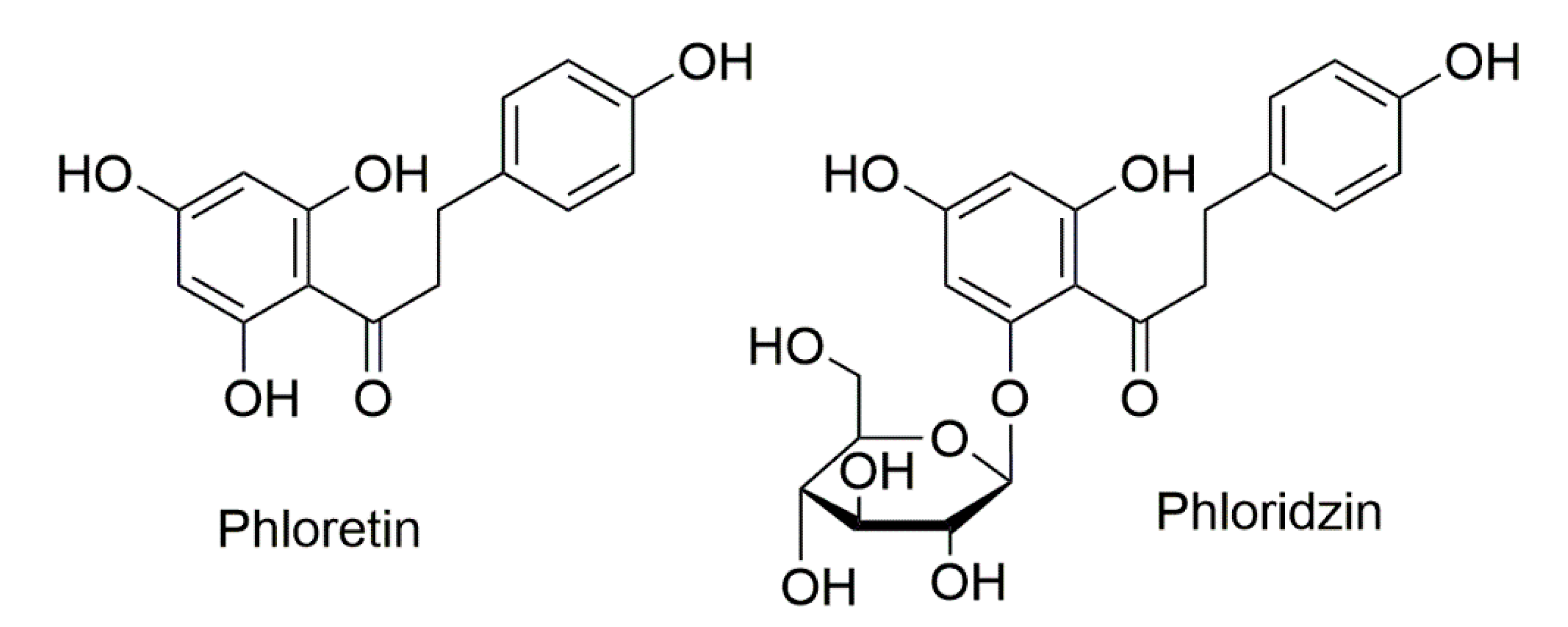
Role of Apple Phytochemicals, Phloretin and Phloridzin, in Modulating Processes Related to Intestinal Inflammation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Apple Extracts’ Preparation for the Phldz and Phlor Analysis
2.3. Analysis of Phlor and Phldz by HPLC-DAD-MS/MS
2.4. Cell Culture Conditions
2.5. Cell Viability Assay
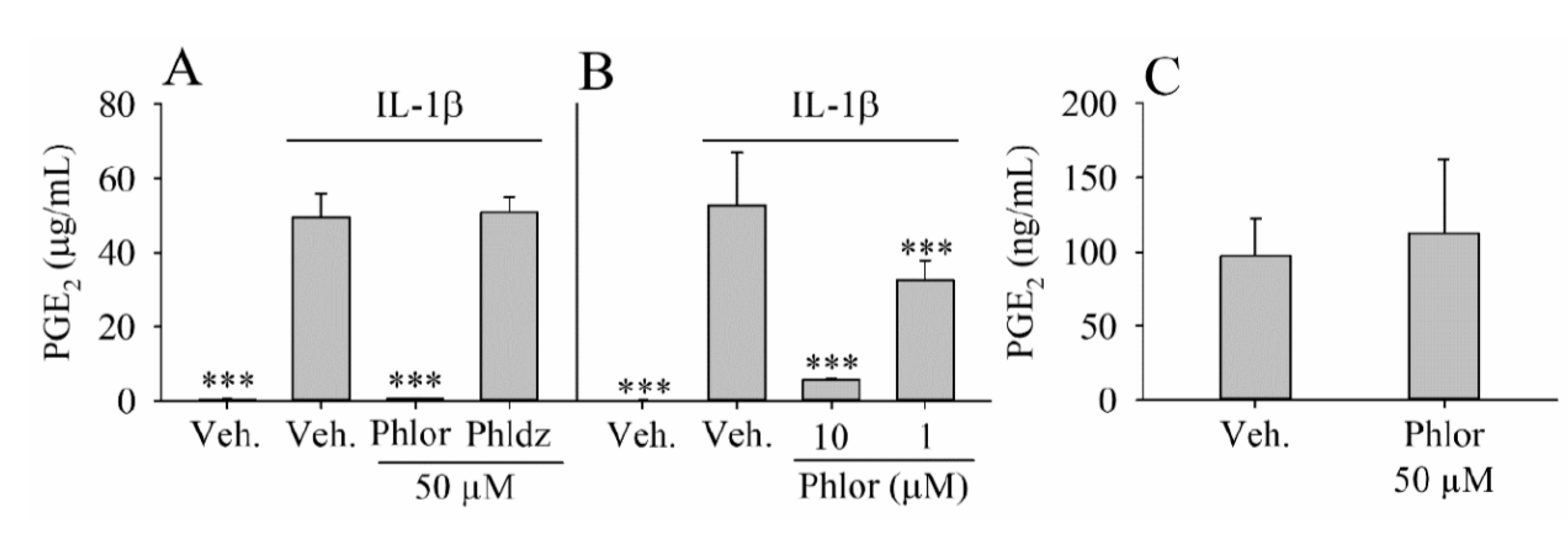
2.6. Effect of Phlor and Phldz on the PGE2 Biosynthesis in IL-1β-Stimulated Myofibroblasts
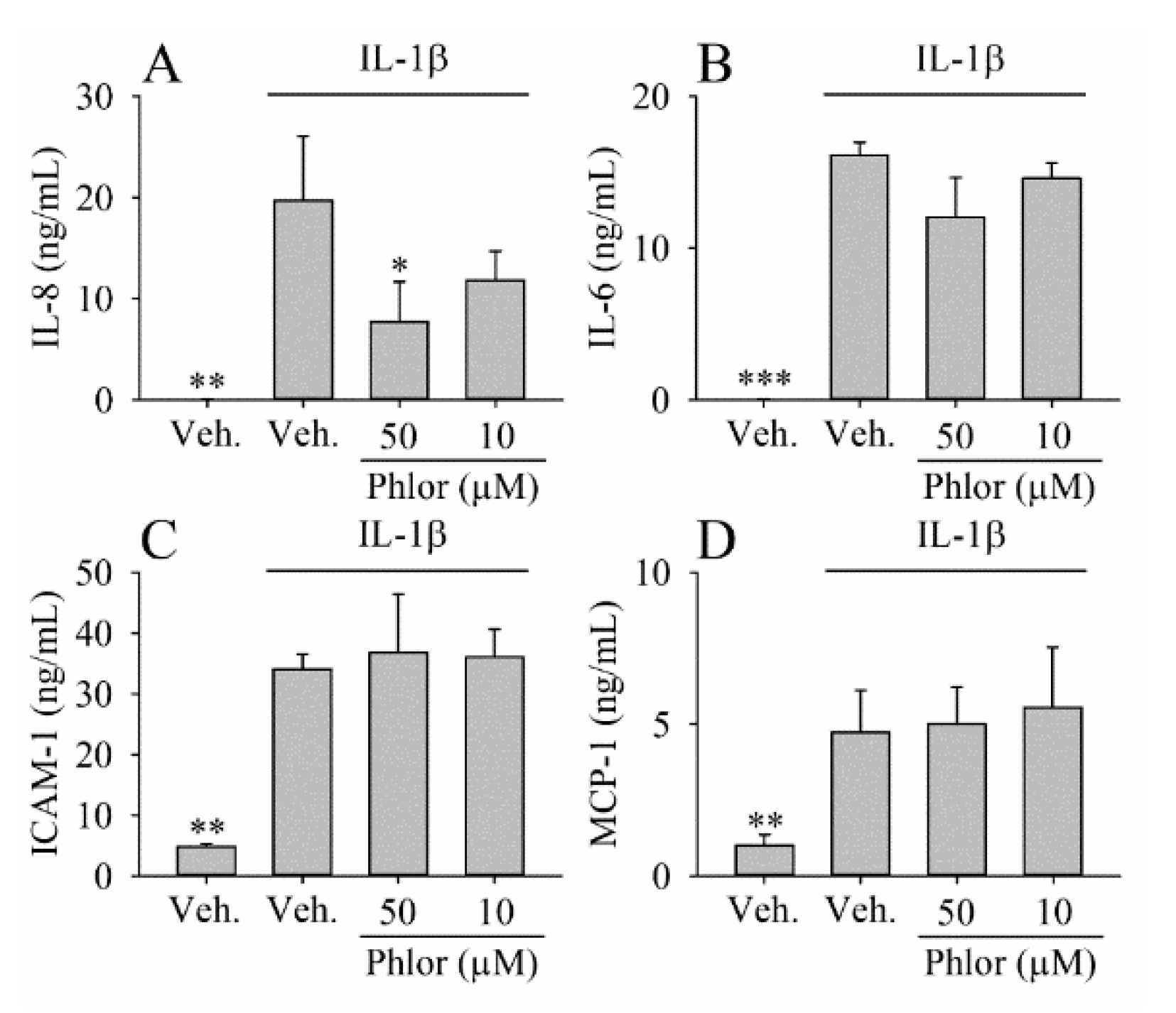
2.7. Effect of Phlor on Cytokine Production in IL-1β-Stimulated Myofibroblasts
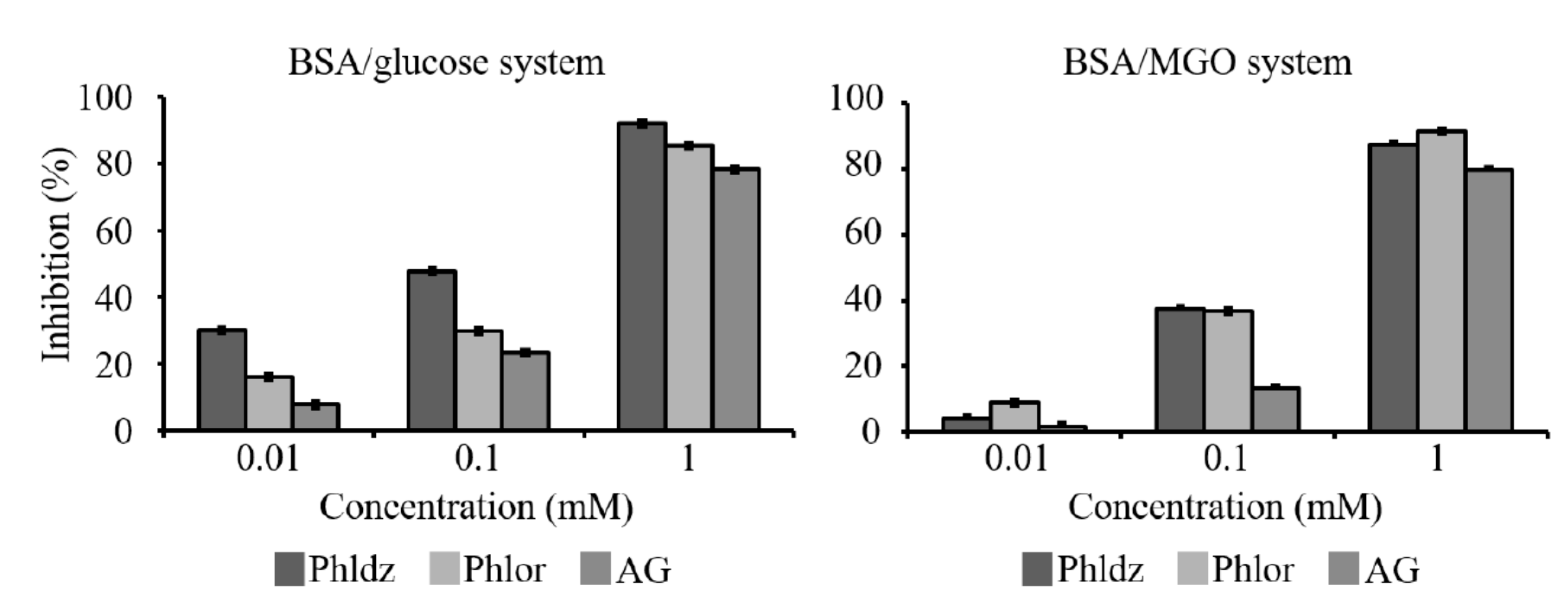
2.8. Inhibition of the Formation of the Advanced Glycation End Products (AGEs)
2.9. Antioxidant Activity
2.10. Reducing Activity
2.11. Ferric-Reducing/Antioxidant Power (FRAP) Activity
2.12. Chelating Activity
2.13. Statistical Analysis
3. Results
3.1. Phlor and Phldz Contents in Apple Peel and Flesh
3.2. Inhibition of PGE2 Biosynthesis by Phlor and Phldz
3.3. Effect of Phlor on Cytokines Biosynthesis in IL-1β-Stimulated Myofibroblasts
3.4. Inhibition of In Vitro AGEs’ Formation by Phlor and Phldz
3.5. The Antioxidant, Reducing and Chelating Activity of Phlor and Phldz
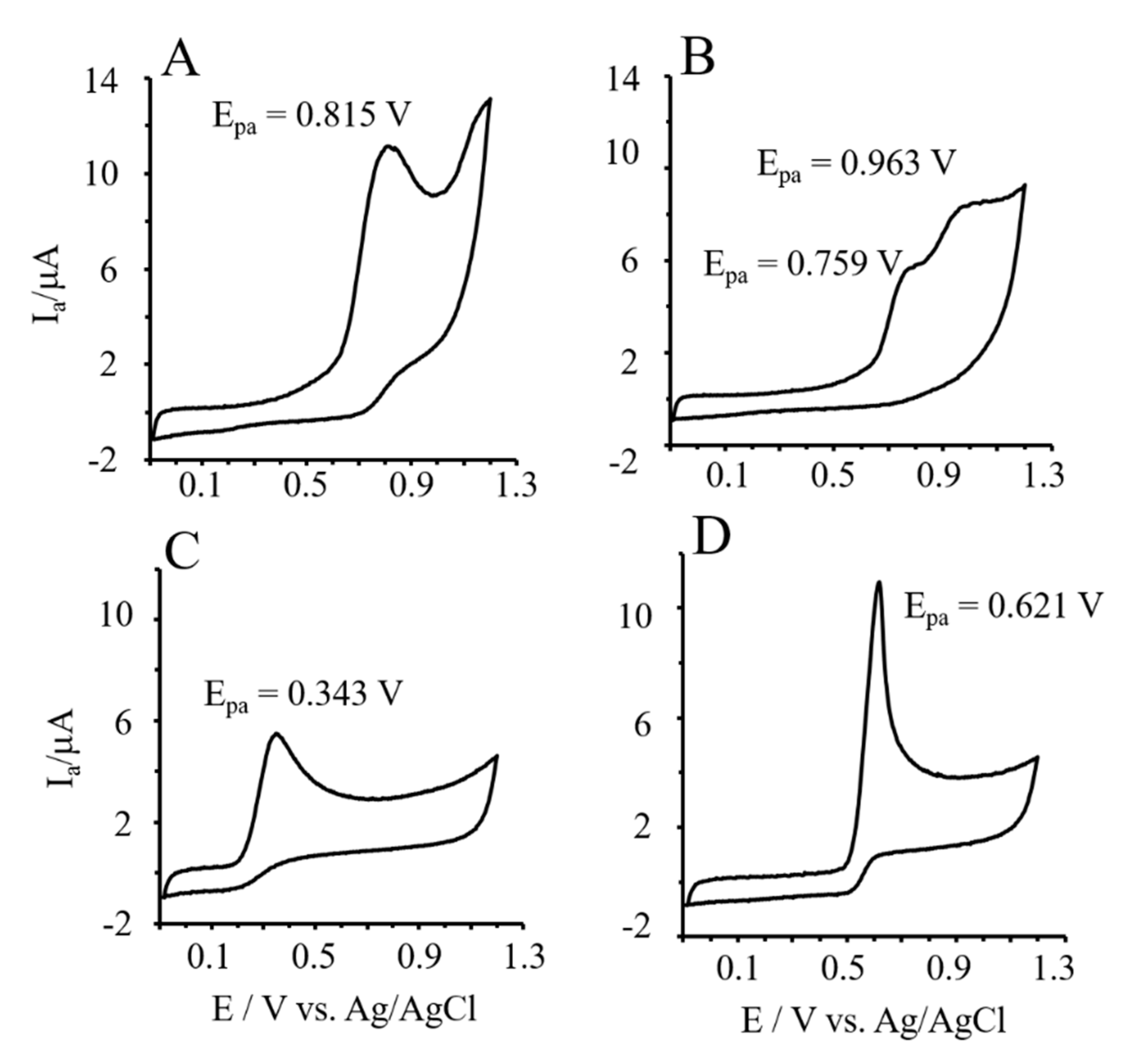
3.6. Reducing Power of Phlor and Phldz by Cyclic Voltammetry
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Chiba, M.; Nakane, K.; Komatsu, M. Westernized diet is the most ubiquitous environmental factor in inflammatory bowel disease. Perm. J. 2019, 23, 18–107. [Google Scholar] [CrossRef]
- Yuan, X.; Zhao, J.; Qu, W.; Zhang, Y.; Jia, B.; Fan, Z.; He, Q.; Li, J. Accumulation and effects of dietary advanced glycation end products on the gastrointestinal tract in rats. Int. J. Food Sci. Technol. 2018, 53, 2273–2281. [Google Scholar] [CrossRef]
- Cario, E. Toll-like receptors in inflammatory bowel diseases: A decade later. Inflamm. Bowel. Dis. 2010, 16, 1583–1597. [Google Scholar] [CrossRef]
- Ciccocioppo, R.; Vanoli, A.; Klersy, C.; Imbesi, V.; Boccaccio, V.; Manca, R.; Betti, E.; Cangemi, G.C.; Strada, E.; Besio, R.; et al. Role of the advanced glycation end products receptor in Crohn’s disease inflammation. World J. Gastroenterol. 2013, 19, 8269–8281. [Google Scholar] [CrossRef]
- Blander, J.M. Death in the intestinal epithelium-basic biology and implications for inflammatory bowel disease. FEBS J. 2016, 283, 2720–2730. [Google Scholar] [CrossRef] [PubMed]
- Dreher, M.L. Whole Fruits and Fruit Fiber Emerging Health Effects. Nutrients 2018, 10, 1833. [Google Scholar] [CrossRef]
- Wang, S.; Li, Q.; Zang, Y.; Zhao, Y.; Liu, N.; Wang, Y.; Xu, X.; Liu, L.; Mei, Q. Apple Polysaccharide inhibits microbial dysbiosis and chronic inflammation and modulates gut permeability in HFD-fed rats. Int. J. Biol. Macromol. 2017, 99, 282–292. [Google Scholar] [CrossRef] [PubMed]
- Yeganeh, P.R.; Leahy, J.; Spahis, S.; Patey, N.; Desjardins, Y.; Roy, D.; Delvin, E.; Garofalo, C.; Leduc-Gaudet, J.P.; St-Pierre, D.; et al. Apple peel polyphenols reduce mitochondrial dysfunction in mice with DSS-induced ulcerative colitis. J. Nutr. Biochem. 2018, 57, 56–66. [Google Scholar] [CrossRef]
- Sies, H. Polyphenols and health: Update and perspectives. Arch. Biochem. Biophys. 2010, 501, 2–5. [Google Scholar] [CrossRef] [PubMed]
- Hertog, M.G.; Hollman, P.C.H.; Katan, M.B. Content of potentially anticarcinogenic flavonoids of 28 vegetables and 9 fruits commonly consumed in the Netherlands. J. Agric. Food Chem. 1992, 12, 2379–2383. [Google Scholar] [CrossRef]
- Tsao, R.; Yang, R.; Young, J.C.; Zhu, H. Polyphenolic profiles in eight apple cultivars using high-performance liquid chromatography (HPLC). J. Agric. Food Chem. 2003, 51, 6347–6353. [Google Scholar] [CrossRef]
- Crespy, V.; Aprikian, O.; Morand, C.; Besson, C.; Manach, C.; Demigne, C.; Remesy, C. Bioavailability of phloretin and phloridzin in rats. J. Nutr. 2001, 131, 3227–3230. [Google Scholar] [CrossRef] [PubMed]
- Mennen, L.I.; Sapinho, D.; Ito, H.; Bertrais, S.; Galan, P.; Hercberg, S.; Scalbert, A. Urinary flavonoids and phenolic acids as biomarkers of intake for polyphenol-rich foods. Br. J. Nutr. 2006, 96, 191–198. [Google Scholar] [CrossRef]
- Jeon, D.; Jeong, M.C.; Jnawali, H.N.; Kwak, C.; Ryoo, S.; Jung, I.D.; Kim, Y. Phloretin exerts anti-tuberculosis activity and suppresses lung inflammation. Molecules 2017, 22, 183. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Yang, T.; Wang, T.; Hao, N.; Shen, Y.; Wu, Y.; Yuan, Z.; Chen, L.; Wen, F. Phloretin attenuates mucus hypersecretion and airway inflammation induced by cigarette smoke. Int. Immunopharmacol. 2018, 55, 112–119. [Google Scholar] [CrossRef]
- Khalifa, M.M.A.; Bakr, A.G.; Osman, A.T. Protective effects of phloridzin against methotrexate-induced liver toxicity in rats. Biomed. Pharm. 2017, 95, 529–535. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Regmi, S.C.; Kim, J.A.; Cho, M.H.; Yun, H.; Lee, C.S.; Lee, J. Apple flavonoid phloretin inhibits Escherichia coli O157:H7 biofilm formation and ameliorates colon inflammation in rats. Infect. Immun. 2011, 79, 4819–4827. [Google Scholar] [CrossRef]
- Zhang, Z.; Li, S.; Cao, H.; Shen, P.; Liu, J.; Fu, Y.; Cao, Y.; Zhang, N. The protective role of phloretin against dextran sulfate sodium-induced ulcerative colitis in mice. Food Funct. 2019, 10, 422–431. [Google Scholar] [CrossRef]
- Gimenez-Bastida, J.A.; Zielinski, H.; Piskula, M.; Zielinska, D.; Szawara-Nowak, D. Buckwheat bioactive compounds, their derived phenolic metabolites and their health benefits. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Thornalley, P.J. Use of aminoguanidine (Pimagedine) to prevent the formation of advanced glycation endproducts. Arch. Biochem. Biophys. 2003, 419, 31–40. [Google Scholar] [CrossRef]
- Franke, A.A.; Custer, L.J.; Arakaki, C.; Murphy, S.P. Vitamin C and flavonoid levels of fruits and vegetables consumed in Hawaii. J. Food Comp. Anal. 2004, 17, 1–35. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free-radical method to evaluate antioxidant activity. Food Sci. Technol. Leb. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Zielinska, D.; Zielinski, H. Antioxidant activity of flavone C-glucosides determined by updated analytical strategies. Food Chem. 2011, 124, 672–678. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. Ferric reducing antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Method Enzym. 1999, 299, 15–27. [Google Scholar]
- Mladenka, P.; Zatloukalova, L.; Filipsky, T.; Hrdina, R. Cardiovascular effects of flavonoids are not caused only by direct antioxidant activity. Free Radic. Biol. Med. 2010, 49, 963–975. [Google Scholar] [CrossRef]
- Hossain, M.A.; Salehuddin, S.M.; Kabir, M.J.; Rahman, S.M.M.; Rupasinghe, H.P.V. Sinensetin, rutin, 3’-hydroxy-5,6,7,4’-teramethoxyflavone and rosmarinic acid contents and antioxidative effect of the skin of apple fruit. Food Chem. 2009, 113, 185–190. [Google Scholar] [CrossRef]
- Kondo, S.; Tsuda, K.; Muto, N.; Ueda, J. Antioxidant activity of apple skin or flesh extracts associated with fruit development on selected apple cultivars. Sci. Hortic. 2002, 96, 177–185. [Google Scholar] [CrossRef]
- Lu, Y.; Foo, L.Y. Antioxidant and radical scavenging activities of polyphenols from apple pomace. Food Chem. 2000, 68, 81–85. [Google Scholar] [CrossRef]
- Oszmiański, J.; Wolniak, M.; Wojdyło, A.; Wawer, I. Influence of apple puree preparation and storage on polyphenol contents and antioxidant activity. Food Chem. 2008, 107, 1473–1484. [Google Scholar] [CrossRef]
- Dragovic-Uzelac, V.; Pospisil, J.; Levaj, B.; Delonga, K. The study of phenolic profiles of raw apricots and apples and their purees by HPLC for the evaluation of apricot nectars and jams authenticity. Food Chem. 2005, 91, 373–383. [Google Scholar] [CrossRef]
- Giomaro, G.; Karioti, A.; Bilia, A.R.; Bucchini, A.; Giamperi, L.; Ricci, D.; Fraternale, D. Polyphenols profile and antioxidant activity of skin and pulp of a rare apple from Marche region (Italy). Chem. Cent. J. 2014, 8, 45. [Google Scholar] [CrossRef]
- Espley, R.V.; Butts, C.A.; Laing, W.A.; Martell, S.; Smith, H.; McGhie, T.K.; Zhang, J.; Paturi, G.; Hedderley, D.; Bovy, A.; et al. Dietary flavonoids from modified apple reduce inflammation markers and modulate gut microbiota in mice. J. Nutr. 2014, 144, 146–154. [Google Scholar] [CrossRef]
- Castagnini, C.; Luceri, C.; Toti, S.; Bigagli, E.; Caderni, G.; Femia, A.P.; Giovannelli, L.; Lodovici, M.; Pitozzi, V.; Salvadori, M.; et al. Reduction of colonic inflammation in HLA-B27 transgenic rats by feeding Marie Menard apples, rich in polyphenols. Br. J. Nutr. 2009, 102, 1620–1628. [Google Scholar] [CrossRef]
- Schaefer, S.; Baum, M.; Eisenbrand, G.; Dietrich, H.; Will, F.; Janzowski, C. Polyphenolic apple juice extracts and their major constituents reduce oxidative damage in human colon cell lines. Mol. Nutr. Food Res. 2006, 50, 24–33. [Google Scholar] [CrossRef]
- Jung, M.; Triebel, S.; Anke, T.; Richling, E.; Erkel, G. Influence of apple polyphenols on inflammatory gene expression. Mol. Nutr. Food Res. 2009, 53, 1263–1280. [Google Scholar] [CrossRef]
- Kahle, K.; Huemmer, W.; Kempf, M.; Scheppach, W.; Erk, T.; Richling, E. Polyphenols are intensively metabolized in the human gastrointestinal tract after apple juice consumption. J. Agric. Food Chem. 2007, 55, 10605–10614. [Google Scholar] [CrossRef]
- Marks, S.C.; Mullen, W.; Borges, G.; Crozier, A. Absorption, metabolism, and excretion of cider dihydrochalcones in healthy humans and subjects with an ileostomy. J. Agric. Food Chem. 2009, 57, 2009–2015. [Google Scholar] [CrossRef]
- De Oliveira, M.R. Phloretin-induced cytoprotective effects on mammalian cells: A mechanistic view and future directions. Biofactors 2016, 42, 13–40. [Google Scholar] [CrossRef]
- Avila-Galvez, M.A.; Gonzalez-Sarrias, A.; Espin, J.C. In vitro research on dietary polyphenols and health: A call of caution and a guide on how to proceed. J. Agric. Food Chem. 2018, 66, 7857–7858. [Google Scholar] [CrossRef]
- Andoh, A.; Fujino, S.; Okuno, T.; Fujiyama, Y.; Bamba, T. Intestinal subepithelial myofibroblasts in inflammatory bowel diseases. J. Gastroenterol. 2002, 37 (Suppl. 14), 33–37. [Google Scholar] [CrossRef]
- Wang, D.; Dubois, R.N. The role of COX-2 in intestinal inflammation and colorectal cancer. Oncogene 2010, 29, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Levenson, A.S.; Biswas, P.K. Structural insights into Resveratrol’s antagonist and partial agonist actions on estrogen receptor alpha. BMC Struct. Biol. 2013, 13, 27. [Google Scholar] [CrossRef]
- Gonzalez-Sarrias, A.; Larrosa, M.; Tomas-Barberan, F.A.; Dolara, P.; Espin, J.C. NF-kappaB-dependent anti-inflammatory activity of urolithins, gut microbiota ellagic acid-derived metabolites, in human colonic fibroblasts. Br. J. Nutr. 2010, 104, 503–512. [Google Scholar] [CrossRef]
- Gimenez-Bastida, J.A.; Larrosa, M.; Gonzalez-Sarrias, A.; Tomas-Barberan, F.; Espin, J.C.; Garcia-Conesa, M.T. Intestinal ellagitannin metabolites ameliorate cytokine-induced inflammation and associated molecular markers in human colon fibroblasts. J. Agric. Food Chem. 2012, 60, 8866–8876. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Wu, S.J.; Tu, R.S.; Lai, Y.R.; Liou, C.J. Phloretin inhibits interleukin-1beta-induced COX-2 and ICAM-1 expression through inhibition of MAPK, Akt, and NF-kappaB signaling in human lung epithelial cells. Food Funct. 2015, 6, 1960–1967. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Chadee, K. Prostaglandin E2 stimulates IL-8 gene expression in human colonic epithelial cells by a posttranscriptional mechanism. J. Immunol. 1998, 161, 3746–3752. [Google Scholar]
- Cho, J.S.; Han, I.H.; Lee, H.R.; Lee, H.M. Prostaglandin E2 induces IL-6 and IL-8 production by the EP receptors/Akt/NF-kappaB pathways in nasal polyp-derived fibroblasts. Allergy Asthma Immunol. Res. 2014, 6, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.T.; Huang, W.C.; Liou, C.J. Evaluation of the anti-inflammatory effects of phloretin and phlorizin in lipopolysaccharide-stimulated mouse macrophages. Food Chem. 2012, 134, 972–979. [Google Scholar] [CrossRef]
- Huang, W.C.; Dai, Y.W.; Peng, H.L.; Kang, C.W.; Kuo, C.Y.; Liou, C.J. Phloretin ameliorates chemokines and ICAM-1 expression via blocking of the NF-kappaB pathway in the TNF-alpha-induced HaCaT human keratinocytes. Int. Immunopharmacol. 2015, 27, 32–37. [Google Scholar] [CrossRef]
- Body-Malapel, M.; Djouina, M.; Waxin, C.; Langlois, A.; Gower-Rousseau, C.; Zerbib, P.; Schmidt, A.M.; Desreumaux, P.; Boulanger, E.; Vignal, C. The RAGE signaling pathway is involved in intestinal inflammation and represents a promising therapeutic target for Inflammatory Bowel Diseases. Mucosal. Immunol. 2019, 12, 468–478. [Google Scholar] [CrossRef]
- Sampath, C.; Sang, S.; Ahmedna, M. In vitro and in vivo inhibition of aldose reductase and advanced glycation end products by phloretin, epigallocatechin 3-gallate and [6]-gingerol. Biomed. Pharm. 2016, 84, 502–513. [Google Scholar] [CrossRef] [PubMed]
- Shao, X.; Bai, N.; He, K.; Ho, C.T.; Yang, C.S.; Sang, S. Apple polyphenols, phloretin and phloridzin: New trapping agents of reactive dicarbonyl species. Chem. Res. Toxicol. 2008, 21, 2042–2050. [Google Scholar] [CrossRef] [PubMed]
- Shao, X.; Chen, H.; Zhu, Y.; Sedighi, R.; Ho, C.T.; Sang, S. Essential structural requirements and additive effects for flavonoids to scavenge methylglyoxal. J. Agric. Food Chem. 2014, 62, 3202–3210. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.H.; Huang, S.M.; Lin, J.A.; Yen, G.C. Inhibition of advanced glycation endproduct formation by foodstuffs. Food Funct. 2011, 2, 224–234. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Ma, J.; Chen, F.; Wang, M. Naturally occurring inhibitors against the formation of advanced glycation end-products. Food Funct. 2011, 2, 289–301. [Google Scholar] [CrossRef] [PubMed]
- Gulcin, I. Antioxidant activity of food constituents: An overview. Arch. Toxicol. 2012, 86, 345–391. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Chen, B.; Xie, H.; He, Y.; Zhong, D.; Chen, D. Antioxidant structure(-)activity relationship analysis of five dihydrochalcones. Molecules 2018, 23, 1162. [Google Scholar] [CrossRef]
- Vieira, F.G.; Di Pietro, P.F.; da Silva, E.L.; Borges, G.S.; Nunes, E.C.; Fett, R. Improvement of serum antioxidant status in humans after the acute intake of apple juices. Nutr. Res. 2012, 32, 229–232. [Google Scholar] [CrossRef]
- Lotito, S.B.; Frei, B. Relevance of apple polyphenols as antioxidants in human plasma: Contrasting in vitro and in vivo effects. Free Radic. Biol. Med. 2004, 36, 201–211. [Google Scholar]
| Apple Cultivar/Compound | Phloretin | Phloridzin | ||
|---|---|---|---|---|
| Peel | Flesh | Peel | Flesh | |
| Quinte | 0.96 ± 0.02 d | 0.72 ± 0.03 b | 23.91 ± 0.48 d | 18.89 ± 0.98 d |
| Jonagored | 0.91 ± 0.08 d | 0.42 ± 0.01 g | 24.65 ± 1.63 d | 7.17 ± 0.17 h |
| Early Geneva | 0.85 ± 0.02 e | 0.72 ± 0.01 b | 24.45 ± 0.49 d | 20.63 ± 0.80 c |
| Paulared | 0.64 ± 0.01 g | 0.51 ± 0.02 e | 16.43 ± 0.76 e | 10.37 ± 0.36 f |
| Sunrise | 1.38 ± 0.08 b | 0.60 ± 0.03 d | 48.08 ± 0.80 b | 14.68 ± 0.84 e |
| Gloster | 1.32 ± 0.02 b | 0.41 ± 0.00 g | 43.45 ± 2.59 c | 5.86 ± 0.16 i |
| Delikates | 0.73 ± 0.03 f | 0.47 ± 0.01 ef | 18.30 ± 1.74 e | 9.14 ± 0.06 g |
| Papierówka | 2.25 ± 0.05 a | 0.75 ± 0.03 b | 84.10 ± 1.60 a | 23.46 ± 0.77 b |
| Rubinola | 1.24 ± 0.05 c | 0.65 ± 0.03 c | 42.90 ± 3.21 c | 18.09 ± 0.22 d |
| Antonówka | 1.20 ± 0.01 c | 1.38 ± 0.08 a | 23.37 ± 0.45 d | 45.05 ± 1.79 a |
| Ligol | 0.66 ± 0.04 g | 0.43 ± 0.01 fg | 16.51 ± 0.73 e | 6.61 ± 0.14 hi |
| Compound/Assay | Antioxidant Activity (mM Trolox) | Reducing Activity (mM Trolox) | Chelating Activity (%) | |
|---|---|---|---|---|
| DPPH RSA | FRAP | CV | FZ | |
| Phloretin | 0.19 ± 0.01 c | 0.95 ± 0.02 c | 0.56 ± 0.02 b | 3.78 ± 0.47 b |
| Phloridzin | 0.06 ± 0.01 d | 0.18 ± 0.01 d | 0.36 ± 0.03 c | 3.86 ± 0.34 a |
| L-ascorbic acid | 0.70 ± 0.01 b | 1.13 ± 0.02 b | 1.04 ± 0.01 a | 1.79 ± 0.36 c |
| Uric acid | 1.08 ± 0.01 a | 1.27 ± 0.01 a | 0.98 ± 0.01 a | 0.99 ± 0.11 d |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zielinska, D.; Laparra-Llopis, J.M.; Zielinski, H.; Szawara-Nowak, D.; Giménez-Bastida, J.A. Role of Apple Phytochemicals, Phloretin and Phloridzin, in Modulating Processes Related to Intestinal Inflammation. Nutrients 2019, 11, 1173. https://doi.org/10.3390/nu11051173
Zielinska D, Laparra-Llopis JM, Zielinski H, Szawara-Nowak D, Giménez-Bastida JA. Role of Apple Phytochemicals, Phloretin and Phloridzin, in Modulating Processes Related to Intestinal Inflammation. Nutrients. 2019; 11(5):1173. https://doi.org/10.3390/nu11051173
Chicago/Turabian StyleZielinska, Danuta, José Moisés Laparra-Llopis, Henryk Zielinski, Dorota Szawara-Nowak, and Juan Antonio Giménez-Bastida. 2019. "Role of Apple Phytochemicals, Phloretin and Phloridzin, in Modulating Processes Related to Intestinal Inflammation" Nutrients 11, no. 5: 1173. https://doi.org/10.3390/nu11051173
APA StyleZielinska, D., Laparra-Llopis, J. M., Zielinski, H., Szawara-Nowak, D., & Giménez-Bastida, J. A. (2019). Role of Apple Phytochemicals, Phloretin and Phloridzin, in Modulating Processes Related to Intestinal Inflammation. Nutrients, 11(5), 1173. https://doi.org/10.3390/nu11051173







