Predicting Glycemic Index and Glycemic Load from Macronutrients to Accelerate Development of Foods and Beverages with Lower Glucose Responses
Abstract
1. Introduction
2. Materials and Methods
2.1. Products
2.2. In-Vivo Testing
2.3. FAO Predictive Model
2.4. Development of a New Model to Predict GI
3. Results and Discussion
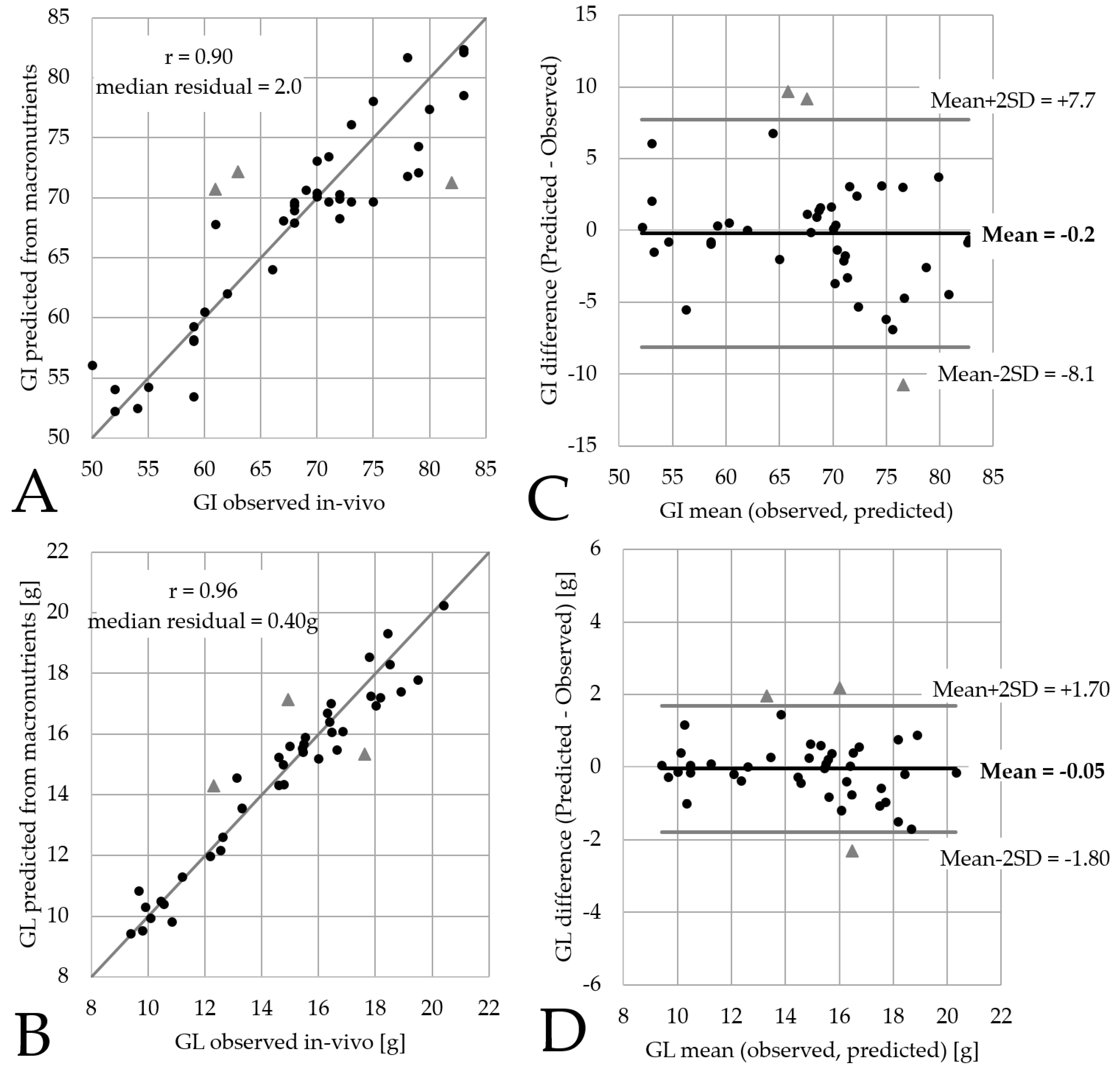
3.1. Precision of Predictions for 42 Breakfast Cereals
3.2. Limitations of the Model
3.3. Potential Use of Model Beyond Breakfast Cereals
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- World Health Organisation. Guideline: Sugars Intake for Adults and Children; World Health Organisation: Geneva, Switzerland, 2015. [Google Scholar]
- Lamothe, L.; Lê, K.; Samra, R.; Roger, O.; Green, H.; Macé, K. The scientific basis for healthful carbohydrate profile. Crit. Rev. Food Sci. Nutr. 2017, 1058–1070. [Google Scholar] [CrossRef]
- Vinoy, S.; Laville, M.; Feskens, E.J.M. Slow-release carbohydrates: Growing evidence on metabolic responses and public health interest. Summary of the symposium held at the 12th European Nutrition Conference (FENS 2015). Food Nutr. Res. 2016, 60, 31662. [Google Scholar] [CrossRef]
- Saulnier, L.; Ducasse, M.; Chiron, H.; Della Valle, G.; Martin, C.; Issanchou, S.; Rouau, X.; Rizkalla, S.W. Impact of texture modification and dietary fibre content on the glycemic index and the acceptability of French bread. Diet. Fibre New Front. Food Health 2010, 115–120. [Google Scholar] [CrossRef]
- AbuMweis, S.; Thandapilly, S.J.; Storsley, J.; Ames, N. Effect of barley β-glucan on postprandial glycaemic response in the healthy human population: A meta-analysis of randomized controlled trials. J. Funct. Foods 2016, 27, 329–342. [Google Scholar] [CrossRef]
- Quek, R.; Bi, X.; Henry, C.J. Impact of protein-rich meals on glycaemic response of rice. Br. J. Nutr. 2016, 115, 1194–1201. [Google Scholar] [CrossRef]
- Owen, B.; Wolever, T.M.S. Effect of fat on glycaemic responses in normal subjects: A dose-response study. Nutr. Res. 2003, 23, 1341–1347. [Google Scholar] [CrossRef]
- Meng, H.; Matthan, N.R.; Ausman, L.M.; Lichtenstein, A.H. Effect of macronutrients and fiber on postprandial glycemic responses and meal glycemic index and glycemic load value determinations. Am. J. Clin. Nutr. 2017, 105, 842–853. [Google Scholar] [CrossRef]
- Augustin, L.S.A.; Kendall, C.W.C.; Jenkins, D.J.A.; Willett, W.C.; Astrup, A.; Barclay, A.W.; Björck, I.; Brand-Miller, J.C.; Brighenti, F.; Buyken, A.E.; et al. Glycemic index, glycemic load and glycemic response: An International Scientific Consensus Summit from the International Carbohydrate Quality Consortium (ICQC). Nutr. Metab. Cardiovasc. Dis. 2015, 25, 795–815. [Google Scholar] [CrossRef]
- Barclay, A.W.; Petocz, P.; McMillan-Price, J.; Flood, V.M.; Prvan, T.; Mitchell, P.; Brand-Miller, J.C. Glycemic index, glycemic load, and chronic disease risk—A metaanalysis of observational studies. Am. J. Clin. Nutr. 2008, 87, 627–637. [Google Scholar] [CrossRef]
- Mirrahimi, A.; Chiavaroli, L.; Srichaikul, K.; Augustin, L.S.A.; Sievenpiper, J.L.; Kendall, C.W.C.; Jenkins, D.J.A. The role of glycemic index and glycemic load in cardiovascular disease and its risk factors: A review of the recent literature. Curr. Atheroscler. Rep. 2014, 16, 380. [Google Scholar] [CrossRef]
- Brand-Miller, J.; Hayne, S.; Petocz, P.; Colagiuri, S. Low-glycemic index diets in the management of diabetes: A meta-analysis of randomized controlled trials. Diabetes Care 2003, 26, 2261–2267. [Google Scholar] [CrossRef]
- Gilbertson, H.R.; Brand-Miller, J.C.; Thorburn, A.W.; Evans, S.; Chondros, P.; Werther, G.A. The effect of flexible low glycemic index dietary advice versus measured carbohydrate exchange diets on glycemic control in children with type 1 diabetes. Diabetes Care 2001, 24, 1137–1143. [Google Scholar] [CrossRef]
- Salmerón, J.; Manson, J.E.; Stampfer, M.J.; Colditz, G.A.; Wing, A.L.; Willett, W.C. Dietary fiber, glycemic load, and risk of non-insulin-dependent diabetes mellitus in women. J. Am. Med. Assoc. 1997, 277, 472–477. [Google Scholar] [CrossRef]
- Ludwig, D.S. The glycemic index: Physiological mechanisms relating to obesity, diabetes, and cardiovascular disease. J. Am. Med. Assoc. 2002, 287, 2414–2423. [Google Scholar] [CrossRef]
- Liu, S.; Willett, W.C.; Stampfer, M.J.; Hu, F.B.; Franz, M.; Sampson, L.; Hennekens, C.H.; Manson, J.E. A prospective study of dietary glycemic load, carbohydrate intake, and risk of coronary heart disease in US women. Am. J. Clin. Nutr. 2000, 71, 1455–1461. [Google Scholar] [CrossRef]
- Food and Agriculture Organization. Carbohydrates in Human Nutrition: Report of a Joint FAO/WHO Expert Consultation. Rome, 1997; FAO: Rome, Italy, 1998. [Google Scholar]
- Atkinson, F.S.; Foster-Powell, K.; Brand-Miller, J.C. International tables of glycemic index and glycemic load values: 2008. Diabetes Care 2008, 31, 2281–2283. [Google Scholar] [CrossRef]
- Flint, A.; Møller, B.; Raben, A.; Pedersen, D.; Tetens, I.; Holst, J.; Astrup, A. The use of glycaemic index tables to predict glycaemic index of composite breakfast meals. Br. J. Nutr. 2004, 91, 979–989. [Google Scholar] [CrossRef]
- Wolever, T.M.S.; Yang, M.; Zeng, X.Y.; Atkinson, F.; Brand-Miller, J.C. Food glycemic index, as given in Glycemic Index tables, is a significant determinant of glycemic responses elicited by composite breakfast meals. Am. J. Clin. Nutr. 2006, 83, 1306–1312. [Google Scholar] [CrossRef]
- Bland, J.M.; Altman, D.G. Measuring agreement in method comparison studies. Stat. Methods Med. Res. 1999, 8, 135–160. [Google Scholar] [CrossRef]
- AOAC International. Official Methods of Analysis of AOAC International; AOAC International: Arlington, VA, USA, 2011. [Google Scholar]
- International Standard Organization. Food Products—Determination of the Glycaemic Index (GI) and Recommendation for Food Classification; International Standard Organization: Geneva, Switzerland, 2010; ISO 26642:2010(E). [Google Scholar]
- Jenkins, D.J.A.; Wolever, T.M.S.; Taylor, R.H. Glycemic index of foods: A physiological basis for carbohydrate exchange. Am. J. Clin. Nutr. 1981, 34, 362–366. [Google Scholar] [CrossRef]
- Zeevi, D.; Korem, T.; Zmora, N.; Israeli, D.; Rothschild, D.; Weinberger, A.; Ben-Yacov, O.; Lador, D.; Avnit-Sagi, T.; Lotan-Pompan, M.; et al. Personalized Nutrition by Prediction of Glycemic Responses. Cell 2015, 163, 1079–1095. [Google Scholar] [CrossRef]
- Online Glycemic Index Database University of Sidney. Available online: www.glycemicindex.com (accessed on 9 February 2018).
- Deibert, P.; König, D.; Kloock, B.; Groenefeld, M.; Berg, A. Glycaemic and insulinaemic properties of some German honey varieties. Eur. J. Clin. Nutr. 2010, 64, 762–764. [Google Scholar] [CrossRef]
- Granito, M.; Pérez, S.; Valero, Y. Quality of cooking, acceptability and glycemic index of enriched pasta with legumes. Rev. Chil. Nutr. 2014, 41, 425–432. [Google Scholar] [CrossRef]
- Sonia, S.; Witjaksono, F.; Ridwan, R. Effect of cooling of cooked white rice on resistant starch content and glycemic response. Asia Pac. J. Clin. Nutr. 2015, 24, 620–625. [Google Scholar]
- Englyst, K.N.; Englyst, H.N.; Hudson, G.J.; Cole, T.J.; Cummings, J.H. Rapidly available glucose in foods: An in vitro measurement that reflects the glycemic response. Am. J. Clin. Nutr. 1999, 69, 448–454. [Google Scholar] [CrossRef]
- Nuttall, F.Q.; Mooradian, A.D.; Gannon, M.C.; Billington, C.J.; Krezowski, P. Effect of protein ingestion on the glucose and insulin response to a standardized oral glucose load. Diabetes Care 1984, 7, 465–470. [Google Scholar] [CrossRef]
- Brand-Miller, J.; Atkinson, F.; Rowan, A. Effect of added carbohydrates on glycemic and insulin responses to children’s milk products. Nutrients 2013, 5, 23–31. [Google Scholar] [CrossRef]
- Lilly, L.N.; Heiss, C.J.; Maragoudakis, S.F.; Braden, K.L.; Smith, S.E. The Effect of Added Peanut Butter on the Glycemic Response to a High–Glycemic Index Meal: A Pilot Study. J. Am. Coll. Nutr. 2019, 38, 351–357. [Google Scholar] [CrossRef]
- Meynier, A.; Goux, A.; Atkinson, F.; Brack, O.; Vinoy, S. Postprandial glycaemic response: How is it influenced by characteristics of cereal products? Br. J. Nutr. 2015, 113, 1931–1939. [Google Scholar] [CrossRef]
- Ramdath, D.D.; Padhi, E.; Hawke, A.; Sivaramalingam, T.; Tsao, R. The glycemic index of pigmented potatoes is related to their polyphenol content. Food Funct. 2014, 5, 909–915. [Google Scholar] [CrossRef]
- Jenkins, D.J.A.; Wolever, T.M.S.; Jenkins, A.L.; Lee, R.; Wong, G.S.; Josse, R. Glycemic response to wheat products: Reduced response to pasta but no effect of fiber. Diabetes Care 1983, 6, 155–159. [Google Scholar] [CrossRef]
- Shafaeizadeh, S.; Muhardi, L.; Henry, C.J.; van de Heijning, B.J.M.; van der Beek, E.M. Macronutrient composition and food form affect glucose and insulin responses in humans. Nutrients 2018, 10, 188. [Google Scholar] [CrossRef]
- Argyri, K.; Athanasatou, A.; Bouga, M.; Kapsokefalou, M. The potential of an in vitro digestion method for predicting glycemic response of foods and meals. Nutrients 2016, 8, 42. [Google Scholar] [CrossRef]
- Bellmann, S.; Minekus, M.; Sanders, P.; Bosgra, S.; Havenaar, R. Human glycemic response curves after intake of carbohydrate foods are accurately predicted by combining in vitro gastrointestinal digestion with in silico kinetic modeling. Clin. Nutr. Exp. 2018, 17, 8–22. [Google Scholar] [CrossRef]
- Ranawana, V.; Kaur, B. Role of Proteins in Insulin Secretion and Glycemic Control. Adv. Food Nutr. Res. 2013, 70, 1–47. [Google Scholar]
- Jenkins, D.J.A.; Kendall, C.W.C.; Axelsen, M.; Augustin, L.S.A.; Vuksan, V. Viscous and nonviscous fibres, nonabsorbable and low glycaemic index carbohydrates, blood lipids and coronary heart disease. Curr. Opin. Lipidol. 2000, 11, 49–56. [Google Scholar] [CrossRef]
- Wolever, T.M.S.; Jenkins, A.L.; Prudence, K.; Johnson, J.; Duss, R.; Chu, Y.; Steinert, R.E. Effect of adding oat bran to instant oatmeal on glycaemic response in humans-a study to establish the minimum effective dose of oat β-glucan. Food Funct. 2018, 9, 1692–1700. [Google Scholar] [CrossRef]
- Wood, P.J.; Beer, M.U.; Butler, G. Evaluation of role of concentration and molecular weight of oat β-glucan in determining effect of viscosity on plasma glucose and insulin following an oral glucose load. Br. J. Nutr. 2000, 84, 19–23. [Google Scholar]
- Lau, E.; Zhou, W.; Henry, C.J. Effect of fat type in baked bread on amylose-lipid complex formation and glycaemic response. Br. J. Nutr. 2016, 115, 2122–2129. [Google Scholar] [CrossRef]
- Rytz, A.; Moser, M.; Lepage, M.; Mokdad, C.; Perrot, M.; Antille, N.; Pineau, N. Using fractional factorial designs with mixture constraints to improve nutritional value and sensory properties of processed food. Food Qual. Prefer. 2017, 58, 71–75. [Google Scholar] [CrossRef]
| Process | Sucrose (g/100) | Starch (g/100) | Fiber Sol. (g/100) | Fiber Ins. (g/100) | Fat (g/100) | Protein (g/100) | GI | GI pred. | GL (g) | GL pred. (g) | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| P01 | Extruded | 11.8 | 61.2 | 2.4 | 7.1 | 2.2 | 9.1 | 83 | 79 | 18.2 | 17.2 |
| P02 | Extruded | 10.9 | 71.0 | 0.7 | 2.0 | 2.1 | 6.9 | 83 | 82 | 20.4 | 20.2 |
| P03 | Extruded | 6.3 | 68.0 | 1.9 | 5.8 | 3.3 | 8.3 | 83 | 82 | 18.5 | 18.3 |
| P04 | Flakes | 14.9 | 56.8 | 2.8 | 8.3 | 2.2 | 10.3 | 82 | 71 | 17.6 | 15.3 |
| P05 | Extruded | 16.8 | 57.5 | 2.2 | 6.7 | 2.1 | 8.5 | 80 | 77 | 17.8 | 17.3 |
| P06 | Flakes | 11.8 | 64.2 | 1.7 | 5.1 | 1.4 | 9.3 | 79 | 74 | 18.0 | 16.9 |
| P07 | Flakes | 28.6 | 53.7 | 0.8 | 2.3 | 2.5 | 6.3 | 79 | 72 | 19.5 | 17.8 |
| P08 | Extruded | 4.1 | 74.7 | 0.7 | 2.0 | 1.4 | 12.3 | 78 | 82 | 18.4 | 19.3 |
| P09 | Extruded | 28.8 | 52.0 | 1.1 | 3.3 | 2.7 | 6.3 | 78 | 72 | 18.9 | 17.4 |
| P10 | Extruded | 4.0 | 62.6 | 3.2 | 9.7 | 2.1 | 12.3 | 75 | 78 | 15.0 | 15.6 |
| P11 | Extruded | 25.2 | 48.8 | 2.2 | 6.5 | 4.0 | 7.8 | 75 | 70 | 16.7 | 15.5 |
| P12 | Extruded | 30.0 | 46.9 | 1.5 | 4.4 | 4.8 | 6.2 | 73 | 70 | 16.8 | 16.1 |
| P13 | Flakes | 11.4 | 69.8 | 1.0 | 2.9 | 2.1 | 7.1 | 73 | 76 | 17.8 | 18.5 |
| P14 | Bar | 27.4 | 41.0 | 1.4 | 4.1 | 7.1 | 6.3 | 72 | 70 | 14.8 | 14.3 |
| P15 | Extruded | 29.8 | 44.3 | 1.9 | 5.7 | 3.9 | 8.4 | 72 | 68 | 16.0 | 15.2 |
| P16 | Extruded | 25.0 | 51.2 | 1.6 | 4.8 | 5.0 | 7.2 | 72 | 70 | 16.5 | 16.1 |
| P17 | Bar | 27.9 | 40.6 | 1.5 | 4.5 | 7.3 | 6.2 | 71 | 70 | 14.6 | 14.3 |
| P18 | Flakes | 17.2 | 60.0 | 1.6 | 4.7 | 1.2 | 8.6 | 71 | 73 | 16.4 | 17.0 |
| P19 | Bar | 21.3 | 48.2 | 1.3 | 3.8 | 6.5 | 5.8 | 70 | 73 | 14.6 | 15.2 |
| P20 | Extruded | 29.7 | 48.3 | 1.5 | 4.6 | 3.0 | 7.6 | 70 | 70 | 16.4 | 16.4 |
| P21 | Extruded | 20.9 | 52.6 | 1.9 | 5.8 | 3.7 | 8.8 | 70 | 70 | 15.4 | 15.5 |
| P22 | Extruded | 29.8 | 49.0 | 1.3 | 4.0 | 5.0 | 4.9 | 69 | 71 | 16.3 | 16.7 |
| P23 | Bar | 22.5 | 42.7 | 1.4 | 4.3 | 9.8 | 6.3 | 68 | 69 | 13.3 | 13.6 |
| P24 | Extruded | 28.7 | 47.0 | 1.4 | 4.1 | 7.4 | 7.6 | 68 | 68 | 15.4 | 15.4 |
| P25 | Extruded | 28.8 | 47.0 | 1.6 | 4.7 | 4.5 | 8.0 | 68 | 69 | 15.5 | 15.7 |
| P26 | Flakes | 28.4 | 47.7 | 2.2 | 6.6 | 1.7 | 8.7 | 68 | 70 | 15.5 | 15.9 |
| P27 | Extruded | 24.8 | 48.6 | 1.3 | 3.9 | 10.1 | 5.7 | 67 | 68 | 14.8 | 15.0 |
| P28 | Bar | 30.0 | 33.4 | 1.0 | 3.0 | 12.9 | 7.4 | 66 | 64 | 12.6 | 12.2 |
| P29 | Extruded | 24.2 | 54.9 | 1.3 | 3.9 | 3.2 | 7.0 | 63 | 72 | 14.9 | 17.1 |
| P30 | Flakes | 0.7 | 67.1 | 3.1 | 9.3 | 2.2 | 11.5 | 62 | 62 | 12.6 | 12.6 |
| P31 | Extruded | 26.9 | 44.7 | 2.0 | 5.9 | 4.9 | 9.0 | 61 | 68 | 13.1 | 14.6 |
| P32 | Extruded | 4.7 | 62.7 | 2.1 | 6.4 | 7.5 | 11.1 | 61 | 71 | 12.3 | 14.3 |
| P33 | Muesli | 14.9 | 47.3 | 2.6 | 7.8 | 6.7 | 11.9 | 60 | 61 | 11.2 | 11.3 |
| P34 | Muesli | 11.4 | 45.5 | 2.0 | 6.1 | 11.1 | 11.8 | 59 | 58 | 10.1 | 9.9 |
| P35 | Muesli | 14.9 | 44.1 | 2.2 | 6.5 | 9.5 | 10.6 | 59 | 59 | 10.4 | 10.5 |
| P36 | Granola | 22.3 | 38.9 | 1.4 | 4.2 | 13.7 | 14.4 | 59 | 53 | 10.8 | 9.8 |
| P37 | Granola | 25.5 | 43.3 | 1.6 | 4.7 | 9.8 | 10.0 | 59 | 58 | 12.2 | 12.0 |
| P38 | Granola | 22.3 | 41.6 | 1.9 | 5.8 | 10.5 | 11.6 | 55 | 54 | 10.5 | 10.4 |
| P39 | Granola | 18.7 | 41.8 | 1.9 | 5.8 | 12.4 | 13.0 | 54 | 52 | 9.8 | 9.5 |
| P40 | Granola | 18.5 | 41.7 | 1.9 | 5.7 | 12.8 | 13.1 | 52 | 52 | 9.4 | 9.4 |
| P41 | Granola | 22.1 | 41.4 | 1.9 | 5.7 | 10.3 | 12.0 | 52 | 54 | 9.9 | 10.3 |
| P42 | Granola | 22.2 | 42.3 | 1.8 | 5.4 | 9.8 | 13.5 | 50 | 56 | 9.7 | 10.8 |
| Min | 0.7 | 33.4 | 0.7 | 2.0 | 1.2 | 4.9 | 50 | 52 | 9.4 | 9.4 | |
| Max | 30.0 | 74.7 | 3.2 | 9.7 | 13.7 | 14.4 | 83 | 82 | 20.4 | 20.2 | |
| Mean | 20.1 | 51.1 | 1.7 | 5.2 | 5.9 | 9.0 | 68 | 68 | 14.7 | 14.7 | |
| SD | 8.5 | 10.0 | 0.6 | 1.7 | 3.8 | 2.5 | 9.2 | 8.2 | 3.1 | 2.9 | |
| Glycemic Carbohydrates | GIi | ai | |
|---|---|---|---|
| Monosaccharides | Glucose | 100 | 1 |
| Fructose | 20 | 1 | |
| Galactose | 20 | 1 | |
| Disaccharides | Maltose | 105 | 1 |
| Trehalose | 70 | 1 | |
| Sucrose | 62 | 1 | |
| Lactose | 47 | 1 | |
| Isomaltulose | 32 | 1 | |
| Polysaccharides | Maltotriose | 110 | 1 |
| Maltotetraose | 110 | 1 | |
| Starch | 110 | %RDS/100 | |
| Maltodextrin | 110 | 1 | |
| Sugar alcohols | Maltitol | 35 | 1 |
| Xylitol | 12 | 1 | |
| GI-lowering Macronutrients | bi | |
|---|---|---|
| Carbohydrates | β-glucan | 0.6 |
| Fiber soluble | 0.3 | |
| Fiber insoluble | 0.1 | |
| Others | Fat | 0.6 |
| Protein | 0.6 | |
| Ashes | 0.1 | |
| Water | 0.0 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rytz, A.; Adeline, D.; Lê, K.-A.; Tan, D.; Lamothe, L.; Roger, O.; Macé, K. Predicting Glycemic Index and Glycemic Load from Macronutrients to Accelerate Development of Foods and Beverages with Lower Glucose Responses. Nutrients 2019, 11, 1172. https://doi.org/10.3390/nu11051172
Rytz A, Adeline D, Lê K-A, Tan D, Lamothe L, Roger O, Macé K. Predicting Glycemic Index and Glycemic Load from Macronutrients to Accelerate Development of Foods and Beverages with Lower Glucose Responses. Nutrients. 2019; 11(5):1172. https://doi.org/10.3390/nu11051172
Chicago/Turabian StyleRytz, Andreas, Dorothée Adeline, Kim-Anne Lê, Denise Tan, Lisa Lamothe, Olivier Roger, and Katherine Macé. 2019. "Predicting Glycemic Index and Glycemic Load from Macronutrients to Accelerate Development of Foods and Beverages with Lower Glucose Responses" Nutrients 11, no. 5: 1172. https://doi.org/10.3390/nu11051172
APA StyleRytz, A., Adeline, D., Lê, K.-A., Tan, D., Lamothe, L., Roger, O., & Macé, K. (2019). Predicting Glycemic Index and Glycemic Load from Macronutrients to Accelerate Development of Foods and Beverages with Lower Glucose Responses. Nutrients, 11(5), 1172. https://doi.org/10.3390/nu11051172





