Anaemia and Its Relation to Demographic, Socio-economic and Anthropometric Factors in Rural Primary School Children in Hai Phong City, Vietnam
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. School Selection
2.3. Participant Recruitment
2.4. Blood Sample Collection and Haematological Measures
2.5. Anthropometric Measures
2.6. Socio-economic Status Data
2.7. Data Analysis
3. Results
3.1. Participants
3.2. The Prevalence of Anaemia and Associated Factors
3.3. The Prevalence of Microcytic Anaemia and Associated Factors
3.4. The Prevalence of Normocytic Anaemia and Associated Factors
3.5. Relationship between Hb and MCV, BMI-For-Age Z-Score, and Socio-Economic Factors
3.6. Intracluster Correlation Coefficients
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Worldwide Prevalence of Anaemia 1993–2005: WHO Global Database on Anaemia; World Health Organization: Geneva, Switzerland, 2008. [Google Scholar]
- Lopez, A.D.; Mathers, C.D.; Ezzati, M.; Jamison, D.T.; Murray, C.J.L. Global and regional burden of disease and risk factors, 2001: Systematic analysis of population health data. Lancet 2006, 367, 1747–1757. [Google Scholar] [CrossRef]
- Subramaniam, G.; Girish, M. Iron Deficiency Anemia in Children. Indian J. Pediatr. 2015, 82, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Balarajan, Y.; Ramakrishnan, U.; Özaltin, E.; Shankar, A.H.; Subramanian, S.V. Anaemia in low-income and middle-income countries. Lancet 2011, 378, 2123–2135. [Google Scholar] [CrossRef]
- Bobonis, G.J.; Miguel, E.; Puri-Sharma, C. Anemia and school participation. J. Hum. Resour. 2006, 41, 692–721. [Google Scholar] [CrossRef]
- Roy, C.N. Anemia of inflammation. ASH Educ. Prog. Book 2010, 2010, 276–280. [Google Scholar] [CrossRef] [PubMed]
- Mockenhaupt, F.P.; Rong, B.; Günther, M.; Beck, S.; Till, H.; Kohne, E.; Thompson, W.N.; Bienzle, U. Anaemia in pregnant Ghanaian women: Importance of malaria, iron deficiency, and haemoglobinopathies. Trans. R. Soc. Trop. Med. Hyg. 2000, 94, 477–483. [Google Scholar] [CrossRef]
- WHO/UNICEF/UNU. Iron Deficiency Anaemia Assessment, Prevention, and Control. A Guide for Programme Managers. Available online: http://www.who.int/nutrition/publications/micronutrients/anaemia_iron_deficiency/WHO_NHD_01.3/en/ (accessed on 6 May 2019).
- Viện Dinh Dưỡng. Tổng điều tra dinh dưỡng 2009–2010.; Nhà xuất bản Y học: Hanoi, Vietnam, 2010. [Google Scholar]
- Hall, A.; Bobrow, E.; Brooker, S.; Jukes, M.; Nokes, K.; Lambo, J.; Guyatt, H.; Bundy, D.; Adjei, S.; Wen, S.-T. Anaemia in schoolchildren in eight countries in Africa and Asia. Public Health Nutr. 2001, 4, 749–756. [Google Scholar] [PubMed]
- Choi, H.-J.; Lee, H.-J.; Jang, H.B.; Park, J.Y.; Kang, J.-H.; Park, K.-H.; Song, J. Effects of maternal education on diet, anemia, and iron deficiency in Korean school-aged children. BMC Public Health 2011, 11, 870. [Google Scholar] [CrossRef]
- Alderman, H.; Linnemayr, S. Anemia in Low-Income Countries is Unlikely to be Addressed by Economic Development without Additional Programs. Food Nutr. Bull. 2009, 30, 265–269. [Google Scholar] [CrossRef]
- UNICEF. Strategy for Improved Nutrition of Children and Women in Developing Countries: A UNICEF Policy Review; UNICEF: New York, NY, USA, 1990. [Google Scholar]
- Burchard, G.D.; Cramer, J.P.; Ehrhardt, S.; Mantel, C.; Bienzle, U.; Mockenhaupt, F.P.; Kubo, M.; Kaiser, S.; Otchwemah, R.N. Malaria, Anemia, and Malnutrition in African Children—Defining Intervention Priorities. J. Infect. Dis. 2006, 194, 108–114. [Google Scholar]
- Zimmermann, M.B.; Zeder, C.; Muthayya, S.; Winichagoon, P.; Chaouki, N.; Aeberli, I.; Hurrell, R.F. Adiposity in women and children from transition countries predicts decreased iron absorption, iron deficiency and a reduced response to iron fortification. Int. J. Obes. 2008, 32, 1098. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, J.; Smuts, C.M.; Aeberli, I.; Malan, L.; Tjalsma, H.; Zimmermann, M.B. Overweight impairs efficacy of iron supplementation in iron-deficient South African children: A randomised controlled intervention. Int. J. Obes. 2013, 37, 24–30. [Google Scholar] [CrossRef] [PubMed]
- DeMaeyer, E.M.; Dallman, P.; Gurney, J.M.; Hallberg, L.; Sood, S.; Srikantia, S.; Organization, W.H. Preventing and Controlling Iron Deficiency Anaemia through Primary Health Care: A Guide for Health Administrators and Programme Managers; World Health Organization: Geneva, Switzerland, 1989. [Google Scholar]
- Pinhas-Hamiel, O.; Newfield, R.S.; Koren, I.; Agmon, A.; Lilos, P.; Phillip, M. Greater prevalence of iron deficiency in overweight and obese children and adolescents. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 416–418. [Google Scholar] [CrossRef] [PubMed]
- Manios, Y.; Moschonis, G.; Chrousos, G.P.; Lionis, C.; Mougios, V.; Kantilafti, M.; Tzotzola, V.; Skenderi, K.P.; Petridou, A.; Tsalis, G.; et al. The double burden of obesity and iron deficiency on children and adolescents in Greece: The Healthy Growth Study. J. Hum. Nutr. Diet. 2013, 26, 470–478. [Google Scholar] [CrossRef] [PubMed]
- Cepeda-Lopez, A.C.; Osendarp, S.J.; Melse-Boonstra, A.; Aeberli, I.; Gonzalez-Salazar, F.; Feskens, E.; Villalpando, S.; Zimmermann, M.B. Sharply higher rates of iron deficiency in obese Mexican women and children are predicted by obesity-related inflammation rather than by differences in dietary iron intake. Am. J. Clin. Nutr. 2011, 93, 975–983. [Google Scholar] [CrossRef] [PubMed]
- Le, H.T.; Brouwer, I.D.; Verhoef, H.; Nguyen, K.C.; Kok, F.J. Anemia and intestinal parasite infection in school children in rural Vietnam. Asia Pac. J. Clin. Nutr. 2007, 16, 716–723. [Google Scholar] [PubMed]
- Nhien, N.V.; Khan, N.C.; Yabutani, T.; Ninh, N.X.; Chung le, T.K.; Motonaka, J.; Nakaya, Y. Relationship of Low Serum Selenium to Anemia among Primary School Children Living in Rural Vietnam. J. Nutr. Sci. Vitaminol. 2008, 54, 454–459. [Google Scholar] [CrossRef] [PubMed]
- Hop, L.T.; Berger, J. Multiple micronutrient supplementation improves anemia, micronutrient nutrient status, and growth of Vietnamese infants: Double-blind, randomised, placebo-controlled trial. J. Nutr. 2005, 135, 660S–665S. [Google Scholar] [CrossRef] [PubMed]
- Hieu, N.T.; Sandalinas, F.; de Sesmaisons, A.S.; Laillou, A.; Tam, N.P.; Khan, N.C.; Bruyeron, O.; Wieringa, F.T.; Berger, J. Multi-micronutrient-fortified biscuits decreased the prevalence of anaemia and improved iron status, whereas weekly iron supplementation only improved iron status in Vietnamese school children. Br. J. Nutr. 2012, 108, 1419–1427. [Google Scholar] [CrossRef] [PubMed]
- National Institute of Nutrition. Results of the National Nutrition Survey and Introduction of the National Nutrition Strategies Period 2011–2020. Available online: http://viendinhduong.vn/vi/chien-luoc-dinh-duong-2011-2020/phan-thu-hai-chien-luoc-quoc-gia-ve-dinh-duong-giai-doan-2011-den-2020-va-tam-nhin-den-nam-2030.html (accessed on 27 May 2013).
- Hoang, N.; Orellana, L.; Le, T.; Gibson, R.; Worsley, A.; Sinclair, A.; Szymlek-Gay, E. Anthropometric Status among 6–9-Year-Old School Children in Rural Areas in Hai Phong City, Vietnam. Nutrients 2018, 10, 1431. [Google Scholar] [CrossRef] [PubMed]
- WHO. Haemoglobin Concentrations for the Diagnosis of Anaemia and Assessment of Severity. Available online: http://www.who.int/vmnis/indicators/haemoglobin/en/ (accessed on 2 March 2016).
- Gibson, R.S. Principles of Nutritional Assessment, 2nd ed.; Oxford University Press: New York, NY, USA, 2005. [Google Scholar]
- Fulgoni, V.L., III; Agarwal, S.; Kellogg, M.D.; Lieberman, H.R. Establishing Pediatric and Adult RBC Reference Intervals With NHANES Data Using Piecewise Regression. Am. J. Clin. Pathol. 2018, 151, 128–142. [Google Scholar] [CrossRef] [PubMed]
- CDC/National Center for Health Statistics. NHANES: Anthropometry Procedures Manual; CDC/National Center for Health Statistics: Hyattsville, MD, USA, 2004.
- WHO. WHO Anthro (version 3.2.2, January 2011) and Macros. Available online: http://www.who.int/childgrowth/software/en/ (accessed on 19 October 2016).
- WHO. BMI-for-Age (5–19 Years). Available online: http://www.who.int/growthref/who2007_bmi_for_age/en/index.html (accessed on 14 June 2012).
- Nandy, S.; Irving, M.; Gordon, D.; Subramanian, S.; Smith, G.D. Poverty, child undernutrition and morbidity: New evidence from India. Bull. World Health Organ. 2005, 83, 210–216. [Google Scholar] [PubMed]
- Huong, L.T. Anemia among School Children in Vietnam: The Efficacy of Iron Fortification; Wageningen University: Wageningen, The Netherlands, 2006. [Google Scholar]
- Trần Thúy Nga; Nguyễn Xuân Ninh; Nguyễn Thị Lâm; Nguyễn Công Khẩn. Tình trạng dinh dưỡng, thiếu máu, nhiễm ký sinh trùng đường ruột và một số yếu tố liên quan của trẻ em tiểu học ở một số vùng nông miền Bắc. Tạp chí DD&TP 2006, 2, 15–19. [Google Scholar]
- People Committee of Hai Phong City. Competitive Advantage; People Committee of Hai Phong City: Hai Phong, Vietnam, 2017.
- NIN. Đánh giá hoạt động và hiệu quả của dự án Phòng chống suy dinh dưỡng trẻ em sau 5 năm triển khai (1999-2004). Báo cáo đề tài nghiên cứu cấp Bộ [Evaluation the PEMC for five years of implementing (1999–2004). Report on the Ministry Level Study; NIN: Hanoi, Vietnam, 2006; pp. 14–16. [Google Scholar]
- Bhutta, Z.A.; Salam, R.A.; Das, J.K. Meeting the challenges of micronutrient malnutrition in the developing world. Br. Med. Bull. 2013, 106, 7–17. [Google Scholar] [CrossRef] [PubMed]
- Rasha Aziz, S.; Meray, R.L. The Prevalence of anemia among informal primary school children: A community based study in Rural Upper Egypt. Epidemiol. Biostat. Public Health 2016, 13. [Google Scholar] [CrossRef]
- UNICEF. The Faces of Malnutrition. Available online: https://www.unicef.org/nutrition/index_faces-of-malnutrition.html (accessed on 28 April 2015).
- Ngui, R.; Lim, Y.A.L.; Chong Kin, L.; Sek Chuen, C.; Jaffar, S. Association between Anaemia, Iron Deficiency Anaemia, Neglected Parasitic Infections and Socioeconomic Factors in Rural Children of West Malaysia. PLoS Negl. Trop. Dis. 2012, 6, e1550. [Google Scholar] [CrossRef] [PubMed]
- Bhaskaram, P. Micronutrient Malnutrition, Infection, and Immunity: An Overview. Nutr. Rev. 2002, 60, S40–S45. [Google Scholar] [CrossRef] [PubMed]
- Bulliyya, G.; Mallick, G.; Sethy, G.S.; Kar, S.K. Hemoglobin status of non-school going adolescent girls in three districts of Orissa, India. Int. J. Adolesc. Med. Health 2007, 19, 395–406. [Google Scholar] [CrossRef]
- Amarasinghe, G.S.; Naottunna, N.P.G.C.R.; Agampodi, T.C.; Agampodi, S.B. Factors associated with anemia among Sri Lankan primary school children in rural North Central Province. BMC Pediatr. 2017, 17, 87. [Google Scholar] [CrossRef]
- Le Nguyen Bao, K.; Tran Thuy, N.; Nguyen Huu, C.; Khouw, I.; Deurenberg, P. Anemia and iron deficiency in Vietnamese children, 6 to 11 years old. Asia Pac. J. Public Health 2016, 28, 94S–102S. [Google Scholar] [CrossRef]
- Hoàng Thị Đức Ngàn; Lê Thị Hợp. Tỷ lệ TC-BP và một số yếu tố liên quan của trẻ em tại một số trường tiểu học tại Hải Phòng năm 2012; Viện Dinh dưỡng: Hanoi, Vietnam, 2012. [Google Scholar]
- Khatiwada, S.; Gelal, B.; Gautam, S.; Tamang, M.K.; Shakya, P.R.; Lamsal, M.; Baral, N. Anemia among school children in eastern Nepal. J. Trop. Pediatr. 2015, 61, 231–233. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nguyen, B.K.L.; Thi, H.L.; Do, V.A.N.; Thuy, N.T.; Huu, C.N.; Do, T.T.; Deurenberg, P.; Khouw, I. Double burden of undernutrition and overnutrition in Vietnam in 2011: Results of the SEANUTS study in 0·5–11-year-old children. Br. J. Nutr. 2013, 110, S45–S56. [Google Scholar] [CrossRef] [PubMed]
- Alelign, T.; Degarege, A.; Erko, B. Prevalence and factors associated with undernutrition and anaemia among school children in Durbete Town, northwest Ethiopia. Arch. Public Health 2015, 73, 34. [Google Scholar] [CrossRef] [PubMed]
 , n = 77, 9.0%), normocytic anaemia as Hb < 115 g/L and MCV 80–90 fL (
, n = 77, 9.0%), normocytic anaemia as Hb < 115 g/L and MCV 80–90 fL ( , n = 59, 6.9%), macrocytic anaemia as Hb < 115 g/L and MCV > 90 fL (n = 0, 0.0%), and no anaemia was defined as Hb ≥ 115 g/L (
, n = 59, 6.9%), macrocytic anaemia as Hb < 115 g/L and MCV > 90 fL (n = 0, 0.0%), and no anaemia was defined as Hb ≥ 115 g/L ( , n = 724, 84.1 %).
, n = 724, 84.1 %).
 , n = 77, 9.0%), normocytic anaemia as Hb < 115 g/L and MCV 80–90 fL (
, n = 77, 9.0%), normocytic anaemia as Hb < 115 g/L and MCV 80–90 fL ( , n = 59, 6.9%), macrocytic anaemia as Hb < 115 g/L and MCV > 90 fL (n = 0, 0.0%), and no anaemia was defined as Hb ≥ 115 g/L (
, n = 59, 6.9%), macrocytic anaemia as Hb < 115 g/L and MCV > 90 fL (n = 0, 0.0%), and no anaemia was defined as Hb ≥ 115 g/L ( , n = 724, 84.1 %).
, n = 724, 84.1 %).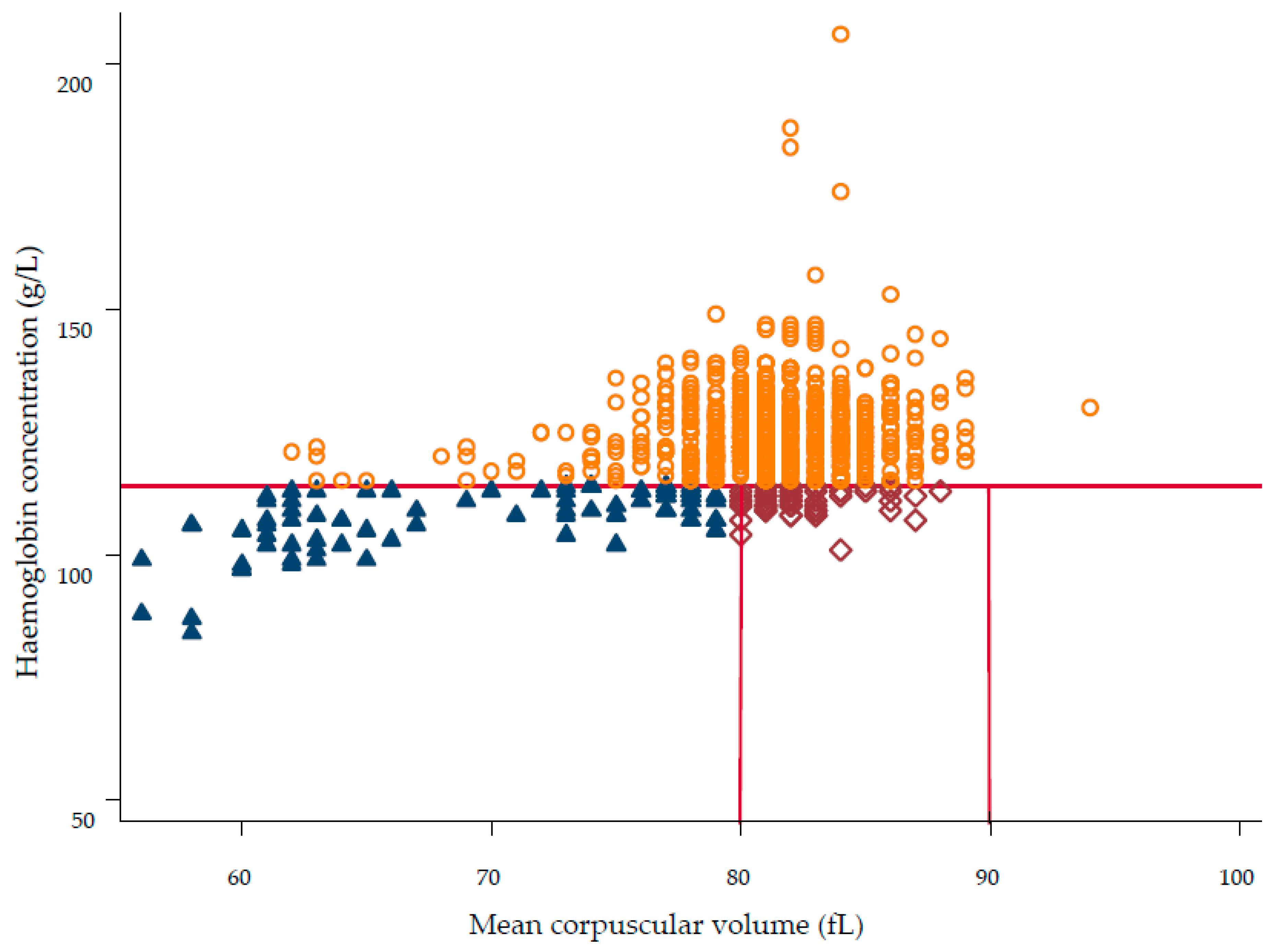
| N | % | ||
|---|---|---|---|
| All Children | 893 | ||
| Sex | Girls | 450 | 50.4 |
| Boys | 443 | 49.6 | |
| Age (months) | 69 to <81 | 303 | 33.9 |
| 81 to <92 | 294 | 32.9 | |
| 92 to ≤108 | 296 | 33.2 | |
| Stunting a | Not stunted | 841 | 94.7 |
| Stunted | 47 | 5.3 | |
| Underweight b | Not underweight | 812 | 91.2 |
| Underweight | 78 | 8.8 | |
| Wasting c | Not wasted | 833 | 93.8 |
| Wasted | 55 | 6.2 | |
| Overweight/ Obesity d | Not overweight/obese | 722 | 81.3 |
| Overweight/obese | 166 | 18.7 | |
| CIAF e | No anthropometric failure | 772 | 86.7 |
| Anthropometric failure e | 118 | 13.3 | |
| Wasting only | 28 | 3.2 | |
| Wasting + underweight | 23 | 2.6 | |
| Wasting + stunting + underweight | 4 | 0.4 | |
| Stunting + underweight | 31 | 3.5 | |
| Stunting only | 12 | 1.3 | |
| Underweight only | 20 | 2.3 | |
| Maternal employment status | Full time employment | 424 | 63.0 |
| Self-employed | 117 | 17.4 | |
| Farmer | 75 | 11.1 | |
| Unemployed | 57 | 8.5 | |
| Maternal education | Above high school | 178 | 26.3 |
| High school | 229 | 33.8 | |
| Below high school | 271 | 39.9 | |
| Monthly household income (million VND) | <6 | 199 | 31.1 |
| 6 to <10 | 209 | 32.7 | |
| ≥10 | 232 | 36.2 | |
| Total (N) | Anaemia (N) | Prevalence, % (95% CI) | Univariate Analysis a | ||||
|---|---|---|---|---|---|---|---|
| OR (95% CI) | P-Value | Global P-Value b | |||||
| All children | 893 | 138 | 12.9 (8.1, 19.9) | ||||
| Sex | Girls (reference) | 450 | 66 | 12.4 (7.5, 19.8) | 1 | ||
| Boys | 443 | 72 | 13.3 (8.2, 21.1) | 1.09 (0.75, 1.58) | 0.664 | ||
| Age (months) | 69 to <81 (reference) | 303 | 54 | 15.4 (9.3, 24.5) | 1 | 0.158 | |
| 81 to <92 | 294 | 45 | 12.9 (7.6, 21.0) | 0.81 (0.52, 1.27) | 0.366 | ||
| 92 to ≤108 | 296 | 39 | 10.4 (5.9, 17.5) | 0.64 (0.40, 1.00) | 0.055 | ||
| Maternal employment status | Self-employed (reference) | 117 | 15 | 11.2 (5.4, 21.7) | 1 | 0.676 | |
| Full time employment | 424 | 67 | 11.8 (6.6, 20.0) | 1.06 (0.57, 1.99) | 0.853 | ||
| Farmer | 75 | 13 | 12.2 (5.6, 24.5) | 1.11 (0.47, 2.58) | 0.815 | ||
| Unemployed | 57 | 11 | 17.2 (8.0, 33.2) | 1.65 (0.68, 4.02) | 0.268 | ||
| Maternal education | Above high school (reference) | 178 | 26 | 13.8 (7.8, 23.2) | 1 | 0.559 | |
| High school | 229 | 33 | 11.1 (6.0, 19.5) | 0.98 (0.54, 1.76) | 0.939 | ||
| Below high school | 271 | 47 | 11.3 (6.0, 20.3) | 1.26 (0.72, 2.20) | 0.425 | ||
| Monthly household income (million VND) | <6 (reference) | 199 | 31 | 13.0 (7.1, 22.5) | 1 | 0.538 | |
| 6 to <10 | 209 | 28 | 10.4 (5.5, 18.6) | 0.78 (0.44, 1.37) | 0.383 | ||
| ≥10 | 232 | 39 | 13.4 (7.5, 22.8) | 1.04 (0.61, 1.77) | 0.893 | ||
| Stunting c | Not stunted (reference) | 841 | 125 | 12.3 (7.7, 19.2) | 1 | ||
| Stunted | 47 | 13 | 24.5 (12.6, 42.4) | 2.31 (1.15, 4.61) | 0.018 | ||
| Underweight d | Not underweight (reference) | 812 | 117 | 12.0 (7.5, 18.6) | 1 | ||
| Underweight | 78 | 21 | 23.6 (13.2, 38.4) | 2.27 (1.30, 3.97) | 0.004 | ||
| Wasting e | Not wasted (reference) | 833 | 122 | 12.1 (7.5, 18.8) | 1 | ||
| Wasted | 55 | 16 | 26.0 (14.0, 43.1) | 2.55 (1.35, 4.84) | 0.004 | ||
| Overweight/ Obesity f | Not overweight/obese (reference) | 722 | 124 | 14.4 (9.2, 21.7) | 1 | ||
| Overweight/obese | 166 | 14 | 7.9 (4.0,14.8) | 0.51 (0.28, 0.92) | 0.026 | ||
| CIAF g | No anthropometric failure (reference) | 772 | 108 | 11.6 (7.2, 18.1) | 1 | ||
| Anthropometric failure | 118 | 30 | 22.0 (12.9, 34.9) | 2.16 (1.34, 3.48) | 0.002 | ||
| Total (N) | Microcytic Anaemia (N) | Prevalence, % (95% CI) | Univariate Analysis a | ||||
|---|---|---|---|---|---|---|---|
| OR (95% CI) | P-Value | Global P-Value b | |||||
| All children | 860 | 77 | 7.9 (5.3, 11.6) | ||||
| Sex | Girls (reference) | 429 | 32 | 6.5 (4.0, 10.4) | 1 | ||
| Boys | 431 | 45 | 9.2 (5.9, 14.0) | 1.45 (0.90, 2.35) | 0.127 | ||
| Age (months) | 69 to <81 (reference) | 272 | 26 | 8.2 (4.9, 13.4) | 1 | 0.444 | |
| 81 to <92 | 292 | 29 | 9.0 (5.5, 14.3) | 1.11 (0.63, 1.96) | 0.720 | ||
| 92 to ≤108 | 296 | 22 | 6.4 (3.7, 10.8) | 0.76 (0.42, 1.39) | 0.377 | ||
| Maternal employment status | Full time employment (reference) | 412 | 39 | 4.3 (1.7, 10.5) | 1 | 0.167 | |
| Self-employed | 110 | 5 | 8.5 (5.5 13.0) | 2.06 (0.79, 5.43) | 0.142 | ||
| Farmer | 72 | 5 | 5.8 (2.2, 14.3) | 1.37 (0.38, 5.02) | 0.632 | ||
| Unemployed | 57 | 8 | 13.7 (6.6, 26.3) | 3.54 (1.09, 11.52) | 0.036 | ||
| Maternal education | Above high school (reference) | 173 | 15 | 8.2 (4.7, 14.1) | 1 | 0.986 | |
| High school | 221 | 19 | 7.8 (4.5, 13.0) | 0.94 (0.45, 1.94) | 0.865 | ||
| Below high school | 262 | 23 | 8.0 (4.8, 12.9) | 0.97 (0.48, 1.95) | 0.929 | ||
| Monthly household income (million VND) | <6 (reference) | 192 | 14 | 7.0 (3.9, 12.1) | 1 | 0.771 | |
| 6 to <10 | 204 | 19 | 8.8 (5.3, 14.3) | 1.30 (0.63, 2.67) | 0.483 | ||
| ≥10 | 224 | 18 | 7.6 (4.5, 12.5) | 1.10 (0.53, 2.28) | 0.797 | ||
| Stunting c | Not stunted (reference) | 809 | 70 | 7.6 (5.1, 11.2) | 1 | ||
| Stunted | 46 | 7 | 13.5 (6.1, 27.5) | 1.90 (0.81, 4.45) | 0.141 | ||
| Underweight d | Not underweight (reference) | 783 | 67 | 7.5 (5.0, 11.1) | 1 | ||
| Underweight | 74 | 10 | 12.0 (6.0, 22.5) | 1.67 (0.81, 3.43) | 0.163 | ||
| Wasting e | Not wasted (reference) | 804 | 71 | 7.7 (5.2, 11.4) | 1 | ||
| Wasted | 51 | 6 | 10.6 (4.5, 23.1) | 1.42 (0.58, 3.44) | 0.448 | ||
| Overweight/ obesity f | Not overweight/obese (reference) | 695 | 67 | 8.5 (5.7, 12.4) | 1 | ||
| Overweight/obese | 160 | 10 | 6.0 (3.0, 11.6) | 0.69 (0.34, 1.40) | 0.307 | ||
| CIAF g | No anthropometric failure (reference) | 744 | 63 | 7.4 (4.9, 11.1) | 1 | ||
| Anthropometric failure | 113 | 14 | 11.0 (6.0, 19.3) | 1.54 (0.82, 2.86) | 0.178 | ||
| Total (N) | Normocytic Anaemia (N) | Prevalence, % (95% CI) | Univariate Analysis a | ||||
|---|---|---|---|---|---|---|---|
| OR (95% CI) | P-Value | Global P-Value b | |||||
| All children | 860 | 59 | 5.3 (2.9, 9.5) | ||||
| Sex | Girls (reference) | 429 | 32 | 5.9 (3.1, 11.0) | 1 | ||
| Boys | 431 | 27 | 4.7 (2.4, 9.0) | 0.78 (0.46, 1.34) | 0.373 | ||
| Age (months) | 69 to <81 (reference) | 272 | 26 | 7.5 (3.9, 14.1) | 1 | 0.102 | |
| 81 to <92 | 292 | 16 | 4.2 (2.0, 8.7) | 0.54 (0.28, 1.04) | 0.069 | ||
| 92 to ≤108 | 296 | 17 | 4.4 (2.1, 8.9) | 0.56 (0.29, 1.07) | 0.077 | ||
| Maternal employment status | Full time employment (reference) | 412 | 27 | 4.9 (1.4, 16.3) | 1 | 0.477 | |
| Self-employed | 110 | 9 | 3.1 (0.9, 9.9) | 0.62 (0.27, 1.41) | 0.258 | ||
| Farmer | 72 | 8 | 5.4 (1.4, 18.5) | 1.10 (0.38, 3.16) | 0.866 | ||
| Unemployed | 57 | 3 | 3.1 (0.6, 14.1) | 0.62 (0.15, 2.47) | 0.495 | ||
| Maternal education | Above high school (reference) | 173 | 11 | 3.0 (0.8, 10.2) | 1 | 0.309 | |
| High school | 221 | 13 | 3.0 (0.9, 9.9) | 1.01 (0.42, 2.41) | 0.989 | ||
| Below high school | 262 | 23 | 4.8 (1.5, 14.4) | 1.64 (0.73, 3.36) | 0.230 | ||
| Monthly household income(million VND) | <6 (reference) | 192 | 16 | 4.6 (1.4, 14.5) | 1 | 0.126 | |
| 6 to <10 | 204 | 9 | 2.1 (0.6, 7.6) | 0.45 (0.19, 1.06) | 0.066 | ||
| ≥10 | 224 | 20 | 4.5 (1.3, 14.0) | 0.97 (0.48, 1.99) | 0.942 | ||
| Stunting c | Not stunted (reference) | 809 | 54 | 5.1 (2.8, 9.3) | 1 | ||
| Stunted | 46 | 5 | 9.0 (3.2, 22.9) | 1.82 (0.68, 4.90) | 0.236 | ||
| Underweight d | Not underweight (reference) | 783 | 48 | 4.7 (2.6, 8.6) | 1 | ||
| Underweight | 74 | 11 | 12.0 (5.4, 24.3) | 2.74 (1.33, 5.64) | 0.006 | ||
| Wasting e | Not wasted (reference) | 804 | 49 | 4.7 (2.6, 8.5) | 1 | ||
| Wasted | 51 | 10 | 15.7 (7.0, 31.4) | 3.76 (1.73, 8.17) | 0.001 | ||
| Overweight/ obesity f | Not overweight/obese (reference) | 695 | 55 | 6.2 (3.5, 10.9) | 1 | ||
| Overweight/obese | 160 | 4 | 2.2 (0.7, 6.4) | 0.33 (0.12, 0.94) | 0.038 | ||
| CIAF g | No anthropometric failure (reference) | 744 | 44 | 4.6 (2.5, 8.3) | 1 | ||
| Anthropometric failure | 113 | 15 | 10.5 (5.1, 20.5) | 2.45 (1.30, 4.64) | 0.006 | ||
| ICC (95% CI) | |
|---|---|
| Anaemia b | 0.146 (0.088, 0.204) |
| Microcytic anaemia c | 0.085 (0.053, 0.116) |
| Normocytic anaemia d | 0.065 (0.034, 0.096) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hoang, N.T.D.; Orellana, L.; Le, T.D.; Gibson, R.S.; Worsley, A.; Sinclair, A.J.; Hoang, N.T.T.; Szymlek-Gay, E.A. Anaemia and Its Relation to Demographic, Socio-economic and Anthropometric Factors in Rural Primary School Children in Hai Phong City, Vietnam. Nutrients 2019, 11, 1478. https://doi.org/10.3390/nu11071478
Hoang NTD, Orellana L, Le TD, Gibson RS, Worsley A, Sinclair AJ, Hoang NTT, Szymlek-Gay EA. Anaemia and Its Relation to Demographic, Socio-economic and Anthropometric Factors in Rural Primary School Children in Hai Phong City, Vietnam. Nutrients. 2019; 11(7):1478. https://doi.org/10.3390/nu11071478
Chicago/Turabian StyleHoang, Ngan T.D., Liliana Orellana, Tuyen D. Le, Rosalind S. Gibson, Anthony Worsley, Andrew J. Sinclair, Nghien T.T. Hoang, and Ewa A. Szymlek-Gay. 2019. "Anaemia and Its Relation to Demographic, Socio-economic and Anthropometric Factors in Rural Primary School Children in Hai Phong City, Vietnam" Nutrients 11, no. 7: 1478. https://doi.org/10.3390/nu11071478
APA StyleHoang, N. T. D., Orellana, L., Le, T. D., Gibson, R. S., Worsley, A., Sinclair, A. J., Hoang, N. T. T., & Szymlek-Gay, E. A. (2019). Anaemia and Its Relation to Demographic, Socio-economic and Anthropometric Factors in Rural Primary School Children in Hai Phong City, Vietnam. Nutrients, 11(7), 1478. https://doi.org/10.3390/nu11071478





