Glycine Metabolism and Its Alterations in Obesity and Metabolic Diseases
Abstract
1. Introduction
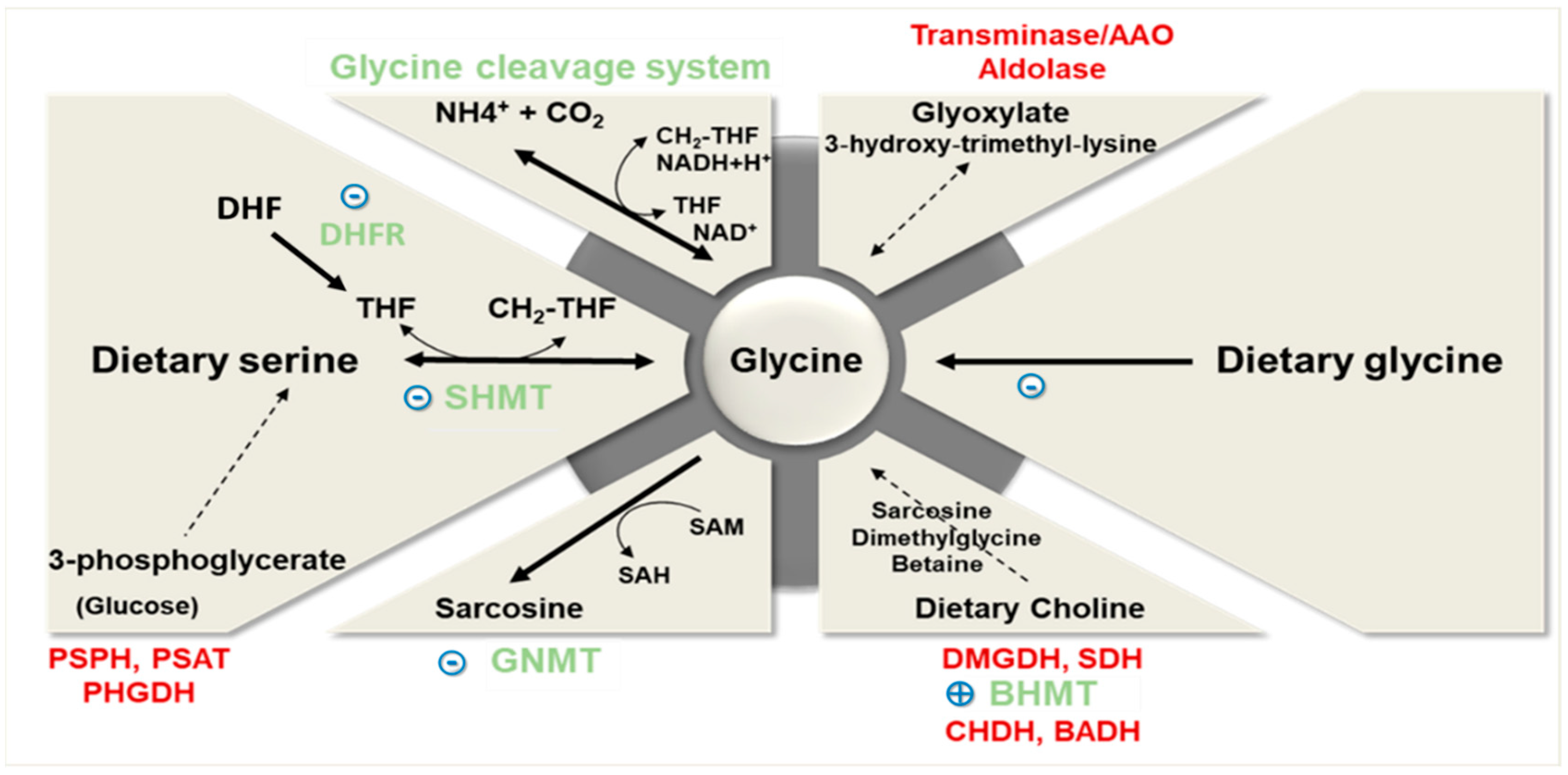
2. Glycine Dietary Intake and Metabolism
2.1. Glycine Synthesis
2.2. Glycine Catabolism
2.3. Glycine Uptake
2.4. Glycine Conjugation and Excretion
3. Plasma Concentrations of Glycine are Decreased in Obesity and Associated Metabolic Disorders, Although Dietary Intake are Unaltered
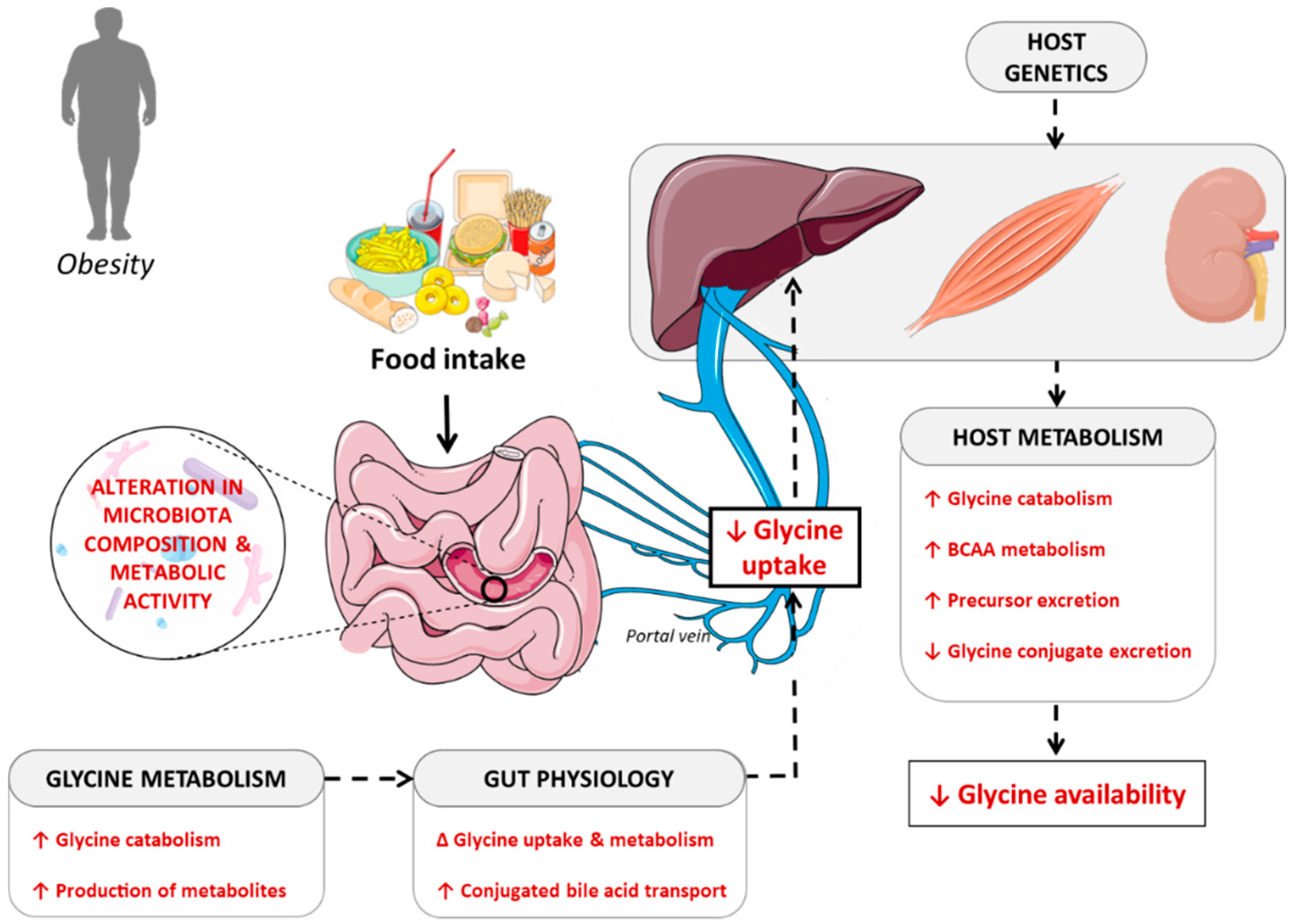
4. Potential Causes of Decreased Glycine Availability
4.1. Importance of Dietary Patterns in Determining Glycine Availability
4.2. Contribution of the Gut Microbiota in Determining Glycine Bioavailability
4.3. Interaction between Host Metabolism and Fate of Glycine
5. Potential Benefits of Glycine Supplementation in Obesity and Associated Metabolic Disorders
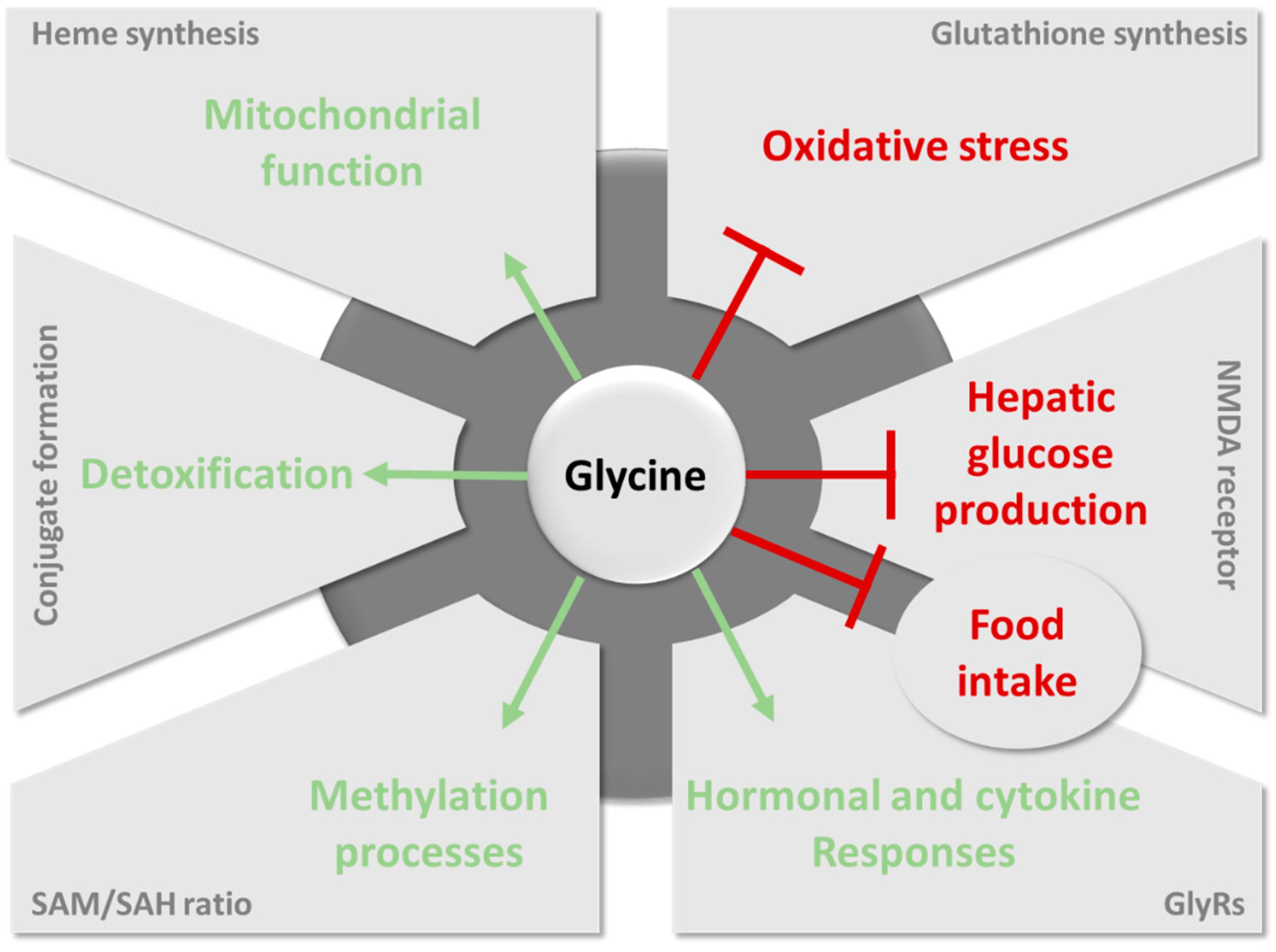
6. The Contribution of Glycine to Host Metabolism and the Pathogenesis of Metabolic Disorders
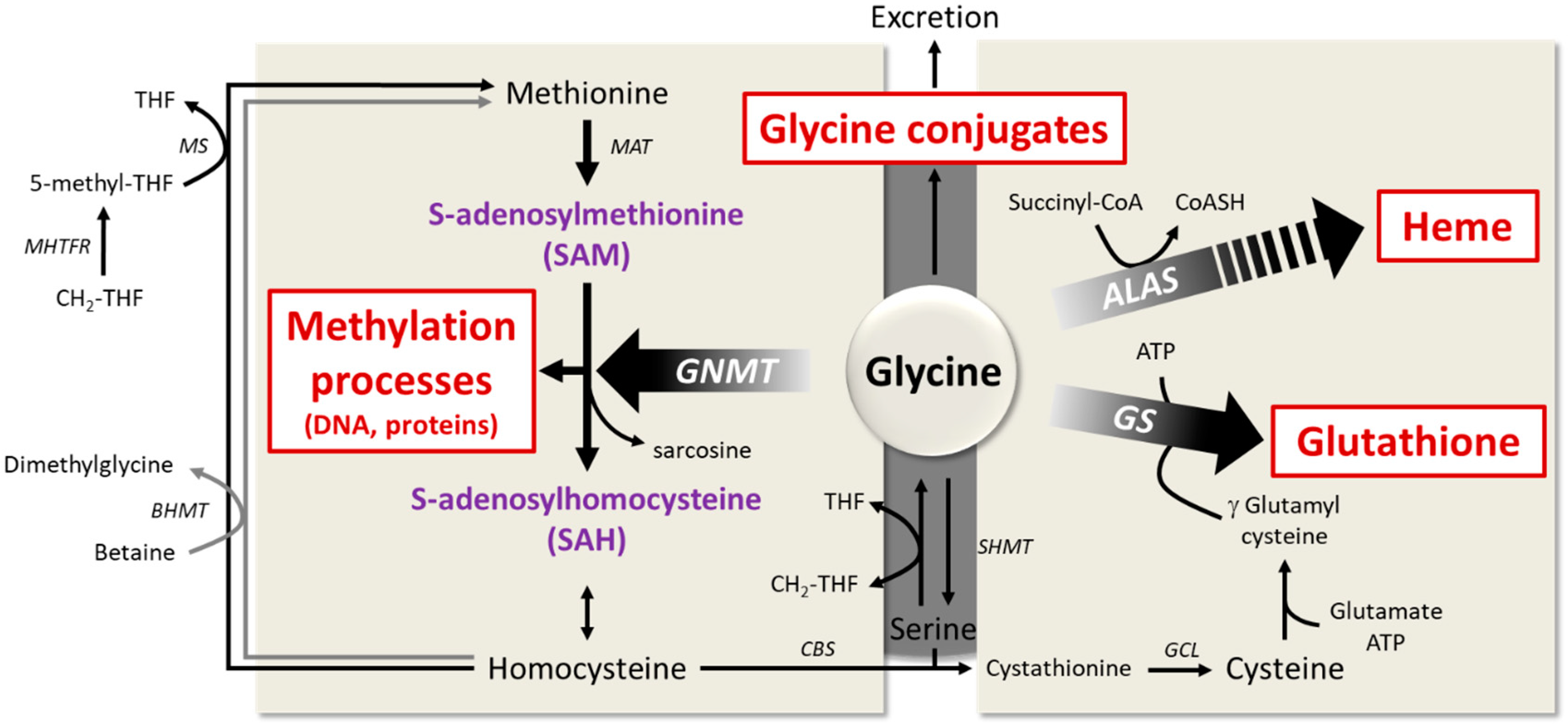
6.1. Importance of Glycine for Antioxidant Protection by Glutathione
6.2. The Role of Glycine in Heme Biosynthesis
6.3. Importance of Glycine Conjugation and Urinary Excretion
6.4. The Key Role of Glycine in the One-Carbon Metabolism
6.5. Glycine as a Neurotransmitter
7. Conclusions
Funding
Conflicts of Interest
Abbreviations
| AGAT | Alanine:glyoxylate aminotransferase |
| ALA | delta-aminolevulinic acid |
| ALAS | delta-aminolevulinic acid synthase |
| BA | Bile acids |
| BAAT | Bile acid-coenzyme A: amino acid N-acyltransferase |
| BCAA | Branched chain amino acids |
| BCKDH | Branched-chain keto acid dehydrogenase |
| BHMT | Betaine-homocysteine S-methyltransferase |
| CH2-THF | 5,10-methylene tetrahydrofolate |
| CPS1 | Carbamoyl-Phosphate Synthase 1 |
| DHF | dihydrofolate |
| DHFR | dihydrofolate reductase |
| DMGDH | Dimethylglycine dehydrogenase |
| EPIC | European Prospective Investigation into Cancer and Nutrition |
| GLYAT | Glycine N-acyltransferases |
| GlyR | Glycine receptor |
| GNMT | Glycine N-methyltransferase |
| GSG index | Glutamate-serine-glycine index |
| GWAS | Genome-wide association study |
| HOMA index | Homeostatic Model Assessment index |
| NAFLD | Non-alcoholic fatty liver disease |
| NMDA | N-methyl-D-aspartate |
| SAM | S-adenosylmethionine |
| SAH | S-adenosylhomocysteine |
| SDH | Sarcosine dehydrogenase (SDH) |
| SHMT | Serine hydroxymethyltransferase |
| SNP | Single nucleotide polymorphism |
| T2DM | Type 2 diabetes mellitus |
| THF | tetrahydrofolate |
References
- Jackson, A.A.; Badaloo, A.V.; Forrester, T.; Hibbert, J.M.; Persaud, C. Urinary excretion of 5-oxoproline (pyroglutamic aciduria) as an index of glycine insufficiency in normal man. Br. J. Nutr. 1987, 58, 207–214. [Google Scholar] [CrossRef] [PubMed]
- Gersovitz, M.; Bier, D.; Matthews, D.; Udall, J.; Munro, H.N.; Young, V.R. Dynamic aspects of whole body glycine metabolism: Influence of protein intake in young adult and elderly males. Metabolism 1980, 29, 1087–1094. [Google Scholar] [CrossRef]
- Yu, Y.M.; Yang, R.D.; Matthews, D.E.; Wen, Z.M.; Burke, J.F.; Bier, D.L.; Young, V.R. Quantitative aspects of glycine and alanine nitrogen metabolism in postabsorptive young men: Effects of level of nitrogen and dispensable amino acid intake. J. Nutr. 1985, 115, 399–410. [Google Scholar] [CrossRef]
- Meléndez-Hevia, E.; De Paz-Lugo, P.; Cornish-Bowden, A.; Cárdenas, M.L. A weak link in metabolism: The metabolic capacity for glycine biosynthesis does not satisfy the need for collagen synthesis. J. Biosci. 2009, 34, 853–872. [Google Scholar] [CrossRef] [PubMed]
- Luka, Z.; Cerone, R.; Phillips, J.A., 3rd; Mudd, H.S.; Wagner, C. Mutations in human glycine N-methyltransferase give insights into its role in methionine metabolism. Hum Genet. 2002, 110, 68–74. [Google Scholar] [CrossRef]
- Martínez-Chantar, M.L.; Vázquez-Chantada, M.; Ariz, U.; Martínez, N.; Varela, M.; Luka, Z.; Capdevila, A.; Rodríguez, J.; Aransay, A.M.; Matthiesen, R.; et al. Loss of the glycine N-methyltransferase gene leads to steatosis and hepatocellular carcinoma in mice. Hepatology 2008, 47, 1191–1199. [Google Scholar] [CrossRef] [PubMed]
- Durkin, P.J.; Friedberg, F. The synthesis of glutathione in torula utilis studied with 14C-carboxyl labeled glycine. Biochim. Biophys. Acta 1952, 9, 105–106. [Google Scholar] [CrossRef]
- Guasch-Ferre, M.; Hruby, A.; Toledo, E.; Clish, C.B.; Martınez-Gonzalez, M.A.; Salas-Salvado, J.; Hu, F.B. Metabolomics in prediabetes and diabetes: A systematic review and meta-analysis. Diabetes Care 2016, 39, 833–846. [Google Scholar] [CrossRef]
- Gaggini, M.; Carli, F.; Rosso, C.; Buzzigoli, E.; Marietti, M.; Della Latta, V.; Ciociaro, D.; Abate, M.L.; Gambino, R.; Cassader, M.; et al. Altered amino acid concentrations in NAFLD: Impact of obesity and insulin resistance. Hepatology 2018, 67, 145–158. [Google Scholar] [CrossRef]
- Wang, W.; Wu, Z.; Dai, Z.; Yang, Y.; Wang, J.; Wu, G. Glycine metabolism in animals and humans: Implications for nutrition and health. Amino Acids 2013, 45, 463–477. [Google Scholar] [CrossRef]
- Adeva-Andany, M.; Souto-Adeva, G.; Ameneiros-Rodríguez, E.; Fernández-Fernández, C.; Donapetry-García, C.; Domínguez-Montero, A. Insulin resistance and glycine metabolism in humans. Amino Acids 2018, 50, 11–27. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Torres, I.; Zuniga-Munoz, A.M.; Guarner-Lans, V. Beneficial effects of the amino acid glycine. Mini-Rev. Med. Chem. 2017, 17, 15–32. [Google Scholar] [CrossRef] [PubMed]
- Razak, M.A.; Begum, P.S.; Viswanath, B.; Rajagopal, S. Multifarious beneficial effect of nonessential amino acid, glycine: A review. Oxid. Med. Cell. Longev. 2017, 2017, 1716701. [Google Scholar] [CrossRef] [PubMed]
- Gar, C.; Rottenkolber, M.; Prehn, C.; Adamski, J.; Seissler, J.; Lechner, A. Serum and plasma amino acids as markers of prediabetes, insulin resistance, and incident diabetes. Crit. Rev. Clin. Lab. Sci. 2018, 55, 21–32. [Google Scholar] [CrossRef] [PubMed]
- Gall, W.E.; Beebe, K.; Lawton, K.A.; Adam, K.P.; Mitchell, M.W.; Nakhle, P.J.; Ryals, J.A.; Milburn, M.V.; Nannipieri, M.; Camastra, S.; et al. α-hydroxybutyrate is an early biomarker of insulin resistance and glucose intolerance in a nondiabetic population. PLoS ONE 2010, 5, e10883. [Google Scholar] [CrossRef] [PubMed]
- Thalacker-Mercer, A.E.; Ingram, K.H.; Guo, F.; Ilkayeva, O.; Newgard, C.B.; Garvey, W.T. BMI, RQ, diabetes, and sex affect the relationships between amino acids and clamp measures of insulin action in humans. Diabetes 2014, 63, 791–800. [Google Scholar] [CrossRef]
- Tulipani, S.; Palau-Rodriguez, M.; Miñarro Alonso, A.; Cardona, F.; Marco-Ramell, A.; Zonja, B.; Lopez de Alda, M.; Muñoz-Garach, A.; Sanchez-Pla, A.; Tinahones, F.J.; et al. Biomarkers of Morbid Obesity and Prediabetes by Metabolomic Profiling of Human Discordant Phenotypes. Clin. Chim. Acta 2016, 463, 53–61. [Google Scholar] [CrossRef]
- Cheng, S.; Rhee, E.P.; Larson, M.G.; Lewis, G.D.; McCabe, E.L.; Shen, D.; Palma, M.L.; Roberts, L.D.; Dejam, A.; Souza, A.L.; et al. Metabolite profiling identifies pathways associated with metabolic risk in humans. Circulation 2012, 125, 2222–2231. [Google Scholar] [CrossRef]
- Takashina, C.; Tsujino, I.; Watanabe, T.; Sakaue, S.; Ikeda, D.; Yamada, A.; Sato, T.; Ohira, H.; Otsuka, Y.; Oyama-Manabe, N.; et al. Associations among the plasma amino acid profile, obesity, and glucose metabolism in Japanese adults with normal glucose tolerance. Nutr. Metab. (Lond.) 2016, 13, 5. [Google Scholar] [CrossRef]
- Labonte, C.C.; Farsijani, S.; Marliss, E.B.; Bougeon, R.; Morais, J.A.; Pereira, S.; Bassil, M.; Winter, A.; Murphy, J.; Combs, T.P.; et al. Plasma Amino Acids vs Conventional Predictors of Insulin Resistance Measured by the Hyperinsulinemic Clamp. J. Endocr. Soc. 2017, 1, 861–873. [Google Scholar] [CrossRef]
- US Department of Agriculture, Agricultural Research Service, Nutrient Data Laboratory. USDA National Nutrient Database for Standard Reference, Release 28. Version Current: September 2015. Internet: /nea/bhnrc/ndl.
- Schmidt, J.A.; Rinaldi, S.; Scalbert, A.; Ferrari, P.; Achaintre, D.; Gunter, M.J.; Appleby, P.N.; Key, T.J.; Travis, R.C. Plasma concentrations and intakes of amino acids in male meat-eaters, fish-eaters, vegetarians and vegans: A cross-sectional analysis in the EPIC-Oxford cohort. Eur. J. Clin. Nutr. 2016, 70, 306–312. [Google Scholar] [CrossRef] [PubMed]
- Lamers, Y.; Williamson, J.; Gilbert, L.R.; Stacpoole, P.W.; Gregory, J.F., 3rd. Glycine turnover and decarboxylation rate quantified in healthy men and women using primed, constant infusions of [1,2-(13)C2]glycine and [(2)H3]leucine. J. Nutr. 2007, 137, 2647–2652. [Google Scholar] [CrossRef] [PubMed]
- Robert, J.J.; Bier, D.M.; Zhao, X.H.; Matthews, D.E.; Young, V.R. Glucose and insulin effects on the novo amino acid synthesis in young men: Studies with stable isotope labeled alanine, glycine, leucine, and lysine. Metabolism 1982, 31, 1210–1218. [Google Scholar] [CrossRef]
- Robert, J.J.; Beaufrere, B.; Koziet, J.; Desjeux, J.F.; Bier, D.M.; Young, V.R.; Lestradet, H. Whole body de novo amino acid synthesis in type I (insulin-dependent) diabetes studied with stable isotope-labeled leucine, alanine, and glycine. Diabetes 1985, 34, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Ducker, G.S.; Rabinowitz, J.D. One-carbon metabolism in health and disease. Cell Metab. 2017, 25, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Garrow, T.A.; Brenner, A.A.; Whitehead, V.M.; Chen, X.N.; Duncan, R.G.; Korenberg, J.R.; Shane, B. Cloning of human cDNAs encoding mitochondrial and cytosolic serine hydroxymethyltransferases and chromosomal localization. J. Biol. Chem. 1993, 268, 11910–11916. [Google Scholar] [PubMed]
- Pfendner, W.; Pizer, L.I. The metabolism of serine and glycine in mutant lines of Chinese hamster ovary cells. Arch. Biochem. Biophys. 1980, 200, 503–512. [Google Scholar] [CrossRef]
- Melendez-Hevia, E.; Paz-Lugo, P.D. Branch-point stoichiometry can generate weak links in metabolism: The case of glycine biosynthesis. J. Biosci. 2008, 33, 771–780. [Google Scholar] [CrossRef]
- Mardinoglu, A.; Agren, R.; Kampf, C.; Asplund, A.; Uhlen, M.; Nielsen, J. Genome-scale metabolic modelling of hepatocytes reveals serine deficiency in patients with non-alcoholic fatty liver disease. Nat. Commun. 2014, 5, 3083. [Google Scholar] [CrossRef]
- Lewis, R.M.; Godfrey, K.M.; Jackson, A.A.; Cameron, I.T.; Hanson, M.A. Low serine hydroxymethyltransferase activity in the human placenta has important implications for fetal glycine supply. J. Clin. Endocrinol. Metab. 2005, 90, 1594–1598. [Google Scholar] [CrossRef]
- Mardinoglu, A.; Bjornson, E.; Zhang, C.; Klevstig, M.; Söderlund, S.; Ståhlman, M.; Adiels, M.; Hakkarainen, A.; Lundbom, N.; Kilicarslan, M.; et al. Personal model-assisted identification of NAD(+)and glutathione metabolism as intervention target in NAFLD. Mol. Syst. Biol. 2017, 13, 916. [Google Scholar] [CrossRef] [PubMed]
- Soloway, S.; Stetten, D., Jr. The metabolism of choline and its conversion to glycine in the rat. J. Biol. Chem. 1953, 204, 207–214. [Google Scholar] [PubMed]
- Ahlhoff, C.; Worsch, S.; Sailer, M.; Hummel, B.A.; Fiamoncini, J.; Uebel, K.; Obeid, R.; Scherling, C.; Geisel, J.; Bader, B.L.; et al. Methyl-donor supplementation in obese mice prevents the progression of NAFLD, activates AMPK and decreases acyl-carnitine levels. Mol. Metab. 2014, 3, 565–580. [Google Scholar] [CrossRef] [PubMed]
- Dahlhoff, C.; Desmarchelier, C.; Sailer, M.; Fürst, R.W.; Haag, A.; Ulbrich, S.E.; Hummel, B.; Obeid, R.; Geisel, J.; Bader, B.L.; et al. Hepatic methionine homeostasis is conserved in C57BL/6N mice on high-fat diet despite major changes in hepatic one-carbon metabolism. PLoS ONE 2013, 8, e57387. [Google Scholar] [CrossRef]
- Holmes, R.P.; Assimos, D.G. Glyoxylate synthesis, and its modulation and influence on oxalate synthesis. J. Urol. 1998, 160, 1617–1624. [Google Scholar] [CrossRef]
- Eastman, H.B.; Swick, A.G.; Schmitt, M.C.; Azizkhan, J.C. Stimulation of dihydrofolate reductase promoter activity by antimetabolic drugs. Proc. Natl. Acad. Sci. USA 1991, 88, 8572–8576. [Google Scholar] [CrossRef] [PubMed]
- Urlaub, G.; Chasin, L.A. Isolation of Chinese hamster cell mutants deficient in dihydrofolate reductase activity. Proc. Natl. Acad. Sci. USA 1980, 77, 4216–4220. [Google Scholar] [CrossRef]
- Meierhofer, D.; Weidner, C.; Sauer, S. Integrative analysis of transcriptomics, proteomics, and metabolomics data of white adipose and liver tissue of high-fat diet and rosiglitazone-treated insulin-resistant mice identified pathway alterations and molecular hubs. J. Proteome Res. 2014, 13, 5592–5602. [Google Scholar] [CrossRef]
- Yamamoto, E.; Nakamura, T.; Kataoka, K.; Tokutomi, Y.; Dong, Y.F.; Fukuda, M.; Nako, H.; Yasuda, O.; Ogawa, H.; Kim-Mitsuyama, S. Nifedipine prevents vascular endothelial dysfunction in a mouse model of obesity and type 2 diabetes, by improving eNOS dysfunction and dephosphorylation. Biochem. Biophys. Res. Commun. 2010, 403, 258–263. [Google Scholar] [CrossRef]
- Kikuchi, G.; Motokawa, Y.; Yoshida, T.; Hiraga, K. Glycine cleavage system: Reaction mechanism, physiological significance, and hyperglycinemia. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2008, 84, 246–263. [Google Scholar] [CrossRef]
- Matthews, D.E.; Conway, J.M.; Young, V.R.; Bier, D.M. Glycine nitrogen metabolism in man. Metabolism 1981, 30, 886–893. [Google Scholar] [CrossRef]
- Skamarauskas, J.T.; Oakley, F.; Smith, F.E.; Bawn, C.; Dunn, M.; Vilder, D.S.; Clemence, M.; Blain, P.G.; Taylor, R.; Gamcsik, M.P.; et al. Noninvasive in vivo magnetic resonance measures of glutathione synthesis in human and rat liver as an oxidative stress biomarker. Hepatology 2014, 59, 2321–2330. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Kikuchi, G. Physiological significance of glycine cleavage system in human liver as revealed by the study of a case of hyperglycinemia. Biochem. Biophys. Res. Commun. 1969, 35, 577–583. [Google Scholar] [CrossRef]
- Dasarathy, S.; Kasumov, T.; Edmison, J.M.; Gruca, L.L.; Bennett, C.; Duenas, C.; Marczewski, S.; McCullough, A.J.; Hanson, R.W.; Kalhan, S.C. Glycine and urea kinetics in nonalcoholic steatohepatitis in human: Effect of intralipid infusion. Am. J. Physiol. Gastrointest. Liver Physiol. 2009, 297, G567–G575. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.B. Repression of arginine-glycine transamidinase activity by dietary creatine. Biochim. Biophys. Acta 1959, 36, 574–575. [Google Scholar] [CrossRef]
- McGuire, D.M.; Gross, M.D.; Elde, R.P.; van Pilsum, J.F. Localization of L-arginine-glycine amidinotransferase protein in rat tissues by immunofluorescence microscopy. J. Histochem. Cytochem. 1986, 34, 429–435. [Google Scholar] [CrossRef] [PubMed]
- Pacana, T.; Cazanave, S.; Verdianelli, A.; Patel, V.; Min, H.K.; Mirshahi, F.; Quinlivan, E.; Sanyal, A.J. Dysregulated hepatic methionine metabolism drives homocysteine elevation in diet-induced nonalcoholic fatty liver disease. PLoS ONE 2015, 10, e0136822. [Google Scholar] [CrossRef]
- Hoyles, L.; Fernández-Real, J.M.; Federici, M.; Serino, M.; Abbott, J.; Charpentier, J.; Heymes, C.; Luque, J.L.; Anthony, E.; Barton, R.H.; et al. Molecular phenomics and metagenomics of hepatic steatosis in non-diabetic obese women. Nat. Med. 2018, 24, 1070–1080. [Google Scholar] [CrossRef]
- Boll, M.; Daniel, H.; Gasnier, B. The SLC36 family: Proton-coupled transporters for the absorption of selected amino acids from extracellular and intracellular proteolysis. Pflug. Arch. 2004, 447, 776–779. [Google Scholar] [CrossRef]
- Guastella, J.; Brechna, N.; Weigmann, C.; Lester, H.A.; Davidson, N. Cloning, expression and localization of a rat brain high-affinity glycine transporter. Proc. Natl. Acad. Sci. USA 1992, 89, 7189–7193. [Google Scholar] [CrossRef]
- Liu, Q.R.; López-Corcuera, B.; Mandiyan, S.; Nelson, H.; Nelson, N. Cloning and expression of a spinal cord- and brain-specific glycine transporter with novel structural features. J. Biol. Chem. 1993, 268, 22802–22808. [Google Scholar] [PubMed]
- Zafra, F.; Aragon, C.; Gimenez, C. Molecular biology of glycinergic neurotransmission. Mol. Neurobiol. 1997, 14, 117–142. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.M.; Kingsmore, S.F.; Han, H.; Yang-Feng, T.L.; Godinot, N.; Seldin, M.F.; Caron, M.G.; Giros, B. Cloning of the human glycine transportertype 1: Molecular and pharmacological characterization of novel isoform variants and chromosomal localization of the gene in thehuman and mouse genomes. Mol. Pharmacol. 1994, 45, 608–617. [Google Scholar] [PubMed]
- Bergeron, R.; Meyer, T.M.; Coyle, J.T.; Greene, R.W. Modulation of N-methyl-D-aspartate receptor function by glycine transport. Proc. Natl. Acad. Sci. USA 1998, 95, 15730–15734. [Google Scholar] [CrossRef] [PubMed]
- Yue, J.T.; Abraham, M.A.; Bauer, P.V.; LaPierre, M.P.; Wang, P.; Duca, F.A.; Filippi, B.M.; Chan, O.; Lam, T.K. Inhibition of glycine transporter-1 in the dorsal vagal complex improves metabolic homeostasis in diabetes and obesity. Nat. Commun. 2016, 7, 13501. [Google Scholar] [CrossRef] [PubMed]
- Howard, A.; Tahir, I.; Javed, S.; Waring, S.M.; Ford, D.; Hirst, B.H. Glycine transporter GLYT1 is essential for glycine-mediated protection of human intestinal epithelial cells against oxidative damage. J. Physiol. 2010, 588, 995–1009. [Google Scholar] [CrossRef]
- Aroeira, R.I.; Sebastião, A.M.; Valente, C.A. GlyT1 and GlyT2 in brain astrocytes: Expression, distribution and function. Brain Struct. Funct. 2014, 219, 817–830. [Google Scholar] [CrossRef]
- Xu, T.X.; Gong, N.; Xu, T.L. Inhibitors of GlyT1 and GlyT2 differentially modulate inhibitory transmission. Neuroreport 2005, 16, 1227–1231. [Google Scholar] [CrossRef]
- Bröer, S. The SLC38 family of sodium-amino acid co-transporters. Pflug. Arch. 2014, 466, 155–172. [Google Scholar] [CrossRef]
- Van der Sluis, R.; Badenhorst, C.P.; Erasmus, E.; van Dyk, E.; van der Westhuizen, F.H.; van Dik, A.A. Conservation of the coding regions of the glycine N-acyltransferase gene further suggests that glycine conjugation is an essential detoxification pathway. Gene 2015, 571, 126–134. [Google Scholar] [CrossRef]
- Badenhorst, C.P.S.; Erasmus, E.; van der Sluis, R.; Nortje, C.; van Dijk, A.A. A new perspective on the importance of glycine conjugation in the metabolism of aromatic acids. Drug Metab. Rev. 2014, 46, 343–361. [Google Scholar] [CrossRef] [PubMed]
- Yan-Do, R.; MacDonald, P.E. Impaired “glycine”-mia in type 2 diabetes and potential mechanisms contributing to glucose homeostasis. Endocrinology 2017, 158, 1064–1073. [Google Scholar] [CrossRef] [PubMed]
- Badenhorst, C.P.; van der Sluis, R.; Erasmus, E.; van Dijk, A.A. Glycine conjugation: Importance in metabolism, the role of glycine N-acyltransferase, and factors that influence interindividual variation. Expert Opin. Drug Metab. Toxicol. 2013, 9, 1139–1153. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, M.; Terai, K.; Kameda, N.; Matsumoto, A.; Kurokawa, Y.; Funase, Y.; Nishikawa, K.; Sugaya, N.; Hitua, N.; Kishimoto, T. Designation of enzyme activity of glycine-N-acyltransferase family genes and depression of glycine-N-acyltransferase in human hepatocellular carcinoma. Biochem. Biophys. Res. Commun. 2012, 420, 901–906. [Google Scholar] [CrossRef] [PubMed]
- Guo, C.; Xie, C.; Ding, P.; Qin, G.; Mo, W.; Cao, X.; Zheng, S. Quantification of glycocholic acid in human serum by stable isotope dilution ultra performance liquid chromatography electrospray ionization tandem mass spectrometry. J. Chromatogr. B 2018, 1072, 315–319. [Google Scholar] [CrossRef] [PubMed]
- Falany, C.N.; Johnson, M.R.; Barnes, S.; Diasio, R.B. Glycine and Taurine Conjugation of Bile Acids by a Single Enzyme. Molecular cloning and expression of human liver bile acid CoA: Amino acid N-acyltransferase. J. Biol. Chem. 1994, 269, 19375–19379. [Google Scholar] [PubMed]
- Solaas, K.; Ulvestad, A.; Söreide, O.; Kase, B.F. Subcellular organization of bile acid amidation in human liver: A key issue in regulating the biosynthesis of bile salts. J. Lipid Res. 2000, 41, 1154–1162. [Google Scholar]
- Pellicoro, A.; van den Heuvel, F.A.J.; Geuken, M.; Moshage, H.; Jansen, P.L.M.; Faber, K.N. Human and rat bile acid-CoA:amino acid N -acyltransferase are liver-specific peroxisomal enzymes: Implications for intracellular bile salt transport. Hepatology 2007, 45, 340–348. [Google Scholar] [CrossRef]
- Haeusler, R.A.; Camastra, S.; Nannipieri, M.; Astiarraga, B.; Castro-Perez, J.; Xie, D.; Wang, L.; Chakravarthy, M.; Ferrannini, E. Increased bile acid synthesis and impaired bile acid transport in human obesity. J. Clin. Endocrinol. Metab. 2016, 101, 1935–1944. [Google Scholar] [CrossRef]
- Caspary, W.F. Increase of active transport of conjugated bile salts in streptozotocin-diabetic rat small intestine. Gut 1973, 14, 949–955. [Google Scholar] [CrossRef][Green Version]
- Okekunle, A.P.; Li, Y.; Liu, L.; Du, S.; Wu, X.; Chen, Y.; Li, Y.; Qi, J.; Sun, C.; Feng, R. Abnormal circulating amino acid profiles in multiple metabolic disorders. Diabetes Res. Clin. Pract. 2017, 132, 45–58. [Google Scholar] [CrossRef] [PubMed]
- Perseghin, G.; Ghosh, S.; Gerow, K.; Shulman, G.I. Metabolic defects in lean nondiabetic offspring of NIDDM parents: A cross-sectional study. Diabetes 1997, 46, 1001–1009. [Google Scholar] [CrossRef] [PubMed]
- Tulipani, S.; Griffin, J.; Palau-Rodriguez, M.; Mora-Cubillos, X.; Bernal-Lopez, R.M.; Tinahones, F.J.; Corkey, B.E.; Andres-Lacueva, C. Metabolomics-guided insights on bariatric surgery versus behavioral interventions for weight loss. Obesity (Silver Spring) 2016, 24, 2451–2466. [Google Scholar] [CrossRef] [PubMed]
- Gralka, E.; Luchinat, C.; Tenori, L.; Ernst, B.; Thurnheer, M.; Schultes, B. Metabolomic fingerprint of severe obesity is dynamically affected by bariatric surgery in a procedure-dependent manner. Am. J. Clin. Nutr. 2015, 102, 1313–1322. [Google Scholar] [CrossRef] [PubMed]
- Magkos, F.; Bradley, D.; Schweitzer, G.G.; Finck, B.N.; Eagon, J.C.; Ilkayeva, O.; Newgard, C.B.; Klein, S. Effect of Roux-en-Y gastric bypass and laparoscopic adjustable gastric banding on branched-chain amino acid metabolism. Diabetes 2013, 62, 2757–2761. [Google Scholar] [CrossRef] [PubMed]
- Laferrère, B.; Reilly, D.; Arias, S.; Swerdlow, N.; Gorroochum, P.; Bawa, B.; Bose, M.; Teixeira, J.; Stevens, R.D.; et al. Differential metabolic impact of gastric bypass surgery versus dietary intervention in obese diabetic subjects despite identical weight loss. Sci. Transl. Med. 2011, 3, 80re2. [Google Scholar] [CrossRef] [PubMed]
- Glynn, E.L.; Piner, L.W.; Huffman, K.M.; Slentz, C.A.; Elliot-Penry, L.; AbouAssi, H.; White, P.J.; Bain, J.R.; Muehlbauer, M.J.; Ilkayeva, O.R.; et al. Impact of combined resistance and aerobic exercise training on branched-chain amino acid turnover, glycine metabolism and insulin sensitivity in overweight humans. Diabetologia 2015, 58, 2324–2335. [Google Scholar] [CrossRef]
- Wang-Sattler, R.; Yu, Z.; Herder, C.; Messias, A.C.; Floegel, A.; He, Y.; Heim, K.; Campillos, M.; Holzapfel, C.; Thorand, B.; et al. Novel biomarkers for pre-diabetes identified by metabolomics. Mol. Syst. Biol. 2012, 8, 615. [Google Scholar] [CrossRef]
- Floegel, A.; Stefan, N.; Yu, Z.; Mühlenbruch, K.; Drogan, D.; Joost, H.G.; Fritsche, A.; Häring, H.U.; Hrabe de Angelis, M.; Peters, A.; et al. Identification of serum metabolites associated with risk of type 2 diabetes using a targeted metabolomic approach. Diabetes 2013, 62, 639–648. [Google Scholar] [CrossRef]
- Palmer, N.D.; Stevens, R.D.; Antinozzi, P.A.; Anderson, A.; Bergman, R.N.; Wagenknecht, L.E.; Newgard, C.B.; Bowden, D.W. Metabolomic profile associated with insulin resistance and conversion to diabetes in the Insulin Resistance Atherosclerosis Study. J. Clin. Endocrinol. Metab. 2015, 100, E463–E468. [Google Scholar] [CrossRef]
- Lever, M.; Slow, S.; McGregor, D.O.; Dellow, W.J.; George, P.M.; Chambers, S.T. Variability of plasma and urine betaine in diabetes mellitus and its relationship to methionine load test responses: An observational study. Cardiovasc. Diabetol. 2012, 11, 34. [Google Scholar] [CrossRef] [PubMed]
- Lever, M.; George, P.M.; Slow, S.; Bellamy, D.; Young, J.M.; Ho, M.; McEntyre, C.J.; Emslie, J.L.; Atkinson, W.; Molyneux, S.L.; et al. Betaine and trimethylamine-N-oxide as predictors of cardiovascular outcomes show different patterns in diabetes mellitus: An observational study. PLoS ONE 2014, 9, e114969. [Google Scholar] [CrossRef] [PubMed]
- Magnusson, M.; Wang, T.J.; Clish, C.; Engström, G.; Nilsson, P.; Gerszten, R.E.; Melander, O. Dimethylglycine deficiency and the development of diabetes mellitus. Diabetes 2015, 64, 3010–3016. [Google Scholar] [CrossRef] [PubMed]
- Lever, M.; Slow, S. The clinical significance of betaine, an osmolyte with a key role in methyl group metabolism. Clin. Biochem. 2010, 43, 732–744. [Google Scholar] [CrossRef] [PubMed]
- Yan, J.; Winter, L.B.; Burns-Whitmore, B.; Vermeylen, F.; Caudill, M.A. Plasma choline metabolites associate with metabolic stress among young overweight men in a genotype-specific manner. Nutr. Diabetes. 2012, 2, e49. [Google Scholar] [CrossRef] [PubMed]
- Ejaz, A.; Martinez-Guino, L.; Goldfine, A.B.; Ribas-Aulinas, F.; De Nigrus, V.; Ribo, S.; Gonzalez-Franquesa, A.; Garcia-Roves, P.M.; Li, E.; Dreyfuss, J.M.; et al. Dietary Betaine Supplementation Increases Fgf21 Levels to Improve Glucose Homeostasis and Reduce Hepatic Lipid Accumulation in Mice. Diabetes 2016, 65, 902–912. [Google Scholar] [CrossRef] [PubMed]
- Walford, G.A.; Ma, Y.; Clish, C.; Florez, J.C.; Wang, T.J.; Gerszten, R.E.; Diabetes Prevention Program Research Group. Metabolite Profiles of Diabetes Incidence and Intervention Response in the Diabetes Prevention Program. Diabetes 2016, 65, 1424–1433. [Google Scholar] [CrossRef] [PubMed]
- Svingen, G.F.; Schartum-Hansen, H.; Pedersen, E.R.; Ueland, P.M.; Tell, G.S.; Mellgren, G.; Njolstad, P.R.; Seifert, R.; Strand, E.; Karlsson, T.; et al. Prospective Associations of Systemic and Urinary Choline Metabolites with Incident Type 2 Diabetes. Clin. Chem. 2016, 62, 755–765. [Google Scholar] [CrossRef]
- Stechemesser, L.; Eder, S.K.; Wagner, A.; Patsch, W.; Feldman, A.; Strasser, M.; Auer, S.; Niederseer, D.; Huber-Schönauer, U.; Paulweber, B.; et al. Metabolomic profiling identifies potential pathways involved in the interaction of iron homeostasis with glucose metabolism. Mol. Metab. 2016, 6, 38–47. [Google Scholar] [CrossRef]
- Schartum-Hansen, H.; Ueland, P.M.; Pedersen, E.R.; Meyer, K.; Ebbing, M.; Bleie, O.; Svingen, G.F.; Seifert, R.; Nygard, O. Assessment of urinary betaine as a marker of diabetes mellitus in cardiovascular patients. PLoS ONE 2013, 8, e69454. [Google Scholar] [CrossRef]
- Mirmiran, P.; Bahadoran, Z.; Esfandyari, S.; Azizi, F. Dietary Protein and Amino Acid Profiles in Relation to Risk of Dysglycemia: Findings from a Prospective Population-Based Study. Nutrients 2017, 9, 971. [Google Scholar] [CrossRef] [PubMed]
- Altorf-van der Kuil, W.; Brink, E.J.; Boetje, M.; Seibelink, E.; Bijlsma, S.; Engerink, M.F.; van’t Veer, P.; Tomé, D.; Bakker, S.J.; van Baak, M.A.; et al. Identification of biomarkers for intake of protein from meat, dairy products and grains: A controlled dietary intervention study. Br. J. Nutr. 2013, 110, 810–822. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, A.; Gibney, M.J.; Brennan, L. Dietary intake patterns are reflected in metabolomic profiles: Potential role in dietary assessment studies. Am. J. Clin. Nutr. 2011, 93, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Wittenbecher, C.; Mühlenbruch, K.; Kröger, J.; Jacobs, S.; Kuxhaus, O.; Floegel, A.; Fritsche, A.; Pischon, T.; Prehn, C.; Adanski, J.; et al. Amino acids, lipid metabolites, and ferritin as potential mediators linking red meat consumption to type 2 diabetes. Am. J. Clin. Nutr. 2015, 101, 1241–1250. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Wang, Y.; Sun, G. High dietary choline and betaine intake is associated with low insulin resistance in the Newfoundland population. Nutrition 2017, 33, 28–34. [Google Scholar] [CrossRef]
- Beaumont, M.; Portune, K.J.; Steuer, N.; Lan, A.; Cerrudo, V.; Audebert, M.; Dumont, F.; Mancano, G.; Khodorova, N.; Andriamihaja, M.; et al. Quantity and source of dietary protein influence metabolite production by gut microbiota and rectal mucosa gene expression: A randomized, parallel, double-blind trial in overweight humans. Am. J. Clin. Nutr. 2017, 106, 1005–1019. [Google Scholar] [CrossRef] [PubMed]
- Dai, Z.L.; Li, X.L.; Xi, P.B.; Zhang, J.; Wu, G.; Zhu, W.Y. Metabolism of select amino acids in bacteria from the pig small intestine. Amino Acids 2012, 42, 1597–1608. [Google Scholar] [CrossRef]
- Dai, Z.L.; Zhang, J.; Wu, G.; Zhu, W.Y. Utilization of amino acids by bacteria from the pig small intestine. Amino Acids 2010, 39, 1201–1215. [Google Scholar] [CrossRef]
- Dai, Z.L.; Wu, G.; Zhu, W.Y. Amino acid metabolism in intestinal bacteria: Links between gut ecology and host health. Front. Biosci. (Landmark Ed.) 2011, 16, 1768–1786. [Google Scholar] [CrossRef]
- Forslund, K.; Hildebrand, F.; Nielsen, T.; Falony, G.; Le Chatelier, E.; Sunagawa, S.; Prifti, E.; Vieira-Silva, S.; Gudmundsdottir, V.; Pedersen, H.K.; et al. Disentangling type 2 diabetes and metformin treatment signatures in the human gut microbiota. Nature 2015, 528, 262–266. [Google Scholar] [CrossRef]
- Metges, C.C. Contribution of Microbial Amino Acids to Amino Acid Homeostasis of the Host. J. Nutr. 2000, 130, 1857S–1864S. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.; Nyman, M. Prebiotic and synbiotic effects on rats fed malted barley with selected bacteria strains. Food Nutr. Res. 2014, 58. [Google Scholar] [CrossRef] [PubMed]
- Ndagijimana, M.; Laghi, L.; Vitali, B.; Placucci, G.; Brigidi, P.; Guerzoni, E. Effect of a synbiotic food consumption on human gut metabolic profiles evaluated by 1H Nuclear Magnetic Resonance spectroscopy. Int. J. Food Microbiol. 2009, 134, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.P.; Wang, Y.; Sprenger, N.; Holmes, E.; Lindon, J.C.; Kochhar, S.; Nicholson, J.K. Effects of probiotic Lactobacillus paracasei treatment on the host gut tissue metabolic profiles probed via magic-angle-spinning NMR spectroscopy. J. Proteome Res. 2007, 6, 1471–1481. [Google Scholar] [CrossRef] [PubMed]
- Ma, N.; Tian, Y.; Wu, Y.; Ma, X. Contributions of the Interaction Between Dietary Protein and Gut Microbiota to Intestinal Health. Curr. Protein Pept. Sci. 2017, 18, 795–808. [Google Scholar] [CrossRef] [PubMed]
- Beaumont, M.; Jaoui, D.; Douard, V.; Mat, D.; Koeth, F.; Goustard, B.; Mayeur, C.; Mondot, S.; Hovaghimian, A.; Le Feunteun, S.; et al. Structure of protein emulsion in food impacts intestinal microbiota, caecal luminal content composition and distal intestine characteristics in rats. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Neis, E.P.J.G.; Dejong, C.H.C.; Rensen, S.S. The Role of Microbial Amino Acid Metabolism in Host Metabolism. Nutrients 2015, 7, 2930–2946. [Google Scholar] [CrossRef]
- Sommer, F.; Nookaew, I.; Sommer, N.; Fogelstrand, P.; Bäckhed, F. Site-specific programming of the host epithelial transcriptome by the gut microbiota. Genome Biol. 2015, 16, 62. [Google Scholar] [CrossRef]
- Mardinoglu, A.; Shoaie, S.; Bergentall, M.; Ghaffari, P.; Zhang, C.; Larsson, E.; Bäckhed, F.; Nielsen, J. The gut microbiota modulates host amino acid and glutathione metabolism in mice. Mol. Syst. Biol. 2015, 11, 834. [Google Scholar] [CrossRef]
- Jois, M.; Hall, B.; Fewer, K.; Brosnan, J.T. Regulation of hepatic glycine catabolism by glucagon. J. Biol. Chem. 1989, 264, 3347–3351. [Google Scholar]
- Boden, G.; Rezvani, I.; Owen, O.E. Effects of glucagon on plasma amino acids. J. Clin. Investig. 1984, 73, 785–793. [Google Scholar] [CrossRef] [PubMed]
- Gosmanov, N.R.; Gosmanov, A.R.; Gerich, J.E. Glucagon Physiology. In Endotext [Internet]; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., Dungan, K., Grossman, A., Hershman, J.M., Kaltsas, G., Koch, C., Kopp, P., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- White, P.J.; Lapworth, A.L.; An, J.; Wang, L.; McGarrah, R.W.; Stevens, R.D.; Ilkayeva, O.; George, T.; Muehlbauer, M.J.; Bain, J.R.; et al. Branched-chain amino acid restriction in Zucker-fatty rats improves muscle insulin sensitivity by enhancing efficiency of fatty acid oxidation and acyl-glycine export. Mol. Metab. 2016, 5, 538–551. [Google Scholar] [CrossRef] [PubMed]
- She, P.; Van Horn, C.; Reid, T.; Hutson, S.M.; Cooney, R.N.; Lynch, C.J. Obesity-related elevations in plasma leucine are associated with alterations in enzymes involved in branched-chain amino acid metabolism. Am. J. Physiol. Endocrinol. Metab. 2007, 293, E1552–E1563. [Google Scholar] [CrossRef] [PubMed]
- Lian, K.; Du, C.; Liu, Y.; Zhy, D.; Yan, W.; Zhang, H.; Hong, Z.; Liu, P.; Zhang, L.; Pei, H.; et al. Impaired adiponectin signaling contributes to disturbed catabolism of branched-chain amino acids in diabetic mice. Diabetes 2015, 64, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Lever, M.; Sizeland, P.C.; Bason, L.M.; Hayman, C.M.; Robson, R.A.; Chambers, S.T. Abnormal glycine betaine content of the blood and urine of diabetic and renal patients. Clin. Chim. Acta 1994, 230, 69–79. [Google Scholar] [CrossRef]
- Csernus, K.; Lanyi, E.; Erhardt, E.; Molnar, D. Effect of childhood obesity and obesity-related cardiovascular risk factors on glomerular and tubular protein excretion. Eur. J. Pediatr. 2005, 164, 44–49. [Google Scholar] [CrossRef] [PubMed]
- Xie, W.; Wood, A.R.; Lyssenko, V.; Weedon, M.N.; Knowles, J.W.; Alkayyali, S.; Assimes, T.L.; Questermous, T.; Abbasi, F.; Paananen, J.; et al. Genetic variants associated with glycine metabolism and their role in insulin sensitivity and type 2 diabetes. Diabetes 2013, 62, 2141–2150. [Google Scholar] [CrossRef]
- Matone, A.; Scott-Boyer, M.P.; Carayol, J.; Fazelzadeh, P.; Fazelzadeh, P.; Lefebvre, G.; Valsesia, A.; Charon, C.; Vervoort, J.; Astrup, A.; et al. Network Analysis of Metabolite GWAS Hits: Implication of CPS1 and the Urea Cycle in Weight Maintenance. PLoS ONE 2016, 11, e0150495. [Google Scholar] [CrossRef]
- Jia, Q.; Han, Y.; Huang, P.; Woodward, N.C.; Gukasyan, J.; Kettunen, J.; Ala-Korpela, M.; Anufrieva, O.; Wang, Q.; Perola, M.; et al. Genetic Determinants of Circulating Glycine Levels and Risk of Coronary Artery Disease. J. Am. Heart Assoc. 2019, 8, e011922. [Google Scholar] [CrossRef]
- Gannon, M.C.; Nuttall, J.A.; Nuttall, F.Q. The metabolic response to ingested glycine. Am. J. Clin. Nutr. 2002, 76, 1302–1307. [Google Scholar] [CrossRef]
- González-Ortiz, M.; Medina-Santillán, R.; Martínez-Abundis, E.; von Drateln, C.R. Effect of glycine on insulin secretion and action in healthy first-degree relatives of type 2 diabetes mellitus patients. Horm. Metab. Res. 2001, 33, 358–360. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, D.; Hsu, J.W.; Jahoor, F.; Sekhar, R.V. Effect of increasing glutathione with cysteine and glycine supplementation on mitochondrial fuel oxidation, insulin sensitivity, and body composition in older HIV-infected patients. J. Clin. Endocrinol. Metab. 2014, 99, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Flores, M.; Cruz, M.; Duran-Reyes, G.; Munquia-Miranda, C.; Loza-Rodriguez, H.; Pulido-Casas, E.; Torres-Ramirez, N.; Gaja-Rodriguez, O.; Kumate, J.; Baiza-Gutman, L.A.; et al. Oral supplementation with glycine reduces oxidative stress in patients with metabolic syndrome, improving their systolic blood pressure. Can. J. Physiol. Pharmacol. 2013, 91, 855–860. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Shen, L.; Tan, Z.; Zhang, P.; Zhao, X.; Xu, Y.; Gan, M.; Yang, Q.; Ma, J.; Jiang, A.; et al. Betaine Supplementation Enhances Lipid Metabolism and Improves Insulin Resistance in Mice Fed a High-Fat Diet. Nutrients 2018, 10, 131. [Google Scholar] [CrossRef] [PubMed]
- Grizales, A.M.; Patti, M.E.; Lin, A.P.; Beckman, J.A.; Sahni, V.A.; Cloutier, E.; Fowler, K.M.; Dreyfuss, J.M.; Pan, H.; Kozuka, C.; et al. Metabolic Effects of Betaine: A Randomized Clinical Trial of Betaine Supplementation in Prediabetes. J. Clin. Endocrinol. Metab. 2018, 103, 3038–3049. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Han, D.; Xu, R.; Wu, H.; Qu, C.; Wang, F.; Wang, X.; Zhao, Y. Glycine protects against high sucrose and high fat-induced non-alcoholic steatohepatitis in rats. Oncotarget 2016, 7, 80223–80237. [Google Scholar] [CrossRef] [PubMed]
- Takashima, S.; Ikejima, K.; Arai, K.; Yokokawa, J.; Kon, K.; Yamashina, S.; Watanabe, S. Glycine prevents metabolic steatohepatitis in diabetic KK-Ay mice through modulation of hepatic innate immunity. Am. J. Physiol. Gastrointest. Liver Physiol. 2016, 311, G1105–G1113. [Google Scholar] [CrossRef] [PubMed]
- McCarty, M.F.; O’Keefe, J.H.; DiNicolantonio, J.J. Dietary Glycine Is Rate-Limiting for Glutathione Synthesis and May Have Broad Potential for Health Protection. Ochsner J. 2018, 18, 81–87. [Google Scholar] [PubMed]
- Rani, V.; Deep, G.; Singh, R.K.; Palle, K.; Yadav, U.C. Oxidative stress and metabolic disorders: Pathogenesis and therapeutic strategies. Life Sci. 2016, 148, 183–193. [Google Scholar] [CrossRef]
- Lutchmansingh, F.K.; Hsu, J.W.; Bennett, F.I.; Bardaloo, A.V.; McFarlane-Anderson, N.; Gordon-Strachan, G.M.; Wright-Pascoe, R.A.; Jahoor, F.; Boyne, M.S. Glutathione metabolism in type 2 diabetes and its relationship with microvascular complications and glycemia. PLoS ONE 2018, 13, e0198626. [Google Scholar] [CrossRef]
- Furfaro, A.L.; Nitti, M.; Marengo, B.; Domenicotti, C.; Cottalasso, D.; Marinari, U.M.; Pronzato, M.A.; Traverso, N. Impaired synthesis contributes to diabetes-induced decrease in liver glutathione. Int. J. Mol. Med. 2012, 29, 899–905. [Google Scholar] [PubMed]
- Sekhar, R.V.; McKay, S.V.; Patel, S.G.; Guthikonda, A.P.; Reddy, V.T.; Balasubramanyam, A.; Jahoor, F. Glutathione synthesis is diminished in patients with uncontrolled diabetes and restored by dietary supplementation with cysteine and glycine. Diabetes Care 2011, 34, 162–167. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhang, J.; Li, C.; Wang, Z.; Li, J.; Zhao, D.; Wang, S.; Zhang, H.; Huang, Y.; Guo, X. Glycine Transporter-1 and glycine receptor mediate the antioxidant effect of glycine in diabetic rat islets and INS-1 cells. Free Radic. Biol. Med. 2018, 123, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Bonkovsky, H.L.; Guo, J.T.; Hou, W.; Li, T.; Narang, T.; Thapar, M. Porphyrin and heme metabolism and the porphyrias. Compr. Physiol. 2013, 3, 365–401. [Google Scholar] [PubMed]
- Garcia-Santos, D.; Schranzhofer, M.; Bergeron, R.; Sheftel, A.D.; Ponka, P. Extracellular glycine is necessary for optimal hemoglobinization of erythroid cells. Haematologica 2017, 102, 1314–1323. [Google Scholar] [CrossRef] [PubMed]
- Converso, D.P.; Taillé, C.; Carreras, M.C.; Jaitovich, A.; Poderoso, J.J.; Boczkowski, J. HO-1 is located in liver mitochondria and modulates mitochondrial heme content and metabolism. FASEB J. 2006, 20, 1236–1238. [Google Scholar] [CrossRef] [PubMed]
- Ridaura, V.K.; Faith, J.J.; Rey, F.E.; Cheng, J.; Duncan, A.E.; Kau, A.L.; Griffin, N.W.; Lombard, V.; Henrissat, B.; Bain, J.R.; et al. Gut microbiota from twins discordant for obesity modulate metabolism in mice. Science 2013, 341, 1241214. [Google Scholar] [CrossRef]
- Koves, T.R.; Ussher, J.R.; Noland, R.C.; Slentz, D.; Mosedale, M.; Ilkayeva, O.; Bain, J.; Stevens, R.; Dyck, J.R.; Newgard, C.B.; et al. Mitochondrial overload and incomplete fatty acid oxidation contribute to skeletal muscle insulin resistance. Cell Metab. 2008, 7, 45–56. [Google Scholar] [CrossRef]
- Pelantová, H.; Bugáňová, M.; Holubová, M.; Sediva, B.; Zemenova, J.; Sykora, D.; Kavalkova, P.; Haluzik, M.; Zelezna, B.; Maletinska, L.; et al. Urinary metabolomic profiling in mice with diet-induced obesity and type 2 diabetes mellitus after treatment with metformin, vildagliptin and their combination. Mol. Cell. Endocrinol. 2016, 431, 88–100. [Google Scholar] [CrossRef]
- Mays, L.L.; Borek, E.; Finch, C.E. Glycine N-methyltransferase is a regulatory enzyme which increases in ageing animals. Nature 1973, 243, 411–413. [Google Scholar] [CrossRef]
- Wang, Y.C.; Chen, Y.M.; Lin, Y.J.; Liu, S.P.; Chiang, E.P. GNMT expression increases hepatic folate contents and folate-dependent methionine synthase-mediated homocysteine remethylation. Mol. Med. 2011, 17, 486–494. [Google Scholar] [CrossRef] [PubMed]
- Wagner, C.; Briggs, W.T.; Cook, R.J. Inhibition of glycine N-methyltransferase activity by folate derivatives: Implications for regulation of methyl group metabolism. Biochem. Biophys. Res. Commun. 1985, 127, 746–752. [Google Scholar] [CrossRef]
- Tseng, T.L.; Shih, Y.P.; Huang, Y.C.; Wang, C.K.; Chen, P.H.; Chang, J.G.; Yeh, K.T.; Chen, Y.M.; Buetow, K.H. Genotypic and phenotypic characterization of a putative tumor susceptibility gene, GNMT, in liver cancer. Cancer Res. 2003, 63, 647–654. [Google Scholar] [PubMed]
- Borowa-Mazgaj, B.; de Conti, A.; Tryndyak, V.; Steward, C.R.; Jimenez, L.; Melnyk, S.; Seneshaw, M.; Mirshahi, F.; Rusyn, I.; Beland, F.A.; et al. Gene expression and DNA methylation alterations in the glycine N-methyltransferase gene in diet-induced nonalcoholic fatty liver disease-associated carcinogenesis. Toxicol. Sci. 2019, kfz110. [Google Scholar] [CrossRef] [PubMed]
- Huidobro, C.; Toraño, E.G.; Fernández, A.F.; Urdinguio, R.G.; Rodríguez, R.M.; Ferrero, C.; Martínez-Camblor, P.; Boix, L.; Bruix, J.; García-Rodríguez, J.L.; et al. A DNA methylation signature associated with the epigenetic repression of glycine N-methyltransferase in human hepatocellular carcinoma. J. Mol. Med. (Berl.) 2013, 91, 939–950. [Google Scholar] [CrossRef] [PubMed]
- Luka, Z.; Capdevila, A.; Mato, J.M.; Wagner, C. A glycine N-methyltransferase knockout mouse model for humans with deficiency of this enzyme. Transgenic Res. 2006, 15, 393–397. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.J.; Liu, S.P.; Lee, C.M.; Yen, C.H.; Chuang, P.C.; Chen, C.Y.; Tsai, T.F.; Huang, S.F.; Lee, Y.H.; Chen, Y.M. Characterization of a glycine N-methyltransferase gene knockout mouse model for hepatocellular carcinoma: Implications of the gender disparity in liver cancer susceptibility. Int. J. Cancer 2009, 124, 816–826. [Google Scholar] [CrossRef]
- Liao, Y.J.; Lee, T.S.; Twu, Y.C.; Hsu, S.M.; Yang, C.P.; Wang, C.K.; Liang, Y.C.; Chen, Y.A. Glycine N-methyltransferase deficiency in female mice impairs insulin signaling and promotes gluconeogenesis by modulating the PI3K/Akt pathway in the liver. J. Biomed. Sci. 2016, 23, 69. [Google Scholar] [CrossRef]
- Hughey, C.C.; Trefts, E.; Bracy, D.P.; James, F.D.; Donahue, E.P.; Wasserman, D.H. Glycine N-methyltransferase deletion in mice diverts carbon flux from gluconeogenesis to pathways that utilize excess methionine cycle intermediates. J. Biol. Chem. 2018, 293, 11944–11954. [Google Scholar] [CrossRef]
- Young, A.B.; Snyder, S.H. Strychnine binding associated with glycine receptors of the central nervous system. Proc. Natl. Acad. Sci. USA 1973, 70, 2832–2836. [Google Scholar] [CrossRef]
- Johnson, J.W.; Ascher, P. Glycine potentiates the NMDA response in cultured mouse brain neurons. Nature 1987, 325, 529–531. [Google Scholar] [CrossRef]
- Lam, C.K.; Chari, M.; Su, B.B.; Cheung, G.W.; Kokorovic, A.; Yang, C.S.; Wang, P.Y.; Lai, T.Y.; Lam, T.K. Activation of N-methyl-D-aspartate (NMDA) receptors in the dorsal vagal complex lowers glucose production. J. Biol. Chem. 2010, 285, 21913–21921. [Google Scholar] [CrossRef] [PubMed]
- Sorrels, T.L.; Bostock, E. Induction of feeding by 7-chlorokynurenic acid, a strychnine-insensitive glycine binding site antagonist. Brain Res. 1992, 572, 265–268. [Google Scholar] [CrossRef]
- Manousopoulou, A.; Koutmani, Y.; Karaliota, S.; Woelk, C.H.; Manolakos, E.S.; Karalis, K.; Garbis, S.D. Hypothalamus proteomics from mouse models with obesity and anorexia reveals therapeutic targets of appetite regulation. Nutr. Diabetes 2016, 6, e204. [Google Scholar] [CrossRef] [PubMed]
- Gameiro, A.; Reimann, F.; Habib, A.M.; O’Malley, D.; Williams, L.; Simpson, A.K.; Gribble, F.M. The neurotransmitters glycine and GABA stimulate glucagon-like peptide-1 release from the GLUTag cell line. J. Physiol. 2005, 569, 761–772. [Google Scholar] [CrossRef] [PubMed]
- Yan-Do, R.; Duong, E.; Manning Fox, J.E.; Dai, X.; Suzuki, K.; Khan, S.; Bautista, A.; Ferdaoussi, M.; Lyon, J.; Wu, X.; et al. A Glycine-Insulin Autocrine Feedback Loop Enhances Insulin Secretion from Human β-Cells and Is Impaired in Type 2 Diabetes. Diabetes 2016, 65, 2311–2321. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, M.D.; Ikejema, K.; Enomoto, N.; Stacklewitz, R.F.; Seabra, V.; Zhong, Z.; Yin, M.; Schemmer, P.; Rose, M.L.; Rusyn, I.; et al. Glycine: A new anti-inflammatory immunonutrient. Cell. Mol. Life Sci. 1999, 56, 843–856. [Google Scholar] [CrossRef] [PubMed]
- Jain, M.; Nilsson, R.; Sharma, S.; Madhusudhan, N.; Kitami, T.; Souza, A.L.; Kafri, R.; Kirschner, M.W.; Clish, C.B.; Mootha, V.K. Metabolite profiling identifies a key role for glycine in rapid cancer cell proliferation. Science 2012, 336, 1040–1044. [Google Scholar] [CrossRef]
- Pirola, L.; Ciesielski, O.; Balcerczyk, A. The Methylation Status of the Epigenome: Its Emerging Role in the Regulation of Tumor Angiogenesis and Tumor Growth, and Potential for Drug Targeting. Cancers 2018, 10, 268. [Google Scholar] [CrossRef]
| Study Group | Health Status | Glycine Concentration (µmol/L) | Level of Significance | Reference | |
|---|---|---|---|---|---|
| Control Group | Study Group | ||||
| 20 control subjects, 15 subjects with obesity with NAFLD | Controls vs. obesity with NAFLD | Mean: 205.9 ± 9.7 | Mean obesity with NAFLD: 179.2 ± 7.6 | p = 0.03 | [9] |
| The observational, prospective cohort PPSDiab: 151 women with gestational diabetes or normoglycemia during pregnancy | NGT vs. PGT | Median NGT: 272.6 | Median PGT: 224.6 | p < 0.01 | [14] |
| 399 nondiabetic adults | IS vs. IR | NA | 0.85 fold vs. controls | p = 2.79 × 10−11 | [15] |
| 124 adults (63 European American and 60 African American) | IS vs. IR and T2DM | Mean IS: 306.8 | Mean IR: 257.0 | p < 0.01 for the two comparisons vs. IS | [16] |
| Mean T2DM: 246.8 | |||||
| [Glycine concentration was correlated to GDR in a hyperinsulinemic-euglycemic clamp] | |||||
| 64 adults | sex-matched groups for BMI [lean vs. morbid obesity] and risk of developing T2DM [IS vs. IR] | Glycine concentration is negatively associated to fasting insulin and HOMA-IR | R = −0.51, p = 0.0017; and R = −0.49, p = 0.0033 | [17] | |
| Framingham Heart Study (n = 1015) and the Malmö Diet and Cancer Study (n = 746) | 45% of individuals meeting the criteria for metabolic syndrome | Mean NGT = 270 | Mean PGT = 220 | p = 0.0005 | [18] |
| 73 control subjects, 10 subjects with obesity | Controls vs. obesity | Mean: 223.7 ± 33.0 | Mean: 197.9 ± 41.4 | p = 0.027 | [19] |
| 51 healthy control subjects; 31 overweight or obese subjects; 52 subjects with T2DM | Controls vs. obesity and T2DM Men and women were analyzed separately | Mean men: 211 ± 30 | Mean men with obesity: 186 ± 30 | p < 0.05 for all comparisons vs. controls | [20] |
| Mean men with T2DM: 187 ± 44 | |||||
| Mean women: 231 ± 67 | Mean women with obesity: 203 ± 48 | ||||
| Mean women with T2DM: 184 ± 48 | |||||
| Population | Health Status | Dose and Duration | Health Impacts of Glycine Supplementation | Reference |
|---|---|---|---|---|
| Glycine dietary supplementation | ||||
| Clinical studies | ||||
| Adult humans: | Healthy patients | Single oral morning dose of 5 g glycine +/− 25 g glucose vs. water +/− 25 g glucose | Improves insulin response and glucose tolerance in response to glucose ingestion | [122] |
| 4 Women | ||||
| 5 Men | ||||
| Age: 21 to 52 y | ||||
| Adult humans: | Healthy lean patients with first degree relatives of T2DM | Single oral morning dose of 5 g glycine vs. magnesium oxide (placebo) | Improves insulin response, measured during an euglycemic-hyperinsulinemic clamp; No significant alteration in insulin action | [123] |
| 8 Women | ||||
| 4 Men | ||||
| Age: 23.7 ± 4.1 y | ||||
| Adult humans: | Patients with MetS (NCEP/ATP III criteria) | 15 g glycine/day (3 times 5 g/d) dissolved in water vs. starch (placebo) for 3 months | Improves systolic blood pressure in men; Protects against oxidative damages determined from antioxidant enzymes activity in erythrocytes and leukocytes, and thiobarbituric acid reactive substances (TBARS) in plasma | [125] |
| 29 Women | ||||
| 23 Men | ||||
| Age: 35 to 65 y | ||||
| Elderly patients: | Patients with HIV | 1.33 mmol glycine/kg/day with 0.81 mmol/kg/day N-acetylcysteine for 14 days | Improves insulin sensitivity, measured by hyperinsulinemic-euglycemic clamp before and after supplementation | [124] |
| 9 Men | ||||
| Age: 56.1 ± 1.0 y | ||||
| Preclinical studies | ||||
| Male Sprague Dawley rats: n = 48 | High fat/high sucrose feeding vs. standard chow for 24 weeks | 3.5 g glycine/kg/day in water vs. water (placebo) for 24 weeks | Improves hepatic steatosis assessed histologically | [128] |
| Age: NA | ||||
| Male KK-Ay mice: n = 5/group | Animal model of obesity and T2DM | Semisynthetic diet containing 5% glycine vs. casein (placebo) for 4 weeks | Improves hepatic steatosis assessed histologically Improves glucose tolerance measured during a glucose tolerance test | [129] |
| Age: 7 weeks | ||||
| Betaine dietary supplementation | ||||
| Clinical studies | ||||
| Adult humans: | patients with obesity and pre-diabetes | 3.30 g betaine, twice daily for 10 days, followed by 4.95 g twice daily for 12 weeks vs. microcrystalline cellulose (placebo) | No major effects on glucose homeostasis (euglycemic hyperinsulinemic clamp) and liver fat deposition | [127] |
| 8 Women | ||||
| 20 Men | ||||
| Age: 21 to 70 y | ||||
| Preclinical studies | ||||
| Female | High-fat feeding for 13 weeks | 1% weight/volume betaine, in water vs. water for 1 week | Improves insulin resistance and glucose homeostasis measured using glucose/insulin tolerance tests | [126] |
| Kunming | ||||
| Mice: n = 40 | ||||
| Age: 6 weeks | ||||
| Male C57Bl6 mice: n = 24 | High-fat feeding for 16 weeks | 1% weight/volume betaine, in water vs. water for 1 week | Improves insulin resistance and glucose homeostasis measured using glucose/insulin tolerance test and euglycemic hyperinsulinemic clamp; Reduces liver fat deposition quantified on chloroform-methanol extracts | [87] |
| Age: NA | ||||
| Male C57BL6/N mice: n = 46 | High-fat feeding for 12 weeks, methyl-donor supplementation was given during the last 4 weeks | 15 g/kg betaine, 15 g/kg choline chloride, 7.5 g/kg methionine, 15 mg/kg folic acid, 1.5 mg/kg vitamin B12, 150 mg/kg ZnSO4 | Prevented the progression of hepatic steatosis Increases phosphorylation of AMPK-α together with enhanced β-HAD activity, suggesting increased fatty acid oxidation | [34] |
| Age: 8 weeks | ||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alves, A.; Bassot, A.; Bulteau, A.-L.; Pirola, L.; Morio, B. Glycine Metabolism and Its Alterations in Obesity and Metabolic Diseases. Nutrients 2019, 11, 1356. https://doi.org/10.3390/nu11061356
Alves A, Bassot A, Bulteau A-L, Pirola L, Morio B. Glycine Metabolism and Its Alterations in Obesity and Metabolic Diseases. Nutrients. 2019; 11(6):1356. https://doi.org/10.3390/nu11061356
Chicago/Turabian StyleAlves, Anaïs, Arthur Bassot, Anne-Laure Bulteau, Luciano Pirola, and Béatrice Morio. 2019. "Glycine Metabolism and Its Alterations in Obesity and Metabolic Diseases" Nutrients 11, no. 6: 1356. https://doi.org/10.3390/nu11061356
APA StyleAlves, A., Bassot, A., Bulteau, A.-L., Pirola, L., & Morio, B. (2019). Glycine Metabolism and Its Alterations in Obesity and Metabolic Diseases. Nutrients, 11(6), 1356. https://doi.org/10.3390/nu11061356





