Prolonged Collagen Peptide Supplementation and Resistance Exercise Training Affects Body Composition in Recreationally Active Men
Abstract
1. Introduction
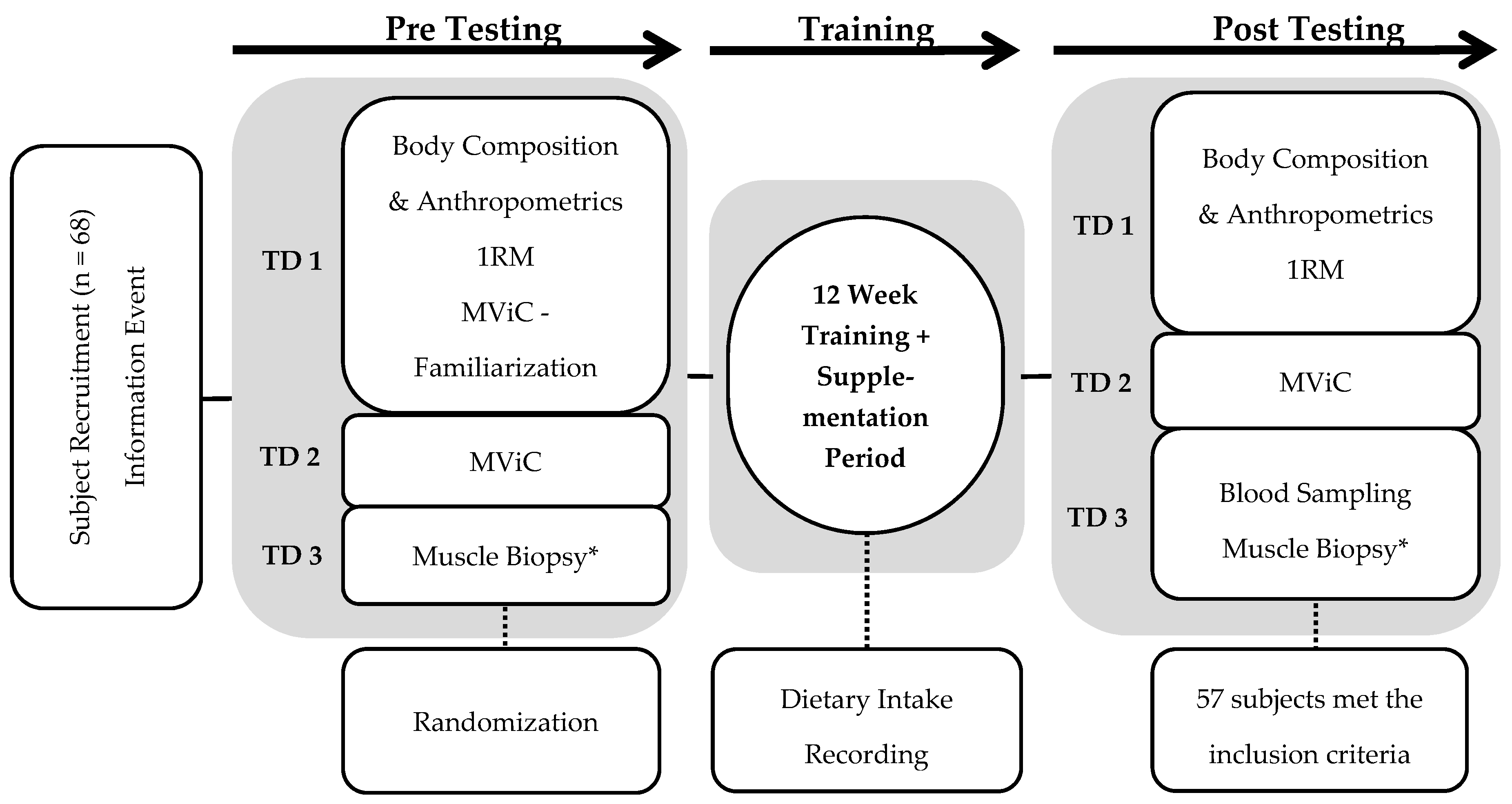
2. Materials and Methods
2.1. Subjects
2.2. Resistance Training Protocol and External Load Calculation
2.3. Dietary Protein Supplementation
2.4. Dietary Intake Recording
2.5. Body Composition Testing
2.6. Ultrasound and Anthropometric Measurement
2.7. Strength Testing
2.8. Muscle Biopsies and Venous Blood Sampling
2.9. Immunohistochemical Stains of Skeletal Muscle
2.10. Subjective Perception
2.11. Statistical Analysis
3. Results
3.1. External Load, Ultrasound, Leg Circumference, and Subjective Perception
3.2. Body Composition
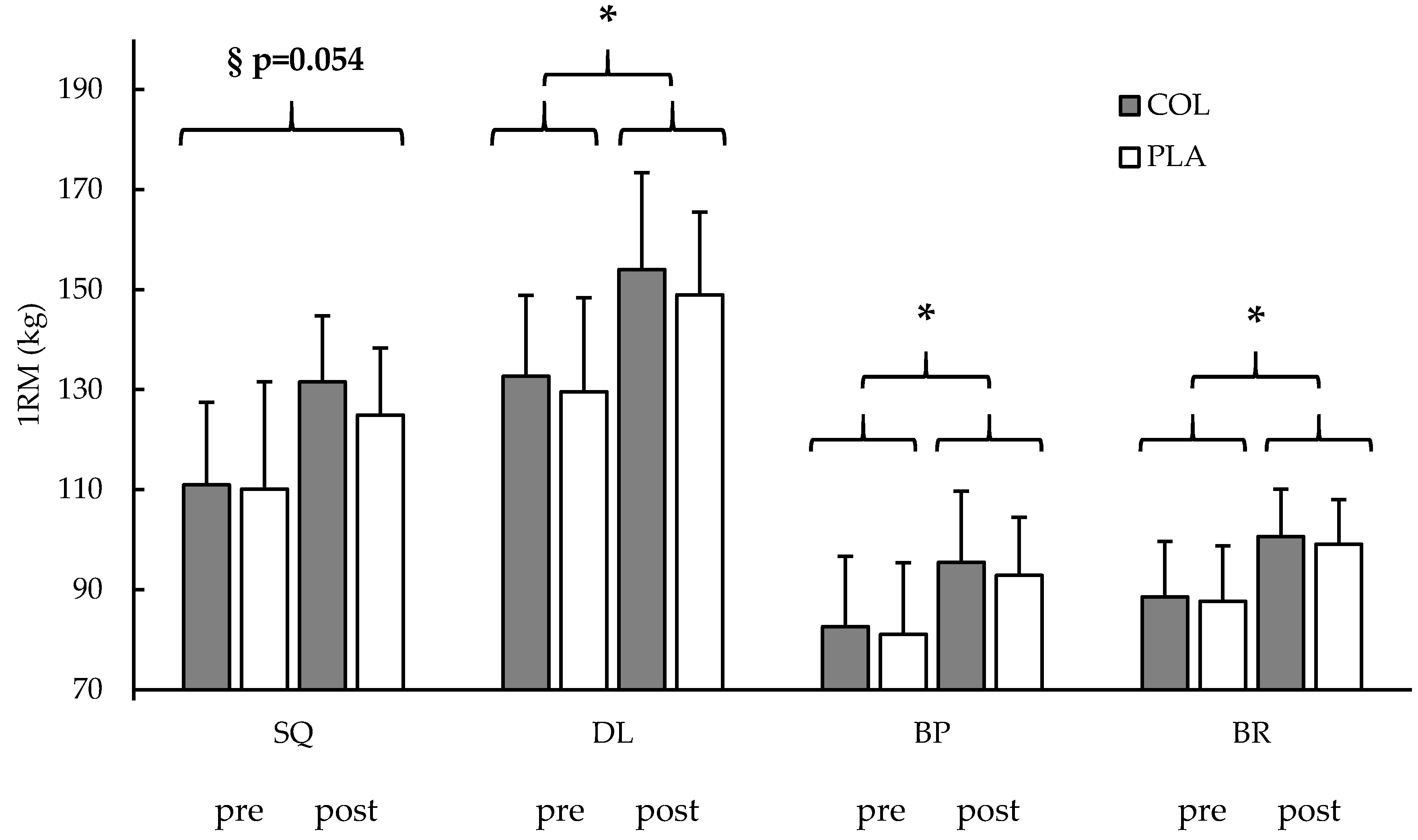
3.3. Strength Testing
3.4. Dietary Intake Recording
3.5. Venous Blood Sampling
3.6. Muscle Biopsy
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 1RM | One-repetition maximum |
| BFM | Body fat mass |
| BP | Bench press |
| BR | Bent-over row |
| BW | Body weight |
| COL | Hydrolyzed collagen peptide supplementation group |
| CP | collagen peptide |
| CSA | Cross-sectional area |
| DL | Deadlift |
| DOMS | Delayed onset of muscle soreness |
| ECM | Extracellular matrix |
| fCSA | Muscle fiber cross-sectional area |
| FFM | Fat-free mass |
| KE | Knee extension |
| LC | Leg circumference |
| Leg-Ex | Leg extension |
| MViC | Maximal voluntary isometric contraction |
| PLA | Placebo-controlled supplementation group |
| RDA | Recommended dietary allowance |
| RET | Resistance exercise training |
| RPE | Rate of perceived exertion |
| SQ | Squat |
References
- Phillips, S.M. Dietary protein requirements and adaptive advantages in athletes. Br. J. Nutr. 2012, 108 (Suppl. 2), S158–S167. [Google Scholar] [CrossRef] [PubMed]
- Cermak, N.M.; Res, P.T.; de Groot, L.C.; Saris, W.H.; van Loon, L.J. Protein supplementation augments the adaptive response of skeletal muscle to resistance-type exercise training: A meta-analysis. Am. J. Clin. Nutr. 2012, 96, 1454–1464. [Google Scholar] [CrossRef]
- Morton, R.W.; Murphy, K.T.; McKellar, S.R.; Schoenfeld, B.J.; Henselmans, M.; Helms, E.; Aragon, A.A.; Devries, M.C.; Banfield, L.; Krieger, J.W.; et al. A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. Br. J. Sports Med. 2017, 52, 376–384. [Google Scholar] [CrossRef]
- Heaton, L.E.; Davis, J.K.; Rawson, E.S.; Nuccio, R.P.; Witard, O.C.; Stein, K.W.; Baar, K.; Carter, J.M.; Baker, L.B. Selected In-Season Nutritional Strategies to Enhance Recovery for Team Sport Athletes: A Practical Overview. Sports Med. 2017, 47, 2201–2218. [Google Scholar] [CrossRef]
- Hoppeler, H. Molecular networks in skeletal muscle plasticity. J. Exp. Biol. 2016, 219, 205–213. [Google Scholar] [CrossRef]
- Chiquet, M. Regulation of extracellular matrix gene expression by mechanical stress. Matrix Biol. 1999, 18, 417–426. [Google Scholar] [CrossRef]
- Mackey, A.L.; Donnelly, A.E.; Turpeenniemi-Hujanen, T.; Roper, H.P. Skeletal muscle collagen content in humans after high-force eccentric contractions. J. Appl. Physiol. 2004, 97, 197–203. [Google Scholar] [CrossRef] [PubMed]
- Moore, D.R.; Phillips, S.M.; Babraj, J.A.; Smith, K.; Rennie, M.J. Myofibrillar and collagen protein synthesis in human skeletal muscle in young men after maximal shortening and lengthening contractions. Am. J. Physiol. Endocrinol. Metab. 2005, 288, E1153–E1159. [Google Scholar] [CrossRef]
- Alexander, R.M.; Bennet-Clark, H.C. Storage of elastic strain energy in muscle and other tissues. Nature 1977, 265, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Kjaer, M. Role of extracellular matrix in adaptation of tendon and skeletal muscle to mechanical loading. Physiol. Rev. 2004, 84, 649–698. [Google Scholar] [CrossRef]
- Zdzieblik, D.; Oesser, S.; Baumstark, M.W.; Gollhofer, A.; König, D. Collagen peptide supplementation in combination with resistance training improves body composition and increases muscle strength in elderly sarcopenic men: A randomised controlled trial. Br. J. Nutr. 2015, 114, 1237–1245. [Google Scholar] [CrossRef]
- Kjaer, M.; Magnusson, P.; Krogsgaard, M.; Boysen Møller, J.; Olesen, J.; Heinemeier, K.; Hansen, M.; Haraldsson, B.; Koskinen, S.; Esmarck, B.; et al. Extracellular matrix adaptation of tendon and skeletal muscle to exercise. J. Anat. 2006, 208, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Bello, A.E.; Oesser, S. Collagen hydrolysate for the treatment of osteoarthritis and other joint disorders: A review of the literature. Curr. Med. Res. Opin. 2006, 22, 2221–2232. [Google Scholar] [CrossRef]
- Clark, K.L.; Sebastianelli, W.; Flechsenhar, K.R.; Aukermann, D.F.; Meza, F.; Millard, R.L.; Deitch, J.R.; Sherbondy, P.S.; Albert, A. 24-Week study on the use of collagen hydrolysate as a dietary supplement in athletes with activity-related joint pain. Curr. Med. Res. Opin. 2008, 24, 1485–1496. [Google Scholar] [CrossRef]
- Ramanathan, G.; Muthukumar, T.; Tirichurapalli Sivagnanam, U. In vivo efficiency of the collagen coated nanofibrous scaffold and their effect on growth factors and pro-inflammatory cytokines in wound healing. Eur. J. Pharmacol. 2017. [Google Scholar] [CrossRef]
- Dressler, P.; Gehring, D.; Zdzieblik, D.; Oesser, S.; Gollhofer, A.; König, D. Improvement of Functional Ankle Properties Following Supplementation with Specific Collagen Peptides in Athletes with Chronic Ankle Instability. J. Sports Sci. Med. 2018, 17, 298–304. [Google Scholar] [CrossRef]
- Praet, S.F.E.; Purdam, C.R.; Welvaert, M.; Vlahovich, N.; Lovell, G.; Burke, L.M.; Gaida, J.E.; Manzanero, S.; Hughes, D.; Waddington, G. Oral Supplementation of Specific Collagen Peptides Combined with Calf-Strengthening Exercises Enhances Function and Reduces Pain in Achilles Tendinopathy Patients. Nutrients 2019, 11, 76. [Google Scholar] [CrossRef] [PubMed]
- Zdzieblik, D.; Oesser, S.; Gollhofer, A.; König, D. Improvement of activity-related knee joint discomfort following supplementation of specific collagen peptides. Appl. Physiol. Nutr. Metab. 2017, 42, 588–595. [Google Scholar] [CrossRef]
- Baar, K. Stress Relaxation and Targeted Nutrition to Treat Patellar Tendinopathy. Int. J. Sport Nutr. Exerc. Metab. 2019, 1–5. [Google Scholar] [CrossRef]
- Shaw, G.; Lee-Barthel, A.; Ross, M.L.; Wang, B.; Baar, K. Vitamin C-enriched gelatin supplementation before intermittent activity augments collagen synthesis. Am. J. Clin. Nutr. 2017, 105, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Fry, C.S.; Kirby, T.J.; Kosmac, K.; McCarthy, J.J.; Peterson, C.A. Myogenic Progenitor Cells Control Extracellular Matrix Production by Fibroblasts during Skeletal Muscle Hypertrophy. Cell Stem Cell 2017, 20, 56–69. [Google Scholar] [CrossRef] [PubMed]
- Mackey, A.L.; Magnan, M.; Chazaud, B.; Kjaer, M. Human skeletal muscle fibroblasts stimulate in vitro myogenesis and in vivo muscle regeneration. J. Physiol. 2017, 595, 5115–5127. [Google Scholar] [CrossRef] [PubMed]
- Pawlikowski, B.; Vogler, T.O.; Gadek, K.; Olwin, B.B. Regulation of skeletal muscle stem cells by fibroblast growth factors. Dev. Dyn. 2017, 246, 359–367. [Google Scholar] [CrossRef]
- Chow, S.-C. Adaptive clinical trial design. Annu. Rev. Med. 2014, 65, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Faul, F.; Erdfelder, E.; Lang, A.-G.; Buchner, A. G* Power 3: A flexible statistical power analysis program for the social, behavioral, and biomedical sciences. Behav. Res. Methods 2007, 39, 175–191. [Google Scholar] [CrossRef]
- Halson, S.L. Monitoring training load to understand fatigue in athletes. Sports Med. 2014, 44 (Suppl. 2), S139–S147. [Google Scholar] [CrossRef]
- Chen, Z.; Wang, Z.; Lohman, T.; Heymsfield, S.B.; Outwater, E.; Nicholas, J.S.; Bassford, T.; LaCroix, A.; Sherrill, D.; Punyanitya, M.; et al. Dual-energy X-ray absorptiometry is a valid tool for assessing skeletal muscle mass in older women. J. Nutr. 2007, 137, 2775–2780. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.J.; Erceg, D.N.; Schroeder, E.T. Utility of multifrequency bioelectrical impedance compared with dual-energy X-ray absorptiometry for assessment of total and regional body composition varies between men and women. Nutr. Res. 2012, 32, 479–485. [Google Scholar] [CrossRef]
- Miyatani, M.; Kanehisa, H.; Ito, M.; Kawakami, Y.; Fukunaga, T. The accuracy of volume estimates using ultrasound muscle thickness measurements in different muscle groups. Eur. J. Appl. Physiol. 2004, 91, 264–272. [Google Scholar] [CrossRef]
- Kreamer, W.J.; Fry, A.C. Strength testing: Development and evaluation of methodology. In Physiological Assessment of Human Fitness; Maud, P.J., Foster, C., Eds.; Human Kinetics: Champaign, IL, USA, 1995; Volume 2, pp. 119–150. [Google Scholar]
- Bergstrom, J. Percutaneous needle biopsy of skeletal muscle in physiological and clinical research. Scand. J. Clin. Lab. Investig. 1975, 35, 609–616. [Google Scholar] [CrossRef]
- Baar, K. Minimizing Injury and Maximizing Return to Play: Lessons from Engineered Ligaments. Sports Med. 2017, 47, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-S.; Kosek, D.J.; Petrella, J.K.; Cross, J.M.; Bamman, M.M. Resting and load-induced levels of myogenic gene transcripts differ between older adults with demonstrable sarcopenia and young men and women. J. Appl. Physiol. 2005, 99, 2149–2158. [Google Scholar] [CrossRef]
- Cleather, D.J.; Guthrie, S.R. Quantifying delayed-onset muscle soreness: A comparison of unidimensional and multidimensional instrumentation. J. Sports Sci. 2007, 25, 845–850. [Google Scholar] [CrossRef] [PubMed]
- Borg, G.; Hassmén, P.; Lagerström, M. Perceived exertion related to heart rate and blood lactate during arm and leg exercise. Eur. J. Appl. Physiol. Occup. Physiol. 1987, 56, 679–685. [Google Scholar] [CrossRef]
- Jendricke, P.; Centner, C.; Zdzieblik, D.; Gollhofer, A.; König, D. Specific Collagen Peptides in Combination with Resistance Training Improve Body Composition and Regional Muscle Strength in Premenopausal Women: A Randomized Controlled Trial. Nutrients 2019, 11, 892. [Google Scholar] [CrossRef]
- Haun, C.T.; Vann, C.G.; Roberts, B.M.; Vigotsky, A.D.; Schoenfeld, B.J.; Roberts, M.D. A Critical Evaluation of the Biological Construct Skeletal Muscle Hypertrophy: Size Matters but So Does the Measurement. Front. Physiol. 2019, 10, 613. [Google Scholar] [CrossRef]
- Kurobe, K.; Huang, Z.; Nishiwaki, M.; Yamamoto, M.; Kanehisa, H.; Ogita, F. Effects of resistance training under hypoxic conditions on muscle hypertrophy and strength. Clin. Physiol. Funct. Imaging 2015, 35, 197–202. [Google Scholar] [CrossRef]
- Abe, T.; DeHoyos, D.V.; Pollock, M.L.; Garzarella, L. Time course for strength and muscle thickness changes following upper and lower body resistance training in men and women. Eur. J. Appl. Physiol. 2000, 81, 174–180. [Google Scholar] [CrossRef]
- Ogborn, D.; Schoenfeld, B.J. The Role of Fiber Types in Muscle Hypertrophy. Strength Cond. J. 2014, 36, 20–25. [Google Scholar] [CrossRef]
- Mackey, A.L.; Brandstetter, S.; Schjerling, P.; Bojsen-Moller, J.; Qvortrup, K.; Pedersen, M.M.; Doessing, S.; Kjaer, M.; Magnusson, S.P.; Langberg, H. Sequenced response of extracellular matrix deadhesion and fibrotic regulators after muscle damage is involved in protection against future injury in human skeletal muscle. FASEB J. 2011, 25, 1943–1959. [Google Scholar] [CrossRef] [PubMed]
- Mackey, A.L.; Heinemeier, K.M.; Koskinen, S.O.A.; Kjaer, M. Dynamic adaptation of tendon and muscle connective tissue to mechanical loading. Connect. Tissue Res. 2008, 49, 165–168. [Google Scholar] [CrossRef] [PubMed]
- Folland, J.P.; Williams, A.G. The adaptations to strength training: Morphological and neurological contributions to increased strength. Sports Med. 2007, 37, 145–168. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.D.; Sacks, G.; Chandramohan, D.; Chow, C.C.; Wang, Y.C.; Gortmaker, S.L.; Swinburn, B.A. Quantification of the effect of energy imbalance on bodyweight. Lancet 2011, 378, 826–837. [Google Scholar] [CrossRef]
- Yang, Y.J.; Kim, M.K.; Hwang, S.H.; Ahn, Y.; Shim, J.E.; Kim, D.H. Relative validities of 3-day food records and the food frequency questionnaire. Nutr. Res. Pract. 2010, 4, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Tieland, M.; Borgonjen-Van den Berg, K.J.; van Loon, L.J.; de Groot, L.C. Dietary protein intake in community-dwelling, frail, and institutionalized elderly people: Scope for improvement. Eur. J. Nutr. 2012, 51, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Hays, N.P.; Kim, H.; Wells, A.M.; Kajkenova, O.; Evans, W.J. Effects of whey and fortified collagen hydrolysate protein supplements on nitrogen balance and body composition in older women. J. Am. Diet. Assoc. 2009, 109, 1082–1087. [Google Scholar] [CrossRef]
- Thomas, D.K.; Quinn, M.A.; Saunders, D.H.; Greig, C.A. Protein Supplementation Does Not Significantly Augment the Effects of Resistance Exercise Training in Older Adults: A Systematic Review. J. Am. Med. Dir. Assoc. 2016, 17, 959.e1–959.e9. [Google Scholar] [CrossRef]
- Clifford, T.; Ventress, M.; Allerton, D.M.; Stansfield, S.; Tang, J.C.Y.; Fraser, W.D.; Vanhoecke, B.; Prawitt, J.; Stevenson, E. The effects of collagen peptides on muscle damage, inflammation and bone turnover following exercise: A randomized, controlled trial. Amino Acids 2019, 1–14. [Google Scholar] [CrossRef]
- Oesser, S.; Adam, M.; Babel, W.; Seifert, J. Oral administration of (14)C labeled gelatin hydrolysate leads to an accumulation of radioactivity in cartilage of mice (C57/BL). J. Nutr. 1999, 129, 1891–1895. [Google Scholar] [CrossRef]
| Characteristics | Whole Cohort | COL | PLA | ANOVA (Time × Group) | Unpaired t-Test (Post) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n | Pre | Post | n | Pre | Post | n | Pre | Post | p-Value | p-Value | ||
| Body Composition | BW (kg) | 57 | 78.8 ± 7.7 | 80.5 ± 7.1 § | 29 | 79.3 ± 8.4 | 81.3 ± 8.1 * | 28 | 78.2 ± 6.3 | 79.6 ± 6.0 * | n.s. 0.314 | - |
| BFM (kg) | 57 | 9.0 ± 3.5 | 9.3 ± 3.5 § | 29 | 9.2 ± 3.8 | 9.2 ± 3.9 * | 28 | 8.8 ± 3.2 | 9.5 ± 3.0 * | 0.027 | n.s. 0.672 | |
| FFM (kg) | 57 | 69.8 ± 6.4 | 71.1 ± 6.3 § | 29 | 70.1 ± 6.7 | 72.1 ± 6.6 * | 28 | 69.4 ± 6.2 | 70.1 ± 5.9 * | 0.002 | n.s. 0.227 | |
| LC (cm) | 57 | 57.2 ± 3.3 | 58.4 ± 3.0 § | 29 | 57.4 ± 3.8 | 58.7 ± 3.5 * | 28 | 57.1 ± 2.7 | 58.0 ± 2.4 * | n.s. 0.386 | - | |
| Ultra- sound | Rec (mm) | 56 | 25.51 ± 3.25 | 26.08 ± 2.83 § | 28 | 25.45 ± 3.42 | 26.10 ± 3.09 | 28 | 25.57 ± 3.15 | 26.06 ± 2.60 | n.s. 0.770 | - |
| Int (mm) | 56 | 19.10 ± 3.52 | 20.57 ± 3.42 § | 28 | 19.31 ± 3.70 | 21.21 ± 3.89 | 28 | 18.88 ± 3.37 | 19.92 ± 2.78 | n.s. 0.194 | - | |
| Lat (mm) | 56 | 26.36 ± 4.08 | 28.69 ± 3.96 § | 28 | 26.10 ± 3.82 | 28.75 ± 3.35 | 28 | 26.63 ± 4.37 | 28.62 ± 4.56 | n.s. 0.177 | - | |
| External load | Squat (kg) | 56 | 2735 ± 544 | 3280 ± 429 § | 29 | 2861 ± 463 | 3339 ± 457 | 27 | 2599 ± 598 | 3217 ± 395 | n.s. 0.198 | - |
| Deadlift (kg) | 56 | 3251 ± 478 | 3783 ± 550 § | 29 | 3338 ± 430 | 3836 ± 537 | 27 | 3158 ± 517 | 3726 ± 568 | n.s. 0.503 | - | |
| Bench press (kg) | 56 | 2052 ± 379 | 2356 ± 360 § | 29 | 2069 ± 376 | 2380 ± 409 | 27 | 2034 ± 388 | 2330 ± 409 | n.s. 0.803 | - | |
| Bent-over row (kg) | 56 | 2198 ± 360 | 2526 ± 347 § | 29 | 2177 ± 374 | 2564 ± 349 | 27 | 2177 ± 374 | 2564 ± 349 | n.s. 0.070 | - | |
| Strength | Leg-Ex (N×m) | 55 | 266.4 ± 46.5 | 291.8 ± 54.4 § | 28 | 271.5 ± 51.4 | 299.5 ± 61.6 * | 27 | 261.2 ± 41.2 | 283.7 ± 45.6 * | n.s. 0.477 | - |
| Squat (kg) | 53 | 110.5 ± 14.9 | 128.4 ± 18.2 § | 28 | 110.9 ± 16.5 | 131.5 ± 21.4 * | 25 | 110.1 ± 13.2 | 124.9 ± 13.4 * | n.s. 0.054 | n.s. 0.180 | |
| Deadlift (kg) | 54 | 131.2 ± 17.7 | 151.6 ± 17.8 § | 28 | 132.7 ± 16.2 | 154.0 ± 18.8 * | 26 | 129.6 ± 19.4 | 148.9 ± 16.6 * | n.s. 0.576 | - | |
| Bench press (kg) | 56 | 81.8 ± 14.0 | 94.2 ± 13.0 § | 28 | 82.6 ± 14.0 | 95.4 ± 14.3 * | 28 | 81.0 ± 14.2 | 92.9 ± 11.6 * | n.s. 0.474 | - | |
| Bent-over row (kg) | 56 | 88.1 ± 10.2 | 99.9 ± 10.0 § | 29 | 88.5 ± 11.0 | 100.6 ± 11.1 * | 27 | 87.6 ± 9.5 | 99.1 ± 8.9 * | n.s. 0.768 | - | |
| Food protocol | Total Energy (kcal/day) | 54 | 3014 ± 634 | 27 | 2958 ± 676 | 27 | 3070 ± 597 | - | n.s. 0.521 | |||
| Carbohydrate (kcal/day) | 54 | 1442 ± 367 | 27 | 1386 ± 390 | 27 | 1498 ± 341 | - | n.s. 0.269 | ||||
| Fat (kcal/day) | 54 | 1001 ± 332 | 27 | 986 ± 370 | 27 | 1016 ± 396 | - | n.s. 0.745 | ||||
| Protein (kcal/day) | 54 | 570 ± 153 | 27 | 585 ± 139 | 27 | 556 ± 167 | - | n.s. 0.491 | ||||
| Protein (g/kg/day) | 54 | 1.77 ± 0.46 | 27 | 1.81 ± 0.42 | 27 | 1.74 ± 0.50 | - | n.s. 0.610 | ||||
| COL (n = 10) | PLA (n = 11) | ANOVA | ||||
|---|---|---|---|---|---|---|
| Variable | Pre | Post | Pre | Post | Time (p) | Time × Group (p) |
| Type I % | 40 ± 10 | 37 ± 11 | 37 ± 13 | 38 ± 9 | n.s. 0.692 | n.s. 0.649 |
| Type II (%) | 60 ± 10 | 63 ± 11 | 63 ± 13 | 62 ± 9 | n.s. 0.692 | n.s. 0.649 |
| Type I fCSA (µm2) | 6455 ± 1462 | 6883 ± 1650 | 6419 ± 1094 | 6886 ± 1120 | 0.099 | n.s. 0.941 |
| Type II fCSA (µm2) | 7258 ± 1444 | 8330 ± 2076 | 7501 ± 1604 | 8484 ± 1812 | <0.001 | n.s. 0.865 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kirmse, M.; Oertzen-Hagemann, V.; de Marées, M.; Bloch, W.; Platen, P. Prolonged Collagen Peptide Supplementation and Resistance Exercise Training Affects Body Composition in Recreationally Active Men. Nutrients 2019, 11, 1154. https://doi.org/10.3390/nu11051154
Kirmse M, Oertzen-Hagemann V, de Marées M, Bloch W, Platen P. Prolonged Collagen Peptide Supplementation and Resistance Exercise Training Affects Body Composition in Recreationally Active Men. Nutrients. 2019; 11(5):1154. https://doi.org/10.3390/nu11051154
Chicago/Turabian StyleKirmse, Marius, Vanessa Oertzen-Hagemann, Markus de Marées, Wilhelm Bloch, and Petra Platen. 2019. "Prolonged Collagen Peptide Supplementation and Resistance Exercise Training Affects Body Composition in Recreationally Active Men" Nutrients 11, no. 5: 1154. https://doi.org/10.3390/nu11051154
APA StyleKirmse, M., Oertzen-Hagemann, V., de Marées, M., Bloch, W., & Platen, P. (2019). Prolonged Collagen Peptide Supplementation and Resistance Exercise Training Affects Body Composition in Recreationally Active Men. Nutrients, 11(5), 1154. https://doi.org/10.3390/nu11051154






