Plasma Trimethylamine-N-oxide following Cessation of L-carnitine Supplementation in Healthy Aged Women
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects
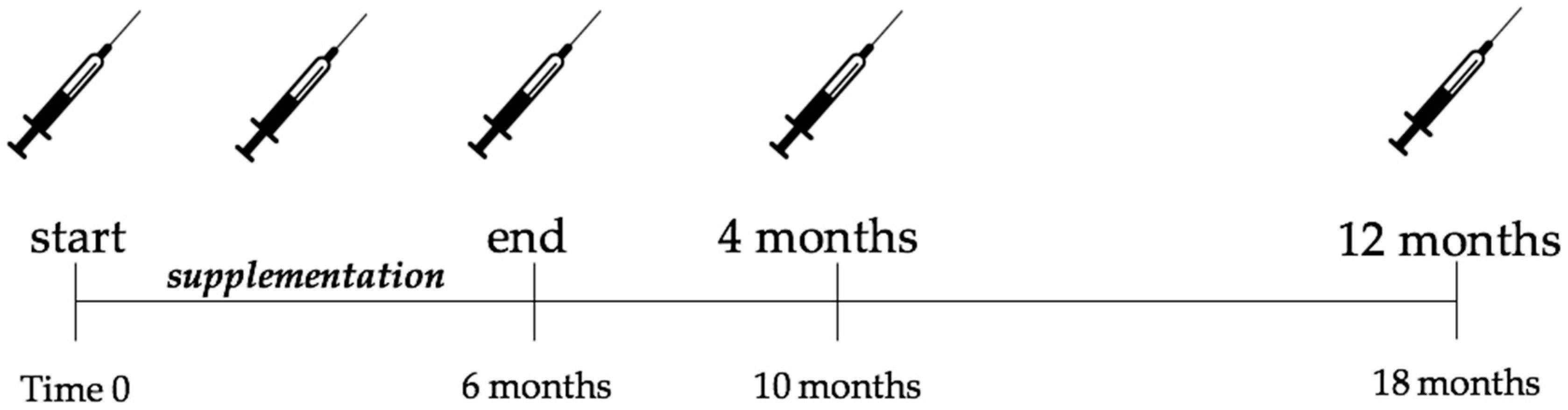
2.2. Study Procedure
2.3. Fish Consumption Habits
2.4. Blood Sampling
2.5. Biochemical Determination
2.6. Statistical Analyses
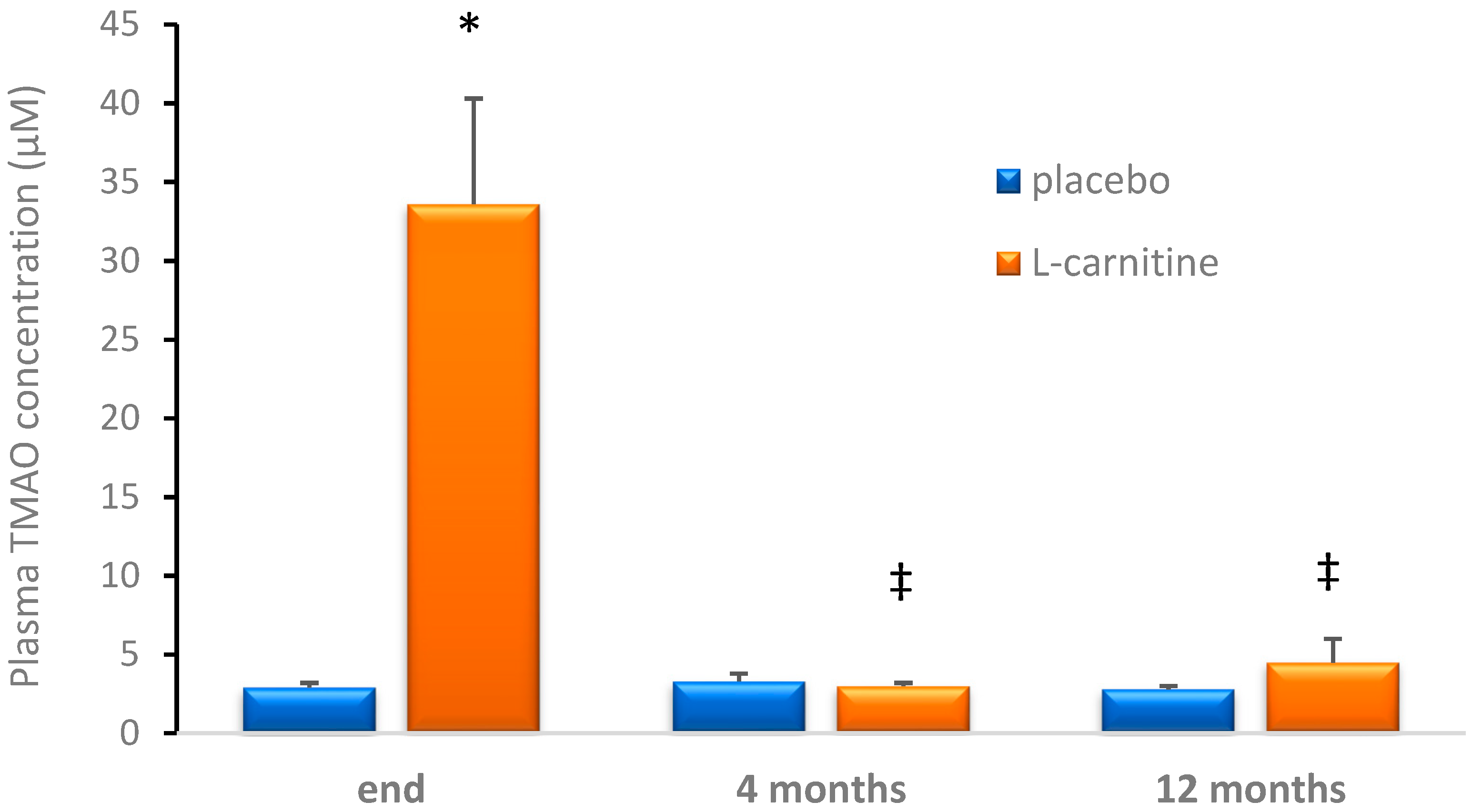
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Herrington, W.; Lacey, B.; Sherliker, P.; Armitage, J.; Lewington, S. Epidemiology of Atherosclerosis and the Potential to Reduce the Global Burden of Atherothrombotic Disease. Circ. Res. 2016, 118, 535–546. [Google Scholar] [CrossRef]
- Yurdagul, A., Jr.; Finney, A.C.; Woolard, M.D.; Orr, A.W. The Arterial Microenvironment: The Where and Why of Atherosclerosis. Biochem. J. 2006, 473, 1281–1295. [Google Scholar] [CrossRef]
- Daniels, T.F.; Killinger, K.M.; Michal, J.J.; Wright, R.W., Jr.; Jiang, Z. Lipoproteins, Cholesterol Homeostasis and Cardiac Health. Int. J. Biol. Sci. 2009, 5, 474–488. [Google Scholar] [CrossRef]
- Wang, Z.; Klipfell, E.; Bennett, B.J.; Koeth, R.; Levison, B.S.; Dugar, B.; Feldstein, A.E.; Britt, E.B.; Fu, X.; Chung, Y.M.; et al. Gut Flora Metabolism of Phosphatidylcholine Promotes Cardiovascular Disease. Nature 2011, 472, 57–63. [Google Scholar] [CrossRef]
- Koeth, R.A.; Wang, Z.; Levison, B.S.; Buffa, J.A.; Org, E.; Sheehy, B.T.; Britt, E.B.; Fu, X.; Wu, Y.; Li, L.; et al. Intestinal Microbiota Metabolism of L-Carnitine, a Nutrient in Red Meat, Promotes Atherosclerosis. Nat. Med. 2013, 19, 576–585. [Google Scholar] [CrossRef]
- Jakopin, Z. Risks Associated with Fat Burners: A Toxicological Perspective. Food Chem. Toxicol. 2018, 123, 205–224. [Google Scholar] [CrossRef]
- Fukami, K.; Yamagishi, S.; Sakai, K.; Kaida, Y.; Yokoro, M.; Ueda, S.; Wada, Y.; Takeuchi, M.; Shimizu, M.; Yamazaki, H.; et al. Oral L-Carnitine Supplementation Increases Trimethylamine-N-Oxide but Reduces Markers of Vascular Injury in Hemodialysis Patients. J. Cardiovasc. Pharmacol. 2015, 65, 289–295. [Google Scholar] [CrossRef]
- Miller, M.J.; Bostwick, B.L.; Kennedy, A.D.; Donti, T.R.; Sun, Q.; Sutton, V.R.; Elsea, S.H. Chronic Oral L-Carnitine Supplementation Drives Marked Plasma Tmao Elevations in Patients with Organic Acidemias Despite Dietary Meat Restrictions. JIMD Rep. 2016, 30, 39–44. [Google Scholar]
- Vallance, H.D.; Koochin, A.; Branov, J.; Rosen-Heath, A.; Bosdet, T.; Wang, Z.; Hazen, S.L.; Horvath, G. Marked Elevation in Plasma Trimethylamine-N-Oxide (Tmao) in Patients with Mitochondrial Disorders Treated with Oral L-Carnitine. Mol. Genet. Metab. Rep. 2018, 15, 130–133. [Google Scholar] [CrossRef]
- Samulak, J.J.; Sawicka, A.K.; Hartmane, D.; Grinberga, S.; Pugovics, O.; Lysiak-Szydlowska, W.; Olek, R.A. L-Carnitine Supplementation Increases Trimethylamine-N-Oxide but Not Markers of Atherosclerosis in Healthy Aged Women. Ann. Nutr. Metab. 2019, 74, 11–17. [Google Scholar] [CrossRef]
- Sawicka, A.K.; Hartmane, D.; Lipinska, P.; Wojtowicz, E.; Lysiak-Szydlowska, W.; Olek, R.A. L-Carnitine Supplementation in Older Women. A Pilot Study on Aging Skeletal Muscle Mass and Function. Nutrients 2018, 10, 255. [Google Scholar] [CrossRef]
- Zhang, A.Q.; Mitchell, S.C.; Smith, R.L. Dietary Precursors of Trimethylamine in Man: A Pilot Study. Food Chem. Toxicol. 1999, 37, 515–520. [Google Scholar] [CrossRef]
- Kasielski, M.; Eusebio, M.O.; Pietruczuk, M.; Nowak, D. The Relationship between Peripheral Blood Mononuclear Cells Telomere Length and Diet—Unexpected Effect of Red Meat. Nutr. J. 2016, 15, 68. [Google Scholar] [CrossRef]
- Jaworska, K.; Huc, T.; Samborowska, E.; Dobrowolski, L.; Bielinska, K.; Gawlak, M.; Ufnal, M. Hypertension in Rats Is Associated with an Increased Permeability of the Colon to Tma, a Gut Bacteria Metabolite. PLoS ONE 2017, 12, e0189310. [Google Scholar] [CrossRef]
- Tang, W.H.; Wang, Z.; Levison, B.S.; Koeth, R.A.; Britt, E.B.; Fu, X.; Wu, Y.; Hazen, S.L. Intestinal Microbial Metabolism of Phosphatidylcholine and Cardiovascular Risk. N. Engl. J. Med. 2013, 368, 1575–1584. [Google Scholar] [CrossRef] [Green Version]
- Tang, W.H.; Wang, Z.; Kennedy, D.J.; Wu, Y.; Buffa, J.A.; Agatisa-Boyle, B.; Li, X.S.; Levison, B.S.; Hazen, S.L. Gut Microbiota-Dependent Trimethylamine N-Oxide (Tmao) Pathway Contributes to Both Development of Renal Insufficiency and Mortality Risk in Chronic Kidney Disease. Circ. Res. 2015, 116, 448–455. [Google Scholar] [CrossRef]
- Suzuki, T.; Heaney, L.M.; Bhandari, S.S.; Jones, D.J.; Ng, L.L. Trimethylamine N-Oxide and Prognosis in Acute Heart Failure. Heart 2016, 102, 841–848. [Google Scholar] [CrossRef]
- Gruppen, E.G.; Garcia, E.; Connelly, M.A.; Jeyarajah, E.J.; Otvos, J.D.; Bakker, S.J.L.; Dullaart, R.P.F. Tmao Is Associated with Mortality: Impact of Modestly Impaired Renal Function. Sci. Rep. 2017, 7, 13781. [Google Scholar] [CrossRef]
- Gansevoort, R.T.; Correa-Rotter, R.; Hemmelgarn, B.R.; Jafar, T.H.; Heerspink, H.J.; Mann, J.F.; Matsushita, K.; Wen, C.P. Chronic Kidney Disease and Cardiovascular Risk: Epidemiology, Mechanisms, and Prevention. Lancet 2013, 382, 339–352. [Google Scholar] [CrossRef]
- Damman, K.; Valente, M.A.; Voors, A.A.; O’Connor, C.M.; van Veldhuisen, D.J.; Hillege, H.L. Renal Impairment, Worsening Renal Function, and Outcome in Patients with Heart Failure: An Updated Meta-Analysis. Eur. Heart J. 2017, 35, 455–469. [Google Scholar] [CrossRef]
- Bain, M.A.; Faull, R.; Fornasini, G.; Milne, R.W.; Evans, A.M. Accumulation of Trimethylamine and Trimethylamine-N-Oxide in End-Stage Renal Disease Patients Undergoing Haemodialysis. Nephrol. Dial. Transplant. 2006, 21, 1300–1304. [Google Scholar] [CrossRef]
- Hauet, T.; Baumert, H.; Gibelin, H.; Godart, C.; Carretier, M.; Eugene, M. Citrate, Acetate and Renal Medullary Osmolyte Excretion in Urine as Predictor of Renal Changes after Cold Ischaemia and Transplantation. Clin. Chem. Lab. Med. 2000, 38, 1093–1098. [Google Scholar] [CrossRef]
- Alshahrani, S.M.; Fraser, G.E.; Sabate, J.; Knutsen, R.; Shavlik, D.; Mashchak, A.; Lloren, J.I.; Orlich, M.J. Red and Processed Meat and Mortality in a Low Meat Intake Population. Nutrients 2019, 11, 622. [Google Scholar] [CrossRef]
- Rohrmann, S.; Linseisen, J.; Allenspach, M.; von Eckardstein, A.; Muller, D. Plasma Concentrations of Trimethylamine-N-Oxide Are Directly Associated with Dairy Food Consumption and Low-Grade Inflammation in a German Adult Population. J. Nutr. 2016, 146, 283–289. [Google Scholar] [CrossRef]
- Cheung, W.; Keski-Rahkonen, P.; Assi, N.; Ferrari, P.; Freisling, H.; Rinaldi, S.; Slimani, N.; Zamora-Ros, R.; Rundle, M.; Frost, G.; et al. A Metabolomic Study of Biomarkers of Meat and Fish Intake. Am. J. Clin. Nutr. 2017, 105, 600–608. [Google Scholar] [CrossRef]
- Veeravalli, S.; Karu, K.; Scott, F.; Fennema, D.; Phillips, I.R.; Shephard, E.A. Effect of Flavin-Containing Monooxygenase Genotype, Mouse Strain, and Gender on Trimethylamine N-Oxide Production, Plasma Cholesterol Concentration, and an Index of Atherosclerosis. Drug Metab. Dispos. 2018, 46, 20–25. [Google Scholar] [CrossRef]
- Schiattarella, G.G.; Sannino, A.; Toscano, E.; Giugliano, G.; Gargiulo, G.; Franzone, A.; Trimarco, B.; Esposito, G.; Perrino, C. Gut Microbe-Generated Metabolite Trimethylamine-N-Oxide as Cardiovascular Risk Biomarker: A Systematic Review and Dose-Response Meta-Analysis. Eur. Heart J. 2017, 38, 2948–2956. [Google Scholar] [CrossRef]
- Shang, R.; Sun, Z.; Li, H. Effective Dosing of L-Carnitine in the Secondary Prevention of Cardiovascular Disease: A Systematic Review and Meta-Analysis. BMC Cardiovasc. Disord. 2014, 14, 88. [Google Scholar] [CrossRef]
- Song, X.; Qu, H.; Yang, Z.; Rong, J.; Cai, W.; Zhou, H. Efficacy and Safety of L-Carnitine Treatment for Chronic Heart Failure: A Meta-Analysis of Randomized Controlled Trials. BioMed Res. Int. 2017, 2017, 6274854. [Google Scholar] [CrossRef]
- Wang, Z.Y.; Liu, Y.Y.; Liu, G.H.; Lu, H.B.; Mao, C.Y. L-Carnitine and Heart Disease. Life Sci. 2018, 194, 88–97. [Google Scholar] [CrossRef]
- Iliceto, S.; Scrutinio, D.; Bruzzi, P.; D’Ambrosio, G.; Boni, L.; di Biase, M.; Biasco, G.; Hugenholtz, P.G.; Rizzon, P. Effects of L-Carnitine Administration on Left Ventricular Remodeling after Acute Anterior Myocardial Infarction: The L-Carnitine Ecocardiografia Digitalizzata Infarto Miocardico (Cedim) Trial. J. Am. Coll. Cardiol. 1995, 26, 380–387. [Google Scholar] [CrossRef]
- Grube, M.; Ameling, S.; Noutsias, M.; Köck, K.; Triebel, I.; Bonitz, K.; Meissner, K.; Jedlitschky, G.; Herda, L.R.; Reinthaler, M.; et al. Selective regulation of cardiac organic cation transporter novel type 2 (OCTN2) in dilated cardiomyopathy. Am. J. Pathol. 2011, 178, 2547–2559. [Google Scholar] [CrossRef] [PubMed]
- Acet, H.; Ertas, F.; Akil, M.A.; Oylumlu, M.; Polat, N.; Yildiz, A.; Bilik, M.Z.; Yuksel, M.; Kaya, Z.; Ulgen, M.S. New Inflammatory Predictors for Non-Valvular Atrial Fibrillation: Echocardiographic Epicardial Fat Thickness and Neutrophil to Lymphocyte Ratio. Int. J. Cardiovasc. Imaging 2014, 30, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Demirkol, S.; Balta, S.; Unlu, M.; Arslan, Z.; Cakar, M.; Kucuk, U.; Celik, T.; Arslan, E.; Turker, T.; Iyisoy, A.; et al. Neutrophils/Lymphocytes Ratio in Patients with Cardiac Syndrome X and Its Association with Carotid Intima-Media Thickness. Clin. Appl. Thromb. Hemost. 2014, 20, 250–255. [Google Scholar] [CrossRef] [PubMed]
- Ussher, J.R.; Lopaschuk, G.D.; Arduini, A. Gut Microbiota Metabolism of L-Carnitine and Cardiovascular Risk. Atherosclerosis 2013, 231, 456–461. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Rodriguez, E.; Fernandez-Prado, R.; Esteras, R.; Perez-Gomez, M.V.; Gracia-Iguacel, C.; Fernandez-Fernandez, B.; Kanbay, M.; Tejedor, A.; Lazaro, A.; Ruiz-Ortega, M.; et al. Impact of Altered Intestinal Microbiota on Chronic Kidney Disease Progression. Toxins 2018, 10, 300. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Prado, R.; Esteras, R.; Perez-Gomez, M.V.; Gracia-Iguacel, C.; Gonzalez-Parra, E.; Sanz, A.B.; Ortiz, A.; Sanchez-Nino, M.D. Nutrients Turned into Toxins: Microbiota Modulation of Nutrient Properties in Chronic Kidney Disease. Nutrients 2017, 9, 489. [Google Scholar] [CrossRef] [PubMed]
- Nowinski, A.; Ufnal, M. Trimethylamine N-Oxide: A Harmful, Protective or Diagnostic Marker in Lifestyle Diseases? Nutrition 2018, 46, 7–12. [Google Scholar] [CrossRef]
- Janeiro, M.H.; Ramirez, M.J.; Milagro, F.I.; Martinez, J.A.; Solas, M. Implication of Trimethylamine N-Oxide (Tmao) in Disease: Potential Biomarker or New Therapeutic Target. Nutrients 2018, 10, 1398. [Google Scholar] [CrossRef]
| L-carnitine | Placebo | |||||
|---|---|---|---|---|---|---|
| End | 4 Months | 12 Months | End | 4 Months | 12 Months | |
| TCh (mg·dL−1) | 217 ± 12 | 213 ± 16 | 211 ± 13 | 199 ± 15 | 211 ± 10 | 202 ± 11 |
| HDL (mg·dL−1) | 67 ± 5 | 72 ± 6 | 70 ± 5 | 61 ± 4 | 72 ± 7 | 67 ± 5 |
| LDL (mg·dL−1) | 127 ± 10 | 119 ± 14 | 115 ± 12 | 117 ± 13 | 120 ± 10 | 117 ± 7 |
| TG (mg·dL−1) | 114 ± 18 | 116 ± 14 | 130 ± 22 | 103 ± 18 | 98 ± 12 | 89 ± 14 |
| L-carnitine | Placebo | |||||
|---|---|---|---|---|---|---|
| End | 4 Months | 12 Months | End | 4 Months | 12 Months | |
| Leuko (109·L−1) | 5.7 ± 0.5 | 6.1 ± 0.6 | 6.6 ± 0.8 | 5.4 ± 0.4 | 5.7 ± 0.5 | 5.5 ± 0.4 |
| Neutro (109·L−1) | 3.0 ± 0.3 | 3.4 ± 0.5 | 3.7 ± 0.5 | 3.0 ± 0.2 | 3.0 ± 0.4 | 2.7 ± 0.2 |
| Lympho (109·L−1) | 2.0 ± 0.2 | 2.0 ± 0.2 | 2.1 ± 0.2 | 1.8 ± 0.2 | 2.1 ± 0.2 | 2.1 ± 0.2 |
| NLR | 1.6 ± 0.2 | 1.7 ± 0.2 | 1.8 ± 0.2 | 1.7 ± 0.2 | 1.5 ± 0.2 | 1.4 ± 0.1 |
| Mono (109·L−1) | 0.51 ± 0.04 | 0.51 ± 0.03 | 0.54 ± 0.05 | 0.45 ± 0.06 | 0.46 ± 0.06 | 0.45 ± 0.04 |
| Platelets (109·L−1) | 279 ± 16 | 272 ± 12 | 291 ± 13 | 249 ± 19 | 248 ± 18 | 253 ± 23 |
| L-carnitine | Placebo | |||
|---|---|---|---|---|
| Median | Range | Median | Range | |
| Cod | F1 | F0–F3 | F1 | F0–F3 |
| Salmon | F1 | F0–F2 | F2 | F0–F3 |
| Mackerel | F1 | F0–F2 | F1 | F0–F2 |
| Herring | F1 | F0–F3 | F1 | F0–F3 |
| Trout | F0 | F0–F1 | F1 | F0–F3 |
| Tuna | F0 | F0–F1 | F0 | F0–F2 |
| Flounder | F0 | F0–F1 | F0 | F0–F1 |
| Hake | F0 | F0–F1 | F0 | F0–F1 |
| Eel | F0 | F0–F1 | F0 | F0–F1 |
| Pollock | F0 | F0–F1 | F0 | F0–F1 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samulak, J.J.; Sawicka, A.K.; Samborowska, E.; Olek, R.A. Plasma Trimethylamine-N-oxide following Cessation of L-carnitine Supplementation in Healthy Aged Women. Nutrients 2019, 11, 1322. https://doi.org/10.3390/nu11061322
Samulak JJ, Sawicka AK, Samborowska E, Olek RA. Plasma Trimethylamine-N-oxide following Cessation of L-carnitine Supplementation in Healthy Aged Women. Nutrients. 2019; 11(6):1322. https://doi.org/10.3390/nu11061322
Chicago/Turabian StyleSamulak, Joanna J., Angelika K. Sawicka, Emilia Samborowska, and Robert A. Olek. 2019. "Plasma Trimethylamine-N-oxide following Cessation of L-carnitine Supplementation in Healthy Aged Women" Nutrients 11, no. 6: 1322. https://doi.org/10.3390/nu11061322
APA StyleSamulak, J. J., Sawicka, A. K., Samborowska, E., & Olek, R. A. (2019). Plasma Trimethylamine-N-oxide following Cessation of L-carnitine Supplementation in Healthy Aged Women. Nutrients, 11(6), 1322. https://doi.org/10.3390/nu11061322





