The Effect of Ketoanalogues on Chronic Kidney Disease Deterioration: A Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy and Eligibility Criteria
2.2. Data Extraction
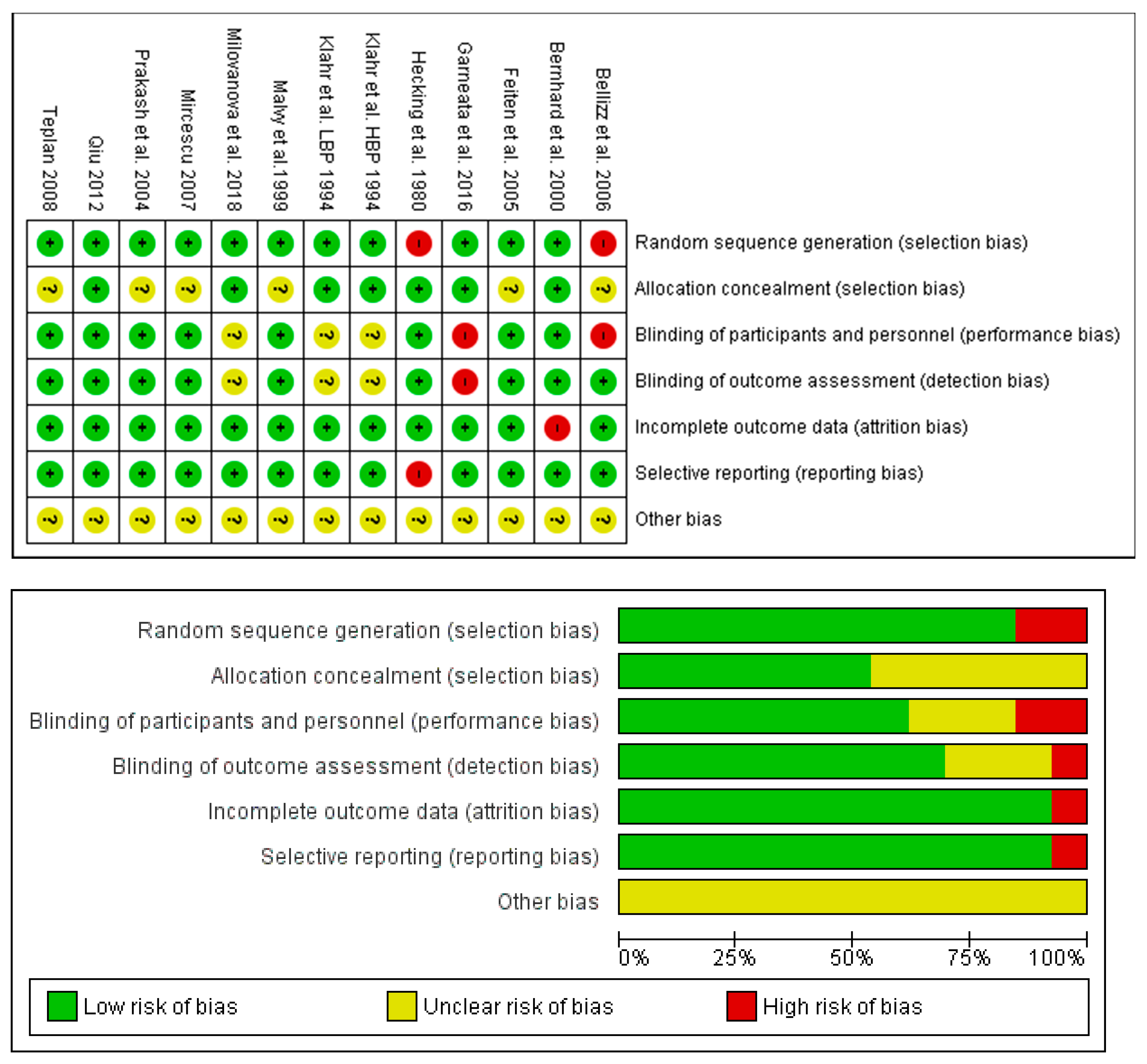
2.3. Quality Assessment
2.4. Statistical Analysis
3. Results
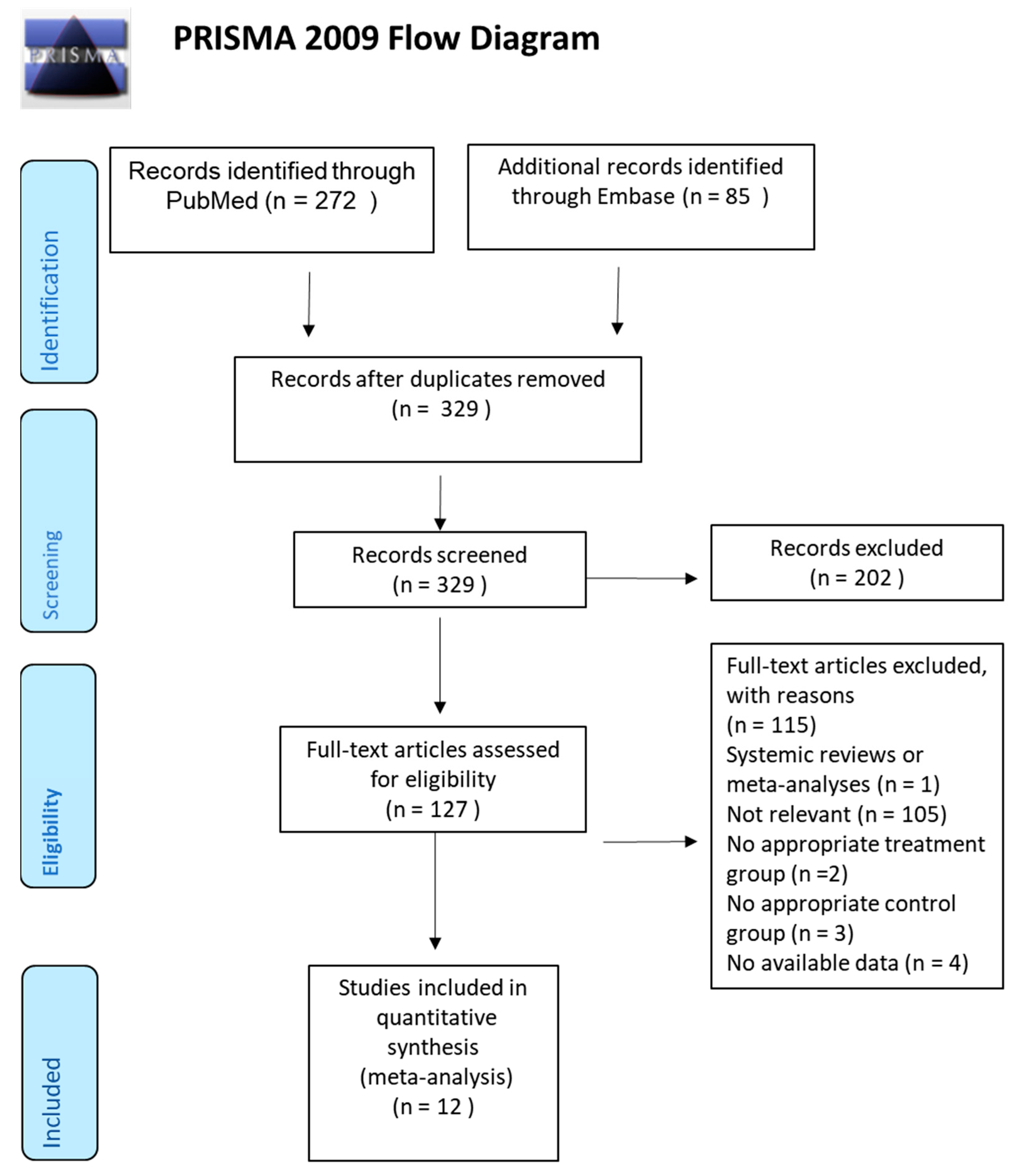
3.1. Study Selection
3.2. Renal Function
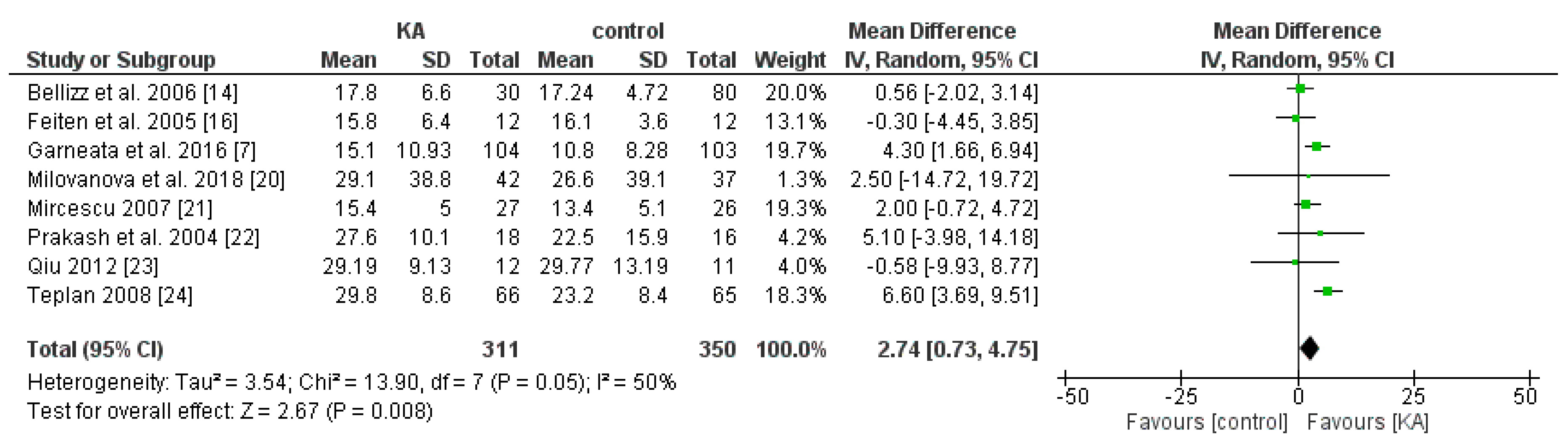
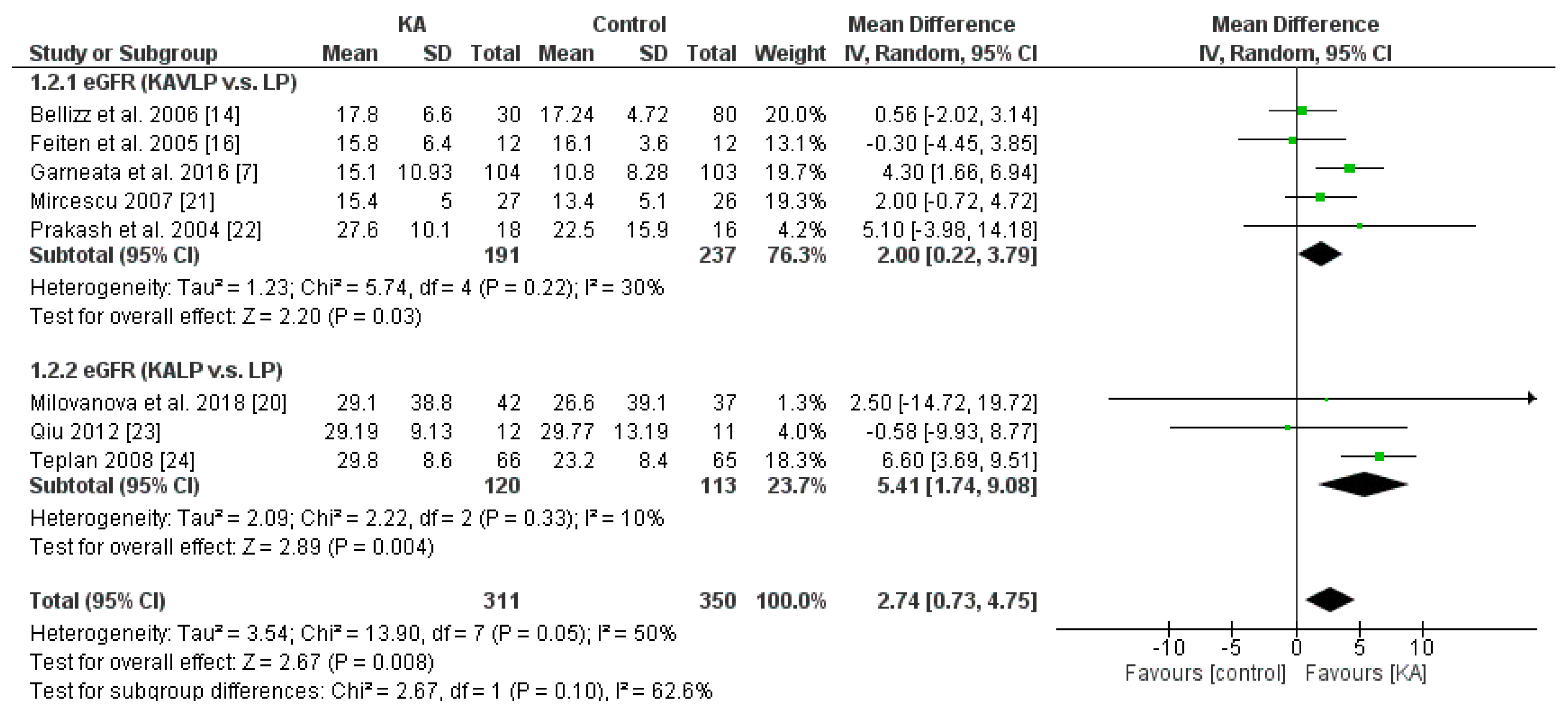
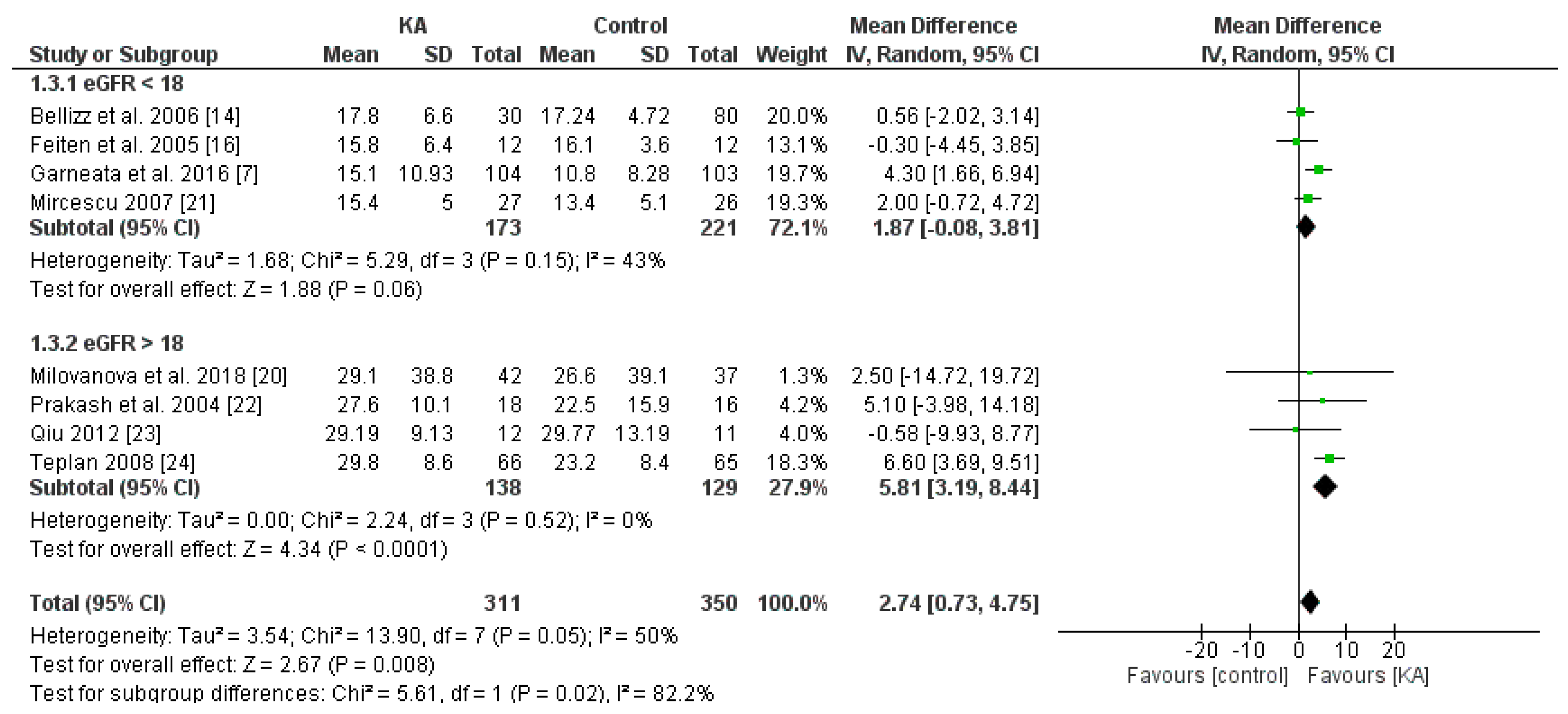
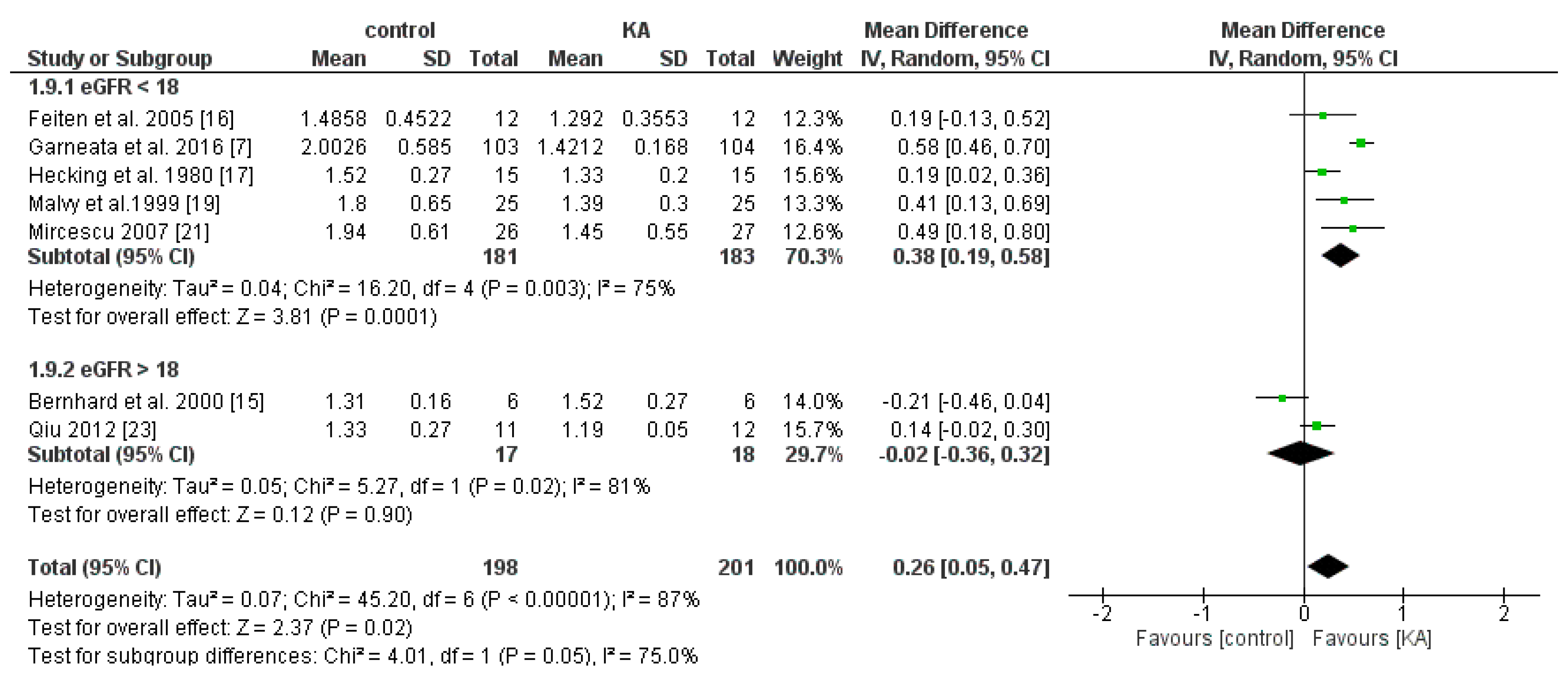
3.2.1. Effects of KA on Preventing eGFR Deterioration
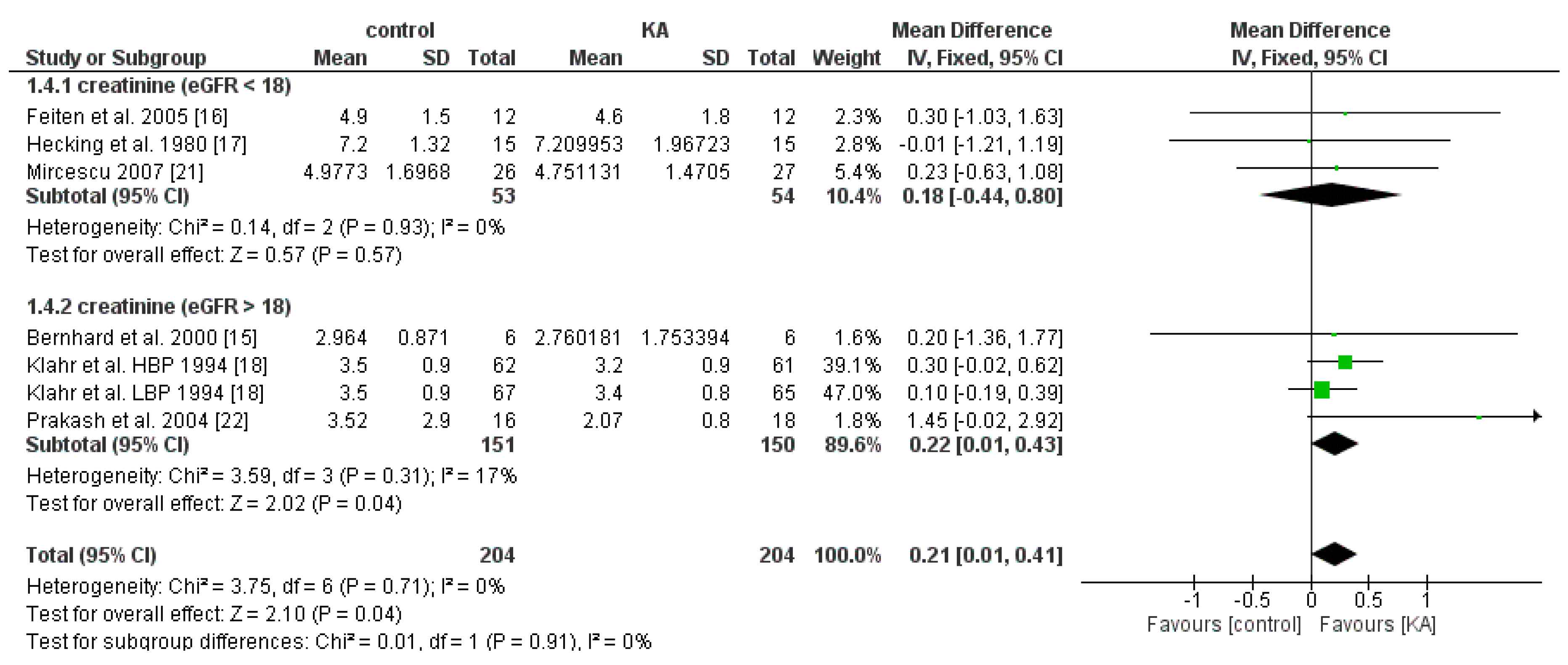
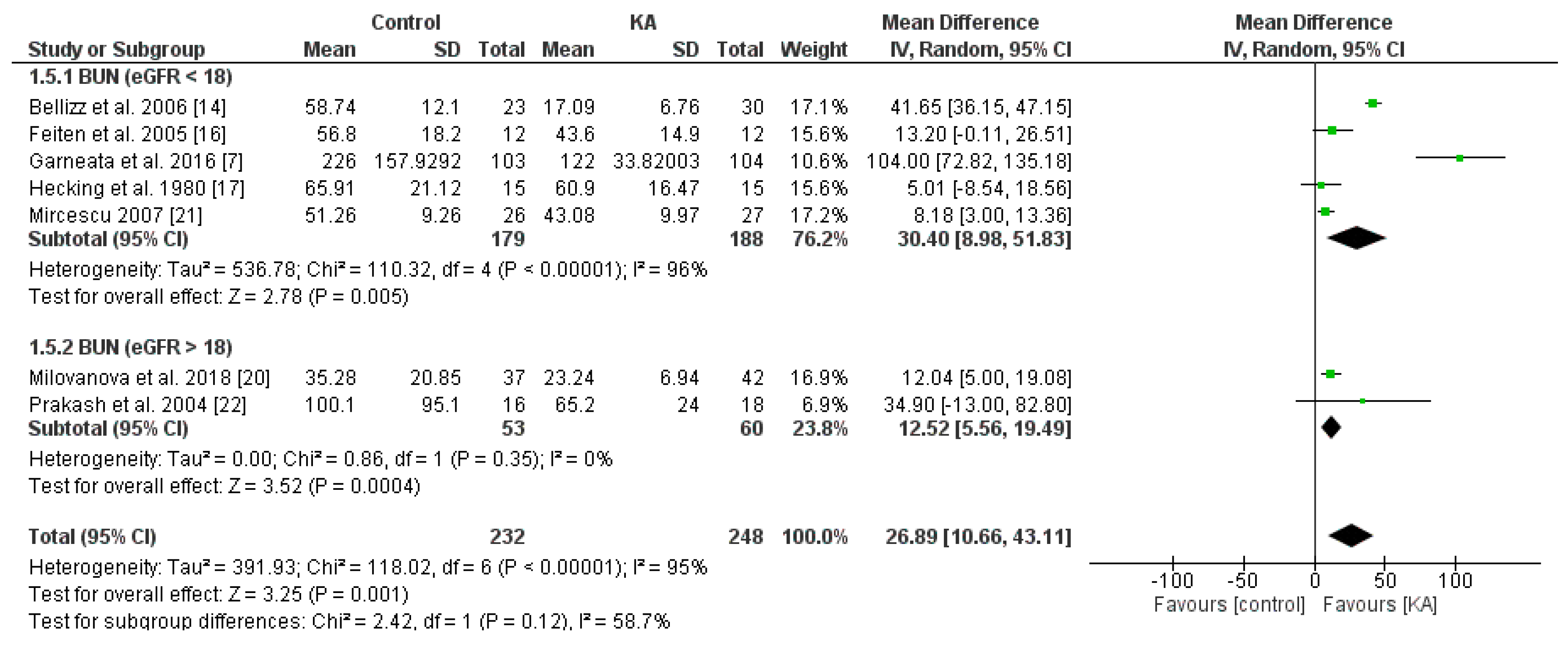
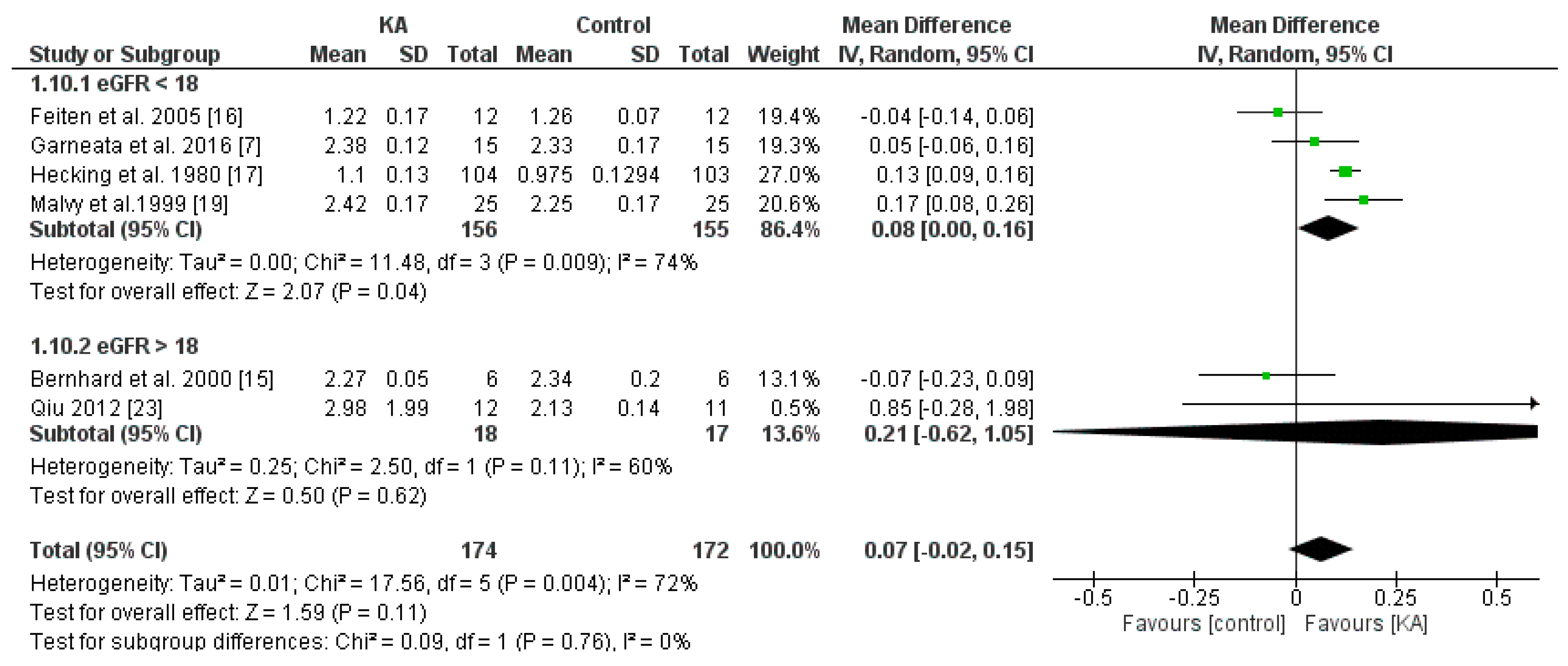
3.2.2. Effects of KA on Serum Creatinine and Blood Urea Nitrogen (BUN) Level
3.3. Status of Protein-Energy Wasting (PEW)
3.3.1. Clinical Importance
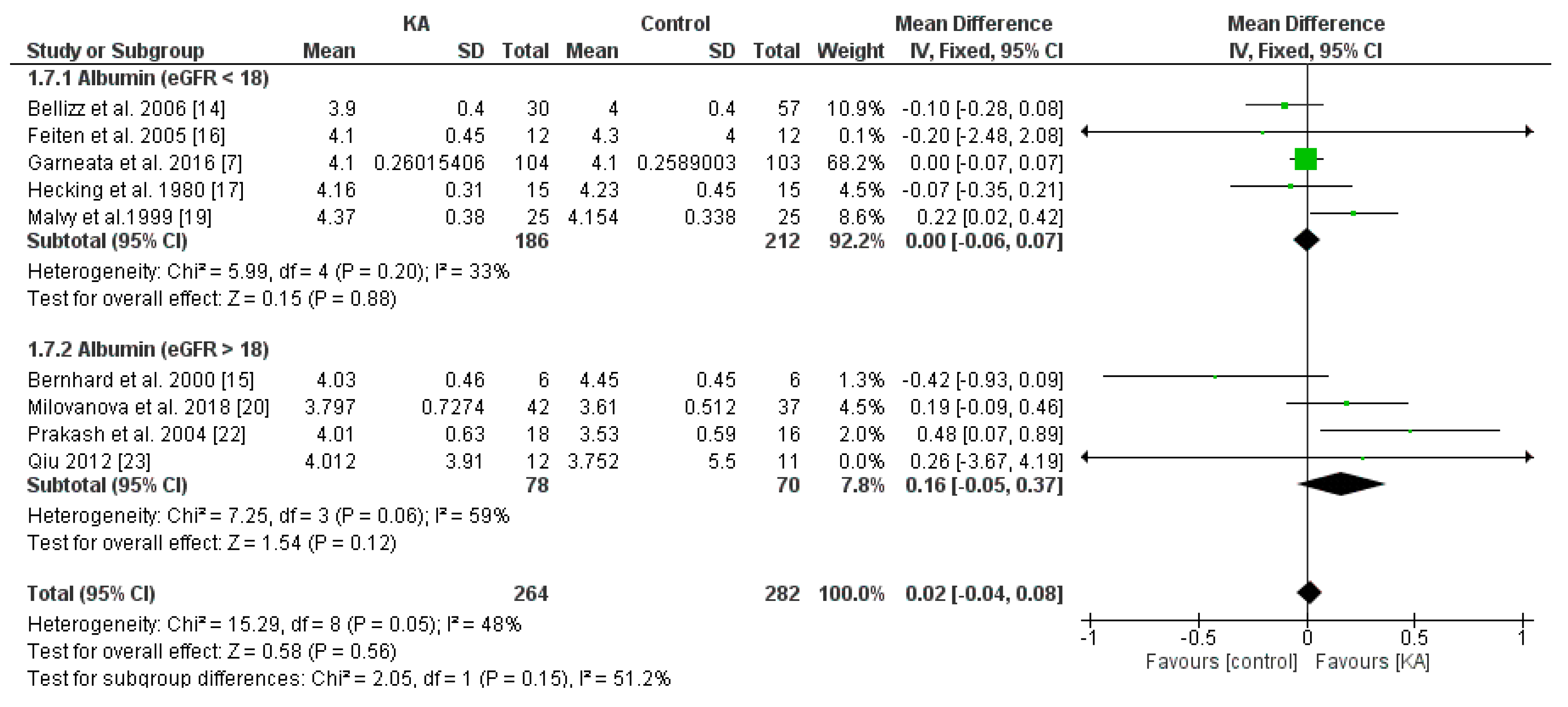
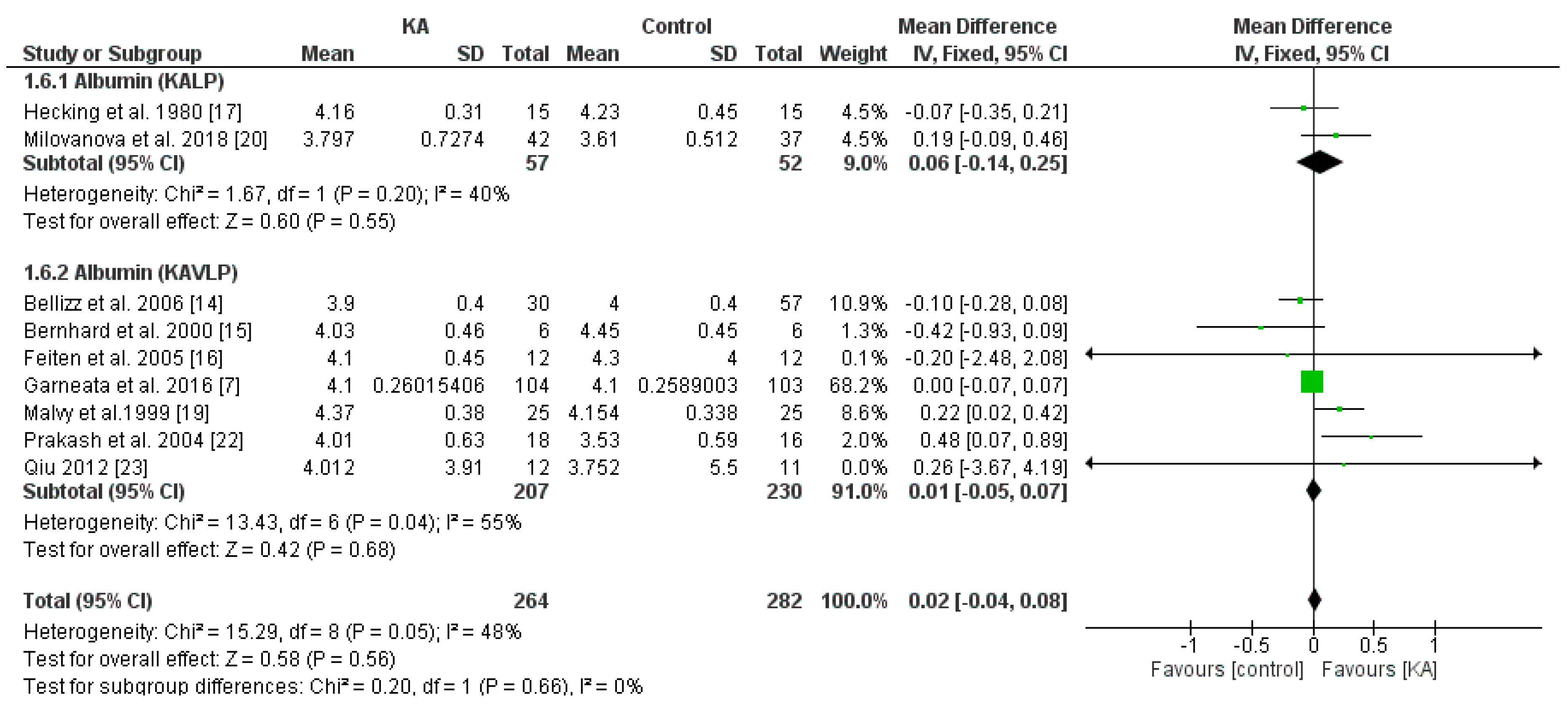
3.3.2. Effects of KA on Serum Albumin
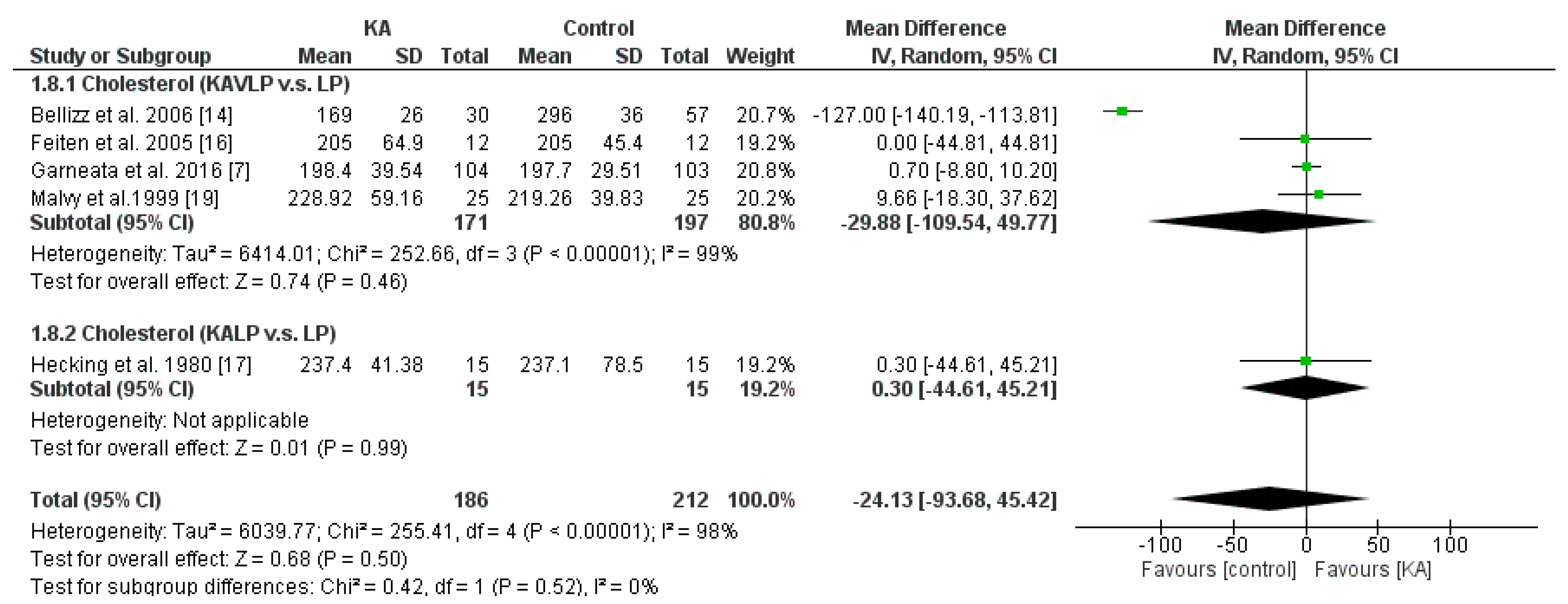
3.3.3. Effects of KA on Serum Cholesterol
3.4. Mineral and Bone Disorder
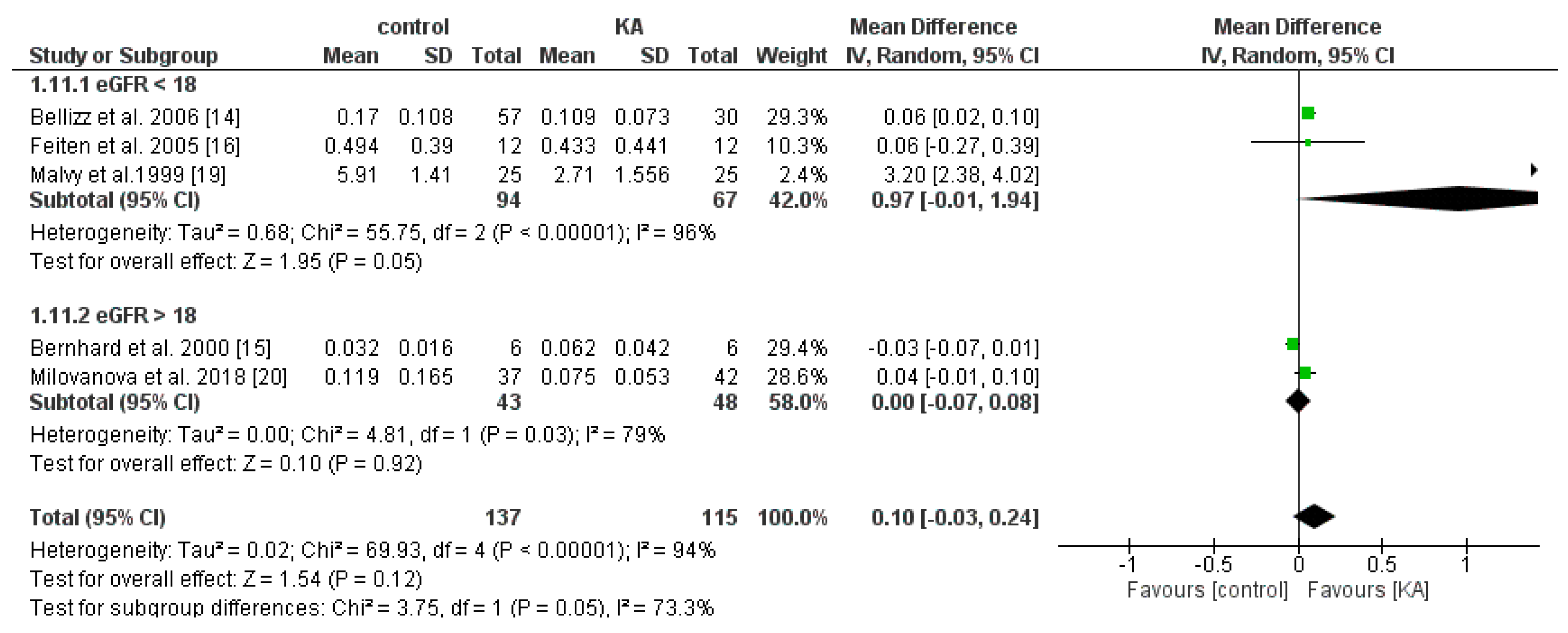
3.4.1. Effects of KA on Serum Phosphorus
3.4.2. Effects of KA on Serum Calcium
3.4.3. Effects of KA on Serum Parathyroid Hormone (PTH)
3.5. Risk of Bias and Quality Assessment
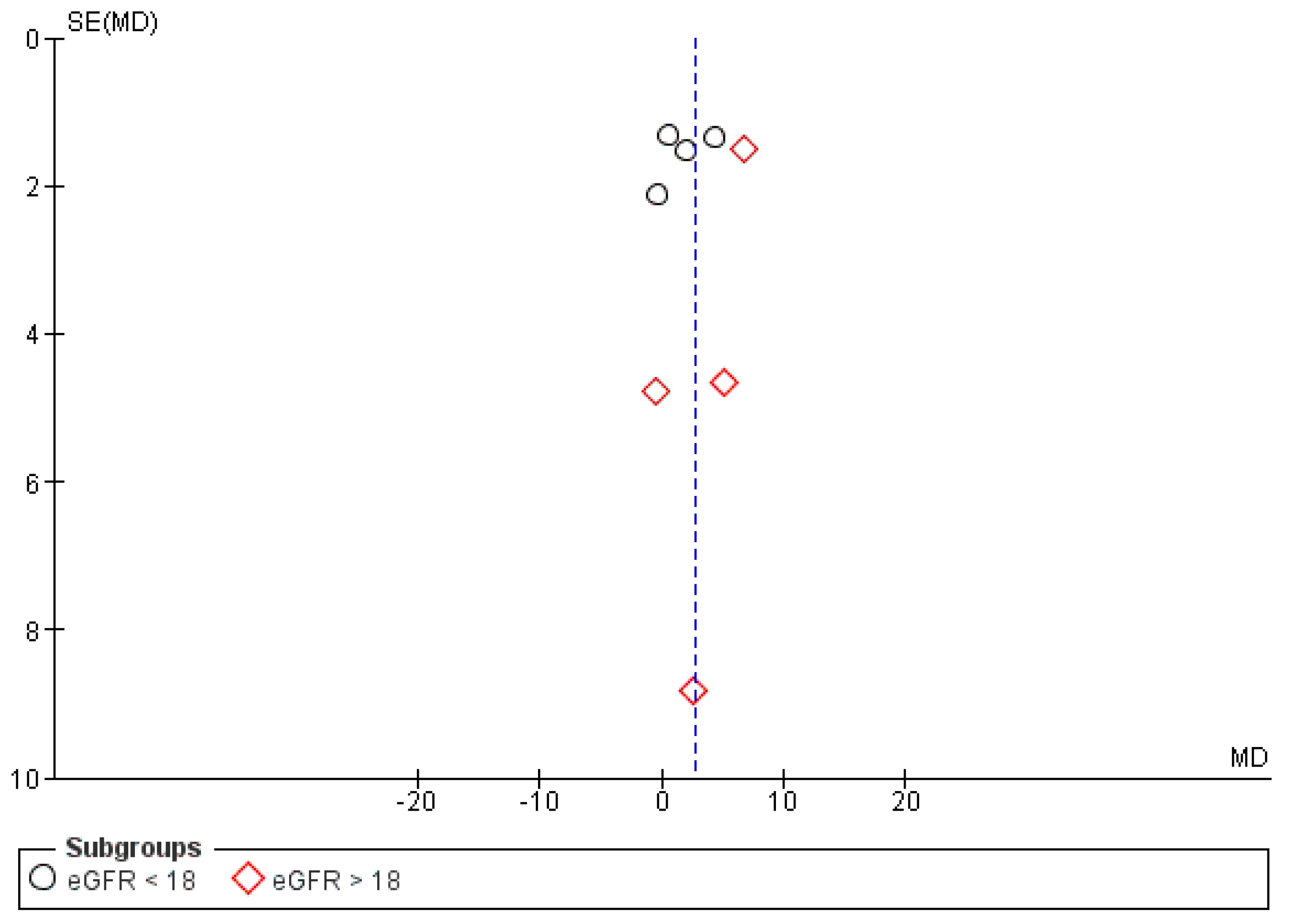
3.6. Publication Bias
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Levin, A. The clinical epidemiology of cardiovascular diseases in chronic kidney disease: Clinical epidemiology of cardiovascular disease in chronic kidney disease prior to dialysis. Semin. Dial. 2003, 16, 101–105. [Google Scholar] [CrossRef]
- Liu, M.; Li, X.C.; Lu, L.; Cao, Y.; Sun, R.R.; Chen, S.; Zhang, P.Y. Cardiovascular disease and its relationship with chronic kidney disease. Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 2918–2926. [Google Scholar] [PubMed]
- Mitch, W.E. Dietary protein restriction in chronic renal failure: Nutritional efficacy, compliance, and progression of renal insufficiency. J. Am. Soc. Nephrol. 1991, 2, 823–831. [Google Scholar]
- Campbell, J.; Porter, J. Dietary mobile apps and their effect on nutritional indicators in chronic renal disease: A systematic review. Nephrology 2015, 20, 744–751. [Google Scholar] [CrossRef]
- Bellizzi, V.; Calella, P.; Carrero, J.J.; Fouque, D. Very low-protein diet to postpone renal failure: Pathophysiology and clinical applications in chronic kidney disease. Chronic Dis. Transl. Med. 2018, 4, 45–50. [Google Scholar] [CrossRef]
- Khan, I.A.; Nasiruddin, M.; Haque, S.F.; Khan, R.A. Comparative evaluation of efficacy and safety profile of rhubarb and alpha-keto analogs of essential amino acids supplementation in patients with diabetic nephropathy. Saudi. J. Kidney Dis. Transpl. 2016, 27, 710–716. [Google Scholar] [CrossRef] [PubMed]
- Garneata, L.; Stancu, A.; Dragomir, D.; Stefan, G.; Mircescu, G. Ketoanalogue-Supplemented Vegetarian Very Low-Protein Diet and CKD Progression. J. Am. Soc. Nephrol. 2016, 27, 2164–2176. [Google Scholar] [CrossRef]
- Jiang, Z.; Zhang, X.; Yang, L.; Li, Z.; Qin, W. Effect of restricted protein diet supplemented with keto analogues in chronic kidney disease: A systematic review and meta-analysis. Int. Urol. Nephrol. 2018, 50, 687–694. [Google Scholar] [CrossRef] [PubMed]
- Higgins, J.P. Cochrane Handbook for Systematic Reviews of Interventions. Version 5.1.0 [updated March 2011]. The Cochrane Collaboration. Available online: http://handbook-5-1.cochrane.org/ (accessed on 13 November 2018).
- Shamseer, L.; Moher, D.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: Elaboration and explanation. Br. Med. J. 2015, 349, g7647. [Google Scholar] [CrossRef]
- Higgins, J.P.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A. Cochrane Bias Methods Group; Cochrane Statistical Methods Group. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef] [PubMed]
- Stang, A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 2010, 25, 603–605. [Google Scholar] [CrossRef] [PubMed]
- Review Manager (RevMan) [Computer Program]. Version 5.3; The Nordic Cochrane Centre, The Cochrane Collaboration: Copenhagen, Denmark, 2014; Available online: https://community.cochrane.org/help/tools-and-software/revman-5 (accessed on 12 November 2018).
- Bellizzi, V.; Di Iorio, B.R.; De Nicola, L.; Minutolo, R.; Zamboli, P.; Trucillo, P.; Catapano, F.; Cristofano, C.; Scalfi, L.; Conte, G. Very low protein diet supplemented with ketoanalogs improves blood pressure control in chronic kidney disease. Kidney Int. 2007, 71, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Bernhard, J.; Beaufrere, B.; Laville, M.; Fouque, D. Adaptive response to a low-protein diet in predialysis chronic renal failure patients. J. Am. Soc. Nephrol. 2001, 12, 1249–1254. [Google Scholar]
- Feiten, S.F.; Draibe, S.A.; Watanabe, R.; Duenhas, M.R.; Baxmann, A.C.; Nerbass, F.B.; Cuppari, L. Short-term effects of a very-low-protein diet supplemented with ketoacids in nondialyzed chronic kidney disease patients. Eur. J. Clin. Nutr. 2005, 59, 129–136. [Google Scholar] [CrossRef]
- Hecking, E.; Andrzejewski, L.; Prellwitz, W.; Opferkuch, W.; Muller, D. Double-blind cross-over study with oral alpha-ketoacids in patients with chronic renal failure. Am. J. Clin. Nutr. 1980, 33, 1678–1681. [Google Scholar] [CrossRef] [PubMed]
- Klahr, S.; Levey, A.S.; Beck, G.J.; Caggiula, A.W.; Hunsicker, L.; Kusek, J.W.; Striker, G. The effects of dietary protein restriction and blood-pressure control on the progression of chronic renal disease. N. Engl. J. Med. 1994, 330, 877–884. [Google Scholar] [CrossRef]
- Malvy, D.; Maingourd, C.; Pengloan, J.; Bagros, P.; Nivet, H. Effects of severe protein restriction with ketoanalogues in advanced renal failure. J. Am. Coll. Nutr. 1999, 18, 481–486. [Google Scholar] [CrossRef]
- Milovanova, L.; Fomin, V.; Moiseev, S.; Taranova, M.; Milovanov, Y.; Lysenko Kozlovskaya, L.; Kozlov, V.; Kozevnikova, E.; Milovanova, S.; Lebedeva, M.; et al. Effect of essential amino acid ketoanalogues and protein restriction diet on morphogenetic proteins (FGF-23 and Klotho) in 3b-4 stages chronic kidney disease patients: A randomized pilot study. Clin. Exp. Nephrol. 2018, 22, 1351–1359. [Google Scholar] [CrossRef]
- Mircescu, G.; Garneata, L.; Stancu, S.H.; Capusa, C. Effects of a supplemented hypoproteic diet in chronic kidney disease. J. Ren. Nutr. 2007, 17, 179–188. [Google Scholar] [CrossRef]
- Prakash, S.; Pande, D.P.; Sharma, S.; Sharma, D.; Bal, C.S.; Kulkarni, H. Randomized, double-blind, placebo-controlled trial to evaluate efficacy of ketodiet in predialytic chronic renal failure. J. Ren. Nutr. 2004, 14, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Qiu, H.Y.; Liu, F.; Zhao, L.J.; Huang, S.M.; Zuo, C.; Zhong, H.; Chen, F. Comparison of the effects of alpha-keto/amino acid supplemented low protein diet and diabetes diet in patients with diabetic nephropathy. Sichuan Da Xue Xue Bao Yi Xue Ban 2012, 43, 425–428. [Google Scholar]
- Teplan, V.; Schück, O.; Racek, J.; Mareckova, O.; Stollova, M.; Hanzal, V.; Malý, J. Reduction of plasma asymmetric dimethylarginine in obese patients with chronic kidney disease after three years of a low-protein diet supplemented with keto-amino acids: A randomized controlled trial. Wien. Klin. Wochenschr. 2008, 120, 478–485. [Google Scholar] [CrossRef]
- Kalantar-Zadeh, K.; Cano, N.J.; Budde, K.; Chazot, C.; Kovesdy, C.P.; Mak, R.H.; Mehrotra, R.; Raj, D.S.; Sehgal, A.R.; Stenvinkel, P.; et al. Diets and enteral supplements for improving outcomes in chronic kidney disease. Nat. Rev. Nephrol. 2011, 7, 369–384. [Google Scholar] [CrossRef]
- Rambod, M.; Bross, R.; Zitterkoph, J.; Benner, D.; Pithia, J.; Colman, S.; Kovesdy, C.P.; Kopple, J.D.; Kalantar-Zadeh, K. Association of Malnutrition-Inflammation Score with quality of life and mortality in hemodialysis patients: A 5-year prospective cohort study. Am. J. Kidney Dis. 2009, 53, 298–309. [Google Scholar] [CrossRef]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. Br. Med. J. 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Pugliese, G.; Solini, A.; Bonora, E.; Orsi, E.; Zerbini, G.; Giorgino, F.; Cavalot, F.; Pontiroli, A.E.; Baroni, M.G.; Morano, S.; et al. The Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation provides a better definition of cardiovascular burden associated with CKD than the Modification of Diet in Renal Disease (MDRD) Study formula in subjects with type 2 diabetes. Atherosclerosis 2011, 218, 194–199. [Google Scholar] [CrossRef]
- Whitman, I.R.; Feldman, H.I.; Deo, R. CKD and sudden cardiac death: Epidemiology, mechanisms, and therapeutic approaches. J. Am. Soc. Nephrol. 2012, 23, 1929–1939. [Google Scholar] [CrossRef]
- Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Work Group. KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int. Suppl. 2009, 76, S1–S2. [Google Scholar]
- Tonelli, M.; Wiebe, N.; Culleton, B.; House, A.; Rabbat, C.; Fok, M.; McAlister, F.; Garg, A.X. Chronic kidney disease and mortality risk: A systematic review. J. Am. Soc. Nephrol. 2006, 17, 2034–2047. [Google Scholar] [CrossRef]
- McClellan, W.M.; Jurkovitz, C.; Abramson, J. The epidemiology and control of anaemia among pre-ESRD patients with chronic kidney disease. Eur. J. Clin. Invest. 2005, 35, 58–65. [Google Scholar] [CrossRef]
- Garofalo, C.; Borrelli, S.; De Stefano, T.; Provenzano, M.; Andreucci, M.; Cabiddu, G.; La Milia, V.; Vizzardi, V.; Sandrini, M.; Cancarini, G.; et al. Incremental dialysis in ESRD: Systematic review and meta-analysis. J. Nephrol. 2019, 2, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Garneata, L.; Stancu, A.; Luca, P.; Stefan, G.; Mircescu, G. Vegetarian very low protein diet supplemented with ketoanalogues may reduce nephrotic-range proteinuria in predialysis CKD patients. Nephrol. Dials. Transpl. 2016, 31, 202. [Google Scholar] [CrossRef][Green Version]
- Noce, A.; Vidiri, M.F.; Marrone, G.; Moriconi, E.; Bocedi, A.; Capria, A.; Rovella, V.; Ricci, G.; De Lorenzo, A.; Di Daniele, N. Is low-protein diet a possible risk factor of malnutrition in chronic kidney disease patients? Cell Death Discov. 2016, 2, 16026. [Google Scholar] [CrossRef]
- Fouque, D.; Aparicio, M. Eleven reasons to control the protein intake of patients with chronic kidney disease. Nat. Rev. Nephrol. 2007, 3, 383–392. [Google Scholar] [CrossRef] [PubMed]
- Raphael, K.L.; Zhang, Y.; Ying, J.; Greene, T. Prevalence of and risk factors for reduced serum bicarbonate in chronic kidney disease. Nephrology 2014, 19, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Driver, T.H.; Shlipak, M.G.; Katz, R.; Goldenstein, L.; Sarnak, M.J.; Hoofnagle, A.N.; Siscovick, D.S.; Kestenbaum, B.; de Boer, I.H.; Ix, J.H. Low serum bicarbonate and kidney function decline: The Multi-Ethnic Study of Atherosclerosis (MESA). Am. J. Kidney Dis. 2014, 64, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Feneberg, R.; Schaefer, F.; Veldhuis, J.D. Neuroendocrine adaptations in renal disease. Pediatr Nephrol 2003, 18, 492–497. [Google Scholar] [CrossRef] [PubMed]
- Pupim, L.B.; Flakoll, P.J.; Majchrzak, K.M.; Aftab Guy, D.L.; Stenvinkel, P.; Ikizler, T.A. Increased muscle protein breakdown in chronic hemodialysis patients with type 2 diabetes mellitus. Kidney Int. 2005, 68, 1857–1865. [Google Scholar] [CrossRef]
- Tonshoff, B.; Blum, W.F.; Mehls, O. Derangements of the somatotropic hormone axis in chronic renal failure. Kidney Int. Suppl. 1997, 58, S106–S113. [Google Scholar]
- Handelsman, D.J.; Dong, Q. Hypothalamo-pituitary gonadal axis in chronic renal failure. Endocrinol. Metab. Clin. N. Am. 1993, 22, 145–161. [Google Scholar] [CrossRef]
- Amdur, R.L.; Feldman, H.I.; Gupta, J.; Yang, W.; Kanetsky, P.; Shlipak, M.; Rahman, M.; Lash, J.P.; Townsend, R.R.; Ojo, A.; et al. Inflammation and Progression of CKD: The CRIC Study. Clin. J. Am. Soc. Nephrol. 2016, 11, 1546–1556. [Google Scholar] [CrossRef] [PubMed]
- Ramezani, A.; Raj, D.S. The gut microbiome, kidney disease, and targeted interventions. J. Am. Soc. Nephrol. 2014, 25, 657–670. [Google Scholar] [CrossRef]
- Vaziri, N.D.; Wong, J.; Pahl, M.; Piceno, Y.M.; Yuan, J.; DeSantis, T.Z.; Ni, Z.; Nguyen, T.H.; Andersen, G.L. Chronic kidney disease alters intestinal microbial flora. Kidney Int. 2013, 83, 308–315. [Google Scholar] [CrossRef]
- Ikizler, T.A.; Cano, N.J.; Franch, H.; Fouque, D.; Himmelfarb, J.; Kalantar-Zadeh, K.; Kuhlmann, M.K.; Stenvinkel, P.; TerWee, P.; Teta, D.; et al. Prevention and treatment of protein energy wasting in chronic kidney disease patients: A consensus statement by the International Society of Renal Nutrition and Metabolism. Kidney Int. 2013, 84, 1096–1107. [Google Scholar] [CrossRef]
- Kovesdy, C.P.; Kopple, J.D.; Kalantar-Zadeh, K. Management of protein-energy wasting in non-dialysis-dependent chronic kidney disease: Reconciling low protein intake with nutritional therapy. Am. J. Clin. Nutr. 2013, 97, 1163–1177. [Google Scholar] [CrossRef]
- Menon, V.; Kopple, J.D.; Wang, X.; Beck, G.J.; Collins, A.J.; Kusek, J.W.; Greene, T.; Levey, A.S.; Sarnak, M.J. Effect of a very low-protein diet on outcomes: Long-term follow-up of the Modification of Diet in Renal Disease (MDRD) Study. Am. J. Kidney Dis. 2009, 53, 208–217. [Google Scholar] [CrossRef]
- Ikizler, T.A. The use and misuse of serum albumin as a nutritional marker in kidney disease. Clin. J. Am. Soc. Nephrol. 2012, 7, 1375–1377. [Google Scholar] [CrossRef]
- Gama-Axelsson, T.; Heimbürger, O.; Stenvinkel, P.; Bárány, P.; Lindholm, B.; Qureshi, A.R. Serum albumin as predictor of nutritional status in patients with ESRD. Clin. J. Am. Soc. Nephrol. 2012, 7, 1446–1453. [Google Scholar] [CrossRef]
- Moe, S.M.; Drueke, T.; Lameire, N.; Eknoyan, G. Chronic kidney disease-mineral-bone disorder: A new paradigm. Adv. Chronic Kidney Dis. 2007, 14, 3–12. [Google Scholar] [CrossRef]
- Young, E.W.; Albert, J.M.; Satayathum, S.; Goodkin, D.A.; Pisoni, R.L.; Akiba, T.; Akizawa, T.; Kurokawa, K.; Bommer, J.; Piera, L.; et al. Predictors and consequences of altered mineral metabolism: The Dialysis Outcomes and Practice Patterns Study. Kidney Int. 2005, 67, 1179–1187. [Google Scholar] [CrossRef]
- Block, G.A.; Klassen, P.S.; Lazarus, J.M.; Ofsthun, N.; Lowrie, E.G.; Chertow, G.M. Mineral metabolism, mortality, and morbidity in maintenance hemodialysis. J. Am. Soc. Nephrol. 2004, 15, 2208–2218. [Google Scholar] [CrossRef]
- Stevens, L.A.; Djurdjev, O.; Cardew, S.; Cameron, E.C.; Levin, A. Calcium, phosphate, and parathyroid hormone levels in combination and as a function of dialysis duration predict mortality: Evidence for the complexity of the association between mineral metabolism and outcomes. J. Am. Soc. Nephrol. 2004, 15, 770–779. [Google Scholar] [CrossRef]
- Shrier, I.; Boivin, J.F.; Steele, R.J.; Platt, R.W.; Furlan, A.; Kakuma, R.; Brophy, J.; Rossignol, M. Should meta-analyses of interventions include observational studies in addition to randomized controlled trials? A critical examination of underlying principles. Am. J. Epidemiol. 2007, 166, 1203–1209. [Google Scholar] [CrossRef]
| First Author, Year | Country | Regimen | Sample Size (Male,Female) | Treatment Duration | Study Design | Age (years) |
|---|---|---|---|---|---|---|
| Bellizzi, 2006 [14] | Italy | T: KAVLP (1 pill/5 kg/day) C: FD | T: 30 C: 23 | 3–6 months | Non-RCT | T: 58.0 ± 16.1 C: 56.3 ± 15.6 |
| Bernhard, 2000 [15] | France | T: KALP (1 pill/5 kg/day) C: Placebo | T: 6 (4,2) C: 6 (6,0) | 3 months | RCT | T: 49.5 ± 7.0 C: 39.0 ± 5.8 |
| Feiten, 2005 [16] | Brazil | T: KAVLP (1 pill/5 kg/day) C: Placebo | T: 12 (7,5) C: 12 (8,4) | 4 months | RCT | T: 49.7 ± 11.3 C: 43.9 ± 16.3 |
| Garneata, 2016 [7] | Romania | T: KAVLP (1 pill/5 kg/day) C: Placebo | T: 104 C: 103 | 12 months | RCT | NA |
| Hecking, 1980 [17] | Germany | T: KALP (1.05 g/10 kg/day) C: Placebo | T: 15 (7,8) C: 15 (7,8) | 6 weeks | Non-RCT | T: 43.7 ± 12.6 C: 43.7 ± 12.6 |
| Klahr, 1994 (HBP) [18] | USA | T: KAVLP (0.28 g/kg/day) C: Placebo | T: 61 C: 62 | 18–45 months | RCT | NA |
| Klahr, 1994 (LBP) [18] | USA | T: KAVLP (0.28 g/kg/day) C: Placebo | T: 65 C: 67 | 18–45 months | RCT | NA |
| Malvy, 1999 [19] | France | T: KAVLP (0.17 g/kg/day) C: Placebo | T: 25 (11,14) C: 25 (10,15) | 3 months | RCT | T: 53.6 ± 11.0 C: 56.0 ± 14.0 |
| Milovanova, 2018 [20] | Russian Federation | T: KALP (0.1 g/kg/day) C: Placebo | T: 42 C: 37 | 14 months | RCT | NA |
| Mircescu, 2007 [21] | Romania | T: KAVLP (1 pill/5 kg/day) C: Placebo | T: 27 (17,10) C: 26 (15,11) | 15 months | RCT | T: 55.0 ± 12.7 C: 53.6 ± 11.0 |
| Prakash, 2004 [22] | India | T: KAVLP (1 pill/5 kg/day) C: Placebo | T: 18 (10,8) C: 16 (7,9) | 9 months | RCT | T: 52.8 ± 14.1 C: 55.9 ± 17.6 |
| Qiu, 2012 [23] | China | T: KAVLP (1 pill/5 kg/day) C: Placebo | T: 12 C: 11 | 52 months | RCT | T: 63.0 ± 8.9 C: 61.60 ± 9.67 |
| Teplan, 2008 [24] | Czech | T: KAVLP (0.1 g/5 kg/day) C: Placebo | T: 66 C: 65 | 36 months | RCT | T: 52 ± 7 C: 52 ± 7 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, A.; Lee, H.-Y.; Lin, Y.-C. The Effect of Ketoanalogues on Chronic Kidney Disease Deterioration: A Meta-Analysis. Nutrients 2019, 11, 957. https://doi.org/10.3390/nu11050957
Li A, Lee H-Y, Lin Y-C. The Effect of Ketoanalogues on Chronic Kidney Disease Deterioration: A Meta-Analysis. Nutrients. 2019; 11(5):957. https://doi.org/10.3390/nu11050957
Chicago/Turabian StyleLi, Albert, Hsiang-Yen Lee, and Yen-Chung Lin. 2019. "The Effect of Ketoanalogues on Chronic Kidney Disease Deterioration: A Meta-Analysis" Nutrients 11, no. 5: 957. https://doi.org/10.3390/nu11050957
APA StyleLi, A., Lee, H.-Y., & Lin, Y.-C. (2019). The Effect of Ketoanalogues on Chronic Kidney Disease Deterioration: A Meta-Analysis. Nutrients, 11(5), 957. https://doi.org/10.3390/nu11050957





