Habitual Breakfast Patterns Do Not Influence Appetite and Satiety Responses in Normal vs. High-Protein Breakfasts in Overweight Adolescent Girls
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Study Participants
2.3. Breakfast Patterns
2.4. Specific Testing Procedures on Day 4 of Each Pattern
2.5. Appetite and Satiety Questionnaires
2.6. Repeated Blood Sampling and Hormonal Analyses
2.7. Data and Statistical Analyses
3. Results
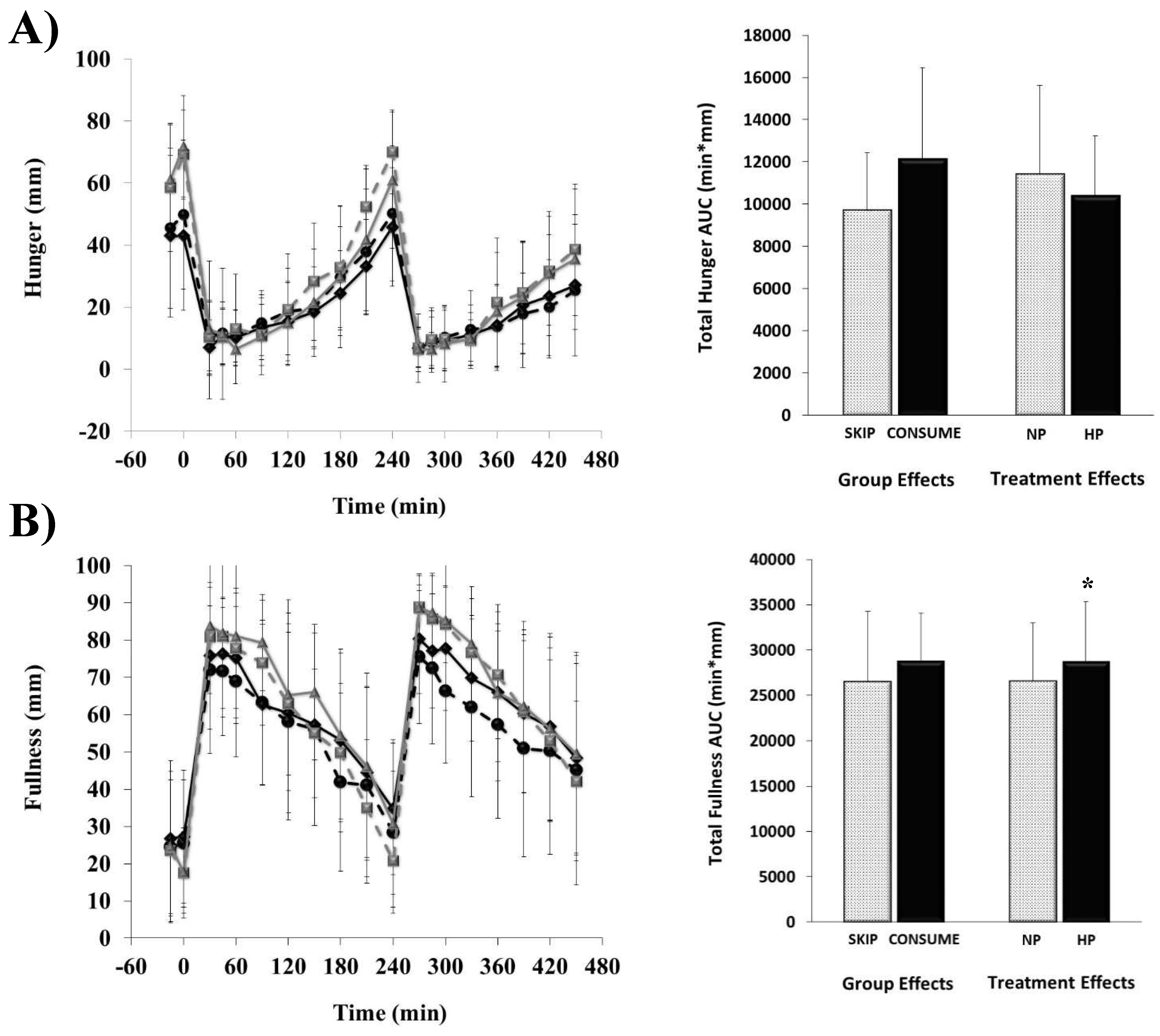
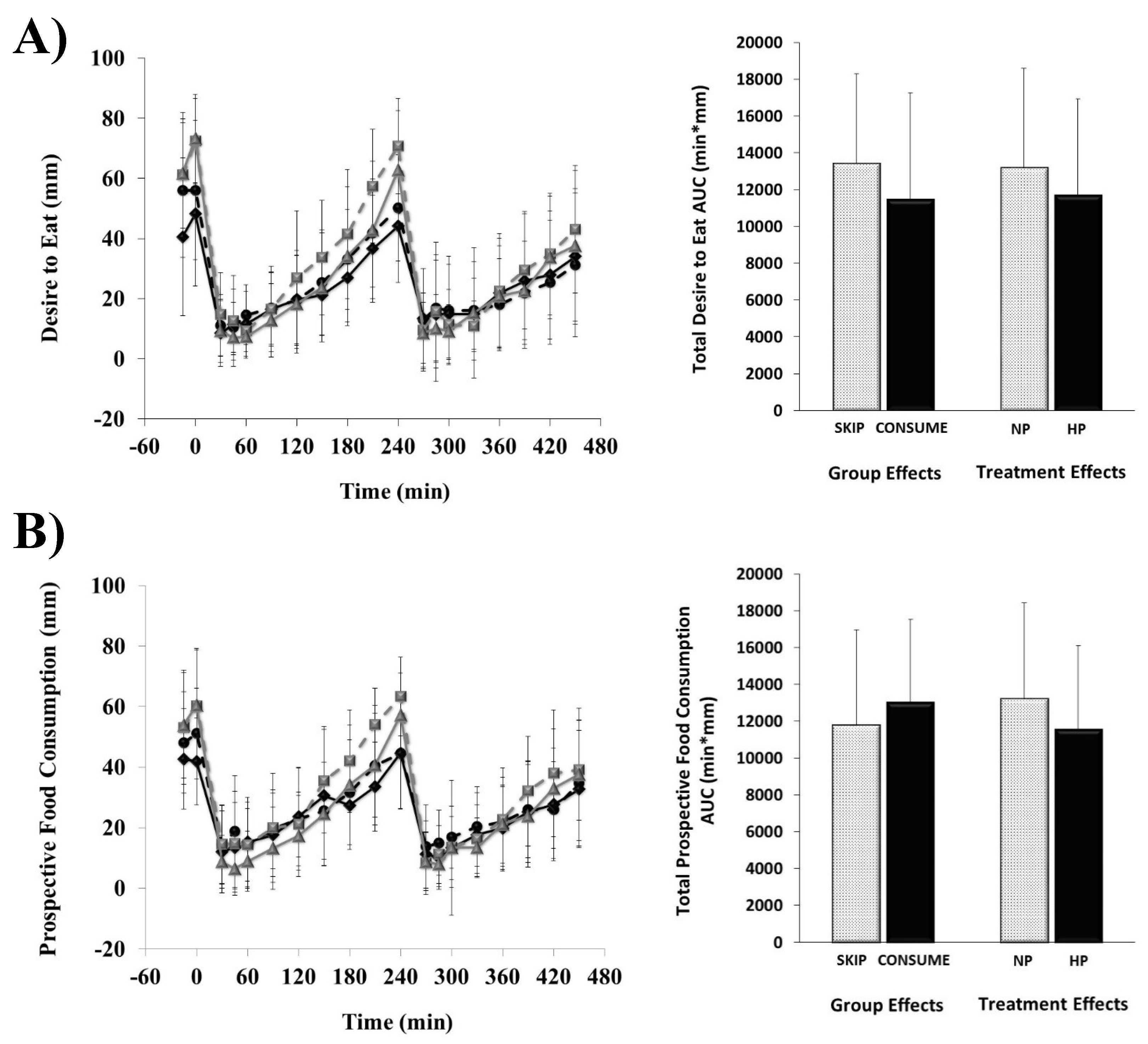
3.1. Perceived Appetite Responses
3.2. Hormonal Responses
3.3. Energy Intake Responses
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ogden, C.L.; Carroll, M.D.; Lawman, H.G.; Fryar, C.D.; Kruszon-Moran, D.; Kit, B.K.; Flegal, K.M. Trends in Obesity Prevalence Among Children and Adolescents in the United States, 1988–1994 Through 2013–2014. JAMA 2016, 315, 2292–2299. [Google Scholar] [CrossRef]
- National Center for Health Statistics. Health, United States, 2016: With Chartbook on Long-Term Trends in Health; Government Printing Offic: Hyattsville, MD, USA, 2017.
- Timlin, M.T.; Pereira, M.A.; Story, M.; Neumark-Sztainer, D. Breakfast eating and weight change in a 5-year prospective analysis of adolescents: Project EAT (Eating Among Teens). Pediatrics 2008, 121, e638–e645. [Google Scholar] [CrossRef] [PubMed]
- Rampersaud, G.C.; Pereira, M.A.; Girard, B.L.; Adams, J.; Metzl, J.D. Breakfast habits, nutritional status, body weight, and academic performance in children and adolescents. J. Am. Diet. Assoc. 2005, 105, 743–760. [Google Scholar] [CrossRef] [PubMed]
- Leidy, H.J.; Gwin, J.A.; Roenfeldt, C.A.; Zino, A.Z.; Shafer, R.S. Evaluating the Intervention-Based Evidence Surrounding the Causal Role of Breakfast on Markers of Weight Management, with Specific Focus on Breakfast Composition and Size. Adv. Nutr. 2016, 7, 563S–575S. [Google Scholar] [CrossRef]
- Gwin, J.A.; Leidy, H.J. Breakfast Consumption Augments Appetite, Eating Behavior, and Exploratory Markers of Sleep Quality Compared with Skipping Breakfast in Healthy Young Adults. Curr. Dev. Nutr. 2018, 2, nzy074. [Google Scholar] [CrossRef]
- Leidy, H.J.; Ortinau, L.C.; Douglas, S.M.; Hoertel, H.A. Beneficial effects of a higher-protein breakfast on the appetitive, hormonal, and neural signals controlling energy intake regulation in overweight/obese, “breakfast-skipping,” late-adolescent girls. Am. J. Clin. Nutr. 2013, 97, 677–688. [Google Scholar] [CrossRef]
- Alwattar, A.Y.; Thyfault, J.P.; Leidy, H.J. The effect of breakfast type and frequency of consumption on glycemic response in overweight/obese late adolescent girls. Eur. J. Clin. Nutr. 2015, 69, 885–890. [Google Scholar] [CrossRef] [PubMed]
- Leidy, H.J.; Hoertel, H.A.; Douglas, S.M.; Higgins, K.A.; Shafer, R.S. A high-protein breakfast prevents body fat gain, through reductions in daily intake and hunger, in “Breakfast skipping” adolescents. Obesity (Silver Spring) 2015, 23, 1761–1764. [Google Scholar] [CrossRef] [PubMed]
- Bauer, L.B.; Reynolds, L.J.; Douglas, S.M.; Kearney, M.L.; Hoertel, H.A.; Shafer, R.S.; Thyfault, J.P.; Leidy, H.J. A pilot study examining the effects of consuming a high-protein vs normal-protein breakfast on free-living glycemic control in overweight/obese ‘breakfast skipping’ adolescents. Int. J. Obes. (Lond.) 2015, 39, 1421–1424. [Google Scholar] [CrossRef] [PubMed]
- Schlundt, D.G.; Hill, J.O.; Sbrocco, T.; Pope-Cordle, J.; Sharp, T. The role of breakfast in the treatment of obesity: A randomized clinical trial. Am. J. Clin. Nutr. 1992, 55, 645–651. [Google Scholar] [CrossRef]
- Thomas, E.A.; Higgins, J.; Bessesen, D.H.; McNair, B.; Cornier, M.A. Usual breakfast eating habits affect response to breakfast skipping in overweight women. Obesity (Silver Spring) 2015, 23, 750–759. [Google Scholar] [CrossRef] [PubMed]
- Long, S.J.; Jeffcoat, A.R.; Millward, D.J. Effect of habitual dietary-protein intake on appetite and satiety. Appetite 2000, 35, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, G.; Moreno, L.A.; Sarria, A.; Fleta, J.; Bueno, M. Resting energy expenditure in children and adolescents: Agreement between calorimetry and prediction equations. Clin. Nutr. 2002, 21, 255–260. [Google Scholar] [CrossRef]
- Blundell, J.; de Graaf, C.; Hulshof, T.; Jebb, S.; Livingstone, B.; Lluch, A.; Mela, D.; Salah, S.; Schuring, E.; van der Knaap, H.; et al. Appetite control: Methodological aspects of the evaluation of foods. Obes. Rev. 2010, 11, 251–270. [Google Scholar] [CrossRef] [PubMed]
- Flint, A.; Raben, A.; Blundell, J.E.; Astrup, A. Reproducibility, power and validity of visual analogue scales in assessment of appetite sensations in single test meal studies. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 38–48. [Google Scholar] [CrossRef]
- Brown, A.W.; Bohan Brown, M.M.; Allison, D.B. Belief beyond the evidence: Using the proposed effect of breakfast on obesity to show 2 practices that distort scientific evidence. Am. J. Clin. Nutr. 2013, 98, 1298–1308. [Google Scholar] [CrossRef]
- Natalucci, G.; Riedl, S.; Gleiss, A.; Zidek, T.; Frisch, H. Spontaneous 24-h ghrelin secretion pattern in fasting subjects: Maintenance of a meal-related pattern. Eur. J. Endocrinol. 2005, 152, 845–850. [Google Scholar] [CrossRef] [PubMed]
- Frecka, J.M.; Mattes, R.D. Possible entrainment of ghrelin to habitual meal patterns in humans. Am. J. Physiol. Gastrointest. Liver Physiol. 2008, 294, G699–G707. [Google Scholar] [CrossRef]
- Gwin, J.A.; Leidy, H.J. A Review of the Evidence Surrounding the Effects of Breakfast Consumption on Mechanisms of Weight Management. Adv. Nutr. 2018, 9, 717–725. [Google Scholar] [CrossRef]
- U.S. Department of Agriculture; Agricultural Research Service; Beltsville Human Nutrition Research Center; Food Surveys Research Group (Beltsville, MD); U.S. Department of Health and Human Services; Centers for Disease Control and Prevention; National Center for Health Statistics (Hyattsville, MD). What We Eat in America, NHANGES 2007–2010. Available online: https://data.nal.usda.gov/dataset/what-we-eat-america-wweia-database (accessed on 23 January 2019).
- Leidy, H.J.; Clifton, P.M.; Astrup, A.; Wycherley, T.P.; Westerterp-Plantenga, M.S.; Luscombe-Marsh, N.D.; Woods, S.C.; Mattes, R.D. The role of protein in weight loss and maintenance. Am. J. Clin. Nutr. 2015, 101, 1320S–1329S. [Google Scholar] [CrossRef]
- Batterham, R.L.; Cowley, M.A.; Small, C.J.; Herzog, H.; Cohen, M.A.; Dakin, C.L.; Wren, A.M.; Brynes, A.E.; Low, M.J.; Ghatei, M.A.; et al. Gut hormone PYY(3-36) physiologically inhibits food intake. Nature 2002, 418, 650–654. [Google Scholar] [CrossRef]
- Mars, M.; Stafleu, A.; de Graaf, C. Use of satiety peptides in assessing the satiating capacity of foods. Physiol. Behav. 2012, 105, 483–488. [Google Scholar] [CrossRef]
- Hoertel, H.A.; Will, M.J.; Leidy, H.J. A randomized crossover, pilot study examining the effects of a normal protein vs. high protein breakfast on food cravings and reward signals in overweight/obese “breakfast skipping”, late-adolescent girls. Nutr. J. 2014, 13, 80. [Google Scholar] [CrossRef]
- Drapeau, V.; King, N.; Hetherington, M.; Doucet, E.; Blundell, J.; Tremblay, A. Appetite sensations and satiety quotient: Predictors of energy intake and weight loss. Appetite 2007, 48, 159–166. [Google Scholar] [CrossRef]
- Mattes, R. Hunger Ratings Are Not a Valid Proxy Measure of Reported Food-Intake in Humans. Appetite 1990, 15, 103–113. [Google Scholar] [CrossRef]
- Holt, G.M.; Owen, L.J.; Till, S.; Cheng, Y.; Grant, V.A.; Harden, C.J.; Corfe, B.M. Systematic Literature Review Shows That Appetite Rating does Not Predict Energy Intake. Crit. Rev. Food Sci. Nutr. 2007, 57, 3577–3582. [Google Scholar] [CrossRef]
 ), SKIP-HP (
), SKIP-HP ( ), CONSUME-NP (
), CONSUME-NP ( ), CONSUME-HP (
), CONSUME-HP ( ).
).
 ), SKIP-HP (
), SKIP-HP ( ), CONSUME-NP (
), CONSUME-NP ( ), CONSUME-HP (
), CONSUME-HP ( ).
).
 ), SKIP-HP (
), SKIP-HP ( ), CONSUME-NP (
), CONSUME-NP ( ), CONSUME-HP (
), CONSUME-HP ( ).
).
 ), SKIP-HP (
), SKIP-HP ( ), CONSUME-NP (
), CONSUME-NP ( ), CONSUME-HP (
), CONSUME-HP ( ).
).
 ), SKIP-HP (
), SKIP-HP ( ), CONSUME-NP (
), CONSUME-NP ( ), CONSUME-HP (
), CONSUME-HP ( ).
).
 ), SKIP-HP (
), SKIP-HP ( ), CONSUME-NP (
), CONSUME-NP ( ), CONSUME-HP (
), CONSUME-HP ( ).
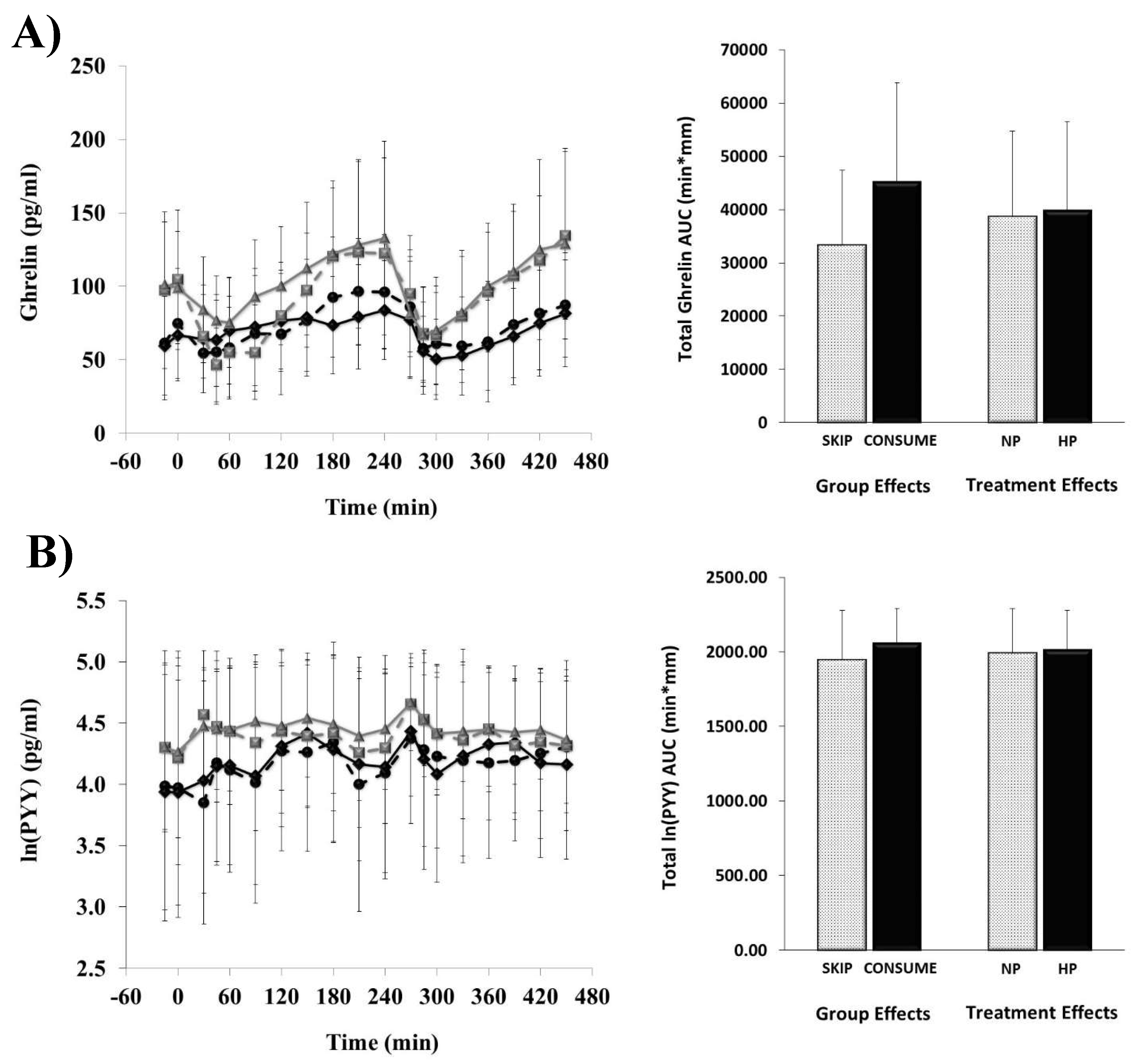
).
| SKIP (n = 19) | CONSUME (n = 18) | |
|---|---|---|
| Age (year) | 19 ± 1 | 19 ± 1 |
| Height (cm) | 166 ± 5.4 | 167 ± 7.0 |
| Weight (kg) | 80.3 ± 9.9 | 80.3 ± 9.9 |
| BMI (kg/m2) | 29.0 ± 3.8 | 28.9 ± 2.9 |
| Frequency of breakfast consumption (no/week) | 1 ± 1 | 6 ± 1 |
| First eating or drinking occasion of the day | 12:30 ± 0:15 p.m. | 8:15 ± 0:10 a.m. |
| SKIP | CONSUME | Group Effect | Protein Effect | Group × Protein | |||
|---|---|---|---|---|---|---|---|
| NP | HP | NP | HP | ||||
| Energy (kcal) | 2360 ± 138 | 2312 ± 132 | 2554 ± 145 | 2542 ± 138 | 0.49 | 0.22 | 0.29 |
| Protein (g) | 76 ± 4 | 95 ± 3 | 79 ± 4 | 101 ± 3 | 0.92 | <0.01 | 0.15 |
| Carbohydrates (g) | 346 ± 20 | 325 ± 21 | 366 ± 21 | 343 ± 22 | 0.71 | <0.01 | 0.14 |
| Fat (g) | 65 ± 5 | 61 ± 5 | 79 ± 6 | 79 ± 6 | 0.12 | <0.01 | <0.01 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Douglas, S.M.; Byers, A.W.; Leidy, H.J. Habitual Breakfast Patterns Do Not Influence Appetite and Satiety Responses in Normal vs. High-Protein Breakfasts in Overweight Adolescent Girls. Nutrients 2019, 11, 1223. https://doi.org/10.3390/nu11061223
Douglas SM, Byers AW, Leidy HJ. Habitual Breakfast Patterns Do Not Influence Appetite and Satiety Responses in Normal vs. High-Protein Breakfasts in Overweight Adolescent Girls. Nutrients. 2019; 11(6):1223. https://doi.org/10.3390/nu11061223
Chicago/Turabian StyleDouglas, Steve M., Adam W. Byers, and Heather J. Leidy. 2019. "Habitual Breakfast Patterns Do Not Influence Appetite and Satiety Responses in Normal vs. High-Protein Breakfasts in Overweight Adolescent Girls" Nutrients 11, no. 6: 1223. https://doi.org/10.3390/nu11061223
APA StyleDouglas, S. M., Byers, A. W., & Leidy, H. J. (2019). Habitual Breakfast Patterns Do Not Influence Appetite and Satiety Responses in Normal vs. High-Protein Breakfasts in Overweight Adolescent Girls. Nutrients, 11(6), 1223. https://doi.org/10.3390/nu11061223




