Waist Circumference and Abdominal Volume Index Can Predict Metabolic Syndrome in Adolescents, but only When the Criteria of the International Diabetes Federation are Employed for the Diagnosis
Abstract
1. Introduction
2. Materials and Methods
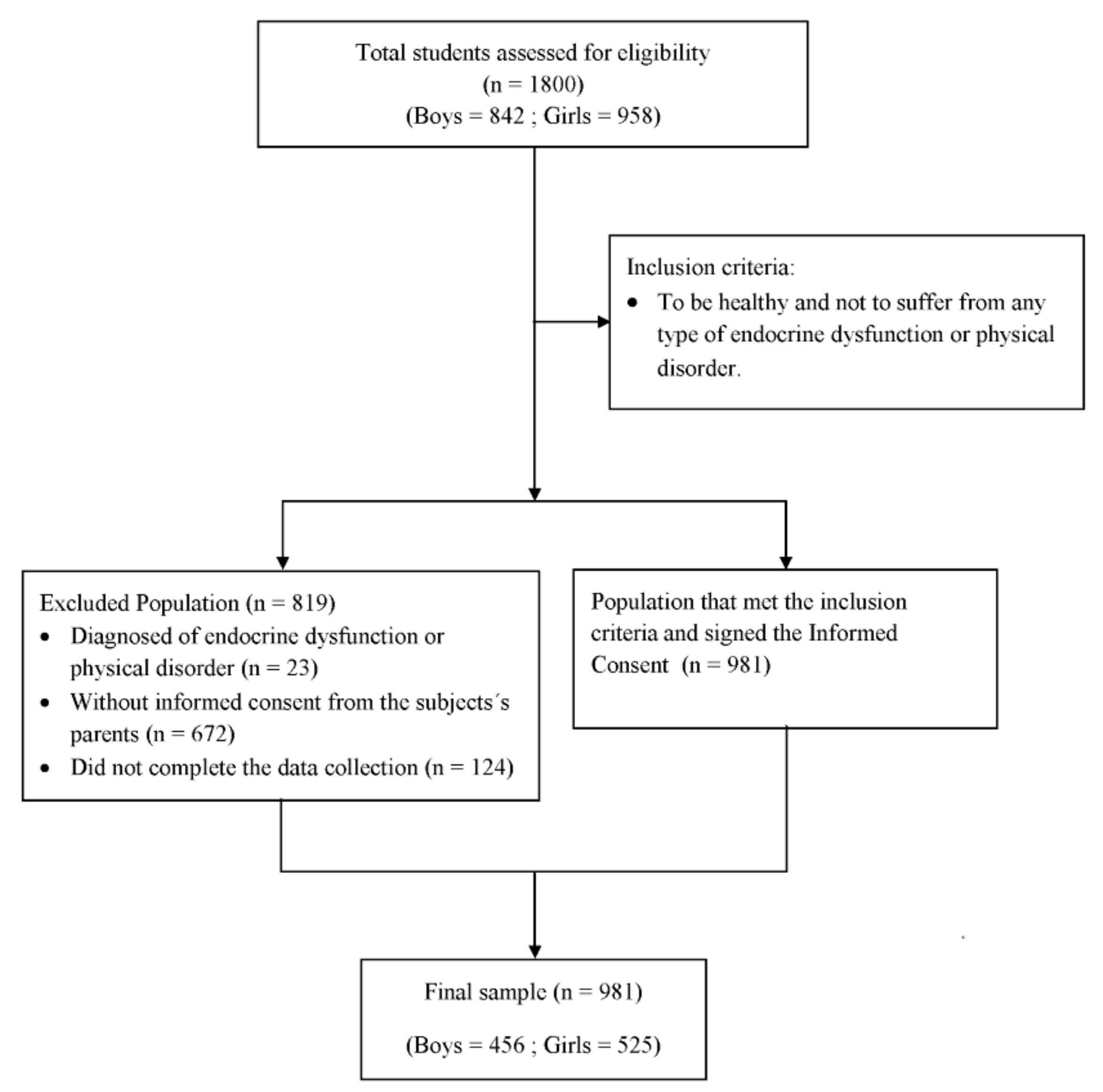
2.1. Study Design and Participants
2.2. Anthropometric Measurements
2.3. Serum Biochemical Examination
2.4. Blood Pressure Determination
2.5. Diagnostic Criteria of Metabolic Syndrome
2.6. Statistical Analysis
3. Results
3.1. Baseline Characteristics of the Participants
3.2. Area under the Curve Values of the Anthropometric Indexes for the Diagnosis of Metabolic Syndrome
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Eisenmann, J.C.; Laurson, K.R.; Dubose, K.D.; Smith, B.K.; Donnelly, J.E. Construct validity of a continuous metabolic syndrome score in children. Diabetol. Metab. Syndr. 2010, 2, 8. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.F.; Santos, N.S.; Goldraich, N.P.; Barski, T.F.; Andrade, K.S.; Kruel, L.F.M. Metabolic syndrome in obese adolescents: A comparison of three different diagnostic criteria. J. Pediatr. 2012, 88, 303–309. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kim, J.; Lee, I.; Lim, S. Overweight or obesity in children aged 0 to 6 and the risk of adultmetabolic-syndrome: A systematic review and meta-analysis. J. Clin. Nurs. 2017, 26, 3869–3880. [Google Scholar] [CrossRef] [PubMed]
- Kaur, J. A comprehensive review on metabolic syndrome. Cardiol. Res. Pract. 2014, 2014, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Vanlancker, T.; Schaubroeck, E.; Vyncke, K.; Cadenas-Sanchez, C.; Breidenassel, C.; González-Gross, M.; Gottrand, F.; Moreno, L.A.; Beghin, L.; Molnár, D.; et al. Comparison of definitions for the metabolic syndrome in adolescents. The HELENA study. Eur. J. Pediatr. 2017, 176, 241–252. [Google Scholar] [CrossRef]
- Perona, J.S.; Schmidt Rio-Valle, J.; Ramírez-Vélez, R.; Correa-Rodríguez, M.; Fernández-Aparicio, Á.; González-Jiménez, E. Waist circumference and abdominal volume index are the strongest anthropometric discriminators of metabolic syndrome in Spanish adolescents. Eur. J. Clin. Investig. 2019, 49, e13060. [Google Scholar] [CrossRef]
- Zimmet, P.; Alberti, K.G.; Kaufman, F.; Tajima, N.; Silink, M.; Arslanian, S.; Wong, G.; Bennett, P.; Shaw, J.; Caprio, S. The metabolic syndrome in children and adolescents—An IDF consensus report. Pediatr. Diabetes 2007, 8, 299–306. [Google Scholar] [CrossRef]
- Pergher, R.N.; Melo, M.E.; Halpern, A. Is a diagnosis of metabolic syndrome applicable to children? J. Pediatr. 2010, 86, 101–108. [Google Scholar] [CrossRef][Green Version]
- Weiss, R.; Bremer, A.; Lustig, R.H. What is metabolic syndrome, and why are children getting it? Ann. N. Y. Acad. Sci. 2013, 1281, 123–140. [Google Scholar] [CrossRef]
- Goodman, E.; Daniels, S.R.; Meigs, J.B.; Dolan, L.M. Instability in the diagnosis of metabolic syndrome in adolescents. Circulation 2007, 115, 2316–2322. [Google Scholar] [CrossRef]
- National Institutes of Health/National Heart, Lung, and Blood Institute. Third Report of the National Cholesterol Education Program (NCEP). In Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III); Final Report; National Institutes of Health: Bethesda, MD, USA, 2002. [Google Scholar]
- Cook, S.; Weitzman, M.; Auinger, P.; Nguyen, M.; Dietz, W.H. Prevalence of a metabolic syndrome phenotype in adolescents: Findings from the third National Health and Nutrition Examination Survey, 1988-1994. Arch. Pediatr. Adolesc. Med. 2003, 157, 821–827. [Google Scholar] [CrossRef] [PubMed]
- Weiss, R.; Dziura, J.; Burgert, T.S.; Tamborlane, W.V.; Taksali, S.E.; Yeckel, C.W.; Allen, K.; Lopes, M.; Savoye, M.; Morrison, J.; et al. Obesity and the metabolic syndrome in children and adolescents. N. Engl. J. Med. 2004, 350, 2362–2374. [Google Scholar] [CrossRef] [PubMed]
- Duncan, G.E.; Li, S.M.; Zhou, X.H. Prevalence and trends of a metabolic syndrome phenotype among U.S. adolescents, 1999–2000. Diabetes Care 2004, 27, 2438–2443. [Google Scholar] [CrossRef] [PubMed]
- De Ferranti, S.D.; Gauvreau, K.; Ludwig, D.S.; Neufeld, E.J.; Newburger, J.W.; Rifai, N. Prevalence of the metabolic syndrome in American adolescents: Findings from the Third National Health and Nutrition Examination Survey. Circulation 2004, 110, 2494–2497. [Google Scholar] [CrossRef] [PubMed]
- Cruz, M.L.; Goran, M.I. The metabolic syndrome in children and adolescents. Curr. Diabetes Rep. 2004, 4, 53–62. [Google Scholar] [CrossRef]
- Rodríguez, M.; Salazar, B.; Violante, R.; Guerrero, F. Metabolic syndrome among children and adolescents aged 10–18 years. Diabetes Care 2004, 27, 2516–2517. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Viner, R.M.; Segal, T.Y.; Lichtarowicz-Krynska, E.; Hindmarsh, P. Prevalence of the insulin resistance syndrome in obesity. Arch. Dis. Child. 2005, 90, 10–14. [Google Scholar] [CrossRef]
- Alberti, K.G.; Zimmet, P.Z. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: Diagnosis and classification of diabetes mellitus provisional report of a WHO consultation. Diabetes Med. 1998, 15, 539–553. [Google Scholar] [CrossRef]
- Marfell-Jones, M.; Olds, T.; Stewart, A. International Standards for Anthropometric Assessment; ISAK: Potchefstroom, South Africa, 2006. [Google Scholar]
- Brook, C.G.D. Determination of body composition of children from skinfold measurements. Arch. Dis. Child. 1971, 46, 182–184. [Google Scholar] [CrossRef]
- Siri, W.E. Body composition from fluid spaces and density: Analysis of methods. In Techniques for Measuring Body Composition; Brozeck, J., Henschel, A., Eds.; National Academies Sciences National Research Council: Washington, DC, USA, 1961. [Google Scholar]
- Guerrero-Romero, F.; Rodríguez-Morán, M. Abdominal volume index. An anthropometry-based index for estimation of obesity is strongly related to impaired glucose tolerance and type 2 diabetes mellitus. Arch. Med. Res. 2003, 34, 428–432. [Google Scholar] [CrossRef]
- Thomas, D.M.; Bredlau, C.; Bosy-Westphal, A.; Mueller, M.; Shen, W.; Gallagher, D.; Maeda, Y.; McDougall, A.; Peterson, C.M.; Ravussin, E.; et al. Relationships between body roundness with body fat and visceral adipose tissue emerging from a new geometrical model. Obesity 2013, 21, 2264–2271. [Google Scholar] [CrossRef] [PubMed]
- Bergman, R.N.; Stefanovski, D.; Buchanan, T.A.; Sumner, A.E.; Reynolds, J.C.; Sebring, N.G.; Xiang, A.H.; Watanabe, R.M. A better index of body adiposity. Obesity 2011, 19, 1083–1089. [Google Scholar] [CrossRef] [PubMed]
- El Aarbaoui, T.; Samouda, H.; Zitouni, D.; di Pompeo, C.; de Beaufort, C.; Trincaretto, F.; Mormentyn, A.; Hubert, H.; Lemdani, M.; Guinhouya, B.C. Does the body adiposity index (BAI) apply to paediatric populations? Ann. Hum. Biol. 2013, 40, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Valdez, R. A simple model-based index of abdominal adiposity. J. Clin. Epidemiol. 1991, 44, 955–956. [Google Scholar] [CrossRef]
- Krakauer, N.Y.; Krakauer, J.C. A new body shape index predicts mortality hazard independently of body mass index. PLoS ONE 2012, 7, e39504. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.; Hosker, J.; Rudenski, A.; Naylor, B.; Treacher, D.; Turner, R. Homeostasis model assessment: Insulin resistance and B-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Pickering, T.G.; Hall, J.E.; Appel, L.J.; Falkner, B.E.; Graves, J.; Hill, M.N.; Jones, D.W.; Kurtz, T.; Sheps, S.G.; Roccella, E.J. Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Recommendations for blood pressure measurement in humans and experimental animals, part 1: Blood pressure measurement in humans: A statement for professionals from the Subcommittee of Professional and Public Education of the American Heart Association Council on High Blood Pressure Research. Hypertension 2005, 45, 142–161. [Google Scholar] [PubMed]
- DeLong, E.R.; DeLong, D.M.; Clarke-Pearson, D.L. Comparing the areas under two or more correlated receiver operating characteristic curves: A nonparametric approach. Biometrics 1988, 44, 837–845. [Google Scholar] [CrossRef]
- Braga-Tavares, H.; Fonseca, H. Prevalence of metabolic syndrome in a Portuguese obese adolescent population according to three different definitions. Eur. J. Pediatr. 2010, 169, 935–940. [Google Scholar] [CrossRef]
- Saffari, F.; Jalilolghadr, S.; Esmailzadehha, N.; Azinfar, P. Metabolic syndrome in a sample of the 6- to 16-year-old overweight or obese pediatric population: A comparison of two definitions. Ther. Clin. Risk Manag. 2012, 8, 55–63. [Google Scholar] [CrossRef][Green Version]
- Sarrafzadegan, N.; Gharipour, M.; Sadeghi, M.; Nouri, F.; Asgary, S.; Zarfeshani, S. Differences in the prevalence of metabolic syndrome in boys and girls based on various definitions. ARYA Atheroscler. 2013, 9, 70–76. [Google Scholar] [PubMed]
- Ramírez-Vélez, R.; Anzola, A.; Martinez-Torres, J.; Vivas, A.; Tordecilla-Sanders, A.; Prieto-Benavides, D.; Izquierdo, M.; Correa-Bautista, J.E.; Garcia-Hermoso, A. Metabolic Syndrome and Associated Factors in a Population-Based Sample of Schoolchildren in Colombia: The FUPRECOL Study. Metab. Syndr. Relat. Disord. 2016, 14, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Nasreddine, L.; Naja, F.; Tabet, M.; Habbal, M.; El-Aily, A.; Haikal, C.; Sidani, S.; Adra, N.; Hwalla, N. Obesity is associated with insulin resistance and components of the metabolic syndrome in Lebanese adolescents. Ann. Hum. Biol. 2012, 39, 122–128. [Google Scholar] [CrossRef] [PubMed]
- Shashaj, B.; Bedogni, G.; Graziani, M.P.; Tozzi, A.E.; DiCorpo, M.L.; Morano, D.; Tacconi, L.; Veronelli, P.; Contoli, B.; Manco, M. Origin of cardiovascular risk in overweight preschool children: A cohort study of cardiometabolic risk factors at the onset of obesity. JAMA Pediatr. 2014, 168, 917–924. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Liu, J.; Liu, J.; Zhu, G.; Han, S. Relation between metabolic syndrome and body compositions among Chinese adolescents and adults from a large-scale population survey. BMC Public Health 2017, 17, 337. [Google Scholar] [CrossRef] [PubMed]
- Zaki, M.E.; El-Bassyouni, H.T.; El-Gammal, M.; Kamal, S. Indicators of the metabolic syndrome in obese adolescents. Arch. Med. Sci. 2015, 11, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Haghighatdoost, F.; Sarrafzadegan, N.; Mohammadifard, N.; Asgary, S.; Boshtam, M.; Azadbakht, L. Assessing body shape index as a risk predictor for cardiovascular diseases and metabolic syndrome among Iranian adults. Nutrition 2014, 30, 636–644. [Google Scholar] [CrossRef]
- Behboudi-Gandevani, S.; Ramezani Tehrani, F.; Cheraghi, L.; Azizi, F. Could, “a body shape index” and “waist to height ratio” predict insulin resistance and metabolic syndrome in polycystic ovary syndrome? Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 205, 110–114. [Google Scholar] [CrossRef]
| IDF | Cook | De Ferranti | Weiss | Viner | Duncan | Rodríguez-Moran | Cruz & Goran | |
|---|---|---|---|---|---|---|---|---|
| Age (years) | 10–16 | 12–19 | ≥12 | 4–20 | 2–18 | 12–19 | 10–18 | 8–13 |
| Number of components | Obesity + 2 components | ≥3 | ≥3 | ≥3 | ≥4 | ≥3 | ≥3 | ≥3 |
| Obesity | WC > 90 percentile | WC > 90 percentile | WC > 75 percentile | BMI z-score ≥ 2 | BMI ≥ 95 percentile | WC ≥ 90 percentile | WC ≥ 90 percentile | WC ≥ 90 percentile |
| Glucose (mg/dL) | ≥100 | ≥110 | ≥110 | ≥140 | ≥110 | ≥110 | ≥110 | <100 |
| TG (mg/dL) | ≥150 | ≥110 | ≥100 | >95 percentile | ≥150 | ≥110 | ≥90 percentile | ≥90 percentile |
| HDL-cholesterol (mg/dL) | ≤40 | ≤40 | <50 girls <45 boys | <5 percentile | ≤35 | <40 | - | <10 percentile |
| SBP (mmHg) | ≥130 | >90 percentile | >90 percentile | >95 percentile | >95 percentile | ≥90 percentile | ≥90 percentile | ≥90 percentile |
| DBP (mmHg) | ≥85 | - | - | - | - | - | - | - |
| Variables | Boys (n = 456) | Girls (n = 525) | ||
|---|---|---|---|---|
| Mean | SD | Mean | SD | |
| Age (years) | 13.2 | 1.2 | 13.3 | 1.2 |
| Weight (kg) | 57.1 | 14.1 | 53.1 | 11.0 *** |
| Fat (%) | 27.3 | 8.3 | 29.6 | 7.8 *** |
| BMI (kg/m2) | 21.5 | 4.0 | 21.1 | 3.6 |
| WC (cm) | 73.7 | 11.8 | 71.3 | 9.6 *** |
| Glucose (mg/dL) | 86.2 | 31.2 | 85.2 | 28.7 |
| TG (mg/dL) | 129.2 | 59.3 | 125.0 | 46.2 |
| Cholesterol (mg/dL) | 81.8 | 17.3 | 81.4 | 15.7 |
| LDL-c (mg/dL) | 93.4 | 23.6 | 92.9 | 22.5 |
| HDL-c (mg/dL) | 40.1 | 2.8 | 40.0 | 3.1 |
| SBP (mmHg) | 119.6 | 15.7 | 116.9 | 15.1 ** |
| DBP (mmHg) | 64.5 | 9.2 | 63.9 | 8.8 |
| Insulin (mU/mL) | 21.0 | 10.2 | 20.2 | 9.0 |
| HOMA-IR | 4.5 | 2.9 | 4.3 | 3.1 |
| Variables | IDF | Cook | De Ferranti | Weiss | Viner | Duncan | Rodríguez-Moran | Cruz & Goran | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| MetS (number) | 41 | 39 | 63 | 32 | 25 | 68 | 68 | 68 | ||||||||
| Age (y) | 13.6 a | 1.0 | 12.9 a | 1.2 | 13.0 a | 1.0 | 13.2 a | 1.2 | 13.0 a | 1.2 | 13.1 a | 1.1 | 13.1 a | 1.1 | 13.1 a | 1.1 |
| Weight (kg) | 69.3 a | 13.6 | 54.9 b | 14.3 | 59.8 b | 15.6 | 59.3 ab | 17.4 | 59.1 ab | 17.0 | 60.3 ab | 15.3 | 60.3 ab | 15.3 | 60.3 ab | 15.3 |
| Fat (%) | 34.1 a | 6.5 | 29.3 a | 7.8 | 32.1 a | 7.7 | 29.4 a | 8.4 | 30.3 a | 8.1 | 31.3 a | 7.8 | 31.3 a | 7.8 | 31.3 a | 7.8 |
| BMI (kg/m2) | 25.2 a | 4.3 | 21.8 a | 4.2 | 23.6 a | 4.7 | 22.4 a | 5.5 | 22.4 a | 5.0 | 23.4 a | 4.5 | 23.4 a | 4.5 | 23.4 a | 4.5 |
| WC (cm) | 86.0 a | 9.9 | 74.6 b | 11.7 | 80.2 ab | 12.7 | 76.9 b | 14.0 | 77.8 ab | 13.5 | 79.5 ab | 12.4 | 79.5 ab | 12.4 | 79.5 ab | 12.4 |
| Glucose (mg/dL) | 128.4 a | 53.5 | 172.9 b | 47.4 | 139.5 a | 56.9 | 191.4 b | 27.8 | 193.7 b | 24.4 | 136.0 a | 56.2 | 136.0 a | 56.2 | 136.0 a | 56.2 |
| TG (mg/dL) | 193.0 a | 120.1 | 257.9 a | 146.4 | 210.1 ac | 133.9 | 295.0 bc | 143.9 | 338.9 b | 132.5 | 204.0 a | 130.6 | 204.0 a | 130.6 | 204.0 a | 130.6 |
| Chol (mg/dL) | 129.6 a | 35.6 | 151.6 ab | 27.1 | 134.6 a | 35.7 | 163.0 b | 7.3 | 163.2 b | 8.1 | 133.4 a | 35.4 | 133.4 a | 35.4 | 133.4 a | 35.4 |
| LDL-c (mg/dL) | 101.9 a | 23.8 | 119.7 b | 24.9 | 107.2 ab | 26.7 | 126.3 c | 19.8 | 126.0 c | 21.3 | 107.2 ab | 25.3 | 107.2 ab | 25.3 | 107.2 ab | 25.3 |
| HDL-c (mg/dL) | 35.7 a | 3.4 | 33.1 bc | 2.2 | 35.0 a | 3.4 | 32.3 c | 1.6 | 32.4 c | 1.8 | 34.9 ab | 3.2 | 34.9 ab | 3.2 | 34.9 ab | 3.2 |
| SBP (mmHg) | 137.2 a | 14.7 | 121.6 b | 17.0 | 123.5 b | 17.3 | 122.9 b | 17.9 | 123.4 b | 18.8 | 124.0 b | 16.6 | 124.0 b | 16.6 | 124.0 b | 16.6 |
| DBP mmHg) | 74.1 a | 11.4 | 64.6 b | 8.6 | 65.5 b | 9.0 | 65.1 b | 9.0 | 63.9 b | 9.2 | 66.3 b | 8.7 | 66.3 b | 8.7 | 66.3 b | 8.7 |
| Insulin (mU/mL) | 28.7 a | 15.1 | 20.9 a | 10.4 | 25.5 a | 13.6 | 23.7 a | 14.2 | 24.0 a | 14.3 | 24.9 a | 13.3 | 24.9 a | 13.3 | 24.9 a | 13.3 |
| HOMA-IR | 8.8 a | 6.0 | 9.1 a | 5.7 | 8.3 a | 5.1 | 10.7 a | 5.6 | 11.1 a | 6.2 | 8.0 a | 5.0 | 8.0 a | 5.0 | 8.0 a | 5.0 |
| Variables | IDF | Cook | de Ferranti | Weiss | Viner | Duncan | Rodríguez-Moran | Cruz & Goran | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| MetS (number) | 32 | 58 | 97 | 18 | 21 | 134 | 86 | 171 | ||||||||
| Age (years) | 13.3 a | 1.1 | 12.9 a | 1.1 | 12.9 a | 1.1 | 13.1 a | 1.4 | 13.2 a | 1.3 | 13.0 a | 1.1 | 13.1 a | 1.1 | 13.1 a | 1.1 |
| Weight (kg) | 67.1 a | 10.1 | 51.8 b | 12.8 | 55.4 bc | 11.9 | 57.0 ab | 18.0 | 59.2 ab | 16.6 | 54.1 b | 10.4 | 60.3 ac | 15.3 | 54.8 b | 11.0 |
| Fat (%) | 38.1 a | 6.6 | 30.0 a | 8.4 | 32.6 a | 8.0 | 31.9 a | 11.3 | 33.0 a | 9.9 | 31.9 a | 7.5 | 31.3 a | 7.8 | 32.0 a | 7.6 |
| BMI (kg/m2) | 25.8 a | 3.5 | 21.1 b | 4.3 | 22.5 bc | 4.0 | 22.4 ab | 5.9 | 23.1 ab | 5.2 | 22.1 bc | 3.6 | 23.4 a | 4.5 | 22.1 bc | 3.7 |
| WC (cm) | 83.2 a | 7.5 | 70.5 b | 10.4 | 75.2 bc | 10.3 | 73.7 bc | 14.8 | 75.5 ab | 13.3 | 73.6 b | 9.1 | 79.5 ac | 12.4 | 73.7 b | 9.5 |
| Glucose (mg/dL) | 132.0 abc | 52.2 | 148.3 b | 51.8 | 121.1 a | 52.3 | 194.4 d | 7.1 | 188.2 d | 22.5 | 110.0 c | 48.2 | 136.0 ab | 56.2 | 104.5 | 43.9 c |
| TG (mg/dL) | 196.2 ab | 120.9 | 191.6 ab | 114.9 | 164.5 ab | 98.1 | 329.8 c | 121.9 | 312.9 c | 128.1 | 151.9 a | 85.9 | 204.0 b | 130.6 | 145.5 a | 77.0 |
| Chol (mg/dL) | 132.3 ab | 33.2 | 139.9 a | 30.9 | 122.1 b | 35.8 | 161.4 a | 4.8 | 160.0 a | 4.9 | 114.1 c | 34.8 | 133.4 a | 35.4 | 109.3 c | 32.5 |
| LDL-c (mg/dL) | 106.3 ab | 20.0 | 109.3 a | 21.2 | 98.2 b | 23.3 | 126.1 a | 16.1 | 122.8 a | 15.9 | 92.9 c | 22.3 | 107.2 a | 25.3 | 90.1 c | 21.2 |
| HDL-c (mg/dL) | 35.3 ab | 2.6 | 33.7 a | 4.5 | 36.1 b | 4.8 | 32.8 a | 1.3 | 33.1 a | 1.3 | 37.2 c | 4.6 | 34.9 | 3.2 a | 38.0 | 4.3 c |
| SBP (mmHg) | 132.3 a | 10.2 | 113.4 b | 17.1 | 117.7 bc | 17.0 | 116.4 bc | 20.8 | 118.8 ab | 19.5 | 117.2 b | 15.5 | 124.0 ac | 16.6 | 117.5 b | 15.3 |
| DBP mmHg) | 70.7 a | 8.8 | 62.1 a | 8.8 | 62.6 a | 9.5 | 64.4 a | 10.2 | 64.4 a | 10.1 | 63.0 a | 9.0 | 66.3 a | 8.7 | 63.7 a | 8.9 |
| Insulin (mU/mL) | 11.0 a | 8.0 | 8.4 b | 6.6 | 7.1 bc | 5.6 | 13.8 a | 8.7 | 13.6 a | 8.5 | 6.1 b | 5.0 | 8.0 ab | 5.0 | 5.8 c | 4.6 |
| HOMA-IR | 32.5 a | 15.8 | 22.0 b | 12.2 | 23.6 b | 12.3 | 28.8 ab | 17.9 | 28.8 ab | 17.1 | 21.7 b | 10.4 | 24.9 ab | 13.3 | 21.8 b | 10.5 |
| Variables | IDF | Cook | De Ferranti | Weiss | Viner | Duncan | Rodríguez-Moran | Cruz & Goran | Maximum | Author | Minimum | Author |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fat (%) | 0.757 | 0.582 | 0.696 | 0.576 | 0.612 | 0.666 | 0.666 | 0.666 | 0.757 | IDF | 0.576 | Weiss |
| BMI | 0.783 | 0.523 | 0.663 | 0.523 | 0.542 | 0.657 | 0.657 | 0.657 | 0.783 | IDF | 0.523 | Weiss and Cook |
| WC | 0.831 | 0.526 | 0.680 | 0.564 | 0.601 | 0.669 | 0.669 | 0.669 | 0.831 | IDF | 0.526 | Cook |
| WHR | 0.789 | 0.609 | 0.715 | 0.635 | 0.690 | 0.655 | 0.655 | 0.655 | 0.789 | IDF | 0.609 | Cook |
| ABSI | 0.663 | 0.559 | 0.652 | 0.564 | 0.617 | 0.615 | 0.615 | 0.615 | 0.663 | IDF | 0.559 | Cook |
| BAI | 0.686 | 0.559 | 0.689 | 0.496 | 0.512 | 0.664 | 0.664 | 0.664 | 0.689 | de Ferranti | 0.496 | Weiss |
| AVI | 0.831 | 0.524 | 0.678 | 0.562 | 0.599 | 0.668 | 0.668 | 0.668 | 0.831 | IDF | 0.524 | Cook |
| BRI | 0.800 | 0.590 | 0.728 | 0.572 | 0.615 | 0.700 | 0.700 | 0.700 | 0.800 | IDF | 0.572 | Weiss |
| CI | 0.767 | 0.577 | 0.706 | 0.593 | 0.644 | 0.675 | 0.675 | 0.675 | 0.767 | IDF | 0.577 | Cook |
| BAIp | 0.752 | 0.521 | 0.671 | 0.499 | 0.515 | 0.668 | 0.668 | 0.668 | 0.752 | IDF | 0.499 | Weiss |
| Variables | IDF | Cook | De Ferranti | Weiss | Viner | Duncan | Rodríguez-Moran | Cruz & Goran | Maximum | Author | Minimum | Author |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Fat (%) | 0.812 | 0.638 | 0.638 | 0.559 | 0.605 | 0.619 | 0.557 | 0.634 | 0.812 | IDF | 0.557 | Rodriguez-Moran |
| BMI | 0.855 | 0.635 | 0.635 | 0.517 | 0.607 | 0.627 | 0.557 | 0.636 | 0.855 | IDF | 0.517 | Weiss |
| WC | 0.866 | 0.645 | 0.645 | 0.535 | 0.614 | 0.616 | 0.554 | 0.626 | 0.866 | IDF | 0.535 | Weiss |
| WHR | 0.717 | 0.662 | 0.662 | 0.528 | 0.557 | 0.536 | 0.533 | 0.537 | 0.717 | IDF | 0.528 | Weiss |
| ABSI | 0.585 | 0.590 | 0.590 | 0.489 | 0.496 | 0.541 | 0.511 | 0.539 | 0.590 | de Ferranti | 0.489 | Weiss |
| BAI | 0.797 | 0.651 | 0.651 | 0.513 | 0.585 | 0.714 | 0.590 | 0.702 | 0.797 | IDF | 0.513 | Weiss |
| AVI | 0.867 | 0.643 | 0.643 | 0.535 | 0.616 | 0.619 | 0.554 | 0.629 | 0.867 | IDF | 0.535 | Weiss |
| BRI | 0.848 | 0.677 | 0.677 | 0.519 | 0.594 | 0.663 | 0.575 | 0.661 | 0.848 | IDF | 0.519 | Weiss |
| CI | 0.730 | 0.642 | 0.642 | 0.520 | 0.552 | 0.597 | 0.542 | 0.593 | 0.730 | IDF | 0.520 | Weiss |
| BAIp | 0.840 | 0.624 | 0.624 | 0.525 | 0.608 | 0.687 | 0.575 | 0.688 | 0.840 | IDF | 0.525 | Weiss |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Perona, J.S.; Schmidt-RioValle, J.; Fernández-Aparicio, Á.; Correa-Rodríguez, M.; Ramírez-Vélez, R.; González-Jiménez, E. Waist Circumference and Abdominal Volume Index Can Predict Metabolic Syndrome in Adolescents, but only When the Criteria of the International Diabetes Federation are Employed for the Diagnosis. Nutrients 2019, 11, 1370. https://doi.org/10.3390/nu11061370
Perona JS, Schmidt-RioValle J, Fernández-Aparicio Á, Correa-Rodríguez M, Ramírez-Vélez R, González-Jiménez E. Waist Circumference and Abdominal Volume Index Can Predict Metabolic Syndrome in Adolescents, but only When the Criteria of the International Diabetes Federation are Employed for the Diagnosis. Nutrients. 2019; 11(6):1370. https://doi.org/10.3390/nu11061370
Chicago/Turabian StylePerona, Javier S., Jacqueline Schmidt-RioValle, Ángel Fernández-Aparicio, María Correa-Rodríguez, Robinson Ramírez-Vélez, and Emilio González-Jiménez. 2019. "Waist Circumference and Abdominal Volume Index Can Predict Metabolic Syndrome in Adolescents, but only When the Criteria of the International Diabetes Federation are Employed for the Diagnosis" Nutrients 11, no. 6: 1370. https://doi.org/10.3390/nu11061370
APA StylePerona, J. S., Schmidt-RioValle, J., Fernández-Aparicio, Á., Correa-Rodríguez, M., Ramírez-Vélez, R., & González-Jiménez, E. (2019). Waist Circumference and Abdominal Volume Index Can Predict Metabolic Syndrome in Adolescents, but only When the Criteria of the International Diabetes Federation are Employed for the Diagnosis. Nutrients, 11(6), 1370. https://doi.org/10.3390/nu11061370









