Protein-Reduced Complementary Foods Based on Nordic Ingredients Combined with Systematic Introduction of Taste Portions Increase Intake of Fruits and Vegetables in 9 Month Old Infants: A Randomised Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
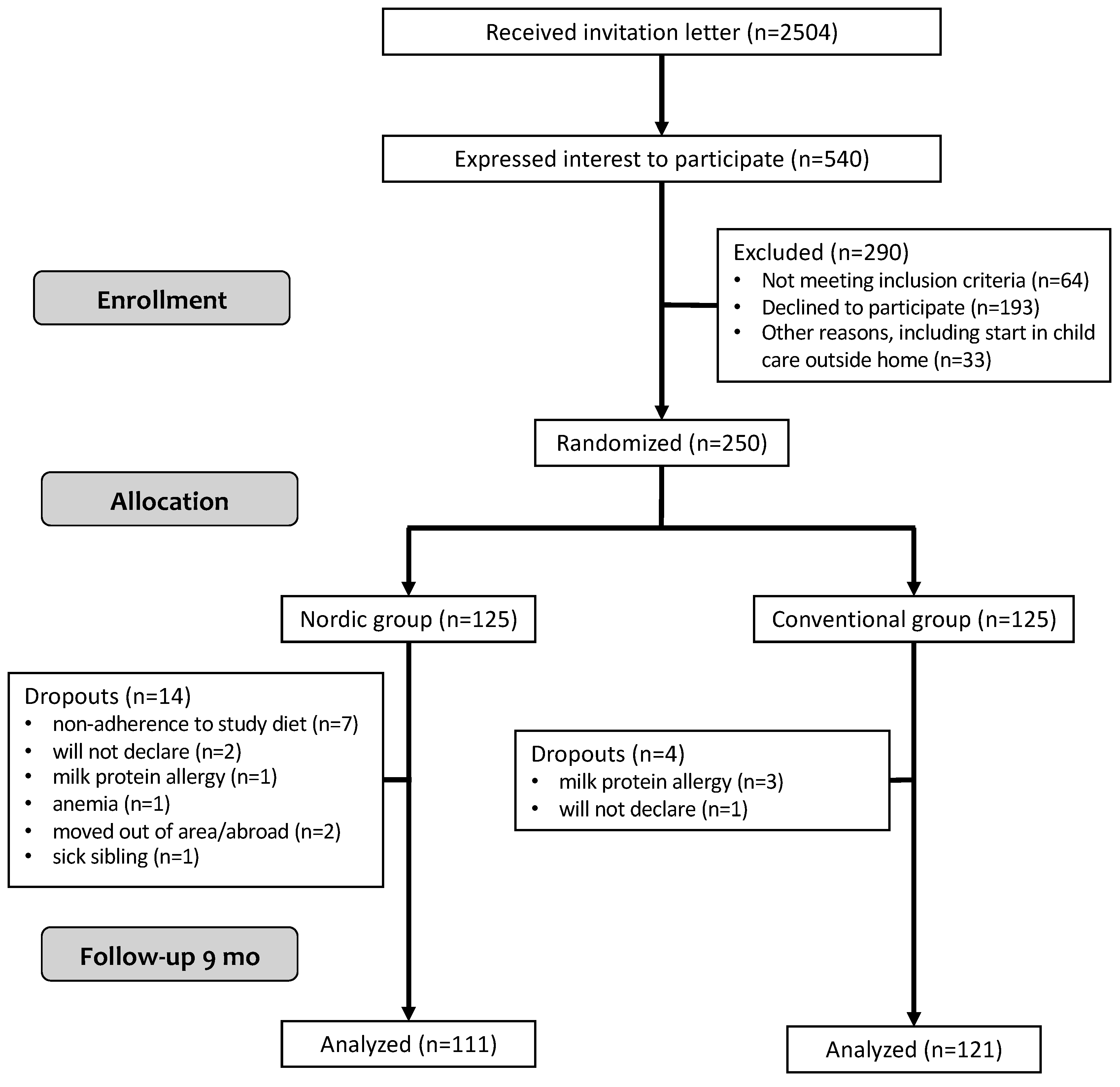
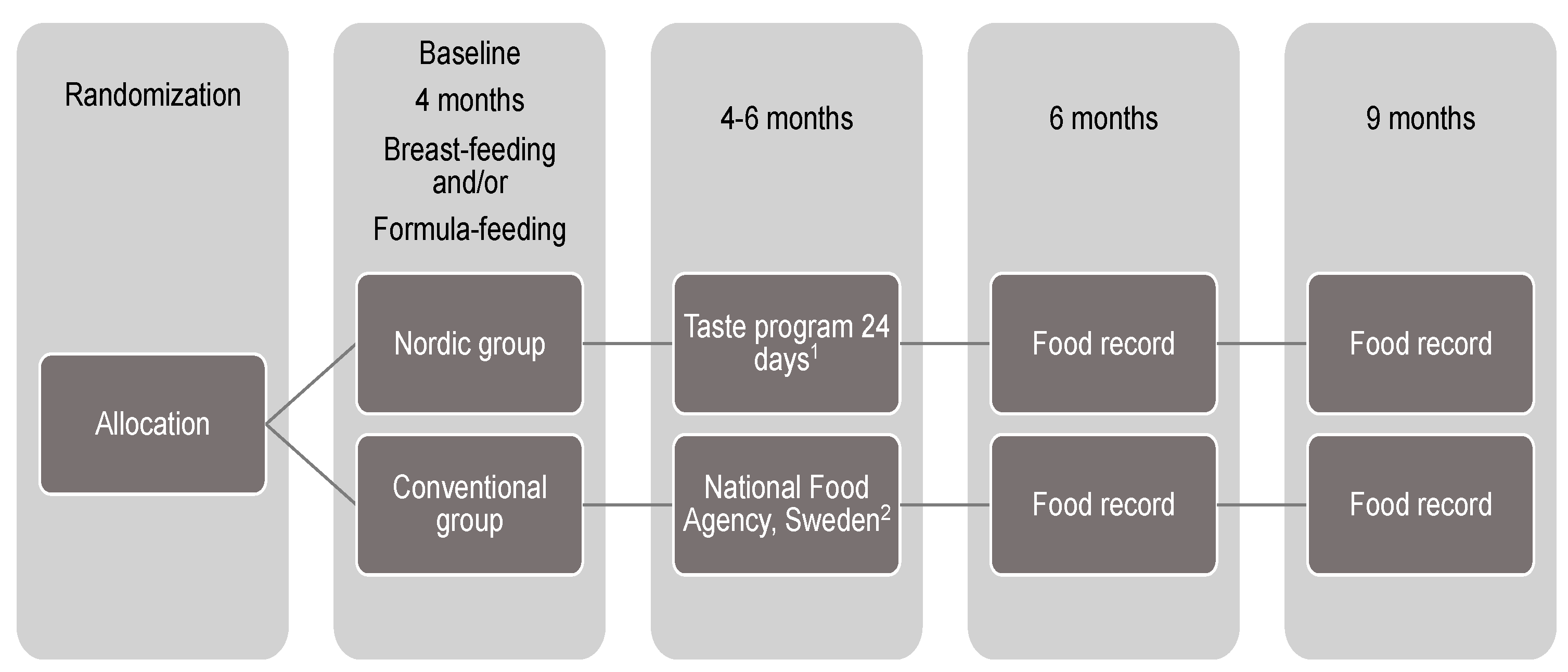
2.1. Study Participants, Design, Allocation and Blinding
2.2. New Nordic Food and Diet
2.3. Introduction of Taste Portions in the Nordic Group
2.4. Adherence to the Taste Portions in the Nordic Group
2.5. Support through Social Media in the Nordic Group
2.6. Baby Food Recipes in the Nordic Group
2.7. Baby Food Products in the Nordic Group
2.8. The Conventional Group and Taste Portion Introduction
2.9. Baby Food Products in the Conventional Group
2.10. Food Records and Dietary Assessment
2.11. Anthropometry
2.12. Blood Samples and Laboratory Analyses
2.13. Demographic Variables
2.14. Group Size Calculation
2.15. Ethical Considerations
2.16. Statistical Analyses
3. Results
3.1. Study Participants
3.2. The Taste Portion Schedule in the Nordic Group
3.3. Energy and Macronutrient Intake and Breast-Feeding Duration
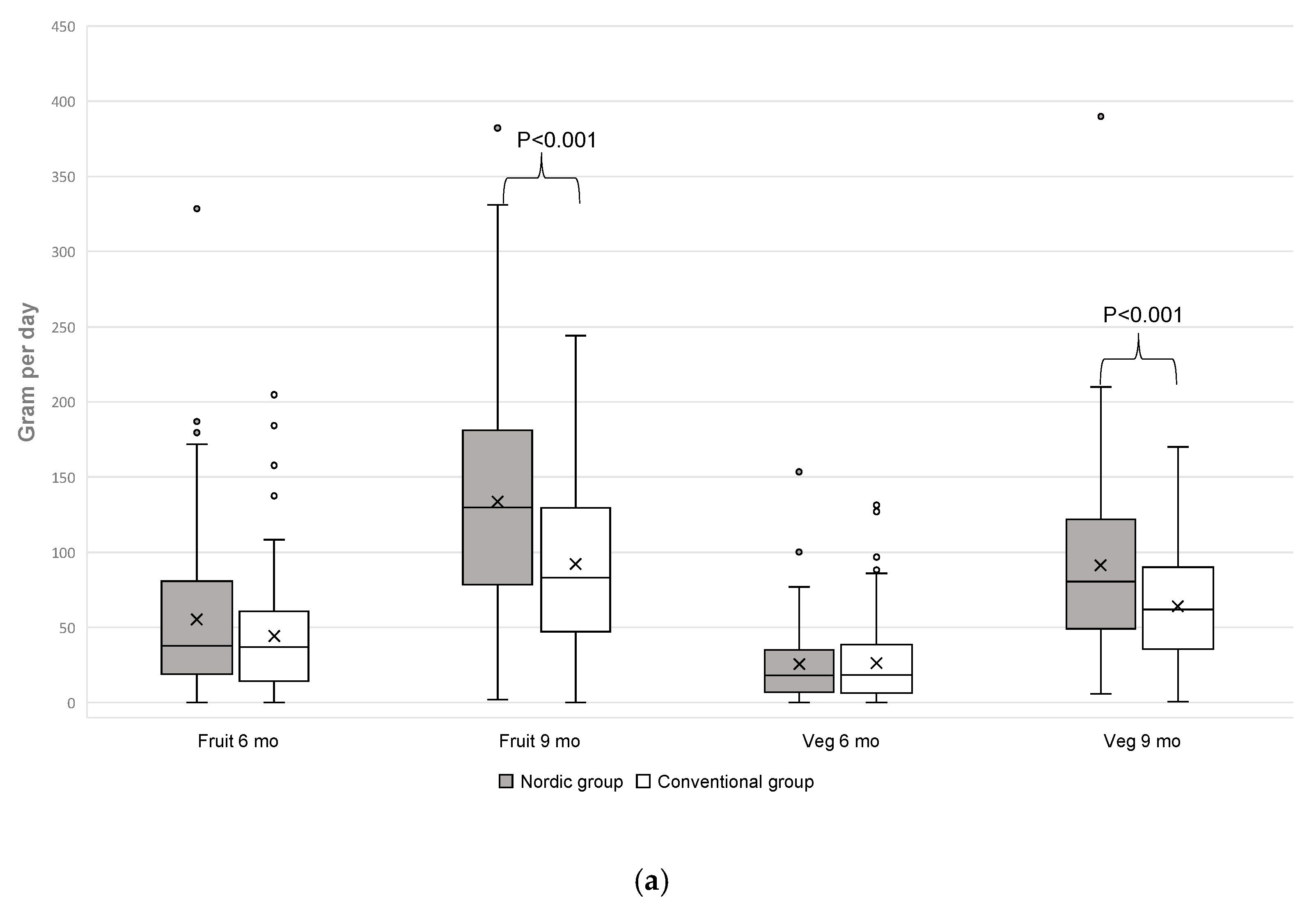
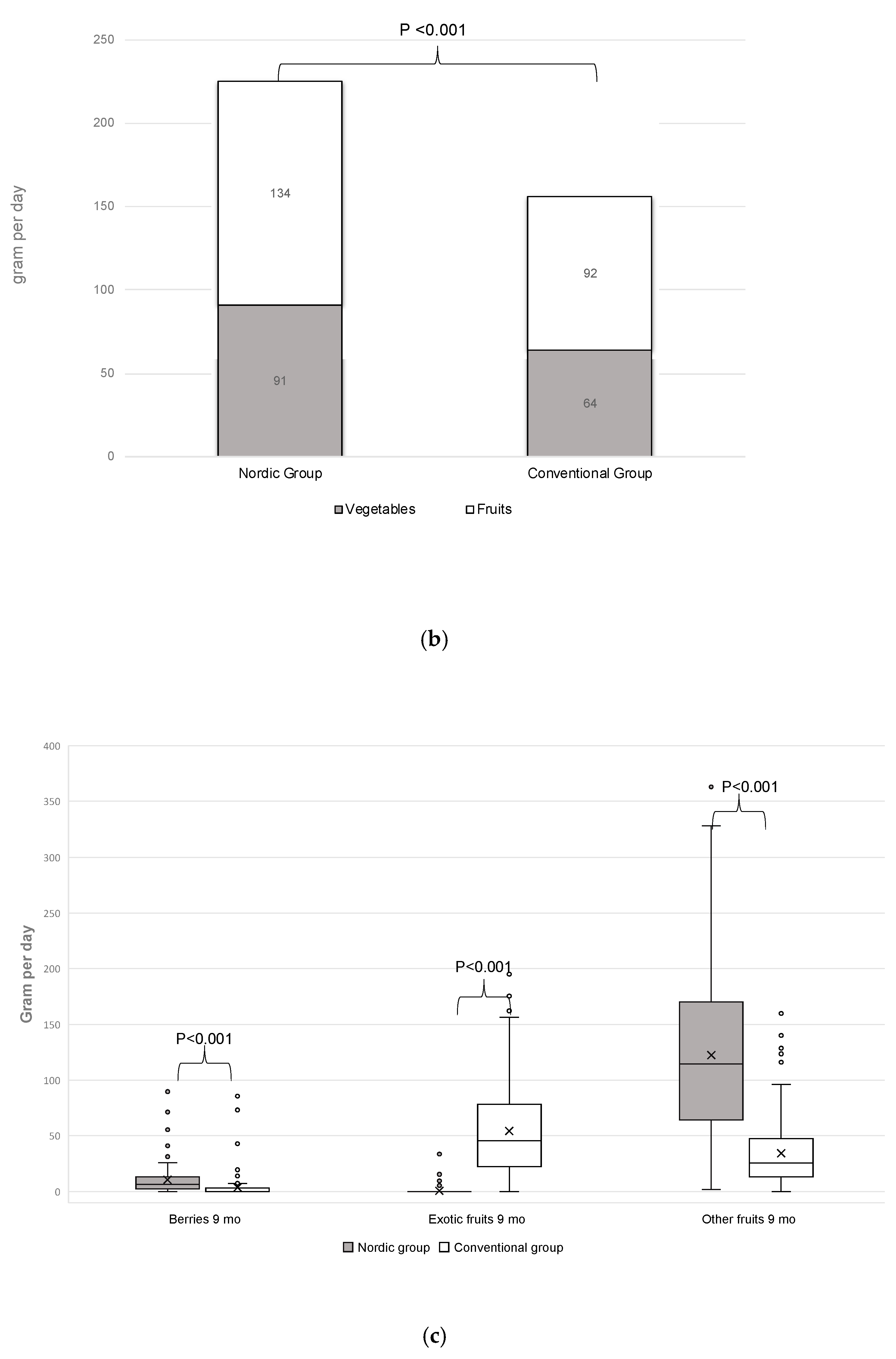
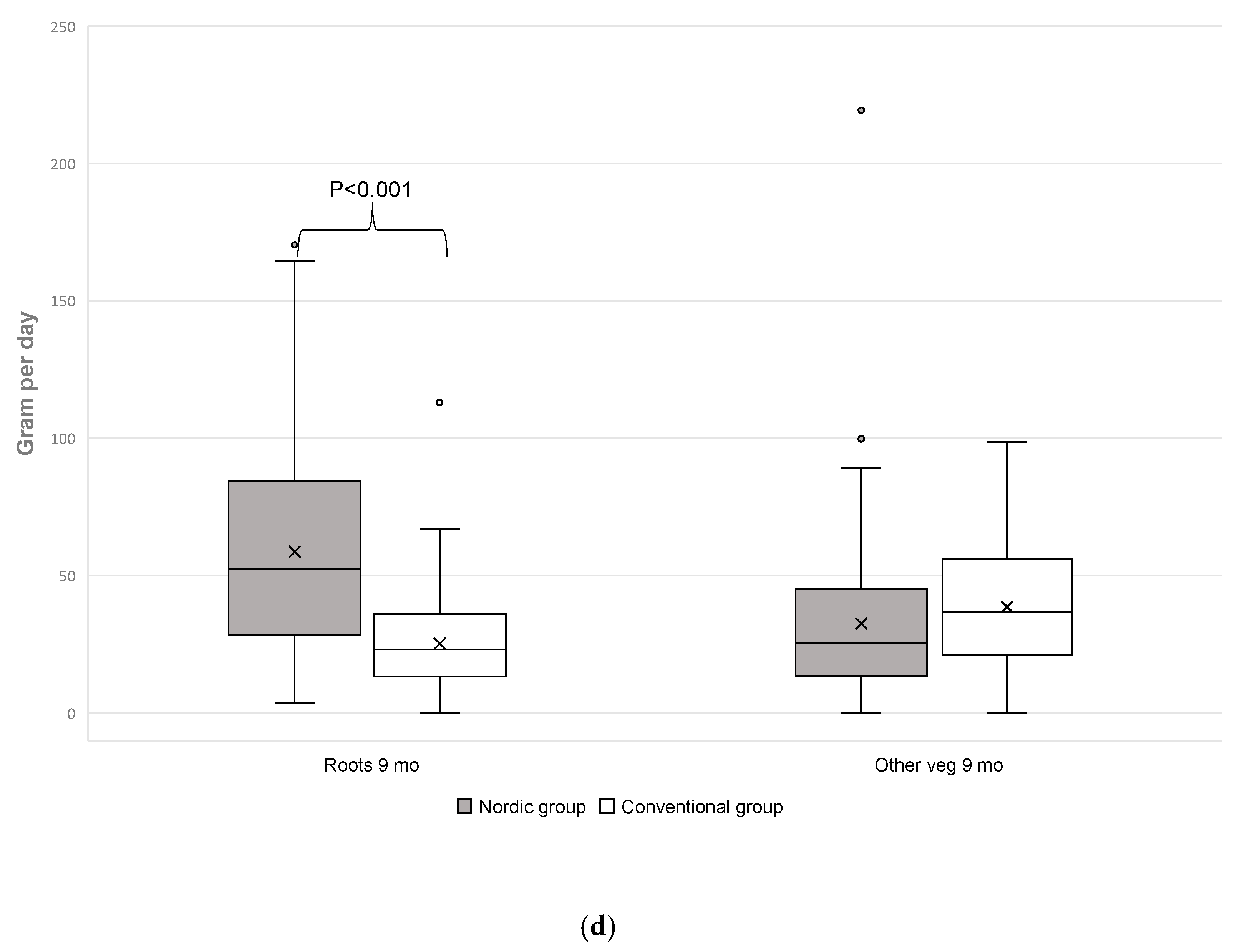
3.4. Fruit and Vegetable Intake
3.5. Main Course Meals
3.6. Anthropometry and Biochemistry
4. Discussion
Strengths and Weaknesses
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviation
| FR: Food record |
| NG: Nordic group |
| CG: Control group |
| EI: Energy intake |
| BMI: Body mass index |
| CF: Complementary feeding |
| SD: Standard deviation |
| OTIS: Optimised complementary feeding study |
| BIG: Baby food in glass jars |
| ND: Nordic diet |
| KJ: Kilojoules |
| MJ: Megajoules |
References
- Mikkilä, V.; Räsänen, L.; Raitakari, O.T.; Pietinen, P.; Viikari, J. Consistent dietary patterns identified from childhood to adulthood: The Cardiovascular Risk in Young Finns Study. Br. J. Nutr. 2005, 93, 923–931. [Google Scholar] [CrossRef]
- Craigie, A.M.; Lake, A.A.; Kelly, S.A.; Adamson, A.J.; Mathers, J.C. Tracking of obesity-related behaviours from childhood to adulthood: A systematic review. Maturitas 2011, 70, 266–284. [Google Scholar] [CrossRef]
- Saavedra, J.M.; Deming, D.; Dattilo, A.; Reidy, K. Lessons from the Feeding Infants and Toddlers Study in North America: What Children Eat, and Implications for Obesity Prevention. Ann. Nutr. Metab. 2013, 62, 27–36. [Google Scholar] [CrossRef]
- Birch, L.; Savage, J.S.; Ventura, A. Influences on the Development of Children’s Eating Behaviours: From Infancy to Adolescence. Can. J. Diet. Pract. Res. 2007, 68, s1–s56. [Google Scholar]
- Young, B.E.; Krebs, N.F. Complementary Feeding: Critical Considerations to Optimize Growth, Nutrition, and Feeding Behavior. Curr. Pediatr. Rep. 2013, 1, 247–256. [Google Scholar] [CrossRef]
- World Health Organization. Global Status Report on Non Communicable Diseases 2010; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Amcoff, E.; Edberg, A.; Enghardt Barbieri, H.; Lindroos, A.K.; Nälsén, C.; Pearson, M.; Warensjö Lemming, E. Riksmaten - vuxna 2010-11. Livsmedels- och näringsintag bland vuxna i Sverige; Livsmedelsverket (Swedish National Food Agency): Uppsala, Sweden, 2012.
- Barbieri Enghardt, H.; Pearson, M.; Becker, W. Riksmaten - barn 2003. Livsmedels- och näringsintag bland barn i Sverige; Livsmedelsverket (Swedish National Food Agency): Uppsala, Sweden, 2003.
- Inchley, J.; Currie, D.; Young, T.; Samdal, O.; Torsheim, T.; Augustson, L.; Mathison, F.; Aleman Diaz, A.; Molcho, M.; Weber, M.; et al. Growing up Unequal: Gender and Socioeconomic Differences in Young People’s Health and Wellbeing. Health Behaviour in School-Aged Children (HBSC) Study: International Report from the 2013/2014 Survey; WHO Regional Office for Europe: Copenhagen, Denmark, 2016. [Google Scholar]
- National Cancer Institute. Usual Dietary Intakes: Food Intakes, US Population, 2007–2010. Usual Dietary Intakes: Food Intakes, US Population, 2007–2010; National Cancer Institute: Bethesda, MD, USA, 2015.
- Ness, A.R.; Maynard, M.; Frankel, S.; Davey Smith, G.; Frobisher, C.; Leary, S.D.; Emmett, P.M.; Gunnell, D. Diet in childhood and adult cardiovascular and all cause mortality: The Boyd Orr cohort. Heart 2005, 91, 894–898. [Google Scholar] [CrossRef]
- Maynard, M.; Gunnell, D.; Emmett, P.; Frankel, S.; Davey, S. Fruit, vegetables, and antioxidants in childhood and risk of adult cancer: The Boyd Orr cohort. J. Epidemiol. Community Health 2003, 57, 218–225. [Google Scholar] [CrossRef]
- Boeing, H.; Bechthold, A.; Bub, A.; Ellinger, S.; Haller, D.; Kroke, A.; Leschik-Bonnet, E.; Müller, M.J.; Oberritter, H.; Schulze, M.; et al. Critical review: Vegetables and fruit in the prevention of chronic diseases. Eur. J. Nutr. 2012, 51, 637–663. [Google Scholar] [CrossRef]
- World Health Organization. Global Strategy on Diet, Physical Activity and Health: Promoting Fruit and Vegetable Consumption around the World; World Health Organization: Geneva, Switzerland, 2003; Available online: htpps://who.int/dietphysicalactivity/fruit/en/ (accessed on 5 December 2018).
- Mennella, J.A.; Reiter, A.R.; Daniels, L.M. Vegetable and Fruit Acceptance during Infancy: Impact of Ontogeny, Genetics, and Early Experiences. Adv. Nutr. Int. Rev. J. 2016, 7, 211S–219S. [Google Scholar] [CrossRef]
- Mennella, J.A. Ontogeny of taste preferences: Basic biology and implications for health12345. Am. J. Clin. Nutr. 2014, 99, 704S–711S. [Google Scholar] [CrossRef]
- Forestell, C.A.; Mennella, J.A. The Relationship Between Infant Facial Expressions and Food Acceptance. Curr. Nutr. Rep. 2017, 6, 141–147. [Google Scholar] [CrossRef] [PubMed]
- Harris, G. Development of taste and food preferences in children. Clin. Nutr. Metab. Care 2008, 11, 315–319. [Google Scholar] [CrossRef] [PubMed]
- Coulthard, H.; Harris, G.; Emmett, P. Long-term consequences of early fruit and vegetable feeding practices in the United Kingdom. Public Health Nutr. 2010, 13, 2044–2051. [Google Scholar] [CrossRef] [PubMed]
- Mennella, J.A.; Daniels, L.M.; Reiter, A.R. Learning to like vegetables during breastfeeding: A randomized clinical trial of lactating mothers and infants. Am. J. Clin. Nutr. 2017, 106, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Hodder, R.K.; O’Brien, K.M.; Stacey, F.G.; Wyse, R.J.; Clinton-McHarg, T.; Tzelepis, F.; James, E.L.; Bartlem, K.M.; Nathan, N.K.; Sutherland, R.; et al. Interventions for increasing fruit and vegetable consumption in children aged five years and under. Cochrane Database Syst. Rev. 2018. [Google Scholar] [CrossRef] [PubMed]
- Caton, S.J.; Ahern, S.M.; Remy, E.; Nicklaus, S.; Blundell, P.; Hetherington, M.M. Repetition counts: Repeated exposure increases intake of a novel vegetable in UK pre-school children compared to flavour–flavour and flavour–nutrient learning. Br. J. Nutr. 2013, 109, 2089–2097. [Google Scholar] [CrossRef]
- Coulthard, H.; Harris, G.; Emmett, P. Delayed introduction of lumpy foods to children during the complementary feeding period affects child’s food acceptance and feeding at 7 years of age. Matern. Child. Nutr. 2009, 5, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Hetherington, M.M.; Schwartz, C.; Madrelle, J.; Croden, F.; Nekitsing, C.; Vereijken, C.M.J.L.; Weenen, H. A step-by-step introduction to vegetables at the beginning of complementary feeding. The effects of early and repeated exposure. Appetite 2015, 84, 280–290. [Google Scholar] [CrossRef]
- Mok, E.; Vanstone, C.A.; Gallo, S.; Li, P.; Constantin, E.; Weiler, H.A. Diet diversity, growth and adiposity in healthy breastfed infants fed homemade complementary foods. Int. J. Obes. 2017, 41, 776–782. [Google Scholar] [CrossRef]
- Hörnell, A.; Lagström, H.; Lande, B.; Thorsdottir, I. Protein intake from 0 to 18 years of age and its relation to health: A systematic literature review for the 5th Nordic Nutrition Recommendations. Food Nutr. Res. 2013, 57, 21083. [Google Scholar] [CrossRef]
- Patro-Gołąb, B.; Zalewski, B.M.; Kołodziej, M.; Kouwenhoven, S.; Poston, L.; Godfrey, K.M.; Koletzko, B.; van Goudoever, J.B.; Szajewska, H. Nutritional interventions or exposures in infants and children aged up to three years and their effects on subsequent risk of overweight, obesity, and body fat: A systematic review of systematic reviews. Obes. Rev. Off. J. Int. Assoc. Study Obes. 2016, 17, 1245–1257. [Google Scholar] [CrossRef] [PubMed]
- Öhlund, I.; Hernell, O.; Hörnell, A.; Stenlund, H.; Lind, T. BMI at 4 years of age is associated with previous and current protein intake and with paternal BMI. Eur. J. Clin. Nutr. 2009, 64, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Gunnarsdottir, I.; Thorsdottir, I. Relationship between growth and feeding in infancy and body mass index at the age of 6 years. Int. J. Obes. 2003, 27, 1523–1527. [Google Scholar] [CrossRef]
- Hoppe, C.; Mølgaard, C.; Thomsen, B.L.; Juul, A.; Michaelsen, K.F. Protein intake at 9 mo of age is associated with body size but not with body fat in 10-y-old Danish children. Am. J. Clin. Nutr. 2004, 79, 494–501. [Google Scholar] [CrossRef] [PubMed]
- Thorisdottir, B.; Gunnarsdottir, I.; Thorisdottir, A.V.; Palsson, G.I.; Halldorsson, T.I.; Thorsdottir, I. Nutrient intake in infancy and body mass index at six years in two population-based cohorts recruited before and after revision of infant dietary recommendations. Ann. Nutr. Metab. 2013, 63, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Nordic Council of Ministers. Nordic Nutrition Recommendations 2012, 5th ed.; Nordic Council of Ministers: Copenhagen, Denmark, 2013. [Google Scholar]
- World Health Organization. Protein and Amino Acid Requirements in Human Nutrition Report of a Joint WHO/FAO/UNU Expert Consultation; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Thorisdottir, B.; Gunnarsdottir, I.; Palsson, G.I.; Halldorsson, T.I.; Thorsdottir, I. Animal protein intake at 12 months is associated with growth factors at the age of six. Acta Paediatr. 2014, 103, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Mithril, C.; Dragsted, L.O.; Meyer, C.; Blauert, E.; Holt, M.K.; Astrup, A. Guidelines for the new Nordic diet. Public Health Nutr. 2012, 15, 1941–1947. [Google Scholar] [CrossRef]
- Damsgaard, C.T.; Dalskov, S.-M.; Petersen, R.A.; Sørensen, L.B.; Mølgaard, C.; Biltoft-Jensen, A.; Andersen, R.; Thorsen, A.V.; Tetens, I.; Sjödin, A.; et al. Design of the OPUS School Meal Study: A randomised controlled trial assessing the impact of serving school meals based on the New Nordic Diet. Scand. J. Public Health 2012, 40, 693–703. [Google Scholar] [CrossRef]
- Adamsson, V.; Reumark, A.; Fredriksson, I.B.; Hammarstrom, E.; Vessby, B.; Johansson, G.; Risérus, U. Effects of a healthy Nordic diet on cardiovascular risk factors in hypercholesterolaemic subjects: A randomized controlled trial (NORDIET). J. Intern. Med. 2011, 269, 150–159. [Google Scholar] [CrossRef]
- Perälä, M.-M.; von Bonsdorff, M.; Männistö, S.; Salonen, M.K.; Simonen, M.; Kanerva, N.; Pohjolainen, P.; Kajantie, E.; Rantanen, T.; Eriksson, J.G. A healthy Nordic diet and physical performance in old age: Findings from the longitudinal Helsinki Birth Cohort Study. Br. J. Nutr. 2016, 115, 878–886. [Google Scholar] [CrossRef]
- Skreden, M.; Hillesund, E.R.; Wills, A.K.; Brantsæter, A.L.; Bere, E.; Øverby, N.C. Adherence to the New Nordic Diet during pregnancy and subsequent maternal weight development: A study conducted in the Norwegian Mother and Child Cohort Study (MoBa). Br. J. Nutr. 2018, 119, 1286–1294. [Google Scholar] [CrossRef] [PubMed]
- Lacoppidan, S.A.; Kyrø, C.; Loft, S.; Helnæs, A.; Christensen, J.; Hansen, C.P.; Dahm, C.C.; Overvad, K.; Tjønneland, A.; Olsen, A. Adherence to a healthy Nordic food index is associated with a lower risk of type-2 diabetes—The Danish diet, cancer and health cohort study. Nutrients 2015, 7, 8633–8644. [Google Scholar] [CrossRef] [PubMed]
- Uusitupa, M.; Hermansen, K.; Savolainen, M.J.; Schwab, U.; Kolehmainen, M.; Brader, L.; Mortensen, L.S.; Cloetens, L.; Johansson-Persson, A.; Önning, G.; et al. Effects of an isocaloric healthy Nordic diet on insulin sensitivity, lipid profile and inflammation markers in metabolic syndrome—A randomized study (SYSDIET). J. Intern. Med. 2013, 274, 52–66. [Google Scholar] [CrossRef] [PubMed]
- Lind, T.; Johansson, U.; Öhlund, I.; Lindberg, L.; Lönnerdal, B.; Tennefors, C.; Hernell, O. Study protocol: Optimized complementary feeding study (OTIS): A randomized controlled trial of the impact of a protein-reduced complementary diet based on Nordic foods. BMC Public Health 2019, 19, 134. [Google Scholar] [CrossRef] [PubMed]
- Adamsson, V.; Reumark, A.; Cederholm, T.; Vessby, B.; Riserus, U.; Johansson, G. What is a healthy Nordic diet? Foods and nutrients in the NORDIET study. Food Nutr. Res. 2012, 56, 18189. [Google Scholar] [CrossRef] [PubMed]
- Ministers NC of New Nordic Food—A Sustainable Choice. 2011. Available online: https://www.norden.org/en/publications/publikationer/2011-410 (accessed on 11 May 2017).
- Mithril, C.; Dragsted, L.O.; Meyer, C.; Tetens, I.; Biltoft-Jensen, A.; Astrup, A. Dietary composition and nutrient content of the New Nordic Diet. Public Health Nutr. 2013, 16, 777–785. [Google Scholar] [CrossRef] [PubMed]
- Mennella, J.A.; Bobowski, N.K. The sweetness and bitterness of childhood: Insights from basic research on taste preferences. Physiol. Behav. 2015, 152, 502–507. [Google Scholar] [CrossRef]
- Textalk—Media and Commerce Development Since 1987. Available online: https://www.textalk.com (accessed on 1 October 2017).
- Sweden Food Agency. Good Food for Infants Under One Year; Sweden Food Agency. 2011. Available online: https://slv.se (accessed on 21 October 2018).
- Michaelsen, K.F.; Skafte, L.; Badsberg, J.H.; Jorgensen, M. Variation in Macronutrients in Human Bank Milk: Influencing Factors and Implications for Human Milk Banking. J. Pediatr. Gastroenterol. Nutr. 1990, 11, 229–239. [Google Scholar] [CrossRef]
- Paul, A.A.; Black, A.E.; Evans, J.; Cole, T.J.; Whitehead, R.G. Breastmilk intake and growth in infants from two to ten months. J. Hum. Nutr. Diet. 1988, 1, 437–450. [Google Scholar] [CrossRef]
- Swedish Food Agency. National Food Administration; 2017 Swedish Food Database. Available online: https://www.livsmedelsverket.se (accessed on 1 January 2017).
- Lohman, T.G.; Roche, A.F.; Martorell, R. Anthropometric Standardization Reference Manual; Human Kinetics Publications: Champaign, IL, USA, 1988. [Google Scholar]
- World Health Organization. WHO Child Growth Standards: Length/Height-for-Age, Weight-for-Age, Weight-for-Length, Weight-for-Height and Body Mass Index-for-Age: Methods and Development; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- Rustad, P.; Felding, P.; Lahti, A.; Petersen, P.H. Descriptive analytical data and consequences for calculation of common reference intervals in the Nordic reference interval project 2000. Scand. J. Clin. Lab. Invest. 2004, 64, 343–370. [Google Scholar] [CrossRef]
- Hedges, L.V. Distribution theory for Glass’s estimator of effect size and related estimators. J. Educ. Stat. 1981, 6, 107–128. [Google Scholar] [CrossRef]
- Willett, W.; Rockström, J.; Loken, B.; Springmann, M.; Lang, T.; Vermeulen, S.; Garnett, T.; Tilman, D.; DeClerck, F.; Wood, A.; et al. Food in the Anthropocene: The EAT–Lancet Commission on healthy diets from sustainable food systems. Lancet 2019, 393, 447–492. [Google Scholar] [CrossRef]
- Fildes, A.; van Jaarsveld, C.H.M.; Wardle, J.; Cooke, L. Parent-Administered Exposure to Increase Children’s Vegetable Acceptance: A Randomized Controlled Trial. J. Acad. Nutr. Diet. 2014, 114, 881–888. [Google Scholar] [CrossRef] [PubMed]
- Hausner, H.; Hartvig, D.L.; Reinbach, H.C.; Wendin, K.; Bredie, W.L.P. Effects of repeated exposure on acceptance of initially disliked and liked Nordic snack bars in 9–11 year-old children. Clin. Nutr. 2012, 31, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Ahern, S.M.; Caton, S.J.; Blundell, P.; Hetherington, M.M. The root of the problem: Increasing root vegetable intake in preschool children by repeated exposure and flavour flavour learning. Appetite 2014, 80, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Mennella, J.A.; Beauchamp, G.K. Flavor experiences during formula feeding are related to preferences during childhood. Early Hum. Dev. 2002, 68, 71–82. [Google Scholar] [CrossRef]
- Barends, C.; de Vries, J.; Mojet, J.; de Graaf, C. Effects of repeated exposure to either vegetables or fruits on infant’s vegetable and fruit acceptance at the beginning of weaning. Food Qual. Prefer. 2013, 29, 157–165. [Google Scholar] [CrossRef]
- Campbell, K.J.; Lioret, S.; McNaughton, S.A.; Crawford, D.A.; Salmon, J.; Ball, K.; McCallum, Z.; Gerner, B.E.; Spence, A.C.; Cameron, A.J.; et al. A Parent-Focused Intervention to Reduce Infant Obesity Risk Behaviors: A Randomized Trial. Pediatrics 2013, 131, 652–660. [Google Scholar] [CrossRef]
- Fildes, A.; Lopes, C.; Moreira, P.; Moschonis, G.; Oliveira, A.; Mavrogianni, C.; Manios, Y.; Beeken, R.; Wardle, J.; Cooke, L. An exploratory trial of parental advice for increasing vegetable acceptance in infancy. Br. J. Nutr. 2015, 114, 328–336. [Google Scholar] [CrossRef]
- Hörnell, A.; Hofvander, Y.; Kylberg, E. Introduction of solids and formula to breastfed infants: A longitudinal prospective study in Uppsala, Sweden. Acta Pædiatrica 2001, 90, 477–482. [Google Scholar] [CrossRef]
| Nordic Group n = 125 | Conventional Group n = 125 | |
|---|---|---|
| Girls/boys (n, %) | 52/73 (42)/(58) | 56/69 (45)/(55) |
| mean ± SD | mean ± SD | |
| Age at baseline (months) | 4.5 ± 0.5 | 4.5 ± 0.5 |
| Neonatal characteristics | ||
| Birth weight (kg) | 3.6 ± 0.4 | 3.6 ± 0.5 |
| Birth length (cm) | 50.7 ± 1.9 | 50.6 ± 2.1 |
| Birth head circumference (cm) | 35.4 ± 1.3 | 35.5 ± 1.4 |
| Gestational age (weeks) | 39.6 ± 1.3 | 39.8 ± 1.3 |
| Anthropometry | ||
| Weight (kg) | 7.3 ± 0.8 | 7.3 ± 0.8 |
| Weight-for-age z score | 0.31 ± 0.87 | 0.35 ± 0.89 |
| Weight-for-length z score | 0.07 ± 0.98 | 0.12 ± 0.96 |
| Length (cm) | 65.2 ± 3.0 | 65.1 ± 2.4 |
| Length-for-age z score | 0.47 ± 0.85 | 0.50 ± 0.94 |
| Head circumstance (cm) | 42.4 ± 1.3 | 42.5 ± 1.3 |
| Head circumstance-for-age z-score | 0.69 ± 0.89 | 0.75 ± 0.90 |
| Body mass index (BMI; kg/m2) | 17.2 ± 1.5 | 17.2 ± 1.4 |
| BMI-for-age z score | 0.05 ± 0.96 | 0.09 ± 0.95 |
| Breast-feeding | n (%) | n (%) |
| Breast-feeding | 95 (76) | 94 (75) |
| Never breast-fed | 2 (1.6) | 2 (1.6) |
| mean ± SD | mean ± SD | |
| Duration of exclusive breast-feeding (months) | 4.1 ± 1.5 | 4.2 ± 1.4 |
| Laboratory markers | mean ± SD | mean ± SD |
| Haemoglobin (g/L) | 116 ± 7 | 116 ± 8 |
| Mean corpuscular volume (fL) | 77.7 ± 3.3 | 77.7 ± 3.0 |
| S-Transferrin (g/L) | 2.3 ± 0.3 | 2.3 ± 0.4 |
| Transferrin saturation (%) | 16.5 ± 5.5 | 16.3 ± 6.2 |
| S-Ferritin (μg/L) (geometric mean) | 107.6 ± 2.1 | 105.7 ± 2.1 |
| S-Urea (mmol/L) | 2.0 ± 0.5 | 2.0 ± 0.5 |
| S-Folate (nmol/L) (median, IQR) | 44.0 (38–45) | 41.0 (36.8–45) |
| Family characteristics | mean ± SD | mean ± SD |
| No siblings (n, %) | 63 (52) | 69 (55) |
| Mothers’ age (years) | 31 ± 4.6 | 31 ± 4.9 |
| Partners’ age (years) | 34 ± 5.0 | 32 ± 5.4 |
| Education level Mother | n (%) | n (%) |
| Elementary school | 2 (1.6) | 4 (3.2) |
| High school | 34 (27.9) | 36 (28.8) |
| University | 86 (70.5) | 84 (67.2) |
| Education level Partner | ||
| Elementary school | 10 (8.2) | 9 (7.2) |
| High school | 44 (36.1) | 48 (39.2) |
| University | 68 (55.7) | 67 (53.6) |
| Ethnicity (born in Sweden) | ||
| Mother | 120 (98.4) | 118 (95.2) |
| Partner | 110 (88.0) | 104 (83.2) |
| Health | ||
| Smoking mother | 2 (1.6) | 1 (0.8) |
| Smoking partner | 13 (10.7) | 13 (10.4) |
| Food allergy mother (self-estimated) | 25 (20.3) | 20 (16) |
| Food allergy partner (self-estimated) | 13 (10.6) | 17 (13.6) |
| Food allergy siblings | 4 (7) 1 | 5 (9) 1 |
| Annual income per household (Euro €/per thousand) | ||
| <10: | 1 (0.8) | 2 (1.6) |
| 10.0–19.9: | 8 (6.6) | 10 (8.0) |
| 20.0–29.9: | 30 (24.6) | 32 (25.6) |
| 30.0–39.9: | 35 (28.7) | 34 (27.2) |
| 40.0–49.9: | 30 (24.6) | 30 (24.0) |
| 50.0–59.9: | 11 (9.0) | 8 (6.4) |
| >60: | 7 (5.7) | 7 (5.6) |
| Taste | Total no. of Exposures (n) | Total Amounts of Purée (g) | Amounts of Purée per Exposure (g) | Total no. of Refuses (n) |
|---|---|---|---|---|
| mean ± SD | mean ± SD | mean ± SD | mean ± SD | |
| Apple | 8.3 ± 1.3 | 72.1 ± 51.2 | 8.6 ± 5.7 | 0.3 ± 1.0 |
| Green peas | 8.1 ± 1.3 | 68.7 ± 45.9 | 8.5 ± 5.3 | 0.4 ± 0.9 |
| Raspberry | 8.0 ± 1.4 | 67.9 ± 47.2 | 8.4 ± 7.1 | 0.5 ± 1.4 |
| Cauliflower | 7.9 ± 1.5 | 76.3 ± 46.7 | 9.6 ± 5.4 | 0.4 ± 1.4 |
| Buckthorn/Lingonberry 1 | 8.0 ± 1.7 | 68.4 ± 47.0 | 8.3 ± 5.1 | 0.4 ± 1.0 |
| Turnip | 7.9 ± 1.7 | 77.4 ± 44.6 | 9.7 ± 5.1 | 0.3 ± 0.7 |
| Cranberry | 7.7 ± 1.7 | 63.9 ± 44.8 | 8.2 ± 5.4 | 0.5 ± 1.4 |
| White radish | 7.8 ± 2.0 | 73.3 ± 46.7 | 9.4 ± 5.4 | 0.4 ± 1.2 |
| Intake | Nordic Group Mean ± SD | Conventional Group Mean ± SD | p for Difference 1 |
|---|---|---|---|
| Energy and macronutrient at 6 months | |||
| Age at follow-up (months) | 6.3 ± 0.6 | 6.2 ± 0.5 | 0.30 |
| Energy (kJ) | 2941 ± 525 | 2941 ± 497 | 0.76 |
| Protein (g) | 11.7 ± 3.0 | 13.1 ± 4.0 | 0.003 |
| Fat (g) | 32.3 ± 6.0 | 33.1 ± 6.7 | 0.38 |
| Carbohydrate (g) | 87.6 ± 20.9 | 83.1 ± 16.8 | 0.07 |
| Energy and macronutrient at 9 months | |||
| Age at follow-up (months) | 8.7 ± 0.4 | 8.7 ± 0.3 | 0.99 |
| Energy (kJ) | 3472 ± 498 | 3432 ± 538 | 0.56 |
| Energy/bodyweight (kJ/kg) | 379 ± 62 | 372 ± 58 | 0.38 |
| Protein (g) | 15.9 ± 3.6 | 21.5 ± 5.7 | <0.001 |
| Protein/bodyweight (g/kg) | 1.7 ± 0.4 | 2.3 ± 0.6 | <0.001 |
| Fat (g) | 32.8 ± 5.4 | 33.7 ± 6.9 | 0.27 |
| Carbohydrate (g) | 113.6 ± 20.6 | 103.4 ± 18.5 | <0.001 |
| Nordic Group Mean ± SD | Conventional Group Mean ± SD | p for Difference 1,2 | |
|---|---|---|---|
| Anthropometry | n = 108 | n = 122 | |
| Age at follow-up (months) | 8.7 ± 0.4 | 8.7 ± 0.4 | 0.97 1 |
| Body weight (kg) | 9.2 ± 1.0 | 9.3 ± 1.0 | 0.75 1 |
| Weight-for-age z score | 0.62 ± 0.91 | 0.68 ± 0.91 | 0.60 1 |
| Weight-for-length z score | 0.60 ± 0.97 | 0.60 ± 0.96 | 0.80 1 |
| Body length (cm) | 71.8 ± 2.1 | 72.0 ± 2.5 | 0.51 1 |
| Length-for-age z score | 0.41 ± 0.79 | 0.57 ± 0.99 | 0.24 1 |
| Head circumstance (cm) | 45.7 ± 1.3 | 45.7 ± 1.5 | 0.87 1 |
| Head circumstance-for-age z-score | 0.99 ± 0.87 | 1.05 ± 1.01 | 0.82 1 |
| BMI (kg/m2) | 17.9 ± 1.5 | 17.8 ± 1.5 | 0.97 1 |
| BMI-for-age z score | 0.51 ± 0.98 | 0.49 ± 0.98 | 0.92 1 |
| Laboratory markers | n = 101 | n = 114 | |
| Haemoglobin (g/L) | 114.7 ± 6.4 | 116.3 ± 7.5 | 0.10 1 |
| Mean corpuscular volume (fL) | 75.7 ± 2.5 | 75.8 ± 2.7 | 0.69 1 |
| S-Transferrin (g/L) | 2.54 ± 0.31 | 2.58 ± 0.34 | 0.40 1 |
| S-Transferrin saturation (%) | 13.8 ± 5.9 | 14.4 ± 5.2 | 0.40 1 |
| S-Ferritin (ug/L) | 40.7 ± 1.82 3 | 40.2 ± 1.90 3 | 0.88 1 |
| S-Urea (mmol/L) | 2.33 ± 0.66 | 3.26 ± 0.87 | <0.001 1 |
| S-Folate (nmol/L) | 42 (37.8–45) 4 | 39 (34.8–45) 4 | <0.05 2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Johansson, U.; Öhlund, I.; Hernell, O.; Lönnerdal, B.; Lindberg, L.; Lind, T. Protein-Reduced Complementary Foods Based on Nordic Ingredients Combined with Systematic Introduction of Taste Portions Increase Intake of Fruits and Vegetables in 9 Month Old Infants: A Randomised Controlled Trial. Nutrients 2019, 11, 1255. https://doi.org/10.3390/nu11061255
Johansson U, Öhlund I, Hernell O, Lönnerdal B, Lindberg L, Lind T. Protein-Reduced Complementary Foods Based on Nordic Ingredients Combined with Systematic Introduction of Taste Portions Increase Intake of Fruits and Vegetables in 9 Month Old Infants: A Randomised Controlled Trial. Nutrients. 2019; 11(6):1255. https://doi.org/10.3390/nu11061255
Chicago/Turabian StyleJohansson, Ulrica, Inger Öhlund, Olle Hernell, Bo Lönnerdal, Lene Lindberg, and Torbjörn Lind. 2019. "Protein-Reduced Complementary Foods Based on Nordic Ingredients Combined with Systematic Introduction of Taste Portions Increase Intake of Fruits and Vegetables in 9 Month Old Infants: A Randomised Controlled Trial" Nutrients 11, no. 6: 1255. https://doi.org/10.3390/nu11061255
APA StyleJohansson, U., Öhlund, I., Hernell, O., Lönnerdal, B., Lindberg, L., & Lind, T. (2019). Protein-Reduced Complementary Foods Based on Nordic Ingredients Combined with Systematic Introduction of Taste Portions Increase Intake of Fruits and Vegetables in 9 Month Old Infants: A Randomised Controlled Trial. Nutrients, 11(6), 1255. https://doi.org/10.3390/nu11061255






