The Consumption of Nuts is Associated with Better Dietary and Lifestyle Patterns in Polish Adults: Results of WOBASZ and WOBASZ II Surveys
Abstract
1. Introduction
2. Materials and Methods
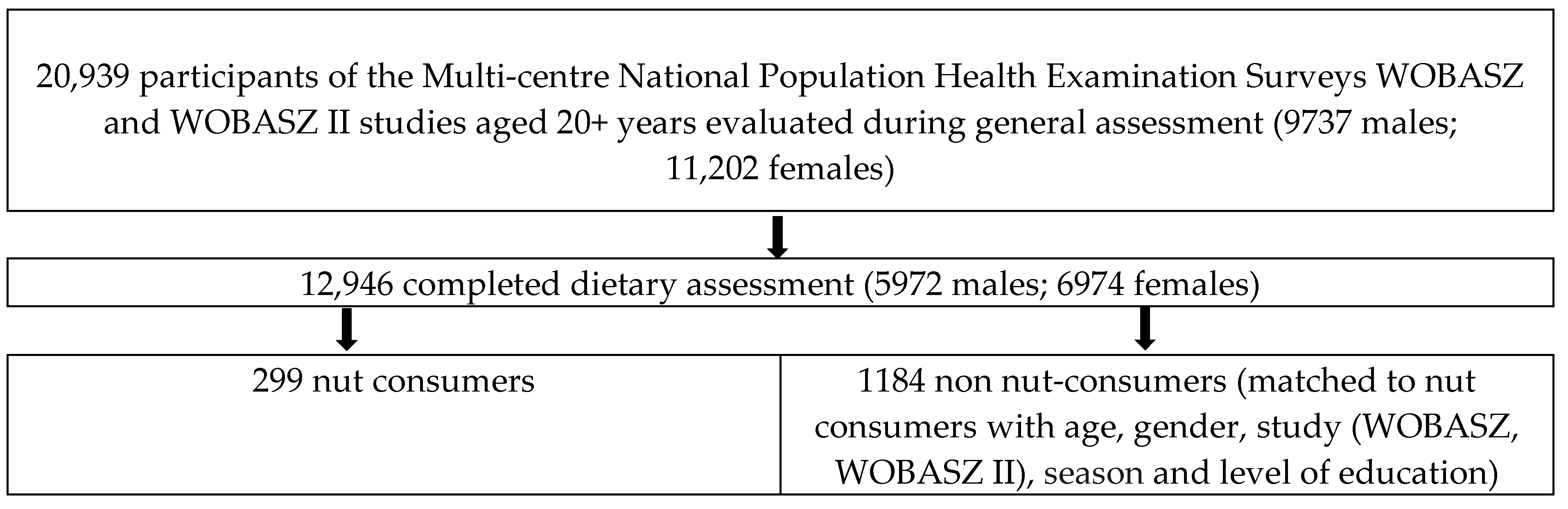
2.1. Study Participants
2.2. Control Matching Procedure
2.3. Data Collection
2.4. Nutritional Assessment
2.4.1. 24-h Dietary Recall Method
2.4.2. Determination of Nut Consumption
2.4.3. Diet Quality Assessment
2.4.4. Estimation of Dietary Total Antioxidant Capacity and Dietary Polyphenol Intake
2.4.5. Food Frequency Questionnaire (FFQ)
2.4.6. Determination of Atherogenic and Antiatherogenic Food Consumption
2.4.7. Determination of Dietary Practices and Self-Assessment of Nutrition
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nash, S.D.; Nash, D.T. Nuts as part of a healthy cardiovascular diet. Curr. Atheroscler. Rep. 2008, 10, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Ros, E. Nuts and CVD. Br. J. Nutr. 2015, 113 (Suppl. 2), S111–S120. [Google Scholar] [CrossRef] [PubMed]
- Luo, C.; Zhang, Y.; Ding, Y.; Shan, Z.; Chen, S.; Yu, M.; Hu, F.B.; Liu, L. Nut consumption and risk of type 2 diabetes, cardiovascular disease, and all-cause mortality: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2014, 100, 256–269. [Google Scholar] [CrossRef] [PubMed]
- Hosseinpour-Niazi, S.; Hosseini, S.; Mirmiran, P.; Azizi, F. Prospective study of nut consumption and incidence of metabolic syndrome: Tehran Lipid and Glucose Study. Nutrients 2017, 9, 1056. [Google Scholar] [CrossRef] [PubMed]
- Ibarrola-Jurado, N.; Bulló, M.; Guasch-Ferré, M.; Ros, E.; Martínez-González, M.A.; Corella, D.; Fiol, M.; Wärnberg, J.; Estruch, R.; Román, P.; et al. PREDIMED Study Investigators. Cross-sectional assessment of nut consumption and obesity, metabolic syndrome and other cardiometabolic risk factors: The PREDIMED study. PLoS ONE 2013, 8, e57367. [Google Scholar] [CrossRef] [PubMed]
- Bitok, E.; Sabaté, J. Nuts and cardiovascular disease. Prog. Cardiovasc. Dis. 2018, 61, 33–37. [Google Scholar] [CrossRef]
- Piepoli, M.F.; Hoes, A.W.; Agewall, S.; Albus, C.; Brotons, C.; Catapano, A.L.; Cooney, M.T.; Corrà, U.; Cosyns, B.; Deaton, C.; et al. 2016 European Guidelines on cardiovascular disease prevention in clinical practice: The Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of 10 societies and by invited experts) Developed with the special contribution of the European Association for Cardiovascular Prevention & Rehabilitation (EACPR). Eur. Heart J. 2016, 37, 2315–2381. [Google Scholar] [CrossRef]
- Eckel, R.H.; Jakicic, J.M.; Ard, J.D.; de Jesus, J.M.; Houston Miller, N.; Hubbard, V.S.; Lee, I.M.; Lichtenstein, A.H.; Loria, C.M.; Millen, B.E.; et al. 2013 AHA/ACC guideline on lifestyle management to reduce cardiovascular risk: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 2014, 129 (Suppl. 2), S76–S99. [Google Scholar] [CrossRef]
- Relja, A.; Miljković, A.; Gelemanović, A.; Bošković, M.; Hayward, C.; Polašek, O.; Kolčić, I. Nut consumption and cardiovascular risk factors: A cross-sectional study in a Mediterranean population. Nutrients 2017, 9, 1296. [Google Scholar] [CrossRef]
- Brown, R.C.; Tey, S.L.; Gray, A.R.; Chisholm, A.; Smith, C.; Fleming, E.; Blakey, C.; Parnell, W. Patterns and predictors of nut consumption: Results from the 2008/09 New Zealand Adult Nutrition Survey. Br. J. Nutr. 2014, 112, 2028–2040. [Google Scholar] [CrossRef]
- Brown, R.C.; Gray, A.R.; Tey, S.L.; Chisholm, A.; Burley, V.; Greenwood, D.C.; Cade, J. Associations between nut consumption and health vary between omnivores, vegetarians, and vegans. Nutrients 2017, 9, 1219. [Google Scholar] [CrossRef] [PubMed]
- Sterling, S.R.; Bertrand, B.; Judd, S.; Carson, T.L.; Chandler-Laney, P.; Baskin, M.L. Longitudinal analysis of nut-inclusive diets and body mass index among overweight and obese African American women living in rural Alabama and Mississippi, 2011–2013. Prev. Chronic Dis. 2017, 14, E82. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Brown, R.C.; Tey, S.L.; Gray, A.R.; Chisholm, A.; Smith, C.; Fleming, E.; Parnell, W. Nut consumption is associated with better nutrient intakes: Results from the 2008/09 New Zealand Adult Nutrition Survey. Br. J. Nutr. 2016, 115, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Witkowska, A.; Zujko, M.E.; Mirończuk-Chodakowska, I. The effect of a Mediterranean diet model on serum beta-carotene concentration. A preliminary assessment. Rocz. Państwowego Zakładu Hig. 2013, 64, 123–127. [Google Scholar]
- O’Neil, C.E.; Nicklas, T.A.; Fulgoni, V.L., III. Tree nut consumption is associated with better nutrient adequacy and diet quality in adults: National Health and Nutrition Examination Survey 2005–2010. Nutrients 2015, 7, 595–607. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.L.; Brown, R.; Gray, A.; Delahunty, C. Nuts improve diet quality compared to other energy-dense snacks while maintaining body weight. J. Nutr. Metab. 2011, 2011, 357350. [Google Scholar] [CrossRef]
- O’Neil, C.E.; Keast, D.R.; Nicklas, T.A.; Fulgoni, V.L., III. Out-of-hand nut consumption is associated with improved nutrient intake and health risk markers in US children and adults: National Health and Nutrition Examination Survey 1999–2004. Nutr. Res. 2012, 32, 185–194. [Google Scholar] [CrossRef]
- Jacobs, D.R., Jr.; Gross, M.D.; Tapsell, L.C. Food synergy: An operational concept for understanding nutrition. Am. J. Clin. Nutr. 2009, 89, 1543S–1548S. [Google Scholar] [CrossRef]
- Broda, G.; Rywik, S. Multicenter national Polish population health status tests – WOBASZ project with defined problems and treatment goals. Kardiol. Pol. 2005, 63, 601–604. (In Polish) [Google Scholar]
- Rywik, S.; Kupść, W.; Piotrowski, W.; Broda, G.; Piwoński, J.; Kurjata, P.; Waśkiewicz, A.; Gaździk, D. Multicenter national Polish population health status tests—WOBASZ project. Establishment of methods and logistics. Kardiol. Pol. 2005, 63, 605–613. (In Polish) [Google Scholar]
- Drygas, W.; Niklas, A.A.; Piwońska, A.; Piotrowski, W.; Flotyńska, A.; Kwaśniewska, M.; Nadrowski, P.; Puch-Walczak, A.; Szafraniec, K.; Bielecki, W.; et al. Multi-center National Population Health Examination Survey (WOBASZ II study): Assumptions, methods and implementation. Kardiol. Pol. 2016, 74, 681–690. [Google Scholar] [CrossRef] [PubMed]
- Broda, G.; Kurjata, P.; Piwońska, A.; Polakowska, M.; Waśkiewicz, A.; Sygnowska, E.; Piotrowski, W.; Istynowicz, K. National HES Manual, Poland. The Cardinal Stefan Wyszyński Institute of Cardiology. Available online: http://www.ehes.info/manuals/national_manuals/national_manual_Poland_EN.pdf (accessed on 20 February 2019).
- Alberti, K.G.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.C.; James, W.P.; Loria, C.M.; Smith, S.C., Jr. Harmonizing the metabolic syndrome. A joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009, 120, 1640–1645. [Google Scholar] [CrossRef]
- Szponar, L.; Wolnicka, K.; Rychlik, E. Album of Photographs of Food Products and Dishes; National Food and Nutrition Institute Press: Warsaw, Poland, 2000. (In Polish) [Google Scholar]
- World Health Organisation. Diet, Nutrition, and the Prevention of Chronic Diseases; WHO Technical Report Series No. 797; Report of a WHO Study Group; World Health Organisation: Geneva, Switzerland, 1990. [Google Scholar]
- Fransen, H.P.; Beulens, J.W.; May, A.M.; Struijk, E.A.; Boer, J.M.; de Wit, G.A.; Onland-Moret, N.C.; van der Schouw, Y.T.; Bueno-de-Mesquita, H.B.; Hoekstra, J.; et al. Dietary patterns in relation to quality-adjusted life years in the EPIC-NL cohort. Prev. Med. 2015, 77, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Kunachowicz, H.; Nadolna, I.; Przygoda, B.; Iwanow, K. Food Composition Tables; PZWL Medical Publishing House: Warsaw, Poland, 2005. [Google Scholar]
- Witkowska, A.M.; Waśkiewicz, A.; Zujko, M.E.; Szcześniewska, D.; Pająk, A.; Stepaniak, U.; Drygas, W. Dietary Polyphenol Intake, but Not the Dietary Total Antioxidant Capacity, Is Inversely Related to Cardiovascular Disease in Postmenopausal Polish Women: Results of WOBASZ and WOBASZ II Studies. Oxid. Med. Cell. Longev. 2017, 2017, 5982809. [Google Scholar] [CrossRef] [PubMed]
- Zujko, M.E.; Waśkiewicz, A.; Witkowska, A.M.; Szcześniewska, D.; Zdrojewski, T.; Kozakiewicz, K.; Drygas, W. Dietary total antioxidant capacity and dietary polyphenol intake and prevalence of metabolic syndrome in polish adults: A nationwide study. Oxid. Med. Cell. Longev. 2018, 2018, 7487816. [Google Scholar] [CrossRef] [PubMed]
- Zujko, M.E.; Witkowska, A.M. Antioxidant potential and polyphenol content of selected food. Int. J. Food Prop. 2011, 14, 300–308. [Google Scholar] [CrossRef]
- Zujko, M.E.; Witkowska, A.M. Antioxidant potential and polyphenol content of beverages, chocolates, nuts, and seeds. Int. J. Food Prop. 2014, 17, 86–92. [Google Scholar] [CrossRef]
- Carlsen, M.H.; Halvorsen, B.L.; Holte, K.; Bøhn, S.K.; Dragland, S.; Sampson, L.; Willey, C.; Senoo, H.; Umezono, Y.; Sanada, C.; et al. The total antioxidant content of more than 3100 foods, beverages, spices, herbs and supplements used worldwide. Nutr. J. 2010, 9, 3. [Google Scholar] [CrossRef]
- Neveu, V.; Perez-Jiménez, J.; Vos, F. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. Database 2010, 2010, bap024. [Google Scholar] [CrossRef]
- Jenab, M.; Sabaté, J.; Slimani, N.; Ferrari, P.; Mazuir, M.; Casagrande, C.; Deharveng, G.; Tjønneland, A.; Olsen, A.; Overvad, K.; et al. Consumption and portion sizes of tree nuts, peanuts and seeds in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohorts from 10 European countries. Br. J. Nutr. 2006, 96 (Suppl. 2), S12–S23. [Google Scholar] [CrossRef]
- Jankowski, P. Zasady profilaktyki chorób układu krążenia w 2018 roku. [Principles of cardiovascular disease prevention in 2018]. Kardiol. Inwazyjna 2017, 12, 42–48. (In Polish) [Google Scholar]
- Gili, R.V.; Leeson, S.; Montes-Chañi, E.M.; Xutuc, D.; Contreras-Guillén, I.A.; Guerrero-Flores, G.N.; Martins, M.C.T.; Pacheco, F.J.; Pacheco, S.O.S. Healthy vegan lifestyle habits among Argentinian vegetarians and non-vegetarians. Nutrients 2019, 11, 154. [Google Scholar] [CrossRef] [PubMed]
- Bibiloni, M.M.; Julibert, A.; Bouzas, C.; Martínez-González, M.A.; Corella, D.; Salas-Salvadó, J.; Zomeño, M.D.; Vioque, J.; Romaguera, D.; Martínez, J.A.; et al. Nut Consumptions as a Marker of Higher Diet Quality in a Mediterranean Population at High Cardiovascular Risk. Nutrients 2019, 11, 754. [Google Scholar] [CrossRef] [PubMed]
- US Food and Drug Administration. Qualified Health Claims: Letter of Enforcement Discretion—Nuts and Coronary Heart Disease (Docket No 02P-0505). 2003. Available online: http://www.fda.gov/Food/IngredientsPackagingLabeling/LabelingNutrition/ucmucm072926.htm (accessed on 18 December 2018).
- Estruch, R.; Ros, E.; Salas-Salvado, J.; Covas, M.I.; Corella, D.; Aros, F.; Gomez-Gracia, E.; Ruiz-Gutierrez, V.; Fiol, M.; Lapetra, J.; et al. PREDIMED Study Investigators. Primary prevention of cardiovascular disease with a Mediterranean diet. N. Engl. J. Med. 2013, 368, 1279–1290. [Google Scholar] [CrossRef] [PubMed]
- Stefler, D.; Pikhart, H.; Jankovic, N.; Kubinova, R.; Pajak, A.; Malyutina, S.; Simonova, G.; Feskens, E.J.M.; Peasey, A.; Bobak, M. Healthy diet indicator and mortality in Eastern European populations: Prospective evidence from the HAPIEE cohort. Eur. J. Clin. Nutr. 2014, 68, 1346–1352. [Google Scholar] [CrossRef] [PubMed]
- Jankovic, N.; Geelen, A.; Streppel, M.T.; de Groot, L.C.; Orfanos, P.; van den Hooven, E.H.; Pikhart, H.; Boffetta, P.; Trichopoulou, A.; Bobak, M.; et al. Adherence to a healthy diet according to the World Health Organization guidelines and all-cause mortality in elderly adults from Europe and the United States. Am. J. Epidemiol. 2014, 180, 978–988. [Google Scholar] [CrossRef] [PubMed]
- Bellavia, A.; Stilling, F.; Wolk, A. High red meat intake and all-cause cardiovascular and cancer mortality: Is the risk modified by fruit and vegetable intake? Am. J. Clin. Nutr. 2016, 104, 1137–1143. [Google Scholar] [CrossRef]
- Pan, A.; Sun, Q.; Bernstein, A.M.; Schulze, M.B.; Manson, J.E.; Stampfer, M.J.; Willett, W.C.; Hu, F.B. Red meat consumption and mortality: Results from 2 prospective cohort studies. Arch. Intern. Med. 2012, 172, 555–563. [Google Scholar] [CrossRef]
- Bouvard, V.; Loomis, D.; Guyton, K.; Grosse, Y.; Ghissassi, F.E.; Benbrahim-Tallaa, L.; Guha, N.; Mattock, H.; Straif, K.; International Agency for Research on Cancer Monograph Working Group. Carcinogenicity of consumption of red and processed meat. Lancet Oncol. 2015, 16, 1599–1600. [Google Scholar] [CrossRef]
- Abete, I.; Romaguera, D.; Vieira, A.R.; Lopez de Munain, A.; Norat, T. Association between total, processed, red and white meat consumption and all-cause, CVD and IHD mortality: A meta-analysis of cohort studies. Br. J. Nutr. 2014, 112, 762–775. [Google Scholar] [CrossRef]
- Birt, C.; Buzeti, T.; Grosso, G.; Justesen, L.; Lachat, C.; Lafranconi, A.; Mertanen, E.; Rangelov, N.; Sarlio-Lähteenkorva, S.; European Public Health Association—EUPHA. Healthy and Sustainable Diets for European Countries. EUPHA, 2017. Available online: https://eupha.org/repository/advocacy/EUPHA_report_on_healthy_and_sustainable_diets_20-05-2017.pdf (accessed on 6 March 2019).
- US Department of Health and Human Services & US Department of Agriculture. 2015–2020 Dietary Guidelines for Americans, 8th ed.; DHHS and USDA: Washington, DC, USA, 2015. Available online: http://www.cnpp.usda.gov/2015-2020-dietary-guidelines-americans (accessed on 6 March 2019).
- Serino, A.; Salazar, G. Protective role of polyphenols against vascular inflammation, aging and cardiovascular disease. Nutrients 2018, 11, 53. [Google Scholar] [CrossRef] [PubMed]
- Witkowska, A.M.; Zujko, M.E.; Waśkiewicz, A.; Terlikowska, K.M.; Piotrowski, W. Comparison of various databases for estimation of dietary polyphenol intake in the population of Polish adults. Nutrients 2015, 7, 9299–9308. [Google Scholar] [CrossRef] [PubMed]
- Tey, S.L.; Robinson, T.; Gray, A.R.; Chisholm, A.W.; Brown, R.C. Do dry roasting, lightly salting nuts affect their cardioprotective properties and acceptability? Eur. J. Nutr. 2017, 56, 1025–1036. [Google Scholar] [CrossRef] [PubMed]
| Nut Consumers N = 299 | Non-Nut Consumers N = 1184 | ||||
|---|---|---|---|---|---|
| Characteristics | mean ± SD | median (25–75 percentile) | mean ± SD | median (25–75 percentile) | p |
| Age (years) | 44.1 ± 14.1 | 43.0 (33–55) | 43.2 ± 14.0 | 43.0 (31–54) | 0.3421 |
| % | % | ||||
| Gender (%) | 0.974 | ||||
| men | 45.27 | 45.48 | |||
| women | 54.73 | 54.52 | |||
| Age (%) | 0.732 | ||||
| 20–44 years | 51.52 | 53.98 | |||
| 45–64 years | 40.74 | 39.00 | |||
| 65+ years | 7.74 | 7.02 | |||
| Level of education (%) | 0.904 | ||||
| under middle | 27.76 | 28.80 | |||
| middle | 42.47 | 41.13 | |||
| university | 29.77 | 30.07 | |||
| Smoking status (%) | 0.336 | ||||
| current smokers | 22.07 | 26.02 | |||
| past smokers | 24.75 | 22.35 | |||
| never smokers | 53.18 | 51.63 | |||
| BMI [kg/m2] (%): | 0.755 | ||||
| underweight (BMI<18.5) | 1.37 | 1.39 | |||
| normal (BMI 18.5–24.99) | 42.47 | 42.53 | |||
| overweight (BMI 25–29.99) | 37.67 | 35.04 | |||
| obese (BMI>30) | 18.49 | 21.04 | |||
| PAL (%) | 0.026 | ||||
| low level | 34.78 | 43.34 | |||
| middle level | 32.44 | 27.40 | |||
| high level | 32.78 | 29.26 | |||
| Diseases (%) | |||||
| Hypertension | 33.22 | 33.48 | 0.934 | ||
| Myocardial infarction | 3.01 | 2.70 | 0.774 | ||
| Stroke | 1.68 | 1.11 | 0.386* | ||
| Diabetes | 7.67 | 6.71 | 0.658 | ||
| Hypercholesterolemia (%) | 61.86 | 62.71 | 0.789 | ||
| Central obesity (%) | 29.69 | 32.04 | 0.439 | ||
| Metabolic syndrome (%) | 27.76 | 28.38 | 0.832 | ||
| Regular adding extra salt during the meal (%) | 18.39 | 21.88 | 0.1881 | ||
| Removal of visible fat from meat and cold cuts, removal of poultry skin (%) | 52.51 | 48.48 | 0.2131 | ||
| Special diets: | 0.0394 | ||||
| low-fat, low-cholesterol, or diabetic diet (%) | 7.36 | 5.15 | |||
| weight-loss diet (%) | 1.67 | 0.84 | |||
| other diet (%) | 3.68 | 1.77 | |||
| Recognizing their nutrition as appropriate (%) | 58.19 | 53.63 | 0.2976 | ||
| Nut Consumers N = 299 | Non-Nut Consumers N = 1184 | ||||
|---|---|---|---|---|---|
| Intake | mean ± SD | Median (25–75 percentile) | mean ± SD | Median (25–75 percentile) | p |
| Energy (kcal/d) | 2281 ± 888 | 2110 (1659–2755) | 2044 ± 997 | 1872 (1391–2456) | <0.0001 |
| Nut consumption (g/d) | 56.00 ± 52.8 | 30.0 (30.0–70.0) | 0 ± 0 | 0 (0–0) | <0.0001 |
| Red meat (processed, unprocessed) (g/d) | 101.5 ± 115.8 | 60 (0–160) | 123.4 ± 157.6 | 85 (20–184) | 0.0198 |
| Fish (g/d) | 24.5 ± 66.6 | 0 (0–0) | 19.2 ± 64.6 | 0 (0–0) | 0.0593 |
| Poultry (processed, unprocessed) (g/d) | 67.8 ± 108.6 | 0 (0–120) | 53.1 ± 96.8 | 0 (0–75) | 0.0286 |
| Wholemeal bread (g/d) | 30.3 ± 58.0 | 0 (0–50) | 27.6 ± 57.0 | 0 (0–30) | 0.2743 |
| Vegetables (g/d) | 257.2 ± 189 | 230 (128.1–350) | 235.2 ± 172.7 | 204.2 (113.8–320.1) | 0.0750 |
| Fruits (g/d) | 290.5 ± 254.7 | 250 (110–400) | 206.5 ± 241 | 150 (0–310) | <0.0001 |
| Legumes (g/d) | 3.1 ± 12.7 | 0 (0–0) | 4.1 ± 19.2 | 0 (0–0) | 0.9006 |
| Tea (g/d) | 341.6 ± 259.5 | 250 (200–500) | 328.8 ± 263.4 | 250 (200–500) | 0.3075 |
| Coffee (g/d) | 180.1 ± 179.8 | 200 (0–250) | 182.5 ± 198.8 | 200 (0–250) | 0.9450 |
| Alcohol(pure ethanol g/d) | 2.7 ± 9.7 | 0 (0–0) | 3.7 ± 17.7 | 0 (0–0) | 0.9329 |
| Sodium (mg/d) ** | 1903 ± 1051 | 1645 (1177–2423) | 2026 ± 1273 | 1706 (1168–2545) | 0.3254 |
| DPI (mg/d) | 2907.2 ± 1212.7 | 2677.9 (2118.7–3515) | 2040.9 ± 911.4 | 1951.9 (1402.4–2487.7) | <0.0001 |
| DTAC (mmol/d) | 29.3 ± 26.4 | 21.3 (13.4–35.8) | 12 ± 5.9 | 11.4 (8.0–15.3) | <0.0001 |
| HDI (points) | 3.9 ± 1.3 | 4 (3–5) | 3.2 ± 1.3 | 3 (2–4) | <0.0001 |
| Nut Consumers N = 299 | Nut non-Consumers N = 1184 | ||||
|---|---|---|---|---|---|
| Monthly frequency of intake | mean ± SD | Median (25–75 percentile) | mean ± SD | Median (25–75 percentile) | p |
| Atherogenic foods (sum) | 53.1 ± 27.6 | 54.4 (30.0–72.2) | 56.8 ± 25.1 | 55.7 (39.4–73.3) | 0.0501 |
| Antiatherogenic foods (sum) | 66.2 ± 23.6 | 66.4 (50.8–85.0) | 62.2 ± 23.5 | 62.1 (46.5–77.9) | 0.0084 |
| Atherogenic/antiatherogenic food ratio | 1.01 ± 1.42 | 0.8 (0.5–1.3) | 1.19 ± 1.25 | 0.9 (0.6–1.3) | 0.0009 |
| Hard cheese (days/month) | 10.6 ± 9.1 | 10.7 (4.3–10.7) | 10.6 ± 9.07 | 10.7 (4.3–10.7) | 0.9985 |
| Red meats unprocessed (beef, pork, veal, lamb, organ meats) (days/month) | 12.0 ± 9.0 | 10.7 (4.3–21.4) | 12.73 ± 8.67 | 10.7 (4.3–21.4) | 0.1055 |
| Processed meats (pork, beef, veal) (days/month) | 13.1 ± 9.5 | 10.7 (4.3–21.4) | 15.2 ± 9.7 | 10.7 (10.7–21.4) | 0.0009 |
| Butter (days/month) | 15.0 ± 13.7 | 10.7 (0.0–30.0) | 15.8 ± 13.5 | 21.4 (0.0–30.0) | 0.4308 |
| Lard (days/month) | 2.38 ± 5.46 | 0 (0.0–1.5) | 2.59 ± 5.43 | 0 (0–1.5) | 0.1815 |
| Fish, fresh, preserved, smoked (days/month) | 5.75 ± 5.02 | 4.3 (1.5–10.7) | 5.44 ± 4.73 | 4.3 (1.5–4.3) | 0.4689 |
| Vegetables, cooked (days/month) | 15.2 ± 9.9 | 10.7(10.7–21.4) | 14.82 ± 9.91 | 10.7 (10.7–21.4) | 0.5334 |
| Vegetables, raw (days/month) | 17.36 ± 9.97 | 21.4 (10.7–30.0) | 16.22 ± 10.55 | 10.7 (10.7–30.0) | 0.0875 |
| Legumes (days/month) | 3.12 ± 3.76 | 1.5 (1.5–4.3) | 3.16 ± 4.5 | 1.5 (1.5–4.3) | 0.2774 |
| Fruit, fresh (days/month) | 24.75 ± 8.2 | 30 (21.4–30.0) | 22.59 ± 9.67 | 30 (10.7–30.0) | 0.0009 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Witkowska, A.M.; Waśkiewicz, A.; Zujko, M.E.; Szcześniewska, D.; Śmigielski, W.; Stepaniak, U.; Pająk, A.; Drygas, W. The Consumption of Nuts is Associated with Better Dietary and Lifestyle Patterns in Polish Adults: Results of WOBASZ and WOBASZ II Surveys. Nutrients 2019, 11, 1410. https://doi.org/10.3390/nu11061410
Witkowska AM, Waśkiewicz A, Zujko ME, Szcześniewska D, Śmigielski W, Stepaniak U, Pająk A, Drygas W. The Consumption of Nuts is Associated with Better Dietary and Lifestyle Patterns in Polish Adults: Results of WOBASZ and WOBASZ II Surveys. Nutrients. 2019; 11(6):1410. https://doi.org/10.3390/nu11061410
Chicago/Turabian StyleWitkowska, Anna M., Anna Waśkiewicz, Małgorzata E. Zujko, Danuta Szcześniewska, Witold Śmigielski, Urszula Stepaniak, Andrzej Pająk, and Wojciech Drygas. 2019. "The Consumption of Nuts is Associated with Better Dietary and Lifestyle Patterns in Polish Adults: Results of WOBASZ and WOBASZ II Surveys" Nutrients 11, no. 6: 1410. https://doi.org/10.3390/nu11061410
APA StyleWitkowska, A. M., Waśkiewicz, A., Zujko, M. E., Szcześniewska, D., Śmigielski, W., Stepaniak, U., Pająk, A., & Drygas, W. (2019). The Consumption of Nuts is Associated with Better Dietary and Lifestyle Patterns in Polish Adults: Results of WOBASZ and WOBASZ II Surveys. Nutrients, 11(6), 1410. https://doi.org/10.3390/nu11061410







