Saffron: The Golden Spice with Therapeutic Properties on Digestive Diseases
Abstract
1. Introduction
2. Methods
3. Saffron Components’ Bioavailability and Bioactivity
4. Anti-Inflammatory and Anti-Carcinogenic Effects of Saffron
5. Effectiveness of Saffron in Digestive Diseases
5.1. Effects on Gastrointestinal Inflammatory Disorders
5.1.1. Gastritis and Peptic Ulcer
5.1.2. Irritable Bowel Syndrome
5.1.3. Inflammatory Bowel Diseases
5.1.4. Hepatitis
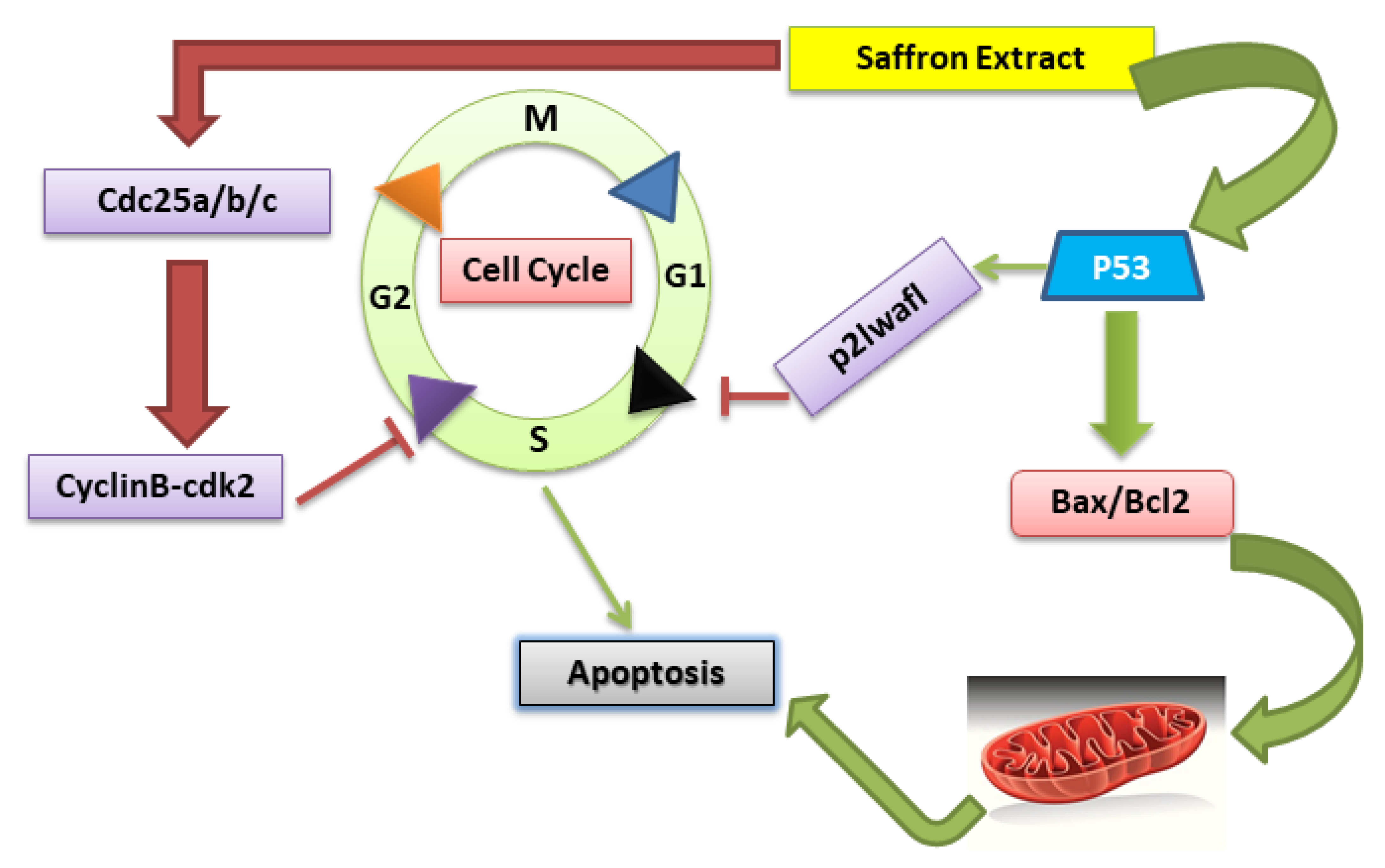
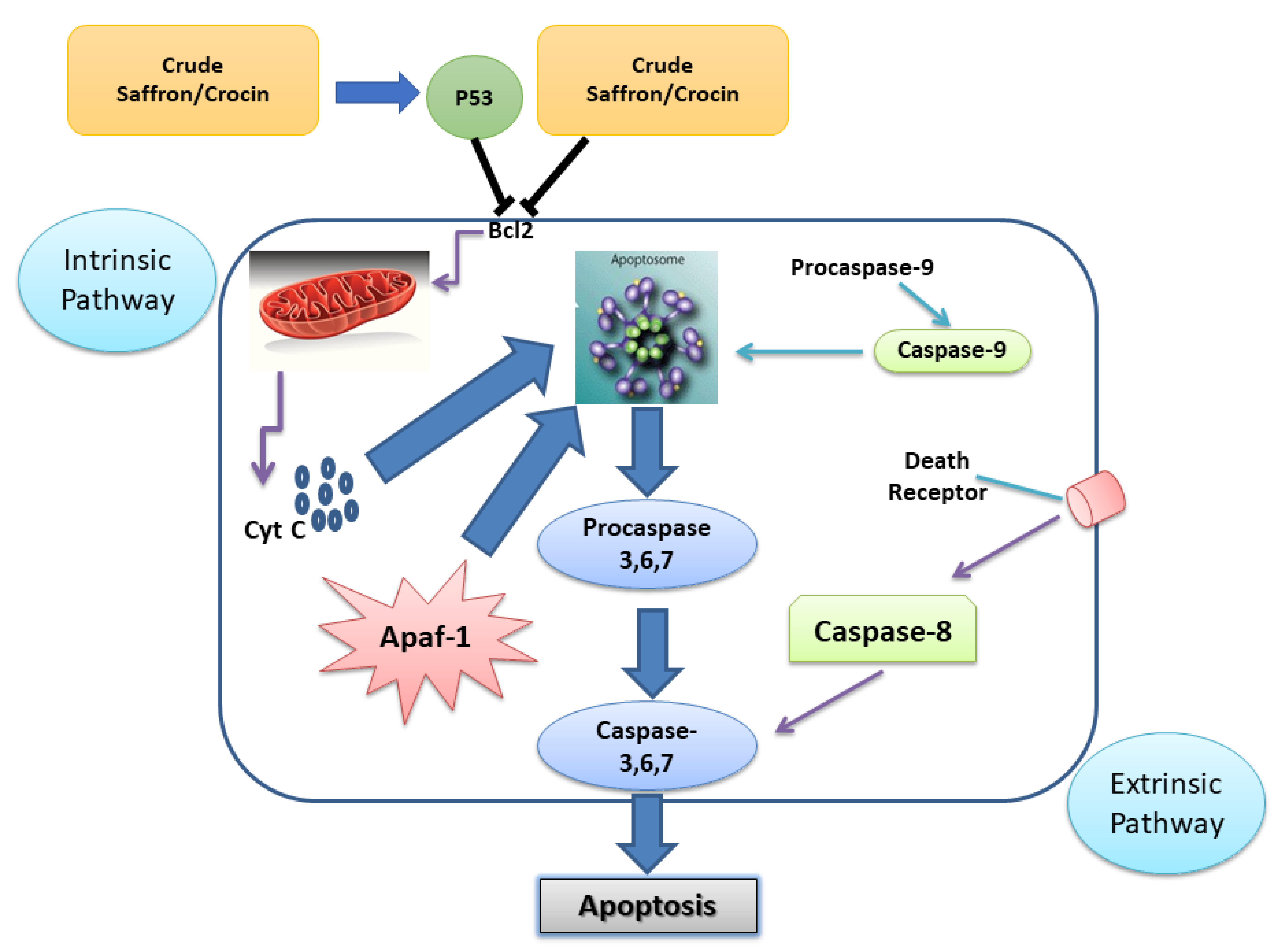
5.2. Effects on Cancer Prevention and Treatment
5.2.1. Gastric Cancer
5.2.2. Colorectal Cancer
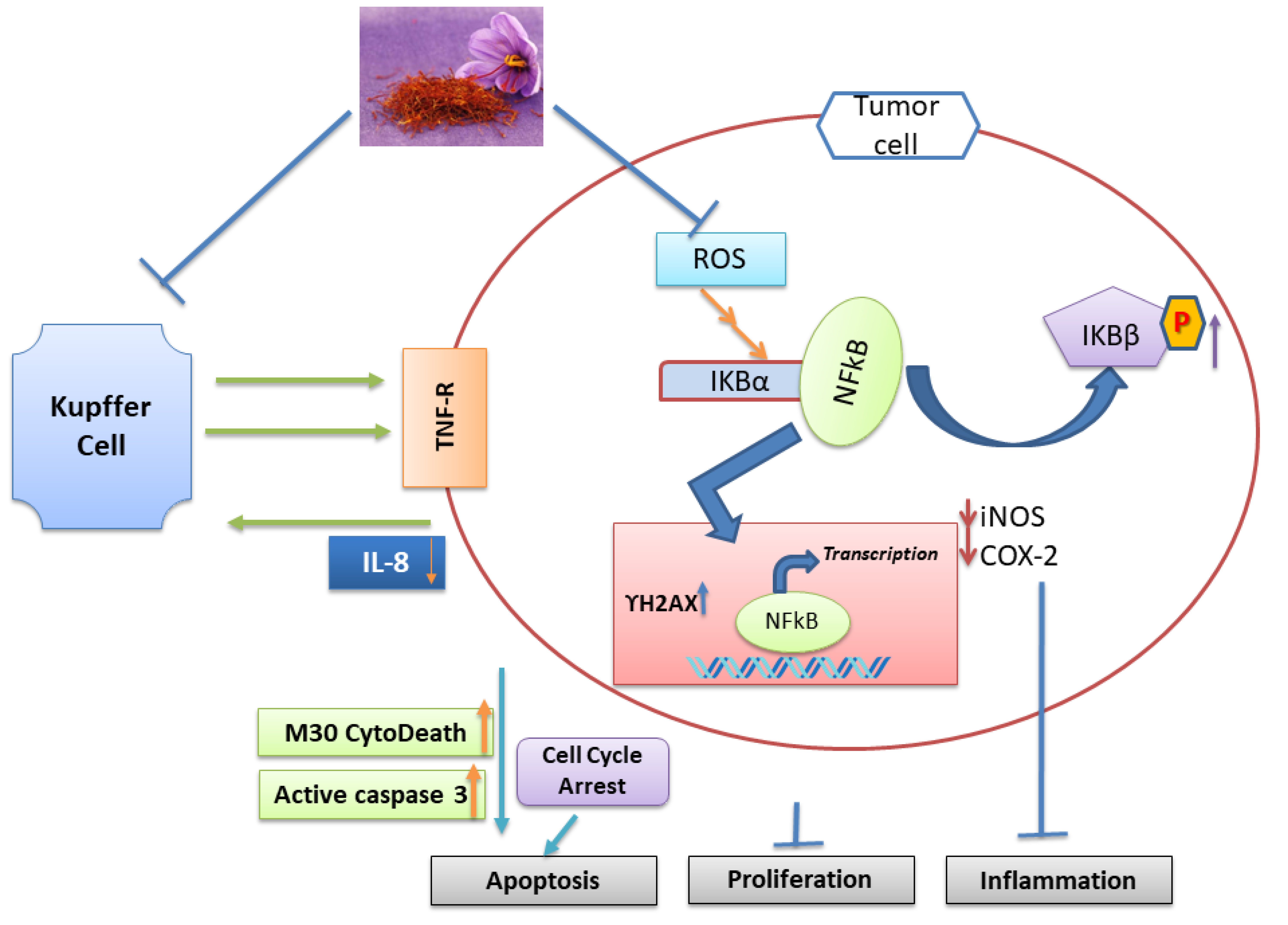
5.2.3. Liver Cancer
5.2.4. Pancreatic Cancer
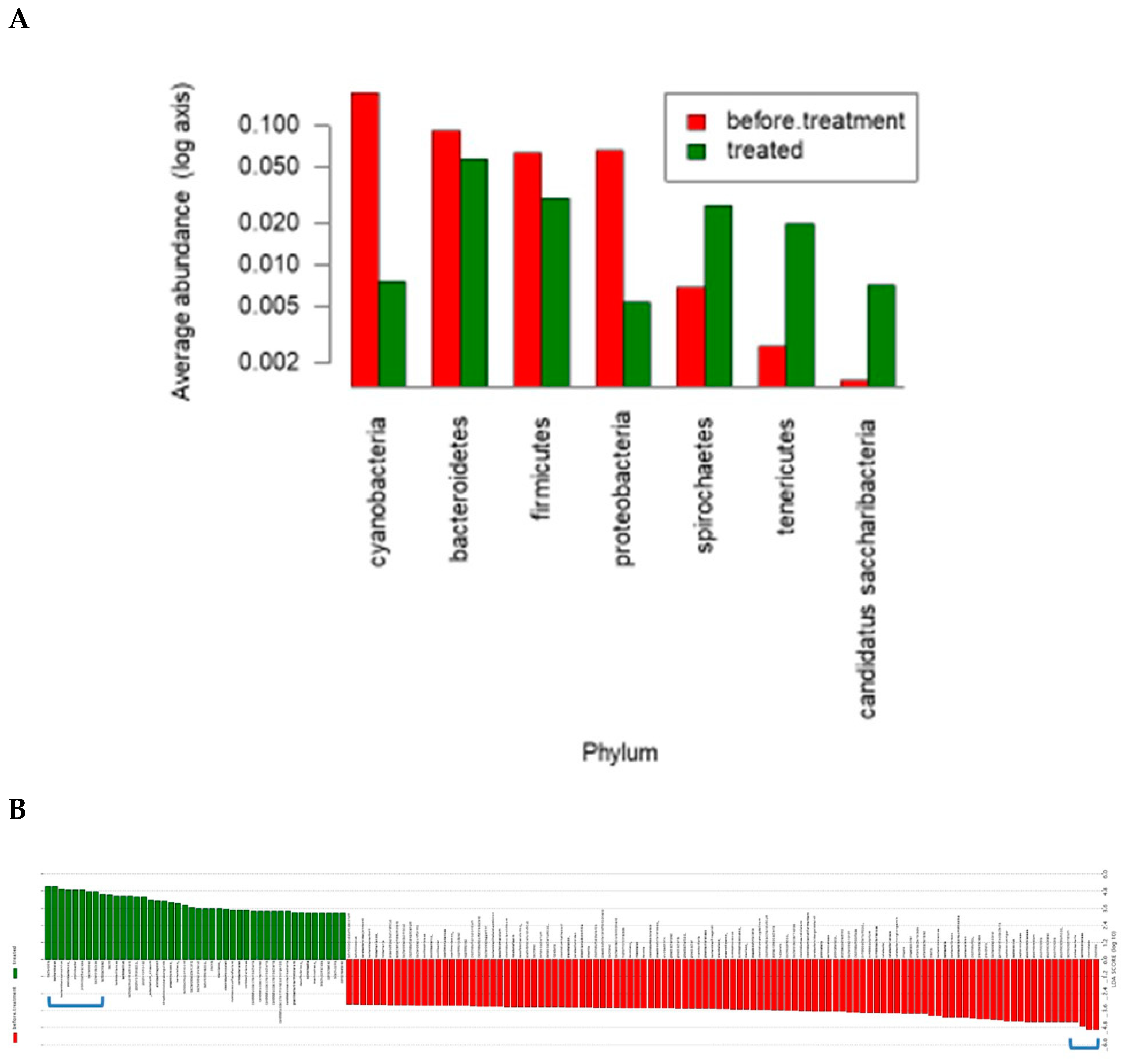
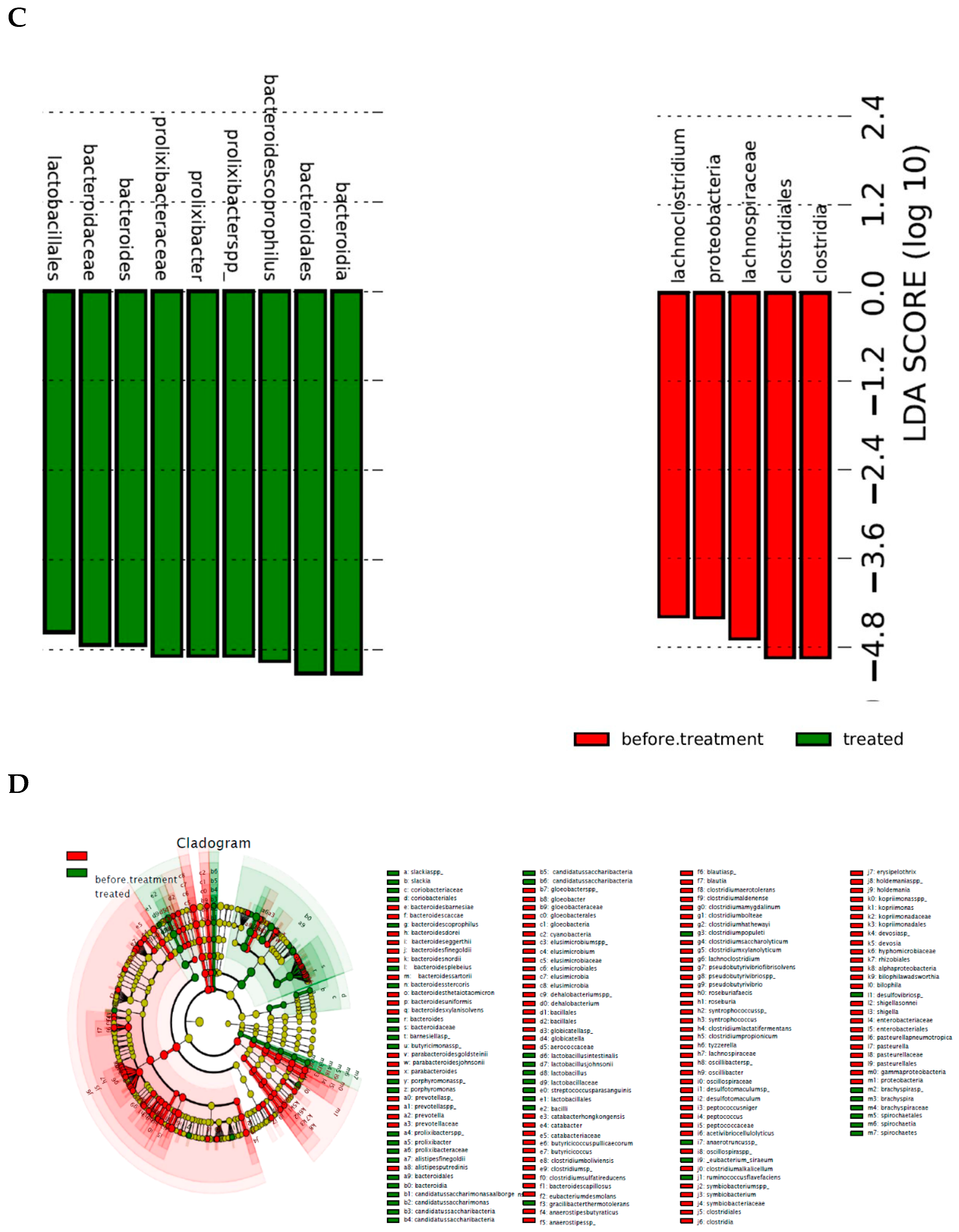
5.3. Effects of Saffron on the Gut Microbiome
6. Conclusion and Perspective of the Review
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khazdair, M.R.; Boskabady, M.H.; Hosseini, M.; Rezaee, R.; Tsatsakis, A.M. The effects of Crocus sativus (saffron) and its constituents on nervous system: A review. Avicenna J. Phytomed. 2015, 5, 376–391. [Google Scholar]
- Kianbakht, S. A Systematic Review on Pharmacology of Saffron and its Active Constituents. J. Med. Plants 2008, 4, 1–27. [Google Scholar]
- Abdullaev, F.I.; Espinosa-Aguirre, J.J. Biomedical properties of saffron and its potential use in cancer therapy and chemoprevention trials. Cancer Detect. Prev. 2004, 28, 426–432. [Google Scholar] [CrossRef] [PubMed]
- Nilakshi, N.; Gadiya, R.V.; Champalal, K.D. Detailed profile of Crocus sativus. Int. J. Pharma Bio Sci. 2011, 2, 189–195. [Google Scholar]
- Akhondzadeh, S.; Sabet, M.S.; Harirchian, M.H.; Togha, M.; Cheraghmakani, H.; Razeghi, S.; Hejazi, S.S.; Yousefi, M.H.; Alimardani, R.; Jamshidi, A.; et al. Saffron in the treatment of patients with mild to moderate Alzheimer’s disease: A 16-week, randomized and placebo-controlled trial. J. Clin. Pharm. Ther. 2010, 35, 581–588. [Google Scholar] [CrossRef] [PubMed]
- Sarris, J. Herbal medicines in the treatment of psychiatric disorders: A systematic review. Phytother. Res. 2007, 21, 703–716. [Google Scholar] [CrossRef]
- Moallem, S.A.; Afshar, M.; Etemad, L.; Razavi, B.M.; Hosseinzadeh, H. Evaluation of teratogenic effects of crocin and safranal, active ingredients of saffron, in mice. Toxicol. Ind. Health 2016, 32, 285–291. [Google Scholar] [CrossRef]
- Kamboj, V.P. Herbal medicine. Curr. Sci. 2000, 78, 35–44. [Google Scholar]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef] [PubMed]
- Jose Bagur, M.; Alonso Salinas, G.L.; Jimenez-Monreal, A.M.; Chaouqi, S.; Llorens, S.; Martínez-Tomé, M.; Alonso, G.L. Saffron: An Old Medicinal Plant and a Potential Novel Functional Food. Molecules 2017, 23, 30. [Google Scholar] [CrossRef]
- Lautenschlager, M.; Sendker, J.; Huwel, S.; Chaouqi, S.; Llorens, S.; Martínez-Tomé, M.; Alonso, G.L. Intestinal formation of trans-crocetin from saffron extract (Crocus sativus L.) and in vitro permeation through intestinal and blood brain barrier. Phytomedicine 2015, 22, 36–44. [Google Scholar] [CrossRef]
- Zhang, Y.; Fei, F.; Zhen, L.; Zhu, X.; Wang, J.; Li, S.; Geng, J.; Sun, R.; Yu, X.; Chen, T.; et al. Sensitive analysis and simultaneous assessment of pharmacokinetic properties of crocin and crocetin after oral administration in rats. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 2017, 1044, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Yoshino, F.; Yoshida, A.; Umigai, N.; Kubo, K.; Lee, M.C. Crocetin reduces the oxidative stress induced reactive oxygen species in the stroke-prone spontaneously hypertensive rats (SHRSPs) brain. J. Clin. Biochem. Nutr. 2011, 49, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Khorasany, A.R.; Hosseinzadeh, H. Therapeutic effects of saffron (Crocus sativus L.) in digestive disorders: A review. Iran. J. Basic Med. Sci. 2016, 19, 455–469. [Google Scholar] [PubMed]
- Kawabata, K.; Tung, N.H.; Shoyama, Y.; Sugie, S.; Mori, T.; Tanaka, T. Dietary Crocin Inhibits Colitis and Colitis-Associated Colorectal Carcinogenesis in Male ICR Mice. Evid. Based Complement. Alternat. Med. 2012, 2012, 820415. [Google Scholar] [CrossRef] [PubMed]
- Khodir, A.E.; Said, E.; Atif, H.; ElKashef, H.A.; Salem, H.A. Targeting Nrf2/HO-1 signaling by crocin: Role in attenuation of AA-induced ulcerative colitis in rats. Biomed. Pharmacother. 2019, 110, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Farahmand, S.K.; Samini, F.; Samini, M.; Samarghandian, S. Safranal ameliorates antioxidant enzymes and suppresses lipid peroxidation and nitric oxide formation in aged male rat liver. Biogerontology 2013, 14, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Yang, T.; Huang, J.; Tian, X.; Zhao, C.; Cai, L.; Feng, L.; Zhang, H. Comparative evaluation of the antioxidant capacity of crocetin and crocin in vivo. Chin. Pharmacol. Bull. 2010, 26, 248–251. [Google Scholar]
- Bandegi, A.R.; Rashidy-Pour, A.; Vafaei, A.A.; Ghadrdoost, B. Protective Effects of Crocus Sativus L. Extract and Crocin against Chronic-Stress Induced Oxidative Damage of Brain, Liver and Kidneys in Rats. Adv. Pharm. Bull. 2014, 4, 493–499. [Google Scholar] [PubMed]
- Rezaee-Khorasany, A.; Razavi, B.M.; Taghiabadi, E.; Tabatabaei Yazdi, A.; Hosseinzadeh, H. Effect of saffron (stigma of Crocus sativus L.) aqueous extract on ethanol toxicity in rats: A biochemical, histopathological and molecular study. J. Ethnopharmacol. 2019, 237, 286–299. [Google Scholar] [CrossRef] [PubMed]
- El-Maraghy, S.A.; Rizk, S.M.; Shahin, N.N. Gastroprotective effect of crocin in ethanol-induced gastric injury in rats. Chem. Biol. Interact. 2015, 229, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Kianbakht, S.; Mozaffari, K. Effects of saffron and its active constituents, crocin and safranal, on prevention of indomethacin induced gastric ulcers in diabetic and non-diabetic rats. J. Med. Plants 2009, 8, 30–38. [Google Scholar]
- Inoue, E.; Shimizu, Y.; Shoji, M.; Tsuchida, H.; Sano, Y.; Ito, C. Pharmacological properties of N-095, a drug containing red ginseng, polygala root, saffron, antelope horn and aloe wood. Am. J. Chin. Med. 2005, 33, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Al-Mofleh, I.A.; Alhaider, A.A.; Mossa, J.S.; Al-Sohaibani, M.O.; Qureshi, S.; Rafatullah, S. Antigastric ulcer studies on ‘saffron’ Crocus sativus L. in rats. Pak. J. Biol. Sci. 2006, 9, 1009–1013. [Google Scholar]
- Everhart, J.E.; Ruhl, C.E. Burden of digestive diseases in the United States Part III: Liver, biliary tract, and pancreas. Gastroenterology 2009, 136, 1134–1144. [Google Scholar] [CrossRef] [PubMed]
- Saito, Y.A.; Schoenfeld, P.; Locke, G.R. The epidemiology of irritable bowel syndrome in North America: A systematic review. Am. J. Gastroenterol. 2002, 97, 1910–1915. [Google Scholar] [CrossRef]
- Hungin, A.P.; Chang, L.; Locke, G.R.; Dennis, E.H.; Barghout, V. Irritable bowel syndrome in the United States: Prevalence, symptom patterns and impact. Aliment. Pharmacol. Ther. 2005, 21, 1365–1375. [Google Scholar] [CrossRef]
- Grover, M.; Drossman, D.A. Centrally acting therapies for irritable bowel syndrome. Gastroenterol. Clin. N. Am. 2011, 40, 183–206. [Google Scholar] [CrossRef]
- Hungin, A.P.; Whorwell, P.J.; Tack, J.; Mearin, F. The prevalence, patterns and impact of irritable bowel syndrome: An international survey of 40,000 subjects. Aliment. Pharmacol. Ther. 2003, 17, 643–650. [Google Scholar] [CrossRef]
- Andrews, E.B.; Eaton, S.C.; Hollis, K.A.; Hopkins, J.S.; Ameen, V.; Hamm, L.R.; Cook, S.F.; Tennis, P.; Mangel, A.W. Prevalence and demographics of irritable bowel syndrome: Results from a large web-based survey. Aliment. Pharmacol. Ther. 2005, 22, 935–942. [Google Scholar] [CrossRef]
- Cain, K.C.; Jarrett, M.E.; Burr, R.L.; Rosen, S.; Hertig, V.L.; Heitkemper, M.M. Gender differences in gastrointestinal, psychological, and somatic symptoms in irritable bowel syndrome. Dig. Dis. Sci. 2009, 54, 1542–1549. [Google Scholar] [CrossRef]
- Wigington, W.C.; Johnson, W.D.; Minocha, A. Epidemiology of irritable bowel syndrome among African Americans as compared with whites: A population-based study. Clin. Gastroenterol. Hepatol. 2005, 3, 647–653. [Google Scholar] [CrossRef]
- Kang, J.Y. Systematic review: The influence of geography and ethnicity in irritable bowel syndrome. Aliment. Pharmacol. Ther. 2005, 21, 663–676. [Google Scholar] [CrossRef] [PubMed]
- Saha, L. Irritable bowel syndrome: Pathogenesis, diagnosis, treatment, and evidence-based medicine. World J. Gastroenterol. 2014, 20, 6759–6773. [Google Scholar] [CrossRef] [PubMed]
- Ng, Q.X.; Soh, A.Y.S.; Loke, W.; Venkatanarayanan, N.; Lim, D.Y.; Yeo, W.S. A Meta-Analysis of the Clinical Use of Curcumin for Irritable Bowel Syndrome (IBS). J. Clin. Med. 2018, 7, 298. [Google Scholar] [CrossRef] [PubMed]
- Najafabadi, B.T.; Kermany, G.; Ranjbar, T.; Noorbala, A.; Daryani, N.E.; Vanaki, E.; Akhondzadeh, S. Therapeutic effects of saffron versus fluoxetine on Irritable Bowel Syndrome: Adouble-blind randomized clinical trial. Adv. Integr. Med. 2019, in press. [Google Scholar]
- Capannolo, A.; Ciccone, F.; Latella, G. Mastocytic Enterocolits and the Role of Mast Cells in Functional and Inflammatory Intestinal Disorders: A Systematic Review. Dig. Dis. 2018, 36, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Altobelli, E.; Del Negro, V.; Angeletti, P.M.; Latella, G. Low-FODMAP Diet Improves Irritable Bowel Syndrome Symptoms: A Meta-Analysis. Nutrients 2017, 9, 940. [Google Scholar] [CrossRef]
- Rahbani, M.; Mohajeri, D.; Rezaie, A.; Nazeri, M. Protective Effect of Ethanolic Extract of Saffron (Dried Stigmas of Crocus sativus L.) on Hepatic Tissue Injury in Streptozotocin-Induced Diabetic Rats. J. Anim. Vet. Adv. 2012, 11, 1985–1994. [Google Scholar] [CrossRef]
- Ramadan, A.; Soliman, G.; Mahmoud, S.S.; Nofal, S.M.; Abdel-Rahman, R.F. Evaluation of the safety and antioxidant activities of Crocus sativus and Propolis ethanolic extracts. J. Saudi Chem. Soc. 2012, 16, 13–21. [Google Scholar] [CrossRef]
- Yahfoufi, N.; Alsadi, N.; Jambi, M.; Matar, C. The Immunomodulatory and Anti-Inflammatory Role of Polyphenols. Nutrients 2018, 10, 1618. [Google Scholar] [CrossRef] [PubMed]
- Bajbouj, K.; Schulze-Luehrmann, J.; Diermeier, S.; Amin, A.; Schneider-Stock, R. The anticancer effect of saffron in two p53 isogenic colorectal cancer cell lines. BMC Complement. Altern. Med. 2012, 12, 69. [Google Scholar] [CrossRef] [PubMed]
- Aung, H.H.; Wang, C.Z.; Ni, M.; Fishbein, A.; Mehendale, S.R.; Xie, J.T.; Shoyama, C.Y.; Yuan, C.S. Crocin from Crocus sativus possesses significant anti-proliferation effects on human colorectal cancer cells. Exp. Oncol. 2007, 29, 175–180. [Google Scholar] [PubMed]
- Amin, A.; Hamza, A.A.; Bajbouj, K.; Ashraf, S.S.; Daoud, S. Saffron: A potential candidate for a novel anticancer drug against hepatocellular carcinoma. Hepatology 2011, 54, 857–867. [Google Scholar] [CrossRef] [PubMed]
- Amin, A.; Hamza, A.A.; Daoud, S.; Khazanehdari, K.; Hrout, A.A.; Baig, B.; Chaiboonchoe, A.; Adrian, T.E.; Zaki, N.; Salehi-Ashtiani, K. Saffron-Based Crocin Prevents Early Lesions of Liver Cancer: In vivo, In vitro and Network Analyses. Recent Pat. Anticancer Drug Discov. 2016, 11, 121–133. [Google Scholar] [CrossRef] [PubMed]
- Bathaie, S.Z.; Hoshyar, R.; Miri, H.; Sadeghizadeh, M. Anticancer effects of crocetin in both human adenocarcinoma gastric cancer cells and rat model of gastric cancer. Biochem. Cell Biol. 2013, 91, 397–403. [Google Scholar] [CrossRef] [PubMed]
- He, K.; Si, P.; Wang, H.; Tahir, U.; Chen, K.; Xiao, J.; Duan, X.; Huang, R.; Xiang, G. Crocetin induces apoptosis of BGC-823 human gastric cancer cells. Mol. Med. Rep. 2014, 9, 521–526. [Google Scholar] [CrossRef] [PubMed]
- Noureini, S.K.; Wink, M. Antiproliferative effects of crocin in HepG2 cells by telomerase inhibition and hTERT down-regulation. Asian Pac. J. Cancer Prev. 2012, 13, 2305–2309. [Google Scholar] [CrossRef] [PubMed]
- Dhar, A.; Mehta, S.; Dhar, G.; Dhar, K.; Banerjee, S.; Van Veldhuizen, P.; Campbell, D.R.; Banerjee, S.K. Crocetin inhibits pancreatic cancer cell proliferation and tumor progression in a xenograft mouse model. Mol. Cancer. Ther. 2009, 8, 315–323. [Google Scholar] [CrossRef]
- Bakshi, H.; Sam, S.; Rozati, R.; Sultan, P.; Islam, T.; Rathore, B.; Lone, Z.; Sharma, M.; Triphati, J.; Saxena, R.C. DNA fragmentation and cell cycle arrest: A hallmark of apoptosis induced by crocin from kashmiri saffron in a human pancreatic cancer cell line. Asian Pac. J. Cancer Prev. 2010, 11, 675–679. [Google Scholar] [PubMed]
- Rangarajan, P.; Subramaniam, D.; Paul, S.; Kwatra, D.; Palaniyandi, K.; Islam, S.; Harihar, S.; Ramalingam, S.; Gutheil, W.; Putty, S.; et al. Crocetinic acid inhibits hedgehog signaling to inhibit pancreatic cancer stem cells. Oncotarget 2015, 6, 27661–27673. [Google Scholar] [CrossRef] [PubMed]
- Li, C.Y.; Huang, W.F.; Wang, Q.L.; Wang, F.; Cai, E.; Hu, B.; Du, J.C.; Wang, J.; Chen, R.; Cai, X.J.; et al. Crocetin induces cytotoxicity in colon cancer cells via p53-independent mechanisms. Asian Pac. J. Cancer Prev. 2012, 13, 3757–3761. [Google Scholar] [CrossRef] [PubMed]
- Jnaneshwari, S.; Hemshekhar, M.; Santhosh, M.S.; Sunitha, K.; Thushara, R.; Thirunavukkarasu, C.; Kemparaju, K.; Girish, K.S. Crocin, a dietary colorant, mitigates cyclophosphamide-induced organ toxicity by modulating antioxidant status and inflammatory cytokines. J. Pharm. Pharmacol. 2013, 65, 604–614. [Google Scholar] [CrossRef] [PubMed]
- Bathaie, S.Z.; Miri, H.; Mohagheghi, M.A.; Mokhtari-Dizaji, M.; Shahbazfar, A.A.; Hasanzadeh, H. Saffron Aqueous Extract Inhibits the Chemically-induced Gastric Cancer Progression in the Wistar Albino Rat. Iran. J. Basic Med. Sci. 2013, 16, 27–38. [Google Scholar] [PubMed]
- Al-Hrout, A.; Chaiboonchoe, A.; Khraiwesh, B.; Murali, C.; Baig, B.; El-Awady, R.; Tarazi, H.; Alzahmi, A.; Nelson, D.R.; Greish, Y.E.; et al. Safranal induces DNA double-strand breakage and ER-stress-mediated cell death in hepatocellular carcinoma cells. Sci. Rep. 2018, 8, 16951. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Yang, J.; Wang, L.Z.; Sun, L.R.; Dong, Q. Crocin attenuates cisplatin-induced liver injury in the mice. Hum. Exp. Toxicol. 2014, 33, 855–862. [Google Scholar] [CrossRef]
- Hoshyar, R.; Bathaie, S.Z.; Sadeghizadeh, M. Crocin triggers the apoptosis through increasing the Bax/Bcl-2 ratio and caspase activation in human gastric adenocarcinoma, AGS, cells. DNA Cell Biol. 2013, 32, 50–57. [Google Scholar] [CrossRef]
- Garcia-Olmo, D.C.; Riese, H.H.; Escribano, J.; Ontañón, J.; Fernandez, J.A.; Atiénzar, M.; García-Olmo, D. Effects of long-term treatment of colon adenocarcinoma with crocin, a carotenoid from saffron (Crocus sativus L.): An experimental study in the rat. Nutr. Cancer 1999, 35, 120–126. [Google Scholar] [CrossRef] [PubMed]
- Amerizadeh, F.; Rezaei, N.; Rahmani, F.; Hassanian, S.M.; Moradi-Marjaneh, R.; Fiuji, H.; Boroumand, N.; Nosrati-Tirkani, A.; Ghayour-Mobarhan, M.; Ferns, G.A.; et al. Crocin synergistically enhances the antiproliferative activity of 5-flurouracil through Wnt/PI3K pathway in a mouse model of colitis-associated colorectal cancer. J. Cell Biochem. 2018, 119, 10250–10261. [Google Scholar] [CrossRef]
- Amin, A.; Bajbouj, K.; Koch, A.; Gandesiri, M.; Schneider-Stock, R. Defective autophagosome formation in p53-null colorectal cancer reinforces crocin-induced apoptosis. Int. J. Mol. Sci. 2015, 16, 1544–1561. [Google Scholar] [CrossRef]
- Rastgoo, M.; Hosseinzadeh, H.; Alavizadeh, H.; Abbasi, A.; Ayati, Z.; Jaafari, M.R. Antitumor activity of PEGylated nanoliposomes containing crocin in mice bearing C26 colon carcinoma. Planta Med. 2013, 79, 447–451. [Google Scholar] [CrossRef] [PubMed]
- Abdullaev, F.I. Cancer chemopreventive and tumoricidal properties of saffron (Crocus sativus L.). Exp. Biol. Med. 2002, 227, 20–25. [Google Scholar] [CrossRef]
- Sasso, A.; Latella, G. Role of Heme Iron in the Association Between Red Meat Consumption and Colorectal Cancer. Nutr. Cancer 2018, 70, 1173–1183. [Google Scholar] [CrossRef] [PubMed]
| Questionnaire | Weeks into Treatment | Treatment Group | |||||
|---|---|---|---|---|---|---|---|
| Saffron | Fluoxetine | ||||||
| Mean | SD | p-Value * | Mean | SD | p-Value * | ||
| IBS-Qol | Baseline | 60.00 | 9.15 | 59.18 | 7.28 | ||
| 2 weeks | 60.61 | 8.07 | 0.033 | 59.27 | 6.21 | 0.753 | |
| 4 weeks | 62.36 | 7.27 | <0.001 | 61.33 | 6.74 | <0.001 | |
| 6 weeks | 68.06 | 7.00 | <0.001 | 67.36 | 7.58 | <0.001 | |
| HADS-Depression domain | Baseline | 7.48 | 1.80 | 7.88 | 1.85 | ||
| 2 weeks | 7.36 | 1.52 | 0.525 | 7.45 | 1.64 | <0.001 | |
| 4 weeks | 6.58 | 1.23 | <0.001 | 6.76 | 1.12 | <0.001 | |
| 6 weeks | 5.91 | 0.98 | <0.001 | 6.21 | 0.86 | <0.001 | |
| HADS-Anxiety domain | Baseline | 7.27 | 1.72 | 7.45 | 1.60 | ||
| 2 weeks | 7.03 | 1.76 | 0.058 | 7.61 | 1.34 | 0.201 | |
| 4 weeks | 6.79 | 1.62 | 0.001 | 7.39 | 1.30 | 0.690 | |
| 6 weeks | 6.55 | 1.50 | <0.001 | 6.94 | 0.90 | 0.019 | |
| Types of Cancers | Cell Lines/Animal Model | Mechanism of Action | Reference |
|---|---|---|---|
| Colon Cancer | HCT116 | Induction of Apoptosis | [42] |
| HCT116, SW480, and HT29 | Induction of cytotoxicity and Inhibition of cell proliferation | [43] | |
| Liver Cancer | HPG2 | Induction of cytotoxicity and Inhibition of cell proliferation | [44] |
| HPG2 | Induction of Apoptosis | [45] |
| Type of Cancer | Secondary Metabolite | Mechanism of Action | Molecular Changes | References |
|---|---|---|---|---|
| Hepatic Cancer | crocin | Apoptosis | Down-regulation of hTERT gene Down-regulation of the expression of catalytic subunit of enzyme telomerase | [44,45,48] |
| Anti-oxidant property and Anti-inflammatory effect | Increased the levels of GST, SOD, and CAT Reduced myeloperoxidase activity, malondialdehyde Inhibition of COX 2, iNOS, NF-κB | |||
| Pancreatic Cancer | crocetin | Cell cycle arrest at G2/M Phase | Reduced expression of Cdc-2 (hyperphosphoryltion) Reduced expression of Cdc-25c phosphatase Inhibition of Cyclin B1 | [49] |
| crocin, crocetin | Apoptosis | Increased expression of Bax protein Suppressed expression of Bcl-2 Elevated Bax/Bcl-2 ratio | [49,50,51] | |
| crocetin | Inhibition of cell proliferation | Reduced activity of EGFR Reduced phosphorylation of Akt | [49,51] | |
| Colorectal Cancer | crocetin | Cell cycle arrest at S Phase | Reduced expression of cyclin A and cdk2 | [52] |
| crocin | Cell cycle arrest at G3 phase | Decrease in the levels of cyclin B1 and pH3 | [42,45] | |
| Crocin, crocetin | Apoptosis | Augmented expression of p53 and P21 | [42,53] | |
| crocin | DNA Damage | Up-regulation of H2AX | [42,45] | |
| Autophagolysis | Formation of LC3-II Decrease in protein levels of Beclin 1 and Atg 7 genes | [45] | ||
| Gastric Cancer | crocin | Apoptosis | Activation of caspases Elevated Bax/Bcl-2 ratio | [54] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ashktorab, H.; Soleimani, A.; Singh, G.; Amin, A.; Tabtabaei, S.; Latella, G.; Stein, U.; Akhondzadeh, S.; Solanki, N.; Gondré-Lewis, M.C.; et al. Saffron: The Golden Spice with Therapeutic Properties on Digestive Diseases. Nutrients 2019, 11, 943. https://doi.org/10.3390/nu11050943
Ashktorab H, Soleimani A, Singh G, Amin A, Tabtabaei S, Latella G, Stein U, Akhondzadeh S, Solanki N, Gondré-Lewis MC, et al. Saffron: The Golden Spice with Therapeutic Properties on Digestive Diseases. Nutrients. 2019; 11(5):943. https://doi.org/10.3390/nu11050943
Chicago/Turabian StyleAshktorab, Hassan, Akbar Soleimani, Gulshan Singh, Amr Amin, Solmaz Tabtabaei, Giovanni Latella, Ulrike Stein, Shahin Akhondzadeh, Naimesh Solanki, Marjorie C. Gondré-Lewis, and et al. 2019. "Saffron: The Golden Spice with Therapeutic Properties on Digestive Diseases" Nutrients 11, no. 5: 943. https://doi.org/10.3390/nu11050943
APA StyleAshktorab, H., Soleimani, A., Singh, G., Amin, A., Tabtabaei, S., Latella, G., Stein, U., Akhondzadeh, S., Solanki, N., Gondré-Lewis, M. C., Habtezion, A., & Brim, H. (2019). Saffron: The Golden Spice with Therapeutic Properties on Digestive Diseases. Nutrients, 11(5), 943. https://doi.org/10.3390/nu11050943









