The Effect of Vitamin C (Ascorbic Acid) in the Treatment of Patients with Cancer: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Objectives
- — Vitamin C administration is more effective in the treatment of cancer than placebo or no treatment in susceptible populations.
- — Different routes of vitamin C administration (intravenous/oral) may differ in effectiveness in treating cancer.
2.2. Protocol and Registration
2.3. Eligibility Criteria
2.3.1. Types of Studies
2.3.2. Types of Participant
2.3.3. Types of Intervention
2.3.4. Types of Outcome Measures
2.4. Literature Search
2.5. Data Collection and Analysis
3. Results
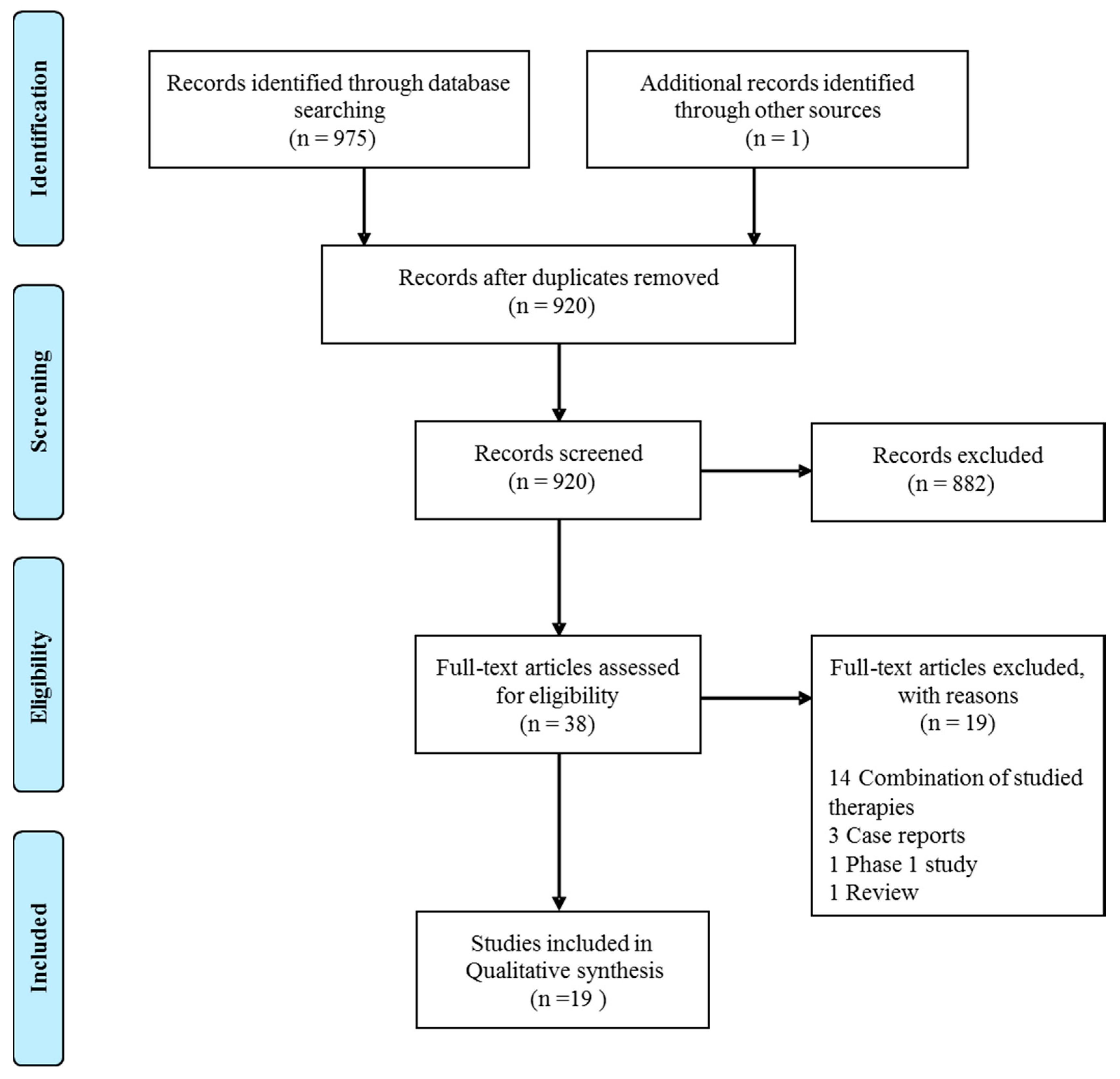
3.1. Study Selection
3.2. Study Characteristics
3.2.1. Participants
3.2.2. Intervention
3.2.3. Other Treatments
3.2.4. Outcome Measures
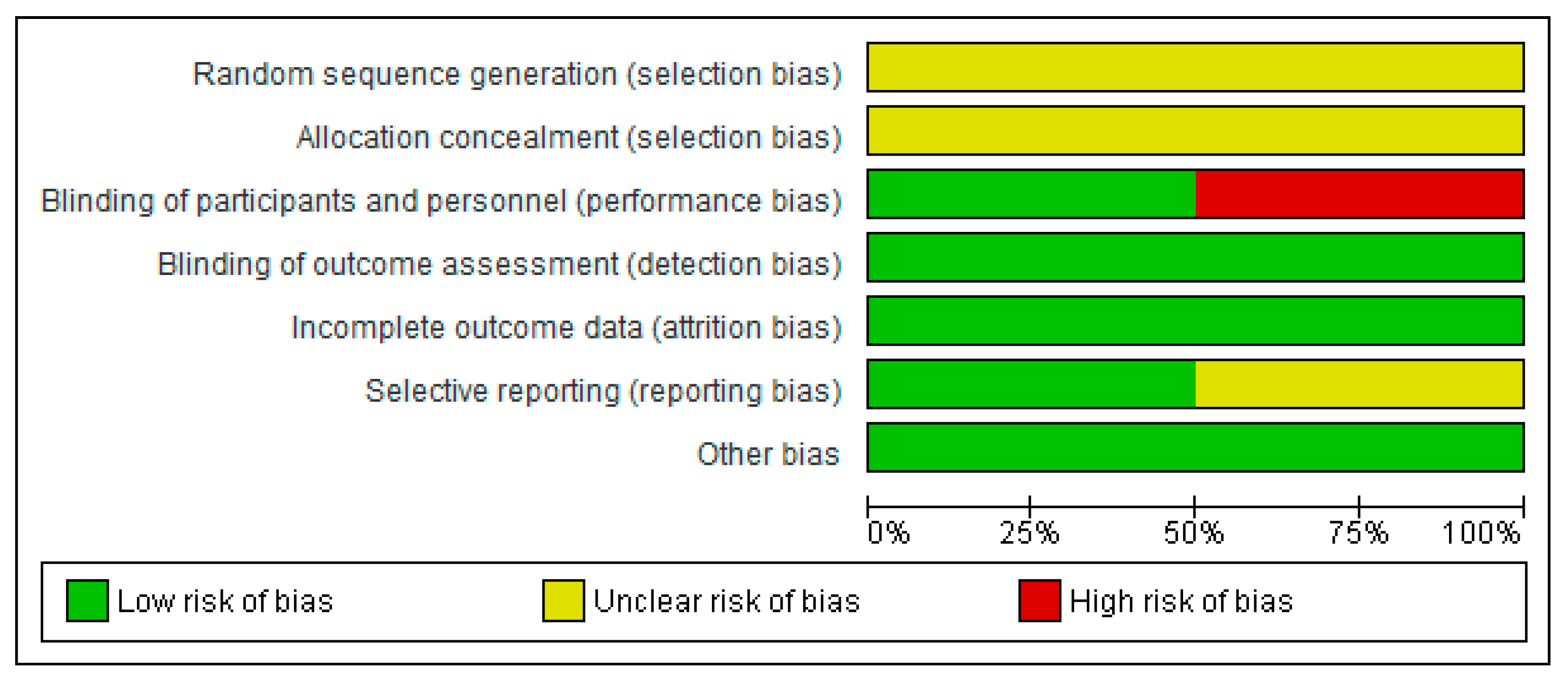
3.3. Risk of Bias in Included Studies
3.4. Results of Individual Studies
3.4.1. Overall Survival
Intravenous versus Oral Vitamin C
3.4.2. Clinical Response
Intravenous Versus Oral Vitamin C
3.4.3. Quality of Life and Performance Status
Intravenous Versus Oral Vitamin C
3.4.4. Safety and Toxicity
3.4.5. Vitamin C Concentrations after Supplementation
4. Discussion
4.1. Summary of the Main Results
4.2. Quality of the Evidence
4.3. Potential Biases in the Review Process
5. Conclusions
Supplementary Materials
Funding
Conflicts of Interest
References
- Cameron, E.; Pauling, L. Supplemental ascorbate in the supportive treatment of cancer: Prolongation of survival times in terminal human cancer. Proc. Natl. Acad. Sci. USA 1976, 73, 3685–3689. [Google Scholar] [CrossRef] [PubMed]
- Cameron, E.; Campbell, A. The orthomolecular treatment of cancer. Ii. Clinical trial of high-dose ascorbic acid supplements in advanced human cancer. Chem. Biol. Interact. 1974, 9, 285–315. [Google Scholar] [CrossRef]
- Creagan, E.T.; Moertel, C.G.; O’Fallon, J.R.; Schutt, A.J.; O’Connell, M.J.; Rubin, J.; Frytak, S. Failure of high-dose vitamin c (ascorbic acid) therapy to benefit patients with advanced cancer. A controlled trial. N. Engl. J. Med. 1979, 301, 687–690. [Google Scholar] [CrossRef] [PubMed]
- Padayatty, S.J.; Sun, H.; Wang, Y.; Riordan, H.D.; Hewitt, S.M.; Katz, A.; Wesley, R.A.; Levine, M. Vitamin c pharmacokinetics: Implications for oral and intravenous use. Ann. Intern. Med. 2004, 140, 533–537. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Espey, M.G.; Sun, A.Y.; Pooput, C.; Kirk, K.L.; Krishna, M.C.; Khosh, D.B.; Drisko, J.; Levine, M. Pharmacologic doses of ascorbate act as a prooxidant and decrease growth of aggressive tumor xenografts in mice. Proc. Natl. Acad. Sci. USA 2008, 105, 11105–11109. [Google Scholar] [CrossRef] [PubMed]
- Young, J.I.; Zuchner, S.; Wang, G. Regulation of the epigenome by vitamin c. Annu. Rev. Nutr. 2015, 35, 545–564. [Google Scholar] [CrossRef] [PubMed]
- Kassouf, W.; Highshaw, R.; Nelkin, G.M.; Dinney, C.P.; Kamat, A.M. Vitamins C and K3 sensitize human urothelial tumors to gemcitabine. J. Urol. 2006, 176, 1642–1647. [Google Scholar] [CrossRef]
- Bober, P.; Alexovic, M.; Talian, I.; Tomkova, Z.; Viscorova, Z.; Benckova, M.; Andrasina, I.; Ciccocioppo, R.; Petrovic, D.; Adamek, M.; et al. Proteomic analysis of the vitamin C effect on the doxorubicin cytotoxicity in the MCF-7 breast cancer cell line. J. Cancer Res. Clin. Oncol. 2017, 143, 35–42. [Google Scholar] [CrossRef]
- Jung, S.A.; Lee, D.H.; Moon, J.H.; Hong, S.W.; Shin, J.S.; Hwang, I.Y.; Shin, Y.J.; Kim, J.H.; Gong, E.Y.; Kim, S.M.; et al. L-Ascorbic acid can abrogate SVCT-2-dependent cetuximab resistance mediated by mutant KRAS in human colon cancer cells. Free. Radic. Biol. Med. 2016, 95, 200–208. [Google Scholar] [CrossRef]
- Kalita, S.; Verma, A.K.; Prasad, S.B. Chlorambucil and ascorbic acid-mediated anticancer activity and hematological toxicity in Dalton’s ascites lymphoma-bearing mice. Indian J. Exp. Biol. 2014, 52, 112–124. [Google Scholar]
- Kurbacher, C.M.; Wagner, U.; Kolster, B.; Andreotti, P.E.; Krebs, D.; Bruckner, H.W. Ascorbic acid (vitamin C) improves the antineoplastic activity of doxorubicin, cisplatin, and paclitaxel in human breast carcinoma cells in vitro. Cancer Lett. 1996, 103, 183–189. [Google Scholar] [CrossRef]
- Huijskens, M.J.; Walczak, M.; Koller, N.; Briede, J.J.; Senden-Gijsbers, B.L.; Schnijderberg, M.C.; Bos, G.M.; Germeraad, W.T. Technical advance: ascorbic acid induces development of double-positive T cells from human hematopoietic stem cells in the absence of stromal cells. J. leukoc. biol. 2014, 96, 1165–1175. [Google Scholar] [CrossRef]
- Huijskens, M.J.; Walczak, M.; Sarkar, S.; Atrafi, F.; Senden-Gijsbers, B.L.; Tilanus, M.G.; Bos, G.M.; Wieten, L.; Germeraad, W.T. Ascorbic acid promotes proliferation of natural killer cell populations in culture systems applicable for natural killer cell therapy. Cytotherapy 2015, 17, 613–620. [Google Scholar] [CrossRef]
- Van Gorkom, G.N.Y.; Klein Wolterink, R.G.J.; Van Elssen, C.; Wieten, L.; Germeraad, W.T.V.; Bos, G.M.J. Influence of Vitamin C on Lymphocytes: An Overview. Antioxidants 2018, 7, 41. [Google Scholar] [CrossRef]
- Huijskens, M.J.; Wodzig, W.K.; Walczak, M.; Germeraad, W.T.; Bos, G.M. Ascorbic acid serum levels are reduced in patients with hematological malignancies. Result. Immun. 2016, 6, 8–10. [Google Scholar] [CrossRef]
- Mayland, C.R.; Bennett, M.I.; Allan, K. Vitamin C deficiency in cancer patients. Palliat. Med. 2005, 19, 17–20. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gotzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009, 6, e1000100. [Google Scholar] [CrossRef]
- Cochrane Consumers and Communication. Data Extraction Template for Included Studies. Version 1.8, updated 20 November 2016. Available online: http://cccrg.cochrane.org/author-resources (accessed on 1 August 2018).
- Higgins, J.P.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Higgins, J.P.T.; Reeves, B.C.; On behalf of the development group for ACROBAT-NRSI. A Cochrane Risk of Bias Assessment Tool: For Non-Randomized Studies of Interventions (ACROBAT-NRSI), Version 1.0.0 24 September 2014. Available online: http://www.bristol.ac.uk/media-library/sites/social-community-medicine/images/centres/cresyda/ACROBAT-NRSI%20Version%201_0_0.pdf (accessed on 1 August 2018).
- West, S.; King, V.; Carey, T.S.; Lohr, K.N.; McKoy, N.; Sutton, S.F.; Lux, L. Systems to rate the strength of scientific evidence. Evid. Rep. Technol. Assess. 2002, 47, 1–11. [Google Scholar]
- Bazzan, A.J.; Zabrecky, G.; Wintering, N.; Newberg, A.B.; Monti, D.A. Retrospective Evaluation of Clinical Experience With Intravenous Ascorbic Acid in Patients With Cancer. Integr. Cancer Ther. 2018, 17, 912–920. [Google Scholar] [CrossRef]
- Cameron, E.; Pauling, L. Supplemental ascorbate in the supportive treatment of cancer: Reevaluation of prolongation of survival times in terminal human cancer. Proc. Natl. Acad. Sci. USA 1978, 75, 4538–4542. [Google Scholar] [CrossRef]
- Cameron, E.; Campbell, A. Innovation vs. quality control: an ’unpublishable’ clinical trial of supplemental ascorbate in incurable cancer. Med. Hypotheses 1991, 36, 185–189. [Google Scholar] [CrossRef]
- Hoffer, L.J.; Robitaille, L.; Zakarian, R.; Melnychuk, D.; Kavan, P.; Agulnik, J.; Cohen, V.; Small, D.; Miller, W.H. High-dose intravenous vitamin C combined with cytotoxic chemotherapy in patients with advanced cancer: a phase I-II clinical trial. PloS ONE 2015, 10, e0120228. [Google Scholar] [CrossRef]
- Gunes-Bayir, A.; Kiziltan, H.S. Palliative Vitamin C Application in Patients with Radiotherapy-Resistant Bone Metastases: A Retrospective Study. Nutr. Cancer 2015, 67, 921–925. [Google Scholar] [CrossRef]
- Ma, Y.; Chapman, J.; Levine, M.; Polireddy, K.; Drisko, J.; Chen, Q. High-dose parenteral ascorbate enhanced chemosensitivity of ovarian cancer and reduced toxicity of chemotherapy. Sci. Transl. Med. 2014, 6, 222ra218. [Google Scholar] [CrossRef]
- Mikirova, N.; Casciari, J.; Rogers, A.; Taylor, P. Effect of high-dose intravenous vitamin C on inflammation in cancer patients. J. Transl. Med. 2012, 10, 189. [Google Scholar] [CrossRef]
- Moertel, C.G.; Fleming, T.R.; Creagan, E.T.; Rubin, J.; O’Connell, M.J.; Ames, M.M. High-dose vitamin C versus placebo in the treatment of patients with advanced cancer who have had no prior chemotherapy. A randomized double-blind comparison. The New England journal of medicine. N. Engl. J. Med. 1985, 312, 137–141. [Google Scholar] [CrossRef]
- Murata, A.; Morishige, F.; Yamaguchi, H. Prolongation of survival times of terminal cancer patients by administration of large doses of ascorbate. Int. J. Vitam. Nutr. Res. 1982, 23, 103–113. [Google Scholar]
- Nielsen, T.K.; Hojgaard, M.; Andersen, J.T.; Jorgensen, N.R.; Zerahn, B.; Kristensen, B.; Henriksen, T.; Lykkesfeldt, J.; Mikines, K.J.; Poulsen, H.E. Weekly ascorbic acid infusion in castration-resistant prostate cancer patients: A single-arm phase II trial. Transl. Androl. Urol. 2017, 6, 517–528. [Google Scholar] [CrossRef]
- Poulter, J.M.; White, W.F.; Dickerson, J.W. Ascorbic acid supplementation and five year survival rates in women with early breast cancer. Acta. Vitaminol. Enzymol. 1984, 6, 175–182. [Google Scholar]
- Riordan, H.D.; Casciari, J.J.; Gonzalez, M.J.; Riordan, N.H.; Miranda-Massari, J.R.; Taylor, P.; Jackson, J.A. A pilot clinical study of continuous intravenous ascorbate in terminal cancer patients. Puerto Rico Health Sci. J. 2005, 24, 269–276. [Google Scholar]
- Takahashi, H.M.H.; Ynagisawa, A. High-dose intravenous vitamin C improves quality of life in cancer patients. Pers. Med. Univ. 2012, 1, 49–53. [Google Scholar] [CrossRef]
- Vollbracht, C.; Schneider, B.; Leendert, V.; Weiss, G.; Auerbach, L.; Beuth, J. Intravenous vitamin C administration improves quality of life in breast cancer patients during chemo-/radiotherapy and aftercare: results of a retrospective, multicentre, epidemiological cohort study in Germany. In Vivo 2011, 25, 983–990. [Google Scholar]
- Yeom, C.H.; Jung, G.C.; Song, K.J. Changes of terminal cancer patients’ health-related quality of life after high dose vitamin C administration. J. Korean Med. Sci. 2007, 22, 7–11. [Google Scholar] [CrossRef] [PubMed]
- Zhao, H.; Zhu, H.; Huang, J.; Zhu, Y.; Hong, M.; Zhu, H.; Zhang, J.; Li, S.; Yang, L.; Lian, Y.; et al. The synergy of Vitamin C with decitabine activates TET2 in leukemic cells and significantly improves overall survival in elderly patients with acute myeloid leukemia. Leuk. Res. 2018, 66, 1–7. [Google Scholar] [CrossRef] [PubMed]
| Study | N(Vitamin C/ controls) | Type | Participants | Interven-tion | Concomitant therapy | Main endpoints | Adverse events |
|---|---|---|---|---|---|---|---|
| Bazzan 2018 [22] | 86 (86/0) | Retrospective cohort | All types of cancer in different settings | 50 - 150 g IV, at least 5 times (total 3034 doses) | 32 patients none, 54 patients chemotherapy | 20 of 40 patients improvement of fatigue, 15 of 86 patients improvement of appetite. | Mostly mild adverse events (AEs), like nausea, vomiting and discomfort at injection side (<3% of infusions). Self- limiting to time of infusions. No serious AEs (SAEs) related to vitamin C |
| Cameron 1974 [2] | 50(50/0) | Retrospective cohort | Advanced stage cancer patients | 10 g a day IV for 10 days + oral vitamin C | None | 10 minimal response, 11 growth retardation, 3 stable disease, 5 tumor regression. Less pain, reduction in ascites/pleural effusions. | Fluid retention, edema, dyspeptic symptoms, tumor hemorrhage/necrosis. |
| Cameron 1976 [1] | 1100(100/1000) | Case-control | Incurable cancer patients | 10 g a day IV for 10 days + oral vitamin C | Conventional anti-cancer treatment | Mean overall survival (OS) 210 days vs. 50 days in controls (4.2x more) | Not reported (NR) |
| Cameron 1978 [23] | 1100(100/1000) | Case-control | Terminal cancer patients | 10 g a day IV for 10 days + oral vitamin C | None | Recalculation of Cameron 1976. Average OS (7.7x more = 288 days) | NR |
| Cameron 1991 [24] | 1826(294/1532) | Case-control | Terminal cancer patients | 10 g a day IV for 10 days + oral vitamin C | None | OS 343 days vs. 180 days in controls | NR |
| Creagan 1979 [3] | 123(60/63) | RCT | Advanced stage cancer patients | 10 g a day orally | NR | Identical survival.Performance status (PS) identical. 58% vs. 63% some improvement in symptoms | Nausea, vomiting |
| Hoffer 2015 [25] | 14 (14/0) | Uncontrolled phase II | Advanced stage cancer patients | 1.5g/kg IV 2-3 times per week. | Chemothera-py | 6 transient, partly long-lasting stable disease | Edema, thirst, nausea, vomiting, headache, chills |
| Günes-Bayir 2015 [26] | 39 (15/24) | Case-control | Bone metastases from various types of cancer | 2.5 g IV a day | NR | OS 10 months vs. 2 months in controls. Decrease in pain in 9/15 vs. 5/24 in controls. PS improvement in 4/15 vs. 1/24 in controls. | 40% mild diarrhea, 30% mild oliguria |
| Ma 2014 [27] | 25 (13/12) | RCT | Newly diagnosed stage III and IV ovarian cancer after debulking | IV two times per week using a dose escalating protocol (final dose either 75 or 100g) for 12 months. | Paclitaxel and carboplatin chemotherapy | Trend in improvement OS, 25.5 months vs. 16.75 months in controls, (not significant). | Fewer chemotherapy related side effects with vitamin C, no relevant AEs of vitamin C. |
| Mikirova 2012 [28] | 45 (45/0) | Retrospective cohort | Various types of cancer, mostly metastatic | Escalate to 50 g IV 3 times per week for a median of 9 times | NR | 76% reduction in C –reactive protein, 75% reduction in prostate-specific antigen (PSA) | NR |
| Moertel 1985 [29] | 100 (51/49) | RCT | Advanced colorectal cancer | 10 g a day orally | None | Median OS 2.9 months vs. 4.1 months in controls. 7/11 symptom relief vs. 11/17 in controls | Low incidence of AEs, mild. |
| Murata 1982 [30] | 130 (111/19) | Non randomized clinical trial | Terminal cancer patients | Site 1: 6–30 g a day orally and 10–20g IV. Site 2: 0.5–3 g or 5–30 g per day orally. | NR | Site 1: average OS with high dose vitamin C 246 days vs. 43 days with low dose.Site 2: average OS with high dose vitamin C 115 days vs. 48 days in controls. Less use of narcotic drugs in vitamin C treated patients: 17% in high dose vs. 50% in low dose vs. 79% in controls. Improved state of wellbeing, improved appetite, increased mental alertness | No SAEs |
| Nielsen 2017 [31] | 23 (23/0) | Uncontrolled phase II | Chemothe-rapy-naïve metastatic castration-resistant prostate cancer | Weekly infusions for 12 weeks. Week 1: 5 g, week2: 30 g, week 3–12: 60 g + oral 500 mg/day. | None | 75% of patients PSA increase at 12 weeks, one PSA decrease of 27%. 80% unchanged PS at week 12, 2 improved, 2 worse score.Quality of life (QOL) identical baseline to week 12. | 53 AE, mostly mild and not related to vitamin C. 11 SAEs, explained by progression of prostate cancer. 2 pulmonary embolisms. |
| Poulter 1984 [32] | 66 (27/25) | Non randomized clinical trial | Newly diagnosed breast cancer | 3 g a day orally | NR | No change in survival rates | NR |
| Riordan 2005 [33] | 24 (24/0) | Uncontrolled phase II | Late stage terminal cancer, mostly colorectal (19) | 150 to 710 mg/kg/day IV for 8 weeks | None | 1 patient stable disease, all others progressive disease. | Mild: nausea (46%), edema (29%), dry mouth or skin (29%), fatigue (25%). Serious: 1 kidney stones, 1 hypokalemia. |
| Takaha-shi 2012 [34] | 60 (60/0) | Prospective cohort | Newly diagnosed cancer of various types | IV dose of 12.5–15 g, 25 g and 50 g + vitamin C orally 2–4 g a day. | Chemothera-py (n = 33), radiation therapy (n = 1), none (n = 2) | Improvement in QOL: score 44.6 before treatment vs. 53.2 at 2 weeks and 61.4 at 4 weeks. | Mild (grade 1), most often headache (8.3%), nausea (8.3%) |
| Voll-bracht 2011 [35] | 125 (53/72) | Retrospective cohort | Breast cancer stages IIa–IIIb. | IV 7.5 g once a week for at least 4 weeks | Primary surgical treatment +/- adjuvant chemotherapy +/- adjuvant radiotherapy | Reduced QOL related side effects, slight increase PS during adjuvant treatment (80% vs. 71%) and aftercare (87% vs. 78%). | None |
| Yeom 2007 [36] | 39 (39/0) | Uncontrolled phase II | Terminal cancer of various types, stage IV. | 10 g IV twice + 4 g oral vitamin C daily for a week. | NR | Health score improved from 3637 to 5537 after vitamin C. | No vitamin C supplementation stopped because of side effects. |
| Zhao 2017 [37] | 73 (39/34) | RCT | Newly diagnosis elderly with acute myeloid leukemia | 50–80 mg/kg IV during 10 days/month, at most 10 months | Decitabine, cytarabine and aclarubicin chemotherapy | Median OS 15.3 months vs. 9.3 months in controls (p = 0.039). Complete remission rate higher with Vitamin C: 84.6% vs. 70.6% after 2 courses. | Identical amount of AEs and SAEs in both groups. |
| Study | Evidence of Selection Bias/Prognostic Imbalance | Bias Due to Confounding Factors | Bias in Measurement of Outcomes | Follow-up of Participants Sufficiently Complete | Bias Due to Selection of Reported Results or due to Missing Data | Comparability of Cohorts on Important Confounding Factors |
|---|---|---|---|---|---|---|
| Cameron 1976 [1] | High risk | High risk | Moderate risk | Moderate risk | Low risk | Moderate risk |
| Cameron 1978 [23] | High risk | High risk | Moderate risk | Moderate risk | Low risk | Moderate risk |
| Cameron 1991 [24] | Moderate risk | High risk | Low risk | Moderate risk | Low risk | Moderate risk |
| Günes-Bayir 2015 [26] | Moderate risk | Moderate risk | Moderate risk | Moderate risk | Low risk | Moderate risk |
| Murata 1982 [30] | High risk | High risk | Moderate risk | Moderate risk | Moderate risk | Moderate risk |
| Poulter 1984 [32] | High risk | Moderate risk | Moderate risk | High risk | Moderate risk | Moderate risk |
| Vollbracht 2011 [35] | High risk | Moderate risk | Moderate risk | Low risk | Low risk | High risk |
| Study | Selection | Study Design | Confounders | Blinding | Data Collection Methods | Withdrawals and Drop-Outs |
|---|---|---|---|---|---|---|
| Bazzan 2018 [22] | Weak | Weak | Weak | Weak | Weak | Not applicable |
| Cameron 1974 [2] | Moderate | Weak | Weak | Weak | Moderate | Not applicable |
| Hoffer 2015 [25] | Moderate | Weak | Weak | Weak | Strong | Strong |
| Mikirova 2012 [28] | Weak | Weak | Weak | Weak | Moderate | Strong |
| Nielsen 2017 [31] | Moderate | Weak | Moderate | Weak | Strong | Strong |
| Riordan 2005 [33] | Moderate | Weak | Weak | Weak | Strong | Strong |
| Takahashi 2012 [34] | Moderate | Weak | Weak | Weak | Moderate | Strong |
| Yeom 2007 [36] | Moderate | Weak | Weak | Weak | Moderate | Strong |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
van Gorkom, G.N.Y.; Lookermans, E.L.; Van Elssen, C.H.M.J.; Bos, G.M.J. The Effect of Vitamin C (Ascorbic Acid) in the Treatment of Patients with Cancer: A Systematic Review. Nutrients 2019, 11, 977. https://doi.org/10.3390/nu11050977
van Gorkom GNY, Lookermans EL, Van Elssen CHMJ, Bos GMJ. The Effect of Vitamin C (Ascorbic Acid) in the Treatment of Patients with Cancer: A Systematic Review. Nutrients. 2019; 11(5):977. https://doi.org/10.3390/nu11050977
Chicago/Turabian Stylevan Gorkom, Gwendolyn N.Y., Eline L. Lookermans, Catharina H.M.J. Van Elssen, and Gerard M.J. Bos. 2019. "The Effect of Vitamin C (Ascorbic Acid) in the Treatment of Patients with Cancer: A Systematic Review" Nutrients 11, no. 5: 977. https://doi.org/10.3390/nu11050977
APA Stylevan Gorkom, G. N. Y., Lookermans, E. L., Van Elssen, C. H. M. J., & Bos, G. M. J. (2019). The Effect of Vitamin C (Ascorbic Acid) in the Treatment of Patients with Cancer: A Systematic Review. Nutrients, 11(5), 977. https://doi.org/10.3390/nu11050977






