evaluation experiment | 50 mg melatonin/kg body weight and 5µg Aβ42-1 intracerebroventricularly administrated | The treatment reduced Aβ-induced oxidative stress, related to ROS and proinflammatory cytokines IL6 and IL1-β, and the intracellular calcium levels and acetylcholinesterase activity in the neocortex and hippocampus regions | Masilamoni et al. [132] |
| Melatonin | Review of the in vivo and in vitro studies | Reviews about the effects on the prevention of neurodegenerative diseases and their molecular mechanism | Melatonin is significantly decreased in elderly AD individuals and associated with the emergence of AD, it exhibits a protective effect on the cholinergic system and protects brain neurons from damage and death by increasing viability in hippocampal neurons and glial cells | Hornedo-Ortega et al. [133] |
| Phenolic compounds from beer | In vitro anti-AChE and anti-BChE activities of simple phenolic acids | Phenolic solutions at their beer concentration. IC50 values at 336 and 160 mM calculated for AChE and BChE, respectively | Phenolic acids from beer can play a role in neuroprotection by through an inhibition of cholinesterases | Szwajgier [134] |
| Phenolic compounds from malt | In vitro studies of the phenolic fraction profile from several malt types. | Phenolic solution at different concentrations to inhibit AChE and BChE enzymes (~0.38–1 mM/L) | The main phenolic compounds from malt (ferulic acid, p-coumaric, 4-hydroxybenzoic, and sinapic acids). Among them, the ferulic and p-coumaric acids showed a high neuroprotective role and can be considered as possible anti-AD agents | Szwajgier and Borowiec [135] |
| Phenolic compounds from beer | Female Tg2576 mice on a 14 months trial to evaluate the pathology of AD | Different diets including 0.5% phenolic compounds evaluated through immunohistochemistry and morphometry of Aβ deposits | The extracted phenolic compounds prevent AD pathology development through the regulation of Aβ aggregation pathway | Hamaguchi et al. [136] |
| Xanthohumol | Wild-type murine neuroblastoma Neuro2a cells (N2a/WT) and N2a stably transfected with human APP Swedish mutant (N2a/APP) on a 24-h treatment | 0–25 μM Xanthohumol in cell culture and later comparative proteomics, immunocytochemistry of Aβ1-40 and Aβ1-42 | Xanthohumol suppresses Aβ production and tau hyperphosphorylation via APP processing and the GSK-3β pathway. Thus, it may have potential effects for the treatment of AD | Huang et al. [137] |
| Iso-α-acids | Alzheimer’s model in 5xFAD mice on a three-month period to evaluate cognitive function in the progression of dementia | 0.05% (w/w) of the iso-α-acids | The iso-α-acids suppressed the neuroinflammation markers IL-1β and chemokine, and improve cognitive function | Ano et al. [138] |
| Iso-α-acids | Male Crl:CD1(ICR) mice, vagotomized male ICR mice, and Sprague-Dawley (SD) rats on a 3-month period to evaluate cognitive function test, especially hippocampus-dependent memory | 1 mg/kg iso-α-acids | Iso-α-acids activate dopamine D1 receptor-signaling in the hippocampus and improves spatial and object recognition memory functions | Ano et al. [139,140] |
| Iso-α-acids | Male C57BL/6J mice treated for 3 months | Dietary intake of 0.05% (w/w) iso-α-acids to evaluate episodic and spatial memory and microglia analysis | Reduced inflammation in the brain and prevent the cognitive impairment associated with normal aging | Ano et al. [140] |
| Iso-α-acids | Male C57BL/6J mice on an AD model (5xFAD transgenic) on a 7-day trial | 1 mg/kg iso-α-acids and later transcriptome analysis | Reduced Aβ in the brain and increased the expression of transthyretin in the hippocampus, thus displayed protective effects AD pathologies | Fukuda et al. [141] |
| Beer/iso-α-acids from hops extract | Male C57BL/6J mice on a long-term cognitive evaluation trial | 1 mg extract/kg equivalent to 4.8 mg/day in humans (60 kg body weight) or 0.17–0.3 L/day of beers | Iso-α-acids could improve working memory in dementia and visual/reversal discrimination learning, which are considered high-order cognitive functions. | Ayabe et al. [142] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sánchez-Muniz, F.J.; Macho-González, A.; Garcimartín, A.; Santos-López, J.A.; Benedí, J.; Bastida, S.; González-Muñoz, M.J. The Nutritional Components of Beer and Its Relationship with Neurodegeneration and Alzheimer’s Disease. Nutrients 2019, 11, 1558. https://doi.org/10.3390/nu11071558
Sánchez-Muniz FJ, Macho-González A, Garcimartín A, Santos-López JA, Benedí J, Bastida S, González-Muñoz MJ. The Nutritional Components of Beer and Its Relationship with Neurodegeneration and Alzheimer’s Disease. Nutrients. 2019; 11(7):1558. https://doi.org/10.3390/nu11071558
Chicago/Turabian StyleSánchez-Muniz, Francisco José, Adrián Macho-González, Alba Garcimartín, Jorge Arturo Santos-López, Juana Benedí, Sara Bastida, and María José González-Muñoz. 2019. "The Nutritional Components of Beer and Its Relationship with Neurodegeneration and Alzheimer’s Disease" Nutrients 11, no. 7: 1558. https://doi.org/10.3390/nu11071558
APA StyleSánchez-Muniz, F. J., Macho-González, A., Garcimartín, A., Santos-López, J. A., Benedí, J., Bastida, S., & González-Muñoz, M. J. (2019). The Nutritional Components of Beer and Its Relationship with Neurodegeneration and Alzheimer’s Disease. Nutrients, 11(7), 1558. https://doi.org/10.3390/nu11071558





